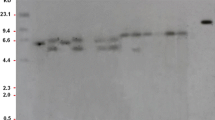Abstract
Convolvulus arvensis is a troublesome weed that is naturally tolerant to glyphosate. This weed tolerates glyphosate at a rate 5.1 times higher than that of glyphosate-susceptible Calystegia hederacea. Glyphosate-treated C. arvensis plants accumulated less shikimic acid than C. hederacea plants. The overexpression of EPSPS genes from the two species in transgenic Arabidopsis thaliana resulted in similar glyphosate tolerance levels. qPCR of genomic DNA revealed that the EPSPS copy number in C. arvensis was approximately 2 times higher than that in C. hederacea. Moreover, glyphosate treatment caused a marked increase in EPSPS mRNA in C. arvensis compared to C. hederacea. GUS activity analysis showed that the promoter of CaEPSPS (CaEPSPS-P) highly improved GUS expression after glyphosate treatment, while no obvious differential GUS expression was observed in ChEPSPS-P transgenic A. thaliana in the presence or absence of glyphosate. Based on the obtained results, two coexisting mechanisms may explain the natural glyphosate tolerance in C. arvensis: (i) high EPSPS copy number and (ii) specific promoter-mediated overexpression of EPSPS after glyphosate treatment.
Similar content being viewed by others
Introduction
Glyphosate is a nonselective, foliar-applied herbicide that has been used to manage annual, perennial, and biennial herbaceous species of grasses, sedges, and broadleaf weeds1,2. It affects aromatic amino acid biosynthesis by inhibiting 5-enolpyruvyl-shikimate-3-phosphate synthase (EPSPS), a nuclear-encoded, plastid-localized enzyme in the shikimate pathway3. Glyphosate has become the most widely used herbicide in the world due to its advantage of broad-spectrum, low toxicity, and low soil residual activity4. However, the widespread and intensive use of glyphosate over years imposes selective pressure on weeds5,6. Since glyphosate resistance was first found in rigid ryegrass (Lolium rigidum)7 in Australia in 1996, 43 weed species with resistance to glyphosate have been detected8.
Mechanisms of glyphosate resistance are classified as target-site and non-target site. Target-site resistance is caused by mutations in EPSPS that decrease its binding affinity for glyphosate, or by EPSPS overexpression, which allows the plant to produce adequate EPSPS to maintain the synthesis of aromatic amino acids. Single amino acid substitutions in EPSPS at position 106 from proline to serine (P106S), alanine (P106A), threonine (P106T), or leucine (P106L) have been identified in Eleusine indica9, L. rigidum10,11, Lolium multiflorum12, Echinochloa colona13, and Amaranthus tuberculatus14,15. Additionally, a double amino acid substitution (T102I + P106S) in E. indica16,17 and Bidens Pilosa18 in certain populations was found and reported to confer a higher glyphosate resistance level than that conferred by the single P106S mutation.
EPSPS overexpression through increased EPSPS copy number confers glyphosate resistance in A. palmeri19, L. multiflorum20, A. spinosus21, and A. tuberculatus22,23. For glyphosate-resistant A. palmeri, increased EPSPS copy number produces abundant enzymes to maintain the shikimate pathway18. Furthermore, EPSPS overexpression through elevated EPSPS transcript levels after glyphosate treatment is associated with glyphosate tolerance in Dicliptera chinensis24 and Ophiopogon japonicus25.
Reduced glyphosate absorption, translocation26,27, and vacuolar sequestration28 are the main non-target glyphosate resistance mechanism. To protect the young meristematic tissue, resistant plants sequester glyphosate within the vacuoles of the leaves29,30. Maintaining glyphosate in vacuolar tissues by ABC transporters to avoid damage was identified to be responsible for glyphosate resistance31,32. Furthermore, studies have reported that chloroplast proteins played an important role in glyphosate resistance in Conyza canadensis33.
