Abstract
Temperature and salinity are important environmental factors affecting the normal functioning of marine animals, particularly animals such as sea urchins living in shallow waters and tide pools. Here, we evaluated the effect of different combinations of temperature and salinity on early embryos of the endemic New Zealand sea urchin Evechinus chloroticus. Animals were collected at Matheson’s Bay (36º18′17′′S; 174º47′51′′E) in north-eastern New Zealand in February 2013. Embryos were exposed to five salinities (29, 31, 34, 35 and 37 ppt) and two temperatures (18 and 21 °C) during the first 24 h of development. Low salinity (29 ppt) affected all parameters (fertilization, development rate, gastrulation and normal development), with ca. 50 % of embryos surviving at 29 ppt, whereas seawater temperature only affected development rate and gastrulation. An increase in temperature from 18 to 21 °C minimized the negative effect of low salinity (≤31 ppt) on development rate and gastrulation of E. chloroticus. Overall, the results of this study suggest that early embryos of E. chloroticus have developmental plasticity to withstand reductions in salinity up to 29 ppt; however, it is still unknown whether the surviving embryos will be able to complete larval development at low salinities, particularly whether the embryos and larvae are carried into extreme environments such as estuaries where salinity is even lower. Multistressor studies are very important for climate change research as multiple environmental factors will act together in the wild, having major consequences for development and recruitment of marine invertebrates.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Environmental conditions play an important role in the normal functioning of all animals (Willmer 1999), with temperature and salinity of greatest importance for marine animals living in intertidal and shallow subtidal environments (Castro and Huber 2005). Extreme differences in seawater temperature can occur daily or seasonally in relation to the intensity of solar radiation that reaches the surface of the oceans (Lalli and Parsons 1997). In addition, changes in salinity occur naturally, either in shallow waters influenced by river discharge, in tide pools after heavy rain events (Lawrence 1975; Russell 2013) or by evaporation when ambient air temperatures are high (Trowbridge 1994).
In addition, at a regional scale, seawater temperature and salinity are also projected to vary globally due to anthropogenic climate change (IPCC 2014). In general, a rise in seawater temperature is projected due to a global increase in atmospheric heat, while salinity of the water is projected to vary regionally due to changes in rainfall patterns, ice melt and evaporation (IPCC 2014). In the specific case of New Zealand, an increase in seawater temperature of 0.7–2.3 °C is projected by 2090, whereas salinity levels are most likely to be affected by changes in precipitation patterns (IPCC 2014), and decreased rainfall/increased salinity for the East coast of the North Island and the opposite pattern on the West coast (Nottage et al. 2010). In addition, the number of extreme rainfall events is projected to increase about 32 % around New Zealand by the next century (Renwick et al. 2013).
Echinoderms are generally affected by changes in temperature and salinity (Moore 1966; Russell 2013), with important effects on fertilization success and cleavage (Greenwood and Bennett 1981; Cameron et al. 1985; Sewell and Young 1999; Kashenko 2006; Byrne et al. 2009; Allen and Pechenik 2010; Delorme and Sewell 2013), survival (Roller and Stickle 1993; Metaxas 1998; Sewell and Young 1999; Li et al. 2011), larval morphology and metamorphosis (George and Walker 2007; Diaz-Perez and Carpizo-Ituarte 2011; Pia et al. 2012; Armstrong et al. 2013; Byrne et al. 2013). In general, temperature and salinity have opposing effects during early development of echinoderms. For example, an increase in temperature (within optimal ranges) results in an increase in development rate (Fujisawa 1995; Sewell and Young 1999; Azad et al. 2010; Li et al. 2011; Delorme and Sewell 2013; Hardy et al. 2014), whereas a decrease in salinity results in a delay of development (Roller and Stickle 1985, 1993; Metaxas 1998; Cowart et al. 2009; Allen and Pechenik 2010; Li et al. 2011; Armstrong et al. 2013). Although there is little research on the combined effect of temperature and salinity on the development of echinoderms (Azad et al. 2010), previous studies suggest that at optimal temperatures, lower salinities delay development. However, when the temperature increase is within the optimal range, the effect of low salinities is diminished (Roller and Stickle 1993; Bressan et al. 1995; Kashenko 2006; Armstrong et al. 2013).
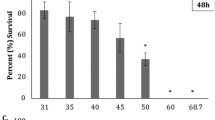
The echinometrid sea urchin Evechinus chloroticus (Valenciennes 1846) is an endemic New Zealand species, distributed from the Three Kings Island in the north (34º10′S) to the Snares Islands in the subantarctic (47º60′S) (Fenwick and Horning 1980; Schiel et al. 1986). Individuals are commonly found in shallow waters (<14 m) in a variety of habitats, including tide pools and fiords (Dix 1970; Barker 2007). As a result of the broad distribution of E. chloroticus, individuals around New Zealand can be exposed to a variation of seawater temperatures (9.5–21 °C) and salinities (34–35 ppt) depending on the latitude and time of the year (Garner 1969). In waters of the Hauraki Gulf, northern New Zealand, seawater temperature varies seasonally between 14 and 21 °C (Leigh Marine Laboratory temperature records 1967–2011), whereas salinity can vary between 34.3 and 35.5 ppt during early spring and late summer (Zeldis 2004; Zeldis et al. 2004). However, lower salinities (>25 psu, Walker and Vaughan 2013) can be encountered in shallow waters during winter and spring due to increased river flow, while higher salinities are present in summer due to reduced freshwater input (Zeldis and Smith 1999). In tide pools of the Hauraki Gulf, temperature and salinity can undergo more extreme seasonal changes, ranging between 10 and 37 °C, and 31 and 60 ppt (Trowbridge 1994). Additionally, in the fiords of the South Island, surface salinity undergoes more extreme changes due to the combination of natural variation of the input of freshwater through precipitation and/or input from hydroelectric power stations. In Doubtful Sound, for example, this results in surface seawater (to 5 m depth) having salinities between 5 and 30 ppt, overlaying more oceanic salinities of 34–35 ppt at depths >10 m (Peake et al. 2001).
