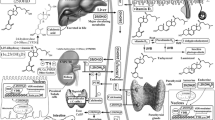
Abstract
During evolution, the ability of many organisms to synthesize vitamin D photochemically represented, and still represents, a major driving factor for the development of life on earth. In humans because not more than 10–20% of the requirement of vitamin D can be satisfied by the diet (under most living conditions in the US and Europe), the remaining 80–90% need to be photochemically synthesized in the skin through the action of solar or artificial ultraviolet-B (UV-B) radiation. The skin is a key organ of the human body’s vitamin D endocrine system (VDES), representing both the site of vitamin D synthesis and a target tissue for biologically active vitamin D metabolites. Human keratinocytes contain the enzymatic machinery (CYP27B1) for the synthesis of the biologically most active natural vitamin D metabolite 1,25-dihydroxyvitamin D3 (1,25(OH)2D3), representing an autonomous vitamin D3 pathway. Cutaneous production of 1,25(OH)2D3 may mediate intracrine, autocrine and paracrine effects on keratinocytes and on neighboring cells. Many skin cells (including keratinocytes, sebocytes, fibroblasts, melanocytes, macrophages and other skin immune cells) express the vitamin D receptor (VDR), an absolute pre-requisite for exerting genomic effects of 1,25(OH)2D3 and analogs. The VDR is a member of the superfamily of trans-acting transcriptional regulatory factors, which also contains the steroid and thyroid hormone receptors as well as the retinoid-X receptors (RXR) and retinoic acid receptors (RAR). A large body of evidence, including cDNA microarray analyses of mRNAs, indicates that as many as 500–1000 genes may be controlled by VDR ligands that regulate a broad variety of cellular functions including growth, differentiation, and apoptosis. Clinical and laboratory investigations, including the observation that 1,25(OH)2D3 is very effective in inducing the terminal differentiation and in inhibiting the proliferation of cultured human keratinocytes have resulted in the use of 1,25(OH)2D3 and analogs for the treatment of psoriasis. Focussing on the UV-induced cutaneous synthesis of vitamin D, this review gives an update on the relevance of the VDES and of UV radiation for the management of psoriasis and other inflammatory skin diseases.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
J. Reichrath, C. C. Zouboulis, T. Vogt, and M. F. Holick, Targeting the vitamin D endocrine system (VDES) for the management of inflammatory and malignant skin diseases: An historical view and outlook, Rev. Endocr. Metab. Disord., 2016, 17, 3 405–417
M. F. Holick, Vitamin D deficiency, N. Engl. J. Med., 2007, 357, 266–281
J. Reichrath, Vitamin D and the skin: an ancient friend, revisited, Exp. Dermatol., 2007, 16, 618–625
R. Saternus, S. Pilz, S. Gräber, M. Kleber, W. März, T. Vogt, and J. Reichrath, A Closer Look at Evolution: Variants (SNPs) of Genes Involved in Skin Pigmentation, Including EXOC2, TYR, TYRP1, and DCT, Are Associated With 25(OH)D Serum Concentration, Endocrinology, 2015, 156, 39–47
W. Rossberg, R. Saternus, S. Wagenpfeil, M. Kleber, W. März, Th. Vogt, and J. Reichrath, Skin pigmentation, cutaneous vitamin D synthesis and evolution: variants of genes (SNPs) involved in skin pigmentation are associated with 25(OH)D serum concentration, Anticancer Res., 2016, 36, 1429–1438
