Abstract
Aim
It is recognized that stereotactic body radiotherapy (SBRT) for centrally located lung metastases is affected by higher rates of severe toxicity. In the present study, we report the clinical outcomes following a novel intensity-modulated radiotherapy prescription dose, termed simultaneous integrated protection (SIP), for nearby organs at risk (OARs).
Materials and methods
The prescribed total doses of SBRT were 70 Gy in 10 fractions and 60 Gy in 8 fractions. For ultra-centrally located lesions, a dose of 60 Gy in 10 fractions was delivered. The main planning instructions were: (1) to remain within the limits of the given dose constraints for an OAR; (2) to make use of the maximum possible dose to the OARs to minimize dose inhomogeneity for the Planning Target Volume (PTV). SBRT-related toxicity was prospectively assessed according to the Common Terminology Criteria for Adverse Events (CTCAE) v4.0. The primary clinical endpoint was the SBRT-related toxicity. Secondary endpoint was local control.
Results
Forty patients affected by a single central malignancy were analyzed. The median follow-up was 20 months (range, 6–58 months). Acute and late clinical pulmonary toxicity ≥grade 2 was recorded in 2 out of 40 patients (5%) and 3 out of 40 patients (7%), respectively. No patient experienced cardiac toxicity. No narrowing or stenosis of any airway or vessel was registered. One-year local control rate was 91%. The median time to local progression was 13 months (range, 6–46 months).
Conclusion
SBRT using a PTV-SIP approach for single central lung metastases achieved low SBRT-related toxicity with acceptable local control.
Zusammenfassung
Ziel
Die extrakranielle Körperstereotaxie (SBRT) von zentral gelegenen Lungentumoren weist höhergradige Toxizitäten auf. In der vorliegenden Studie berichten wir über die klinischen Ergebnisse einer neuen intensitätsmodulierten Technik, welche zur Reduktion der Toxizität eine simultan integrierte Protektion (SIP) von Risikoorganen (OARs) anwendet.
Material und Methoden
Die Verschreibungsdosis der SBRT war 70 Gy in 10 Fraktionen und 60 Gy in 8 Fraktionen. Bei sehr zentral gelegenen Läsionen wurde eine Dosis von 60 Gy in 10 Fraktionen appliziert. Die wichtigsten Punkte bei der Bestrahlungsplanung waren folgende: (1) innerhalb der Toleranzgrenzen der OAR zu bleiben; (2) die maximale Dosis eines OARs auszureizen, um die Dosisinhomogenität für das PTV zu minimieren. Die therapiebezogene Toxizität wurde prospektiv nach den Common Terminology Criteria for Adverse Events (CTCAE) v4.0 erhoben. Der primäre klinische Endpunkt war die SBRT-bezogene Toxizität. Sekundärer Endpunkt war die lokale Kontrolle.
Ergebnisse
Insgesamt wurden 40 Patienten an einem singulären zentralen Lungentumor behandelt. Das mediane Follow-up betrug 20 Monate (Spanne 6–58 Monate). Akute pulmonale Nebenwirkungen ≥Grad 2 wurden bei 2 von 40 Patienten (5%) festgestellt und pulmonale Spätnebenwirkungen (≥Grad 2) bei 3 von 40 Patienten (7%). Kein Patient wies eine kardiale Toxizität auf. Es wurde keine Verengung oder Stenose der Atemwege oder Gefäße festgestellt. Die lokale 1‑Jahres-Kontrollrate lag bei 91%. Die mediane Zeit bis zum lokalen Progress betrug 13 Monate (Spanne 6–46 Monate).
