Abstract
The risk of venous thromboembolism following major orthopaedic procedures, such as joint arthroplasty and hip fracture surgery, are well recognised and represent one of the major challenges in orthopaedic practice, having in mind the increasing number of arthroplasties of the hip and knee done worldwide per year and their successful outcome. This potentially fatal complication remains a challenge in orthopaedic practice. The percentage of patients in whom antithrombotic prophylaxis has not been administrated or has been inadequate may reach 50 %. Until recently, anticoagulant prophylaxis with low molecular weight heparins (LMWHs) has been a “gold standard”. LMWHs are indirect inhibitors of the clotting factors Xa and thrombin and are administered by daily subcutaneous injection. Their efficacy has been proven in numerous clinical trials and the rate of complications with their use is relatively low. However these compounds are associated with a failure rate and are inconvenient to administer, requiring subcutaneous injection, leading to inadequate compliance. For these reasons postoperative thrombembolism continues to occur in up to 10 % of this patient population. Recently, novel oral anticoagulants have been introduced into practice for thromboprophylaxis after joint arthroplasy and hip fracture surgery. These drugs are direct thrombin inhibitors (dabigatran) or direct factor Xa inhibitors (rivaroxaban, apixaban and edoxaban). These oral drugs have the same efficacy as the LMWHs with the same or slightly more clinically significant haemorrhage as their main side effect. Their ease of administration and favourable clinical profile makes them an important addition to the therapeutic armamentarium available for venous thromboprophylaxis. In this paper we review the aetiology and pathogenesis of venous thromboembolism and present the various alternatives for its prevention after major orthopaedic surgical procedures with emphasis on the new oral drugs.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Venous thromboembolism (VTE) refers to a single pathological process—thrombosis of a peripheral vein (deep venous thrombosis/DVT), embolisation and thrombosis of a branch of the pulmonary artery (pulmonary embolism/PE). The increased risk of development of venous thromboembolic disease and its concomitant complications and mortality after major orthopaedic interventions, arthroplasty of hip and knee joint (THA, TKA), and surgical intervention of hip joint fractures is well established and continues to be a challenge in orthopaedic practice [1]. The incidence of deep venous thrombosis when no prophylaxis is administered is 42–57 % with total hip arthroplasty and 41–85 % with total knee arthroplasty. Fatal PE occurs in 0.1–2 % of patients after hip THA and in 0.1–1.7 % of the patients with TKA [2]. It is recognised that symptomatic VTE, which occurs in about 4 % of patients, is more frequent than the complications such as luxation and postoperative infection. These data demonstrate the need for effective and safe thromboprophylaxis. Anticoagulant prophylaxis initially using unfractionated heparin and subsequently using low molecular weight heparins in major orthopaedic surgery became widespread in the last quarter of the 20th century. Recommendations for antithrombotic prophylaxis published by different healthcare and scientific organisations and committees (American College of Chest Physicians [ACCP], American Academy of Orthopedic Surgeons [AAOS], National Institute for Health and Clinical Excellence, etc.) resulted in extensive administration of anticoagulants in practice [3–6]. Until recently anticoagulant prophylaxis with low molecular weight heparin has been a “gold standard”. Nevertheless, the percentage of patients in whom antithrombotic prophylaxis has not been administered or has been inadequate may reach 50 % [7–10]. Recently, new orally available pharmacological agents with effectiveness comparable to low molecular weight heparin have been introduced into practice.
Historical review of the development of thromboprophylaxis
After the proposal of the main mechanisms for development of the thrombosis and embolism by Rudolf Virchow in the 1880s, attempts at treatment and prevention of venous thromboembolism have been undertaken since the beginning of the twentieth century. The first author who described emergency pulmonary embolectomy was Trendelenburg in 1908 through thoracotomy and removal of embolus via incision of the pulmonary artery [11]. The initial results were disappointing and the first two successful embolectomies were described in 1928 [12]. Diagnosis of pulmonary thromboembolism at that time was purely clinical until the introduction of the pulmonary angiography in 1963 [13], which together with the development of cardiopulmonary bypass increased the success of pulmonary embolectomy. Currently this procedure is reserved for patients with massive pulmonary thromboembolism leading to shock.
