Abstract
Background
Magnetic resonance imaging (MRI) is often the first modality of choice in the detection of pituitary pathologies due to its excellent contrast resolution and high spatial resolution. However, evaluating the size of the pituitary gland is somewhat difficult since the gland morphology varies widely among individuals.
Objective
The aim of this study was to provide normal reference values for pituitary volumes in the pediatric population using three-dimensional (3-D) MRI data. We also aimed to compare the volumetric data measured through 3-D images with the mathematical volumes obtained by the ellipsoid formula from 2-D images, considering the demographic subgroups of age and sex.
Materials and methods
This study was performed retrospectively using PACS-archived brain MRIs obtained in the Pediatric Radiology Unit. Eight hundred children under the age of 18 who underwent brain MRI between January 2014 and May 2018 for headaches, seizures or trauma and who had no brain anomaly or endocrine pathology were recruited. Pituitary volumes were separately calculated from those brain MRIs by means of ellipsoid formula and volumetry. The mean, standard deviation, maximum and minimum and median values were achieved for each 1-year age group in both sexes. Normal reference ranges containing 5th -10th -25th -50th -75th -90th -95th percentiles of volumetric pituitary values were tabulated.
Results
Pituitary gland volumes in both sexes were found to increase gradually with age. The average 3-D volume values were greater in girls than boys except for the age groups 1–2 years and 8–9 years. Pituitary volumes showed a growth spurt during early puberty, which was more prominent in girls. Moreover, a strong positive correlation was noted between the 2-D calculated and 3-D measured volumes in both sexes.
Conclusion
Normative data obtained through this study can be used for clinical diagnostic purposes as well as a reference for future research. The 2-D calculated volumes be used where it is not possible to obtain 3-D volumetric values.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
The use of magnetic resonance imaging (MRI), especially in the evaluation of neurological and endocrine disorders, has increased dramatically in the last two decades. Advances in MRI technology together with the emergence of novel techniques now allow better delineation of small structures like the pituitary gland. Although most of the pediatric pituitary gland disorders such as tumors and midline malformations can be easily detected on routine MRI, disorders related to gland size are somewhat difficult to evaluate due to wide variation in normal gland morphology [1, 2]. It is also a well-known fact that gland size changes considerably over the course of life, based on age, sex and hormonal status [3,4,5,6,7,8]. Therefore, volumetric normative data are strongly needed, especially in the pediatric population, to evaluate pituitary gland size.
The majority of reports to date regarding pituitary gland size or volume are mainly based on a surrogate marker such as the gland height or rough volume estimates indirectly calculated from two-dimensional (2-D) data sets and have mostly been obtained in adults [3,4,5,6,7,8,9,10,11,12,13]. Only a few studies have been conducted in children using 3-D volumetry. Two of them are limited to a specific ethnicity, namely Japanese and Chinese, and the other two either considered only the anterior pituitary gland volume or pituitary volumes in prepubertal children [1, 2, 14, 15].
In this retrospective study, we aimed to provide normal reference values for pituitary volumes in the pediatric population using 3-D MRI data. The secondary aim was to compare the 3-D volumes with the mathematical volumes obtained by ellipsoid formula from 2-D data sets, considering the demographic subgroups of age and sex.
Materials and methods
The study received ethics approval from the ethics in human research committee (Decree No: 08–516-18 and Date: 07.05.2018). Written informed consent from the parents or guardians of the children was waived due to the retrospective nature of the study.
Subjects
After reviewing the relevant patient files, 800 children under the age of 18 undergoing brain MRI at our institution between January 2014 and May 2018 for headache, idiopathic seizure or trauma and who had no obvious brain anomaly were included. Any child with a history of preterm delivery at <35 weeks, breech presentation or asphyxia at birth was excluded. Those children with known endocrinologic abnormality or genetic syndrome were not included in the study. MR images degraded by means of any artifacts were excluded.
MR acquisition and measurements
All MR examinations were performed using a 1.5-T system (Philips Ingenia; Philips Medical Systems, Eindhoven, The Netherlands) with a 16-channel quadrature head coil. Thin section volumetric studies were obtained with 3-D isovolumetric T1-weighted imaging (T1-WI) sequence without application of contrast agent, using the following parameters: repetition time (TR)/echo time (TE) of 25/4.6 ms; matrix 148×145, reconstruction matrix 336, 200-mm field of view (FOV), 30° flip angle, section thickness of 1–1.3 mm; reconstruction voxel size of 0,56 mm without interslice gap. Total imaging time was approximately 15–20 min for each patient.