Schlussfolgerung
Eine SBRT unter Verwendung eines PTV-SIP-Ansatzes für zentrale Lungenmalignome ermöglicht eine akzeptable lokale Tumorkontrolle bei niedriger therapieassoziierter Toxizität.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
In the management of lung malignancies, stereotactic body radiotherapy (SBRT) represents an effective treatment option with acceptable adverse events [1,2,3,4]. In daily clinical practice, the real challenge for radiation oncologists is represented by the so-called central lung tumors, mainly due to two reasons: (i) the increased levels of severe toxicity, as reported by the Radiation Therapy and Oncology Group (RTOG) 0236 study [5] and (ii) the need to provide an adequate dose to the tumor to guarantee high rates of effectiveness. Starting from the last point, it is recognized that a biologically effective dose (BED) of at least 100 Gy remains a strong predictive factor of long-term local control (LC) after SBRT [6, 7]. On the other hand, following the first concern, a sort of dogma was formulated for centrally located tumors, defining this last clinical scenario as a “no fly zone” or SBRT-exclusion area.
Notably, over the last years, the adoption of more fractionated treatment schedules was shown to be safer in case of SBRT for central lung malignancies, maintaining acceptable local control rates [8, 9].
Technical and technological advances including on-board imaging and intensity-modulated irradiation allowed reduction of high doses to surrounding healthy tissue and improved the tolerability profile in several settings, including lung malignancies [10,11,12]. Additionally, an interesting prescription modality termed simultaneous integrated protection (PTV-SIP), was investigated for patients in which SBRT was performed close to high-risk OARs. To date, the clinical data following this last prescription strategy are mainly reported by studies evaluating the role of SBRT for gastrointestinal cancers [13, 14].
The main planning instructions of PTV-SIP are: (1) to remain within the limits of the given dose constraints for an OAR; (2) to make use of the maximum possible dose to the OARs to minimize dose inhomogeneity for the PTV [13].
To the best of our knowledge, no clinical data are available regarding the impact of SBRT using a PTV-SIP approach in case of central lung malignancies. Herein, we report our experience in terms of the tolerability and effectiveness of PTV-SIP in a cohort of patients affected by central lung tumors receiving SBRT.
Materials and methods
Study design
The present study is a mono-institutional prospective observational study. The inclusion criteria were: single central lung metastasis receiving SBRT; absence of extra-thoracic disease; Karnofsky performance status >70; maximum tumor diameter <5 centimeters (cm); at least 6 months of follow-up after SBRT. Lung metastases were defined as central according to the International Association for the Study of Lung Cancer (IASLC) recommendations (i. e., tumor within 2 cm of any mediastinal critical structure) [15].
Stereotactic body radiotherapy
The prescribed total doses of SBRT were: 60/70 Gy in 10 fractions and 60 Gy in 8 fractions according to a risk-adapted dose prescription [16]. More deeply, regarding the fractionation schedule for each patient (10 × 7 Gy or 8 × 7.5 Gy) were used indifferently. In case of problems to achieve the OAR constraints in our clinical practice, a dose of 60 Gy in 10 fractions instead of 70 Gy in 10 fractions was chosen.
The following organs at risk (OAR) were delineated during treatment planning: healthy lungs (total lung minus PTV), main bronchus, esophagus, main vessels, heart, ribs, and spinal cord. For all of the above-listed OARs, except for the lungs and the heart, a planning risk volume (PRV) was derived by a 3 mm isotropic expansion.
The near-maximum doses to the PRVs, referred to an organ-specific threshold volume (cm3), had to be smaller than 50 Gy for the main bronchus (4 cm3), 30 Gy for the esophagus (0.5 cm3), 47 Gy for major vessels (10 cm3), 40 Gy for the heart (1 cm3) and the trachea (1 cm3), 60 Gy for the ribs (1.4 cm3), and 27 Gy for the spinal cord (0.3 cm3) [17]. Furthermore, for total lung minus PTV, the mean lung dose constraint was <4 Gy.