Prophylaxis of pulmonary embolism by ligation of the vein above the site of thrombosis was described for the first time in 1934 by Homans [14]. Because at that time there were no pharmacological agents for prevention of the thromboembolism, bilateral ligation of the femoral vein proved to be the main method for prophylaxis of pulmonary thromboembolism. Later, ligation of inferior vena cava was also introduced. The use of the two procedures decreased sharply with the introduction of heparin and oral anticoagulants. Insertion of a filter in the inferior vena cava was introduced in the 1970s. The indications for its use are VTE with concomitant active bleeding or other contraindications for anticoagulant therapy.
Howell and Holt [15] discovered a substance in 1918, which had the property of slowing coagulation. It was named heparin due to its finding in large quantity in the liver. Heparin is a mucopolysaccharide, which is present in plasma in small quantities and has weak intrinsic anticoagulant properties. However, in combination with antithrombin, it forms a complex which binds to and inhibits the activity of certain activated clotting factors, namely, factors XIIa, XIa, Xa, IXa and thrombin. Intravenous heparin was found to be effective in the prevention of postoperative VTE [16, 17], although the initial studies were not controlled and events were detected both clinically and at post mortem. Subcutaneous heparin in low doses for the prophylaxis of venous thromboembolism after surgical intervention was first reported by Sharnoff et al. [18]. Randomised studies demonstrated a risk reduction of postoperative DVT of up to 68 %. Although the frequency of wound haematomas was increased major postoperative haemorrhages were a rare complication. Low dose subcutaneous heparin was also found to be effected for VTE prophylaxis in orthopaedic operations such as arthroplasty of major joints and operations for hip fractures which have a significantly higher incidence of the DVT than general surgical operations.
Low molecular weight heparins (LMWHs) were introduced in the 1980s. These drugs are derivatives of unfractionated heparin, obtained by chemical or enzymatic depolymerisation, and have a molecular weight of one third of that of unfractionated heparin. LMWHs have less anti thrombin activity than anti factor Xa activity, which may improve their safety profile [19]. LMWHs in contrast to unfractionated heparin are given in fixed doses by daily subcutaneous injection and they do not require routine laboratory monitoring. This makes them more suitable for use after hospital discharge which is beneficial after major joint arthroplasy. Furthermore, osteoporosis and heparin induced thrombocytopenia are rarely encountered with LMWH use [20]. A disadvantage of LMWH is its subcutaneous administration, which may reduce patient compliance in out-patient conditions. This potential drawback of LMWH is significant, because according to a series of studies, the reduction of DVT (symptomatic or proved with venography) in two thirds of the cases of the hip arthroplasty is achieved by administration of LMWH for a minimum of four weeks after surgery [7, 21, 22].
The first reports of the use of oral anticoagulants for prevention of venous thromboembolism were published 60 years ago [23, 24]. The drugs used were vitamin K antagonists, which block the synthesis of coagulation factors—VII, IX, X and II. Despite their proven efficacy [25], and the convenience of their oral administration, they have significant disadvantages limiting their use in orthopaedic practice. The late onset of their anticoagulant activity results in an unprotected period of two to three days after the surgery. Their use requires accurate monitoring of the international normalised ratio, because of the variable levels of dietary vitamin K and multiple interactions with medications and food. The last is true especially in patients receiving therapy for concomitant diseases. For example, warfarin, which is used frequently in North America and the United Kingdom for prophylaxis in orthopaedic surgery, has an unpredictable pharmacological profile and its dose must be individualised for every patient due to the risk of haemorrhagic complications.
