Abstract
Purpose
To evaluate the prognostic value of metabolic parameters and bone marrow uptake (BMU) patterns on pretherapeutic 18-F-fluorodeoxyglucose (18F–FDG) positron emission tomography/computed tomography (PET/CT) in pediatric patients with neuroblastoma (NB).
Patients and methods
Forty-seven pediatric patients with newly diagnosed neuroblastoma who underwent 18F–FDG PET/CT were retrospectively reviewed. Clinicopathological factors and metabolic parameters including maximum standardized uptake value (SUVmax), metabolic tumor volume (MTV), total lesion glycolysis (TLG) and bone marrow uptake patterns on PET/CT were compared to predict recurrence-free survival (RFS) and overall survival (OS) by univariate and multivariate analysis.
Results
During the follow-up period, 27 (57.4%) patients experienced recurrence. MTV (P = 0.001), TLG (P = 0.004) and BMU patterns (P = 0.025) remained significant predictive factors for tumor recurrence, along with tumor size, histology, stage, lactate dehydrogenase (LDH) and other distant metastasis (except bone metastasis). Univariate analysis showed that histology, stage, tumor size (>37.25 cm), other distant metastasis, MTV (>88.10cm3) and TLG (>1045.2 g) and BMU patterns correlated with both RFS and OS (P < 0.05). On multivariate analysis, TLG remained the only independent prognostic factor for RFS (P = 0.016) and OS (P = 0.012), and BMU patterns and MTV were statistically significant for OS (P = 0.024 and P = 0.038, respectively).
Conclusion
Pretherapeutic 18F-FDG PET/CT can provide reliable prognostic information for neuroblastoma pediatric patients, and patients with high MTV, TLG and focal bone marrow (unifocal and multifocal) uptake on PET/CT may have inferior outcomes during subsequent treatment.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Neuroblastoma is the most common extracranial solid tumor in children, and accounts for 7% of children’s malignancies and 15% of pediatric oncology deaths [1,2,3]. More than half of patients with neuroblastoma are diagnosed as high-risk categories with poor prognosis. The unfavorable prognostic features include age (≥18 mo at presentation), stage (distant metastases in lymph nodes, cortical bone, bone marrow, and liver), laboratory test parameters (high levels of ferritin, neuron-specific enolase (NSE), LDH, and urinary catecholamine metabolites) and molecular pathology (MYCN oncogene amplification and chromosomal loss of 1p36) [3,4,5,6,7,8]. Bone and bone marrow are the most frequently involved sites, and as a predictor of poor prognosis, bone marrow involvement can be used to evaluate the disease response instead of observing the development of the greater tumor burden [9,10,11].
18F–FDG PET/CT has been increasingly used in pediatric neuroblastoma for diagnosis staging and predicting prognosis [12,13,14,15,16]. Although the maximum standardized uptake value (SUVmax) on 18F–FDG PET/CT can estimate a tumor’s metabolic activity, it cannot reflect the metabolic activity of the whole tumor. The volumetric imaging parameters based on 18F–FDG PET/CT, including metabolic tumor volume (MTV) and total lesion glycolysis (TLG), have also been suggested as prognostic factors for various tumors [17,18,19,20]. To our knowledge, there are few studies that have evaluated the parameters of volume and bone marrow on PET/CT as prognostic factors in patients with neuroblastoma [14]. Hence, the aim of this study was to evaluate the prognostic value of metabolic parameters and BMU patterns on pretherapeutic 18F–FDG PET/CT in pediatric patients with neuroblastoma, comparing with clinical indices.
Methods
Patients
We retrospectively reviewed the record of 248 pediatric patients (age < 18 years) with newly diagnosed neuroblastoma from January 2012 to December 2016 in our department of Xinhua Hospital affiliated to Shanghai Jiaotong University. The exclusion criteria included: patients with a history of surgery (n = 53), receiving chemotherapy before 18F–FDG PET/CT imaging (n = 127) or incomplete clinical information (n = 31). Forty-seven consecutive patients with pathologically confirmed NB were enrolled in this study. The diagnosis was established by ultrasonography-guided fine-needle aspiration or biopsy and the final post-operation pathology. All patients underwent 18F–FDG PET/CT imaging within 2 weeks before the beginning of treatment. The data obtained from the clinical medical records included age, sex, stage, pathological characteristics, laboratory test parameters, treatment and follow-up information.
