Abstract
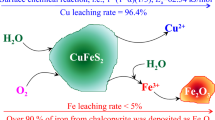
The literature on the ferric ion leaching of chalcopyrite has been surveyed to identify those leaching parameters which are well established and to outline areas requiring additional study. New experimental work was undertaken to resolve points still in dispute. It seems well established that chalcopyrite dissolution in either ferric chloride or ferric sulfate media is independent of stirring speeds above those necessary to suspend the particles and of acid concentrations above those required to keep iron in solution. The rates are faster in the chloride system and the activation energy in that medium is about 42 kJ/mol; the activation energy is about 75 kJ/mol in ferric sulfate solutions. It has been confirmed that the rate is directly proportional to the surface area of the chalcopyrite in both chloride and sulfate media. Sulfate concentrations, especially FeSO4 concentrations, decrease the leaching rate substantially; furthermore, CuSO4 does not promote leaching in the sulfate system. Chloride additions to sulfate solutions accelerate slightly the dissolution rates at elevated temperatures. It has been confirmed that leaching in the ferric sulfate system is nearly independent of the concentration of Fe3+, ka[Fe3+]0.12. In ferric chloride solutions, the ferric concentration dependence is greater and appears to be independent of temperature over the interval 45 to 100 °C.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
L. A. Haas and J. L. Shafer:J. Metals, 1979, vol. 31, no. 7, pp. 40–47.
H. W. Sheffer and L. G. Evans: USBM I.C.-8341, 1968.
F. Letowski, B. Kolodziej, M. Czernecki, A. Jedrczak and Z. Adamski:Hydromel., 1979, vol. 4, pp. 169–184.
L. J. Cabri:Econ. Geol., 1973, vol. 68, no. 4, pp. 443–54.
D. L. Jones and E. Peters:Extractive Metallurgy of Copper, J. C. Yannopoulos and J. C. Agarwal, eds., vol. 2, pp. 633–53, AIME, N.Y., 1976.
L. W. Beckstead, P. B. Munoz, J. L. Sepulveda, J. A. Herbst, J. D. Miller, F. A. Olson and M. E. Wadsworth:Extractive Metallurgy of Copper, vol. 2, J. C. Yannopoulos and J. C. Agarwal, eds., 1976 AIME, N.Y. pp. 611–32.
P. B. Munoz, J. D. Miller and M. E. Wadsworth:Metall. Trans. B, 1979, vol. 10B, pp. 149–58.
J. E. Dutrizac, R. J. C. MacDonald and T. R. Ingraham:Trans TMS-AIME, 1969, vol. 245, pp. 955–59.
J. E. Dutrizac:Metall. Trans. B, 1978, vol. 9B, pp. 431–39.
G. W. Warren: Ph.D. Thesis, University of Utah, 1978.
J. E. Dutrizac and R. J. C. MacDonald:Metall. Trans. B, 1971, vol. 2B, pp. 2310–12.
M. Ammou-Chokroum, D. Steinmetz, and A. Malve:Bull. Mineral (Fγ.), 1978, vol. 101, pp. 26–43.
J. E. Dutrizac and S. Kaiman:Can. Mineral., 1976, vol. 14, pp. 151–58.
J. E. Dutrizac and R. J. C. MacDonald:Can. Metal. Quart., 1973, vol. 12(4), pp. 409–20.
J. E. Dutrizac and R. J. C. MacDonald:Minerals Sci. Eng., 1974, vol. 6(2), pp. 3–100.
M. Ammou-Chokroum, M. Cambozoglu and D. Steinmetz:Bull. Soc. Mineral. Cristallogr., 1977, vol. 100, pp. 149–61.
D. F. Lowe: Ph.D. Thesis, University of Arizona, 1970.
J. P. Baur, H. L. Gibbs and M. E. Wadsworth: USBM RI-7823, 1974.
H. G. Linge:Hydrometallurgy, 1976, vol.2, pp. 51–64.
J. D. Sullivan:Trans. AIME, 1933, vol. 106, pp. 515–546.
V. V. Ermilov, O. B. Tkachenko and A. L. Tseft:Tr. Inst. Met., Obogashch, Alma Ata, 1979, vol. 30, pp. 3–14.
B. L. Tiwari, J. Kolbe and H. W. Hayden:Metall. Trans. B, 1980, vol. 11B, pp. 89–93.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Dutrizac, J.E. The dissolution of chalcopyrite in ferric sulfate and ferric chloride media. Metall Trans B 12, 371–378 (1981). https://doi.org/10.1007/BF02654471
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF02654471