Abstract
Chalcopyrite is one of the difficult-to-leach minerals as passive films of copper polysulphide form with mild leaching in sulfate media. Also, molten sulfur wets the mineral surface and can also inhibit leaching [1]. Chloride hydrometallurgy could be a viable option for chalcopyrite leaching as it has several advantages over sulfate systems and chalcopyrite does not passivate in chloride leach [2–6]. In this work, copper was extracted from chalcopyrite using a mixture of electrolytes containing relatively dilute HCl and nonassociated chloride salts such as NaCl, CaCl2, CuCl2, FeCl3, and MgCl2 rather than using pure HCl or pure binary electrolytes. This resulted in proton activities as high or higher than those obtained in pure concentrated HCl solutions.
Access provided by Autonomous University of Puebla. Download conference paper PDF
Similar content being viewed by others
Keywords
- Mineral processing
- Hydrometallurgy
- Chalcopyrite ore
- Copper
- Iron disposal
- Hematite
- Chloride media
- High pressure
- High temperature
- Autoclave
1 Introduction
Chalcopyrite is one of the difficult-to-leach minerals as passive films of copper polysulphide form with mild leaching in sulfate media. Also, molten sulfur wets the mineral surface and can also inhibit leaching [1]. Chloride hydrometallurgy could be a viable option for chalcopyrite leaching as it has several advantages over sulfate systems and chalcopyrite does not passivate in chloride leach [2,3,4,5,6]. In this work, copper was extracted from chalcopyrite using a mixture of electrolytes containing relatively dilute HCl and nonassociated chloride salts such as NaCl, CaCl2, CuCl2, FeCl3, and MgCl2 rather than using pure HCl or pure binary electrolytes. This resulted in proton activities as high or higher than those obtained in pure concentrated HCl solutions.
The basic approach of this process was to leach copper from chalcopyrite concentrate in a strong brine solution containing copper and iron chloride salts. As shown in Fig. 1, after leaching in a 4-stage countercurrent cascade, free acid in the pregnant leach solution (PLS) was neutralized with limestone addition to form gypsum followed by solvent extraction (SX) of copper using a combined oxidation/extraction process. Copper was stripped from loaded organic using a sulfate-based spent electrolyte from the copper electrowinning circuit. The conventional deposition of copper cathode on stainless steel was performed. Iron was removed from the SX raffinate by oxidation and hydrolytic precipitation at high temperatures before recycling the raffinate back to leaching.
This paper discussed the conversion of iron in an autoclave where one third of the ferrous iron in the effluent was rejected as hematite for safe disposal and the rest was converted to ferric ions in the solution that was recycled back to the leaching circuit. A suitable condition for hematite formation was found to be 175 °C and 40 psig oxygen overpressure.
2 Iron Removal as Hematite
The copper raffinate solution contains predominantly cupric and ferrous ions. Autoclave oxidation was performed to oxidize the ferrous ion to ferric ion and by hydrolysis and precipitation as hematite, which is the preferred option to dispose of iron from the environmental point of view. As shown in Table 1, all residual iron in the product solution remained as ferric ions. Negligible losses of copper with hematite as coprecipitate were observed.
The best conditions for maximum amounts (58 g) of hematite rejection from 1 L brine solution containing about 116 g/L iron (Fe2+) and 40 g/L copper (Cu2+) are: 175 °C, 40 psig O2 overpressure and ½-1 h agitation at 800 rpm.
Ferrous oxidation reactions are given in Eqs. 1–3.
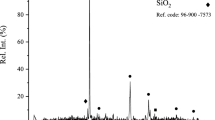
One third of ferrous in the autoclave feed goes for hematite formation and two third is converted to ferric ions. Since a greater amount of ferric ions is recycled back to the leaching circuit, it is assumed that the oxidation of pyrite and sulfur is due to FeCl3 rather than oxygen. XRD data confirmed that iron was precipitated as hematite. The kinetics of the oxyhydrolysis were fast with highly crystalline, filterable hematite as the reaction product. Figure 2 shows the oxygen consumption against time which suggests that the reaction is completed within 0.5 h.
References
Dreisinger D (2006) New developments in Cu and Ni hydrometallurgy. Presentation to JOGMEC (Japan)
Winand R (1991) Chloride hydrometallurgy. Hydrometallurgy 27:285–316
Dutrizac JE (1992) The leaching of sulphide minerals in chloride media. Hydrometallurgy 29:1–45
Lu J, Dreisinger D (2013) Pressure oxidation of ferrous ions by oxygen and hematite precipitation from concentrated solution of calcium, copper and iron chlorides. Hydrometallurgy 140:59–65
Lu J, Dreisinger D (2013) Copper chloride leaching from chalcopyrite and bornite concentrates containing high levels of impurities and minor elements. Hydrometallurgy 138:40–47
Lu J, Dreisinger D (2013) Copper leaching from chalcopyrite concentrate in Cu(II)/Fe(III) chloride system. Miner Eng 45:185–190
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2023 The Author(s), under exclusive license to Springer Nature Switzerland AG
About this paper
Cite this paper
Alam, S., Dreisinger, D. (2023). Iron Rejection as Hematite in an Autoclave During Processing of Chalcopyrite in Chloride Media. In: Proceedings of the 62nd Conference of Metallurgists, COM 2023. COM 2023. Springer, Cham. https://doi.org/10.1007/978-3-031-38141-6_23
Download citation
DOI: https://doi.org/10.1007/978-3-031-38141-6_23
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-031-38140-9
Online ISBN: 978-3-031-38141-6
eBook Packages: Chemistry and Materials ScienceChemistry and Material Science (R0)