Abstract
Estuaries in coastal South Texas feature naturally extreme salinity fluctuations that are increasing in magnitude due to anthropogenic activities, with salinities exceeding 70 parts per thousand (ppt) in some areas during periods of recurrent drought. Red drum (Sciaenops ocellatus) are a recreationally valuable estuarine dependent fish species that rely on these increasingly saline estuaries to complete development. Though juvenile and adult life stages of red drum are known to be euryhaline, current evidence suggests this species is relatively stenohaline during early development. Therefore, the goal of this study was to evaluate the impacts of salinity stress on hatch and survival of early life stage (ELS) red drum. Hatch success at 24-h post fertilization (hpf) was reduced by 29% relative to controls when embryos were exposed to salinities elevated by as little as 2 parts per thousand (ppt) relative to spawning conditions. Larval attrition continued over the course of the 72-h study in all hypersaline treatments, with a ≥ 40% reduction in survival of yolk sac larvae relative to controls observed by test termination. Median lethal time (LT50) studies were also conducted to evaluate the relative sensitivities of embryonic and larval stages to osmotic shock. Results indicate that embryonic stages are more tolerant of extremely hypersaline conditions (68.7 ppt) relative to yolk sac larvae; however, both stages are considered stenohaline. As our experimental salinity concentrations incorporate the range of current summertime conditions in Texas estuaries, these findings have very real implications for ELS red drum populations in the Gulf of Mexico.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Estuaries act as receiving waters for terrestrially influenced freshwater inflows and organic matter and facilitate mixing with water from the open ocean (Canuel and Hardison 2016). This combination of factors drives many biologically important processes that are reliant on upwelling and the availability of nutrients and oxygen (Barbier et al. 2011; Canuel and Hardison 2016; Pesce et al. 2018; Wilson and Dunton 2018). Combined, these dynamics support high productivity and generate salinity gradients that are required by many estuarine-dependent species to complete key life history/developmental processes (Barbier et al. 2011; Buskey et al. 2012; Montagna and Kalke 1992). This includes many species of early life stage (ELS) marine fish and invertebrates that utilize these heterogeneous habitats as nurseries and feeding grounds (Barbier et al. 2011; Lewis et al. 2020).
The majority of commercially and recreationally valuable fisheries in the USA are comprised of estuarine-dependent species (Lellis-Dibble et al. 2008; Lewis et al. 2020; TPWD 2010). In fact, nearly 85% of native estuarine dependent fish species found in the Gulf of Mexico (GoM) are valued as a commercial or recreational fishery species (TPWD n.d. - a, b). Recreational fishing alone contributes over $125 billion USD to the national economy each year, with Texas ranked first in terms of single state contributions (DOI 2008; Lellis-Dibble et al. 2008; Lewis et al. 2020). Nearly two-thirds of Texas’ $3.2 billion USD recreational fishing industry is generated by only two estuarine-dependent Sciaenids, the spotted seatrout (Cynoscion nebulosus) and the red drum (Sciaenops ocellatus) (Buskey et al. 2012; Pulich et al. 2001; Puritz et al. 2014; Vega et al. 2012). The Texas Parks and Wildlife’s Coastal Fisheries Division estimates that recreational anglers catch approximately 225,000 red drum annually, making it the most valuable recreational fishery in Texas (TPWD n.d. - b).
In the often-arid months of late summer and fall, Texas red drum broadcast spawn in nearshore waters, following which the rapidly developing offspring are passively transported through tidal inlets to shallow estuarine nursery habitats (Brown et al. 2004; Davis 1990; Holt 2008). Though red drum can be found throughout Texas’ seven major estuaries, their abundance is highly variable between systems, with the Southernmost estuaries accounting for some of nation’s most robust populations.
These estuaries, including the Mission-Aransas Estuary, the Nueces Estuary, and the Upper Laguna Madre, are located primarily within a geographic region known as the Texas Coastal Bend (TCB). Estuaries in the TCB remain among some of the most well-preserved and productive estuarine habitats in the USA; however, a combination of industrialization, watershed urbanization, increasingly arid conditions, and other anthropogenic impacts is stressing freshwater inflows to TCB estuaries (Quammen and Onuf 1993; Engle et al. 2000; Buskey et al. 2012; Bugica et al. 2020; Lewis et al. 2020). Consequently, rates of evaporation often exceed recharge in these estuaries, leading to rising average salinities and recurrent bouts of extreme hypersalinity (Montagna et al. 2018; CBBEP 2020). This is particularly true for the Nueces Estuary, where increasing pressures on inflow and a changing climate have resulted in hypersaline conditions as high as 39 parts per thousand (ppt) for over 37% of the last two decades (Buskey et al. 2012; CBBEP 2020; Pulich et al. 2001; Wilson and Dunton 2018). Thus, it is reasonable to suggest that the hypersaline conditions in the Laguna Madre (which has no major riverine influences and commonly reaches salinities of 70 ppt or more; TPWD, n.d. - c) represent future extreme salinities that ELS red drum may encounter if current trends in the TCB continue.