Field bindweed (Convolvulus arvensis L) is a perennial weed in the morning-glory family. It is considered one of the most troublesome weeds threatening wheat and cotton production in China34. C. arvensis was the first weed reported to be naturally tolerant to glyphosate35. Previous studies aimed at illuminating the glyphosate tolerance mechanism in C. arvensis have mainly focused on glyphosate absorption and translocation. However, there were no obvious differences in absorption and translocation36,37. Until recently, the tolerance mechanism has not been fully understood. As C. arvensis is naturally tolerant to glyphosate, and a susceptible population in China was not obtained in our previous studies. Therefore, glyphosate-susceptible Calystegia hederacea was used as a control because C. hederacea belongs to the Convolvulaceae family and shares similar biological characteristics with C. arvensis in many aspects, such as perennial, vine climbing, and rapid growth38. In this article, we investigated the mechanism of glyphosate tolerance in C. arvensis with physiological (shikimic acid accumulation) and molecular (EPSPS cloning, overexpression of EPSPS gene, and EPSPS gene expression pattern) approaches. We cloned the EPSPS genes of C. hederacea and C. arvensis and inserted the EPSPS gene into the common model plant Arabidopsis thaliana, which is an excellent tool for research in plant biology39. We examined the glyphosate tolerance of EPSPS-transgenic A. thaliana. We also compared the basal and glyphosate-induced mRNA levels of EPSPS from the two species.
Materials and Methods
Plant material and growth conditions
Seeds of C. arvensis and C. hederacea collected in Beijing, China were germinated in Petri dishes with moist filter paper in an illumination incubator (25 °C day/night temperature). Individual seedlings in the cotyledon growth stage were transplanted into pots (5 cm radius; 6 seedlings per pot) containing a 1:1 (V ⁄ V) peat: sand sterile potting mix. The plants were placed in a greenhouse with an average day/night temperature of 25/20 °C and a 12-h photoperiod under artificial illumination (300 μmol m−2 s−1). The plants were watered as needed.
Glyphosate dose–response assay
Plants at the 5–6 leaf stage were sprayed with glyphosate (Roundup Ultra, 41% glyphosate isopropylammonium, Monsanto, USA) at doses of 0, 250, 500, 1000, 2000, 4000 and 8000 g ha−1 using a research track sprayer (3WPSH-500D), which delivered 450 L ha−1 spray solution at 0.3 MPa. All treatments contained 3 replicate pots (6 plants per pot). Plants were assessed 14 days after treatment (DAT). All aboveground plant materials were cut and dried at 60 °C for 72 h. Dry weight was measured when constant weight was achieved. The experiment was arranged in a completely randomized design and was repeated two times with three replications each.
Shikimate accumulation in vivo assay
Plants at the 5–6 leaf stage sprayed with 1000 g ha−1 glyphosate were harvested at 2, 4, 6, 8, 10 and 12 DAT, and foliar tissue samples were stored at −80 °C until further processed. Determination of shikimate accumulation in C. hederacea and C. arvensis tissue was conducted spectrophotometrically according to Chen40. Shikimic acid was detected using a double-beam spectrophotometer at 380 nm. The determination of the shikimic acid concentration was based on a shikimate (Sigma-Aldrich, Saint Louis, MO, USA). 99% purity) standard curve.
EPSPS gene cloning and sequence analysis
Leaves of C. hederacea and C. arvensis were sampled and ground to fine powders in liquid nitrogen, and the total RNA was extracted with the RNAprep Pure Plant Kit (Tiangen Biotech Co., Ltd., China) following the manufacturer’s protocol. First-strand complementary DNA (cDNA) was amplified with random primers using EasyScript First-Strand cDNA Synthesis SuperMix (TransGen Biotech, China). The final cDNA was stored at −20 °C.
The primer pair EPSPS-cf and EPSPS-cr was designed from plant EPSPS gene sequences in NCBI. PCR was performed in a thermal cycler as follows: 5 min at 95 °C; 30 s at 95 °C; 30 s at 57 °C; 35 s at 72 °C (35 cycles); and 10 min at 72 °C. The amplified product was purified and cloned into the pMD19-T vector (Takara, Japan) for sequencing. The sequence obtained from the conserved region was used to design the 5′-end and the 3′-end primers. Fragments amplified by 5′ and 3′ RACE were purified, cloned into the pMD19-T vector and sequenced. Because of their high homology, ChEPSPS-f and ChEPSPS-r were designed to amplify the full-length EPSPS gene of C. hederacea according to that of C. arvensis. Sequence assembly and comparative analyses of the EPSPS genes from the two species were conducted using DNAMAN (Version 5.0).
