Abstract
The California Current Ecosystem (CCE) is a productive eastern boundary upwelling system that supports a wide variety of forage stocks. Decadal and interannual variability in the environment influence forage species, which in turn affects predators. The recent diet of California sea lions (Zalophus californianus; CSL) from Central California was determined using identification of hard parts from scat samples (n = 785) collected on Año Nuevo Island (37.1083°N 122.3378°W) in 2010 and 2012–2016. Comparisons were made with previously reported data from the late 1990’s and with prey availability as measured by fishery-independent surveys. A significant shift in diet was seen between the two decades where diet from the 1990’s was dominated by Pacific sardine (Sardinops sagax) and northern anchovy (Engraulis mordax). By 2010, diet was more diverse, characterized by rockfishes, Pacific hake, and market squid. There were also strong interannual differences in diet during the most recent decade, a time of substantial climate variability in the North Pacific Ocean. In general, prey were consumed in relation to what was available in the environment.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The California Current Ecosystem (CCE) is a productive eastern boundary current system characterized by seasonal wind-driven upwelling. As such, this region has a strong seasonality in primary and secondary productions that support a wide variety of forage stocks (Checkley and Barth 2009 and references therein). Sardine, anchovy, rockfishes, and squid are all examples of important forage in this system. Forage stocks, however, are known to vary with climate shifts on both decadal and interannual scales (Hilborn et al. 2017). For instance, Pacific sardine (Sardinops sagax) and Northern anchovy (Engraulis mordax) biomass cycle to low levels on a roughly 60-year periodicity (Baumgartner et al. 1992; Field et al. 2009) possibly related to large-scale sea surface temperature (SST) variability (Lindergren et al. 2013). While exact mechanisms are not understood, sardine and anchovy recruitments are thought to depend on SST, sea surface height (SSH), and upwelling (Cury et al. 1995; Zwolinski et al. 2011; Stachura et al. 2014) and, therefore, may vary on interannual time scales with the seasonal occurrence of El Niño–Southern Oscillation (ENSO) events. The general trend of warming ocean waters and the possibility of more frequent ENSO events may have influenced large-scale changes in the dominant forage species in the CCE with the decline of sardine and anchovy, and the rise of mackerel, hake, and rockfishes in recent years (Zwolinski and Demer 2012; Koslow et al. 2013; Ralston et al. 2015).
Forage species such as sardine, rockfish, and squid are important prey for many seabirds and marine mammals in the CCE (Szoboszlai et al. 2015). Large-scale changes in forage populations can have profound effects on predators that rely on them, whether those predators are specialists with diets dominated by several prey, or generalists with diverse diets. Several species of seabirds have been shown to rely heavily on forage fish and may, therefore, be sensitive to reductions in forage stocks (Koehn et al. 2016). One model predicts depleted sardine populations, from either natural cycles or fishing pressure, will lead to strong declines of dolphins and halibut, and moderate declines in seabirds (Kaplan et al. 2017). Reductions in abundance of seabirds at sea in the CCE (Sydeman et al. 2015) and poor seabird reproductive success (Anderson et al. 1982; Velarde et al. 2004; Tompkins et al. 2017) have been linked to declines of sardine and anchovy populations. These reductions of energy-rich forage have also forced California sea lions in Southern California to shift their diet to less energy-rich prey species which has coincided with years of low pup growth rates and increased mortality (Melin et al. 2010; McClatchie et al. 2016a, b).
California sea lions have been described as “plastic specialists” or “opportunistic feeders” that have a diverse diet of 35–40 + possible species but is dominated by 3–4 key prey, the proportions of which shift depending on availability (Lowry et al. 1991; Weise and Harvey 2008; Orr et al. 2011). CSL at Año Nuevo Island in Central California are on the northern edge of their breeding range. The increased use of this area by CSL as a haul-out, and more recently as a small rookery, is likely the result of the growing population in Southern California expanding to Central California (Lowry et al. 2017; Laake et al. 2018), and greater numbers of animals moving into Central California during warm-water periods (Weise 2000). Año Nuevo Island is located near the productive Monterey Bay and Gulf of the Farallones, which provide a great deal of potential prey availability for wide foraging CSL (Weise et al. 2006). Here, we characterize the diet of CSL in Central California from the early to mid-2010s. As this was a period of great climate variability (García-Reyes and Sydeman 2017), we hypothesized that CSL diet composition would change in strong El Niño years as well as in years when the warm water “Blob” had dramatic effects on the oceanography of the North Pacific (Bond et al. 2015). Due to decadal-scale changes in fish populations, we further hypothesized that CSL diet in Central California would have undergone significant changes since the late 1990’s when the last diet study from this region was published (Weise and Harvey 2008). We also examined whether CSL diet varies from an index of local prey availability that we derived from National Marine Fisheries Service (NMFS) midwater trawl survey data. We hypothesized that diet of CSL would track local forage abundance estimates.
Materials and methods
Study area and sample collection/processing
Año Nuevo Island (37.1083°N 122.3378°W) is located 1 km offshore of the Central California mainland, midway between Monterey Bay and Gulf of the Farallones/San Francisco Bay (Fig. 1). Año Nuevo Island is one of the largest haul-out sites along this stretch of coastline for California sea lions with almost 10,000 individuals in some years, and due to an increasing number of births, has recently been further classified as a rookery (Lowry et al. 2017).
California sea lion scat samples were collected weekly from April to August during 6 years (2010; 2012–2016) when researchers visited Año Nuevo Island for multiple other studies, to minimize disturbance. Scats were chosen from island locations known to be free of other pinniped species to ensure they were deposited by CSL. We attempted to sample from a wide variety of scat sizes and locations to represent various CSL age classes and sexes. However, we have assumed an unequal sex ratio based on recent visual surveys which indicate that even though female use of Año Nuevo has been increasing, the island is still predominantly utilized by sub-adult and adult males.