All patients were followed up at least 6 months, and the mean duration was 24.34 ± 15.71 (range, 6-69mo). For histology results, tumors were classified as undifferentiated neuroblastoma, poorly differentiated neuroblastoma, differentiated neuroblastoma, and ganglioneuroblastoma. Bone marrow involvement (BMI) was verified by bone marrow aspirate or biopsy (BMB) or trephine biopsy containing unequivocal tumor cells. Metastatic lesions found on any image through the study period were recorded. Blood and urine tests and diagnostic imaging examinations were performed every 3–6 months during the follow-up period after therapy. This retrospective study was approved by the Institutional Review Board of our hospital, and requirement to obtain informed consent was waived.
18F–Fdg pet/ct
All PET/CT scans were performed on the PET/CT scanner (Siemens Biograph MCT). All patients were asked to fast for at least 6 h before FDG PET/CT scan. Children underwent whole-body scan after approximately 1 h after intravenous injection of FDG (5 MBq/kg). The whole-body low-dose CT imaging was performed from the skull to the upper part of the thigh or feet without contrast medium. The PET scans were obtained immediately after a whole body CT scan. Sedation was performed 30–40 min before scanning if necessary.
Image interpretation
18F–FDG PET/CT images were independently reviewed by two experienced nuclear medicine physicians on the workstation. The volumes of interest were placed over the tumor based on axial, coronal, and sagittal projections. Cross-sectional circles were drawn in all three projections to ensure three-dimensional (3D) coverage of the entire primary tumor. SUVmax, the average of SUV (SUVmean), and MTV were measured by volume viewer software on PET/CT images. SUVmax was calculated as (decay-corrected activity/tissue volume) / (injected dose/body weight). MTV were drawn by using the SUV-based threshold method on the primary tumor without the metastatic lesions. In our study, 40 % SUVmax was chosen as the threshold for generating the volume of interest based on methods which were recommended previously [21,22,23]. If 40% of the primary tumor SUVmax was below 2.5, an SUV of 2.5 was used as the threshold. TLG is the product of SUVmean and MTV, which calculate the volume of the metabolically active areas of the tumor. All the parameters of SUV were calculated by SUVmax of the lesion divided by the mean SUV of the liver.
Based on clinical observations and bone marrow uptake patterns on PET/CT imaging in Hodgkin’s lymphoma (HL) [24], the patterns of BMU on PET/CT imaging were classified into three types: normal uptake (absence of uptake or intensity equal to or lower than the liver with homogeneous metabolism), diffusely increased uptake (diffuse uptake of the whole bone marrow with an intensity higher than the liver), and focal bone lesion (focally increased uptake with one or more lesions and with or without highly diffuse uptake of the whole bone marrow). Since diffuse BMU of 18F–FDG could reflect two phenomena, tumor infiltration or myeloid hyperplasia reacted by inflammation, patients who underwent 18F–FDG PET/CT and had abnormal hematological parameters (especially CRP) with or without a fever within a week were excluded [25]. No patient had a history of using drugs to increase leukocytes in our study. Hence, the criteria of BMI on 18F–FDG PET/CT image included focal bone lesion or diffuse uptake in bone marrow with an intensity higher than the liver.
Statistical analysis
Statistical analyses were performed using SPSS Software (version 20.0 for Windows; SPSS INC.). Contiguous data were describe as mean ± standard deviation (mean ± SD) or median and range, based on whether they followed the normal distribution, and categorical variables as numbers. PET/CT parameters were compared with clinicopathological factors between recurrence group and non-recurrence group using independent t texts, Mann-Whitney U tests, X2tests, and Fisher exact tests. Recurrence-free survival (RFS) was defined as the time from the day of diagnosis to evident tumor recurrence on any radiographic examination. Over survival (OS) was defined as the time between the date of the initial 18F-FDG PET/CT scan and the date of death or the last follow-up.
Receiver operating characteristic (ROC) analysis was used to evaluate tumor size, SUVmax, MTV and TLG in predicting disease recurrence. Survival curves were estimated using the Kaplan-Meier method to calculate cumulative RFS rates and OS rates. The significance of the predictive value of each variable was evaluated using log-rank tests on univariate analysis and COX proportional hazards regression tests were used in multivariate analysis. The uptake of bone marrow on 18F–FDG PET/CT image, including SUVmax and the patterns of lesions between the positive bone marrow biopsy group and the negative group, were estimated using Mann-Whitney U tests and χ2 tests, respectively. All tests were two-sided, and a probability of less than 0.05 was considered statistically significant.
