Abstract
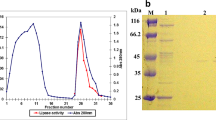
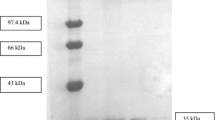
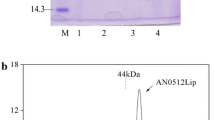
An extracellular lipase from Aureobasidium pullulans was obtained and purified with a specific activity of 17.7 U/mg of protein using ultrafiltration and a DEAE-Sepharose Fast Flow column. Characterization of the lipase indicated that it is a novel finding from the species A. pullulans. The molecular weight of the lipase was 39.5 kDa, determined by sodium dodecyl sulfonate-polyacrylamide gel electrophoresis (SDS-PAGE). The enzyme exhibited its optimum activity at 40 °C and pH of 7. It also showed a remarkable stability in some organic solutions (30%, v/v) including n-propanol, isopropanol, dimethyl sulfoxide (DMSO), and hexane. The catalytic activity of the lipase was enhanced by Ca2+ and was slightly inhibited by Mn2+ and Zn2+ at a concentration of 10 mmol/L. The lipase was activated by the anionic surfactant SDS and the non-ionic surfactants Tween 20, Tween 80, and Triton X-100, but it was drastically inhibited by the cationic surfactant cetyl trimethyl ammonium bromide (CTAB). Furthermore, the lipase was able to hydrolyze a wide variety of edible oils, such as peanut oil, corn oil, sunflower seed oil, sesame oil, and olive oil. Our study indicated that the lipase we obtained is a potential biocatalyst for industrial use.
概 要
目 的
从出芽短梗霉所产的脂肪酶中筛选具有独特酶学 性质的脂肪酶
创新点
发现了一种新的产自出芽短梗霉的脂肪酶, 并对 其酶学性质进行了研究。
方 法
通过超滤和 DEAE-Sepharose Fast Flow 阴离子层 析柱方法对脂肪酶进行纯化, 随后分别用对硝基 酚邻酸盐(pNPP) 法对纯化得到的脂肪酶进行了 酶学性质研究, 并用酸碱中和法检测了脂肪酶对 可食用油脂的水解。
结 论
对分离纯化得到的脂肪酶的酶学性质研究表明, 该酶的分子量为39.5 kDa, 具有一个亚基, 为胞外酶。最佳催化温度为40 °C, 最佳催化pH 为7。该酶对一些有机溶剂、表面活性剂和离子具有优良的抗性。此外, 它可以水解常见的食用油。这些良好的特性使该脂肪酶有可能被应用于洗涤剂生产、生物柴油合成和食品制造等一些工业领域。
Article PDF
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
References
Antonov VK, Dyakov VL, Mishin AA, et al., 1988. Catalytic activity and association of pancreatic lipase. Biochimie, 70(9):1235–1244. https://doi.org/10.1016/0300-9084(88)90190-3
Aouf C, Durand E, Lecomte J, et al., 2014. The use of lipases as biocatalysts for the epoxidation of fatty acids and phenolic compounds. Green Chem, 16(4):1740–1754. https://doi.org/10.1039/C3GC42143K
Aref HL, Mosbah H, Fekih A, et al., 2014. Purification and biochemical characterization of lipase from Tunisian Euphorbia peplus latex. J Am Oil Chem Soc, 91(6): 943–951. https://doi.org/10.1007/s11746-014-2444-z
Bradford MM, Williams WL, 1976. New, rapid, sensitive method for protein determination. Fed Proc, 35(3):274.
