Abstract
Summary
This study examined whether 24 months of weight training exercises enhanced the effectiveness of risedronate, calcium, and vitamin D in maintaining or improving bone mineral density (BMD) in 223 postmenopausal breast cancer survivors. Subjects who were ≥50% adherent to exercise had no improvement in BMD but were less likely to lose BMD.
Introduction
This study examined whether (1) postmenopausal breast cancer survivors (BCS) with bone loss taking 24 months of risedronate, calcium, and vitamin D had increased bone mineral density (BMD) at the total hip, femoral neck, L1-L4 spine, total radius and 33% radius, and decreased bone turnover; (2) subjects who also participated in strength/weight training (ST) exercises had greater increases in BMD and greater decreases in bone turnover; and (3) subjects who also exercised were more likely to preserve (at least maintain) BMD.
Methods
Postmenopausal BCS (223) were randomly assigned to exercise plus medication or medication only groups. Both groups received 24 months of 1,200 mg of calcium and 400 IU of vitamin D daily and 35 mg of risedronate weekly, and the exercise group additionally had ST exercises twice weekly.
Results
After 24 months, women who took medications without exercising had significant improvements in BMD at the total hip (+1.81%) and spine (+2.85%) and significant decreases in Alkphase B (−8.7%) and serum NTx (−16.7%). Women who also exercised had additional increases in BMD at the femoral neck (+0.29%), total hip (+0.34%), spine (+0.23%), total radius (+0.30%), and additional decreases in Alkphase B (−2.4%) and Serum NTx (−6.5%). Additional changes in BMD and bone turnover with exercise were not significant. Subjects who were ≥50% adherent to exercise were less likely to lose BMD at the total hip (chi-square [1] = 4.66, p = 0.03) and femoral neck (chi-square [1] = 4.63, p = 0.03).
Conclusion
Strength/weight training exercises may prevent loss of BMD in postmenopausal BCS at risk for bone loss.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
In 2009, an estimated 192,569 United States women will be diagnosed with breast cancer, and approximately 75% of these women will be long-term survivors of their disease [1]. However, breast cancer survivors (BCS) are at risk for another debilitating disease, osteoporosis, due to premature ovarian failure and treatment with glucocorticoids, aromatase inhibitors, and bone-wasting agents such as doxorubicin, cyclophosphamide, and methotrexate [2–7]. In addition, postmenopausal BCS are, generally, not candidates for hormone replacement therapy due to concerns about cancer recurrence [6]. Although breast cancer is the disease most feared by women, more women die from consequences of osteoporosis than from breast, endometrial, and ovarian cancer combined [1, 8].
To prevent osteoporosis and fractures, postmenopausal BCS are encouraged to increase intake of calcium and vitamin D and to participate in strength/weight training (ST) exercises [9]. Treatment with bisphosphonates is recommended as first-line therapy for postmenopausal women with BMD T-scores of −2 or less or T-scores of −1.5 or less with other risk factors for bone loss [10]. Numerous randomized controlled trials (RCT) have supported the effectiveness of calcium, vitamin D, and bisphosphonates in maintaining and improving bone mineral density (BMD) in postmenopausal women [11–14]. However, findings of studies on the impact of ST exercises on BMD have been inconsistent. Strength/weight training exercise interventions designed to increase BMD usually result in modest gains of 2–8.8% of bone mass after 1 to 2 years of training, while controls often lose 1% [15–17].
Bergstrom et al. [16] reported preservation of total hip BMD in postmenopausal women with bone loss randomized to 12 months of weight training. Women were between the ages of 45 and 65, had no history of breast cancer, and had T-scores of ≤−1 at the hip or spine. All women were given a supplement of calcium and vitamin D, and no women were taking bisphosphonates. In the Bergstrom et al. study, patients less than 50% adherent to training were not included in the per protocol analysis. While women in the training group who were ≥50% adherent to exercises had less than 1% (+0.58%) increase in total hip BMD; women in the control group (calcium and vitamin D but no weight training) had a 0.36% decrease in BMD at the hip.