Although juvenile and adult stage of estuarine-dependent fishes is often tolerant to a wide range of salinities and/or can move to more favorable conditions, this is often not true for ELS fish, which are typically more sensitive to physical and chemical environmental stressors (Alloy et al. 2017; Crocker et al. 1983; Damare et al. 2018; Khursigara et al. 2017; Magnuson et al. 2018; Nielsen et al. 2022; Nielsen et al. 2018). The tolerance of ELS fish to hypersaline conditions can further vary between species and developmental stage. For example, embryos are often less affected by variations in salinity than larvae, as there is often less overall exchange occurring across the chorion (Lein et al. 1997). Generally, salinities that are too far outside of a species’ tolerated upper or lower range result in increased larval mortality, with effects demonstrating dependence on number of days post hatch (Lein et al. 1997; Estudillo et al. 2000; Dhaneesh et al. 2012; Garrido et al. 2015; Martin and Esbaugh 2021).
Those that initially survive may still experience adverse effects that lead to latent mortality or reduced fitness (e.g., increased metabolic rates, reduced neural tissue mass, smaller eye size/poor visual acuity, smaller size), as the energetic burden of osmoregulation under extreme conditions can meaningfully reduce the overall energy budget available to complete development (Garrido et al. 2015; Gong et al. 2018; Moran et al. 2015; McKenzie et al. 2007; Passow et al. 2015; Banks et al. 1991). Therefore, the goal of the present study was to investigate the survival and growth implications of anthropogenic driven salinity stress for ELS red drum that are dependent upon South Texas’ estuarine habitats, across multiple stages of development.
We exposed ELS red drum to a range of environmentally relevant and predicted future extreme salinities, while manipulating the timing of exposure to represent different tide- and wind-driven exposure scenarios.
Methods
All experimental protocols were approved by the Institutional Animal Care and Use Committee (IACUC) at The University of Texas at Austin (AUP-2021–00,068 & AUP-2021–00,078).
Embryo Collection
Embryos were collected from wild-caught spawning broodstock red drum maintained by Texas Parks and Wildlife Department (TPWD) – CCA Marine Development Center (Corpus Christi, Texas, USA) or the University of Texas Marine Science Institute (UTMSI; Port Aransas, Texas, USA) 3- to 6-h post fertilization (hpf). Embryos obtained from TPW were transported to UTMSI in water sourced from spawning tanks that were continuously aerated and held at a constant temperature during transport. All embryos were disinfected in 1-ppt formalin for 1 h, rinsed with sterilized isotonic (to transport water) seawater and checked for buoyancy/viability. All tests were terminated prior to first exogenous feeding of red drum, which occurs at approximately 96-hpf. Hatching success, survival parameters, and morphological changes were evaluated to assess the potential future impacts of hypersalinity on red drum populations, via reduced recruitment to the breeding population.
Median Lethal Salinity Studies
Control salinity for each exposure was adjusted to reflect that of spawning tanks. To assess the impacts of salinity on hatching success and early development, an initial set of embryos was exposed to the following salinities: 31-ppt (control), 35-ppt, 40-ppt, 45-ppt, 50-ppt, 60-ppt, and 68.7-ppt (20 embryos/replicate; n = 5 replicates/salinity), allowing us to identify a preliminary median lethal concentration (LC50) at nominal test hours 24, 48, and 72. At the conclusion of preliminary testing, morphological changes associated with salinity stress in survivors were identified. The highest salinity, 68.7-ppt, was chosen to reflect potential future anthropogenic-driven salinities that may be encountered by ELS red drum in the TCB.
To refine our LC50 value from the first test, a second set of embryos was exposed to a narrower range of salinities, which included a 35-ppt control (to match spawning tanks) and five hypersaline treatments that incrementally increased by 2-ppt intervals (i.e., 37-ppt, 39-ppt, 41-ppt, 43-ppt, and 45-ppt). To further reduce potential uncertainty associated with LC50 derivation, replication was also increased for the second study (20 eggs/replicate; n = 10/salinity). The lowest observed effect concentrations (LOEC) and no observed effect concentrations (NOEC) were derived for mortality at each of the previously identified nominal timepoints. A median effect concentration (EC50) was also calculated for morphological changes in survivors at nominal test hour 72.
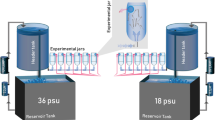
All exposure solutions were prepared by thoroughly mixing Instant Ocean® salts into 18-Ω ultrapure water (ELGA LabWater®, Woodridge, IL) to target salinities. A transfer pipette was used to gently load viable red drum embryos into pre-filled glass test chambers (250-mL) containing 200-mL aerated exposure solutions (> 7.67 mg/L, pH: 8.03–8.32). Test chambers were maintained in an environmentally controlled room (28 °C, 14-h light:10-h dark photoperiod) to ensure environmental parameters did not fluctuate. Water quality parameters (dissolved oxygen, salinity, temperature, pH) were measured in all stocks and test chambers daily. All water quality parameters remained within acceptable ranges, as previously described, for the entirety of the 72-h study.