Results
This study consisted of 32 males and 15 females with a median age of 2 years (range, 2.0 months-8.0 years). The tumors were staged based on the International Neuroblastoma Staging System (INNC), and all patients belonged to stage III or IV, except one patient who was in stage I. All children received therapy according to their clinical staging and risk stratification as follows: 15 children underwent surgical resection with adjuvant chemotherapy, 22 patients underwent preoperative chemotherapy before surgery and postoperative adjuvant chemotherapy, 3 patients twice underwent surgical resections and chemotherapy, 5 patients underwent chemotherapy and radiotherapy after surgical resection, and 2 patients had surgery, chemotherapy and autologous peripheral blood stem cell transplantation. During the period of the clinical follow up, 27 (57.4%) patients experienced recurrence, and of all patients, 18 (38.3%) died. The characteristics of the patients are shown in Table 1.
In our study, there are significant differences in tumor size, histology, stage, LDH, other distant metastasis, BMU patterns, MTV and TLG between patients with recurrence and those without (P < 0.05); age, sex, ferritin, NSE, urinary catecholamine (VMA and HVA), tumor location, lymph node metastasis, bone marrow metastasis, and SUVmax showed no significance between the two groups (P > 0.05).
Survival analysis
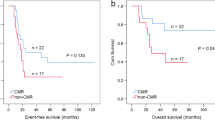
Histology, stage, age, LDH, tumor size, lymph node metastasis, bone metastasis, BMU patterns, other distant metastasis (except bone metastasis), infiltrating across the midline, SUVmax, MTV and TLG were evaluated as variables in the survival analysis of NB. From the receiver-operating-characteristic curve (ROC) analyses, the optimal cutoff values for tumor size, LDH, SUVmax, MTV, and TLG were 37.25 mm, 1011 IU/L, 4.15, 88.10cm3, and 1045.20 g, with the AUCs of 0.715, 0.782, 0.542, 0.778 and 0.748, respectively. The significance of variables for predicting RFS and OS on univariate analysis is shown in Table 2. The histology, stage, tumor size, other distant metastasis, MTV (Fig. 1a), TLG (Fig. 1c) and BMU patterns (Fig. 1e) showed significant differences on univariate analysis for RFS. Similarly, the histology, stage, other distant metastasis, MTV (Fig. 1b), TLG (Fig. 1d) and BMU patterns (Fig. 1f) were the significant prognostic factors for OS. For the univariate analysis, the children with an MTV greater than 88.10cm3 showed lower RFS rates (41.9% vs.87.5%, P = 0.005) and OS rates (48.3%vs. 87.5%, P = 0.008) than those with an MTV of 88.10 cm3 or less; the children with a TLG greater than 1045.20 g also had lower RFS rates (48.6% vs.81.4%, P = 0.008) and OS rates (51.8% vs. 84.2%, P = 0.022) than those with a TLG of 1045.20 g or less. In our study, the index of SUVmax was not a significant prognosis factor for either RFS (P = 0.303) or OS (P = 0.333).
Cumulative RFS curves according to MTV (a), TLG (c) and BMU patterns (e) and cumulative OS curves according to MTV (b), TLG (d)and BMU patterns (f) of neuroblastoma lesions in enrolled patients (n = 47). (a and b) Upper line MTV ≤ 88.10 cm3, N = 17, lower line MTV > 88.10 cm3, N = 30. (c and d) Upper line TLG ≤ 1045.20 g, N = 18, lower line TLG > 1045.20 g, N = 29. (e and f) Upper line normal uptake, N = 17, middle line diffuse uptake, N = 10, lower line focal uptake, N = 20
Variables with a P value of less than 0.05 were enrolled in multivariate analysis using Cox proportional hazards regression. As expected, TLG was calculated by multiplying MTV and the mean SUV, and there was a significant correlation between MTV and TLG (r = 0.943,P < 0.001). Therefore, the two different variables of MTV and TLG were assessed separately for multivariate analyses. TLG was determined to be the only independent significant prognosis factor for RFS and OS, and BMU patterns, MTV and TLG were statistically significant for OS. Histology, stage, BMU patterns showed marginal significance for RFS, and histology and other distant metastasis showed marginal significance for OS. The significant variables for predicting RFS and OS on multivariate analysis are shown in Tables 3 and 4. There were three patterns of BMU of FDG, and we found that focal bone lesions on PET/CT scan had lower survival rate than diffuse uptake pattern.