In their study of 164 postmenopausal women, Uusi-Rasi et al. [18] found no improvement in BMD with 12 months of weight-bearing jumping exercises. However, subjects who exercised did have improved bone structure (as measured by peripheral quantitative computed tomography) at some of the most heavily loaded sites.
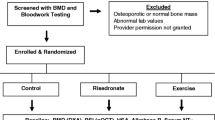
This is a report of the analysis of 24 month data collected in a federally-funded (2002–2007) RCT, testing a multicomponent intervention (calcium, vitamin D, risedronate, and ST exercises) for prevention or treatment of bone loss in 223 postmenopausal BCS with either osteopenia or osteoporosis. The 223 BCS were randomized to exercise plus medication (n = 110) or medication only (n = 113) treatment groups, and randomization was stratified by years of post menopause (<5 years; ≥5 years). Both groups received 24 months of 1,200 mg of calcium and 400 IU of vitamin D supplements daily and 35 mg of risedronate weekly. Exercise subjects also performed ST exercises twice weekly for upper and lower extremities and the spine. Institutional Review Board approval was obtained prior to the RCT, and informed consent was obtained from subjects at the time of their enrollment.
Purposes of this 24-month analysis were:
-
1.
To examine whether over 24 months, subjects on medications alone (risedronate, calcium, and vitamin D but no ST exercises) had increases in BMD at the total hip, femoral neck, L1-L4 spine, total radius and 33% radius, and decreases in serum markers for bone resorption (serum NTx) and formation (Alkphase B);
-
2.
To examine whether over 24 months, subjects in the exercise plus medication group had greater increases in BMD and greater decreases in serum markers for bone resorption (NTx) and formation (Alkphase B) compared to subjects on medications alone;
-
3.
To examine whether over 24 months, subjects in the exercise plus medication group were more likely to preserve (maintain or improve), BMD (Dual Energy X-ray Absorptiometry, DXA) at the total hip, femoral neck, and L1-L4 spine compared to subjects on medications alone.
To promote bone health, the American College of Sports Medicine recommends that individuals perform resistance training 2–3 times per week with 2–3 sets [19]. Effects of ST exercises are more pronounced if exercises are site-specific, high intensity, and when combined with calcium and vitamin D intake [17–21]. A 24-month ST exercise intervention is important because a bone turnover cycle takes 200 days [22]. Resistance training should last at least twice as long (400 days) to ensure the training effect on BMD can be measured.
Strength/weight training exercises impact both nonskeletal (impaired balance, muscle weakness, and falls) and skeletal (bone loss) causes of fractures. Numerous studies have documented the effectiveness of weight training in improving muscle strength and balance [19, 20]. In their study of elderly women, Swanenburg et al. reported an 89% reduction of falls due to improved muscle strength and balance with ST exercises [20]. Strength/weight training exercises are believed to prevent bone loss due to the positive relationship between muscular and bone strength. Increased muscular strength results in greater demands on bones and reduces the imbalance between bone resorption and formation [16].
Methods
Sample
The sample size for the RCT was 223 subjects. According to power calculations, a sample size of 218 subjects or 109 participants per treatment group provided 85% power to detect a difference in the true means of major outcome variables of 0.4 standard deviations (SD) [23].
Women could be aged 35 to 75; had completed breast cancer treatment (except tamoxifen and aromatase inhibitors) at least 6 months earlier; had amenorrhea for at least 12 months; had a BMD T-score of −1 standard deviation or lower; and had physician agreement for participation. They were excluded for conditions that prevented ST exercises, risedronate, calcium, or vitamin D intake. More specific eligibility criteria were reviewed elsewhere [24].
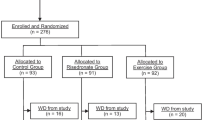
Recruitment procedures and participant flow are shown in Fig. 1. A total of 708 postmenopausal BCS were recruited through media ads, support group meetings, and clinician offices. Of the 708 women, 249 met screening criteria, were enrolled in the study and randomized to the two treatment groups. The 24 month analysis included 223 women (exercise: n = 110 and medication only: n = 113). Challenges in recruitment are described elsewhere [25].