K. Matsumoto, Y. Azuma, M. Kiyoki, H. Okumura, K. Hashimoto, and K. Yoshikawa, Involvement of endogenously produced 1,25-dihydroxyvitamin D-3 in the growth and differentiation of human keratinocytes, Biochim. Biophys. Acta, 1991, 1092, 311–318
J. H. Prystowsky, P. J. Muzio, S. Sevran, and T. L. Clemens, Effect of UVB phototherapy and oral calcitriol (1,25-dihydroxyvitamin D3) on vitamin D photosynthesis in patients with psoriasis, J. Am. Acad. Dermatol., 1996, 35, 690–695
B. Lehmann, T. Genehr, P. Knuschke, J. Pietzsch and M. Meurer, UVB-induced conversion of 7-dehydrocholesterol to 1α,25-dihydroxyvitamin D3 in an in vitro human skin equivalent model, J. Invest. Dermatol., 2001, 117, 1179–1185
B. Lehmann, W. Sauter, P. Knuschke, S. Dreßler, and M. Meurer, Demonstration of UVB-induced synthesis of 1α,25-dihydroxyvitamin D3 (calcitriol) in human skin by microdialysis, Arch. Dermatol. Res., 2003, 295, 24–28
K. Vatieghem, P. Dehaes, R. Bouillon and S. Segaert, Cultured fibroblasts produce non-active vitamin D metabolites that can be activated by cultured keratinocytes, in Abstracts Twelfth Workshop on Vitamin D, 2003, p. 27
C. Y. Cheng, A. T. Slominski, and R. C. Tuckey, Hydroxylation of 20-hydroxyvitamin D3 by human CYP3A4, J. Steroid Biochem. Mol. Biol., 2016, 159, 131–141
A. T. Slominski, T. K. Kim, J. V. Hobrath, A. S. Oak, E. K. Tang, E. W. Tieu, W. Li, R. C. Tuckey, and A. M. Jetten, Endogenously produced nonclassical vitamin D hydroxy-metabolites act as “biased” agonists on VDR and inverse agonists on RORα and RORγ, J. Steroid Biochem. Mol. Biol., 2016, 10.1016/j.jsbmb.2016.09.024
B. Bittiner, S. S. Bleehen and S. Mac Neil, 1α-25-(OH)2 Vitamin D3 increases intracellular calcium in human keratinocytes, Br. J. Dermatol., 1991, 124, 12230–12235
J. A. MacLaughlin, L. C. Cantley, and M. F. Holick, 1,25(OH)2D3 increases calcium and phosphatidylinositol metabolism in differentiating cultured human keratinocytes, J. Nutr. Biochem., 1990, 1, 81–87
M. Doroudi, R. Olivares-Navarrete, B. D. Boyan and Z. Schwartz, A review of 1α,25(OH)2D3 dependent Pdia3 receptor complex components in Wnt5a non-canonical pathway signaling, J. Steroid Biochem. Mol. Biol., 2015, 152, 84–88
J. Reichrath, S. Reichrath, K. Heyne, T. Vogt and K. Roemer, Tumor suppression in skin and other tissues via cross-talk between vitamin D- and p53-signaling, Front. Physiol., 2014, 5, 1–10
M. R. Haussler, G. K. Whitfield, I. Kaneko, C. A. Haussler, D. Hsieh, J. C. Hsieh and P. W. Jurutka, Molecular mechanisms of vitamin D action, Calcif. Tissue Int., 2013, 92, 77–98
A. R. Baker, D. P. Mc Donnell, M. Hughes, T. M. Crisp, D. J. Mangelsdorf, M. R. Haussler, J. W. Pike, J. Shine and B. W. O’Malley, Cloning and expression of full-length cDNA encoding human vitamin D receptor, Proc. Natl. Acad. Sci. U. S. A., 1988, 85, 3294–3298
V. C. Yu, C. Deisert, B. Andersen, J. M. Holloway, O. V. Devary, A. M. Näär, S. Y. Kim, J. M. Boutin, C. K. Glass and M. G. Rosenfeld, RXRβ: a coregulator that enhances binding of retinoic acid, thyroid hormone and vitamin D receptors to their cognate response elements, Cell, 1991, 67, 1251–1266
K. Heyne, T. C. Heil, B. Bette, J. Reichrath and K. Roemer, MDM2 binds and inhibits vitamin D receptor, Cell Cycle, 2015, 14, 2003–2010
P. Milde, U. Hauser, R. Simon, G. Mall, V. Ernst, M. R. Haussler, P. Frosch and E. W. Rauterberg, Expression of 1,25-dihydroxyvitamin D3 receptors in normal and psoriatic skin, J. Invest. Dermatol., 1991, 97, 230–239
