Abstract
Gas-phase heats of formation of 48 commonly used nitrogen-rich cations in energetic salts composed of H, C, N, and O atoms have been calculated at the Gaussian-3 (G3), Gaussian-4 (G4), and G4 (MP2) theories using the atomization reaction. The discrepancies between the predicted and the reported HOFs vary in the range of 0.28–470.78 kJ/mol. The reported HOFs for twenty cations were observed within 5 kJ/mol deviation, ten cations within 6–10 kJ/mol deviation, eleven cations within 11–50 kJ/mol, five cations within 51–100 kJ/mol, while two cations differed by more than 420 kJ/mol with respect to G4 data. The calculated HOFs with the G4 composite method at 298.15 K were recommended for selected cations as being more reliable than the reported data.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
1 Introduction
In modern civil and military uses, explosives with high performance with lower sensitivity to thermal shock, friction, and electric discharge have been a principal requirement and always desirable. However, the combination of performance and insensitivity is challenging, complicated, and also mutually exclusive because improvement in performance brings upsurge in sensitivity (e.g., in CL–20) [1, 2] and vice versa. One promising approach to overcome this challenge is to explore energetic materials based on ionic salts containing nitrogen-rich compounds with large number of C–N and N–N bonds in the chemical structure [3,4,5]. Ionic salts have been established as a diverse class of materials due to their possible chemical manipulation to achieve desired energetic properties. These salts can be chemically altered with selection of cations, anions, functional groups such as –NO2, –NH2, and –N3 allowing tuning energy content, altering the physical properties, and developing a library of compounds with extreme diversity. Performance parameters (detonation velocity and pressure) are more relying on the heat of formation (HOF) and reflect the energy content of explosive, and the density, which indicates amount of material that can be packed into the charge. Our specific interest is in the design and assessment of new energetic materials for which the HOF plays a crucial role in the preliminary assessment of energy content, stability, reactivity, and potential performance. Considering the hazards of explosives, it is a complicated and unsafe task to measure HOF experimentally, and hence, theoretical methods have received considerable attention. To obtain HOF experimentally, differential scanning calorimetry is commonly employed [6]; however, these calorimetric measurements are resource consuming and difficult due to unknown intermediates and hazardous nature of explosives. Several methods have been proposed for neutral energetic molecules including group additive contributions, isodesmic reactions, atomic or group corrections to Hartree–Fock molecular energies, bond additivity corrections, and composite methods [7,8,9,10,11,12,13,14,15,16,17,18,19,20,21,22,23,24,25]. For judging the energy content of the ionic salts, it is most significant to know the HOFs of anion and cation in gas phase. Despite the extensive studies on ionic salts, available thermochemical data for cations are frequently scarce and reveal significant inconsistency among the reported results. In case of ionic compounds, Born–Haber energy cycle (Fig. 1) has been used to predict the HOF of salts [26,27,28] and is given by the formula:
where ∆HL is the lattice energy of the salt. The HOFs of cations and anions are mostly predicted with isodesmic reactions. Most of the reported salts in the literature [29,30,31,32,33,34,35,36,37,38,39,40,41,42,43,44,45,46] show the variation in anion, while cations are commonly the nitrogen-rich compounds. To investigate the HOF of ionic salt, knowledge of the reliable HOF of cation and anion is an essential requirement. In recent years, there have been several accurate first-principle techniques proposed to evaluate the thermochemistry of molecules. In particular, G3, G4, and G4 (MP2) theories [47,48,49,50,51,52,53,54,55,56,57,58,59,60,61,62,63,64,65] are popular composite theoretical techniques which have been successfully applied and established as an accurate method to calculate the HOF of a wide variety of molecules. Experimental data of HOFs for cations are rare, and reliable values are required for quantitative interpretation of performance and energetic nature of material. In this work, we report the calculation of HOFs for commonly used cations in the formation of ionic salts by G3, G4, and G4 (MP2) methods. Curtiss et al. [60, 61] reported the HOFs for different classes of compounds using G3, G4, and G4 (MP2) methods where G4 theory shows minimum deviation from experiment values. The calculated HOFs of cation were compared with that of reported results and recommend in thermochemical models to compute HOF of ionic salts precisely.