Evechinus chloroticus has an annual breeding cycle, spawning during the austral summer when temperatures are highest, 13 °C for the South and 21 °C for the North Island (Garner 1969; Barker 2007). During the reproductive season, embryos may also experience acute exposure to either lower or higher salinities due to sporadic storm events resulting in a high input of freshwater, or by evaporation in shallow coastal waters. Previous research has shown that the early developmental stages of a north-eastern population of E. chloroticus are very sensitive to a temperature increase of 3 °C above ambient (Delorme and Sewell 2013); however, no studies have been published on how the interaction of temperature with other variables such as salinity will affect early life stages. Therefore, the aim of the present study was to evaluate the capacity of the early embryonic stages to develop under a combination of five salinity levels and two temperatures within the optimal range for development of the study population of this species in northern New Zealand (Delorme and Sewell 2013). Overall, this study found that low salinities (<31 ppt) had a negative effect on fertilization and early development of E. chloroticus, and that an increase in temperature from 18 to 21 °C helps to minimize the effects of low salinity.
Materials and methods
Animal collection and laboratory maintenance
Mature adult animals were collected from shallow depths (2 m) at Matheson’s Bay (36º18′17′′S; 174º47′51′′E) in north-eastern New Zealand in February 2013. Seawater temperature and salinity at the time of collection were 21 °C and 34 ppt, measured with a submersible mercury thermometer and a refractometer, respectively. Average seawater temperature during the previous weeks of collection was 20.7 ± 0.5 °C (Leigh Marine Laboratory temperature dataset). After collection, the animals were transported ca. 5 km to the Leigh Marine Laboratory (36º16′09′′S; 174º47′54′′E) and maintained in ambient flow through for 2 weeks in a 460-L tank and fed with fresh Ecklonia radiata.
Temperature and salinity effects on early development
The complete experiment was run in three separate experimental trials using multiple males and females with no differences in sea urchin size (test diameter) between trials (average size ± SD: 6.2 cm ± 0.4 cm, N = 15 ANOVA, F (2,12) = 0.113, P = 0.894). In each trial, eggs from 3 females and sperm from 2 males were pooled to reduce inter-individual variation from single male–female crosses (Byrne et al. 2010, 2011). Sea urchins were induced to spawn by intracoelomic injection of 5 mL of KCl (0.55 M) and gentle agitation. Eggs were collected in 250-mL plastic containers with 1-µm filtered and UV-treated fresh seawater (FSW), whereas sperm were collected “dry” from the gonopore of the males and stored in 1.5-mL Eppendorf tubes at 4 °C until use. Eggs were washed 3 × with FSW and filtered through a 200-µm mesh to remove extraneous material such as feces or spines. Egg quality was checked by observing the eggs under a microscope to check for roundness; additionally, eggs were also checked for fertilization success in a test assay, and only eggs with >95 % fertilization success were used for pooling and experimentation. Eggs were pooled in a 1-L container with FSW, and then, the concentration was determined based on three subsamples of 100 µL. Sperm quality was checked by observing the motility of diluted sperm under the compound microscope at 40 × magnification. Suitable dry sperm were pooled in equal amounts in a petri dish, and then, 20 µL of the pooled sperm was diluted in 60 mL of FSW to obtain a sperm concentration suitable to ensure fertilization and avoid polyspermy (50,000–100,000 sperm mL−1, Franke 2005). The final sperm concentration determined from hemocytometer counts was 85,556 ± 11,706 sperm mL−1 (±SD, n = 3).
Experimental design
A combination of five salinities (29, 31, 34, 35 and 37 ppt) and two temperatures (18 and 21 °C) was used for experiments on early developmental stages. The salinity levels used included the ambient salinity for coastal and ocean waters in New Zealand (34 and 35 ppt, respectively, Garner 1969), with two salinities below and one salinity above the ambient levels. The temperatures used correspond to optimal temperatures for development of this species in north-eastern New Zealand, with 18 °C being a temperature which results in a high percent of normal development (>90 %), and 21 °C being the upper pejus temperature for development of the study population of E. chloroticus (Delorme and Sewell 2013). Thus, the control treatments for this study were considered to be 34 and 35 ppt and 18 °C.
Eggs were fertilized, and embryos cultured in 35-mL plastic containers with FSW at the different salinities (three replicates per salinity). The containers were held by a polypropylene rack inside a 14-L tank with flowing seawater at the desired temperature controlled by a heating unit. Salinity treatments were prepared using deionized water and sea salt (Red Sea Salt) according to the package instructions and based on refractometer measurements. Temperature and salinity were checked before and after the experiment with a submersible mercury thermometer and a refractometer, respectively. In addition, measurements of dissolved oxygen and pH were measured at the end of the experiment in containers with embryos at the same experimental conditions and with FSW-only using a Hach IntelliCAL™ LDO101 probe and a pH tester with spear electrode, respectively.