Mean (SD) subject age was 58.69 (7.5) years, time since menopause was 7.52 (7.7) years, and time since completion of breast cancer treatment (surgery, radiation therapy, and/or chemotherapy) was 5.95 (6.1) years. Ninety-eight percent of the subjects had breast cancer surgery, 68% had cancer chemotherapy, and 45% had radiation therapy. Women were 98.7% Caucasian. Two subjects were African-American, and one subject was American Indian. At baseline, 158 of the subjects had a diagnosis of osteopenia and 65 had a diagnosis of osteoporosis.
In addition to breast cancer treatments, other variables have been found to impact bone loss, and these include age, time since menopause, body mass index (BMI), hours of hard activity (jogging 3–5 mph, dancing, and heavy house work) per week, calcium intake, cigarette smoking, and use of tamoxifen or aromatase inhibitors [26–29]. There were no differences in these selected subject variables when comparing exercise plus medication and medication only subjects. See Table 1.
This study was conducted at four sites, both rural and urban, across 500 miles in one Midwestern state. Strength/weight training exercises were performed in subjects’ homes using free weights, the first 9 months of the study, and at fitness centers, the last 15 months. Outcomes (except serum NTx and Alkphase B) were obtained and analyzed at laboratories and diagnostic centers at each of the four sites. All serum NTx and Alkphase B analyses were conducted at one regional research center.
ST exercises
Strength/weight training exercises were chosen to load bones at the hip, spine, and forearm and were modified with permission from exercises in Nelson’s (1997) book entitled Strong Women Stay Young [30]. For the first 9 months, participants exercised twice weekly for 30–45 min in their home, and they were not to lift beyond a 20-pound hand or ankle weights due to safety concerns. From 10 to 24 months, subjects exercised using weight machines at a nearby fitness center. A monthly stipend was provided to the fitness center for each exercise subject. Upper extremity exercises included biceps curl, overhead triceps or press and upward row, and lower extremity exercises included back and knee extensions, side hip raise, and hip flexion and extension. Subjects performed two sets of each exercise with 8–12 repetitions. After two consecutive training sessions at the maximum set and repetition, more weights were added. Potential goals for progressive training were increases in weights of 20% the first 3 months of exercises, 10% at 6 and 9 months, 5% at 12, 15, and 18 months, and 3% at 21 and 24 months. However, subjects increased weights based on individual response to training. In addition to exercises that required lifting weights, subjects performed two balance exercises without weights—toe stand and heel stand.
Long-term adherence to exercises in postmenopausal women is generally low [21]. Thus, facilitative strategies incorporating education, goal-setting, feedback, and symptom management, organized around Bandura’s (1997) self-efficacy theory [31] were used by research nurses and exercise trainers (ETs) to promote adherence to medications and ST exercises. Certified ETs demonstrated exercises to subjects and safety precautions in performing exercises, monitored performance in exercises, instructed subjects how to progressively increase weights lifted, and assisted subjects to transition from home-based to fitness center exercises. Exercise trainers made home or fitness center visits to subjects every 2 weeks at the beginning of home-based and fitness center exercises and every 2 months for the remainder of the 24 month study. Adverse effects of exercises generally involved temporary muscle soreness for up to 2 days following initiation of exercises or increase in weights lifted. If subjects complained of discomfort beyond the expected muscle soreness, exercises were not performed until the discomfort resolved. There were no serious, long-term, adverse effects from exercises for any of the 110 women exercising.
Measures
During the RCT, measures of BMD, bone formation and resorption, muscle strength, and balance were obtained at baseline and every 6 or 12 months. However, data reported in this study only compares baseline measures to those obtained at 12 and 24 months.