Survival was assessed daily (i.e., every 24 h following initial exposure) in every replicate, at which time dead larvae were removed from exposure vessels. As red drum typically hatch between 24 and 30-hpf (Holt et al. 1981; Davis 1990; TPWD 2010), percent survival at the 24-h time point (i.e., number of larvae alive relative to the number of eggs at test start) was also representative of a successful hatch. Following the final survival assessment at test hour 72, larvae were humanely euthanized in buffered MS-222, placed in a methyl cellulose solution, and then imaged using a Nikon SMZ800N microscope (Nikon Metrology, Inc., Brighton, MI) fitted with a camera and accompanying software. To estimate differences in growth among treatments, body area, length, and eye size were quantified in surviving individuals from all replicates, using ImageJ (National Institutes of Health, version 1.8.0_172).
Median Lethal Time Studies
Depending on the spawning location and wind and tidal influences, ELS red drum may drift from nearshore spawning sites with typical seawater salinities into extremely hypersaline estuaries as embryos, or as yolk sac larvae. Therefore, the first median lethal time (LT50) study was designed to investigate the relative sensitivities of both embryonic and yolk sac larvae from the same spawn.
At test initiation, 20 embryos were transferred into replicate dishes (n = 8 replicates/salinity) containing either 28-ppt (control) or 68.7-ppt reconstituted seawater and survival was monitored hourly until hatch. Shortly after hatch, larvae from control dishes were equally divided into two cohorts for the larval component of the study. One larval cohort was transferred from control test chambers to a separate set of chambers containing control salinity seawater (to determine baseline survival), while the second cohort of newly hatched larvae was transferred to dishes containing 68.7-ppt seawater. Survival of larvae was continuously monitored until all individuals in the hypersaline treatment died. From these data, embryonic and larval LT50 values were calculated.
Due to the considerably higher relative sensitivity of yolk sac larvae observed during the initial round of LT50 testing, follow up testing did not include an embryonic component. Rather, the larval component of the study was repeated using a second set of embryos from TCB wild-caught broodstock that were maintained in spawning tanks with a higher relative salinity (35-ppt). This study design allowed us to investigate the importance of spawning conditions on the salinity tolerance of newly hatched red drum.
Statistical Analyses
Statistical tests were performed using JMP Pro (Version 15), using an α of 0.05. A Shapiro–Wilk test was used to test normality at all time points. If data were non-normally distributed, non-parametric tests were used.
At nominal test hour 24, percent successful hatch was calculated by dividing the number of living newly hatched larvae by the number of viable embryos loaded at test start (n = 20 embryos/test chamber). Larval survival (48 h and 72 h) was assessed, with percent survival calculated as the number of remaining (living) larvae relative to the number of embryos at study initiation. For the first set of embryos (i.e., those exposed to a wide range of salinities), survival and growth parameters, including body length, body area, and relative eye size, were determined to be non-normally distributed. Therefore, a Kruskal–Wallis one-way analysis of variance (ANOVA), followed by a Steel with control (28-ppt) post hoc test, was used to evaluate treatment effects at each assessment time point. LC50 values were estimated by fitting survival data from the appropriate time point with a logistic curve bounded between 0 and 1, followed by an inverse prediction of 50% survival.
For the second set of embryos (i.e., those exposed to a narrower range of salinities), survival data collected at test hours 24 and 72 were determined to be normally distributed, while survival data collected at test hour 48 was found to be non-normally distributed. LC50 and LT50 measures were estimated as described previously. The 48-h NOEC and LOEC values were calculated using a Wilcoxon non-parametric test followed by a Steel with control (35-ppt) post hoc test. NOEC and LOEC values collected at 72 h were assessed with a one-way ANOVA followed by a Dunnett’s post hoc test. Growth parameters were determined to be non-normally distributed and were analyzed using a Wilcoxon non-parametric test followed by a Steel with control (35-ppt) post hoc. LT50 values for embryos and larvae exposed to extreme hypersalinity (68.7-ppt) were calculated by fitting survival data over time with a logistic curve bounded between 0 and 1. An inverse prediction was used to predict the time to 50% survival.
Results
Embryonic Exposures and Survival
In the first set of embryos (i.e., those exposed to salinities ranging from 28 to 68.7-ppt), 24-h survival was 91.5% in the control group. Significant treatment effects on successful hatch/survival were apparent at this timepoint, with salinities > 50-ppt demonstrating significantly lower hatch success relative to those in less saline treatments (p < 0.001, F = 159.7, DF = 6). A complete failure to hatch was observed in the highest salinity treatment, with only one moribund hatched fish present in a 60-ppt replicate at the 24-h assessment (Fig. 1a).
