Abstract
Purpose
This randomized controlled trial compared changes in bone mineral density (BMD) and bone turnover in postmenopausal women with low bone mass randomized to 12 months of either risedronate, exercise, or a control group.
Methods
Two hundred seventy-six women with low bone mass, within 6 years of menopause, were included in analysis. Treatment groups were 12 months of (a) calcium and vitamin D supplements (CaD) (control), (b) risedronate + CaD (risedronate), or (c) bone-loading exercises + CaD (exercise). BMD and serum markers for bone formation (Alkphase B) and resorption (Serum Ntx) were analyzed at baseline, 6, and 12 months.
Results
Using hierarchical linear modeling, a group by time interaction was found for BMD at the spine, indicating a greater improvement in the risedronate group compared to exercise (p ≤ .010) or control groups (p ≤ .001). At 12 months, for women prescribed risedronate, changes in BMD at the spine, hip, and femoral neck from baseline were + 1.9%, + 0.9%, and + .09%; in exercise group women, + 0.2%, + 0.5%, and − 0.4%; and in control group women, − 0.7%, + 0.5%, and − 0.5%. There were also significant differences in reductions in Alkphase B (RvsE, p < .001, RvsC, p < .001) and Serum Ntx (RvsE, p = .004, RvsC, p = .007) in risedronate women compared to exercise and control groups. For risedronate, 12-month changes in Alkphase B and Serum Ntx were − 20.3% and − 19.0%; for exercise, − 6.7% and − 7.0%; and for control, − 6.3% and − 9.0%.
Conclusion
Postmenopausal women with low bone mass should obtain adequate calcium and vitamin D and participate in bone-loading exercises. Additional use of BPs will increase BMD, especially at the spine.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Osteoporosis, characterized by bone loss (low bone mineral density [BMD], alterations in bone structure) and resultant fractures, is a major health problem of crisis proportions in postmenopausal women [1,2,3]. Postmenopausal women with fractures experience a decrease in quality of life and increased financial burden due to cost of care, and after an osteoporotic hip fracture, the 1-year mortality rate is approximately 21% [4, 5]. According to the National Osteoporosis Foundation (NOF), 43 million postmenopausal women in the USA have low bone mass (T-score − 1 to − 2.49) and are at risk for osteoporosis (< − 2.5) [1, 4]. Preserving and improving BMD in these patients are critical. Compared to patients with low bone mass, patients with osteoporosis have a twofold higher risk of developing fractures [6].
Calcium and vitamin D supplementation, lifestyle modifications such as smoking cessation, prevention of falls, and exercises are recommended for all women with both low bone mass and osteoporosis [3, 7]. Bisphosphonates (BPs) such as risedronate have been found to be effective in improving BMD and preventing fractures and are considered first line-treatment for women with osteoporosis [7]. Women with low bone mass can also be prescribed BPs [7, 8]. However, prescribing BPs for women with low bone mass who do not yet have osteoporosis has been controversial. This controversy is related to the rare incidences of adverse effects (AEs) with long-term use of BPs, such as development of atypical femoral fractures [9, 10].
Participating in weight-bearing plus resistance training (bone-loading) exercises has also been shown to improve BMD and prevent fractures in postmenopausal women at risk for bone loss [11,12,13,14]. Results of this present study can inform decision-making as to whether postmenopausal women with low bone mass can be prescribed bone-loading exercises in place of medication. This study compared changes in BMD and biomarkers of bone turnover in postmenopausal women with low bone mass randomized to either 12 months of the BP “risedronate,” bone-loading exercises, or a control group of women obtaining calcium and vitamin D supplements only. The following are study aims:
-
Aim 1: To compare changes in BMD at the total hip, femoral neck, and spine (dual-energy X-ray absorptiometry [DXA]) in control, risedronate, and exercise group participants after 6 and 12 months of treatment.
-
Aim 2: To compare changes in biomarkers of bone formation and resorption in control, risedronate, and exercise group participants after 6 and 12 months of treatment (formation: Alkphase B, resorption: Serum Ntx).
-
Aim 3: To describe women’s reports of adverse effects (AEs) to treatments as well as 12-month adherence to prescriptions for bone-loading exercises and risedronate.
-
Research question related to Aim 3: Were there relationships between adherence rates to exercises or to risedronate and changes in BMD and serum biomarkers of bone turnover after 12 months of the treatment?
Background and significance
While women are encouraged to engage in exercise to improve bone health, clinicians are reluctant to prescribe the more intensive bone-loading exercises due to concerns about safety with exercises, women’s adherence to exercises, and the effectiveness of the exercises [14].
The LIFTMOR (Lifting Intervention for Training Muscle and Osteoporosis Rehabilitation) clinical trial was one of the first studies to address these clinicians’ concerns [14]. Results of this study were that 8 months of 30-min, twice-weekly, supervised high-intensity resistance and impact training were safe for postmenopausal women with low bone mass, were effective in improving BMD, and most women were adherent to the program. In the LIFTMOR study, after 8 months of exercise, BMD at the lumbar spine increased by 2.9% (SD = 3.1, p < 0.001) and BMD at the femoral neck increased by 0.3% (SD = 3.0, p = 0.025) [14].