J. Reichrath, T. Münssinger, A. Kerber, C. Rochette-Egly, P. Chambon, F. A. Bahmer and H. P. Baum, In situ detection of retinoid-X receptor expression in normal and psoriatic human skin, Br. J. Dermatol., 1995, 133, 168–175
A. T. Slominski, T. K. Kim, Y. Takeda, Z. Janjetovic, A. A. Brozyna, C. Skobowiat, J. Wang, A. Postlethwaite, W. Li, R. C. Tuckey and A. M. Jetten, RORα and ROR γ are expressed in human skin and serve as receptors for endogenously produced noncalcemic 20-hydroxy- and 20,23-dihydroxyvitamin D, FASEB J., 2014, 7, 2775–2789
E. L. Smith, N. C. Walworth and M. F. Holick, Effect of 1α-25-dihydroxyvitamin D3 on the morphologic and biochemical differentiation of cultured human epidermal keratinocytes grown under serum-free conditions, J. Invest. Dermatol., 1986, 86, 709–714
J. Hosomi, J. Hosoi, E. Abe, T. Suda and T. Kuroki, Regulation of terminal differentiation of cultured mouse epidermal cells by 1-alpha 25-dihydroxy-vitamin D3, Endocrinology, 1983, 113, 1950–1957
R. Gniadecki and J. Serup, Stimulation of epidermal proliferation in mice with 1 alpha, 25-dihydroxyvitamin D3 and receptor-active 20-EPI analogues of 1 alpha, 25-dihydroxyvitamin D3, Biochem. Pharmacol., 1995, 49, 621–624
W. F. C. Rigby, The immunobiology of vitamin D, Immunol. Today, 1988, 9, 54–58
M. Texereau, and J. Viac, Vitamin D, immune system and skin, Eur. J. Dermatol., 1992, 2, 258–264
H. F. Gombard, N. Borregaard and H. P. Koeffler, Human cathelicidin antimicrobial peptide (CAMP) gene is a direct target of the vitamin D receptor and is strongly up-regulated in myeloid cells by 1,25-dihydroxyvitamin D3, FASEB J., 2005, 19, 1067–1077
T.-T. Wang, F. P. Nestel, V. Bourdeau, Y. Nagai, Q. Wang, J. Liao, L. Tavera-Mendoza, R. Lin, J. H. Hanrahan, S. Mader and J. H. White, Cutting Edge: 1,25-Dihydroxyvitamin D3 is a direct inducer of antimicrobial peptide gene expression, J. Immunol., 2004, 173, 2909–2912
P. T. Liu, S. Stenger, H. Li, L. Wenzel, B. H. Tan, S. R. Krutzik, M. T. Ochoa, J. Schauber, K. Wu, C. Meinken, D. L. Kamen, M. Wagner, R. Bals, A. Steinmeyer, U. Zügel, R. L. Gallo, D. Eisenberg, M. Hewison, B. W. Hollis, J. S. Adams, B. R. Bloom and R. L. Modlin, Toll-like receptor triggering of a vitamin D-mediated human antimicrobial response, Science, 2006, 311, 1770–1773
N. Karthaus, A. B. van Spriel, M. W. Looman, S. Chen, L. M. Spilgies, L. Lieben, G. Carmeliet, M. Ansems and G. J. Adema, Vitamin D controls murine and human plasmacytoid dendritic cell function, J. Invest. Dermatol., 2014, 134, 1255–1264
A. Kaukinen, J. Pelkonen and I. T. Harvima, Mast cells express CYP27A1 and CYP27B1 in epithelial skin cancers and psoriasis, Eur. J. Dermatol., 2015, 25, 548–555
A. Gruber-Wackernagel, I. Bambach, F. J. Legat, A. Hofer, S. N. Byrne, F. Quehenberger and P. Wolf, Randomized double-blinded placebo-controlled intra-individual trial on topical treatment with a 1,25-dihydroxyvitamin D3 analogue in polymorphic light eruption, Br. J. Dermatol., 2011, 165, 152–163
M. Ranson, S. Posen and R. S. Mason, Human melanocytes as a target tissue for hormones: in vitro studies with 1α,25-dihydroxyvitamin D3, alpha-melanocyte stimulating hormone, and beta-estradiol, J. Invest. Dermatol., 1988, 91, 593–598
C. Krämer, H. Seltmann, M. Seifert, W. Tilgen, C. C. Zouboulis and J. Reichrath, Characterization of the vitamin D endocrine system in human sebocytes in vitro, J. Steroid Biochem. Mol. Biol., 2009, 113, 9–16