Mean control survival at 48 h was 89.8%, with a subsequent decrease to 78.8% by test termination at 72 h (Fig. 1b, c; Table S1). Though significant effects on hatch were observed in the 50-ppt treatment at the first assessment timepoint, survival had fallen below 50% by 48 h (Fig. 1b; Table S1). By 72 h, survival in all but the lowest hypersaline treatments (35- and 40-ppt) had been significantly reduced relative to controls (p < 0.001, F = 83.2, DF = 76; Figs. 2 and 3; Table S1). Based on these data, a 72-h LC50 of 41.8-ppt was calculated for red drum larvae. Among this set of embryos, treatment effects on the size of larval morphological parameters were evaluated only for salinities ≤ 45-ppt, due to low survival at higher salinities. While a downward trend in larval length was observed, it was not statistically significant (Fig. 2a). However, the average body size of red drum larvae from the 45-ppt treatment was significantly (p < 0.001, F = 6.45, DF = 3) smaller when evaluated as mean lateral surface area (− 27% relative to controls; Fig. 2b). The normalized eye size of larval red drum was also significantly (p < 0.0001; F = 9.9; DF = 3) smaller in larvae from the 45-ppt treatment (− 29% relative to controls; Fig. 2c).
Impacts of salinity on larval morphology. Bars represent the percent change in mean length (A), body size (B), and normalized eye size (C) of red drum larvae, relative to control parameters. Error bars encompass ± 1 standard error around the mean and significant differences (p < 0.05) are indicated by an asterisk (*)
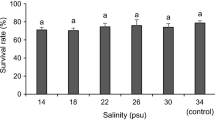
The LC50, NOEC, and LOEC values derived for the second set of embryos (i.e., those exposed to salinities ranging from 35 to 45-ppt) were relatively consistent with those from the first round of testing. However, significant adverse effects on survival were present in all hypersaline treatments, yielding a NOEC value of 35-ppt, which was equivalent to the control salinity (Table 1). Moreover, using a narrower range of salinities allowed us to calculate a refined 72-h LC50 value (37.7-ppt) with a high degree of certainty (Table 1). Survival data across salinities at each time point are shown in Fig. 3 and Table S1. Among these salinities, the lateral surface area of larvae was quantified to assess growth, with results indicating significant (p < 0.001; F = 4.9; DF = 5) negative effects on the mean body size of larvae from the 45-ppt treatment (− 20% relative to controls; Fig. 4).
Larval Exposure and Survival
Hatch success for the first round of LT50 testing was 97% at the 24-h assessment point and no mortality was observed among control animals during either round of larval LT50 testing. The LT50 values derived for embryonic and larval red drum from the 28-ppt spawning tanks indicate that yolk sac larvae are considerably more sensitive to osmotic stress, as it took over 8 times longer for embryos to reach 50% mortality (Table 2). A direct comparison between larval LT50 values for the two rounds of testing indicates that larvae spawned at 35-ppt survived approximately 17 min longer at 68.7-ppt, relative to their counterparts spawned at 28-ppt. These results indicate that spawning salinity appears to play a role in determining outcomes for newly hatched larvae experiencing osmotic stress; however, at extreme salinities, the difference is unlikely to be biologically meaningful (Table 2).
Discussion
Cumulatively, Texas has been in a state of drought for over 12 of the last 20 years (i.e., 60% of the time; CBBEP 2020), though this statewide average is very likely biased low for South Texas, which has a lower relative availability of freshwater and a more arid climate (CBBEP 2020). Moreover, according to the US Census Bureau (2018), Texas has one of the highest population growth rates in the USA, which is disproportionately represented in its coastal counties (Texas Comptroller 2018), further straining limited freshwater supplies and reducing inflow to estuaries. Consequently, an upward trend in estuarine salinities in the TCB has been identified using long-term monitoring data, which has been attributed to anthropogenic impacts such as climate change, coastal development, and urbanization of watersheds (Bugica et al. 2020; Buskey et al. 2012).
Current summertime drought conditions are known to result in extreme estuarine salinities (i.e., 95th percentile) in the Nueces Estuary as high as 39-ppt, with salinities of over 70-ppt in the Upper Laguna Madre (Bugica et al. 2020; CBBEP 2020; TPWD, n.d. - c). The survival data reported herein indicate that the current upper range of salinities in the TCB region is already near the salinity tolerance threshold of native red drum larvae. We also found significant decreases in body size among surviving individuals at higher salinities, indicating that even fairly minimal elevations in salinity (i.e., those currently experienced by larvae during summer drought conditions) may lead to significant adverse effects on the survival, growth, and/or energy homeostasis of larval red drum populations in the TCB (Garrido et al. 2015; Moran et al. 2015).
As red drum hatch < 30-hpf (Holt et al. 1981; Davis 1990; Matlock 1990; TPWD 2010), the effects of salinity changes during this short developmental window can significantly impact both hatching success and development. Our data suggest that exposure to environmentally relevant hypersalinities (both current and predicted) during early development can significantly reduce survival and growth, even if initial spawning conditions are not stressful. Half of the larvae that drift into hypersaline estuaries approximately 24-h post hatch (i.e., 48-hpf) are expected to experience acute lethality at salinities of 44.8-ppt. When hypersaline conditions persist in estuarine nursery grounds for a period of only 2 days, 50% mortality is expected for red drum larvae at salinities as low as 37.7-ppt. Our data further suggest that significant acute effects on the survival of newly hatched larval red drum will occur when salinities in TCB estuaries exceed 35 ppt for two days, even if the animals are spawned at 35-ppt. It is important to note that these values do not include estimates of latent mortality, which is very often an important factor in ELS toxicity studies.