2 Results and discussion
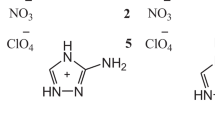
A set of 48 commonly used CHNO cations belonging to a different nitrogen-containing molecules (with –NO2, –NH2, –CH3, –N3, –C(O)NH2, –CN, –OH, and five-membered heterocycles) were taken from the literature [29,30,31,32,33,34,35,36,37,38,39,40,41,42,43,44,45,46] and studied in this work. All ab initio and density functional theory (DFT) computations were performed via the Gaussian 09 package [66]. The structures of selected cations were optimized at the B3PW91/6-31G(d,p) level, and the resulting structures were used as inputs for further G3, G4, and G4 (MP2) calculations. The G3, G4, and G4 (MP2) HOFs at 298.15 K were calculated using the atomization reaction procedures, which includes experimental HOFs of gaseous atoms at 0 K and thermal corrections for elements in their standard states [50]. The optimized Cartesian coordinates of the cations at G3, G4, and G4 (MP2) calculations are summarized in Table S5-148 of Supporting Information.
The computed HOFs for cations using G3, G4, and G4 (MP2) methods are compared in Table 1 together with the reported values from the literature. To the best of our knowledge, HOFs from quantum-chemical calculations for these nitrogen-rich cations are not available in the literature. Figure 2 shows Bland–Altman plot of the calculated HOFs using G4 method and reported data. Figures S1 and S2 in Supporting Information represent the Bland–Altman plots using G3 and G4 (MP2) methods, respectively. 2,2′-Carbonyldihydrazinium and 2,2-dimethyltriazan-2-ium appear as an outlier for which all three methods consistently calculate lower values of the HOF at 298.15 K. In general, the agreement between the calculated HOFs using G3, G4, and G4 (MP2) methods is extremely good. In Table 1, the HOFs computed from G3, G4, and G4 (MP2) atomization reaction are compared and reveal that the HOFs obtained from G3 and G4 (MP2) methods are always overestimated than that of the G4 method. Inspection of Table 1 shows that G3 values differ by 0.84–15.09 kJ/mol and G4 (MP2) values vary from 2.05 to 12.97 kJ/mol relative to G4 HOFs. Overall, the close agreement can be seen in HOFs calculated using G3 and G4 (MP2) methods with the results of G4 level and provides support to assume that G4 method with the atomization reaction will be capable of calculating the accurate HOFs for cations.
The G4 HOFs derived in this work for C+ and N+ containing cations (1–2, 3–5, 6–7, and 8–9) show similar results, while HOFs for cations 10–11, 12–13, and 14–15 differ from each other by 2–37 kJ/mol. As shown in Table 1, the HOFs obtained using G4 method for twenty cations (1–7, 10, 20, 26, 27, 29, 30, 35, 38–41, 46, and 47) show variation within 0.4–5 kJ/mol and close agreement with reported values, while the reported values show 6–10 kJ/mol deviation for ten cations and 11–50 kJ/mol difference for eleven cations concerning to those calculated with the G4 method. The uncertainty in reported values is greater than 50 kJ/mol for seven cations. The HOF obtained for cation 14 and 19 in the present work shows deviation of 55–68 kJ/mol compared to the values reported by Klapotke et al. [30].
The HOFs for cations 12, 14, and 15 predicted in the present work are higher (by 52–66 kJ/mol) than the values of Shreeve et al. [32,33,34]. Similarly, Huang et al. [37] reported HOF for cation 36 to be 73 kJ/mol higher than the G4 value. The HOFs of cation 16 (1721 kJ/mol) and 22 (775 kJ/mol) obtained by Shreeve et al. [39, 40] are substantially higher (> 420 kJ/mol) than calculated in this work. The reason for this considerable disagreement is not quite clear. In summary, the values of HOF obtained by G3, G4, and G4 (MP2) theories with atomization reaction method are very close. Therefore, the obtained G4 HOFs for cations for which no experimental data are available may be used to calculate the energy content of energetic ionic salts and it is hoped that our computed HOFs will serve as reliable estimates.