In cases of an overlap of the PTV and any of the PRVs, at least 95% of the PTV minus PRV (PTVmp) volume had to be covered by 95% of the prescription dose (Dp), while the upper dose constraint of the OAR had to be satisfied by the relative PRV as a necessary condition for planning approval. The dose prescription was at the median PTV dose with assurance from optimization to 95% of the prescribed dose to at least 95% of the PTV and a near-maximum dose not superior to 107% of the prescribed dose.
All plans were performed by volumetric intensity-modulated radiotherapy (VMAT). Before each fraction, image-guided radiotherapy was performed by means of cone beam computed tomography (CBCT).
Evaluation of SBRT response
In all study participants, the clinical outcome was evaluated by both thoracic and abdominal computed tomography (CT) scans and 18F-fluorodeoxyglucose (FDG)-positron-emission tomography (PET)/CT before SBRT. Evaluation of tumor response was then assessed by means of CT scan every 3 months and/or 18FDG-PET/CT, if needed, after SBRT according to RECIST or PERCIST criteria.
Toxicity evaluation
SBRT-related toxicity was prospectively assessed according to Common Terminology Criteria for Adverse Events (CTCAE) v4.0 during SBRT and every 3 months thereafter. Toxicities occurring within 3 months from the beginning of SBRT were defined as acute, those occurring after 3 months as late toxicity. Regarding pneumonitis, radiological and clinical toxicities were defined using Ikezoe et al. criteria [18] and Common Terminology Criteria for Adverse Events (CTCAE) version 4.0, respectively.
Statistical analysis
To summarize the most relevant clinical features of the study participants, descriptive statistics were performed. The primary clinical endpoint was SBRT-related toxicity. Secondary endpoint was local control (LC).
Results
According to the inclusion criteria of the present analysis, 40 patients affected by a single central malignancy were selected. The median follow-up was 20 months (range 6–58 months). The median age was 72 years (range 30–81 years). The patients’ characteristics are summarized in Table 1.
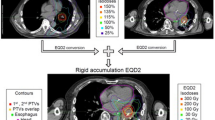
The median distance between tumor and mediastinal healthy structures was 0.6 cm (range 0–1.8 cm). In Fig. 1, an example of central lesion SBRT, with Dp equal to 70 Gy given in 10 fractions and where the right bronchus was the mediastinal critical OAR is presented, with the purpose of illustrating the different volumes involved and their dose coverage/sparing in the SIP approach. On a transverse plane (1a), the different PTVs, i. e., PTVmp (translucent purple) and PTVop (translucent blue), whose union corresponds to the whole PTV, together with the right bronchus and its relative PRV (both in green), are depicted. In (1b), the dose distribution is overlaid to the same transverse plane. In (1c) and (1d), the cumulative DVHs for the bronchus (solid green line) and its PRV (dotted green line), and for the three abovementioned PTVs (1d), are graphed to show that the necessary constraint, D4cc(bronchus) <50 Gy, was satisfied from the bronchial PRV, while an adequate target dose coverage, 95%Dp to >95% of PTVmp, was assured.
An example of central lesion SBRT with Dp equal to 70 Gy given in 10 fractions. In (a) the different planning target volumes (PTVs), i. e., PTVmp (PTV minus PRV; translucent purple) and PTVop (PTV minus overlap; translucent blue), whose union corresponds to the whole PTV, are depicted on a transverse plane. The right bronchus, and its relative planning at risk volume (PRV), are both reported in green. In (b) the dose distribution, by relative isodose lines, is overlaid to the same transverse plane. In (c) and (d) the cumulative DVHs for the bronchus (solid green line) and its PRV (dotted green line), and for the three above PTVs (d), are graphed. The necessary constraint, D4cc(bronchus) <50 Gy, was satisfied by the bronchial PRV together with an adequate target dose coverage: 66.5 Gy (= 95% Dp) was assured to >95% of PTVmp (PTV minus PRV)
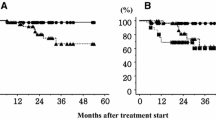
The 1‑year local control rate was 91%. The median time to local progression was 13 months (range 6–46 months). One-year distant progression-free survival rate was 71.7%. At the time of the analysis, the 1‑ and 2‑year overall survival rates were 86.9% and 72.6%, respectively. In Fig. 2, the Kaplan–Maier curves regarding the abovementioned oncologic outcomes are shown.