These findings align with studies examining the salinity tolerance of 3-dph spotted sea trout larvae sharing estuarine habitats in the TCB (Banks et al. 1991). Within the spotted sea trout, the authors suggest this decreased salinity tolerance was associated with first feeding and the lack of osmoregulatory organs (e.g., kidneys, gills) present in 3-dph spotted sea trout (Banks et al. 1991).
The relatively stenohaline nature of yolk sac red drum demonstrated here is likely due to the high metabolic costs of osmoregulation under extreme salinity conditions, as red drum larvae rely on passive diffusion at the skin prior to the development of osmoregulatory (Alderdice et al. 1988). For example, in milkfish (Chanos chanos), Swanson et al. (1998) found that individuals exposed to higher salinities as embryos and larvae utilized significantly more yolk, meaning larvae at lower salinities had larger yolk reserves at hatch for sustenance prior to first exogenous feeding. Differences in yolk reserves at hatch may also significantly impact red drum survival and salinity tolerance over time, as red drum do not begin exogenous feeding until 96-hpf (Davis 1990; Matlock 1990; TPWD 2010).
Results of our median lethal time studies further reveal that yolk sac larvae are considerably more sensitive than red drum embryos when exposed to extreme salinity stress. Moreover, the median lethal time for larvae in the highest salinity tested was nearly 27% shorter for larvae spawned at 28-ppt, relative to those spawned at 35-ppt (i.e., 47.7-m from 65-m). As has been confirmed in other species (Hill 2004; Bourdreau et al. 2005), we predict that the chorion may partially mitigate the rate of water loss experienced by embryos exposed to hypersaline conditions, while newly hatched larvae are reliant on passive diffusion at the skin (Davis 1990; Matlock 1990; Torres et al. 1996). However, it is important to note that salinity tolerance is expected to increase with age as individuals develop osmoregulatory organs, consistent with other species (Estudillo et al. 2000; Faulk and Holt 2006).
Although red drum are known to be particularly salinity tolerant as juveniles and are able to quickly acclimate to hypersalinity both behaviorally and physiologically (Martin and Esbaugh 2021), it is clear that this tolerance is not conserved among early life stages in this species. We found that individuals exposed to salinities > 43-ppt were significantly smaller than individuals exposed to lower salinities, suggesting that growth among surviving fish was significantly reduced at higher salinities. Again, these data suggest a trade-off due to the high metabolic cost of osmoregulation under extreme salinity stress.
In general, fish living in highly stressful environments are known to express phenotypes that favor more efficient energy use and/or lower energy demands, including reduced body size, and metabolic suppression (Moran et al. 2015; Passow et al. 2015). Moreover, fish living in nutrient- and energy-limited environments have been found with reduced, or even completely absent eyes that correspond with reduced neural tissue mass (Moran et al. 2015). This phenotype is thought to reduce the overall energy requirements of development (as neural tissues, including visual sensory tissues, are energetically costly), thus allowing for normal overall growth rates in ELS fish (Moran et al. 2015; Passow et al. 2015).
Consistent with these findings, surviving red drum larvae had significantly reduced eye area relative to controls. This is especially significant for larval fishes, as vision is a critically important aspect of prey detection (especially as larvae transition to exogenous feeding), which is yet another factor underpinning larval growth rates and success in the wild (e.g., Higgs and Fuiman 1996; reviewed by Rønnestad et al. 2013).
While the smaller size of the larvae reared at higher salinities might be a consequence of higher osmoregulatory metabolic demands, it could also have significant implications for the longer-term survival of these larvae. Mortality among larval fish is known to be generally high and driven by phenotype (see review by Johnson et al. 2014), and a link between size at hatch and early survival has been noted among fishes. For example, Garrido et al. (2015) found that larger European sardine (Sardina pilchardus) larvae have better odds of survival. Interestingly, survival in this species was not correlated with egg size, but with an individual’s size at hatch (Garrido et al. 2015). Size has also been shown to significantly influence survival among common damselfish (Neopomacentrus filamentosus), such that larger individuals that grew faster were the fish most likely to survive months after hatching (Vigliola and Meekan 2002).
Together with the findings of previous studies, our data suggest that even transient increases in salinity may significantly affect the long-term survival of ELS red drum in the TCB. Multiple studies have highlighted the complexity of selective mortality on fish larvae, and how it can ultimately impact population dynamics over time (Johnson et al. 2014; Garrido et al. 2015). Thus, additional studies on the salinity tolerance of estuarine-dependent fishes are needed to inform management and conservation decisions in the face of intensifying anthropogenic climate change.
References
Alderdice, D.F. 1988. Osmotic and ionic regulation in teleost eggs and larvae. In: Hoar, W.S., Randall, D.J. Eds., Fish Physiology, vol. XI. Academic Press, San Diego: 163–251.