In this study, BMD (gm/cm2) at the total hip, femoral neck, L1-L4 spine, total radius, and 33% radius were measured by Dual Energy X-ray Absorptiometry (DXA) using Hologic QDR1000+™, Hologic QDR2000™, and GE Lunar Prodigy machines. All DXA results were interpreted by one research team radiologist. Dual Energy X-ray Absorptiometry results provide the most accurate and precise measure of quantity of BMD [32], and the preferred site for diagnosis of osteoporosis are BMD measures at the total hip or femoral neck [33].
Bone resorption (nmol/L BCE) was assessed using serum NTx assay, which measures the excretion of cross-linked N-telopeptides of type I collagen. Bone formation (U/L) was assessed using bone-specific alkaline phosphatase (Alkphase B) serum assay [34, 35]. Numerous studies have correlated serum levels of Alkphase B and NTx with loss of BMD and fracture risk [35, 36]. In one study, after 12 weeks of resistance training in postmenopausal women randomly assigned to exercise or control groups, bone resorption (NTx) decreased by 14.5% (p < 0.05) in exercise women [36].
Physical activity (light, moderate, hard, or very hard) was evaluated by the 7 Day Physical Activity Record-Adapted (7PAR-A) which has an acceptable 2-week test–retest reliability (r = 0.90) [37]. Scores have been correlated (r = 0.62) with perceived levels of physical activity among older adults [38]. Physical activity data were used in this study to assess group differences of more than light activity at baseline and to monitor exercise outside the exercise study protocol in both groups.
Muscle strength (peak torque/body weight at 60 degrees; knee, hip, and wrist flexion and extension) was assessed using Biodex™ velocity spectrum evaluation (Medical Systems, Inc., 1993). Intraclass correlation coefficients for reliability have been reported as 0.99 to 1.00 for the Biodex 3 measures across trial-to-trial and day-to-day reliability [39]. Dynamic balance was assessed using the timed backward tandem walk. Significant repeat measurements with a 1 week interval (r = 0.94; p = 0.001) have been reported [40].
Adherence rates to risedronate, calcium, and Vitamin D were calculated using ratios of reported to desired frequency of medication intake for each month of the study. Medications were dispensed to subjects in monthly medication calendar cards, and frequency of medication intake was measured using pill counts from the calendar cards. Adherence to ST exercises was calculated using ratios of reported-to-desired number of exercises performed. Subjects documented ST exercises and weights in their journals, and journals were monitored by ETs.
Data analysis
For the primary analysis, intent to treat paradigm was used where data from all participants were analyzed according to randomized assignment regardless of protocol adherence. Data were analyzed using a SPSS 15.0 statistical package. Descriptive statistics were used to describe the sample of BCS, and independent t tests were used to determine if there were differences between groups at baseline. The generalized estimating equation (GEE) method (SAS version 9.1) with an exchangeable structure for repeated measures data was used to fit a generalized linear model to examine factors associated with muscle strength, balance, BMD, and bone turnover including time of testing (baseline, 12 and 24 months) and group assignment (exercise or medication only) [41]. Pair wise tests were conducted if the overall test was significant to determine differences. Differences in number of subjects who preserved (maintained or improved) BMD in exercise and medications only groups were analyzed using chi-square. A p value of ≤0.05 was considered to be statistically significant.
Results
For all 223 subjects, average 24 month adherence to risedronate was 96.2% (7.9) and to calcium/vitamin D supplements, 93.7% (10.0), and there were no differences in adherence rates based on randomization to ST exercises. Average 24-month adherence to ST exercises for the 110 women was 69.4% (24.0).
Muscle strength and balance
For the 110 subjects performing 24 months of resistance training, average gains in muscle strength were 9.5% and 28.5% for hip flexion and extension, 21.1% and 11.6% for knee flexion and extension, and 48.6% and 18.1% for wrist flexion and extension. Improvements in muscle strength of the magnitude of 10% to 20% are considered meaningful and related to improved function and fewer falls [39]. Additional findings on muscle strength and balance, as well as more details on the ST exercises used in this RCT are described elsewhere [24]. The remaining results were organized to respond to study purposes:
To examine whether over 24 months subjects on medications alone (risedronate, calcium, and vitamin D, but no ST exercises) had increases in BMD at the total hip, femoral neck, and L1-L4 spine and decreases in serum markers for bone resorption (serum NTx) and formation (Alkphase B).