Chi ZM, Wang F, Chi Z, et al., 2009. Bioproducts from Aureobasidium pullulans, a biotechnologically important yeast. Appl Microbiol Biotechnol, 82(5):793–804. https://doi.org/10.1007/s00253-009-1882-2
de Hoog GS, 1993. Evolution of black yeasts: possible adaptation to the human host. Antonie van Leeuwenhoek, 63(2):105–109. https://doi.org/10.1007/BF00872386
Ebrahimpour A, Rahman RNZRA, Basri M, et al., 2011. High level expression and characterization of a novel thermostable, organic solvent tolerant, 1,3-regioselective lipase from Geobacillus sp. strain ARM. Bioresour Technol, 102(13):6972–6981. https://doi.org/10.1016/j.biortech.2011.03.083
Fauzi AHM, Amin NAS, 2012. An overview of ionic liquids as solvents in biodiesel synthesis. Renewable Sustainable Energy Rev, 16(8):5770–5786. https://doi.org/10.1016/j.rser.2012.06.022
Federici F, 1982. Extracellular enzymatic activities in Aureo-basidium pullulans. Mycologia, 74(5):738–743. https://doi.org/10.1080/00275514.1982.12021580
Ghasemi Y, Rasoul-Amini S, Kazemi A, et al., 2011. Isolation and characterization of some moderately halophilic bacteria with lipase activity. Microbiology, 80(4):483–487. https://doi.org/10.1134/S0026261711040060
Hamdy HS, Abo-Tahon MA, 2012. Extracellular lipase of Aspergillus terreus var. africanus (CBS 130.55): production, purification and characterisation. Ann Microbiol, 62(4): 1723–1736. https://doi.org/10.1007/s13213-012-0429-4
Hasan F, Shah AA, Hameed A, 2006. Industrial applications of microbial lipases. Enzyme Microb Technol, 39(2):235–251. https://doi.org/10.1016/j.enzmictec.2005.10.016
Hasan F, Shah AA, Hameed A, 2009. Methods for detection and characterization of lipases: a comprehensive review. Biotechnol Adv, 27(6):782–798. https://doi.org/10.1016/j.biotechadv.2009.06.001
Hiol A, Jonzo MD, Rugani N, et al., 2000. Purification and characterization of an extracellular lipase from a thermophilic Rhizopus oryzae strain isolated from palm fruit. Enzyme Microb Technol, 26(5–6):421–430. https://doi.org/10.1016/S0141-0229(99)00173-8
Ji QC, Xiao SJ, He BF, et al., 2010. Purification and characterization of an organic solvent-tolerant lipase from Pseudomonas aeruginosa LX1 and its application for biodiesel production. J Mol Catal B Enzym, 66(3–4):264–269. https://doi.org/10.1016/j.molcatb.2010.06.001
Joseph B, Ramteke PW, 2013. Extracellular solvent stable cold-active lipase from psychrotrophic Bacillus sphaericus MTCC 7526: partial purification and characterization. Ann Microbiol, 63(1):363–370. https://doi.org/10.1007/s13213-012-0483-y
Kanjanavas P, Khuchareontaworn S, Khawsak P, et al., 2010. Purification and characterization of organic solvent and detergent tolerant lipase from thermotolerant Bacillus sp. RN2. Int J Mol Sci, 11(10):3783–3792. https://doi.org/10.3390/ijms11103783
Kudanga T, Mwenje E, Mandivenga F, et al., 2007. Esterases and putative lipases from tropical isolates of Aureobasidium pullulans. J Basic Microbiol, 47(2):138–147. https://doi.org/10.1002/jobm.200610207
Laemmli UK, 1970. Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature, 227(5259):680–685. https://doi.org/10.1038/227680a0
Lailaja VP, Chandrasekaran M, 2013. Detergent compatible alkaline lipase produced by marine Bacillus smithii BTMS 11. World J Microbiol Biotechnol, 29(8):1349–1360. https://doi.org/10.1007/s11274-013-1298-0
Leathers TD, Rich JO, Anderson AM, et al., 2013. Lipase production by diverse phylogenetic clades of Aureobasidium pullulans. Biotechnol Lett, 35(10):1701–1706. https://doi.org/10.1007/s10529-013-1268-5