The 3-D volumes were obtained in the sagittal plane since the boundry of the gland is easy to delineate in this orientation. The cross-sectional areas (in mm2) were measured slice-by-slice with a surface area markup tool on our workstations (General Electric, Milwaukee, WI) by manually tracing the borders of the gland with a mouse-guided cursor (Fig. 1). To calculate the 3-D volume (in mm3), all areas measured on each slice were added together and then multiplied by the slice thickness (ΣArean x slice thickness). The marked-up area included the neurohypophisis, but not the pituitary stalk.
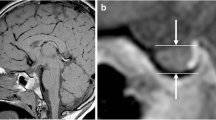
Indirect mathematical volumes were calculated by using the formula [V=(4π/3) (height x length x width)/2], which was originally derived from the formula of an ellipsoid to estimate the the volume of the sella turcica on conventional radiographs in the 1960s [16]. The height was defined as the craniocaudal straight line from the gland’s bottom to the pituitary stalk insertion point on the mid-sagittal image (Fig. 2a). The length was defined as the anteroposterior straight line perpendicular to the height on the same mid-sagittal image (Fig. 2a). The width was defined as the straight line between the cavernous sinuses’ medial borders on the mid-coronal image in which the pituitary stalk is clearly visible (Fig. 2b).
An isovolumetric T1-weighted magnetic resonance (MR) image (a) in a 16-year-old girl presenting with headache shows the height and the length measurements on the mid-sagittal plane. A coronal reformatted image, derived from the three-dimensional isovolumetric T1-weighted MR images, (b) shows the width measurement on the mid-coronal plane
All measurements were performed by a radiologist (A.Ç.) with 6 years of experience who was initially trained for the pituitary MR measurements by a radiologist (O.S.F.) with 23 years of experience in pediatric radiology. A.Ç. was blinded to demographic data of the subjects during the measurements. Three-dimensional volume measurements for 80 children were repeated after a minimum interval of 6 months to assess the intra-observer reliability.
Statistical analyses
All statistical analyses were performed using IBM Statistics Package for the Social Sciences (SPSS) version 23.0 (Armonk, NY). A P-value of <0.05 was considered statistically significant.
Volumes were measured in age-defined subgroups in both sexes. Descriptive statistics were given as numbers and percentages, while statistics regarding distribution were given as mean, standard deviation, median, maximum and minimum values. Normal distribution of the examined variables in each age group was assessed by means of Kolmogorov-Smirnov (n>30) and Shapiro-Wilk (n<30) tests. Once the distribution was stated as normal in all groups, data were analyzed by means of Student’s t-test for differences between sexes, and further investigated by means of one-way analysis of variance (ANOVA) for differences between age-defined subgroups, seperately for each sex. The significance of single differences was assessed by means of post hoc analysis with Tukey and Tamhane tests for equal and unequal sample sizes, respectively. Spearman’s correlation test was performed to assess the association between numerical variables. Reference values with 95% ranges were obtained by linear regression analyses.
The normal range of the pituitary gland volumes was expressed as the mean±standard deviation together with maximum–minimum and median values. The 5th–10th-25th-50th-75th-90th-95th percentiles of 3-D volume were calculated for each age-defined subgroup in both sexes.
Results
The MRIs of 800 children (420 girls, 380 boys) with apparently normal pituitary glands were reviewed. There was high-level intra-observer reliability in the repeated 80 3-D volume measurements, with a correlation coefficient of 0.97 (95% confidence interval [CI] 0.95–0.98).
The mean measured 3-D volume was 488.4±240.5 mm3 (range: 94–1,142 mm3; median: 444 mm3) for girls, and 399.8±187.8 mm3 (range: 85–956 mm3; median: 369.6 mm3) for boys, respectively (Tables 1 and 2, Fig. 3).
The box plots show the three-dimensional (3-D) pituitary volume in girls (a) and boys (b) across age groups. (The dark lines within each boxes represent the median, while the boxes’ lower and upper boundaries represent the 25th and 75th percentile, respectively. The circles represent the extreme values)
The mean calculated 2-D volume was 497.7±234.5 mm3 (range: 91.3–1,262.4 mm3; median: 451.3 mm3) for girls, and 417.9±186.3 mm3 (range: 106–1,078.7 mm3; median: 383.5 mm3) for boys, respectively (Tables 3 and 4).