Women who took risedronate, calcium, and vitamin D but did not exercise demonstrated significant mean increases from baseline to 24 months in BMD at the total hip (+1.81%) and L1-L4 spine (+2.85%). Over 24 months, BMD at the femoral neck increased (+0.63%), and BMD decreased at both the total radius (−0.57%) and 33% radius (−0.16%). Bone mineral density results at the femoral neck, total radius, and 33% radius were not significant. Over 24 months, subjects in the medication only group had significant decreases in Alkphase B (−8.7%, p < 0.001) and serum NTx (−16.7%, p < 0.001). See Table 2.
To examine whether over 24 months, subjects in the exercise group had greater increases in BMD and greater decreases in serum markers for bone resorption (NTx) and formation (Alkphase B) compared to subjects on medications alone.
Compared to the nonexercise group, women who took the medications and also exercised over 24 months had additional increases in BMD of +0.29% at the femoral neck, +0.34% at the total hip, +0.23% at the spine, +0.30 at the total radius, and a decrease of −0.02 at the 33% radius. While changes in BMD were greater in exercise than in medication only subjects, these changes based on treatment group were not significant. At 24 months, exercise group women had a 2.4% greater mean decrease in Alkphase B and a 6.5% greater mean decrease in serum NTx than medication only group women. Although different, the mean decreases were not significant. See Table 2.
To examine whether over 24 months subjects in the exercise group were more likely to preserve (at least maintain) BMD (DXA) at the total hip, femoral neck, and L1-L4 spine compared to subjects on medications alone.
Preservation of BMD over 24 months was operationally defined as no decrease in BMD from baseline greater than least significant change (LSC) [33]. The formula for LSC (1.96 SD/0.7071) was based on SDs calculated for Hologic and Lunar DXA machines at each study site. Over 24 months, 2.8% of subjects in the exercise group lost BMD at the total hip compared to 8.6% of subjects in the medication only group, and this difference approached significance (chi-square [1] = 3.36, p = 0.06).
A per protocol analysis was also performed that included the 81 of 110 exercise subjects who averaged 50% or greater adherence to the 24 months of ST exercises. With the per protocol analysis, only 1.2% of exercise plus medication subjects lost BMD at the total hip compared to 8.6% of medication only subjects (chi-square [1] = 4.66, p = .03). In addition, only 12.3% of exercise plus medication subjects lost BMD at the femoral neck compared to 26.7% of medication only subjects (chi-square [1] = 4.63, p = .03). See Table 3.
Discussion
Although an adherence rate to exercises of 69.4% is far from optimal, adherence to exercises in this study was higher than the 55% adherence reported by Von Stengel et al. [21] in their 24-month study of weight training for postmenopausal women. Adherence in this study may have been impacted by facilitative strategies used to promote ST exercises. Lack of adherence to long-term lifestyle interventions such as weight training remains an ongoing health care concern. Even the twice weekly ST exercises prescribed in this study may not be adequate for maintaining or improving bone health in postmenopausal women. A meta-analysis of effect sizes for 140 studies concluded that three times weekly creates a more effective dose response on strength than either one or two times weekly [42].
Subjects in our study who took 24 months of risedronate, calcium, and vitamin D without exercising had increased BMD at the hip and spine and decreased bone resorption. Implications are that bisphosphonates, calcium, and vitamin D are effective treatments for postmenopausal BCS with bone loss at the hip and spine. Other studies have documented risedronate as effective in preventing bone resorption in postmenopausal women. Studies have reported 0.2% to 6.6% increases in BMD over 24 months in women prescribed with 5 mg of risedronate daily as compared to women taking placebos [23].