Pulmonary toxicity
Acute radiological abnormalities were identified as follows: no changes in 16/40 patients (40%), patch ground glass opacity in 9/40 (22.5%), and patchy consolidation and ground glass opacity in 5/40 (12.5%). Late radiological changes were as follows: no changes in 6/40 cases (15%), scar-like pattern in 8/40 (20%), mass-like pattern in 10/40 (25%), not available in 6/40 cases (15%).
According to CATCE v4.0 criteria, acute and late clinical pulmonary toxicity ≥grade 2 were recorded in 2 out of 40 patients (5%) and 3 out of 40 patients (7%), respectively.
Mediastinal toxicity
In the acute setting, one single case of a grade 1 esophagitis was recorded. However, the patient required no specific medications. No patient experienced a cardiac toxicity. No narrowing or stenosis of any airway or vessel was registered. No other toxicities were registered in either the acute or the late setting.
Discussion
In the case of central lung malignancies, it is recognized that SBRT should be recommended with caution due to the high-risk tumor location and an increased risk of severe toxicities [19]. To date, the discussion concerning SBRT for centrally located malignancies remains controversial for several reasons: firstly, regarding the definition of centrally located tumors resulting from prospective studies [20, 21]; secondly, regarding the proper SBRT prescription dose to minimize the risk of treatment-related toxicity. Concerning the first issue, central tumor location is not evenly defined in literature. In fact, in the RTOG 0236 trial, central lesions were considered as malignancies within 2 cm of the proximal bronchial tree (described as the distal 2 cm of the trachea, carina, main bronchi) [22]. On the other hand, the IASLC defines central tumors as lesions within 2 cm of any mediastinal OAR (i. e., bronchial tree, esophagus, heart, major vessels, spinal cord, brachial plexus, phrenic and recurrent laryngeal nerve) [15]. Finally, the RTOG 0813 study defined central tumor location within 2 cm of the proximal bronchial tree or touching the mediastinal pleura [23].
In the present study, the definition of central lung malignancies referred to the IASLC recommendations [15]. Herein, we investigated the feasibility of a novel SBRT dose delivery technique by means of the so-called PTV-SIP approach. This approach is conceived with the intention to reduce the risk of high-grade toxicities without reducing the entire prescription dose to the PTV. To date, the SBRT prescription dose for centrally located tumors is mainly based on a risk-adapted dose prescription strategy with the intention of reducing the risk of severe adverse events [24]. In particular, a more fractionated prescription seems to guarantee an acceptable toxicity.
In a study by the VU University Medical Center [25], findings from the adoption of eight fractions of 7.5 Gy for centrally located tumors were reported. In their experience, six patients (10%) reported acute grade 2 toxicity and a one patient (2%) reported grade 3 acute toxicity. Regarding late toxicities, nine patients (14%) reported grade 2 toxicity, and four patients (6%) reported grade 3 toxicity. Similarly, Chang et al. evaluated a more fractionated SBRT schedule in a rather heterogeneous study population. They adopted a schedule of 70 Gy in 10 fractions for cases in which dose–volume constraints could not be met with the 50 Gy in 4 fractions regimen [9]. Central location was defined as tumors within 2 cm of the bronchial tree, major vessels, esophagus, heart, trachea, pericardium, brachial plexus, or vertebral body. In the study, no patient experienced grade 4 or 5 toxicity. Unfortunately, the publication does not indicate the difference in toxicity between 50 Gy in 4 fractions (82 patients) and 70 Gy in 10 fractions (18 patients).