J. A. MacLaughlin, W. Gange, D. Taylor, E. Smith and M. F. Holick, Cultured psoriatic fibroblasts from involved and uninvolved sites have partial but not absolute resistance to the proliferation-inhibition activity of 1,25-dihydroxyvitamin D3, Proc. Natl. Acad. Sci. U. S. A., 1985, 82, 5409–5412
S. Morimoto, and Y. Kumahara, A patient with psoriasis cured by 1α-hydroxyvitamin D3, Med. J. Osaka Univ., 1985, 35, 51–54
S. Morimoto, K. Yochikawa, T. Kozuka, Y. Kitano, S. Imawaka, K. Fukuo, E. Koh, and Y. Kumahara, An open study of vitamin D3 treatment in psoriasis vulgaris, Br. J. Dermatol., 1986, 115, 421–429
M. F. Holick, M. L. Chen, X. F. Kong, and D. K. Sanan, Clinical uses for calciotropic hormones 1,25-dihydroxyvitamin D3 and parathyroid hormone related peptide in dermatology: a new perspective, J. Invest. Dermatol. Symp. Proc., 1996, 1, 1–9
A. Perez, T. C. Chen, A. Turner, R. Raab, J. Bhawan, P. Poche, and M. F. Holick, Efficacy and safety of topical calcitriol (1,25-dihydroxyvitamin D3) for the treatment of psoriasis, Br. J. Dermatol., 1996, 134, 238–246
K. Kragballe, H. I. Beck, and H. Sogaard, Improvement of psoriasis by topical vitamin D3 analogue (MC 903) in a double-blind study, Br. J. Dermatol., 1988, 119, 223–230
P. C. M. van de Kerkhof, M. van Bokhoven, M. Zultak, and B. M. Czarnetzki, A double-blind study of topical 1α-25-dihydroxyvitamin D3 in psoriasis, Br. J. Dermatol., 1989, 120, 661–664
J. N. Barker, R. E. Ashton, R. Marks, R. I. Harris, J. Berth-Jones, Topical maxacalcitol for the treatment of psoriasis vulgaris: a placebo-controlled, double-blind, dose-finding study with active comparator, Br. J. Dermatol., 1999, 141, 274–278
C. Durakovic, A. Malabanan, and M. F. Holick, Rationale for use and clinical responsiveness of hexafluoro-1,25-dihydroxyvitamin D3 for the treatment of plaque psoriasis: a pilot study, Br. J. Dermatol., 2001, 144, 500–506
Y. Miyachi, A. Ohkawara, M. Ohkido, S. Harada, K. Tamaki, H. Nakagawa, Y. Hori, and S. Nishiyama, Long-term safety and efficacy of high-concentration (20 microg/g) tacalcitol ointment in psoriasis vulgaris, Eur. J. Dermatol., 2002, 12, 463–468
P. C. van de Kerkhof, J. Berth-Jones, C. E. Griffiths, P. V. Harrison, H. Honigsmann, R. Marks, R. Roelandts, E. Schopf, and C. Trompke, Long-term efficacy and safety of tacalcitol ointment in patients with chronic plaque psoriasis, Br. J. Dermatol., 2002, 146, 414–422
K. Kragballe, B. T. Gjertsen, D. de Hoop, T. Karlsmark, P. C. M. van de Kerhof, O. Larko, C. Nieboer, J. Roed-Petersen, A. Strand, and B. Tikjob, Double-blind right/left comparison of calcipotriol and betametasone valerate in treatment of psoriasis vulgaris, Lancet, 1991, 337, 193–196
L. Binderup, S. Latini, E. Binderup, C. Bretting, M. Calverley, and K. Hansen, 20-epi-vitamin D3 analogues: a novel class of potent regulators of cell growth and immune response, Biochem. Pharmacol., 1991, 42, 1569–1575
C. Queille-Roussel, L. Duteil, A. Parneix-Spake, S. Arsonnaud, and E. Rizova, The safety of calcitriol 3 microg/g ointment. Evaluation of cutaneous contact sensitization, cumulative irritancy, photoallergic contact sensitization and phototoxicity, Eur. J. Dermatol., 2001, 11, 219–224
A. Perez, R. Raab, T. C. Chen, A. Turner, and M. F. Holick, Safety and efficacy of oral calcitriol (1,25-dihydroxyvitamin D3) for the treatment of psoriasis, Br. J. Dermatol., 1996, 134, 1070–1078
J. Reichrath, S. M. Müller, A. Kerber, H. P. Baum, and F. A. Bahmer, Biologic effects of topical calcipotriol (MC 903) treatment in psoriatic skin, J. Am. Acad. Dermatol., 1997, 36, 19–28