After 24 months, subjects who exercised had greater improvements in muscle strength and balance than medication only subjects. These results are consistent with findings of Swanenberg et al. [20] and Karinkanta et al. [43]. In our study, using intention to treat analysis, fewer subjects in the exercise plus medication group lost BMD at the total hip, and this difference between exercise and medication only subjects approached significance (p = 0.06). With per protocol analysis (including only subjects with an average 24-month adherence to exercises of 50% or greater), significantly fewer subjects in the exercise plus medication group lost BMD at the total hip and femoral neck. Bergstrom et al. [16] also reported preservation of total hip BMD in postmenopausal women with bone loss who were at least 50% adherent to 12 months of weight training. Maintaining BMD at the hip and improvements in balance and muscle strength at the knee and hip are critical for prevention of hip fractures. Of all osteoporotic fractures, hip fractures have the greatest morbidity and economic consequences. Magaziner et al. [44] reported that at 12 and 24 months following a hip fracture, 50% of elderly patients continued to be disabled.
While at 24 months, the percentage of subjects who lost BMD at the hip was small in both exercise plus medication and medication only groups, this small number was most likely due to all subjects concurrently taking calcium and vitamin D supplements and a bisphosphonate (risedronate). The small percentage of subjects who were not exercising and who lost BMD on bisphosphonate therapy is still of importance because of the large population of postmenopausal BCS in the United States on bisphosphonates. Even a small percentage of this large population would translate into a large number of women.
Limitations
Women in this study were mostly Caucasian and thus, findings cannot be generalized to minority populations of women. An adherence rate to ST exercises of 69.4% was not optimal, and this was a limitation when assessing efficacy of exercises. Adherence problems are generally observed in long-term weight training studies. However, the use of per protocol analysis where subjects who are nonadherent to exercises are excluded from the analysis may induce a positive bias favoring the effect of the exercises [18]. Thus, use of per protocol analysis should be considered a limitation of the study. One way to address the problem of subject nonadherence would be to select subjects for the study who would be more adherent to ST exercises. However, this selection process would limit generalizability of results of the study [18].
Conclusions
While participation in ST exercises over 24 months did not result in statistically significant improved BMD in BCS with bone loss over medications alone, there was a trend for postmenopausal BCS who participated in ST exercises to at least maintain BMD at the total hip. Also, with per protocol analysis, subjects who were 50% or greater adherent to exercises were significantly less likely, than subjects on medications alone, to lose BMD at the total hip and femoral neck. For postmenopausal BCS with normal BMD or mild osteopenia who are at risk for further bone loss, prevention of loss of BMD may be a satisfactory outcome of treatment. Additional study is needed.
Future studies
More studies should be conducted with three times weekly ST exercises. Also, consideration should be given to recruitment of postmenopausal BCS who are likely to be adherent to ST exercises. Future studies could examine the effectiveness of ST exercises in preserving BMD in BCS with bone loss that cannot or will not take bisphosphonate therapy.
In the Uusi-Rasi study [18], while weight-bearing jumping exercises had no effect on BMD, subjects who exercised did have improved bone structure. There was a mean increase of 3.6% (95% confidence interval (CI), 0.3–7.1%) in bone strength index at the distal tibia site and a mean increase of 3.7% (95% CI, 0.1–7.3%) in the ratio of cortical bone to total bone area (cortical thickening). Thus, future studies testing the effectiveness of ST exercises in preventing fractures should include measures of bone structure along with measures of BMD and bone turnover and should include follow up of women participating in the exercises to document fractures.