Frozen scats were thawed and cleaned using the washing machine method described in Orr et al. (2003). Cephalopod beaks were removed, stored in isopropyl alcohol, and identified to lowest taxonomic level using published guides and the help of W. Walker at the National Marine Fisheries Service–National Marine Mammal Laboratory (NMFS–NMML) in Seattle, Washington. After all remaining materials (rocks, bones, vegetation, etc.) were dehydrated, otoliths were removed and identified to lowest taxonomic level using pertinent literature (Harvey et al. 2000; Lowry 2011). Otolith and beak rostral/hood lengths were measured using either digital calipers or software (Image J, Scion Corporation). Due to the large number of beaks in some years, only a representative subset of 10 within each sample was measured. Otoliths from some related species (rockfishes, surfperches, and sculpins) have very similar morphologies that are difficult to distinguish, particularly after erosion and, therefore, these groups were only identified to family level.
Several studies have investigated the relevance of using an “all-structures” method for determining pinniped diet (Tollit et al. 2003; Phillips and Harvey 2009); however, this was beyond the scope of time and resources for this project and as such, only otoliths and beaks were used to determine CSL prey species. Sweeney and Harvey (2011) compared both methods in controlled feeding trials and calculated numerical correction factors (NCFs) to account for reduced prey recovery, which we used here (among other sources, see below). The earlier study, from which we draw data to make decadal comparisons (Weise and Harvey 2008), did use the “all-structures” method for salmon and cartilaginous prey identification and enumeration. We acknowledge that these particular prey types may have been under-represented in the recent diet, although their relative contributions were expected to be small as Weise and Harvey (2008) found very few of each of these prey in the 1998 summer diet and none in 1999 summer diet.
Diet description
Cumulative species curves were run with Matlab (v. R2015a) to determine if enough scat samples had been collected in each year to properly describe diet. Adequate sample size was determined from the points at which curves reach asymptotes, which indicates that new prey items are introduced only rarely (Ferry and Cailliet 1996; Cortéz 1997).
California sea lion diet was described using two relative measures of prey quantity (RMPQs), number and mass. Traditional methods for calculating the number of fish consumed employ a simple technique where otoliths are assigned orientations (left/right), the total number of each side is counted, and the highest value is used as the minimum number of individuals (MNI) for each species. While this method is simple, it has the potential to underestimate how many individuals were consumed. To provide a more realistic estimate of the number of individuals, we developed a “pairing” method in which all lengths were used to match up pairs of left and right otoliths, resulting in a modified minimum number of individuals (MMNI). Mille et al. (2015) found no significant differences in the lengths of left and right otoliths for several fishes, and only one species had a difference of 0.2 mm which was determined to be biologically inconsequential. For this study, as we were dealing with partially eroded otoliths, we considered 0.5 mm to be a more conservative level of difference, so left and right otoliths were paired if their lengths fell within that range of each other. If any otoliths did not have opposite side length matches less than the designated 0.5-mm cut-off they were left unpaired. Thus, our MMNI became the total number of pairs plus any unpaired otoliths within each sample. Because we did not measure every cephalopod beak, we could not use our pairing method for these structures; therefore, cephalopods were enumerated using the greatest number of upper or lower beaks to calculate a traditional MNI.
To account for differential passage times and hard parts which may have been lost during digestion, we applied published numerical correction factors (NCFs; see Online Resource 1) to otolith and beak counts. Where NCFs were not available for identified fish species, we used values from otoliths with similar robustness and, therefore, resilience to erosion, as substitutes, and for cephalopods a value of 1.1, the NCF for market squid (Doryteuthis opalescens) was substituted. Once NCFs were applied, results were used to enhance MNI counts. Within each sample we determined the percent by number (%N) that each species made up of the total MNI of all species and then averaged across all samples to calculate the mean %N for each species in the diet.
Otolith and beak rostral/hood lengths were used with regression equations from publications and personal communications (see Online Resource 1) to determine the biomass of prey species consumed by CSL. Previous studies have demonstrated that otoliths found in scat have various levels of degradation due to the digestive process (Bowen 2000; Orr and Harvey 2001) and, therefore, we applied length correction factors (LCFs; see Online Resource 1) to all otoliths before back-calculating prey masses. If species-specific LCFs were not available, an average length factor of 1.43 was used for otoliths (Weise and Harvey 2008). As cephalopod beaks are known to withstand effects of digestion, no LCFs were used for these structures. The lengths of otoliths with low levels of erosion (Grade 1 and 2; Lance et al. 2001) were used in regression equations; the resulting masses were averaged within samples and multiplied by MMNI to determine the minimum biomass of individuals (MBI) for each species. For samples in which no Grade 1 or 2 otoliths were present, or where Grade 3 otoliths were greater than 1 mm in length from the largest or smallest Grade 1/2 otolith in a sample, thus representing a wider size range of prey, we added Grade 3 otoliths to calculations when determining MBI (n = 236). While we recognize this method likely underestimated mass for those samples, it was determined to be better than having a truncated size range or no estimate at all. Similarly to calculating number, we determined the percent by mass (%M) that each species made up of total MBI within each sample and then averaged across all samples to calculate a mean %M for all species in CSL diet.
As frequency of occurrence (FO) is already inherently contained in %N and %M calculations (Brown et al. 2012), we made no independent estimates of this metric. Thus, we combined only the two diet metrics, number and mass, into one general measure, the geometric index of importance (% GII), to facilitate comparisons. This approach allows the multidimensional distribution of dietary measures to be condensed into a single vector whose magnitude can be directly compared between prey species (Assis 1996), and was calculated as follows:
where the RMPQs for the j’th prey group are added together and divided by the number of RMPQs used. In addition to the years sampled in this study, we calculated %GII for 1998 and 1999 (“summer” season only; Weise and Harvey 2008) for comparison. Our samples were collected at Año Nuevo Island, and those in Weise and Harvey (2008) came from northern and southern Monterey Bay, roughly 20 and 40 km to the south, respectively. Relative to the wide foraging range of CSL (up to 1450 km2; Weise et al. 2006), sampling sites were in close proximity to each other, and were located within the same upwelling plume (Pt. Año Neuvo Plume; Woodson et al. 2009). Sex ratio was also comparable between sites, being predominantly adult and sub-adult males at Año Nuevo Island as mentioned above, and a mean of 74–83% adult males at the Monterey Bay sites (Weise and Harvey 2008).