Alloy, M., T.R. Garner, K. Nielsen, C. Mansfield, M. Carney, H. Forth, M. Krasnec, C. Lay, R. Takeshita, J. Morris, S. Bonnot, J. Oris, and A. Roberts. 2017. Co-exposure to sunlight enhances the toxicity of naturally weathered Deepwater Horizon oil to early lifestage red drum (Sciaenops ocellatus) and speckled seatrout (Cynoscion nebulosus). Environmental Toxicology and Chemistry 36 (3): 780–785. https://doi.org/10.1002/etc.3640.
Banks, M.A., G.J. Holt, and J.M. Wakeman. 1991. Age-linked changes in salinity tolerance of larval spotted seatrout (Cynoscion nebulosus, Cuvier). Journal of Fish Biology 39: 505–514. https://doi.org/10.1111/j.1095-8649.1991.tb04382.x.
Barbier, E.B., S.D. Hacker, C. Kennedy, E.W. Koch, A.C. Stier, and B.R. Silliman. 2011. The value of estuarine and coastal ecosystem services. Ecological Monographs 81: 169–193. https://doi.org/10.1890/10-1510.1.
Boudreau, M., S.C. Courtenay, D.L. MacLatchy, C.H. Bérubé, L.M. Hewitt, and J.G. Van Der Kraak. 2005. Morphological abnormalities during early-life development of the estuarine mummichog Fundulus heteroclitus as an indicator of androgenic and anti-androgenic endocrine disruption. Aquatic Toxicology 71 (4): 357–369. https://doi.org/10.1016/j.aquatox.2004.12.005.
Brown, C.A., S.A. Holt, G.A. Jackson, D.A. Brooks, and G.J. Holt. 2004. Simulating larval supply to estuarine nursery areas: How important are physical processes to the supply of larvae to the Aransas Pass Inlet? Fisheries Oceanography 13: 181–196.
Bugica, K., B. Sterba-Boatwright, and M.S. Wetz. 2020. Water quality trends in Texas estuaries. Marine Pollution Bulletin 152: 110903. https://doi.org/10.1016/j.marpolbul.2020.110903.
Buskey, E., C. Cook, A. Evans, B. Froeschke, A. Fujarski, C. Heron, Madden, S. Morehead-Palmer, S. Meyers, and C. Rose. 2012. The ecology and sociology of the mission-aransas estuary: An estuarine and watershed profile.
Canuel, E.A., and A.K. Hardison. 2016. Sources, ages, and alteration of organic matter in estuaries. Annual Review of Marine Science 8: 409–434. https://doi.org/10.1146/annurev-marine-122414-034058.
CBBEP. 2020. Coastal bend bays and estuaries program - coastal bend environmental indicators report. https://www.cbbep.org/manager/wp-content/uploads/CBBEP-2020-Indicator-Report-2.4.21.pdf.
Crocker, P.A., C.R. Arnold, J.A. Deboer, and G.J. Holt. 1983. Blood osmolality shift in juvenile red drum, Sciaenops ocellatus L. exposed to fresh water. Journal of Fish Biology 23: 315–319. https://doi.org/10.1111/j.1095-8649.1983.tb02910.x.
Damare, L.M., K. Nielsen, M.M. Alloy, T.E. Curran, B.K. Soulen, H.P. Forth, C.R. Lay, J.M. Morris, J.A. Stoeckel, and A.P. Roberts. 2018. Photo-induced toxicity in early life stage fiddler crab (Uca longisignalis) following exposure to Deepwater Horizon oil. Ecotoxicology 27: 440–447. https://doi.org/10.1007/s10646-018-1908-6.
Davis, J. T. 1990. Red drum: Biology and life history. Center, S. R. A., Ed. SRAC Publication: Vol. 320.
Dhaneesh, K.V., K.N. Devi, T.A. Kumar, T. Balasubramanian, and K. Tissera. 2012. Breeding, embryonic development and salinity tolerance of Skunk clownfish Amphiprion akallopisos. Journal of King Saud University-Science 24: 201–209.
DOI (Department of the Interior). 2008. 2006 National Survey of Fishing, Hunting, and Wildlife Associated Recreation – Texas. U.S. Department of the Interior, Fish and Wildlife Service, U.S. Department of Commerce, and U.S. Census Bureau. Available at https://www.census.gov/library/publications/2006/demo/fhw-06-nat.html. Accessed Nov 2021.
Engle, V., P. Bourgeois, J. Macauley, and J. Summers. 2000. Ecological condition of estuaries in the Gulf of Mexico. USEPA, https://cfpub.epa.gov/si/si_public_record_report.cfm?dirEntryId=60104&Lab=NHEERL.
Estudillo, C.B., M.N. Duray, E.T. Marasigan, and A.C. Emata. 2000. Salinity tolerance of larvae of the mangrove red snapper (Lutjanus argentimaculatus) during ontogeny. Aquaculture 190: 155–167.
Faulk, C.K., and G.J. Holt. 2006. Responses of cobia Rachycentron canadum larvae to abrupt or gradual changes in salinity. Aquaculture 254 (1–4): 275–283. https://doi.org/10.1016/j.aquaculture.2005.10.046.