Li M, Yang LR, Xu G, et al., 2013. Screening, purification and characterization of a novel cold-active and organic solvent-tolerant lipase from Stenotrophomonas maltophilia CGMCC 42542013. Bioresour Technol, 148:114–120. https://doi.org/10.1016/j.biortech.2013.08.101
Li X, Qian P, Wu SG, et al., 2014. Characterization of an organic solvent-tolerant lipase from Idiomarina sp. W33 and its application for biodiesel production using Jatropha oil. Extremophiles, 18(1):171–178. https://doi.org/10.1007/s00792-013-0610-0
Li Y, Cai HY, Zhao MJ, et al., 2015. Screening and identification of high-yield thermostable lipase producing microorganisms. Biotechnol Bull, 31(1): 144–150 (in Chinese). https://doi.org/10.13560/j.cnki.biotech.bull.1985.2015.01.022
Liu ZQ, Li XY, Chi ZM, et al., 2008a. Cloning, characterization and expression of the extracellular lipase gene from Aureobasidium pullulans HN2-3 isolated from sea saltern. Antonie Van Leeuwenhoek, 94(2):245–255. https://doi.org/10.1007/s10482-008-9237-z
Liu ZQ, Chi ZM, Wang L, et al., 2008b. Production, purification and characterization of an extracellular lipase from Aureobasidium pullulans HN2.3 with potential application for the hydrolysis of edible oils. Biochem Eng J, 40(3):445–451. https://doi.org/10.1016/j.bej.2008.01.014
Martinelle M, Holmquist M, Hult K, 1995. On the interfacial activation of Candida antarctica lipase A and B as compared with Humicola lanuginosa lipase. Biochim Biophys Acta, 1258(3):272–276. https://doi.org/10.1016/0005-2760(95)00131-U
Mogensen JE, Sehgal P, Otzen DE, 2005. Activation, inhibition, and destabilization of Thermomyces lanuginosus lipase by detergents. Biochemistry, 44(5):1719–1730. https://doi.org/10.1021/bi0479757
Peng Q, Wang X, Shang M, et al., 2014. Isolation of a novel alkaline-stable lipase from a metagenomic library and its specific application for milkfat flavor production. Microb Cell Fact, 13:1. https://doi.org/10.1186/1475-2859-13-1
Rahman RNZRA, Baharum SN, Basri M, et al., 2005. High-yield purification of an organic solvent-tolerant lipase from Pseudomonas sp. strain S5. Anal Biochem, 341(2): 267–274. https://doi.org/10.1016/j.ab.2005.03.006
Ramakrishnan V, Goveas LC, Suralikerimath N, et al., 2016. Extraction and purification of lipase from Enterococcus faecium MTCC5695 by PEG/phosphate aqueous-two phase system (ATPS) and its biochemical characterization. Biocatal Agric Biotechnol, 6:19–27. https://doi.org/10.1016/j.bcab.2016.02.005
Ramani K, Kennedy LJ, Ramakrishnan M, et al., 2010. Purification, characterization and application of acidic lipase from Pseudomonas gessardii using beef tallow as a substrate for fats and oil hydrolysis. Process Biochem, 45(10):1683–1691. https://doi.org/10.1016/j.procbio.2010.06.023
Ramírez-Zavala B, Mercado-Flores Y, Hernández-Rodríguez C, et al., 2004. Purification and characterization of a lysine aminopeptidase from Kluyveromyces marxianus. FEMS Microbiol Lett, 235(2):369–375. https://doi.org/10.1111/j.1574-6968.2004.tb09612.x
Rúa ML, Díaz-Mauriñob T, Fernández VM, et al., 1993. Purification and characterization of two distinct lipases from Candida cylindracea. Biochim Biophys Acta, 1156(2): 181–189. https://doi.org/10.1016/0304-4165(93)90134-T
Salihu A, Alam MZ, 2015. Solvent tolerant lipases: a review. Process Biochem, 50(1):86–96. https://doi.org/10.1016/j.procbio.2014.10.019