In the present experience, acute and late clinical pulmonary toxicities ≥grade 2 were recorded in 2 out of 40 patients (5%) and 3 out of 40 patients (7%), respectively. Interestingly, mediastinal toxicity was negligible. In fact, in one single case, a grade 1 acute esophagitis was recorded. No patient experienced cardiac toxicity or stenosis of any airway or vessel. On the other hand, acceptable local control was achieved despite that fact that high priority was given to OAR protection instead of PTV coverage. In detail, the 1‑year local control rate was slightly higher than 90% and, thus, similar to literature data [10]. Thus, SBRT is potentially possible in central lung tumors, if well-reasoned dose prescriptions/OAR constraints/fractionation schedules are used that avoid higher toxicities.
In conclusion, in our experience, SBRT using a PTV-SIP approach for single central lung malignancies allowed a low SBRT-related toxicity with acceptable local control rates to be obtained.
References
Alongi F, Arcangeli S, Filippi AR, Ricardi U, Scorsetti M (2012) Review and uses of stereotactic body radiation therapy for oligometastases. Oncologist 17(8):1100–1107
Borm KJ, Oechsner M, Schiller K, Peeken JC, Dapper H, Münch S et al (2018) Prognostic factors in stereotactic body radiotherapy of lung metastases. Strahlenther Onkol. https://doi.org/10.1007/s00066-018-1335-x
Schanne DH, Nestle U, Allgäuer M, Andratschke N, Appold S, Dieckmann U et al (2015) Stereotactic body radiotherapy for centrally located stage I NSCLC: a multicenter analysis. Strahlenther Onkol 191(2):125–132
Mazzola R, Fersino S, Ferrera G, Targher G, Figlia V, Triggiani L et al (2018) Stereotactic body radiotherapy for lung oligometastases impacts on systemic treatment-free survival: a cohort study. Med Oncol 35(9):121
Timmerman R, McGarry R, Yiannoutsos C et al (2006) Excessive toxicity when treating central tumors in a phase II study of stereotactic body radiation therapy for medically inoperable early stage lung cancer. J Clin Oncol 24:4833–4839
Onishi H, Shirato H, Nagata Y, Hiraoka M, Fujino M, Gomi K et al (2011) Stereotactic body radiotherapy (SBRT) for operable stage I non-small-cell lung cancer: can SBRT be comparable to surgery? Int J Radiat Oncol Biol Phys 81(5):1352–1358
Figlia V, Mazzola R, Cuccia F, Alongi F, Mortellaro G, Cespuglio D et al (2018) Hypo-fractionated stereotactic radiation therapy for lung malignancies by means of helical tomotherapy: report of feasibility by a single-center experience. Radiol Med 123(6):406–414
Chaudhuri AA, Tang C, Binkley MS, Jin M, Wynne JF, von Eyben R et al (2015) Stereotactic ablative radiotherapy (SABR) for treatment of central and ultra-central lung tumors. Lung Cancer 89(1):50–56
Chang JY, Li QQ, Xu QY, Allen PK, Rebueno N, Gomez DR et al (2014) Stereotactic ablative radiation therapy for centrally located early stage or isolated parenchymal recurrences of non-small cell lung cancer: how to fly in a “no fly zone”. Int J Radiat Oncol Biol Phys 88(5):1120–1128
Alongi F, Mazzola R, Figlia V, Guckenberger M (2018) Stereotactic body radiotherapy for lung oligometastases: literature review according to PICO criteria. Tumori 104(3):148–156
De Bari B, Filippi AR, Mazzola R, Bonomo P, Trovò M, Livi L, Alongi F (2015) Available evidence on re-irradiation with stereotactic ablative radiotherapy following high-dose previous thoracic radiotherapy for lung malignancies. Cancer Treat Rev 41(6):511–518