Bone marrow metastasis analysis
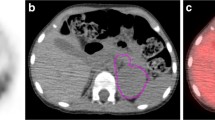
Of 47 pediatric patients, 26 patients underwent bone marrow aspirate or trephine biopsy containing tumor cells, and then BMI were verified in 16 patients. In this study, 10 (62.5%) patients had focal bone marrow lesions (Fig. 2a), and 6 (37.5%) had diffuse FDG uptake (Fig. 2b). When considering only positive BMB as a reference standard, the sensitivity, specificity and accuracy of PET/CT on diagnosis of BMI were 100% (16/16), 50% (5/10) and 80.7% (21/26), respectively. PET also detected two multifocal BMI with negative BMB which were verified by guided biopsy of the PET-positive sites during the follow-up visit in our study. For these patients with confirmed bone marrow results, clinical metastasis highly associated with the patterns of bone marrow on PET/CT image (χ2 =8.53, P = 0.014 < 0.05), and patients with the focal bone lesions were more likely to be metastatic lesions than those with diffuse FDG uptake in bone marrow. No significant difference was noted for SUVmax between patients with or without bone marrow metastasis according to the result of BMB (U = 39.00, P = 0.067).
A. Example of normal BM uptake < liver uptake on 18FDG-PET/CT with negative BMB (coronal slices of CT (a), PET (b), fused PET/CT (c) and MIP (d)). The patient had no recurrence (follow-up: +47 months). B. Example of a pure diffuse BM uptake > liver uptake on 18FDG-PET/CT with negative BMB (coronal slices of CT (a), PET (b), fused PET/CT (c) and MIP (d)). The patient was in complete remission (follow-up: +20 months). C. Example of a multifocal BM lesions on 18FDG-PET/CT and with confirmed positive BMB (coronal slices of CT (a,d), PET (b,e), fused PET/CT (c,f) and MIP (d)). The patient relapsed after 6 courses of chemotherapy (follow-up: +13 months)
Discussion
In the present study of 47 pediatric patients with NB, we investigated the prognostic value of SUVmax, MTV, and TLG of 18F–FDG PET/CT indices, compared with the conventional clinical parameters, and gave a preliminary description of bone marrow FDG uptake patterns for assessing the BMI on pretherapeutic PET/CT in patients with neuroblastoma. Our results demonstrated that TLG is determined to be the only independent significant prognosis factor for RFS and OS, and MTV and BMU patterns are the independent prognostic factors for predicting OS, comparable to histology, stage, other distant metastasis and MTV. In addition, MTV, TLG and BMU pattern are also determined to be statistically significant predictors of recurrence. For bone marrow assessment by PET/CT, the pattern of focal bone lesions should be considered as a harbinger of bone metastases and patients having shorter OS time.
18F–FDG PET/CT has been increasingly used to predict survival prognosis and therapy response for malignancies; nevertheless, only one study has evaluated the value of volumetric parameters on PET/CT for risk stratification in patients with neuroblastoma [14]. Chia-Ju Liu et al. [14] performed pretreatment 18F–DOPA and 18F–FDG PET in 25 children and provided complementary information that lower 18F–DOPA uptake and higher 18F–FDG uptake were associated with poor prognosis, and volumetric indices correlate highly with risk grouping. In this study, we enrolled 47 pediatric patients with newly diagnosed neuroblastoma to evaluate the prognostic value of volumetric parameters measured on PET/CT in different models, compared with conventional prognostic factors. The results of our study showed that TLG is the independent significant prognosis factor, and had a stronger association with RFS (P = 0.016) and OS (P = 0.012) than MTV and SUVmax. SUVmax represents the maximum uptake of region of interest (ROI) on PET/CT. MTV is defined as the volume of the tumor representing metabolic activity and indicating the distribution of18F–FDG uptake. TLG is representative of the metabolic activity throughout the entire tumor lesion above a minimum threshold designed to exclude background activity. A large TLG may reflect a small volume of tumor tissue with high 18F–FDG uptake or a large volume of tumor tissue with low 18F–FDG uptake [20]. Hence, compared with SUVmax, MTV and TLG can more accurately reflect the metabolic burden of tumor lesions and predict survival prognosis. Our results also demonstrated that pediatric patients who showed higher MTV (>88.10 cm3) and TLG (>1045.20 g), focal bone lesions, higher serum level of LDH (>1011.00 IU/L), larger tumor size (>37.20 mm) and other distant metastasis were more likely to relapse than those who did not.