Prey that comprised 5% or greater of diet by number in any year were retained as individual categories for statistical analysis. All other taxa were combined into one of three additional general categories (flatfishes, other fishes, and other cephalopods).
Forage abundance determination
Midwater trawl surveys have been conducted by the National Marine Fisheries Service–Southwest Fisheries Science Center (NMFS–SWFSC) in Central California every May–June since 1983 (Sakuma et al. 2016). The “Rockfish Recruitment and Ecosystem Assessment Survey” (RREAS) core area is made up of 35 stations that stretch along the coast from Monterey Bay to Bodega Bay (Fig. 1) sampled from 1983 to present, and encompasses the same geographic region in which scat samples were collected both for our recent diet analysis and from Weise and Harvey (2008). In general, marine fauna were collected in May–July at night with ~ 15 min tows of a modified Cobb midwater trawl fished at ~ 2 knots at a station-specific standard depth (~ 10 m or ~ 30 m); more survey details can be found in Ralston et al. (2015). For our years of interest (1998–1999, 2010, 2012–2016), catch-per-unit-effort (CPUE) was calculated for the top species identified in CSL diet (Pacific sardine, rockfish spp., market squid, Pacific hake, red octopus, northern anchovy, jack mackerel, and sanddab spp.), averaged by station and then year. Non-standard tows, tows made to non-standard depths, tows for which an error was noted, and stations that were added or dropped mid-time series were not included.
Statistical analysis
Interannual comparison of CSL diet was conducted using %GII values for years within this study as well as with data previously published from the late 1990’s. Interannual differences were initially compared using Spearman Rank Correlations. Percent GII data for prey groups composing the top 90% of diet were then logit-transformed to adjust for non-normality (Collett 2002; Warton and Hui 2011) and comparisons among years were further examined using PRIMER-E (v6.1.5, 2006). Similarity matrices were constructed for each dataset using the Bray–Curtis similarity coefficient, and one-way analysis of similarity (ANOSIM) tests was performed to determine significance of similarity among years (Daly et al. 2009; Thayer et al. 2014). Similarity percentages (SIMPER) were used to identify which taxonomic categories made the greatest contributions to any dissimilarity. All resulting relationships were visualized using non-metric multidimensional scaling (nMDS) plots; stress values less than 0.10 were unlikely to result in misinterpretation of the data (Clarke and Warwick 2001).
To investigate potential correlations between CSL diet and forage abundance indices, we utilized logit-transformed %N values from diet as these values were more directly relatable to CPUE data from trawls. RREAS trawl CPUE were natural log transformed and compared to CSL diet using SIMPER and ANOSIM tests as above.
Results
Diet description
In total, 785 scat samples were collected and processed during the 6 years of this study, of which 40 (5%) contained no otoliths or beaks and were removed from further analysis. Cumulative species curves revealed that to properly determine CSL diet in this time period roughly 50–80 scat samples containing prey items were needed (see Online Resource 2) and that all years, with the exception of 2012, had more than enough samples (n2010 = 69; n2012 = 26; n2013 = 84; n2014 = 129; n2015 = 261; n2016 = 176). Despite having limited samples from 2012, top prey items from this year were similar to others. We retained these samples for analysis and were cautious with interpretation. In the earlier study, Weise and Harvey (2008) reported needing only 31–48 scat samples to adequately characterize summer diet.
From the 745 scats with hard parts, we identified 41 different prey taxa in CSL diet, including 34 fishes and seven cephalopods (see Online Resource 1). Fishes were comprised of epipelagic (14), mesopelagic (5), and demersal species (14), while cephalopods were almost all pelagic squid with one octopus species. All but two prey types (Spiny dogfish, Squalus acanthias, and Salmon spp., Oncorhynchus spp.) from 1998/1999 diet were also found in this study (2010–2016), which may be attributable to our slightly different methods for these species. There were 19 fishes and five squids, however, in the recent CSL diet samples which were not found in the earlier study. For analysis, all prey taxa were reduced to 11 categories: Pacific sardine (Sardinops sagax), rockfishes (Sebastes spp.), market squid, Pacific hake (Merluccius productus), red octopus (Octopus rubescens), northern anchovy (Engraulis mordax), flatfishes (Pleuronectidae), jack mackerel (Trachurus symmetricus), Pacific saury (Cololabis saira), other fishes, and other cephalopods.
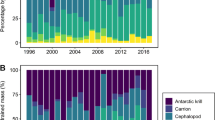
Examination of diet using number, mass, and the combined metric, GII, all revealed similar results where the top 80% of recent diet was composed mostly of rockfish, Pacific hake, market squid, and red octopus (Fig. 2, see Online Resource 3). One exception to this was in 2015, when diet was still diverse but CSL consumed a large amount of jack mackerel (27% by number) which had been virtually absent in all other years. Conversely, diet from the late 1990s was dominated by two prey items, Pacific sardine and northern anchovy, both of were much less prominent in CSL diet in recent years.