Aspirin, due to its antiaggregant properties, was suggested for prophylaxis of venous thrombosis in 1968 [26]. It has a comparatively low thromboprophylactic effect and the slight reduction of the risk of VTE in patients with fractures of the hip joint [8], TKA and THA [20] is accompanied by complications such as gastrointestinal bleeding, bleeding from surgical wounds and the need for red cell transfusion. For these reasons, aspirin is not universally recommended for VTE prophylaxis in orthopaedic patients, according to 2001 and 2006 guidelines [6, 20]. Debates for its use in orthopaedic patients have been in progress since then. The 2012 edition of the American College of Chest Physicians recommendations on antithrombotic treatment includes aspirin as one of the possible medications for prophylaxis [4], stating that the use of one of the pharmacological methods for prophylaxis, including aspirin, is better in comparison with no administration of any pharmacological prophylaxis in orthopaedic patients. Some authors suggest its use in low-risk patients after knee and hip arthroplasty in combination with early rapid mobilisation, pneumatic compression devices and preoperative risk stratification [27].
Clinical studies on the effectiveness of aspirin, warfarin and LMWH
Aspirin and other antiplatelet medicines have weaker effect compared to the other methods of thromboprophylaxis as demonstrated by a study published in 1996, in which patients undergoing TKA received either aspirin and pneumatic compression of the operated limb or aspirin alone. The frequency of DVT in the first group was 27 %, and in the second group—59 % [28]. Another study randomised patients with fractures of the proximal femur to receive either aspirin or danaparoid (a low molecular weight heparinoid). The incidence of VTE was 44 % and 28 %, respectively [29]. In one major study (Pulmonary Embolism Preventing [PEP] trial) with 4,088 patients with TKA and THA, aspirin was compared to a group taking placebo. About 40 % of patients took other forms of prophylaxis such as heparin and low molecular weight heparin. No advantage in prophylaxis against thromboembolic events was found with the use of aspirin [30]. In pooled analysis aspirin demonstrates relative risk reduction of VTE of 26 %, 13 % and 29 % in THA, TKA and fractures of the proximal femur, respectively, which is less than that achieved with anticoagulants [20]. However, a recent study provides new data suggesting a role for aspirin in VTE prophylaxis in orthopaedic surgery. Anderson et al. [31] reported that aspirin is not inferior to dalteparin in prevention of VTE after THA. A total of 778 patients with THA during the period 2007–2010 were included in the study. After ten days of dalteparin postoperative prophylaxis, 400 patients were randomised to receive dalteparin for 28 days and 386 received aspirin for the same period. Only 1.3 % of the group receiving dalteparin and 0.3 % of the group treated with aspirin had VTE on the 90th day. The incidence of bleeding was low and similar in both groups.
Vitamin K antagonists, such as warfarin, possess the advantage of oral administration, however they are less effective than LMWHs. One study from 1993 compared warfarin with LMWH in patients with TKA and THA. Venographically diagnosed that DVT was more common in the warfarin group (37.4 %) than in the LMWH group (31.4 %) [25]. In summary, data from randomised studies show a relative risk reduction of DVT with the use of warfarin in patients after THA, TKA and fractures of the proximal femur to be 59 %, 27 % and 48 %, respectively, indicating their reduced efficacy compared to LMWH [20].
Currently, LMWHs are widely used for VTE prophylaxis in orthopaedic patients. Eight clinical studies have been performed, all demonstrating superior efficacy of LMWH compared to unfractionated heparin in general surgical patients [32]. A randomised study reported in 1986 in 100 patients with THA, who received enoxaparin versus placebo, demonstrated a 12 % incidence of DVT in the LMWH group, compared to 42 % in the placebo group [33]. A similar study of 237 patients with THA demonstrated a 25 % incidence of DVT in patients treated with unfractionated heparin compared to 12.5 % in the group receiving enoxaparin [34]. Regarding knee arthroplasty, DVT occurred in 36.9 % enoxaparin treated patients and in 51.7 % of warfarin treated patients [35]. Data from 30 clinical studies demonstrates a 70 % relative reduction in the risk of DVT in patients with THA, who receive prophylaxis with LMWH, a 52 % relative reduction of DVT after TKA (13 studies) and a 44 % relative reduction after surgery for fractures of the proximal femur (five studies) [20].