Fleckenstein J, Boda-Heggemann J, Siebenlist K, Gudzheva T, Prakofyeva N, Lohr F et al (2018) Non-coplanar VMAT combined with non-uniform dose prescription markedly reduces lung dose in breath-hold lung SBRT. Strahlenther Onkol. https://doi.org/10.1007/s00066-018-1316-0
Brunner TB, Nestle U, Adebahr S, Gkika E, Wiehle R, Baltas D et al (2016) Simultaneous integrated protection: a new concept for high-precision radiation therapy. Strahlenther Onkol 192(12):886–894
Crane CH, Koay EJ (2016) Solutions that enable ablative radiotherapy for large liver tumors: fractionated dose painting, simultaneous integrated protection, motion management, and computed tomography image guidance. Cancer 122(13):1974–1986
Chang JY, Bezjak A, Mornex F, IASLC Advanced Radiation Technology Committee (2015) Stereotactic ablative radiotherapy for centrally located early stage non-small-cell lung cancer: what we have learned. J Thorac Oncol 10(4):577–585
www.nccn.org/professionals/physician_gls/pdf/nscl.pdf accessed on 1 July 2018
Grimm J et al (2011) Dose tolerance limits and dose volume histogram evaluation for stereotactic body radiotherapy. J Appl Clin Med Phys 12(2):267–292
Ikezoe J, Takashima S, Morimoto S, Kadowaki K, Takeuchi N, Yamamoto T et al (1988) CT appearance of acute radiation-induced injury in the lung. AJR Am J Roentgenol 150(4):765–770
Lo SS, Fakiris AJ, Chang EL, Mayr NA, Wang JZ, Papiez L et al (2010) Stereotactic body radiation therapy: a novel treatment modality. Nat Rev Clin Oncol 7(1):44–54
Roesch J, Panje C, Sterzing F, Mantel F, Nestle U, Andratschke N et al (2016) SBRT for centrally localized NSCLC—What is too central? Radiat Oncol 11(1):157
Timmerman R, Paulus R, Galvin J, Michalski J, Straube W, Bradley J et al (2010) Stereotactic body radiation therapy for inoperable early stage lung cancer. JAMA 303(11):1070–1076
Timmerman RD, Paulus R, Galvin J, Michalski J, Straube W, Bradley J et al (2009) Stereotactic body radiation therapy for medically inoperable early-stage lung cancer patients: analysis of RTOG 0236. Int J Radiat Oncol Biol Phys 75:S3
Bezjak A, Paulus R, Gaspar LE, Timmerman RD, Straube WL, Ryan WF et al (2016) Primary study endpoint analysis for NRG oncology/RTOG 0813 trial of Stereotactic Body Radiation Therapy (SBRT) for centrally located Non-Small Cell Lung Cancer (NSCLC). Int J Radiat Oncol Biol Phys 94:5–6
Li Q, Swanick CW, Allen PK, Gomez DR, Welsh JW, Liao Z et al (2014) Stereotactic ablative radiotherapy (SABR) using 70 Gy in 10 fractions for non-small cell lung cancer: exploration of clinical indications. Radiother Oncol 112(2):256–261
Haasbeek CJ, Lagerwaard FJ, Slotman BJ, Senan S (2011) Outcomes of stereotactic ablative radiotherapy for centrally located early-stage lung cancer. J Thorac Oncol 6(12):2036–2043
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
R. Mazzola, R. Ruggieri, V. Figlia, M. Rigo, N.G. Levra, F. Ricchetti, L. Nicosia, S. Corradini, and F. Alongi declare that they have no competing interests.
Ethical standards
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards. Informed consent was obtained from all individual participants included in the study.
Rights and permissions
About this article
Cite this article
Mazzola, R., Ruggieri, R., Figlia, V. et al. Stereotactic body radiotherapy of central lung malignancies using a simultaneous integrated protection approach. Strahlenther Onkol 195, 719–724 (2019). https://doi.org/10.1007/s00066-018-01419-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00066-018-01419-0