Saxena RK, Sheoran A, Giri B, et al., 2003. Purification strategies for microbial lipases. J Microbiol Methods, 52(1):1–18. https://doi.org/10.1016/S0167-7012(02)00161-6
Sharma R, Chisti Y, Banerjee UC, 2001. Production, purification, characterization, and applications of lipases. Biotechnol Adv, 19(8):627–662. https://doi.org/10.1016/S0734-9750(01)00086-6
Shi H, Meng Y, Yang M, et al., 2014. Purification and characterization of a hydrolysis-resistant lipase from Aspergillus terreus. Biotechnol Appl Biochem, 61(2): 165–174. https://doi.org/10.1002/bab.1142
Singh RS, Saini GK, 2008. Pullulan-hyperproducing color variant strain of Aureobasidium pullulans FB-1 newly isolated from phylloplane of Ficus sp. Bioresour Technol, 99(9):3896–3899. https://doi.org/10.1016/j.biortech.2007.08.003
Sulong MR, Rahnian RNZRA, Salleh AB, et al., 2006. A novel organic solvent tolerant lipase from Bacillus sphaericus 205y: extracellular expression of a novel OST-lipase gene. Protein Express Purif, 49(2):190–195. https://doi.org/10.1016/j.pep.2006.04.015
Taher H, Al-Zuhair S, 2017. The use of alternative solvents in enzymatic biodiesel production: a review. Biofuels Bioprod Biorefin, 11(1):168–194. https://doi.org/10.1002/bbb.1727
Tang LL, Xia YL, Wu XL, et al., 2017. Screening and characterization of a novel thermostable lipase with detergent-additive potential from the metagenomic library of a mangrove soil. Gene, 625:64–71. https://doi.org/10.1016/j.gene.2017.04.046
Tomke PD, Rathod VK, 2015. Ultrasound assisted lipase catalyzed synthesis of cinnamyl acetate via transesterification reaction in a solvent free medium. Ultrason Sonochem, 27:241–246. https://doi.org/10.1016/j.ultsonch.2015.04.022
Ugur A, Sarac N, Boran R, et al., 2014. New lipase for biodiesel production: partial purification and characterization of LipSB 25-4. ISRN Biochem, 2014:289749. https://doi.org/10.1155/2014/289749
Wang L, Chi ZM, Wang XH, et al., 2007. Diversity of lipase-producing yeasts from marine environments and oil hydrolysis by their crude enzymes. Ann Microbiol, 57(4): 495–501. https://doi.org/10.1007/BF03175345
Winkler UK, Stuckmann M, 1979. Glycogen, hyaluronate, and some other polysaccharides greatly enhance the formation of exolipase by Serratia marcescens. J Bacteriol, 138(3):663–670.
Yang L, Dordick JS, Garde S, 2004. Hydration of enzyme in nonaqueous media is consistent with solvent dependence of its activity. Biophys J, 87(2):812–821. https://doi.org/10.1529/biophysj.104.041269
Yuan DJ, Lan DM, Xin RP, et al., 2016. Screening and characterization of a thermostable lipase from marine Streptomyces sp. strain W007. Biotechnol Appl Biochem, 63(1): 41–50. https://doi.org/10.1002/bab.1338
Yuan L, Sadiq FA, Liu TJ, et al., 2018. Spoilage potential of psychrotrophic bacteria isolated from raw milk and the thermo-stability of their enzymes. J Zhejiang Univ-Sci B (Biomed & Biotechnol), 19(8):630–642. https://doi.org/10.1631/jzus.B1700352
Author information
Authors and Affiliations
Corresponding author
Additional information
Project supported by the Science & Technology Major Project of Zhejiang Province, China (No. 2012C12005-2)
Rights and permissions
About this article
Cite this article
Li, Y., Liu, Tj., Zhao, Mj. et al. Screening, purification, and characterization of an extracellular lipase from Aureobasidium pullulans isolated from stuffed buns steamers. J. Zhejiang Univ. Sci. B 20, 332–342 (2019). https://doi.org/10.1631/jzus.B1800213
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1631/jzus.B1800213