Our study builds on the LIFTMOR clinical trial and adds further data to inform whether postmenopausal women with low bone mass can effectively maintain or even improve BMD with bone-loading exercises prior to prescriptions for medication. Our long-term goal is to contribute to the development of clinical practice guidelines for the prevention of fractures in postmenopausal women with low bone mass.
Clinical management pathways for women who are at risk for other chronic illnesses such as hypertension or diabetes include trials of lifestyle modifications prior to prescriptions for medications. Thus, due to the potential for adverse effects with long-term use of BPs, the significant consequences of progression to osteoporosis, and the potential bone strength and fracture reduction benefits of exercise, we proposed conduct of our study to determine if, in women with low bone mass, prescriptions for exercises are warranted prior to prescriptions for medications. If an effective exercise program could substitute for or delay the use of BPs such as risedronate in post-menopausal women with low bone mass, not only would bone health improve throughout the lifespan, but women would also benefit from the many other positive effects of exercise.
Methods
Approvals
Data for this study were collected in a larger study entitled: Bone Loading Exercises versus Risedronate on Bone Health in Post-menopausal women supported by the National Institute of Nursing Research under award number R01NR015029. The study was approved by the Institutional Review Boards at both the University of Nebraska and Creighton University Medical Centers, as both were involved in implementation of the study. Written consent for participation and publication of data were obtained from participants at the time of enrollment. Participants received informed consent documents for review prior to their enrollment visit. Careful and thorough explanations were utilized in obtainment of consent to ensure participant comprehension. The R01 study is registered as a clinical trial under the title: Heartland Osteoporosis Prevention Study (see https://clinicaltrials.gov).
Design
We used a prospective stratified (by exercise history), randomized, 3-group repeated measures experimental design with three major data collection points (baseline, 6, and 12 months).
Sample
The power analysis for sample size was based on a small and conservative effect size of f = 0.15 and conducted using G*Power 3 [15]. For a repeated measures analysis of variance, a significance level of 0.05 was assumed for the three time points, with an estimate of 0.5 for the correlation between time points. We believed a sample of 246 participants across three groups would provide sufficient evidence to detect the effects of interest in study calculations. To account for up to 20% attrition, the optimal enrollment goal was 302 participants. We enrolled 276 participants in our study. Data from all 276 women were included in this intent-to-treat analysis.
Women were included in the study if they had low bone mass (T-score of − 1.0 to − 2.49), were in their first 6 years after a diagnosis of menopause and had their health care provider’s approval to be in the study. Menopause was diagnosed as women having no menstrual period for the last 12 months; time of menopause was determined as 12 months after the date when women reported their last menstrual period [16].
Women were excluded from the study if they had a diagnosis of osteoporosis (T-score < − 2.5); had an increased risk of a major or hip fracture; had been on BPs within the last 6 months; were currently on estrogen, tamoxifen, or aromatase inhibitors; had a serum vitamin D level < 10 mg/ml or > 100 mg/ml; had any conditions that prohibited prescriptions for calcium and vitamin D supplements, risedronate, or exercise; or weighed more than 300 lbs. An increased risk of a major or hip fracture was defined as a Fracture Risk Assessment (FRAX) score indicating a 10-year hip fracture probability ≥ 3% or a 10-year probability of a major osteoporotic fracture ≥ 20%. FRAX scores are determined by factors such as age, sex, weight, height, previous fracture, a parent with a fractured hip, currently smoking, on glucocorticoids, alcohol abuse, presence of medical conditions related to secondary osteoporosis, and femoral neck BMD [17, 18]. Women over 300 lbs. were excluded because results of DXA testing with Hologic machines for these women would not be accurate.
Women of all races and ethnicities were encouraged to participate in this study, and participants were recruited through practitioner referral, community presentations, distribution of flyers, postcard mailings, and generation of a website and Facebook page [19].
Randomization
This study used a statistician-generated procedure for randomization to control, risedronate, or exercise groups, and stratification was by exercise history. Prior to randomization, participants reported their usual levels of exercise (≥ 2 high intensity exercise sessions per week; < 2 sessions per week) to ensure equal distribution among groups. Both participants and team members were blinded to group assignment at the time of subject enrollment. After enrollment, participants and research team members learned group assignment by opening a sealed, opaque envelope.
Setting
All BMD testing, blood draws, and participant interviews were completed at the Creighton University Osteoporosis Research Center (CORC) in Omaha, NE. Participants randomized to the exercise group completed the exercise program at community fitness centers in Omaha and Lincoln, NE.
Study treatments
Calcium/vitamin D
The National Osteoporosis Foundation (NOF) recommends that women over age 50 consume 1200 mg of calcium and 800–1000 IU of vitamin D3 per day—either from their diet or from supplements [1]. In our study, dietary intakes of calcium at baseline were calculated for all women using the National Osteoporosis Foundation (NOF) Calcium Intake Estimate. Based on dietary intakes, calcium citrate supplements were prescribed to ensure subject’s total daily intake (dietary and supplemental) was ~ 1200 mg [1]. Participants were prescribed doses of vitamin D based on their serum 25(OH) D levels at baseline, with the goal of women reaching and maintaining a serum level of at least 30 ng/ml [1]. Participants who had serum D levels at 30 ng/ml or greater were prescribed 1000 IU daily, levels of 20–29 ng/ml were prescribed 2000 IU, and levels of 10–19 ng/ml were prescribed 3000 IU. Serum D levels were rechecked at 6 months, and prescriptions were revised as needed.