3 Conclusions
Gas-phase HOFs for a set of widely used nitrogen-rich cations were calculated with a number of methods; G4 performs best against experiment and recommended for calculations. Taking into account the available inconsistent HOF values of cations, significant errors can occur in the HOF calculations for energetic salts. The HOFs computed in the present work will help to avoid significant errors in the calculation of energy content and precisely compute HOF-dependent performance parameters. The major differences between the HOF values for fourteen cations calculated by G4 method and reported results are above 15 kJ/mol. The results of the present calculations is supported by G3 and G4 (MP2) calculations, which shows that the predicted values are consistent and can give reasonable estimations.
4 Supporting information
Deviation in predicted HOFs with G3 and G4 (MP2) methods and reported data from G4 values for selected cations is listed in Table S1. Tables S2 to S4 contain the calculated energies at 0 and 298.15 K for selected cations at G3, G4, and G4 (MP2) level, respectively. Cartesian coordinates (Å) of the cations studied in the present work at the G3, G4, and G4 (MP2) level are summarized in Table S5-S148. Table S149 lists the experimental HOFs (in kcal/mol) and enthalpy corrections of the atomic elements. Total G3, G4, and G4 (MP2) energies (in hartrees) of atomic species are summarized in Table S150-152. Figures S1 and S2 show the Bland–Altman plots between calculated HOFs using G3 and G4 (MP2) methods and reported data.
References
Badgujar DM, Talawar MB, Asthana SN, Mahulikar PP (2008) Advances in science and technology of modern energetic materials: an overview. J Hazard Mater 151:289–305
Ghule VD, Jadhav PM, Patil RS, Radhakrishnan S, Soman T (2010) Quantum-chemical studies on hexaazaisowurtzitanes. J Phys Chem A 114:498–503
Gao H, Shreeve JM (2011) Azole-based energetic salts. Chem Rev 111:7377–7436
Singh RP, Verma RD, Meshri DT, Shreeve JM (2006) Energetic nitrogen-rich salts and ionic liquids. Angew Chem Int Ed 45:3584–3601
Klapötke TM (2015) Chemistry of high-energy materials, 3rd edn. Walter de Gruyter GmbH, Berlin
Hu A, Larade B, Dudiy S, Abou-Rachid H, Lussier LS, Guo H (2007) Theoretical prediction of heats of sublimation of energetic materials using pseudo-atomic orbital density functional theory calculations. Propellants Explos Pyrotech 32:331–337
Vatani A, Mehrpooya M, Gharagheizi F (2007) Prediction of standard enthalpy of formation by a QSPR model. Int J Mol Sci 8:407–432
Albahri TA, Aljasmi AF (2013) SGC method for predicting the standard enthalpy of formation of pure compounds from their molecular structures. Thermochim Acta 568:46–60
Borhani TN, Bagheri M, Manan ZA (2013) Molecular modeling of the ideal gas enthalpy of formation of hydrocarbons. Fluid Phase Equilib 360:423–434
Cao CT, Yuan H, Cao C (2015) New concept of organic homo-rank compounds and its application in estimating enthalpy of formation of mono-substituted alkanes. J Phys Org Chem 28:266–280
Allinger NL, Schmitz LR, Motoc I, Bender C, Labanowski JK (1992) Heats of formation of organic molecules. 2. The basis for calculations using either ab initio or molecular mechanics methods. Alcohols and ethers. J Am Chem Soc 114:2880–2883