References
American Cancer Society (2009) Cancer facts & figures 2009. American Cancer Society, Atlanta
Schwartz AL, Winters-Stone K, Galluci B (2007) Exercise effects on bone mineral density in women with breast cancer receiving adjuvant chemotherapy. Oncol Nurs Forum 34(3):627–633
Waltman N, Ott C, Twiss J et al (2008) Bone mineral density and bone turnover in postmenopausal women treated for breast cancer. Cancer Nurs 31(3):182–190
Shapiro CL, Manola J, Leboff M (2001) Ovarian failure after adjuvant chemotherapy is associated with rapid bone loss in women with early-stage breast cancer. J Clin Oncol 19(14):3306–3311
McCune JS, Games DM, Espirito JS (2005) Assessment of ovarian failure and osteoporosis in premenopausal breast cancer survivors. J Oncol Pharm Pract 11(2):37–43
Winer EP, Hudis C, Burstein HJ et al (2005) American Society of Clinical Oncology technology assessment on the use of aromatase inhibitors as adjuvant therapy for postmenopausal women in hormone receptor-positive breast cancer: status report 2004. J Clin Oncol 23(3):619–629
Lester J, Dodwell D, McCloskey E, Coleman R (2005) The causes and treatment of bone loss associated with carcinoma of the breast. Cancer Treat Rev 31(2):115–142
National Institutes of Health (2003) Osteoporosis and related bone disease. National Resource Center, Bethesda
Swenson KK, Henly SJ, Shapiro AC et al (2005) Interventions to prevent loss of bone mineral density in women receiving chemotherapy for breast cancer. Clin J Oncol Nurs 9(2):177–184
National Osteoporosis Foundation (1998) Osteoporosis: review of the evidence for prevention, diagnosis, and treatment, and cost-effectiveness analysis: executive summary. Osteoporos Int 8(suppl 4):S3–S6
Cranney A, Tugwell P, Adachi J et al (2002) Meta-analysis of risedronate for the treatment of postmenopausal osteoporosis. Endocr Rev 23(4):517–523
Jackson RD, LaCroix AZ, Gass M et al (2006) Calcium plus vitamin D supplementation and the risk of fractures. N Engl J Med 354(7):669–683
Recker R, Barger-Lux J (2005) Risedronate for prevention and treatment of osteoporosis in postmenopausal women. Expert Opin Pharmacother 6(3):465–477
Tang BM, Eslick GD, Nowson C et al (2007) Use of calcium or calcium in combination with vitamin D supplementation to prevent fractures and bone loss in people aged 50 years and older: a meta-analysis. Lancet 370(9588):657–666
Asikainen TM, Kukkonen-Harjula K, Miilunpalo S (2004) Exercise for health for early postmenopausal women: a systematic review of randomized controlled trials. Sports Med 34(11):753–778
Bergstrom I, Landgren B, Brinck J et al (2008) Physical training preserves bone mineral density in postmenopausal women with forearm fractures and low bone mineral density. Osteoporos Int 19(2):177–183
Engelke K, Kemmler W, Lauber D et al (2006) Exercise maintains bone density at spine and hip EFOPS: a 3-year longitudinal study in early postmenopausal women. Osteoporos Int 17(1):133–142
Uusi-Rasi K, Kannus P, Cheng S et al (2003) Effect of alendronate and exercise on bone and physical performance of postmenopausal women: a randomized controlled trial. Bone 33(1):132–143
Kraemer WJ, Adams K, Cafarelli E et al (2002) American college of sports medicine position stand. Progression models in resistance training for healthy adults. Med Sci Sports Exerc 34(2):364–380
Swanenburg J, de Bruin ED, Stauffacher M et al (2007) Effects of exercise and nutrition on postural balance and risk of falling in elderly people with decreased bone mineral density: randomized controlled trial pilot study. Clin Rehabil 21(6):523–534
Von Stengel S, Kemmler W, Kalender WA et al (2007) Differential effects of strength versus power training on bone mineral density in postmenopausal women: a 2-year longitudinal study. Br J Sports Med 41(10):649–655
Erickson E, Axelrod D, Melsen F (1994) Bone histomorphometry. Raven, New York
Fogelman I, Ribot C, Smith R et al (2000) Risedronate reverses bone loss in postmenopausal women with low bone mass: results from a multinational, double-blind, placebo-controlled trial. J Clin Endocrinol Metab 86(5):1895–1901