Risedronate
Risedronate sodium is a pyridinyl bisphosphonate which inhibits osteoclast-mediated bone resorption, thus indirectly increasing BMD [7]. Participants randomized to the risedronate group took the prescribed calcium and vitamin D supplements daily and a 150 mg tablet of oral risedronate once every 4 weeks for 12 months.
Bone-loading exercise program
Bone-loading exercises were 3-times weekly and included jogging using a weighted vest and resistance exercises of the major muscle groups under the direction of the study exercise training and designated exercise trainers at fitness centers (on-site ETs). One Repetitive Maximum (1RM) estimation tests were conducted at baseline and 6 months, and initial weight settings and reps and sets for women were determined by the 1RM or volitional fatigue at 8–12 repetitions. On-site ETs met with women every two weeks during the 12-month study to discuss expected weight loads, reps, and set progressions. Participants were not supervised during sessions; however, there were always ETs available in the fitness centers to respond to questions and concerns. A more detailed exercise protocol and progression program is described in Bilek (2016) [20].
Steps to promote intervention fidelity
Three research team members in addition to principal investigators conducted all procedures for obtaining informed consent and administering interventions. One research exercise trainer had overall responsibility for the exercise intervention and for supervision of the on-site exercise trainers at the five fitness centers—two in Lincoln and three in Omaha. Training sessions were held with research team members and on-site exercise trainers prior to implementing the study to standardize administration of interventions across sites.
Study outcome measures: BMD, serum biomarkers of bone turnover, adherence rates
Outcomes for the study were bone structure measures-peripheral quantitative computed tomography (pQCT) at the tibia and Hip Structural Analysis (HSA); BMD at the total hip, femoral neck, and spine (Hologic QDR2000™ DXA); and serum biomarkers of bone formation (Alkphase B) and resorption (Serum Ntx). In addition, adherence rates and adverse effects were examined for women in all three treatment groups. This report focuses only on outcomes for BMD and biomarkers of bone turnover. Changes in bone structure in groups will be described in a second article soon to be published.
For measuring BMD using DXA scans, patients are irradiated at specific sites (most often the hip and spine) with an X-ray beam of two different energies, enabling bone attenuation to be separated from soft tissue attenuation [21]. In our study, all participants used the same machine throughout, tests were performed by technicians certified in densitometry testing, and all results were interpreted by one research team radiologist.
Rates of bone resorption and formation were measured with Serum Ntx and Alkphase B. Serum was drawn by team members certified in phlebotomy training and/or who had many years of experience with phlebotomy, and serum tests were analyzed by technicians with experience in serum biomarker testing and with ELISA.
Adherence to exercise was defined as the number of sessions women attended at the fitness center/the number of sessions prescribed. Attendance at sessions and AEs with exercises were documented by women in their exercise log and by on-site ETs in their bimonthly documentation forms. Adherence to calcium, vitamin D, and risedronate were measured by pill counts. Women reported AEs with risedronate and supplements by completing investigator-developed forms at 3, 6, and 12 months, or by personal contact with research team members [20].
Study variables, instruments, outcome measures, and data collection time points are included in Table 1.
Data analysis
All statistical analyses were performed using IBM SPSS Version 25. Data were double-entered and inspected for artifacts, missing or out of range values and normality. Consistent with intent to treat analysis, data from all 276 participants were included in this hierarchical linear modeling analysis. All available data were used and there was no data imputation [15]. The primary outcomes of BMD, Alkphase B, and Serum Ntx were compared between groups and over time. A significant time-group interaction would indicate that the three groups being compared had significantly different outcomes at 12 months [15]. Relationships between adherence rates to risedronate and exercises and changes in BMD at 12 months were analyzed using correlations, and AEs associated with risedronate and exercises were examined with descriptive statistics.
Results
Study sample
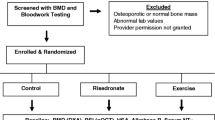
Figure 1 is a CONSORT diagram depicting the flow of participants’ progress through the study. Demographic variables and BMD T-scores at baseline for women randomized to the three groups are described in Table 2. Baseline variables for the three groups of women were compared using analysis of variance and chi-square tests, and there were no significant differences in variables based on group.
Although data from all 276 women were used in this intent-to-treat analysis, 49 of the 276 women withdrew during the study, and only 227 women completed 12-month testing (see Fig. 1). Fourteen of the women were involuntarily withdrawn because they no longer met inclusion criteria for the study (e.g., they were diagnosed with osteoporosis at six months), and 35 women voluntarily withdrew from the study for reasons such as “they experienced AEs from treatment, or they believed they had no time to exercise.” Compared to the 49 women who withdrew, the 227 women retained in the study were older (p = 0.032); were married (p = 0.026); and had a greater increase in BMD at the spine at 6 months (p = 0.02).
All other results were organized to respond to the aims of the study.
-
Aim 1: To compare changes in BMD at the total hip, femoral neck, and spine (dual-energy X-ray absorptiometry [DXA]) in control, risedronate, and exercise group participants after 6 and 12 months of treatment.