B. Dyring-Andersen, C. M. Bonefeld, M. Bzorek, M. B. Løvendorf, J. P. Lauritsen, L. Skov, and C. Geisler, The Vitamin D Analogue Calcipotriol Reduces the Frequency of CD8+ IL-17+ T Cells in Psoriasis Lesions, Scand. J. Immunol., 2015, 82, 84–91
C. Mattozzi, G. Paolino, M. Salvi, L. Macaluso, C. Luci, S. Morrone, S. Calvieri, and A. G. Richetta, Peripheral blood regulatory T cell measurements correlate with serum vitamin D level in patients with psoriasis, Eur. Rev. Med. Pharmacol. Sci., 2016, 20, 1675–1679
S. Cubillos, N. Krieg, and J. Norgauer, Effect of Vitamin D on Peripheral Blood Mononuclear Cells from Patients with Psoriasis Vulgaris and Psoriatic Arthritis, PLoS One, 2016, 11, 1–16
M. L. Chen, A. Perez, D. K. Sanan, G. Heinrich, T. C. Chen, and M. F. Holick, Induction of vitamin D receptor mRNA expression in psoriatic plaques correlates with clinical response to 1,25-dihydroxyvitamin D3, J. Invest. Dermatol., 1996, 106, 637–641
C. Missero, E. Calautti, R. Eckner, J. Chin, L. H. Tsai, D. M. Livingston, and G. P. Dotto, Involvement of the cell-cycle inhibitor Cip1/WAF1 and the E1A-associaated p300 protein in terminal differentiation, Proc. Natl. Acad. Sci. U. S. A., 1995, 92, 5451–5455
B. Visconti, G. Paolino, S. Carotti, A. L. Pendolino, S. Morini, A. G. Richetta, and S. Calvieri, Immunohistochemical expression of VDR is associated with reduced integrity of tight junction complex in psoriatic skin, J. Eur. Acad. Dermatol. Venereol., 2015, 29, 2038–2042
D. Dayangac-Erden, A. Karaduman, H. Erdem-Yurter, Polymorphisms of vitamin D receptor gene in Turkish familial psoriasis patients, Arch. Dermatol. Res., 2007, 299, 487–491
H. Okita, T. Ohtsuka, A. Yamakage, and S. Yamazaki, Polymorphism of the vitamin D(3) receptor in patients with psoriasis, Arch. Dermatol. Res., 2002, 294, 159–162
B. S. Park, J. S. Park, D. Y. Lee, J. I. Youn, and I. G. Kim, Vitamin D receptor polymorphism is associated with psoriasis, J. Invest. Dermatol., 1999, 112, 113–116
H. Saeki, N. Asano, Y. Tsunemi, T. Takekoshi, M. Kishimoto, H. Mitsui, Y. Tada, H. Torii, M. Komine, A. Asahina, and K. Tamaki, Polymorphisms of vitamin D receptor gene in Japanese patients with psoriasis vulgaris, J. Dermatol. Sci., 2002, 30, 167–171
D. Y. Lee, B. S. Park, K. H. Choi, J. H. Jeon, K. H. Cho, K. Y. Song, I. G. Kim, and J. I. Youn, Vitamin D receptor genotypes are not associated with clinical response to calcipotriol in Korean psoriasis patients, Arch. Dermatol. Res., 2002, 294, 1–59
K. Kontula, S. Välimäki, K. Kainulainen, A. M. Viitanen, J. Keski-Oja, Vitamin D receptor polymorphism and treatment of psoriasis with calcipotriol, Br. J. Dermatol., 1997, 136, 147–148
J. B. Mee, and M. J. Cork, Vitamin D receptor polymorphism and calcipotriol response in patients with psoriasis, J. Invest. Dermatol., 1998, 110, 301–302
E. M. Colin, A. E. A. M. Weel, A. G. Uitterlinden, C. J. Buurman, J. C. Birkenhäger, H. A. P. Pols, J. P. T. M. van Leeuwen, Consequences of vitamin D receptor gene polymorphisms for growth inhibition of cultured human peripheral blood mononuclear cells by 1,25-dihydroxyvitamin D3, Clin. Endocrinol., 2000, 52, 211–216
A. G. Richetta, V. Silvestri, S. Giancristoforo, P. Rizzolo, S. D’Epiro, V. Graziano, C. Mattozzi, A. S. Navazio, M. Campoli, C. D’Amico, M. Scarnò, S. Calvieri, and L. Ottini, A-1012G promoter polymorphism of vitamin D receptor gene is associated with psoriasis risk and lower allele-specific expression, DNA Cell Biol., 2014, 33, 102–109