Garrido, S., R. Ben-Hamadou, A.M.P. Santos, S. Ferreira, M.A. Teodósio, U. Cotano, X. Irigoien, M.A. Peck, E. Saiz, and P. Ré. 2015. Born small, die young: Intrinsic, size-selective mortality in marine larval fish. Scientific Reports 5: 17065. https://doi.org/10.1038/srep17065.
Gong, X., X. Huang, and W. Wen. 2018. Influence of salinity on the early development and biochemical dynamics of a marine fish, Inimicus japonicus. Journal of Oceanology and Limnology 36: 427–437. https://doi.org/10.1007/s00343-017-6223-1.
Higgs, D., and L. Fuiman. 1996. Ontogeny of visual and mechanosensory structure and function in Atlantic menhaden Brevoortia tyrannus. The Journal of Experimental Biology 199: 2619–2629.
Hill, A.J. 2004. Water permeability and TCDD-induced edema in zebrafish early-life stages. Toxicological Sciences 78: 78–87.
Holt, J., A.G. Johnson, C.R. Arnold, W.A. Fable Jr, and T.D. Williams. 1981. Description of eggs and larvae of laboratory reared red drum, Sciaenops ocellata. Copeia 751–756.
Holt, S.A. 2008. Distribution of red drum spawning sites identified by a towed hydrophone array. Transactions of the American Fisheries Society 137: 551–561. https://doi.org/10.1577/t03-209.1.
Johnson, D.W., K. Grorud-Colvert, S. Sponaugle, and B.X. Semmens. 2014. Phenotypic variation and selective mortality as major drivers of recruitment variability in fishes. Ecology Letters 17: 743–755.
Khursigara, A.J., P. Petrichon, N.M. Bautista, W.W. Burggren, and A.J. Esbaugh. 2017. Cardiac function and survival are affected by crude oil in larval red drum, Sciaenops ocellatus. Science of the Total Environment 579: 797–804. https://doi.org/10.1016/j.scitotenv.2016.11.026.
Lein, I., S. Tveite, B. Gjerde, and I. Holmefjord. 1997. Effects of salinity on yolk sac larvae of Atlantic halibut (Hippoglossus hippoglossus L.). Aquaculture 156: 291–303.
Lellis-Dibble, K.A., K.E. McGlynn, and T.E. Bigford. 2008. Estuarine fish and shellfish species in US commercial and recreational fisheries: Economic value as an incentive to protect and restore estuarine habitat.
Lewis, N.S., D.R. Young, C.L. Folger, and T.H. DeWitt. 2020. Assessing the relative importance of estuarine nursery habitats - A dungeness crab (cancer magister) case study. Estuaries and Coasts: Journal of the Estuarine Research Federation 44: 1062–1073. https://doi.org/10.1007/s12237-020-00821-1.
Magnuson, J.T., A.J. Khursigara, E.B. Allmon, A.J. Esbaugh, and A.P. Roberts. 2018. Effects of Deepwater Horizon crude oil on ocular development in two estuarine fish species, red drum (Sciaenops ocellatus) and sheepshead minnow (Cyprinodon variegatus). Ecotoxicology and Environmental Safety 166: 186–191. https://doi.org/10.1016/j.ecoenv.2018.09.087.
Martin, L., and A.J. Esbaugh. 2021. Osmoregulatory plasticity during hypersaline acclimation in red drum, Sciaenops ocellatus. Journal of Comparative Physiology B: 1–10.
Matlock G. 1990. The life history of red drum.
Mckenzie, D.J., E. Garofalo, M.J. Winter, S. Ceradini, F. Verweij, N. Day, R. Hayes, R. Van Der Oost, P.J. Butler, J.K. Chipman, and E.W. Taylor. 2007. Complex physiological traits as biomarkers of the sub-lethal toxicological effects of pollutant exposure in fishes. Philosophical Transactions of the Royal Society b: Biological Sciences 362: 2043–2059.
Montagna, P.A., X. Hu, T.A. Palmer, and M. Wetz. 2018. Effect of hydrological variability on the biogeochemistry of estuaries across a regional climatic gradient. Limnology and Oceanography 63: 2465–2478.
Montagna, P.A., and R.D. Kalke. 1992. The effect of freshwater inflow on meiofaunal and macrofaunal populations in the Guadalupe and Nueces Estuaries, Texas. Estuaries 15: 307–326. https://doi.org/10.2307/1352779.
Moran, D., R. Softley, and E.J. Warrant. 2015. The energetic cost of vision and the evolution of eyeless Mexican cavefish. Science Advances 1: e1500363. https://doi.org/10.1126/sciadv.1500363.
Nielsen, K., M. Krasnec, J. Magnuson, J. Morris, M. Gielazyn, J. Chavez, and A. Roberts. 2018. Influence of variable ultraviolet radiation and oil exposure duration on survival of red drum (Sciaenops ocellated) larvae. Environmental Toxicology and Chemistry 37: 2372–2379.