BMD at the spine significantly improved (p < 0.001) over time (baseline to 12 months) in the risedronate group and did not change in control or exercise groups. Based on hierarchical linear modeling analysis, a group by time interaction was found for BMD at the spine, indicating a greater improvement at the spine in the risedronate group compared to exercise (p ≤ 0.010) or control groups (p ≤ 0.001). There were no significant differences in changes in BMD based on assignment to exercise or control groups. At 12 months, for women prescribed risedronate, the changes in BMD at the spine, hip, and femoral neck from baseline were + 1.9%, + 0.9%, and + 0.09%; in exercise group women, + 0.2%, + 0.5%, and − 0.4%; and in control group women, − 0.7%, + 0.5%, and − 0.5% (see Table 3).
-
Aim 2: To compare changes in biomarkers of bone formation and resorption in control, risedronate, and exercise group participants after 6 and 12 months of treatment (formation: Alkphase B, resorption: Serum Ntx).
Using hierarchical linear modeling, there were significant differences in reductions in Alkphase B (RvsE, p < 0.001, RvsC, p < 0.001) and Serum Ntx (RvsE, p = 0.004, RvsC, p = 0.007) in risedronate women compared to women in exercise or control groups. There were no significant differences in reductions in Serum Ntx and Alkphase B based on assignment to exercise or control groups. For women prescribed risedronate, percent changes in Alkphase B and Serum Ntx from baseline were − 20.3% and − 19.0%; in exercise group women, − 6.7% and − 7.0%; and in control group women, − 6.3% and − 9.0% (see Table 4).
-
Aim 3: To describe women’s reports of adverse effects (AEs) to treatments as well as 12-month adherence to prescriptions for bone-loading exercises and risedronate.
The most frequent AE for calcium supplements was constipation (n = 7). Women with constipation discontinued their supplements and reported that they increased their dietary intake of calcium to measures of approximately 1200 mg daily. Women taking risedronate reported gastrointestinal (GI) disturbances (n = 4), muscle or joint pain (n = 11), and chest pain or dizziness (n = 2, and nine of these 17 women who had AEs did discontinue use of risedronate. All women with adverse effects to risedronate did report that their symptoms no longer occurred once they had discontinued the medication. No women complained of adverse effects with vitamin D. A few women complained of muscle soreness with exercises. Adaptations were made in their exercise schedules, and these women were able to continue to exercise. None of the women in the exercise group experienced a serious injury or fracture due to exercises. A greater number of women in the exercise group did withdraw from the study (n = 20) compared to women in control (n = 16) or risedronate (n = 13) groups. Women reported that they did not withdraw because of AEs from exercise, rather they believed they had no time to continue exercising (see Waltman, 2021 [22]).
-
Research question related to Aim 3: Were there relationships between adherence rates to exercises or to risedronate and changes in BMD and serum biomarkers of bone turnover after 12 months of the treatment?
For the 227 of 276 participants who remained in the study and completed 12-month testing, 12-month adherence to calcium was 92.0% (SD = 19.1); vitamin D, 94.2% (SD = 13.5); risedronate, 74.9% (SD = 39.5); and exercises, 58.9% (SD = 27.2). For women in the risedronate group, increased adherence rates to risedronate were correlated with improvements in BMD and decreases in both Serum Ntx and Alkphase B. There were no relationships between adherence to exercises and changes in BMD or bone turnover (see Table 5).
Discussion
Compared to control and exercise group women, women prescribed risedronate along with CaD had greater improvements in BMD at the spine and greater decreases in rates of bone formation and resorption.
Numerous studies have found BPs to be effective both in increasing BMD and in reducing fracture rate in postmenopausal women [8, 23, 24]. In the Waltman et al. (2010) study, postmenopausal women taking risedronate for 2 years had a 1.81% increase in BMD at the total hip and a 2.85% increase at the spine [23]. Harris et al. (1999) reported that risedronate reduced the incidence of vertebral fractures by 41 to 49% and nonvertebral fractures by 36% over 3 years [8]. In the Siris et al. (2008) study of 620 postmenopausal women with low bone mass, risedronate reduced the risk of fragility fractures by 73% over three years versus use of a placebo (p = 0.023) [24].
Although percent increases in BMD have been greater in other studies [23], the sample of women in our study were within their first 6 years of diagnosis of menopause. During early menopause, the average reduction in BMD is 1 to 2% annually [16]. Thus, maintaining BMD and the small increases in BMD in women in our study were positive findings supporting use of BPs.
While there is general agreement that one goal of all treatments for bone loss should be to “at least preserve BMD,” Cefalu (2014) suggests that large increases in BMD may not always be related to significant decreases in fractures. In their review of studies on treatment of bone loss, Cefalu reported the amount of change in BMD with treatment correlated poorly with changes in fracture rate [25]. This finding suggests that while low BMD is one cause of fracture, there are multiple other causes.