Metastasis of NB occurs through lymphatic and hematogenous routes, with involvement of bone, bone marrow, liver and skin [1, 8]. In approximately 70% of patients, metastasis is present at the time of diagnosis and most commonly involves cortical bone and bone marrow. 18F–FDG PET, MIBG scintigraphy [26] and bone scan (BS) have different advantages in detecting bone metastasis, however, there is no consistent conclusion as to which was better [27]. The study of Yun Jung Choi et al. found 18FDG PET was superior to BS in detecting bone metastasis (100% vs. 94.4% insensitivity, 100% vs. 77.8% in specificity) [28]. In our study, the results showed that the sensitivity, specificity and accuracy of PET/CT on diagnosis of bone marrow metastasis were 100%, 50% and 80.7% in 26 patients, considering BMB as the only gold standard for BMI. In addition, PET detected multifocal bone marrow involvement in two NB patients with negative BMB in this study. It is well known that BMB cannot be considered as a histological gold standard for bone marrow infiltration, because it allows the analysis of a very limited area and can consequently miss the lesions not located in the iliac crest. Our results suggest that focal BMU of FDG on PET/CT could detect marrow metastasis more accuracy compared to BMB. The conceivable reasons for the low specificity of PET/CT may include high physiological uptake and non-inflammatory myeloid hyperplasia response in patients. Though there existed some limitations in detecting bone marrow metastasis by FDG PET/CT, Papathanasiou et al. [15] found that FDG uptake of the tumor and the FDG skeletal extent were significant prognostic factors for overall survival in contrast to the I-123 MIBG skeletal score. In the present study, bone/bone marrow metastasis is associated with BMU patterns rather than the SUVmax of bone marrow. The results also demonstrated that the focal bone lesions on PET/CT imaging probably indicate bone marrow metastasis with an accuracy up to 85.7%. Bone/bone marrow involvement pattern was the significant independent prognostic factor for OS, and patients with focal bone marrow lesions on preoperative 18F–FDG PET/CT imaging can be assumed to have lower OS rates, and intensive adjuvant treatment or close follow-up is needed with or without surgical resection for these patients. To our knowledge, this is the first study using the BMU patterns together with other 18F–FDG PET parameters to evaluate the prognostic values at the time of neuroblastoma diagnosis.
The limitations of this study are as follows. First, our study had a relatively small sample size, because the pediatric patients were excluded without certified pathology or with therapy before PET/CT examination. Second, only the primary tumor was calculated in the volumetric parameters as whole-body metabolic burden, without metastatic lesions, because the aforementioned technical difficulty further limited this study. Third, necrotic lesions are commonly seen in huge neuroblastoma and partial-volume effects could affect the 18F–FDG uptake, which would be underestimated in the values of MTV and TLG. Finally, as a retrospective single-center study, the results might be subject to selection bias.
Conclusion
Of the parameters of 18F–FDG PET/CT, TLG is the only independent prognosis factor for the RFS and OS, compared with SUVmax, MTV, and other conventional prognostic factors. MTV and patterns of BMU on PET/CT are the independent prognosis factors for OS. Only focal bone lesion uptake should be considered as a harbinger of bone marrow metastasis in neuroblastoma patients. Pretherapeutic MTV, TLG and patterns of BMU may play important roles on planning treatment schedules and follow-up, and large-scale prospective trials are also needed to validate the results by longer follow-up.
References
Maris JM, Hogarty MD, Bagatell R, Cohn SL. Neuroblastoma Lancet. 2007;369(9579):2106–20. https://doi.org/10.1016/S0140-6736(07)60983-0.
Park JR, Eggert A, Caron H. Neuroblastoma: biology, prognosis, and treatment. Hematol Oncol Clin North Am. 2010;24(1):65–86. https://doi.org/10.1016/j.hoc.2009.11.011.
Spix C, Pastore G, Sankila R, Stiller CA, Steliarova-Foucher E. Neuroblastoma incidence and survival in european children (1978-1997): report from the automated childhood cancer information system project. Eur J Cancer. 2006;42(13):2081–91. https://doi.org/10.1016/j.ejca.2006.05.008.