The promoters of EPSPS from C. hederacea and C. arvensis were amplified with the gwEPS-1, gwEPS-2, and gwEPS-3 primers of the Universal Genome WalkerTM Kit (Clontech, USA) following the manufacturer’s protocol. The sequences of primers used in the present study are listed in Table 1. The prediction of cis-acting elements in the promoters was performed by using the software Plant-CARE.
Quantitative PCR (qPCR) analysis
The relative EPSPS copy number was estimated using genomic DNA. Total DNA from young leaves (100 mg) of the two species from three plants of each replicate was extracted using the New Plant Genome Extraction Kit (Tiangen Biotech Co., Ltd., China). After eluting in double-distilled water, genomic DNA quality and concentration were determined spectrophotometrically, and the DNA samples were stored at −20 °C.
The EPSPS expression level was determined using mRNA extracted from plants after glyphosate treatments. Plants sprayed with 1000 g a.e. ha−1 glyphosate at the 5–6 leaf stage were harvested at 0.5, 1, 2, 4, 6 and 8 DAT. The leaves (the uppermost three leaves, 100 mg) of the two species were sampled from three plants of each replicate and ground to a fine powder in liquid nitrogen, and the total RNA was extracted by using the RNAprep Pure Plant Kit (Tiangen, China) following the manufacturer’s protocol. After elution of total RNA in double-distilled water, DNase I was added to digest any contaminating DNA and then removed. The cDNA was amplified with random primers using the EasyScript First-Strand cDNA Synthesis SuperMix (TransGen Biotech, China).
qPCR was performed in 96-well plates on the ABI 7500 real-time PCR system with the SYBR Green I Master Mix (Invitrogen, USA). To quantify the copy number and expression level of EPSPS, the housekeeping GAPDH gene was used as the internal control gene because the GAPDH gene did not vary across the samples based on our qPCR results (data not shown). The primer sequences used in this study are listed in Table 1. Melting curves were performed before each qPCR experiment to assess the specificity of the primers. The following two-step real-time PCR detection system was used: 15 s at 95 °C and 25 s at 62 °C. Relative gene copy number or expression level was obtained with the formula for fold induction, 2−△△CT. The CT (threshold cycle) value represents the PCR cycle at which the EPSPS copy number or expression level passes the fixed threshold. Two experiments on three independent plant materials were performed to confirm the results, and each time point was repeated three times.
Chimeric vector construction, plant transformation and overexpression of the EPSPS gene in A. thaliana
Total RNA was isolated, and cDNA was synthesized. The coding regions of EPSPS of C. hederacea and C. arvensis were amplified using the EPS-1f and EPS-1r primer pair (Table 1), and the complete EPSPS gene was inserted into the pMD19-T. The vector was verified by sequencing and then digested using XbaI/SmaI. The resulting product was cloned into the pBI121 vector, and the 35S::EPSPS construct was obtained.
The expression vectors 35S::CaEPSPS and 35S::ChEPSPS were introduced into GV3101 Agrobacterium tumefaciens. The transformed A. tumefaciens were used to infect A. thaliana by the floral-dipping method41. T1 seeds were collected and grown under sterile conditions on media containing half-strength MS basal salt mixture, 1% sucrose and 40 mg. L−1 kanamycin. The surviving T2 seedlings showed a ratio of 3:1 KanR/KanS and were selected to produce T3 seeds. T3 lines containing the EPSPS gene were considered homozygous and used for further analysis. Three lines of each transgenic A. thaliana were used for glyphosate dose response analysis or GUS activity assay. Wild-type (WT) A. thaliana was used as a control.
To investigate the role of EPSPS in glyphosate, the seeds of transgenic EPSPS and WT A. thaliana were planted on plates containing half-strength MS salts and glyphosate (1.0 mM), respectively. The subsequent growth of these plants was assessed visually and photographed at 14 d after seeding.