Current prophylaxis
Non-pharmacological agents—early ambulation and mechanical devices
Early ambulation is an important prophylactic measure for DVT after major orthopaedic surgery [36]. Although there are no empirical data for its effectiveness, it has a low cost, no risk for the patient and is compatible with clinical practice. Its realisation depends on patient cooperation, his/her general condition and the concomitant diseases.
Other non-pharmacological agents for thromboprophylaxis are elastic stockings and intermittent pneumatic compression (IPC), which has a limited application—the device must be applied constantly, is generally inconvenient and limits the mobility, which is why it is not tolerated well by patients. Thus, its effectiveness in preventing DVT is controversial, due to incorrect use of the device in many cases [37]. Elastic stockings, which are considered to reduce the risk of thrombosis due to the increase of velocity of venous reflux, are relatively ineffective in reducing the risk of DVT: 6 % relative risk reduction in TKA and 23 % in THA compared to pharmacological methods [2, 20]. Intermittent pneumatic compression in some studies with hip joint arthroplasty shows good efficacy for prevention of all DVT (63 % relative risk reduction), while the effectiveness for proximal deep venous thrombosis is poorer (48 % relative risk reduction), compared to the administration of pharmacological agents. As regards TKA, some authors suggest that IPC is sufficiently effective for the prevention of deep venous thrombosis [28, 38] and more effective compared to aspirin [39]. In summary, intermittent pneumatic compression may be useful when used in addition to pharmacological prophylaxis, mainly during the period of hospitalisation. It can also be considered as an independent prophylactic measure, in patients at high risk of bleeding.
Antithrombotic prophylaxis with pharmacological agents
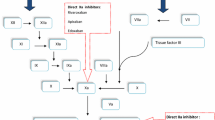
The ideal anticoagulant drug should be efficacious, safe, convenient to use and affordable. During the past few years several new anticoagulants have been approved for DVT prophylaxis after major orthopaedic surgery, namely, fondaparinux, apixaban, dabigartan, and rivaroxaban. These agents provide more targeted inhibition of activated clotting factors than heparins and vitamin K antagonists (Fig. 1). These drugs, like LMWH, have a fixed dose–response relationship which makes routine monitoring of their anticoagulant function unnecessary. Numerous clinical studies have demonstrated the efficacy and safety of these agents compared to LMWH for DVT prophylaxis.
Rivaroxaban
Rivaroxaban is an oral, direct inhibitor of factor Xar, with predictable pharmacokinetics and pharmacodynamics. There is no necessity for dose modification according to age, gender and body mass. It is excreted predominantly by the kidneys (70 %) with a small component excreted by the liver (30 %). There are few reported drug interactions with rivaroxaban and no known food interactions.
Four phase III studies including 12,500 patients with THA and TKA indicated superior efficacy of rivaroxaban compared to enoxaparin (composite endpoint of death and pulmonary embolism 0.5 % and 0.8 %, respectively, P = 0.039) [40–43]. Bleeding risk was the same for both agents. It is important to note that patients with a creatinine clearance below 30 ml/min were not included in the studies and rivaroxaban should not be used in such patients. The approved dose of Rivaroxaban is 10 mg/daily beginning six to ten hours after surgery and continued for five weeks after THA and two weeks after TKA.
Dabigartan
Dabigatran is an oral direct thrombin inhibitor that is administered once daily in a fixed dose for VTE prophylaxis following joint replacement. It has rapid onset of activity and has predictable anticoagulation activity. Excretion of dabigatran is predominantly renal (80 %) and is contraindicated for patients with severe renal failure (creatinine clearance under 30 ml/min), but it can be used at reduced dose in patients with moderate renal impairment with creatinine clearance 30–50 ml/min [44].