In each experimental trial, ~1,000 pooled eggs were placed in each of the three containers (per temperature-salinity treatment), and after 30 min of egg acclimation, 90 µL of pooled diluted sperm was added (without previous acclimation of the sperm to the different salinity treatments). Embryo samples (500 µL) were taken from each container at different time points after fertilization [1, 2, 4, 8 and 24 h post-fertilization (hpf)]. Pre-hatched embryos (1, 2 and 4 hpf) were sampled from the bottom of the container; for post-hatch (8 and 24 hpf), the containers were gently agitated to make an even suspension of the embryos before sampling. Each sample was placed in a 1.5-mL Eppendorf tube and preserved with 5 % formalin in seawater for later analysis. One hundred and fifty samples were collected in each of the three experimental trials (5 salinities, 2 temperatures, 5 times after fertilization, 3 replicates per treatment), with a total of 450 samples collected during the complete experiment (3 separate trials of 3 populations of embryos). Each sample was placed in a Sedgewick Rafter counting cell, and the cell was placed under the microscope for scoring of embryos. The first 100 embryos observed in a random transect across the cell were scored for developmental stage as unfertilized eggs, fertilized eggs, 2-cell, 4-cell, 8-cell and 16-cell embryos, blastula, gastrula and abnormal embryos (degenerating eggs/embryos, asymmetric divisions). Fertilization success was evaluated at 1 hpf, and the sum of all normally developing embryos was used to calculate the percent normal development at each time after fertilization for each treatment. The percent of gastrula stage embryos was determined at 24 hpf in each treatment and graphed as lines to show the pejus points, which were considered to be when gastrulation/performance fell below 75 % (Delorme and Sewell 2013). The average from the three experimental trials was used for all illustrations.
The rate of early development was calculated for each treatment based on the number of divisions of the embryos at each time after fertilization up to 4 hpf. The number of divisions was determined based on the number of cells of each developmental stage, with 1 division = 2-cells, 2 divisions = 4-cells, 3 divisions = 8-cells, and 4 divisions = 16-cells or more. The logarithm of the number of divisions in each treatment was plotted against the logarithm of the time after fertilization in order to linearize the data, with the regression slope being the development rate in each treatment (Cowart et al. 2009). In order to obtain a mean development rate per treatment, a single slope was calculated for each replicate (each population of embryos) allowing further statistical analyses of the development rate in each treatment.
Statistical analyses
Dissolved oxygen and pH data from containers with embryos and with FSW-only were analyzed for each temperature treatment. Dissolved oxygen data met the assumptions of normality and homoscedasticity (Shapiro–Wilk and Levene’s tests, respectively), and a one-way ANOVA was performed. In contrast, pH data did not meet the assumptions and a Kruskal–Wallis nonparametric test was performed.
The percent of embryos in each developmental stage and time after fertilization was calculated from the frequency data obtained from each sample and arcsine transformed for statistical analyses. Statistical analyses were performed on the data resulting from the average of the three experimental trials from fertilization success (1 hpf), number of divisions, early development rate, gastrulation and normal development. Normal development was calculated at each time after fertilization until 24 hpf. As all the data sets met the assumptions of normality and homoscedasticity, they were analyzed with two-way ANOVA with temperature and salinity as factors, and the percent of fertilization, number of divisions, rate of early development, gastrulation or normal development as the dependent variables. The number of divisions and the percent of normal development were analyzed separately for each time point after fertilization. Comparisons between groups were analyzed using the Tukey a posteriori test (α = 0.05). All statistical analyses were run using the statistical software included within Sigma Plot 11.0. (SYSTAT Software, Inc.).
Results
Average temperatures and salinities (±SD) during experimentation across the three experimental trials were 18.0 ± 0.2 °C and 20.8 ± 0.1 °C, and 29 ± 0.3, 31 ± 0.1, 34 ± 0.3, 35 ± 0.1, 37 ± 0.3 ppt. At 24 hpf, oxygen saturation and pH in the containers were >90 % and 8.1 ± 0.1 pH units, respectively, with no significant differences between containers with embryos and with FSW-only (oxygen: ANOVA, F (1,23) = 3.5, P = 0.076 and F (1,23) = 3.8, P = 0.065 for 18 and 21 °C, respectively; pH: Kruskal–Wallis test, H 1 = 2.323, P = 0.128 and H 1 = 0.397, P = 0.529 for 18 and 21 °C, respectively).
Fertilization success
Fertilization of E. chloroticus 1 h after sperm addition was over 80 % in all treatments (Fig. 1). Statistically, fertilization was affected by salinity (ANOVA, F (4,20) = 5.313, P = 0.004), with no significant effect of temperature (ANOVA, F (1,20) = 2.649, P = 0.119) and no significant interaction between factors (ANOVA, F (4,20) = 0.502, P = 0.735). Within salinity treatments, fertilization was lower at 29 ppt compared to 34, 35 and 37 ppt (Table 1; Fig. 1).
Effect of salinity and temperature on fertilization success in Evechinus chloroticus measured by the percent of fertilized eggs observed at 1 h after addition of sperm. Data represent the mean percent of fertilized eggs ± standard error (±SE, n = 3) at each salinity (29, 31, 34, 35 and 37 ppt) and temperature (18 and 21 °C). Significant differences between groups are denoted by lower case letters above bars and in Table 1
Early development
The rate of development of E. chloroticus increased with an increase in temperature from 18 to 21 °C; however, lower salinities (29 and 31 ppt) resulted in a decrease in development rate compared to high salinities at the same temperature (Fig. 2). At 1 hpf, most embryos were fertilized at 18 °C in all salinity treatments, whereas at 21 °C embryos were undergoing cleavage. At 2 and 4 hpf, embryos had more advanced development at higher temperature, but again embryos cleaved fewer times at lower salinities (29 and 31 ppt) compared to high salinities in both temperatures (Fig. 2). At 8 hpf, in all salinities, most embryos were at the 16-cell stage at 18 °C, whereas at 21 °C embryos had reached the blastula stage; however, the percent of 16-cell and blastula was lower at 29 ppt than at higher salinities in both temperatures, reaching only 31 and 45 % at 18 and 21 °C, respectively (Fig. 2).