Quantitative analysis of GUS activity
To further investigate the EPSPS expression pattern, the EPSPS promoters from the two species were amplified using specific primers (ChEPS-Pf × ChEPS-Pr and CaEPS-Pf × CaEPS-Pr) (Table 1). The sequencing-verified promoters were isolated from pMD19-T using HindIII/XbaI digestion and then inserted into the pBI121 vector to generate EPSPS-P::GUS. The recombinant vectors were then verified by restriction digest. Expression vectors of ChEPSPS-P::GUS and CaEPSPS-P::GUS were finally introduced into A. thaliana. The method of plant transformation was described as above.
The GUS activity assay in transgenic A. thaliana seedlings used the methods described by Huang42. The data represent the means ± SD of triplicate measurements.
Statistical analysis
Nonlinear regression analysis and ANOVA were used to determine dose–response curves for each species. The data were expressed as a percentage of dry weight compared to untreated control plants. Data from two repeated experiments with similar results were pooled. The GR50 was estimated by nonlinear regression using the logistic curve model:
In this equation, a is the difference between the upper and lower response limits, GR50 is the glyphosate dose that results in a 50% growth reduction, and b is the slope of the curve around GR50. The estimates were obtained using SigmaPlot software (version 12.0), and Tukey’s multiple range tests were used for comparation.
Data from the EPSPS copy number analysis and other experiment results were subjected to ANOVA, and the means were compared using Student’s t-test or Tukey’s multiple range tests. Means with different letters are significantly different at P = 0.05. All statistical analyses were performed using SPSS software (SPSS 17.0, SPSS Institute Inc.).
Results
Whole-plant bioassay
The responses of C. hederacea and C. arvensis to glyphosate were different (Fig. 1). At the glyphosate field rate (1000 g ha−1), the growth of C. hederacea was reduced by approximately 70%, whereas the growth of C. arvensis was reduced by nearly 30%. The C. arvensis plants were not completely controlled by a glyphosate rate of up to 4000 g ha−1. The GR50 values for C. hederacea and C. arvensis were 562.1 and 2,866.3 g ha−1, respectively, and the calculated tolerant index was 5.1.
Dose–response assay of C. hederacea and C. arvensis treated with different glyphosate doses. Dry weight was expressed as a percentage of the untreated control. Each data point represents the mean ± SE of twice-repeated experiments containing three replicates each, and vertical bars represent the standard error.
Shikimic acid accumulation
Basal shikimate acid levels were similar (55.1–59.2 µg g−1 FW) for C. hederacea and C. arvensis in our study. Shikimic acid accumulation exceeded the initial levels of untreated plants after glyphosate application (1000 g ha−1), and both species accumulated shikimate acid until 6 DAT. However, the two species thereafter differed in shikimate accumulation at 6 DAT, accumulation decreased in C. arvensis but fluctuated in C. hederacea (Fig. 2). Shikimic acid accumulation in C. hederacea (with a peak of 326.2 µg g−1 FW at 6 DAT) was 3.5 times higher than that in C. arvensis at 6 DAT.
Shikimic acid accumulation in C. hederacea and C. arvensis after glyphosate treatments. Each data point represents the mean of twice-repeated experiments containing three replicates each, and vertical bars represent standard errors of the means. Means with different letters are significantly different at P = 0.05.
Sequence analysis of EPSPS
Full-length EPSPS cDNAs were isolated from C. hederacea and C. arvensis (ChEPSPS, EU526078; CaEPSPS, EU698030) using specific primers. Sequence analysis revealed that both ChEPSPS and CaEPSPS consisted of a 1,563 bp open reading frame (ORF) encoding a polypeptide of 520 amino acids. The deduced amino acid sequences shared high similarity (identity was 97.31%). There are 14 different amino acids in EPSPS between the two species, and 6 sites were conserved (Fig. 3a). However, there were no point mutations, such as those mainly found at positions 102 or 106 in EPSPS, which have previously been associated with glyphosate resistance.