The 5th–10th-25th-50th-75th-90th-95th percentiles of 3-D volume were calculated for each year of age in both sexes (Tables 5 and 6).
Spearman’s correlation coefficient and adjusted determination coefficient (R2) were 0.93 (95% CI 0.92–0.94) and 0.87, respectively, between the 3-D volume and 2-D volume in girls, as determined by correlation and regression analyses (Fig. 4). As indicated, the calculated 2-D volume using the formula for the volume of an ellipsoid showed strong agreement with the measured 3-D volume in girls.
Spearman’s correlation coefficient and adjusted determination coefficient (R2) were 0.9 (95% CI 0.88–0.91) and 0.81, respectively, between the 3-D volume and 2-D volume in boys, as determined by correlation and regression analyses (Fig. 5). As indicated, the calculated 2-D volume using the formula for the volume of an ellipsoid showed strong agreement with the measured 3-D volume in boys.
When 3-D volume data were further fitted to wider age groups for ease of reference (as shown in Table 7), a statistically significant difference was noted between these age groups within each sex (P<0.001). Thereafter, separate regression models were applied to the mean 3-D volume to compare the same age groups in girls and boys, and they were found to be almost identical except for the age groups 10–14 years and 15–17 years (Table 7).
In summary, a gradual increase over age in the pituitary volume of the examined children was noted. The gland showed a growth spurt in early puberty, which was more prominent in girls. The pituitary volume in girls was significantly larger than that in boys during early puberty (Fig. 6). And there was a statistically strong correlation between 3-D (measured) and 2-D (calculated) volumes in both sexes.
Discussion
MRI of the pituitary gland is advocated as a part of basal investigations in children with growth retardation, developmental delay or suspected endocrinopathy [17,18,19,20,21]. Although pituitary gland disorders such as tumors and midline malformations can be easily detected on MRI, changes in gland size are somewhat difficult to evaluate due to wide variation in normal gland morphology [1, 2]. Variations in normal pituitary gland shape, together with the paucity of normative volumetric data in the pediatric population, result in diagnostic difficulties and cause subjective judgments [11].
Most of the studies to date have either accepted gland height as a surrogate marker for size or have calculated gland volume from 2-D data sets by using mathematical formulas such as that of the ellipsoid [3,4,5,6,7,8,9,10,11,12,13]. However, taking the aforementioned facts into consideration, the assessment of the gland size may be subject to a high degree of imprecision unless 3-D volumetry is performed. Therefore, more recent studies have focused on 3-D volumetric methods in order to make more precise assessment of the gland size [1, 2, 14, 15, 22].
In this study, we provide normative data for both 3-D measured volumetric volumes and 2-D calculated volumes in age-defined subgroups for each sex. We noted a gradual increase over age in the pituitary volume of the children we examined. The pituitary gland showed a growth spurt in early puberty, which was more prominent in girls. The pituitary volume in girls was significantly larger than that in boys during early puberty. There was a statistically strong correlation between 3-D (measured) and 2-D (calculated) volumes in both sexes.
A relatively old study by Takano et al. [15] including 199 Japanese children, 121 of whom were under the age of 10, also documented that there was a growth spurt in early puberty and this spurt was more prominent in girls. In their study, the pituitary volume was higher in girls except for the age group 1–4 years, which was partly consistent with our results. Another discrepancy compared with our results was that the pituitary volume values reported by Takano et al. [15] were slightly lower. The reasons for the difference may be due to ethnicity, sample size or age group differences.
In a European cohort by Marziali et al. [14], including 49 girls and 46 boys between the age of 2 months and 10 years, only anterior pituitary volumes were measured and the mean values were again given in relatively broad age groups (2–12 months, 1–4 years, 5–10 years). Although this study’s design was different from ours, we could say that their finding of a gradual increase over age in the anterior pituitary volume was in line with our results, taking into consideration that the anterior pituitary constitutes the majority of the gland’s volume.
In a study by Fink et al. [1] including 57 girls and 82 boys under the age of 10, it was demonstrated that volumetric measurements using either sagittal or coronal plane produced almost identical results. Contrary to our results, Fink et al. [1] found a weak correlation (Spearman’s correlation coefficient 0.46; CI 0.37–0.54) between the measured and the calculated pituitary volumes. This conflict may be due to the smaller sample size in their study, which is susceptible to random variation. Other explanations for the discrepancy may be due to the fact that they only included prepubertal children in their study and did not consider sex differences.