Two studies (REMODEL and RE-NOVATE) demonstrated that dabigatran is as effective as enoxaparin in the dose of 40 mg daily in patients with THA and TKA with a similar safety profile [45, 46]. A third study (RE-MOBILIZE) conducted in North America with patients after TKA, comparing dabigatran to enoxaparin 30 mg twice daily, resulted in superior efficacy in the enoxaparin arm, possibly because of the higher dose of this agent—30 mg twice daily—which is the standard North American dose of enoxaparin [47]. The results of a meta-analysis of the three studies showed that the incidence of bleeding was the same for both drugs [48].
Apixaban
Apixaban is an oral direct factor Xa inhibitor, given twice daily. Only 30 % of the drug is renally excreted making this the novel anticoagulant of choice in patients with renal failure. Three studies examined the efficacy and safety of apixaban compared to enoxaparin. In the first study (ADVANCE - 1), apixaban 2.5 mg twice daily compared to twice daily administration of enoxaparin 30 mg in patients with TKA did not show increased efficacy of apixaban [49]. In the second study (ADVANCE - 2), apixaban was compared to enoxaparin 30 mg once daily in TKA and demonstrated superior efficacy [50]. In both studies apixaban demonstrated favourable safety regarding bleeding. ADVANCE - 3 compared apixaban to enoxaparin (30 mg once daily) in THA and in this study apixaban demonstrated superiority compared enoxaparin, and the incidence of bleeding was the same for both medications [51]. The approved dosage of apixaban is 2.5 mg twice daily for a period of 32–38 days after the THA and for 10–14 days after TKA.
Fondaparinux
Fondaparinux is a synthetic pentasaccharide, which is an indirect, specific inhibitor of factor Xa . It is completely eliminated via the kidneys, therefore it must be carefully used in patients with renal disease. Fondaparinux compared to enoxaparin [52] in 7,300 patients with TKA, THA and fractures of proximal femur reduces the overall frequency of VTE on the eleventh day after the operation (6.8 vs 13.7 %). However, fondaparinux is associated with more non clinically significant bleeding compared to enoxaparin and should only be administered six hours after surgery and at least 12 hours after removal of a spinal/epidural catheter in order to avoid surgical or neuroaxial bleeding. Other disadvantages of the medication are its slower elimination (half-life of 13–21 hours), the risk of accumulation of the medication in patients with renal failure, irreversibility of the inhibition of factor Xa, and subcutaneous administration. Overall the advantages of the above-mentioned new anticoagulant agents, compared to the other used medications, are summarised in Table 1.
Conclusions
A large proportion of orthopaedic patients require thrombophylaxis, using either pharmacological agents or non-pharmacological methods. However, not all patients at risk for DVT ultimately receive prophylaxis. The reasons for this discrepancy include an underestimation of the risk of deep venous thrombosis, disagreement with the recommendations and data in literature, “clinical inertia”, apprehension regarding complications and last but not least the inconvenience of anticoagulant prophylaxis, and subsequent poor cooperation of patient and non-optimum results.
The consequences of DVT may be serious and include the post phlebitis syndrome and in patients with pulmonary embolism, chronic pulmonary hypertension, which has a 3–4 % mortality rate from right-sided heart failure [53]. DVT and PE also create a significant financial burden. The cost of therapy of such a patient yearly reach as much as $16,000 [54]. The new inhibitors of thrombin or factor Xa, fondaparinux, dabigatran, rivaroxaban and apixaban represent an alternative to LMWH and vitamin K antagonists in TKA and THA, surgical procedures which represent a major risk of VTE. Randomised controlled trials have demonstrated that these novel agents are at least as effective as enoxaparin and are not associated with an increased incidence of clinically significant bleeding. Their main advantages are oral administration, no need for laboratory monitoring, fixed dosage, predictable pharmacokinetics and pharmacodynamcis, equivalent efficacy to standard therapy, good safety profile, no interaction with food and medications and their postoperative application administration which allows same day admission and operation.