Fragments of 1,077 bp and 1,142 bp upstream of the ChEPSPS and CaEPSPS genes, respectively, were obtained by genome walking and designated as promoter regions (named ChEPSPS-P and CaEPSPS-P, respectively). PlantCARE analysis of ChEPSPS-P showed that a TATA box at −40 to −36 and three CAAT boxes at −350 to −152 were included in the promoter. Furthermore, a putative cis-acting sp1 element was found within the promoter sequence (Fig. 3b). Sequence analysis of CaEPSPS-P with PlantCARE showed the presence of common core promoter elements, including a “TATA-box” (−40 to −36), six “CAAT-box” (−379 to −156) and many cis-acting elements, such as spl, ARE, and GATA motifs. Furthermore, there was a cis-acting TC-rich repeat element, which is involved in defence and stress responsiveness, located in CaEPSPS-P (Fig. 3b).
Response to glyphosate in transgenic A. thaliana
To investigate the role of CaEPSPS and ChEPSPS in response to glyphosate, three independent transgenic A. thaliana lines expressing either EPSPS gene and WT were assayed. Because the three CaEPSPS or ChEPSPS transgenic A. thaliana lines showed similar tolerance to glyphosate (data not shown), one line of CaEPSPS or ChEPSPS transgenic A. thaliana was selected for imaging. Based on Fig. 4, there was no obvious difference in plant growth among the WT, CaEPSPS and ChEPSPS transgenic A. thaliana in the absence of glyphosate. However, in the presence of glyphosate (1 mg L−1), the WT growth was inhibited, and the cotyledons turned yellow and died. In contrast, the CaEPSPS and ChEPSPS transgenic A. thaliana produced normal plants on Petri dishes and showed similar growth. Thus, the CaEPSPS and ChEPSPS genes similarly conferred the ability to withstand higher glyphosate treatments in transgenic A. thaliana. These results indicate that the amino acid differences in EPSPS were not the cause of glyphosate tolerance in C. arvensis.
Comparison of EPSPS gene copy number and expression level
As we found that the amino acid differences did not account for glyphosate tolerance in C. arvensis, the EPSPS gene copy number in both species was evaluated by qPCR using GAPDH as a normalization gene. The EPSPS copy number in the glyphosate-susceptible C. hederacea ranged from 0.64 to 0.75; however, the glyphosate-tolerant C. arvensis had higher relative EPSPS copy numbers, varying from 1.41 to 1.63 (Fig. 5), showing approximately 2 times higher copy number expression than that of C. hederacea. A higher EPSPS copy number indicated that C. arvensis could produce adequate EPSPS to bind glyphosate, thus conferring higher tolerance compared to C. hederacea.
To examine the expression level of the EPSPS transcript in C. arvensis and C. hederacea, we carried out qPCR analysis with template cDNA derived from plants induced by 1000 g ha−1 glyphosate for different times. As shown in Fig. 6, glyphosate treatment induced a remarkable and steady increase of EPSPS expression in C. arvensis from 0.5 to 1 DAT with nearly 12 times higher peak induction than that of the untreated control, and then the EPSPS transcript level declined. In comparison, glyphosate caused a longer but weaker induction of EPSPS in C. hederacea. The induction began at 0.5 DAT and declined at 2 DAT. The peak induction in C. hederacea was much lower than that in C. arvensis (Fig. 6).
GUS expression from the EPSPS promoter
As the expression levels of the EPSPS genes induced by glyphosate in C. arvensis and C. hederacea were obviously different (Fig. 6), we assumed that the specific promoter was likely associated with the differences in EPSPS expression. Hence, we fused the EPSPS promoters to the GUS gene and transformed the recombinant vectors into A. thaliana plants to further investigate the EPSPS expression regulatory mechanism. The GUS activity in three transgenic A. thaliana lines expressing ChEPSPS-P or CaEPSPS-P was examined at 0.5, 1, 2 and 4 days after glyphosate application. The results showed that there was no significant difference of the GUS activity in the ChEPSPS-P transgenic A. thaliana throughout the experiment. In contrast, the GUS activity of the CaEPSPS-P transgenic A. thaliana was induced at much higher levels by glyphosate from 0.5 to 1 days. The peak induction was detected at 1 day after glyphosate application (Fig. 7). These results indicated that some cis-elements likely exist in CaEPSPS-P that are induced by glyphosate and drive GUS overexpression. This result was consistent with our hypothesis that the overexpression of EPSPS after glyphosate treatment in C. arvensis was likely mediated by a specific EPSPS promoter.