Effect of salinity and temperature on early development in Evechinus chloroticus. Data represent the mean percent of each stage ± standard error (±SE, n = 3) at each salinity (29, 31, 34, 35 and 37 ppt) and temperature (18 and 21 °C). U: unfertilized eggs, F: fertilized eggs, 2: 2-cell, 4: 4-cell, 8: 8-cell, 16: 16-cell or more, B: blastula, and A: abnormal embryos
The mean number of divisions was affected by salinity and temperature at all times after fertilization (Table 2; Fig. 3). At 1 hpf, temperature had a significant effect on cleavage as the number of divisions were higher at 21 °C in 31, 34, 35 and 37 ppt (Table 2; Fig. 3). Additionally, at salinities of 29, 31 and 34 ppt, the number of divisions was lower compared to the other salinity treatments (Table 2; Fig. 3). At 2 hpf, the number of divisions was lower at 18 °C and 29 and 31 ppt (Table 2; Fig. 3), whereas at 4 hpf the number of divisions was also lower at 18 °C but only at a salinity of 29 ppt (Table 2; Fig 3). Development rate was significantly lower at 18 °C in all salinities compared to 21 °C, whereas within temperature treatments development rate at 29 was ca. 1/4 and ca. 3/4 of the rate at salinities greater than 34 ppt at 18 and 21 °C (Table 2; Fig. 4).
The effect of salinity and temperature on the number of divisions observed at different hours post-fertilization (hpf) in Evechinus chloroticus. Data represent the mean number of divisions ± standard error (±SE, n = 3) at each salinity (29, 31, 34, 35 and 37 ppt) and temperature (18 and 21 °C). Significant differences in the number of divisions at each time after fertilization are denoted by lower case letters above bars and in Table 2
Effect of salinity and temperature on development rate in Evechinus chloroticus. Data represent the mean development rate ± standard error (±SE, n = 3) at each salinity (29, 31, 34, 35 and 37 ppt) and temperature (18 and 21 °C) obtained from the slope of the regression line between the number of divisions and time after fertilization at each salinity and temperature. Significant differences in development rate are shown in Table 2
Gastrulation in E. chloroticus was affected by salinity (ANOVA, F (4,20) = 73.825, P < 0.001) and temperature (ANOVA, F (4,20) = 58.934, P < 0.001), with a significant interaction between factors (ANOVA, F (4,20) = 8.110, P < 0.001) (Table 1; Fig. 5). Lower numbers of gastrulae were observed at 29 and 31 ppt in both temperatures (Table 1; Fig. 5), whereas the highest percent of gastrulae was observed at 34 and 35 ppt with 79 and 91 % of gastrula observed at 18 and 21 °C, respectively (Table 1; Fig. 5).
Effect of salinity and temperature on the percent of gastrulae observed at 24 h post-fertilization in Evechinus chloroticus. Data represent the mean percent of gastrulae ± standard error (±SE, n = 3) at each salinity (29, 31, 34, 35 and 37 ppt) and temperature (18 and 21 °C). Broken line indicates the 75 % gastrulation point, and arrows show the pejus points in salinity for each temperature. Significant differences in gastrulation are shown in Table 1
Normal development
Normal development decreased with time in all treatments (Fig. 6). At 2 hpf, the percent of normal embryos was only affected by salinity, with no interaction between factors (Table 1). At this time, normal embryos were >80 % in all salinity/temperature treatments (Fig. 6), with a significant ~10 % reduction in normal embryos at 29 ppt compared to 34 and 35 ppt at both temperatures (Table 1; Fig. 6). At 4 hpf, normal development was also only affected by salinity; however, a significant interaction between salinity and temperature occurred (Table 1). At 18 °C, the percent of normal embryos at 29 and 31 ppt (53–68 %, respectively) was significantly lower compared to the normal development observed at 34, 35 and 37 ppt (Table 1; Fig. 6). At 21 °C, the percent of normal embryos at 29 ppt was ~37 % lower than that observed at 31, 34, 35 and 37 ppt (Table 1; Fig 6). In addition, the significant interaction between factors at 4 hpf showed that at 31 ppt, there were ~19 % less normal embryos at 18 °C than at 21 °C (Table 1; Fig. 6).
Effect of salinity and temperature on normal development at different times after fertilization in Evechinus chloroticus. Data represent the mean percent of normal embryos ± standard error (±SE, n = 3) at each salinity (29, 31, 34, 35 and 37 ppt) and temperature (18 and 21 °C). Significant differences in normal development at each time after fertilization are shown in Table 1
At 8 and 24 hpf, there was a significant further reduction in normal development at 29 and 31 ppt at both temperatures, with no interaction between factors (Table 1; Fig. 6). At 8 hpf, normal development at 29 ppt was 49 and 25 % lower than the other treatments at 18 and 21 °C, respectively (Table 1; Fig. 6), whereas at 31 ppt normal development was 44 and 24 % lower than the other salinity treatments at 18 and 21 °C, respectively (Table 1; Fig. 6). At 24 hpf, normal development at 29 and 31 ppt was even lower, reaching less than 45 and 65 %, respectively, in both temperature treatments. At 24 hpf, the highest percent of normal development was observed at 34 and 35 ppt with 89 and 92 % at 18 and 21 °C, respectively (Fig. 6).