Therefore it is hoped that the new oral anticoagulants will improve the antithrombotic prophylaxis administration and will decrease the frequency of the thrombotic complications after a major orthopaedic surgery.
References
Markovic-Denic L, Zivkovic K, Lesic A et al (2012) Risk factors and distribution of symptomatic venous thromboembolism in total hip and knee replacements: prospective study. Int Orthop 36(6):1299–1305
Geerts WH, Pineo GF, Heit JA et al (2004) Prevention of venous thromboembolism: the seventh ACCP Conference on Antithrombotic and Thrombolytic Therapy. Chest 126:338S–400S
American Academy of Orthopaedic Surgeons (2011) Preventing venous thromboembolic disease in patients undergoing elective hip and knee arthroplasty. Evidence based guidelines and evidence report
Guyatt GH, Akl EA, Crowther M, Gutterman DD, Schuünemann HJ (2012) Executive summary: antithrombotic therapy and prevention of thrombosis, 9th ed: American College of Chest Physicians Evidence-Based Clinical Practice Guidelines. Chest 141(Suppl 2):7S–47S
Hill J, Treasure T (2007) Reducing the risk of venous thromboembolism (deep vein thrombosis and pulmonary embolism) in patients having surgery: summary of NICE guidance. BMJ 334:1053–1054
Nicolaides AN, Fareed J, Kakkar AK et al (2006) Prevention and treatment of venous thromboembolism. International consensus statement (Guidelines according to scientific evidence). Int Angiol 25:101–161
Cohen A, Bailey CS, Alikhan R, Cooper DJ (2001) Extended thromboprophylaxis with low molecular weight heparin reduces symptomatic venous thromboembolism following lower limb arthroplasty. Thromb Haemost 85:940–941
Cohen A, Quinlan D (2000) PEP trial. Lancet 356:247–248
Anderson FA Jr, Hirsh J, White K, Fitzgerald RH Jr (2003) Temporal trends in prevention of venous thromboembolism following primary total hip or knee arthroplasty 1996-2001: findings from the hip and knee register. Chest 124(Suppl 6):349S–356S
Friedman RJ, Gallus AS, Cushner FD, Fitzgerald G, Anderson FA Jr (2008) Physician compliance with guidelines for deep-vein thrombosis prevention in total hip and knee arthroplasty. Curr Med Res Opin 24(1):87–97
Trendelenburg F (1908) Ueber die operative Behandlung der Embolie der Lungenarterie. Arch Klin Chir 86:686–700
Meyer JA (1990) Friedrich Trendelenburg and the surgical approach to massive pulmonary embolism. Arch Surg 125:1202–1205
Sautter RD, Lawton BR, Magin OE et al (1963) Pulmonary embolectomy: report of a case with preoperative and postoperative angiograms. N Eng J Med 269:997–999
Homans J (1934) Thrombosis of the deep veins of the lower leg, causing pulmonary embolism. N Engl J Med 211:993–997
Howell WH, Holt E (1918) Two new factors in blood coagulation—heparin and proantithrombin. Am J Physiol 47:328–333
Craaford C (1937) Preliminary report on post-operative treatment with heparin as a preventive of thrombosis. Acta Chir Scand 79:407–426
Murray GDW, Best CH (1938) Heparin and thrombosis: the present situation. JAMA 110:118–122
Sharnoff JG, Kass HH, Mistica BA (1962) A plan of heparinization of the surgical patient to prevent postoperative thromboembolism. Surg Gynecol Obstet 115:75–79
Hirsh J, Bauer KA, Donati MB et al (2008) Parenteral anticoagulants: American College of Chest Physicians evidence-based clinical practice guidelines (8th edition). Chest 133(Suppl 6):141S–159S
Geerts WH, Heit JA, Clagett GP et al (2001) Prevention of venous thromboembolism. Chest 119(1):132S–175S