Langley CH, Lii JH, Allinger NL (2001) Molecular mechanics calculations on carbonyl compounds. IV. Heats of formation. J Comput Chem 22:1476–1483
Lii JH, Liao FX, Hu CH (2011) Accurate prediction of the enthalpies of formation for xanthophylls. J Comput Chem 32:3175–3187
Wu YY, Zhao FQ, Ju XH (2014) A comparison of the accuracy of semi-empirical pm3, pddg and pm6 methods in predicting heats of formation for organic compounds. J Mex Chem Soc 58:223–229
Byrd EFC, Rice BM (2006) Improved prediction of heats of formation of energetic materials using quantum mechanical calculations. J Phys Chem A 110:1005–1013
Byrd EFC, Rice BM (2009) Improved prediction of heats of formation of energetic materials using quantum mechanical calculations. J Phys Chem A 113:5813
Rousseau E, Mathieu D (2000) Atom equivalents for converting DFT energies calculated on molecular mechanics structures to formation enthalpies. J Comput Chem 21:367–379
Wu J, Xu X (2007) The X1 method for accurate and efficient prediction of heats of formation. J Chem Phys 127:214105
Ohlinger WS, Klunzinger PE, Deppmeier BJ, Hehre WJ (2009) Efficient calculation of heats of formation. J Phys Chem A 113:2165–2175
Mathieu D (2018) Atom pair contribution method: fast and general procedure to predict molecular formation enthalpies. J Chem Inf Model 58:12–26
Keshavarz MH (2011) Prediction of the condensed phase heat of formation of energetic compounds. J Hazard Mater 190:330–344
Jafari M, Keshavarz MH, Noorbala MR, Kamalvand M (2016) A reliable method for prediction of the condensed phase enthalpy of formation of high nitrogen content materials through their gas phase information. Chem Sel 1:5286–5296
Keshavarz MH, Tehrani MK (2007) A new method for determining gas phase heat of formation of aromatic energetic compounds. Propellants Explos Pyrotech 32:155–159
Politzer P, Murray JS, Seminario JM, Lane P, Grice ME, Concha MC (2001) Computational characterization of energetic materials. J Mol Struct THEOCHEM 573:1–10
Politzer P, Lane P, Concha MC (2004) Computational determination of nitroaromatic solid phase heats of formation. Struct Chem 15:469–478
Jenkins HDB, Roobottom HK, Passmore J, Glasser L (1999) Relationships among ionic lattice energies, molecular (formula unit) volumes, and thermochemical radii. Inorg Chem 38:3609–3620
Jenkins HDB, Tudela D, Glasser L (2002) Lattice potential energy estimation for complex ionic salts from density measurements. Inorg Chem 41:2364–2367
Jenkins HDB (2005) Thermodynamics of the relationship between lattice energy and lattice enthalpy. J Chem Educ 82:950
Gao H, Ye C, Piekarski CM, Shreeve JM (2007) Computational characterization of energetic salts. J Phys Chem C 111:10718–10731
Fischer N, Klapötke TM, Stierstorfer J (2011) Energetic nitrogen-rich salts of 1-(2-hydroxyethyl)-5-nitriminotetrazole. Eur J Inorg Chem 2011:4471–4480
Gao H, Ye C, Gupta OD, Xiao JC, Hiskey MA, Twamley B, Shreeve JM (2007) 2,4,5-Trinitroimidazole-based energetic salts. Chem Eur J 13:3853–3860
Gao H, Zeng Z, Twamley B, Shreeve JM (2008) Polycyano-anion-based energetic salts. Chem Eur J 14:1282–1290
Zhang Y, Parrish DA, Shreeve JM (2012) 4-Nitramino-3,5-dinitropyrazole-based energetic salts. Chem Eur J 18:987–994
Huang Y, Gao H, Twamley B, Shreeve JM (2008) Nitroamino triazoles: nitrogen-rich precursors of stable energetic salts. Eur J Inorg Chem 2008:2560–2568