Discussion
In the present study, both fertilization and early development of the sea urchin E. chloroticus were affected by lower salinities, whereas the seawater temperatures used had an effect only on development rate and gastrulation. In multistressor studies, the tested environmental stressors can interact to have synergistic, antagonistic or additive effects on performance (Folt et al. 1999). Only an antagonistic effect was observed in this study, where an increase in seawater temperature of 3 °C (within optimal range of the study population in northern New Zealand) minimized the negative effect of low salinities, as previously observed in early development of other sea urchins (Roller and Stickle 1993; Bressan et al. 1995; Kashenko 2006; Armstrong et al. 2013).
As a single stressor, seawater temperature is known to negatively affect optimum performance and survival of ectothermic animals (Willmer 1999). In E. chloroticus, seawater temperature has been reported to affect early development, decreasing normal development at temperatures of 24 °C, i.e., 3 °C above upper pejus temperature (Delorme and Sewell 2013). As the seawater temperatures used in the present study were within the optimal thermal window for early development of this species (Delorme and Sewell 2013), seawater temperature had a positive effect on early development. In contrast, salinity had a negative effect on early development of E. chloroticus, from fertilization success to normal development.
Fertilization success was significantly affected by low salinity [effect size of −0.13 calculated as the LnRR: natural log of the ratio between the treatment response and the control response (Nakagawa and Cuthill 2007)]; however, it had greater effects on early development (effect size of −0.83). Low salinity in E. chloroticus affected cleavage directly (i.e., through mitosis) and increased the occurrence of abnormalities in embryos (i.e., asymmetric cleavage patterns and degenerating embryos; Roller and Stickle 1985, 1993, 1994; Metaxas 1998; Kashenko 2006, 2007, 2009; Allen and Pechenik 2010; Carballeira et al. 2011; Li et al. 2011). In other echinoids, low salinity has been shown to have a greater effect on embryonic development (before hatching) than in the swimming stages (Kashenko 2007, 2009) and may completely inhibit development (Roller and Stickle 1994; Cowart et al. 2009). This sensitivity during early development has also been observed in holothurians (Li et al. 2011) and asteroids (Kashenko 2006; Pia et al. 2012). However, developmental stages of asteroids may be more resilient to decreasing salinities when compared to echinoids with a similar geographical distribution (Sameoto and Metaxas 2008).
Although the mechanism by which salinity affects cleavage is still unknown, it has been suggested that survival may be affected at extreme salinities (low and high) due to a change in the osmotic balance of cells, by changes in the absorption and saturation of dissolved gases (Kinne 1964 fide Li et al. 2011) or by the effect of reduced osmotic pressure either on proteins associated with the microtubules or functioning of the spindles during mitosis (Sharp et al. 2000; Sharp 2002). In addition, it has been recently reported that sea urchin embryos do not develop under absence of external Na+ due to a reduced activation of the mitosis promoting factor (MPF), which may be the cause of developmental arrest, whereas under low Na+ the cell cycle stops due to intracellular acidification, leading to cell death (Ciapa and Philippe 2013).
Low salinity also had a negative effect on development rate, which was slowed down at 29 ppt by 63 % in the 18 °C treatment and by 30 % in the 21 °C treatment, suggesting that complete PLD in E. chloroticus (21–30 days, Dix 1969; Walker 1984) would take 13–19 days longer at 18 °C and 6–9 days longer at 21 °C. An increase in PLD of this magnitude would likely increase mortality of the larvae due to an increase in predation pressure and the dependence on plankton as a food source (Thorson 1950; Morgan 1995; O’ Connor et al. 2007), and an increased risk of being carried out by currents to areas where conditions might be more unfavourable for development (Thorson 1950; Morgan 1995; Shanks 1995).
Previous laboratory experiments have shown that echinoid species and developmental stages differ in their salinity tolerance (Cameron et al. 1985; Kashenko 2007). For instance, the sand dollar Echinarachnius parma only tolerates short periods (hours) in low salinity (Allen and Pechenik 2010), whereas the heart-shaped sea urchin Echinocardium cordatum and sand dollar Scaphechinus mirabilis can tolerate long periods (days) (Kashenko 2007, 2009). In addition to the differences in tolerance, developmental stages might also avoid exposure to low salinity waters by delaying hatching, as observed in E. cordatum (Kashenko 2007) and E. parma (Armstrong et al. 2013). Small numbers of Sterechinus neumayeri and E. chloroticus embryos have also been observed to show delayed hatching in response to pH and seawater temperature, respectively (unpub. data), suggesting that delaying hatching might be a generalized response of the early developmental stages of echinoderms to stress. On the other hand, post-hatched larval stages can avoid low salinity by swimming away from vertically stratified low salinity waters (Metaxas and Young 1998; Sameoto and Metaxas 2008), increasing the likelihood of larval survival (Metaxas 1998). In the wild, Antonie (2003) monitored the fiords of the South Island showing no occurrence of larvae of E. chloroticus in the low salinity layer (LSL), suggesting either an ability of the larvae to avoid low salinities or, alternatively, that all the larvae that swim to the LSL die because of the extreme low salinity of the water.