California sea lion diet composition expressed as percent geometric index of importance (%GII). Data from 1998/1999 were calculated from a previously published study (Weise and Harvey 2008). Recent diet (2010, 2012–2016) was determined from samples collected during this study
Interannual and interdecadal variability
Spearman rank tests between years revealed significant negative correlations between Pacific sardine and (a) rockfish, (b) Pacific hake, and (c) other cephalopods, as well as between northern anchovy and (a) market squid and (b) red octopus (Table 1). These negative correlations indicate that the visually evident shift in CSL diet between the late 1990s and recent years was indeed significant. Additionally, in years when CSL consumed more market squid they also consumed higher levels of red octopus and Pacific saury, and in years when CSL ate more rockfishes there were higher levels of other cephalopods and flatfishes (Table 1).
Overall CSL diet between the 1990s and 2010s was significantly different (R = 0.97, p = 0.04). SIMPER tests revealed 22% dissimilarity between decades mostly due to fewer Pacific sardine and more cephalopods in the 2010s. There was 90% similarity among the recent years, which was attributed to the prevalence of Pacific hake, market squid, and rockfishes in all 6 years. The influx of jack mackerel to CSL diet in 2015 and 2016 set these 2 years apart from 2010 to 2014. With only 2 years of data from the 1990s we were not able to run a similarity test on CSL diet within this decade, but visually could see high levels of sardine and anchovy in the diet (Fig. 2). The non-metric MDS plot also clearly showed the significant dietary differences among and within decades (Fig. 3).
Non-metric multidimensional scaling (MDS) visualization of California sea lion diet annual comparisons based on Bray–Curtis similarity index using percent geometric index of importance (%GII) values for the top eight prey taxa (“other fishes” and “other cephalopods” groups were removed). Maximum stress value was 0.01, values < 0.10 were regarded as being unlikely to result in misinterpretation of the data (Clarke and Warwick 2001). Open triangles represent diet from the late 1990’s obtained from Weise and Harvey (2008). Shaded triangles represent diet from this study of the 2010’s
Forage abundance indices
Trawl survey data from the RREAS core area revealed distinct changes in prey availability between the decades and high interannual variability among recent years (Fig. 4). In 1998, catch was overwhelmingly dominated by northern anchovy and Pacific sardine, the latter of which declined the following year and was not observed again in any great abundance. Northern anchovy, however, did increase in 2015/2016 to levels observed in the late 90’s (Fig. 4). Increases of rockfish, market squid, Pacific hake, and sanddab catches were observed in the 2010s.
Diet versus prey availability
There was a significant relationship between overall CSL diet and the RREAS indices of prey availability (R = 0.50, p = 0.02). A SIMPER test revealed 33% dissimilarity between diet and availability, with Pacific sardine, red octopus, and jack mackerel occurring more in diet than in trawls. Examining individual relationships, northern anchovy and market squid were consumed by CSL in close proportion to availability indices (Fig. 5). Rockfishes and red octopus in CSL diet was higher than availability indexed by trawls. There was no clear relationship between diet and availability for Pacific hake or sanddabs. Jack mackerel in the diet and trawls appeared to have been strongly related; however, in 5 of the 8 years there were no mackerel found in either source. For the years in which jack mackerel was present, in 1 year (2016) mackerel were higher in CSL diet vs. trawls, while in another year (2015), mackerel were lower in CSL diet vs. trawls.
Discussion
We observed a major shift in the diet of CSL in Central California and diet diversity increased significantly. Weise and Harvey (2008) identified 32 prey taxa in diet from the late 1990s, whereas data from the 2010s revealed 41 prey species, many of which were not reported in the earlier study. This increased diversity was also indicated by our cumulative species curves which showed that 60% more samples were needed to characterize diet composition in the 2010s than was needed in the 1990’s. The diet in 1998–1999 was not only less diverse, but was dominated by one species, Pacific sardine, and to a lesser extent northern anchovy. Since that time, there was a substantial decline in sardine and anchovy, with persistently low biomass of both species in recent years (Hill et al. 2017; Thayer et al. 2017). In the absence of these previously important forage stocks, CSL have apparently switched to other prey, primarily rockfishes, Pacific hake, and market squid. An alternate explanation for the increased diversity could be changing sex ratio, i.e., an increase in females sampled, given that adult females have been shown to consume a greater diversity of prey than juveniles (Orr et al. 2011). Comparisons of adult female and juvenile diets, however, have not been quantified in relation to that of adult and sub-adult males.
Past studies have identified CSL as a generalist predator of a wide range of fishes and cephalopod prey (21–58 taxa identified; Ainley et al. 1982; Lowry et al. 1990; Melin et al. 2010, 2012). While our results do not disagree with previous findings that CSL consume many different prey, it is important to recognize that CSL diet has been shown to be dominated by three to five species which have been utilized in changing proportions through time (Lowry et al. 1991; Weise and Harvey 2008; Orr et al. 2011).
CSL diet may vary in terms of location, seasonality, and/or interannual or decadal changes in ocean conditions that influence prey availability. In the earliest records for annual diet in Central California from the 1970s, CSL preyed almost exclusively on Pacific hake and rockfishes (Ainley et al. 1982; Bailey and Ainley 1982). These studies were conducted using only regurgitated prey remains (Ainley et al. 1982) or only reported data on Pacific hake (Bailey and Ainley 1982), and may represent a biased view of diet. Furthermore, these studies were based on diet from the Farallon Islands, located on the edge of the continental shelf with different prey fields than inshore (Santora et al. 2012). Central California CSL diet in the early 2000s was dominated in various years by market squid, rockfish, northern anchovy, or Pacific sardine (Peterson et al. 2006; Weise et al. 2006), although these annual data were only presented for those four species and only in units of percent mass. Studies from Southern California found that annual CSL diet from San Clemente and San Nicholas Islands in the 1980s was devoid of sardine (after collapse of that species; MacCall 1979), but rich in anchovy, hake, and jack mackerel (Lowry et al. 1990, 1991). Melin et al. (2010, 2012) observed that during summer months in the 2000s CSL from San Miguel Island were frequently consuming Pacific hake, sardine, and anchovy, but that by the early 2010s had switched to rockfishes and market squid. Most of these studies, however, focused on frequency of occurrence (FO) diet estimations, which only indicate the presence/absence of a prey type, not how much was consumed. Information on relative consumption is needed to relate diet composition to prey availability in the environment.