Four women taking risedronate did report gastrointestinal (GI) symptoms. However, a recent study by Siris et al. (2008) found that when women participated in blinded placebo studies, there was no difference in number of GI symptoms between women who took risedronate and women who took a placebo (p = 0.78) [24]. The Siris et al. article did not specifically address incidence of muscle or joint pain with risedronate [24]. Due to eleven women in our study (greater than 10%) reporting incidence in muscle and joint pain with risedronate, providers should be alert for these AEs and monitor patients at follow-up visits. None of the women in our study reported that AEs with risedronate continued after they stopped taking the drug.
BPs have consistently been reported as having relatively acceptable risk–benefit and safety profiles [27]. While women in our study were only prescribed risedronate for twelve months, AEs with long-term use, while extremely rare, have included atypical hip and femur fractures [10]. Researchers have suggested that BPs slow the natural ability of bone to repair the cracks occurring with microdamage over time [9, 10]. Due to concerns about atypical fractures with long-term use, providers frequently discontinue prescriptions for BPs after three to five years in patients who are only a modest risk of fracture [26]. However, even with long-term use, AEs are not likely to outweigh the benefits of BPs. Khosla (2016) and others suggest that articles on atypical fractures have resulted in exaggerated concerns about AEs with BPs. They believe these concerns have resulted in providers not prescribing BPs as often as warranted, and patients refusing to take medications when they are prescribed (27, 28).
In this study, after 12 months, rates of bone resorption and bone formation were significantly reduced in women taking risedronate compared to women in exercise or control groups. For reduction in fracture risk, a goal of treatment should be to reduce serum Ntx levels to at least 12.6 nM BCE or below (the mean value for premenopausal women) [28]. According to Garnero (1996), women with both low hip BMD and high rates of bone resorption have a 4-to-fivefold higher risk of fractures compared to the general population [29, 30].
Biomarkers may be more useful for measuring effectiveness of medications than for exercises. This is because biomarkers reflect changes in overall skeletal bone loss and the whole-body rates of bone resorption and formation. Exercises are targeted to specific skeletal sites, and their effectiveness is better measured by changes in BMD at these specific sites [21].
In our study, after twelve months of bone-loading exercises, women had some improvement in BMD at the spine and total hip, although improvements were greater in the risedronate group. Other studies have reported even greater improvements in BMD with exercise as well as decreases in fracture rates [11,12,13,14]. In addition to increasing BMD, exercises have been documented as increasing insulin sensitivity, improving bone structure, balance, gait, and muscle strength, and decreasing risk of falls and depression [31, 32].
Numerous studies of women treated with calcium and vitamin D for bone loss have found greater improvements in BMD over time [33,34,35]. However, in our study, after 12 months of treatment with calcium and vitamin D supplements, while women in the control group did have increased BMD at the total hip, they had decreases in BMD at the lumbar spine and femoral neck. Compared to the other two treatment groups (risedronate and exercise), women in the control (CaD) group did have less calcium intake at baseline and greater time since menopause (see Table 2). We repeated the hierarchical linear model analyses, this time controlling for calcium sufficiency at baseline and time since menopause in all three treatment groups. No changes in significance were observed from initial results. Although this does not explain why the control group had greater loss of BMD at 12 months compared to the other two treatment groups, it does rule out the possibility that the observed results can be explained by calcium intake at baseline or time since menopause.
Our findings support the conclusions from a systematic review and meta-analysis by Tai et al. (2015) reporting that prescriptions for calcium and vitamin D alone are unlikely to result in significant increases in BMD and are most useful when they are combined with other therapies, such as BPs and exercises [32]. Although prescribing calcium and vitamin D is better than no treatment, their use alone is unlikely to result in a meaningful reduction in fracture risk [33,34,35].
In our study, seven women reported that taking calcium supplements caused constipation and other GI symptoms. There have also been reports of renal stones with calcium intake and cardiovascular events with excessive supplements [33]. Because of the potential for adverse events, some experts have recommended that, if possible, elderly women increase their calcium intake through food rather than by taking supplements. Prescriptions for vitamin D supplements were well tolerated by women. Vitamin D supplements have been found to have beneficial effects on muscle performance, balance, and risk of falling as well as bone health [36]. Unfortunately, vitamin D deficiency has been reported as widespread in postmenopausal women and the elderly [38].
Adherence to calcium and vitamin D supplements and risedronate were measured with pill counts. This method has worked well in numerous previous studies of adherence rates, with less than 5% missing pill count data [23]. Adherence to both calcium and vitamin D supplements was optimal—greater than 90%. Mean adherence rates for women participating in exercises were low—58.9%—and no AEs were reported with participation in exercises. Both women and providers fear injuries such as fractures when women with bone loss engage in high-intensity exercises. Our study demonstrated that when women exercise at approved fitness centers, when exercises were monitored by certified ETs, and when weight loads were increased progressively, injuries can be very rare or nonexistent.
Mean adherence to risedronate in this study was 74.9% ± 39.5%, and adherence rates were positively correlated with increases in BMD. There was no relationship between adherence to exercise and changes in BMD. One explanation for this is offered by Beck et al. (2010). According to Beck et al., exercises stimulate bone formation on the outer bone surface (the periosteum). This increase in bone cross-sectional area results in a higher denominator for the calculation of BMD (bone mineral content/region area). Thus, the finding that exercise has minimal or no effect on BMD may be misleading due to the method for calculating measures of BMD [38].
