Summary
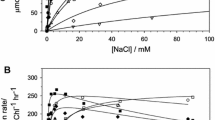
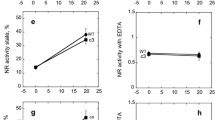
The NADH-nitrate oxidoreductase of Chlorella vulgaris has an inactive form which has previously been shown to be a cyanide complex of the reduced enzyme. This inactive enzyme can be reactivated by treatment with ferricyanide in vitro. In the present study, the activation state of the enzyme was determined after different prior in vivo programs involving environmental variations. Oxygen, nitrate, light and CO2 all affect the in vivo inactivation of the enzyme in an interdependent manner. In general, the inactivation is stimulated by O2 and inhibited by nitrate and CO2. Light may stimulate or inhibit, depending on conditions. Thus, the effects of CO2 and nitrate (inhibition of reversible inactivation) are clearly manifested only in the light. In contrast, light stimulates the inactivation in the presence of oxygen and the absence of CO2 and nitrate. Since the inactivation of the enzyme requires HCN and NADH, and it is improbable that O2 stimulates NADH formation, it is reasonable to conclude that HCN is formed as the result of an oxidation reaction (which is stimulated by light). The formation of HCN is probably stimulated by Mn2+, since the formation of reversibly-inactivated enzyme is impaired in Mn2+-deficient cells. The prevention of enzyme inactivation by nitrate in vivo is in keeping with previous in vitro results showing that nitrate prevents inactivation by maintaining the enzyme in the oxidized form. A stimulation of nitrate uptake by CO2 and light could account for the effect of CO2 (prevention of inactivation) which is seen mainly in the presence of nitrate and light. Ammonia added in the presence of nitrate has the same effect on the enzyme as removing nitrate (promotion of reversible inactivation). Ammonia added in the absence of nitrate has little extra effect. It is therefore likely that ammonia acts by preventing nitrate uptake. The uncoupler, carbonylcyanide-m-chloro-phenylhydrazone, causes enzyme inactivation because it acts as a good HCN precursor, particularly in the light. Nitrite, arsenate and dinitrophenol cause an enzyme inactivation which can not be reversed by ferricyanide in crude extracts. This suggests that there are at least two different ways in which the enzyme can be inactivated rather rapidly in vivo.
Article PDF
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
References
Gewitz, H.-S., Lorimer, G.H., Solomonson, L.P., Vennesland, B.: Presence of HCN inChlorella vulgaris and its possible role in controlling the reduction of nitrate. Nature 249, 79–81 (1974)
Heimer, Y.M.: Nitrate reductase of Dunaliella parva. Electron donor specificity and heat activation. Arch. Microbiol. 103, 181–183 (1975)
Herrera, J., Paneque, A., Maldonado, J.M., Barea, J.L., Losada, M.: Regulation by ammonia of nitrate reductase synthesis and activity in Chlamydomonas reinhardi. Biochem. biophys. Res. Commun. 48, 996–1003 (1972)
Jetschmann, K., Solomonson, L.P., Vennesland, B.: Activation of nitrate reductase by oxidation. Biochim. biophys. Acta (Amst.) 275, 276–278 (1972)
Lorimer, G.H., Gewitz, H.-S., Völker, W., Solomonson, L.P., Vennesland, B.: The presence of bound cyanide in the naturally inactivated form of nitrate reductase of Chlorella vulgaris. J. biol. Chem. 249, 6074–6079 (1974)
Losada, M., Herrera, J., Maldonado, J.M., Paneque, A.: Mechanism of nitrate reductase reversible inactivation by ammonia in Chlamydomonas. Plant Sci. Letters 1, 31–37 (1973)
Losada, M., Paneque, A., Aparicio, P.J., Vega, J.M., Cárdenas, J., Herrera, J.: Inactivation and repression by ammonium of the nitrate reducing system in Chlorella. Biochem. biophys. Res. Commun. 38, 1009–1015 (1970)
Maldonado, J.M., Herrera, J., Paneque, A., Losada, M.: Reversible inactivation by NADH and ADP of Chlorella fusca nitrate reductase. Biochem. biophys. Res. Commun. 51, 27–33 (1973)
Moreno, C.G., Aparicio, P.J., Palacián, E., Losada, M.: Interconversions of the active and inactive forms of Chlorella nitrate reductase. FEBS Letters 26, 11–14 (1972)
Nicholas, D.J.D., Nason, A.: Nitrite determination by diazotization and coupling reactions. In: Methods in Enzymology. Vol. III, pp. 983–984. Eds.: Colowick, S.P. and Kaplan, N.O., New York: Academic Press 1957
Payne, W.J.: Reduction of nitrogenous oxides by microorganisms. Bact. Rev. 37, 409–452 (1973)
Pistorius, E.K., Gewitz, H.-S., Voss, H., Vennesland, B.: Cyanide generation from carbonylcyanide. m-chlorophenyl-hydrazone. FEBS Letters (in press)
Rigano, C., Aliotta, G., Violante, U.: Reversible inactivation by ammonia of assimilatory nitrate reductase in Cyanidium caldarium. Arch. Microbiol. 99, 81–90 (1974)
Rigano, C., Violante, U.: Effect of heat treatment on the activity in vitro of nitrate reductase from Cyanidium caldarium. Biochim. biophys. Acta (Amst.) 256, 524–532 (1972)
Schloemer, R.H., Garrett, R.H.: Nitrate transport system in Neurospora crassa. J. Bact. 118, 259–269 (1974)
Solomonson, L.P.: Regulation of nitrate reductase activity by NADH and cyanide. Biochim. biophys. Acta (Amst.) 334, 297–308 (1974)
Solomonson, L.P., Jetschmann, K., Vennesland, B.: Reversible inactivation of the nitrate reductase of Chlorella vulgaris Beijerinck. Biochim. biophys. Acta (Amst.) 309, 32–43 (1973)
Solomonson, L.P., Lorimer, G.H., Hall, R.L., Borchers, R., Bailey, J.L.: Reduced nicotinamide adenine dinucleotide-nitrate reductase of Chlorella vulgaris: Purification, prosthetic groups and molecular properties. J. biol. Chem. 250, 4120–4127 (1975)
Solomonson, L.P., Vennesland, B.: Properties of nitrate reductase of Chlorella. Biochim biophys. Acta (Amst.) 267, 544–557 (1972a)
Solomonson, L.P., Vennesland, B.: Nitrate reductase and chlorate toxicity in Chlorella vulgaris Beijerinck. Plant Physiol. 50, 421–424 (1972b)
Vennesland, B., Jetschmann, C.: The nitrate reductase of Chlorella vulgaris. Biochim. biophys. Acta (Amst.) 227, 554–564 (1971)
Vennesland, B., Jetschmann, K.: The effect of cyanide and some other carbonyl-binding reagents on glycolate excretion by Chlorella vulgaris. Planta (Berl.) 128, 81–84 (1976)
Warburg, O., Krippahl, G.: Glykolsäurebildung in Chlorella. Z. Naturforsch. 15b, 197–199 (1960)
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Pistorius, E.K., Gewitz, HS., Voss, H. et al. Reversible inactivation of nitrate reductase in Chlorella vulgaris in vivo . Planta 128, 73–80 (1976). https://doi.org/10.1007/BF00397181
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00397181