Eikelboom JW, Quinlan DJ, Douketis JD (2001) Extended duration prophylaxis against venous thromboembolism after total hip or knee replacement: a meta-analysis of the randomised trials. Lancet 358:9–15
Hull RD, Pineo GF, Stein PD et al (2001) Extended out of hospital low-molecular weight heparin prophylaxis against deep venous thrombosis in patients after elective hip arthroplasty: a systematic review. Ann Intern Med 135:858–869
Allen EV, Hines EA, Kvale WF et al (1947) The use of dicumarol as an anticoagulant: experience in 2,307 cases. Ann Intern Med 27:371–381
Sevitt S, Gallagher NG (1959) Prevention of venous thrombosis and pulmonary embolism in injured patients. Lancet 2:981–989
Hull RD, Raskob G, Pineo G et al (1993) A comparison of subcutaneous low-molecular- weight heparin with warfarin sodium for prophylaxis against deep-vein-thrombosis after hip or knee implantation. N Engl J Med 329:1370–1376
O’Brien JR (1968) Effects of salicylates on human platelets. Lancet 2:779–783
Vulcano E, Gesell M, Esposito A et al (2012) Aspirin for elective hip and knee arthroplasty: a multimodal thromboprophylaxis protocol. Int Orthop 36(10):208–210
Westrich GH, Sculco TP (1996) Prophylaxis against deep venous thrombosis after total knee arthroplasty: pneumatic planter compression and aspirin compared with aspirin alone. J Bone Joint Surg Am 78:826–834
Gent M, Hirsh J, Ginsberg JS et al (1996) Low-molecular-weight heparinoid orgaran is more effective than aspirin in the prevention of venous thromboembolism after surgery for hip fracture. Circulation 93:80–84
Pulmonary Embolism Prevention (PEP) trial Collaborative Group (2000) Prevention of pulmonary embolism and deep vein thrombosis with low dose aspirin: Pulmonary embolism prevention (PEP) trial. Lancet 355(9212):1295–1302
Anderson DR, Dunbar MJ, Bohm ER et al (2013) Aspirin versus low-molecular-weight heparin for extended venous thromboembolism prophylaxis after total hip arthroplasty: a randomized trial. Ann Intern Med 158(11):800–806
Clarggett GP, Anderson FA, Levine MN et al (1992) Prevention of venous thromboembolism. Chest 102:391S–407S
Тurpie AGG, Levine MN, Hirsh J et al (1986) A randomized trial of a low-molecular heparin (enoxaparin) to prevent deep-vein thrombosis in patients undergoing elective hip surgery. N Engl J Med 315:925–929
Planes A, Vochelle N, Mazas F et al (1988) Prevention of postoperative venous thrombosis: a randomized trial comparing unfractioned heparin with low molecular weight heparin in patients undergoing total hip replacement. Thromb Haemost 60:407–410
Leclerc JR, Geerts WH, Desjardins L et al (1996) Prevention of venous thromboembolism after knee arthroplasty: a randomized, double-blind trial comparing enoxaparin with warfarin. Ann Intern Med 124:619–626
Lunceford EM, Patel SJ, Niestat HB et al (1978) Prevention of thrombophlebitis in total hip arhtoplasty by early ambulation. Clin Orthop 133:273
Charalambous C, Cleanthous S, Tryfonidis M et al (2003) Foot pump prophylaxis for deep venous thrombosis-rate of effective usage following knee and hip arthroplasty. Int Orthop 27:208–210
Hull R, Delmore TJ, Hirsh J et al (1979) Effectiveness of intermittent pulsatile elastic stockings for the prevention of calf and thigh vein thrombosis in patients undergoing elective knee surgery. Thromb Res 16:37–45