Finally, the effectiveness of exercises in reducing fracture rate may be underestimated if studies only measure outcomes such as BMD and biomarkers of bone turnover as predictors of fractures. There appear to be multiple causes of fractures in postmenopausal women, and low BMD and high bone turnover are just two of them [38]. In a recent study of 2320 Australian women over the age of 50, Mai et al. (2019) reported that almost 53% of women and 90% of men who sustained a fracture had BMD T-scores > − 2.0 [39]. After reviewing findings from numerous studies, Hurley and Armstrong (2012) concluded that both BPs and exercises could improve bone strength and reduce fracture risk. However, they worked through different mechanisms. BPs improved bone strength by increasing BMD at the spine, hip, and femoral neck and by reducing the rate of bone loss. Bone-loading exercises were more likely to strengthen bones and reduce fractures by maintaining BMD, improving bone structure, muscle strength, balance, and gait and by reducing falls [41].
Limitations
The optimal goal of enrolling 302 participants in the study was not met. Another limitation is that 49 of the 276 participants enrolled withdrew during the study, or greater than 17%. Also, a greater number of women in the exercise group (n = 20) withdrew, compared to women in the control (n = 16) or risedronate group (n = 13). Finally, because of the homogenous nature of our sample population (average age of 54 years, majority were white/non-Hispanic and highly educated, all women were from one Midwestern state), results cannot be generalized to other populations of women. Less than 15% of the sample of 276 women enrolled were from minority populations. The underrepresentation of Hispanic and Asian women in osteoporotic studies is especially a concern because these women are considered at increased risk for bone loss and osteoporosis [41].
Future directions
The primary aim of treatment for postmenopausal women with bone loss is to reduce incidence of fractures. A future study for our research team would be to examine whether BMD and bone turnover changes after 12 months of treatment translated into decreased fracture rates. Future studies should be conducted examining risk factors for fractures in postmenopausal and elderly women in addition to low BMD and increased rate of bone turnover. Individual programs could be developed for women that would include prescribing interventions for prevention of fractures most applicable to each of the women’s assessed needs.
Conclusions
Conclusions from our study are that postmenopausal women with low bone mass should be encouraged to obtain adequate calcium and vitamin D and participate in bone-loading exercises. Also, the additional use of BPs will increase BMD, especially at the spine. Decision-making as to whether individual women with low bone mass should be prescribed BPs should be determined by assessments of these women that could include measures of BMD, bone turnover, bone structure, FRAX scores, and other risk factors for fractures including a sedentary lifestyle; concerns about adverse effects of BPs with both short-term and long-term use; and incorporating patient preferences into treatment. Women will be more likely to adhere to their medication and treatment regimen if they have had input into their plan of care [42].
Data availability
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Abbreviations
- BMD:
-
Bone mineral density
- BPs:
-
Bisphosphonates
- CaD:
-
Calcium and vitamin D supplements
- CORC:
-
Creighton University Osteoporosis Research Center
- DXA:
-
Dual-energy X-ray absorptiometry
- ETs:
-
Exercise trainers
- FRAX:
-
Fracture risk assessment
- GI:
-
Gastrointestinal
- HSA:
-
Hip structural assessment
- Lbs.:
-
Pounds
- LIFTMOR:
-
Lifting Intervention for Training Muscle and Osteoporosis Rehabilitation
- NOF:
-
National Osteoporosis Foundation
- pQCT:
-
Peripheral quantitative computed tomography
- 1RM:
-
One-repetition maximum
References
Cosman F, de Beur SJ, LeBoff MS, Lewiecki EM, Tanner B, Randall S, Lindsay R (2014) Clinician’s guide to prevention and treatment of osteoporosis. Osteoporosis International: A Journal Established as Result of Cooperation between the European Foundation for Osteoporosis and the National Osteoporosis Foundation of the USA 25(10):2359–2381. https://doi.org/10.1007/s00198-014-2794-2
Patel, D., Worley, J. R., Volgas, D. A., & Crist, B. D. (2018) The effectiveness of osteoporosis screening and treatment in the Midwest. Geriatric Orthopaedic Surgery & Rehabilitation, 9. https://doi.org/10.1177/2151459318765844
Rizzoli R (2018) Postmenopausal osteoporosis: assessment and management. Best Prac Res Clin Endocrinol Metab 32(5):739–757
Wright NC, Looker AC, Saag KG, Curtis JR, Delzell ES, Randall S, Dawson-Hughes B (2014) The recent prevalence of osteoporosis and low bone mass in the United States based on bone mineral density at the femoral neck or lumbar spine. Journal of Bone and Mineral Research: The Official Journal of the American Society for Bone and Mineral Research 29(11):2520–2526. https://doi.org/10.1002/jbmr.2269