B. Staberg, A. Oxholm, P. Klemp, and C. Christiansen, Abnormal vitamin D metabolism in patients with psoriasis, Acta Derm.-Venereol., 1987, 67, 65–68
A. F. Stewart, J. Battaglini-Sabetta, and L. Millstone, Hypocalcemia-induced pustular psoriasis of von Zumbusch. New experience with an old syndrome, Ann. Intern. Med., 1984, 100, 677–680
O. J. Stone, Chloroquine, ground substance, aggravation of psoriasis, Int. J. Dermatol., 1985, 24, 539
G. P. Lucker, P. C. van de Kerkhof, M. R. van Dijk, and P. M. Steijlen, Effect of topical calcipotriol on congenital ichthyosis, Br. J. Dermatol., 1994, 131, 546–550
M. Okano, Assessment of the clinical effect of topical tacalcitol on ichthyoses with retentive hyperkeratosis, Dermatology, 2001, 202, 116–118
P. Humbert, J. L. Dupond, P. Agache, R. Laurent, A. Rochefort, C. Drobacheff, B. de Wazieres, and F. Aubin, Treatment of scleroderma with oral 1,25-dihydroxyvitamin D3: evaluation of skin involvement using non-invasive techniques. Results of an open prospective trial, Acta Derm.-Venereol., 1993, 73, 449–451
G. Garnier, Vitamin D2 in treatment of acne conglobata, Arch. Belg. Dermatol. Syphiligr., 1964, 20, 105–108
B. Cerri, Attempted therapy for lupus vulgaris and juvenile acne with provitamins of the D group, Minerva Dermatol., 1952, 27, 3 53–57
G. W. Agak, M. Qin, J. Nobe, M. H. Kim, S. R. Krutzik, G. R. Tristan, D. Elashoff, H. J. Garbán, and J. Kim, Propionibacterium acnes Induces an IL-17 Response in Acne Vulgaris that Is Regulated by Vitamin A and Vitamin D, J. Invest. Dermatol., 2014, 34, 366–373
N. Hayashi, H. Watanabe, H. Yasukawa, H. Uratsuji, H. Kanazawa, M. Ishimaru, N. Kotera, M. Akatsuka, and M. Kawashima, Comedolytic effect of topically applied active vitamin D3 analogue on pseudocomedones in the rhino mouse, Br. J. Dermatol., 2006, 155, 895–901
J. Schauber, Antimicrobial peptides, Vitamin D3 and more. How rosacea may develop, Hautarzt, 2011, 62, 815–819
J. D. Heilborn, G. Weber, A. Grönberg, C. Dieterich, M. Ståhle, Topical treatment with the vitamin D analogue calcipotriol enhances the upregulation of the antimicrobial protein hCAP18/LL-37 during wounding in human skin in vivo, Exp. Dermatol., 2010, 19, 4 332–338
J. Reichrath and M. F. Holick, Clinical Utility of 1,25-dihydroxyvitamin D3 and its analogs for the treatment of psoriasis and other skin diseases, Vitamin D. Physiology, Molecular Biology and Clinical Applications, 1999, pp. 357–374
A. M. Carrozzo, S. Gatti, G. Ferranti, G. Primavera, A. P. Vidolin, and G. Nini, Calcipotriol treatment of confluent and reticulated papillomatosis (Gougerot-Carteau syndrome), JEADV, 2000, 14, 131–133
M. Zanolli, Phototherapy treatment of psoriasis today, J. Am. Acad. Dermatol., 2003, 49, 2 suppl S78–S86
W. H. Goeckerman, Treatment of psoriasis, Northwest Med., 1925, 24, 229–231
S. A. Muller, and H. O. Perry, The Goeckerman treatment in psoriasis: six decades of experience at the Mayo Clinic, Cutis, 1984, 34, 3 265–268
C. E. Griffiths, C. M. Clark, R. J. Chalmers, P. A. Li Wan, and H. C. Williams, A systematic review of treatments for severe psoriasis, Health Technol. Assess, 2000, 4, 40 1–125
L. Guenther, R. G. Langley, N. H. Shear, R. Bissonnette, V. Ho, C. Lynde, et al., Integrating biologic agents into management of moderate-to-severe psoriasis: a consensus of the Canadian Psoriasis Expert Panel, J. Cutaneous Med. Surg., 2004, 8, 5 321–337