Nielsen, K.M., L. DeCamp, M. Birgisson, V.P. Palace, K. Kidd, J. Parrott, ... and E.J. Ussery. 2022. Comparative effects of embryonic metformin exposure on wild, and laboratory-spawned fathead minnow (Pimephales promelas) populations. Environmental Science & Technology. https://doi.org/10.1021/acs.est.2c01079.
Passow, C.N., R. Greenway, L. Arias-Rodriguez, P.D. Jeyasingh, and M. Tobler. 2015. Reduction of energetic demands through modification of body size and routine metabolic rates in extremophile fish. Physiological and Biochemical Zoology 88: 371–383.
Pesce, M., A. Critto, S. Torresan, E. Giubilato, M. Santini, A. Zirino, W. Ouyang, and A. Marcomini. 2018. Modelling climate change impacts on nutrients and primary production in coastal waters. Science of the Total Environment 628–629: 919–937. https://doi.org/10.1016/j.scitotenv.2018.02.131.
Pulich, W., J. Tolan, W. Lee, W. Alvis. 2001. Freshwater inflow recommendation for the Nueces Estuary. Austin, TX. Texas Parks and Wildlife.
Puritz, J.B., M.A. Renshaw, D. Abrego, R.R. Vega, and J.R. Gold. 2014. Reproductive variance of brood dams and sires used in restoration enhancement of spotted seatrout in Texas bays and estuaries. North American Journal of Aquaculture 76 (4): 407–414. https://doi.org/10.1080/15222055.2014.920751.
Quammen, M.L., and C.P. Onuf. 1993. Laguna Madre: Seagrass changes continue decades after salinity Reduction. Estuaries 16: 302. https://doi.org/10.2307/1352503.
Rønnestad, I., M. Yúfera, B. Ueberschär, L. Ribeiro, Ø. Sæle, and C. Boglione. 2013. Feeding behaviour and digestive physiology in larval fish: Current knowledge, and gaps and bottlenecks in research. Reviews in Aquaculture 5: S59–S98.
Swanson, C. 1998. Interactive effects of salinity on metabolic rate, activity, growth and osmoregulation in the euryhaline milkfish (Chanos chanos). Journal of Experimental Biology 201: 3355–3366.
Texas Comptroller. 2018. Gulf Coast Regional Snapshot. comptroller.texas.gov/.
Torres, J.J., R.I. Brightman, J. Donnelly, and J. Harvey. 1996. Energetics of larval red drum, Sciaenops ocellatus. Oxygen consumption, specific dynamic action, and nitrogen excretion. Fishery Bulletin 94: 756–765.
TPWD. 2010. Coastal fisheries: Red drum in Texas. https://tpwd.texas.gov/publications/pwdpubs/media/pwd_br_v3400_045_red_drum.pdf.
TPWD. n.d. - a. Texas bays and estuaries. https://tpwd.texas.gov/landwater/water/habitats/bays/ulm/ulm_expand.phtml: Texas Parks and Wildlife.
TPWD. n.d. - b. Red drum - An angler’s delight. https://tpwd.texas.gov/landwater/water/habitats/bays/ulm/ulm_expand.phtml: Texas Parks and Wildlife.
TPWD. n.d. - c. Texas bays: Upper Laguna Madre. https://tpwd.texas.gov/landwater/water/habitats/bays/ulm/ulm_index.phtml: Texas Parks and Wildlife.
US Census Bureau, Census Bureau Reveals Fastest-Growing Large Cities, Census.gov (2018).
Vega R, Neill W, Gold J, Ray M. 2012. Enhancement of Texas Sciaenids (red drum and spotted seatrout). https://agrilifecdn.tamu.edu/gold/files/2012/05/Bulletin-of-the-National-Research-Institute-of-Aquaculture.pdf.
Vigliola, L., and M.G. Meekan. 2002. Size at hatching and planktonic growth determine post-settlement survivorship of a coral reef fish. Oecologia 131: 89–93.
Wilson, S.S., and K.H. Dunton. 2018. Hypersalinity during regional drought drives mass mortality of the seagrass Syringodium filiforme in a subtropical lagoon. Estuaries and Coasts 41: 855–865. https://doi.org/10.1007/s12237-017-0319-x.
Acknowledgements
The authors would like to thank L. Walsh, R. Lopez, A. Fincannon, D. Gatlin, and R. Chavez for their assistance with red drum eggs. We would also like to thank M. McCaffrey for his assistance with fish care and maintenance.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The Principal Investigator (K. Nielsen) consults for the Port Aransas Conservancy (PAC) on potential impacts of desalination effluent on aquatic life in the Corpus Christi Ship Channel. The author also conducts research in areas of interest similar to the conservation interests held by the PAC. The terms of this arrangement have been reviewed and approved by the University of Texas at Austin in accordance with its policy on objectivity in research. The authors declare no additional potential conflicts of interest.
Additional information
Communicated by Henrique Cabral
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Ackerly, K.L., Roark, K.J. & Nielsen, K.M. Short-term Salinity Stress During Early Development Impacts the Growth and Survival of Red Drum (Sciaenops ocellatus). Estuaries and Coasts 46, 541–550 (2023). https://doi.org/10.1007/s12237-022-01124-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12237-022-01124-3