The experimental salinity tolerance in this study for early developmental stages of a north-eastern population of E. chloroticus was 29 ppt. The results obtained for normal development at this salinity are similar to that previously reported for Doubtful Sound populations, where incomplete development was seen at salinities of 27.5 ppt (Antonie 2003). Salinities lower than 29 ppt are currently observed in New Zealand (Zeldis and Smith 1999; Peake et al. 2001), and therefore, it is likely that salinity may be currently limiting the distributional range of E. chloroticus due to poor embryo tolerance, which may be part of the explanation for the absence of this species from estuarine habitats, or alternatively, adult E. chloroticus may be avoiding spawning at low salinity. However, even considering that more extreme and frequent rainfalls are projected throughout New Zealand due to climate change by 2100 (IPCC 2014; Renwick et al. 2013), it is still premature to predict whether E. chloroticus will change its distributional range as a result of changes in salinity. In addition, phenotypic plasticity in embryos of E. chloroticus has been observed at high temperatures (Delorme and Sewell 2013), and now at low salinities with ca. 50 % embryos surviving at 29 ppt (this study), suggesting genetic capacity of this species to deal with environmental change (Byrne and Przeslawski 2013).
In conclusion, we know that seawater temperature as a single stressor can have major effects on this species, particularly for north-eastern populations, which are developing at temperatures very close to their thermal tolerance limit (Delorme and Sewell 2013). However, here we report that the addition of low salinity as another climate change stressor could have even more dramatic effects on early development in E. chloroticus. These results are of particular interest considering that low seawater pH, as another climate change stressor, has also been shown to decrease survival, larval size and calcification index in E. chloroticus (Clark et al. 2009). Climate change stressors will act together in the wild, which may have major consequences for development and recruitment of this species and flow-on effects to the shallow subtidal ecosystem of northeast New Zealand.
References
Allen JD, Pechenik JA (2010) Understanding the effects of low salinity on fertilization success and early development in the sand dollar Echinarachnius parma. Biol Bull 218:189–199
Antonie CR (2003) Effects of low salinity on Evechinus Chloroticus Valenciennes. MSc Thesis, University of Otago, Dunedin
Armstrong AF, Blackburn HN, Allen JD (2013) A novel report of hatching plasticity in the phylum Echinodermata. Am Nat 181:264–272. doi:10.1086/668829
Azad KA, McKinley S, Pearce CM (2010) Factors influencing the growth and survival of larval and juvenile echinoids. Rev Aquac 2:121–137. doi:10.1111/j.1753-5131.2010.01030.x
Barker M (2007) Ecology of Evechinus chloroticus. In: John ML (ed) Developments in aquaculture and fisheries science. Elsevier, Amsterdam, pp 319–338
Bressan M, Marin M, Brunetti R (1995) Influence of temperature and salinity on embryonic development of Paracentrotus lividus (Lmk, 1816). Hydrobiologia 304:175–184. doi:10.1007/bf02329312
Byrne M, Przeslawski R (2013) Multistressor impacts of warming and acidification of the ocean on marine invertebrates’ life histories. Integr Comp Biol 53:582–596. doi:10.1093/icb/ict049
Byrne M, Ho M, Selvakumaraswamy P, Nguyen HD, Symon AD, Davis AR (2009) Temperature, but not pH, compromises sea urchin fertilization and early development under near-future climate change scenarios. Philos Trans R Soc B Biol Sci 276:1883–1888. doi:10.2307/30244023
Byrne M, Soars N, Selvakumaraswamy P, Dworjanyn SA, Davis AR (2010) Sea urchin fertilization in a warm, acidified and high pCO2 ocean across a range of sperm densities. Mar Environ Res 69:234–239. doi:10.1016/j.marenvres.2009.10.014
Byrne M, Selvakumaraswamy P, Ho MA, Woolsey E, Nguyen HD (2011) Sea urchin development in a global change hotspot, potential for southerly migration of thermotolerant propagules. Deep Sea Res Part II 58:712–719. doi:10.1016/j.dsr2.2010.06.010
Byrne M, Foo S, Soars NA, Wolfe KDL, Nguyen HD, Hardy N, Dworjanyn SA (2013) Ocean warming will mitigate the effects of acidification on calcifying sea urchin larvae (Heliocidaris tuberculata) from the Australian global warming hot spot. J Exp Mar Biol Ecol 448:250–257. doi:10.1016/j.jembe.2013.07.016
Cameron R, Boidron-Metairon I, Monterrosa O (1985) Does the embryonic response to temperature and salinity by four species of Caribbean sea urchins parallel the reproductive synchrony? In: Proc 5th Int Coral Reef Congr 5: pp 273–278
Carballeira C, Martín-Díaz L, DelValls TA (2011) Influence of salinity on fertilization and larval development toxicity tests with two species of sea urchin. Mar Environ Res 72:196–203. doi:10.1016/j.marenvres.2011.08.008
Castro P, Huber ME (2005) Marine biology. McGraw-Hill, Boston
Ciapa B, Philippe L (2013) Intracellular and extracellular pH and Ca are bound to control mitosis in the early sea urchin embryo via ERK and MPF activities. PLoS One 8:e66113. doi:10.1371/journal.pone.0066113