A. Juzeniene, P. Brekke, A. Dahlback, S. Andersson-Engels, J. Reichrath, K. Moan, M. Holick, W. B. Grant, and J. Moan, Solar radiation and human health, Rep. Prog. Phys., 2011, 74, 6 1–56
G. M. Murphy, Ultraviolet radiation and immunosuppression, Br. J. Dermatol., 2009, 161, suppl. 3 90–5
M. S. Fisher, and M. L. Kripke, Systemic alteration induced in mice by ultraviolet light irradiation and its relationship to ultraviolet carcinogenesis, Proc. Natl. Acad. Sci. U. S. A., 1977, 74, 1688–1692
S. L. Walker, and A. R. Young, An action spectrum (290–320 nm) for TNFalpha protein in human skin in vivo suggests that basal-layer epidermal DNA is the chromophore, Proc. Natl. Acad. Sci. U. S. A., 2007, 104, 19051–19054
G. M. Murphy, P. G. Norris, A. R. Young, M. F. Corbett, and J. L. Hawk, Low-dose ultraviolet-B irradiation depletes human epidermal Langerhans cells, Br. J. Dermatol., 1993, 129, 674–677
G. F. Murphy, S. Katz, and A. M. Kligman, Topical tretinoin replenishes CD1a-positive epidermal Langerhans cells in chronically photodamaged human skin, J. Cutaneous Pathol., 1998, 25, 30–34
A. V. Powles, G. M. Murphy, A. J. Rutman, G. Haffenden, R. D. Rosin, J. L. Hawk, and L. Fry, Effect of simulated sunlight on Langerhans’ cells in malignant melanoma patients, Acta Derm.-Venereol., 1989, 69, 482–486
L. Timares, S. K. Katiyar, and C. A. Elmets, DNA damage, apoptosis and langerhans cells–Activators of UV-induced immune tolerance, Photochem. Photobiol., 2008, 84, 422–436
S. Lembo, J. Fallon, P. O’Kelly, and G. M. Murphy, Polymorphic light eruption and skin cancer prevalence: is one protective against the other?, Br. J. Dermatol., 2008, 159, 1342–1347
A. S. Jonason, S. Kunala, G. J. Price, R. J. Restifo, H. M. Spinelli, J. A. Persing, D. J. Leffell, R. E. Tarone, and D. E. Brash, Frequent clones of p53-mutated keratinocytes in normal human skin, Proc. Natl. Acad. Sci. U. S. A., 1996, 93, 14025–14029
J. M. Kuchel, R. S. Barnetson, and G. M. Halliday, Cyclobutane pyrimidine dimer formation is a molecular trigger for solar-simulated ultraviolet radiation-induced suppression of memory immunity in humans, Photochem. Photobiol. Sci., 2005, 4, 577–582
D. Leibeling, P. Laspe, and S. Emmert, Nucleotide excision repair and cancer, J. Mol. Histol., 2006, 37, 225–238
S. Arad, N. Konnikov, D. A. Goukassian, and B. A. Gilchrest, T-oligos augment UV-induced protective responses in human skin, FASEB J., 2006, 20, 1895–1897
E. C. De Fabo, and F. P. Noonan, Mechanism of immune suppression by ultraviolet irradiation in vivo. I. Evidence for the existence of a unique photoreceptor in skin and its role in photoimmunology, J. Exp. Med., 1983, 158, 84–98
I. Kurimoto, and J. W. Streilein, cis-urocanic acid suppression of contact hypersensitivity induction is mediated via tumor necrosis factor-alpha, J. Immunol., 1992, 148, 3072–3078
J. A. Ross, S. E. Howie, M. Norval, J. Maingay, and T. J. Simpson, Ultraviolet-irradiated urocanic acid suppresses delayed-type hypersensitivity to herpes simplex virus in mice, J. Invest. Dermatol., 1986, 87, 630–633
Z. Zheng, R. Chen, and J. Prystowsky, UVB radiation induces phosphorylation of the epidermal growth factor receptor decreases EGF binding and blocks EGF induction of ornithine decarboxylase gene expression in SV-40-transfomed human keratinocytes, Exp. Dermatol., 1993, 2, 257–265