Downey C, Kelly M, Quinlan JF (2019) Changing trends in the mortality rate at 1-year post hip fracture–a systematic review. World Journal of Orthopedics 10(3):166–175. https://doi.org/10.5312/wjo.v10.i3.(20166
Delmas, P.D., Eastell, R., Garnero, P., Seibel, M.J. and Stepan, J. for the Committee of Scientific Advisors of the International Osteoporosis Foundation (2000) The use of biochemical markers of bone turnover in osteoporosis. Osteroporosis Int Suppl 6:S2-17
Tu KN, Lie JD, Wan CK et al (2018) Osteoporosis: a review of treatment options. P & T 43(2):92–104
Harris ST, Watts NB, Genant HK et al (1999) Effects of risedronate treatment on vertebral and nonvertebral fractures in women with postmenopausal osteoporosis: a randomized controlled trial. Vertebral efficacy with risedronate therapy (VERT) study group. JAMA 282(14):1344–1352
Lenard L (2009) Bisphosphonates: bone strengtheners or bone hardeners? Townsend Letter. The Examiner of Alternative Medicine 312:76–81
Hellier S, Ross C (2012) Long-term bisphosphonate therapy: possible link to rare femur fracture. Nurse Pract 16(9/10):12–18
Hakestad KA, Torstveit MK, Nordsletten L, Risberg MA (2015) Effect of exercises with weight vests and a patient education programme for women with osteopenia and a healed wrist fracture: a randomized, controlled trial of the OsteoACTIVE programme. BMC Musculoskelet Disord 16:352–360. https://doi.org/10.1186/s12891-015-0811-z
Kemmler W, Bebenek M, Kohl M, von Stengel S (2015) Exercise and fractures in postmenopausal women. Results of the controlled Erlangen Fitness and Osteoporosis Prevention Study (EFOPS). Osteoporos Int 26(10):2491–2499. https://doi.org/10.1007/s00198-015-3165-3
Martyn-St James M, Carroll S (2009) A meta-analysis of impact exercise on postmenopausal bone loss: the case for mixed loading exercise programmes. Br J Sports Med 43(12):898–908. https://doi.org/10.1136/bjsm.2008.052704
Watson S, Weeks BK, Weis LJ, Harding AT, Horan SA, Beck B (2017) High-intensity resistance and impact training improves bone mineral density and physical function in postmenopausal women with osteopenia and osteoporosis: The LIFTMOR Randomized Controlled Trial. J Bone Miner Res 34(3):572–572. https://doi.org/10.1002/jbmr.3284
Polit, D. & Beck, C. (2018) Essentials of nursing research, 9th ed. Lippincott, Williams & Wilkins
Ji MX, Yu Q (2015) Primary osteoporosis in postmenopausal women. Chronic Diseases and Translational Medicine 1(1):9–13. https://doi.org/10.1016/j.cdtm.2015.02.006
Kanis JA, Hans D, Cooper C, Baim S, Bilezikian JP, Binkley N, Cauley JA, Compston JE, Dawson-Hughes B, El-Hajj Fuleihan G, Johansson H, Leslie WD, Lewiecki EM, Luckey M, Oden A, Papapoulos SE, Poiana C, Rizzoli R, Wahl DA, McCloskey EV (2011) Interpretation and use of FRAX in clinical practice. Osteoporos Int 22(9):2395–2411. https://doi.org/10.1007/s00198-011-1713-z
Kanis JA, Adachi JD, Cooper C, Clark P, Cummings SR, Diaz-Curiel M et al (2013) Standardizing the descriptive epidemiology of osteoporosis: recommendations from the Epidemiology and Quality of Life Working Group of IOF. Osteoporosis Int 24(11):2763–2764. https://doi.org/10.1007/s00198-013-2413-7
Waltman NL, Smith KM, Kupzyk KA, Lappe JM, Mack LR, Bilek LD (2019) Approaches to recruitment of postmenopausal women for a community-based study. Nurs Res 68(4):307–316. https://doi.org/10.1097/NNR.0000000000000356
Bilek LD, Waltman NL, Lappe JM, Kupzyk KA, Mack LR, Cullen DM, Berg K, Langel M, Meisinger M, Portelli-Trinidad A, Lang M (2016) Protocol for a randomized controlled trial to compare bone-loading exercises with risedronate for preventing bone loss in osteopenic postmenopausal women. BMC Women’s Health. https://doi.org/10.1186/s12905-016-0339-x
Choplin RH, Lenchik L, Wuertzer S (2014) A practical approach to interpretation of Dual-Energy X-ray Absorptiometry (DXA) for assessment of bone density. Curr Radiol Rep 2:48. https://doi.org/10.1007/s40134-014-0048-x
Waltman N, Cole M, Kupzyk K, Lappe J, Mack L, Bilek L (2021) Promoting adherence to bone loading exercises in postmenopausal women with low bone mass. Journal of American Association of Nurse Practitioners. https://doi.org/10.1097/jXX.00000000000564
Waltman NL, Twiss JJ, Ott CD, Gross GJ, Lindsey AM, Moore TE, Berg K, Kupzyk K (2010) The effect of weight training on bone mineral density and bone turnover in postmenopausal breast cancer survivors with bone loss: a 24-month randomized controlled trial. Osteoporos Int 21(8):1361–1369. https://doi.org/10.1007/s00198-009-1083-y
Siris ES, Simon JA, Barton P, McClung MR, Grauer A (2008) Effects of risedronate on fracture risk of postmenopausal women with osteopenia. Osteoporos Int 19:681–686. https://doi.org/10.1007/s00198-007-0493-y
Cefalu CA (2004) Is bone mineral density predictive of fracture risk reduction? Curr Med Res Opin 20(3):341–349