In a more recent study by Han et al. [2], the control group included 75 healthy Chinese children and young adults between the ages of 1–19 years. In their study, the mean 3-D volumes obtained from sagittal images were given in relatively broad age groups. Han et al. [2] found that the pituitary volume in girls was higher than that in boys except for the age group 1–4 years. They also demonstrated that there was a gradual increase over age in the pituitary volume as well as a growth spurt in the age group 10–14 years being more prominent in girls, which was consistent with our findings.
Another study conducted among Turkish children by Sari et al. [11], including 261 girls and 256 boys under the age of 18, provided normative data for pituitary volume obtained by using the mathematical formula of the ellipsoid. However, there appears to be a noteworthy discrepancy compared with our results in that pituitary volume values reported by Sari et al. [11] are significantly lower. Although it is not clear, we speculate that extreme volume values in their study may be related to possible endocrinopathies, since Sari et al. [11] did not take into account the clinical and laboratory data. In their study, Sari et al. [11] also documented that the pituitary gland showed a gradual increase in volume over age as well as a growth spurt in early puberty and the pituitary volume of girls was greater than that of boys in general.
In a recent study, Aksan et al. [23] provided normative volumetric data for pituitary volume, obtained from the MR images of 240 healthy Turkish children under the age of 12 years. Compared with our results, the pituitary volume values reported by Aksan et al. [23] are slightly higher in some age groups. The reason for this differance may be due to the smaller sample size in their study. Aksan et al. [23] affirmed that the pituitary gland showed a gradual increase in volume over age and the pituitary volume in girls was larger than that in boys above the age of 6 years.
The major strength of this study is the large sample size of 800 subjects from an ethnically diverse population, which is by far the largest study in children. Having a far greater sample size makes our study statistically more powerful in finding significant differences and less vulnerable to random variation. Another strength of this study is the higher image resolution than most of the previous studies, allowing us to make more precise measurements. MR scans included in our study were performed without gadolinium contrast agent. Although this is not a limitation in measuring the pituitary volume and dimensions precisely, we might have recruited children with imperceivable microadenomas on non-contrast-enhanced MR images. However, given the low prevalance of such pathologies among the general pediatric population, we do not think there would be a substantial interference with our results. Another drawback of this study is the lack of a thorough endocrinologic evaluation for all the subjects, as we included MR scans of those children presenting to our institution with headache, seizures or trauma.
Conclusion
In this study, we present normative data for both 3-D measured volumetric volumes and 2-D calculated volumes for each year of age and both sexes. We noted a gradual increase over age in the pituitary volume of children under the age of 18. The pituitary volume in girls was larger than that in boys except for the age groups 1–2 years and 8–9 years. The pituitary gland showed a growth spurt in early puberty, which was more prominent in girls. As opposed to existing literature, we do find it appropriate to use 2-D calculated volume when 3-D volumetry is not possible, given the statistically strong correlation between the two in our study.