C. Sachsenmaier, A. Radler-Pohl, R. Zinck, A. Nordheim, P. Herrlich, and H. J. Rahmsdorf, Involvement of growth factor receptors in the mammalian UVC response, Cell, 1994, 78, 963–972
D. Peus, R. A. Vasa, A. Meves, M. Pott, A. Beyerle, K. Squillace, and M. R. Pittelkow, H2O2 is an important mediator of UVB-induced EGF-receptor phosphorylation in cultured keratinocytes, J. Invest. Dermatol., 1998, 110, 966–971
D. Peus, R. A. Vasa, A. Beyerle, A. Meves, C. Krautmacher, and M. R. Pittelkow, UVB activates ERK1/2 and p38 signaling pathways via reactive oxygen species in cultured keratinocytes, J. Invest. Dermatol., 1999, 112, 751–756
Q. Zhang, Y. Yao, R. L. Konger, A. L. Sinn, S. Cai, K. E. Pollok, and J. B. Travers, UVB radiation-mediated inhibition of contact hypersensitivity reactions is dependent on the platelet-activating factor system, J. Invest. Dermatol., 2008, 128, 1780–1787
P. Wolf, D. X. Nghiem, J. P. Walterscheid, S. Byrne, Y. Matsumura, Y. Matsumura, C. Bucana, H. N. Ananthaswamy, and S. E. Ullrich, Platelet-activating factor is crucial in psoralen and ultraviolet A-induced immune suppression, inflammation, and apoptosis, Am. J. Pathol., 2006, 169, 795–805
L. L. Hill, V. K. Shreedhar, M. L. Kripke, L. B. Owen-Schaub, A critical role for Fas ligand in the active suppression of systemic immune responses by ultraviolet radiation, J. Exp. Med., 1999, 189, 1285–1294
B. Bang, R. Gniadecki, J. K. Larsen, O. Baadsgaard, and L. Skov, In vivo UVB irradiation induces clustering of Fas (CD95) on human epidermal cells, Exp. Dermatol., 2003, 12, 791–798
G. M. Murphy, P. M. Dowd, B. N. Hudspith, J. Brostoff, and M. W. Greaves, Local increase in interleukin-1-like activity following UVB irradiation of human skin in vivo, Photodermatology, 1989, 6, 268–274
M. Ghoreishi, and J. P. Dutz, Tolerance induction by transcutaneous immunization through ultraviolet-irradiated skin is transferable through CD4+CD25+ T regulatory cells and is dependent on host-derived IL-10, J. Immunol., 2006, 176, 2635–2644
J. M. Rivas, and S. E. Ullrich, Systemic suppression of delayed-type hypersensitivity by supernatants from UV-irradiated keratinocytes. An essential role for keratinocyte-derived IL-10, J. Immunol., 1992, 149, 3865–3871
K. Seiffert, and R. D. Granstein, Neuropeptides and neuroendocrine hormones in ultraviolet radiation-induced immunosuppression, Methods, 2002, 28, 97–103
M. C. Scott, I. Suzuki, Z. A. Abdel-Malek, Regulation of the human melanocortin 1 receptor expression in epidermal melanocytes by paracrine and endocrine factors and by ultraviolet radiation, Pigm. Cell Res., 2002, 15, 433–439
G. M. Halliday, and S. Rana, Waveband and dose dependency of sunlight-induced immunomodulation and cellular changes., Photochem. Photobiol., 2008, 84, 35–46
T. S. Poon, R. S. Barnetson, and G. M. Halliday, Sunlight-induced immunosuppression in humans is initially because of UVB, then UVA, followed by interactive effects, J. Invest. Dermatol., 2005, 125, 840–846
M. Norval, The effect of ultraviolet radiation on human viral infections, Photochem. Photobiol., 2006, 82, 1495–1504
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Reichrath, J., Saternus, R. & Vogt, T. Challenge and perspective: the relevance of ultraviolet (UV) radiation and the vitamin D endocrine system (VDES) for psoriasis and other inflammatory skin diseases. Photochem Photobiol Sci 16, 433–444 (2017). https://doi.org/10.1039/c6pp00280c
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1039/c6pp00280c