Prey switching and environmental conditions
Within the timeframe of this study, there have been extreme changes in ocean conditions. During the most recent decade, annual averages of the Central California Multivariate Ocean Climate Indicator (MOCI), an index which synthesizes a number of local and regional oceanic and atmospheric conditions, calculated as summer year x − 1 to spring year x and labeled as year x (e.g., 2010 is summer 2009 to spring 2010) to include El Niño events within the same annual value, showed 2 years of strong upwelling and cold conditions (2012 and 2013), 2 years of moderate, non-anomalous conditions (2010 and 2014), and 2 years of extreme warm conditions (2015 and 2016; Fig. 6 taken with permission from http://www.faralloninstitute.org/moci; García-Reyes and Sydeman 2017). During moderate and cold years, market squid consistently made up roughly 25% of CSL diet in Central California, and during 2013 which contained the coldest point in the MOCI time series, over half of diet was composed of cephalopods. Similarly, Lowry and Carretta (1999) found fewer market squid in CSL diet in Southern California during warm El Niño years in the 1980s and 1990s. Red octopus was another prominent cephalopod in CSL diet, contributing 12% and 22% to the diet in the recent cold years of 2012 and 2013, respectively. Weise and Harvey (2008) also reported an uptick in the number of red octopus in CSL diet during the 1998 El Niño. The increased use of cephalopods is likely indicative of CSL consuming whichever prey is most available when high-energy prey is reduced due to environmental conditions. Although consumption of red octopus did not mirror the prey availability indexed by the RREAS surveys, midwater trawls may not provide the best estimate of abundance for primarily benthic species.
Seasonal Multivariate Ocean Climate Indicator (MOCI; García-Reyes and Sydeman 2017) for Central California taken from http://www.faralloninstitute.org/moci. MOCI synthesizes a number of local and regional oceanic and atmospheric conditions that represent the state of the Central California coastal ocean in a holistic and seasonal manner. Red dots represent MOCI values one standard deviation above the season’s mean (warm conditions and weak upwelling), while blue dots represent values one standard deviation below the seasonal mean (cold conditions and strong upwelling)
During recent warm years, which resulted from the combined effects of El Niño and the Blob (Bond et al. 2015; Gentemann et al. 2017), CSL preyed heavily on jack mackerel (2015) and rockfishes (2015 and 2016), the latter of which have recently had several years of high recruitment (Wells et al. 2017). Jack mackerel, an energy-dense prey source (Sidwell et al. 1974), can be found throughout the CCE, yet they are most abundant offshore and in the Southern California Bight (MacCall and Stauffer 1983). During the strong 1983–1984 El Niño, the jack mackerel fishery catch increased in northern regions (Mason 1989). Likewise, in 2015 and 2016, CPUE of jack mackerel in the RREAS trawls increased, indicating northward expansion and inshore movement, likely tracking warm waters. Even so, mackerel made up a much higher proportion of CSL diet than abundance indexed by trawls in 2015. Trawls may have missed patches of this irregularly distributed pelagic species, or CSL may have obtained mackerel further offshore than trawls were conducted. In 2016, jack mackerel was available as indicated by trawls, yet CSL did not eat them as much as in 2015, possibly because of the higher availability of other prey on or near the shelf, such as juvenile rockfish and hake.
Although the Central California region of the Pacific Ocean experienced warm conditions in 2015–2016 and in 1997–1998 (Fig. 6), small pelagic species such as anchovy and sardine were much less abundant during the recent event, part of declines which began in the mid-2000s (MacCall et al. 2016; Hill et al. 2017). Numbers of Pacific hake caught in 2015–2016 surveys were the largest during our study period. Pacific hake, however, as well as sanddabs, exhibited no clear relationship between proportion in the diet and availability in the environment (Fig. 5), indicating that they were opportunistically consumed rather than selected by CSL. Pacific sardine, however, with its low availability during the majority of years in our study, appeared to be selected for.
Ecological significance of prey switching
The prey switching observed in this study indicates that CSL may now be consuming less energy-dense food, due to distribution and abundance of prey species (Österblom et al. 2008). Sardine and anchovy are high in protein and fat contents (Sidwell et al. 1974; Costa et al. 1991) which before their decline provided CSL with readily available and energy-rich food sources. The sardine and anchovy collapse was followed by a rise of rockfishes, hake, and market squid in the diet, all of which are less energy rich (Costa et al. 1991). The consumption of these species is thought to have had deleterious effects on CSL throughout their range. In Central California, Weise et al. (2006) showed that with warmer waters and shifts in prey populations there were significant changes in mean foraging distance and number of days spent at sea, indicating that male CSL were forced to forage longer and travel further offshore to find sufficient prey. Several studies in Southern California have shown that changes in prey abundance and quality also require breeding female CSL to increase forage trip durations which is linked to increased pup mortality (Feldkamp et al. 1989; Melin et al. 2008, 2010, 2012; McClatchie et al. 2016a, b; McHuron et al. 2017). Further complicating this situation, Zwolinski and Demer (2012) have suggested that the possibility for a near-term sardine recovery is very limited, indicating access to energy-rich prey for CSL may remain restricted in the near future. However, in general, the CSL population has continued to grow over time (Lowry et al. 2017), which could indicate that prey-switching abilities could help buffer CSL from the effects of environmental variability.
