Abstract
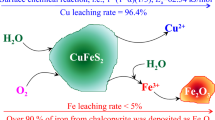
The acid ferric sulfate leaching of chalcopyrite, CuFeS2 + 4Fe+3 = Cu+2 + 5Fe+2 + 2S0 was studied using monosize particles in a well stirred reactor at ambient pressure and dilute solid phase concentration in order to obtain fundamental details of the reaction kinetics. The principal rate limiting step for this electrochemical reaction appears to be a transport process through the elemental sulfur reaction product. This conclusion has been reached in other investigations and is supported by data from this investigation in which the reaction rate was found to have an inverse second order dependence on the initial particle diameter. Furthermore, the reaction kinetics were found to be independent of Fe+3, Fe+2, Cu+2 and H2SO4 in the range of additions studied. The unique aspect of this particular research effort is that data analysis, using the Wagner theory of oxidation, suggests that the rate limiting process may be the transport of electrons through the elemental sulfur layer. Predicted reaction rates calculated from first principles using the physicochemical properties of the system (conductivity of elemental sulfur and the free energy change for the reaction) agree satisfactorily with experimentally determined rates. Further evidence which supports this analysis includes an experimental activation energy of 20 kcal/mol (83.7 kJ/mol) which is approximately the same as the apparent activation energy for the transfer of electrons through elemental sulfur, 23 kcal/ mol (96.3 kJ/mol) calculated from both conductivity and electron mobility measurements reported in the literature.
Article PDF
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
References
R. J. Roman, and B. R. Benner:Minerals Sci. Eng., 1973, vol. 5, no. l,pp. 3–24.
J. Dasher:CIM Trans., 1973, vol. 76, pp. 80–88.
D. F. Lowe: PhD. thesis, University of Arizona, Tucson, Ariz., 1970.
H. G. Linge:Hydrometallurgy, 1976, vol. 2, pp. 51–64.See also H. G. Linge:Hydrometallurgy, 1976, vol. 2, pp. 219-33.
J. E. Dutrizac, R. J. C. MacDonald and T. R. Ingraham:Trans. TMS-AIME, 1969, vol. 245, pp. 955–59.
L. W. Beckstead, P. B. Munoz, J. D. Miller, J. A. Herbst, F. A. Olson, and M. E. Wadsworth:Extractive Metallurgy of Copper II, pp. 611–32, The Metal- lurgical Society of AIME, New York, 1976.
J. D. Sullivan:Trans. AIME, 1933, vol. 106, pp. 515–46.
D. L. Jones, and E. Peters:Extractive Metallurgy of Copper II, pp. 633-53, The Metallurgical Society of AIME, 1976.
G. W. Warren: Ph.D. thesis, Department of Metallurgy and Metallurgical Engineering, University of Utah, Salt Lake City, U., 1978.
J. P. Baur, H. L. Gibbs, and M. E. Wadsworth: USBM RI 7823, 1974.
E. Peters:AIME Internat. Symp. on Hydromet, pp. 205–28, AIME, N.Y., 1973.
L. W. Beckstead and J. D. Miller:Met. Trans. B, 1977, vol. 8B, pp. 19–38.
P. B. Munoz: Ph.D. thesis, Department of Mining, Metallurgical and Fuels Engineering, University of Utah, Salt Lake City, U., 1977.
W. C. Pierce:Quantitative Analysis, pp. 312–15, John Wiley and Sons, New York, 1958.
P. Kofstad:High Temperature Oxidation of Metals, John Wiley and Sons, New York, 1966.
E. Peters: “Hydrometallurgy: Theory and Practice,”First Tutorial Symposium on Hydrometallurgy, AIME, Denver, Co., 1972.
R. Garrelsand and C. Christ:Solutions Minerals and Equilibria, Freeman, Cooper Company, San Francisco, 1965.
W. N. Tuller:The Sulfur Data Book, p. 59, McGraw-Hill Book Company, Inc., 1954.
A. R. Adams and W. E. Spear:J. Phys. Chem. Solids, 1964, vol. 25, pp.1113–18.
D. J. Gibbons and W. E. Spear:J. Phys. Chem. Solids, 1966, vol. 27, pp. 1917–25.
Author information
Authors and Affiliations
Additional information
formerly Metallurgy Graduate Student, University of Utah.
Rights and permissions
About this article
Cite this article
Munoz, P.B., Miller, J.D. & Wadsworth, M.E. Reaction mechanism for the acid ferric sulfate leaching of chalcopyrite. Metall Trans B 10, 149–158 (1979). https://doi.org/10.1007/BF02652458
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF02652458