References
Fink AM, Vidmar S, Kumbla S et al (2005) Age-related pituitary volumes in prepubertal children with normal endocrine function: volumetric magnetic resonance data. J Clin Endocrinol Metab 90:3274–3278
Han X, Xiu J, Huang Z et al (2014) Three-dimensional magnetic resonance volumetry of the pituitary gland is effective in detecting short stature in children. Exp Ther Med 8:551–556
Doraiswamy PM, Potts JM, Axelson DA et al (1992) MR assessment of pituitary gland morphology in healthy volunteers: age- and gender-related differences. AJNR Am J Neuroradiol 13:1295–1299
Elster AD, Chen MY, Williams DW 3rd, Key LL (1990) Pituitary gland: MR imaging of physiologic hypertrophy in adolescence. Radiology 174(3 Pt 1):681–685
Elster AD, Sanders TG, Vines FS, Chen MY (1991) Size and shape of the pituitary gland during pregnancy and post partum: measurement with MR imaging. Radiology 181:531–535
Kato K, Saeki N, Yamaura A (2002) Morphological changes on MR imaging of the normal pituitary gland related to age and sex: main emphasis on pubescent females. J Clin Neurosci 9:53–56
Lurie SN, Doraiswamy PM, Husain MM et al (1990) In vivo assessment of pituitary gland volume with magnetic resonance imaging: the effect of age. J Clin Endocrinol Metab 71:505–508
Tien RD, Kucharczyk J, Bessette J, Middleton M (1992) MR imaging of the pituitary gland in infants and children: changes in size, shape, and MR signal with growth and development. AJR Am J Roentgenol 158:1151–1154
Argyropoulou M, Perignon F, Brunelle F et al (1991) Height of normal pituitary gland as a function of age evaluated by magnetic resonance imaging in children. Pediatr Radiol 21:247–249
Denk CC, Onderoğlu S, Ilgi S, Gürcan F (1999) Height of normal pituitary gland on MRI: differences between age groups and sexes. Okajimas Folia Anat Jpn 76:81–87
Sari S, Sari E, Akgun V et al (2014) Measures of pituitary gland and stalk: from neonate to adolescence. J Pediatr Endocrinol Metab 7:1071–1076
Suzuki M, Takashima T, Kadoya M et al (1990) Height of normal pituitary gland on MR imaging: age and sex differentiation. J Comput Assist Tomogr 14:36–39
Tsunoda A, Okuda O, Sato K (1997) MR height of the pituitary gland as a function of age and sex: especially physiological hypertrophy in adolescence and in climacterium. AJNR Am J Neuroradiol 18:551–554
Marziali S, Gaudiello F, Bozzao A et al (2004) Evaluation of anterior pituitary gland volume in childhood using three-dimensional MRI. Pediatr Radiol 34:547–551
Takano K, Utsunomiya H, Ono H et al (1999) Normal development of the pituitary gland: assessment with three-dimensional MR volumetry. AJNR Am J Neuroradiol 20:312–315
Di Chiro G, Nelson KB (1962) The volume of the Sella turcica. Am J Roentgenol Radium Ther, Nucl Med 87:989–1008
(2000) Consensus guidelines for the diagnosis and treatment of growth hormone (GH) deficiency in childhood and adolescence: summary statement of the GH Research Society. GH Research Society. J Clin Endocrinol Metab 85:3990–3993
Kemp SF, Alter CA, Dana K et al (2002) Use of magnetic resonance imaging in short stature: data from National Cooperative Growth Study (NCGS) substudy 8. J Pediatr Endocrinol Metab 15(Suppl 2):675–679
Maghnie M, Ghirardello S, Genovese E (2004) Magnetic resonance imaging of the hypothalamus-pituitary unit in children suspected of hypopituitarism: who, how and when to investigate. J Endocrinol Investig 27:496–509
Sharafuddin MJ, Luisiri A, Garibaldi LR et al (1994) MR imaging diagnosis of central precocious puberty: importance of changes in the shape and size of the pituitary gland. AJR Am J Roentgenol 162:1167–1173
Tillmann V, Tang VW, Price DA et al (2000) Magnetic resonance imaging of the hypothalamic-pituitary axis in the diagnosis of growth hormone deficiency. J Pediatr Endocrinol Metab 13:1577–1583
Berntsen EM, Haukedal MD, Håberg AK (2021) Normative data for pituitary size and volume in the general population between 50 and 66 years. Pituitary 24:737–745
Aksan IO (2016) The evaluation of pituitary gland volume using three-dimensional MRI in prepubertal children with normal endocrine function [doctoral thesis]. Trabzon, Turkey: Karadeniz Technical University. Accessed 27 June 2022 via: https://tez.yok.gov.tr/UlusalTezMerkezi/tezDetay.jsp?id=evNxT2C604ePyCo-wSkfrg&no=caupto5EOASRsyCURH7kCg
Acknowledgments
I, as the corresponding author, would like to express my gratitude to my mentor, Dr. Suat Fitoz, who guided me throughout this project. I would also like to thank Dr. Seda Kaynak Şahap and Dr. Birsel Şen Akova for their technical support. I wish to extend my special thanks to my wife, Dr. Merve Karaboğa Çolaklar, for the valuable data and statistics she has provided. Authors have contributed equally to this work.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflicts of interest
None
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Çolaklar, A., Fitoz, Ö.S. Pituitary gland volumes in children with normal endocrine function. Pediatr Radiol 53, 450–460 (2023). https://doi.org/10.1007/s00247-022-05505-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00247-022-05505-5