Black DM, Schwartz AV, Ensrud KE et al (2006) Effects of continuing or stopping alendronate after 5 years of treatment: the fracture intervention trial long-term extension (FLEX): a randomized trial. JAMA 296:2927–2938
Khosla, S. & Shane, E. (2016). A crisis in the treatment of osteoporosis. Journal of Bone and Mineral Research. 101002/jbmr.2888
Jha S, Wang Z, Laucis N, Bhattacharyya T (2015) Trends in media reports, oral bisphosphonate prescriptions, and hip fractures 1996–2012: an ecological analysis. Bone Miner Res 30(12):2179–2187
Garnero P, Hausherr E, Chapuy MC et al (1996) Markers of bone resorption predict hip fracture in elderly women: the EPIDOS prospective study. J Bone Miner Res 11(10):1541–1538. https://doi.org/10.1002/jbmr.5650111021
Hlaing TT, Compston JE (2014) Biochemical markers of bone turnover–uses and limitations. The Association of Clinical Biochemistry & Laboratory medicine 51(2):189–202. https://doi.org/10.1177/000456321351515190
Moayyeri A (2008) The association between physical activity and osteoporotic fractures: a review of the evidence and implications for future research. Ann Epidemiol 18(11):827–835. https://doi.org/10.1016/j.annepidem.2008.08.007
Varahra A, Rodrigues IB, MacDermid JC, Bryant D, Birmingham T (2018) Exercise to improve functional outcomes in persons with osteoporosis: a systematic review and meta-analysis. Osteoporos Int 29(2):265–286
Abrahamsen B (2017) The calcium and vitamin D controversy. Therapeutic advances in musculoskeletal disease 9(5):107–114
Tai V, Leung W, Grey A, Reid IR, Bolland MJ (2015) Calcium intake and bone mineral density: systematic review and meta-analysis. BMJ 351:h4183. https://doi.org/10.1136/bmjh4183
Bolland MJ, Leung W, Tai V et al (2015) Calcium intake and risk of fracture: systematic review. BMJ 351:h4580. https://doi.org/10.1136/bmj.h4580
Murad MH, Elamin KB, Abu Elnour NO et al (2011) The effect of vitamin D on falls: a systematic review and meta-analysis. J Clin Endocrinol Metab 96(10):2997–3006
Linnebur SA, Vondracek SF, Vande Griend JP, Ruscin JM, McDermott MT (2007) Prevalence of vitamin D insufficiency in elderly ambulatory outpatients in Denver. Colorado Am J Geriatr Pharmacother 5(1):1–8
Beck TJ, Kohlmeier LA, Petit MA et al (2010) Cofounders in the Association between exercise and femur bone in postmenopausal women. Med Sci Sports Exerc 43(1):80–89
Mai HT, Tran TS, Ho-Le TP, Center JR, Eisman JA, Nguyen TV (2019) Two-thirds of all fractures are not attributable to osteoporosis and advancing age: implications for fracture prevention. J Clin Endocrinol Metab 104:3514–3520
Hurley B, Armstrong TJ (2012) Bisphosphonates vs exercise for the prevention and treatment of osteoporosis. The Journal for Nurse Practitioners 8(3):217–224. https://doi.org/10.1016/j.nurpra.2011.07.029
NIH Osteoporosis and Related bone Diseases: National Resource Center (2018). Osteoporosis and Hispanic women. NIH Pub. No. 18–7924 https://www.bones.nih.gov
Eastell R, Rosen CJ, Black DM, Cheung AM, Murad MH, Shoback D (2019) Pharmacological management of osteoporosis in postmenopausal women: an Endocrine Society Clinical Practice Guideline. J Clin Endocrinol Metab 104(5):1594–1622
Funding
Research reported in this publication was supported by the National Institute of Nursing Research of the National Institutes of Health under award number R01NR015029.
Author information
Authors and Affiliations
Contributions
All authors were involved in the implementation of this R01-funded study. All authors read and approved this final manuscript. Dr. Waltman1 developed the research project, wrote the initial draft of the manuscript, and assisted in analyzing and interpreting results of the study. Dr. Kevin Kupzyk2 was a co-investigator in the study, conducted the power analysis to determine sample size for the study, assisted in data analysis for this study, and reviewed and critiqued the final writing of this manuscript. Laura Flores3 provided valuable information on bone mineral density and bone structure during the analysis of study results and reviewed and critiqued the drafts of the manuscript. Dr. Lynn Mack4 and Dr. Joan Lappe5 were co-investigators for the study and assisted in designing and implementing the study. They reviewed and critiqued the final writing of this manuscript. Dr. Laura Bilek6 was co-principal investigator for the study. In addition to development of this research project, she participated in and supervised recruitment of subjects, data collection, and data entry for the study. She reviewed and critiqued the statistical analysis and the final writing for this manuscript.
Corresponding author
Ethics declarations
Conflicts of interest
None.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Waltman, N., Kupzyk, K.A., Flores, L.E. et al. Bone-loading exercises versus risedronate for the prevention of osteoporosis in postmenopausal women with low bone mass: a randomized controlled trial . Osteoporos Int 33, 475–486 (2022). https://doi.org/10.1007/s00198-021-06083-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00198-021-06083-2