Brodeur GM. Neuroblastoma: biological insights into a clinical enigma. Nat Rev Cancer. 2003;3(3):203–16. https://doi.org/10.1038/nrc1014.
Brodeur GM, Fong CT, Morita M, Griffith R, Hayes FA, Seeger RC. Molecular analysis and clinical significance of N-myc amplification and chromosome 1p monosomy in human neuroblastomas. Prog Clin Biol Res. 1988;271:3–15.
Brodeur GM, Seeger RC, Barrett A, Berthold F, Castleberry RP, D’Angio G, et al. International criteria for diagnosis, staging, and response to treatment in patients with neuroblastoma. J Clin Oncol. 1988;6(12):1874–81. https://doi.org/10.1200/JCO.1988.6.12.1874.
Brodeur GM, Seeger RC, Schwab M, Varmus HE, Bishop JM. Amplification of N-myc in untreated human neuroblastomas correlates with advanced disease stage. Science. 1984;224(4653):1121–4.
Maris JM. Recent advances in neuroblastoma. N Engl J Med. 2010;362(23):2202–11. https://doi.org/10.1056/NEJMra0804577.
Cheung NK, Ostrovnaya I, Kuk D, Cheung IY. Bone marrow minimal residual disease was an early response marker and a consistent independent predictor of survival after anti-GD2 immunotherapy. J Clin Oncol. 2015;33(7):755–63. https://doi.org/10.1200/JCO.2014.57.6777.
Stutterheim J, Zappeij-Kannegieter L, Versteeg R, Caron HN, van der Schoot CE, Tytgat GA. The prognostic value of fast molecular response of marrow disease in patients aged over 1 year with stage 4 neuroblastoma. Eur J Cancer. 2011;47(8):1193–202. https://doi.org/10.1016/j.ejca.2011.02.003.
Viprey VF, Gregory WM, Corrias MV, Tchirkov A, Swerts K, Vicha A, et al. Neuroblastoma mRNAs predict outcome in children with stage 4 neuroblastoma: a european HR-NBL1/SIOPEN study. J Clin Oncol. 2014;32(10):1074–83. https://doi.org/10.1200/JCO.2013.53.3604.
Bleeker G, Tytgat GA, Adam JA, Caron HN, Kremer LC, Hooft L, et al. 123I-MIBG scintigraphy and 18F-FDG-PET imaging for diagnosing neuroblastoma. The Cochrane database of systematic reviews. 2015;9:CD009263. https://doi.org/10.1002/14651858.CD009263.pub2.
Kushner BH, Yeung HW, Larson SM, Kramer K, Cheung NK. Extending positron emission tomography scan utility to high-risk neuroblastoma: fluorine-18 fluorodeoxyglucose positron emission tomography as sole imaging modality in follow-up of patients. J Clin Oncol. 2001;19(14):3397–405. https://doi.org/10.1200/JCO.2001.19.14.3397.
Liu CJ, Lu MY, Liu YL, Ko CL, Ko KY, Tzen KY, et al. Risk stratification of Pediatric patients with Neuroblastoma using volumetric parameters of 18F-FDG and 18F-DOPA PET/CT. Clin Nucl Med. 2017;42(3):e142–e8. https://doi.org/10.1097/RLU.0000000000001529.
Papathanasiou ND, Gaze MN, Sullivan K, Aldridge M, Waddington W, Almuhaideb A, et al. 18F-FDG PET/CT and 123I-metaiodobenzylguanidine imaging in high-risk neuroblastoma: diagnostic comparison and survival analysis. J Nucl Med. 2011;52(4):519–25. https://doi.org/10.2967/jnumed.110.083303.
Sharp SE, Shulkin BL, Gelfand MJ, Salisbury S, Furman WL. 123I-MIBG scintigraphy and 18F-FDG PET in neuroblastoma. J Nucl Med. 2009;50(8):1237–43. https://doi.org/10.2967/jnumed.108.060467.
Fendler WP, Philippe Tiega DB, Ilhan H, Paprottka PM, Heinemann V, Jakobs TF, et al. Validation of several SUV-based parameters derived from 18F-FDG PET for prediction of survival after SIRT of hepatic metastases from colorectal cancer. J Nucl Med. 2013;54(8):1202–8. https://doi.org/10.2967/jnumed.112.116426.