Wang R, Guo Y, Zeng Z, Twamley B, Shreeve JM (2009) Furazan-functionalized tetrazolate-based salts: a new family of insensitive energetic materials. Chem Eur J 15:2625–2634
Ye C, Shreeve JM (2007) Rapid and accurate estimation of densities of room-temperature ionic liquids and salts. J Phys Chem A 111:1456–1461
Huang H, Zhou Z, Song J, Liang L, Wang K, Cao D, Sun W, Dong X, Xue M (2011) Energetic salts based on dipicrylamine and its amino derivative. Chem Eur J 17:13593–13602
Thottempudi V, Shreeve JM (2011) Synthesis and promising properties of a new family of high-density energetic salts of 5-nitro-3-trinitromethyl-1 H-1,2,4-triazole and 5,5′-bis(trinitromethyl)-3,3′-azo-1 H-1,2,4-triazole. J Am Chem Soc 133:19982–19992
Joo YH, Shreeve JM (2009) Energetic ethylene-and propylene-bridged bis (nitroiminotetrazolate) salts. Chem Eur J 15:3198–3203
Gao H, Joo YH, Twamley B, Zhou Z, Shreeve JM (2009) Hypergolic ionic liquids with the 2,2-dialkyltriazanium cation. Angew Chem Int Ed 48:2792–2795
Sabaté CM, Jeanneau E, Delalu H (2012) Energetic ionic compounds based on the 2-tetrazenium cation. Eur J Inorg Chem 2012:2418–2428
Wang K, Parrish DA, Shreeve JM (2011) 3-Azido-N-nitro-1H-1,2,4-triazol-5-amine-based energetic salts. Chem Eur J 17:14485–14492
Wang R, Gao H, Ye C, Twamley B, Shreeve JM (2007) Heterocyclic-based nitrodicyanomethanide and dinitrocyanomethanide salts: a family of new energetic ionic liquids. Inorg Chem 46:932–938
Klapötke TM, Stierstorfer J (2008) The new energetic compounds 1,5-diaminotetrazolium and 5-amino-1-methyltetrazolium dinitramide: synthesis, characterization and testing. Eur J Inorg Chem 2008:4055–4062
Zhang Y, Guo Y, Joo YH, Parrish DA, Shreeve JM (2010) 3,4,5-Trinitropyrazole-based energetic salts. Chem Eur J 16:10778–10784
Klapötke TM, Piercey DG, Stierstorfer J (2012) The 1,4,5-Triaminotetrazolium cation (CN7H6 +): a highly nitrogen-rich moiety. Eur J Inorg Chem 2012:5694–5700
Suntsova MA, Dorofeeva OV (2014) Use of G4 theory for the assessment of inaccuracies in experimental enthalpies of formation of aliphatic nitro compounds and nitramines. J Chem Eng Data 59:2813–2826
Dorofeeva OV, Ryzhova ON, Suchkova TA (2017) Enthalpies of formation of hydrazine and its derivatives. J Phys Chem A 121:5361–5370
Suntsova MA, Dorofeeva OV (2016) Use of G4 theory for the assessment of inaccuracies in experimental enthalpies of formation of aromatic nitro compounds. J Chem Eng Data 61:313–329
Dorofeeva OV, Kolesnikova IN, Marochkin II, Ryzhova ON (2011) Assessment of Gaussian-4 theory for the computation of enthalpies of formation of large organic molecules. Struct Chem 22:1303–1314
Simmie JM (2015) A database of formation enthalpies of nitrogen species by compound methods (CBS-QB3, CBS-APNO, G3, G4). J Phys Chem A 119:10511–10526
Elioff MS, Hoy J, Bumpus JA (2016) Calculating heat of formation values of energetic compounds: a comparative study. Adv Phys Chem 2016:1–11
Dorofeeva OV, Ryzhova ON, Sinditskii VP (2015) Enthalpy of formation of guanidine and its amino and nitro derivatives. Struct Chem 26:1629–1640
Curtiss LA, Raghavachari K, Redfern PC, Rassolov V, Pople JA (1998) Gaussian-3 (G3) theory for molecules containing first and second-row atoms. J Chem Phys 109:7764