References
Ainley DG, Huber HR, Bailey KM (1982) Population fluctuations of California sea lions and the Pacific whiting fishery off central California. Fish Bull 80(2):253–258
Anderson DW, Gress F, Mais KF (1982) Brown pelicans: influence of food supply on reproduction. Oikos 39(1):23–31
Assis CA (1996) A generalized index for stomach contents analysis in fish. Sci Mar 60(2–3):385–389
Bailey KM, Ainley DG (1982) The dynamics of California sea lion predations on Pacific hake. Fish Res 1:163–176
Baumgartner TR, Soutar A, Ferreira-Bartrina V (1992) Reconstruction of the history of Pacific sardine and northern anchovy populations over the past two millennia from sediments on the Santa Barbara Basin, California. CalCOFI Rep 33:24–40
Bond NA, Cronin MF, Freeland H, Mantua N (2015) Causes and impacts of the 2014 warm anomaly in the NE Pacific. Geophys Res Lett 42:3414–3420
Bowen WD (2000) Reconstruction of pinniped diets: accounting for complete digestion of otoliths and cephalopod beaks. Can J Fish Aquat Sci 57:898–905
Brown SC, Bizzarro JJ, Cailliet GM, Ebert DA (2012) Breaking with tradition: redefining measures for diet description with a case study of the Aleutian skate Bathyraja aleutica (Gilbert 1896). Environ Biol Fish 95:3–20
Checkley DM Jr, Barth JA (2009) Patterns and processes in the California Current System. Prog Oceanogr 83:49–64
Clarke KR, Warwick RM (2001) Change in marine communities: an approach to statistical analysis and interpretation, 2nd edn. PRIMER-E, Plymouth
Collett D (2002) Modelling binary data, 2nd edn. Chapman and Hall/CRC, Boca Raton
Cortez E (1997) A critical review of methods of studying fish feeding based on analysis of stomach contents: application to elasmobranch fishes. Can J Fish Aquat Sci 54:726–738
Costa DA, Antonelis DA, DeLong RL (1991) Effects of El Niño on the foraging energetics of the California sea lion. In: Trillmich F, Ono KA (eds) Pinnipeds and El Niño. Responses to Environmental Stress. Springer, Berlin, pp 156–165
Cury P, Roy C, Mendelssohn R, Bakun A, Husby DM, Parrish RH (1995) Moderate is better: exploring nonlinear climatic effects on the Californian northern anchovy (Engraulis mordax). In: Beamish RJ (eds) Climate change and northern fish populations, Can Spec Publ Fish Aquat Sci 121:417–424
Daly EA, Brodeur RD, Weitkamp LA (2009) Ontogenetic shifts in diets of juvenile and subadult coho and Chinook salmon in coastal marine waters: important for marine survival? Trans Am Fish Soc 138:1420–1438
Feldkamp SD, Delong RL, Antonelis GA (1989) Diving patterns of California sea lions, Zalophus californianus. Can J Zool 67:872–883
Ferry LA, Cailliet GM (1996) Sample size and data analysis: are we characterizing and comparing diet properly? In: MacKinlay D, Shearer K (eds) Gutshop’96. Feeding ecology and nutrition on fish symposium proceedings, San Francisco State University, pp 71–80
Field JC, Baumgartner TR, Ferreira V, Gutierrez D, Lozano-Montes H, Salvatteci R, Soutar A (2009) Variability from scales in marine sediments and other historical records. In: Checkley DM Jr, Alheit J, Oozeki Y, Roy C (eds) Climate change and small pelagic fish. Cambridge University Press, Cambridge, pp 45–63
García-Reyes M, Sydeman WJ (2017) California multivariate ocean climate indicator (MOCI) and marine ecosystem dynamics. Ecol Ind 72:521–529
Gentemann CL, Fewings MR, García-Reyes M (2017) Satellite sea surface temperatures along the West Coast of the United States during the 2014–2016 northeast Pacific marine heat wave. Geophys Res Lett 44:312–319
Harvey JT, Loughlin TR, Perez MA, Oxman DS (2000) Relationship between fish size and otolith length for 63 species of fishes from the eastern North Pacific Ocean. NOAA Tech Rep NMFS 150:38
Hilborn R, Amoroso RO, Bogazzi E, Jensen OP, Parma AM, Szuwalski C, Walters CJ (2017) When does fishing forage species affect their predators? Fish Res 191:211–221
Hill KT, Crone PR, Zwolinski JP (2017) Assessment of the Pacific sardine resource in 2017 for U.S. management in 2017–18. Pacific Fishery Management Council, April 2017, Briefing Book, Agenda Item G.5.a, Portland, Oregon, pp 146
Kaplan IC, Koehn LE, Hodgson EE, Marshall KN, Essington TE (2017) Modeling food web effects of low sardine and anchovy abundance in the California Current. Ecol Model 359:1–24
Koehn LE, Essington TE, Marchall KN, Kaplan IC, Sydeman WJ, Szoboszlai AI, Thayer JA (2016) Developing a high taxonomic resolution food web model to assess the functional role of forage fish in the California Current ecosystem. Ecol Model 335:87–100
Koslow JA, Goericke R, Watson W (2013) Fish assemblages in the southern California Current: relationships with climate, 1951–2008. Fish Oceanogr 22(3):207–219
Laake JL, Lowry MS, DeLong RL, Melin SR, Carretta JV (2018) Population growth and status of California sea lions. J Wild Manag 82(3):583–595
Lance MM, Orr AJ, Riemer SD, Weise MJ, Laake JL (2001) Pinniped food habits and prey identification techniques protocol. Alaska Fisheries Science Center/National Marine Fisheries Service Rep, pp 35
Lindergren M, Checkley DM Jr, Rouyer T, MacCall AD, Stenseth N (2013) Climate, fishing, and fluctuations of sardine and anchovy in the California Current. PNAS 110(33):13672–13677
Lowry MS (2011) Photographic catalog of California marine fish otoliths: Prey of California sea lions (Zalophus californianus). NOAA-TM-NMFS-SWFSC-483, pp 256