Haas SB, Insall JN, Scuderi GR, Windsor RE, Ghelman B (1990) Pneumatic sequential compression boots compared with aspirin prophylaxis of deep-vein thrombosis after total knee arthroplasty. J Bone Joint Surg Am 72:27–31
Eriksson BI, Borris LC, Friedman RJ et al (2008) Rivaroxaban versus enoxaparin for thromboprophylaxis after hip arthroplasty. N Engl J Med 358(26):2765–2775
Kakkar AK, Brenner B, Dahl OE et al (2008) Extended duration rivaroxaban versus short-term enoxaparin for the prevention of venous thromboembolism after total hip arthroplasty: a double-blind, randomised controlled trial. Lancet 372(9632):31–39
Lassen MR, Ageno W, Borris LC et al (2008) Rivaroxaban versus enoxaparin for thromboprophylaxis after total knee arthroplasty. N Engl J Med 358(26):2776–2786
Turpie AGG, Lassen MR, Davidson BL et al (2009) Rivaroxaban versus enoxaparin for thromboprophylaxis after total knee arthroplasty (RECORD4): a randomised trial. Lancet 373(9676):1673–1680
Dahl OE, Kurth AA, Rosencher N et al (2012) Thromboprophylaxis in patients older than 75 years or with moderate renal impairment undergoing knee or hip replacement surgery. Int Orthop 36(4):741–748
Eriksson BI, Dahl OE, Rosencher E et al (2007) Oral dabigatran etexilate vs. subcutaneous enoxaparin for the prevention of venous thromboembolism after total knee replacement: the RE-MODEL randomized trial. J Thromb Haemost 5(11):2178–2185
Eriksson BI, Dahl OE, Rosencher N et al (2007) Dabigatran etexilate versus enoxaparin for prevention of venous thromboembolism after total hip replacement: a randomised, double-blind, non-inferiority trial. Lancet 370(9591):949–956
Eriksson BI, Friedman R (2009) Dabigatran etexilate: pivotal trials for venous thromboembolism prophylaxis after hip or knee arthroplasty. Clin Appl Thromb Hemost 15:25S–31S
Wolowacz SE, Roskel NS, Plumb JM, Caprini JA, Eriksson BI (2009) Efficacy and safety of dabigatran etexilate for the prevention of venous thromboembolism following total hip or knee arthroplasty: a meta-analysis. J Thromb Haemost 101(1):77–85
Lassen MR, Raskob GE, Gallus A et al (2009) Apixaban or enoxaparin for thromboprophylaxis after knee replacement. N Engl J Med 361(6):594–604
Lassen MR, Raskob GE, Gallus A et al (2010) Apixaban versus enoxaparin for thromboprophylaxis after knee replacement (ADVANCE-2): a randomised double-blind trial. Lancet 375(9717):807–815
Lassen MR, Gallus A, Raskob GE et al (2010) Apixaban versus enoxaparin for thromboprophylaxis after hip replacement. N Engl J Med 363:2487–2498
Turpie AGG, Bauer KA, Eriksson BI, Lassen MR (2003) Fondaparinux vs enoxaparin for the prevention of venous thromboembolism in major orthopaedic surgery. Arch Intern Med 162:1833–1840
Pengo V, Lensing AWA, Prins MH et al (2004) Incidence of chronic thromboembolic pulmonary hypertension after pulmonary embolism. N Engl J Med 350(22):2257–2323
Spyropoulos AC, Lin J (2007) Direct medical costs of venous thromboembolism and subsequent hospital readmission rates: an administrative claims analysis from 30 managed care organizations. J Manag Care Pharm 13(6):475–486
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kinov, P., Tanchev, P.P., Ellis, M. et al. Antithrombotic prophylaxis in major orthopaedic surgery: an historical overview and update of current recommendations. International Orthopaedics (SICOT) 38, 169–175 (2014). https://doi.org/10.1007/s00264-013-2134-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00264-013-2134-8