Clark D, Lamare M, Barker M (2009) Response of sea urchin pluteus larvae (Echinodermata: Echinoidea) to reduced seawater pH: a comparison among a tropical, temperate, and a polar species. Mar Biol 156:1125–1137. doi:10.1007/s00227-009-1155-8
Cowart D, Ulrich P, Miller D, Marsh A (2009) Salinity sensitivity of early embryos of the Antarctic sea urchin, Sterechinus neumayeri. Polar Biol 32:435–441. doi:10.1007/s00300-008-0536-7
Delorme NJ, Sewell MA (2013) Temperature limits to early development of the New Zealand sea urchin Evechinus chloroticus (Valenciennes, 1846). J Therm Biol 38:218–224. doi:10.1016/j.jtherbio.2013.02.007
Diaz-Perez L, Carpizo-Ituarte E (2011) Effect of thermal stress on survival and delay of metamorphosis in larvae of the purple sea urchin Strongylocentrotus purpuratus. Cienc Mar 37:403–414
Dix T (1969) Larval life span of the echinoid Evechinus chloroticus (val.). New Zeal J Mar Fresh 3:13–16. doi:10.1080/00288330.1969.9515273
Dix T (1970) Biology of Evechinus chloroticus (Echinoidea: Echinometridae) from different localities: 1. General. New Zeal J Mar Fresh 4:91–116. doi:10.1080/00288330.1970.9515331
Fenwick G, Horning D (1980) Echinodermata of the Snares Islands, southern New Zealand. New Zeal J Mar Fresh 14:437–445. doi:10.1080/00288330.1980.9515888
Folt CL, Chen CY, Moore MV, Burnaford J (1999) Synergism and antagonism among multiple stressors. Limnol Oceanogr 44(3):864–877
Franke ES (2005) Aspects of fertilization ecology in Evechinus chloroticus and Coscinasterias muricata. PhD thesis, University of Auckland, Auckland
Fujisawa H (1995) Variation in embryonic temperature sensitivity among groups of the sea urchin, Hemicentrotus pulcherrimus, which differ in their habitats. Zool Sci 12:583–589. doi:10.2108/zsj.12.583
Garner DM (1969) The seasonal range of sea temperature on the New Zealand shelf. New Zeal J Mar Fresh 3:201–208. doi:10.1080/00288330.1969.9515289
George SB, Walker D (2007) Short-term fluctuation in salinity promotes rapid larval development and metamorphosis in Dendraster excentricus. J Exp Mar Biol Ecol 349:113–130. doi:10.1016/j.jembe.2007.05.010
Greenwood PJ, Bennett T (1981) Some effects of temperature-salinity combinations on the early development of the sea urchin Parechinus angulosus (Leske). Fertilization. J Exp Mar Biol Ecol 51:119–131. doi:10.1016/0022-0981(81)90124-6
Hardy NA, Lamare M, Uthicke S, Wolfe K, Doo S, Dworjanyn S, Byrne M (2014) Thermal tolerance of early development in tropical and temperate sea urchins: inferences for the tropicalization of eastern Australia. Mar Biol 161:395–409. doi:10.1007/s00227-013-2344-z
IPCC (2014) Climate change 2014: Impact, adaptation and vulnerability. Working Group II contribution to the IPCC 5th Assessment Report
Kashenko S (2006) The combined effect of temperature and salinity on development of the sea star. Russ J Mar Biol 32:37–44. doi:10.1134/s1063074006010056
Kashenko S (2007) Adaptive responses of embryos and larvae of the heart-shaped sea urchin Echinocardium cordatum to temperature and salinity changes. Russ J Mar Biol 33:381–390. doi:10.1134/S1063074007060041
Kashenko S (2009) Effects of extreme changes of sea water temperature and salinity on the development of the sand dollar Scaphechinus mirabilis. Russ J Mar Biol 35:422–430. doi:10.1134/S1063074009050083
Kinne O (1964) The effects of temperature and salinity on marine and brackish water animals. II. Salinity and temperature-salinity combinations. Oceanogr Mar Biol Annu Rev 2:281–339
Lalli CM, Parsons TR (1997) Biological oceanography: an introduction. Butterworth Heinemann, Oxford
Lawrence JM (1975) The effect of temperature-salinity combinations on the functional well- being of adult Lytechinus variegatus (Lamarck) (Echinodermata, Echinoidea). J Exp Mar Biol Ecol 18:271–275. doi:10.1016/0022-0981(75)90111-2
Li L, Li Q, Sun X, Kong L (2011) Effects of temperature and salinity on larval growth, survival, and development of the sea cucumber Apostichopus japonicus. N Am J Aquacult 73:296–303. doi:10.1080/15222055.2011.598373
Metaxas A (1998) The effect of salinity on larval survival and development in the sea urchin Echinometra lucunter. Invertebr Reprod Dev 34:323–330. doi:10.1080/07924259.1998.9652667
Metaxas A, Young CM (1998) Behaviour of echinoid larvae around sharp haloclines: effects of the salinity gradient and dietary conditioning. Mar Biol 131:443–459. doi:10.1007/s002270050337
Moore HB (1966) General biology of echinoderms. In: Boolootian RA (ed) Physiology of Echinodermata. Interscience Publishers, New York, pp 1–48
Morgan SG (1995) Life and death in the plankton: larval mortality and adaptation. In: McEdward LR (ed) Ecology of marine invertebrate larvae. CRC Press, New York
Nakagawa S, Cuthill IC (2007) Effect size, confidence interval and statistical significance: a practical guide for biologists. Biol Rev 82:591–605. doi:10.1111/j.1469-185x.2007.00027.x