Twiss J, Waltman N, Berg K et al (2009) An exercise intervention for breast cancer survivors with bone loss. J Nurs Scholarship 41(1):20–27
Ott C, Twiss J, Waltman N et al (2006) Challenges of recruitment of breast cancer survivors to a randomized clinical trial for osteoporosis prevention. Cancer Nurs 29(1):21–33
Bass M, Ford M, Mauromoustakos A et al (2006) Variables for the prediction of femoral bone mineral status in American Women. South Med J 99:115–122
Kanis J, Borgstrom F, Laet C et al (2005) Assessment of fracture risk. Osteoporosis Int 16:581–589
MacInnis R, Cassar C, Nowson C et al (2003) Determinants of bone density in 30 to 65 year old women: a co-twin study. J Bone Miner Res 18(2):1650–1656
Ward K, Klesges R (2001) A meta-analysis of the effects of cigarette smoking on bone mineral density. Calcif Tissue Int 68:259–270
Nelson ME (1997) Strong women stay young. Bantam Books, New York
Bandura A (1997) Self-efficacy: the exercise of control. WH Freeman & Co Group, New York
Camacho P, Miller P (2007) Osteoporosis: a guide for clinicians. Lippincott Williams & Wilkins, Philadelphia
Kanis J, Gluer C (2000) An update on the diagnosis and assessment of osteoporosis with densitometry. Osteoporos Int 11:192–202
Caulfield MP, Reitz RE (2004) Biochemical markers of bone turnover and their utility in osteoporosis. Med Lab Obs 36(4):34–37
Leeming DJ, Alexandersen P, Karsdal MA et al (2006) An update on biomarkers of bone turnover and their utility in biomedical research and clinical practice. Eur J Clin Pharmacol 62(10):781–792
Klentrou P, Slack J, Roy B et al (2007) Effects of exercise training with weighted vests on bone turnover and isokinetic strength in postmenopausal women. J Aging Phys Act 15(3):287–299
Blair SN (1984) How to assess exercise habits and physical fitness. In: Matarazzo JD, Weiss SM, Herd JA et al (eds) Behavioral health: a handbook of health enhancement and disease prevention. Wiley, New York, pp 424–447
Hellman EA, Williams MA, Thalken L (1996) Modifications of the 7-day activity interview for use among older adults. J Appl Gerontol 15(1):116–132
Drouin JM, Valovich-mcLeod TC, Shultz SJ et al (2004) Reliability and validity of the Biodex system 3 pro isokinetic dynamometer velocity, torque, and position measurements. Eur J Appl Physiol 91(1):22–29
Nelson ME, Fiatarone MA, Morganti CM et al (1994) Effects of high-intensity strength training on multiple risk factors for osteoporotic fractures. A randomized controlled trial. JAMA 272(24):1909–1914
Zeger SL, Liang KY, Alber PS (1988) Models for longitudinal data: a generalized estimating equation approach. Biometrics 44(4):1049–1060
Rhea M, Alvar B, Burkett L et al (2003) A meta-analysis to determine the dose response of strength development. Med Sci Sports Exerc 35(3):456–464
Karinkanta S, Heinonen A, Sievanen H et al (2007) A multi-component exercise regimen to prevent functional decline and bone fragility in home-dwelling elderly women: randomized controlled trial. Osteoporosis Int 18:453–462
Magaziner J, Fredman L, Hawkes W et al (2003) Changes in functional status attributable to hip fracture: a comparison of hip fracture patients to community-dwelling aged. Am J Epidemiol 157:1023–1031
Acknowledgments
Funding sources include the National Institutes of Health NINR 1 RO1 NR07743-05A1; Wyeth Consumer Health Care; UNMC Clinical Research Center; and Aventis–Proctor and Gamble. Special thanks to Lynette Smith, MS, for her assistance with GEE analyses.
Conflicts of interest
None.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Waltman, N.L., Twiss, J.J., Ott, C.D. et al. The effect of weight training on bone mineral density and bone turnover in postmenopausal breast cancer survivors with bone loss: a 24-month randomized controlled trial. Osteoporos Int 21, 1361–1369 (2010). https://doi.org/10.1007/s00198-009-1083-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00198-009-1083-y