Curtiss LA, Raghavachari K, Redfern PC, Pople JA (2000) Assessment of Gaussian-3 and density functional theories for a larger experimental test set. J Chem Phys 112:7374–7383
Curtiss LA, Redfern PC, Raghavachari K (2005) Assessment of Gaussian-3 and density-functional theories on the G3/05 test set of experimental energies. J Chem Phys 123:124107
Curtiss LA, Redfern PC, Raghavachari K (2007) Gaussian-4 theory. J Chem Phys 126:084108
Rayne S, Forest K (2010) Estimated gas-phase standard state enthalpies of formation for organic compounds using the Gaussian-4 (G4) and W1BD theoretical methods. J Chem Eng Data 55:5359–5364
Rayne S, Forest K (2010) Performance of Gaussian-3 and Gaussian-4 level theoretical methods in estimating gas phase enthalpies of formation for representative and chlorofluorocarbons and hydrochlorofluorocarbons. J Mol Struct THEOCHEM 953:47–48
Curtiss LA, Redfern PC, Raghavachari K (2007) Gaussian-4 theory using reduced order perturbation theory. J Chem Phys 127:124105
Curtiss LA, Redfern PC, Raghavachari K (2011) Gn theory. WIRE Comput Mol Sci 1:810–825
Asatryan R, Bozzelli JW, Simmie JM (2008) Thermochemistry of methyl and ethyl nitro, RNO2, and nitrite, RONO, organic compounds. J Phys Chem A 112:3172–3185
Simmie JM, Somers KP (2015) Benchmarking compound methods (CBS-QB3, CBS-APNO, G3, G4, W1BD) against the active thermochemical tables: a litmus test for cost-effective molecular formation enthalpies. J Phys Chem A 119:7235–7246
Somers KP, Simmie JM (2015) Benchmarking compound methods (CBS-QB3, CBS-APNO, G3, G4, W1BD) against the active thermochemical tables: formation enthalpies of radicals. J Phys Chem A 119:8922–8933
Simmie JM, Sheahan JN (2016) Validation of a database of formation enthalpies and of mid-level model chemistries. J Phys Chem A 120:7370–7384
Frisch MJ, Trucks GW, Schlegel HB, Scuseria GE, Robb MA, Cheeseman JR, Scalmani G, Barone V, Mennucci B, Petersson GA, Nakatsuji H, Caricato M, Li X, Hratchian HP, Izmaylov AF, Bloino J, Zheng G, Sonnenberg JL, Hada M, Ehara M, Toyota K, Fukuda R, Hasegawa J, Ishida M, Nakajima T, Honda Y, Kitao O, Nakai H, Vreven T, Montgomery Jr JA, Peralta JE, Ogliaro F, Bearpark M, Heyd JJ, Brothers E, Kudin KN, Staroverov VN, Keith T, Kobayashi R, Normand J, Raghavachari K, Rendell A, Kurant JC, Iyengar SS, Tomasi J, Cossi M, Rega N, Millam JM, Klene M, Knox JE, Cross JB, Bakken V, Adamo C, Jaramillo J, Gomperts R, Stratmann RE, Yazyev O, Austin AJ, Cammi R, Pomelli C, Ochterski JW, Martin RL, Morokuma K, Zakrzewski VG, Voth GA, Salwador P, Dannenberg JJ, Dapprich S, Daniels AD, Farkas O, Foresman JB, Ortiz JV, Cioslowski J, Fox DJ (2013) Gaussian 09, revision E.01. Gaussian Inc, Wallingford
Czakό G, Mátyus E, Simmonett AC, Császár AG, Schaefer HF III, Allen WD (2008) Anchoring the absolute proton affinity scale. J Chem Theory Comput 4:1220–1229
Acknowledgements
This work is supported by the grant from the DST-SERB, Government of India (Young Scientists, No. SB/FT/CS-110/2014).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Nirwan, A., Ghule, V.D. Estimation of heats of formation for nitrogen-rich cations using G3, G4, and G4 (MP2) theoretical methods. Theor Chem Acc 137, 115 (2018). https://doi.org/10.1007/s00214-018-2300-6
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00214-018-2300-6