Nottage RAC, Wratt DS, Bornman JF, Jones K (2010) Climate change adaptation in New Zealand: future scenarios and some sectoral perspectives. New Zealand Climate Change Centre, Wellington
O’ Connor MI, Bruno JF, Gaines SD, Halpern BS, Lester SE, Kinlan BP, Weiss JM (2007) Temperature control of larval dispersal and the implications for marine ecology, evolution, and conservation. Proc Natl Acad Sci USA 104:1266–1271. doi:10.1073/pnas.0603422104
Peake B, Walls D, Gibbs M (2001) Spatial variations in the levels of nutrients, chlorophyll a, and dissolved oxygen in summer and winter in Doubtful Sound, New Zealand. New Zeal J Mar Fresh 35:681–694. doi:10.1080/00288330.2001.9517035
Pia TS, Johnson T, George SB (2012) Salinity-induced morphological changes in Pisaster ochraceus (Echinodermata: Asteroidea) larvae. J Plankton Res 34:590–601. doi:10.1093/plankt/fbs032
Renwick J, Mullan B, Wilcocks L, Zammit C, Sturman J, Baisden T, Keller L, Kirschbaum M, Meason D, Harrison D, Verkerk G, Cooke A, Marshall P, Clark A (2013) Four degrees of global warming: effects on the New Zealand primary sector. Ministry of Primary Industries, Wellington
Roller RA, Stickle WB (1985) Effects of salinity on larval tolerance and early developmental rates of four species of echinoderms. Can J Zool 63:1531–1538. doi:10.1139/z85-227
Roller RA, Stickle WB (1993) Effects of temperature and salinity acclimation of adults on larval survival, physiology, and early development of Lytechinus variegatus (Echinodermata: Echinoidea). Mar Biol 116:583–591. doi:10.1007/BF00355477
Roller RA, Stickle WB (1994) Effects of adult salinity acclimation on larval survival and early development of Strongylocentrotus droebachiensis and Strongylocentrotus pallidus (Echinodermata: Echinoidea). Can J Zool 72:1931–1939. doi:10.1139/z94-262
Russell MP (2013) Echinoderm responses to variation in salinity. Adv Mar Biol 66:171–212. doi:10.1016/B978-0-12-408096-6.00003-1
Sameoto JA, Metaxas A (2008) Can salinity-induced mortality explain larval vertical distribution with respect to a halocline? Biol Bull 214(3):329–338
Schiel D, Kingsford MJ, Choat JH (1986) Depth distribution and abundance of benthic organisms and fishes at the subtropical Kermadec Islands. New Zeal J Mar Fresh 20:521–535. doi:10.1080/00288330.1986.9516173
Sewell MA, Young CM (1999) Temperature limits to fertilization and early development in the tropical sea urchin Echinometra lucunter. J Exp Mar Biol Ecol 236:291–305. doi:10.1016/S0022-0981(98)00210-X
Shanks AL (1995) Mechanisms of cross-shelf dispersal of larval invertebrates and fish. In: McEdward LR (ed) Ecology of marine invertebrate larvae. CRC Press, New York
Sharp DJ (2002) Cell division: MAST Sails through mitosis. Curr Biol 12:R585–R587. doi:10.1016/S0960-9822(02)01098-9
Sharp DJ, Rogers GC, Scholey JM (2000) Microtubule motors in mitosis. Nature 407:41–47. doi:10.1038/35024000
Thorson G (1950) Reproductive and larval ecology of marine bottom invertebrates. Biol Rev 25:1–45. doi:10.1111/j.1469-185X.1950.tb00585.x
Trowbridge CD (1994) Life at the edge: population dynamics and salinity tolerance of a high intertidal, pool- dwelling ascoglossan opisthobranch on New Zealand rocky shores. J Exp Mar Biol Ecol 182:65–84. doi:10.1016/0022-0981(94)90211-9
Walker MM (1984) Larval life span, larval settlement, and early growth of Evechinus chloroticus (Valenciennes). New Zeal J Mar Fresh 18:393–397. doi:10.1080/00288330.1984.9516060
Walker J, Vaughan M (2013) Marine water quality annual report: 2011. Auckland Council technical report, TR2013/031. Auckland Council, Auckland
Willmer P (1999) Environmental physiology of animals. Blackwell, Massachusetts
Zeldis JR (2004) New and remineralised nutrient supply and ecosystem metabolism on the northeastern New Zealand continental shelf. Cont Shelf Res 24:563–581. doi:10.1016/j.csr.2003.11.008
Zeldis JR, Smith SV (1999) Water, salt and nutrient budgets for Hauraki Gulf New Zealand. In: Smith SV, Crossland CJ (eds) Australasian Estuarine Systems: carbon, nitrogen and phosphorus fluxes, LOICZ Reports and Studies No. 12, 182 pp, LOICZ IPO, Texel, The Netherlands
Zeldis JR, Walters RA, Greig MJN, Image K (2004) Circulation over the northeastern New Zealand continental slope, shelf and adjacent Hauraki Gulf, during spring and summer. Cont Shelf Res 24:543–561. doi:10.1016/j.csr.2003.11.007
Acknowledgments
Thanks to Errol Murray, Peter Browne, for helping with the experimental setup, and to Brady Doak and Richard Taylor for providing diving equipment. Thanks to Leonardo Zamora for helping with animal collection, spawning and sampling. NJD was financially supported by a Chilean Government scholarship (Becas-Chile, National Commission for Scientific and Technological Research, CONICYT).
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by M. Byrne.
Rights and permissions
About this article
Cite this article
Delorme, N.J., Sewell, M.A. Temperature and salinity: two climate change stressors affecting early development of the New Zealand sea urchin Evechinus chloroticus . Mar Biol 161, 1999–2009 (2014). https://doi.org/10.1007/s00227-014-2480-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00227-014-2480-0