Lowry MS, Carretta JV (1999) Market squid (Loligo opalescens) in the diet of California sea lion (Zalophus californianus) in southern California (1981–1995). CalCOFI Rep 40:196–207
Lowry MS, Oliver CW, Macky C, Wexler JB (1990) Food habits of California sea lions Zalophus californianus at San Clemente Island, California, 1981–1986. Fish Bull 88(3):509–521
Lowry MS, Stewart BS, Heath CB, Yochem PK, Francis JM (1991) Seasonal and annual variability in the diet of California sea lions Zalophus californianus at San Nicolas Island, California, 1981–1986. Fish Bull 89:331–336
Lowry MS, Melin SR, Laake JL (2017) Breeding season distribution and population growth of California sea lions, Zalophus californianus, in the United States during 1964–2014. NOAA-TM-NMFS-SWFSC-574, pp 66
MacCall AD (1979) Population estimates for the waning years of the Pacific sardine fishery. CalCOFI Rep 20:72–82
MacCall AD, Stauffer GD (1983) Biology and fishery potential of jack mackerel (Trachurus symmetricus). CalCOFI Rep 24:46–56
MacCall AD, Sydeman WJ, Davison PC, Thayer JA (2016) Recent collapse of northern anchovy biomass off California. Fish Res 175:87–94
Mason JE (1989) The southern California jack mackerel fishery, and the age and length composition of the catch for the 1972–73 through 1983–84 seasons. Calif Dept Fish Game Mar Res Tech Rep 58:38
McClatchie S, Field J, Thompson AR, Gerrodette T, Lowry M, Fiedler PC, Watson W, Nieto KM, Vetter RD (2016a) Food limitation of sea lion pups and the decline of forage off central and southern California. R Soc Open Sci 3:150628
McClatchie S, Goericke R, Leising A, Auth TR, Bjorkstedt E, Robertson RR, Brodeur RD, Du X, Daly EA, Morgan CA, Chavez FP, Debich AJ, Hildebrand J, Field J, Sakuma K, Jacox MG, Kahru M, Kudela R, Anderson C, Largier J, Lavaniegos BE, Gomez-Valdes J, Jimenez-Rosenberg SPA, McCabe R, Melin SR, Ohman MD, Sala LM, Peterson B, Fisher J, Schroeder ID, Bograd SJ, Hazen EL, Schneider SR, Golightly RT, Suryan RM, Gladics AJ, Loredo S, Porquez LM, Thompson AR, Weber ED, Watson W, Trainer W, Warzybok P, Bradley R, Jahncke J (2016b) State of the California Current 2015–16: comparisons with the 1997–98 El Niño. CalCOFI Rep 57:5–61
McHuron EA, Mangel M, Schwarz LK, Costa DP (2017) Energy and prey requirements of California sea lions under variable environmental conditions. Mar Ecol Prog Ser 567:235–247
Melin SR, DeLong RL, Siniff DB (2008) The effects of El Niño on the foraging behavior of lactating California sea lions (Zalophus californianus californianus) during the nonbreeding season. Can J Zool 86:192–206
Melin SR, Orr AJ, Harris JD, Laake JL, DeLong RL, Gulland FMD, Stoudt S (2010) Unprecedented mortality of California sea lion pups associated with anomalous oceanographic conditions along the central California coast in 2009. CalCOFI Rep 51:182–194
Melin SR, Orr AJ, Harris JD, Laake JL, DeLong RL (2012) California sea lions: an indicator for integrated ecosystem assessment of the California Current System. CalCOFI Rep 53:140–152
Mille T, Mahe K, Villanueva MC, De Pontual H, Ernande B (2015) Sagittal otolith morphogenesis asymmetry in marine fishes. J Fish Biol 87:646–663
Orr AJ, Harvey JT (2001) Quantifying errors associated with using fecal samples to determine the diet of the California sea lion (Zalophus californianus). Can J Zool 79:1080–1087
Orr AJ, Laake JL, Dhruv MI, Banks AS, DeLong RL, Huber HR (2003) Comparison of processing pinniped scat samples using a washing machine and nested sieves. Wild Soc Bull 31(1):253–257
Orr AJ, VanBlaricom GR, DeLong RL, Cruz-Escalona VH, Newsome SD (2011) Intraspecific comparison of the diet of California sea lions (Zalophus californianus) assessed using fecal and stable isotope analysis. Can J Zool 89:109–122
Österblom H, Olsson O, Blenckner T, Furness RW (2008) Junk-food in marine ecosystems. Oikos 117:967–977
Peterson WT, Emmett R, Goericke R, Venrick E, Mantyla A, Bograd SJ, Schwing FB, Hewitt R, Lo N, Watson W, Barlow J, Lowry M, Ralston S, Forney KA, Lavaniegos BE, Sydeman WJ, Hyrenbach D, Bradley RW, Warzybok P, Chavez F, Hunter K, Benson S, Weise M, Harvey J, Gaxicola-Castro G, Durazo R (2006) The State of the California Current, 2005-2006: warm in the North, Cool in the South. CalCOFI Rep 47:30–74
Phillips EM, Harvey JT (2009) A captive feeding study with the Pacific harbor seal (Phoca vitulina richardii): implications for scat analysis. Mar Mamm Sci 25(2):373–391
Ralston S, Field JC, Sakuma KM (2015) Ling-term variation in a central California pelagic forage assemblage. J Mar Syst 146:26–37
Sakuma KM, Field JC, Mantua NJ, Ralston S, Marinovic BB, Carrion CN (2016) Anomalous epipelagic micronekton assemblage patterns in the neritic waters of the California Current in spring 2015 during a period of extreme ocean conditions. CalCOFI Rep 57:163–183
Santora JA, Field JC, Schroeder ID, Sakuma KM, Wells BK, Sydeman WJ (2012) Spatial ecology of krill, micronekton and toppredators in the central California Current: implications for defining ecologically important areas. Prog Oceanogr 106:154–174
Sidwell VD, Foncannon PR, Moore NS, Bonnet JC (1974) Composition of the edible portion of raw (fresh or frozen) crustaceans, finish, and mollusks. I. Protein, fat, moisture, ash, carbohydrate, energy value, and cholesterol. Mar Fish Rev 36(3):21–35