Lee JW, Kang CM, Choi HJ, Lee WJ, Song SY, Lee JH, et al. Prognostic value of metabolic tumor volume and total lesion glycolysis on preoperative (1)(8)F-FDG PET/CT in patients with pancreatic cancer. Journal of nuclear medicine : official publication, Society of Nuclear Medicine. 2014;55(6):898–904. https://doi.org/10.2967/jnumed.113.131847.
Oh JR, Seo JH, Chong A, Min JJ, Song HC, Kim YC, et al. Whole-body metabolic tumour volume of 18F-FDG PET/CT improves the prediction of prognosis in small cell lung cancer. Eur J Nucl Med Mol Imaging. 2012;39(6):925–35. https://doi.org/10.1007/s00259-011-2059-7.
Ryu IS, Kim JS, Roh JL, Lee JH, Cho KJ, Choi SH, et al. Prognostic value of preoperative metabolic tumor volume and total lesion glycolysis measured by 18F-FDG PET/CT in salivary gland carcinomas. J Nucl Med. 2013;54(7):1032–8. https://doi.org/10.2967/jnumed.112.116053.
Choi ES, Ha SG, Kim HS, Ha JH, Paeng JC, Han I. Total lesion glycolysis by 18F-FDG PET/CT is a reliable predictor of prognosis in soft-tissue sarcoma. Eur J Nucl Med Mol Imaging. 2013;40(12):1836–42. https://doi.org/10.1007/s00259-013-2511-y.
Lee JA. Segmentation of positron emission tomography images: some recommendations for target delineation in radiation oncology. Radiother Oncol. 2010;96(3):302–7. https://doi.org/10.1016/j.radonc.2010.07.003.
Maffione AM, Ferretti A, Grassetto G, Bellan E, Capirci C, Chondrogiannis S, et al. Fifteen different 18F-FDG PET/CT qualitative and quantitative parameters investigated as pathological response predictors of locally advanced rectal cancer treated by neoadjuvant chemoradiation therapy. Eur J Nucl Med Mol Imaging. 2013;40(6):853–64. https://doi.org/10.1007/s00259-013-2357-3.
Zwarthoed C, El-Galaly TC, Canepari M, Ouvrier MJ, Viotti J, Ettaiche M, et al. Prognostic value of bone marrow tracer uptake pattern in baseline PET scan in Hodgkin lymphoma: results from an international collaborative study. J Nucl Med. 2017; https://doi.org/10.2967/jnumed.116.184218.
Salaun PY, Gastinne T, Bodet-Milin C, Campion L, Cambefort P, Moreau A, et al. Analysis of 18F-FDG PET diffuse bone marrow uptake and splenic uptake in staging of Hodgkin’s lymphoma: a reflection of disease infiltration or just inflammation? Eur J Nucl Med Mol Imaging. 2009;36(11):1813–21. https://doi.org/10.1007/s00259-009-1183-0.
Bombardieri E, Giammarile F, Aktolun C, Baum RP, Bischof Delaloye A, Maffioli L, et al. 131I/123I-metaiodobenzylguanidine (mIBG) scintigraphy: procedure guidelines for tumour imaging. Eur J Nucl Med Mol Imaging. 2010;37(12):2436–46. https://doi.org/10.1007/s00259-010-1545-7.
Brisse HJ, McCarville MB, Granata C, Krug KB, Wootton-Gorges SL, Kanegawa K, et al. Guidelines for imaging and staging of neuroblastic tumors: consensus report from the international Neuroblastoma Risk Group Project. Radiology. 2011;261(1):243–57. https://doi.org/10.1148/radiol.11101352.
Choi YJ, Hwang HS, Kim HJ, Jeong YH, Cho A, Lee JH, et al. (18)F-FDG PET as a single imaging modality in pediatric neuroblastoma: comparison with abdomen CT and bone scintigraphy. Ann Nucl Med. 2014;28(4):304–13. https://doi.org/10.1007/s12149-014-0813-1.
Funding
On behalf of all authors, the corresponding author states that no funding was received for this study.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflicts of interest.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Rights and permissions
About this article
Cite this article
Li, C., Zhang, J., Chen, S. et al. Prognostic value of metabolic indices and bone marrow uptake pattern on preoperative 18F–FDG PET/CT in pediatric patients with neuroblastoma. Eur J Nucl Med Mol Imaging 45, 306–315 (2018). https://doi.org/10.1007/s00259-017-3851-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00259-017-3851-9