Stachura MM, Essington TE, Mantua NA, Hollowed AB, Haltuch MA, Spencer PD, Branch TA, Doyle MJ (2014) Linking Northeast Pacific recruitment synchrony to environmental variability. Fish Oceanogr 23(5):389–408
Sweeny JM, Harvey JT (2011) Diet estimation in California sea lions, Zalophus californianus. Mar Mamm Sci 27(4):E279–E301
Sydeman WJ, Thompson SA, Santora JA, Koslow JA, Goericke R, Ohman MD (2015) Climate-ecosystem change off southern California: time-dependent seabird predator-prey numerical responses. Deep Sea Res II 112:158–170
Szoboszlai AI, Thayer JA, Wood SA, Sydeman WJ, Koehn LE (2015) Forage species in predator diets: synthesis of data from California Current. Ecol Inform 29:45–56
Thayer JA, Field JC, Sydeman WJ (2014) Changes in California Chinook salmon diet over the past 50 years in relation to marine climate: relevance to the recent population crash. Mar Ecol Prog Ser 498:249–261
Thayer JA, MacCall AD, Sydeman WJ, Davidson PC (2017) California anchovy population remains low, 2012–2016. CalCOFI Rep 58:69–76
Tollit DJ, Wong M, Winship AJ, Rosen DAS, Trites AW (2003) Quantifying errors associated with using prey skeletal structures from fecal samples to determine the diet of Stellar’s sea lion (Eumetopias jubatus). Mar Mamm Sci 19(4):724–744
Tompkins EM, Townsend HM, Anderson DJ (2017) Decadal-scale variation in diet forecasts persistently poor breeding under ocean warming in a tropical seabird. PLoS One 12(8):e0182545
Velarde E, Ezcurra E, Cisneros-Mata MA, Lavin MF (2004) Seabird ecology, El Niño anomalies, and prediction of sardine fisheries in the Gulf of California. Ecol Appl 14(2):607–615
Warton DI, Hui FKC (2011) The arcsine and asinine: the analysis of proportions in ecology. Ecol 92(1):3–10
Weise MJ (2000) Abundance, food habits, and annual fish consumption of California sea lion (Zalophus californianus) and its impact on salmonid fisheries in Monterey Bay, California. Master thesis, Moss Landing Marine Laboratories/San Jose State University, Moss Landing
Weise MJ, Harvey JT (2008) Temporal variability in ocean climate and California sea lion diet and biomass consumption: implications for fisheries management. Mar Ecol Prog Ser 373:157–172
Weise MJ, Costa DP, Kudela RM (2006) Movement and diving behavior of male California sea lions (Zalophus californianus) during anomalous oceanographic conditions of 2005 compared to those of 2004. Geophys Res Lett 33:L22S10
Wells BK, Schroeder ID, Bograd SJ, Hazen EL, Jacox MG, Leising A, Mantua N, Santora JA, Fisher J, Peterson WT, Bjorkstedt E, Robertson RR, Chavez FP, Goericke R, Kudela R, Anderson C, Lavaniegos BE, Gomez-Valdes J, Brodeur RD, Daly EA, Morgan CA, Auth TD, Field JC, Sakuma K, McClatchie S, Thompson AR, Weber ED, Watson W, Suryan RM, Parrish J, Dolliver J, Loredo S, Porquez SM, Zamon JE, Schneider SR, Golightly RT, Warzybok P, Bradley R, Jahncke J, Sydeman WJ, Melin SR, Hildebrand JA, Debich AJ, Thayre B (2017) State of the California Current 2016–17: still anything but “normal” in the north. CalCOFI Rep 58:1–55
Woodson CB, Washburn L, Barth JA, Hoover DJ, Kirincich AR, McManus MA, Ryan JP, Tyburczy J (2009) Northern Monterey Bay upwelling shadow front: observations of a coastally and surface-trapped buoyant plume. J Geophys Res 114:C12013
Zwolinski JP, Demer DA (2012) A cold oceanographic regime with high exploitation rates in the Northeast Pacific forecasts a collapse of the sardine stock. PNAS 109(11):4175–4180
Zwolinski JP, Emmett RL, Demer DA (2011) Predicting habitat to optimize sampling of Pacific sardine (Sardinops sagax). ICES J Mar Sci 68(5):867–879
Acknowledgements
This project was supported with funds from United States Office of Naval Research, Research Opportunity for Program Officers Grant # 12122730. All biological samples were collected in accordance with National Marine Fisheries Service Permits #871851 and #17952. We would like to thank P. Morris at University of California, Santa Cruz, for all her hard work collecting scat samples from Año Nuevo Island, J. Hassrick for his initial project coordination, and M. Hester, R. Carle, and J. Beck at Oikonos for boat support and initial sample collection/processing. Our gratitude goes out to K. Hernandez and interns at Moss Landing Marine Laboratories/San Jose State University and Long Marine Lab/UCSC for their endless hours washing scat samples. We thank D. Crocker at Sonoma State University, B. McDonald at MLML/SJSU, and D. Costa and P. Robinson at LMR/UCSC for lab space needed to process our samples. Finally, we thank all reviewers for their thoughtful comments.
Funding
Funding for this study was provided by the United States Office of Naval Research, Research Opportunity for Program Officers Grant # 12122730.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
All applicable international, national, and/or institutional guidelines for the care and use of animals were followed.
Additional information
Responsible Editor: Y. Cherel.
Reviewed by E. McHuron and an undisclosed expert.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Robinson, H., Thayer, J., Sydeman, W.J. et al. Changes in California sea lion diet during a period of substantial climate variability. Mar Biol 165, 169 (2018). https://doi.org/10.1007/s00227-018-3424-x
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00227-018-3424-x