Discussion
Several weeds, including C. arvensis, have been identified with different glyphosate tolerance levels43,44,45,46,47. The recommended glyphosate field doses are commonly 900 to 1500 g ha−1, although these doses vary according to the agronomic management and product marketing of the crops. Thus, C. arvensis (at GR50 level) is tolerant to glyphosate at 1.9–3.2 times the field dose and 5.1 times the level of the glyphosate-susceptible C. hederacea (Fig. 1). To achieve the complete control of C. arvensis, at least double the GR50 rate of glyphosate should be applied; however, this application rate will increase the selection pressure and accelerate the resistance evolution to glyphosate.
Glyphosate affects aromatic amino acid biosynthesis by inhibiting EPSPS, which is a critical enzyme in the shikimate pathway. Previous studies have employed shikimic acid accumulation as a parameter for discriminating glyphosate resistance48,49,50. For example, the inhibition of EPSPS by glyphosate in susceptible weeds usually results in shikimic acid accumulation. Furthermore, glyphosate-tolerant or glyphosate-resistant weeds accumulate shikimate at much lower levels than susceptible plants51. In our study, growth setback and eventual death were observed in C. hederacea owing to shikimic acid accumulation. This effect was due to the complete binding of EPSPS by glyphosate in C. hederacea, resulting in the accumulation of shikimic acid, whereas EPSPS in C. arvensis was not fully inhibited and would still maintain the shikimic pathway, thus leading to normal growth with slight developmental anomalies, such as deformed leaves and shortened internodes. Shikimate accumulation assays indicated that the glyphosate targeting of EPSPS plays a critical role in glyphosate tolerance in C. arvensis. Therefore, EPSPS alteration (mutation or amplification) is likely the major mechanism underlying glyphosate tolerance in C. arvensis.
EPSPS point mutations have been well established as major mechanisms of glyphosate resistance26. Some weeds displaying glyphosate resistance have a site mutation (particularly at the Pro106 codon) in the EPSPS gene26. Recently, E. indica16,17 and Bidens Pilosa18 with a double mutation reported as TIPS (Thr-102-Ile + Pro-106-Ser), have been found to have a high degree of glyphosate resistance. Three amino acid residues (Asp-71-Met, Ala-112-Ile, and Val-201-Met) and a 91Glu deletion in EPSPS were reported to be associated with natural tolerance to glyphosate in three lilyturf species25. In our study, six different amino acid substitutions were discovered in EPSPS in C. arvensis. To investigate the response of different EPSPS proteins to glyphosate, EPSPS genes were inserted into A. thaliana. Glyphosate response assays showed that the two transgenic A. thaliana shared similar glyphosate tolerance levels (Fig. 4). Therefore, target-site mutations are unlikely to account for glyphosate tolerance in C. arvensis.
To examine the possibility of EPSPS overexpression contributing to glyphosate tolerance in C. arvensis, both the basal and induced EPSPS mRNA levels were determined for the two species in this study. The EPSPS copy number for C. arvensis was 2 times higher than that of C. hederacea (Fig. 5). This result alone is not sufficient to explain the tolerance of C. arvensis at the whole plant level. However, the glyphosate-induced expression of the EPSPS gene in C. arvensis was highly enhanced after treatment compared to that in C. hederacea (Fig. 6). Multiple EPSPS copy numbers and/or increased expression of EPSPS have also been reported in other weed species, such as D. chinensis24, O. japonicus25, A. palmeri52,53,54, and Conyza species55,56. Therefore, a higher EPSPS copy number together with increased EPSPS expression likely play an important role in glyphosate tolerance in C. arvensis.
Gene expression is mostly regulated by the promoter. Different promoter regions may have distinctive regulatory functions57. In our study, there was a 77 bp extension in the EPSPS promoter of C. arvensis, which includes three CAAT-boxes. CAAT boxes are known to play important roles in enhancing the transcriptional level of the gene. Moreover, TC-rich repeats, which are involved in defence and stress responsiveness, are located in CaEPSPS-P (Fig. 3b). Thus, cis-elements, such as CAAT-boxes or TC-rich repeats, are likely induced by glyphosate treatment and improve the capacity to respond to glyphosate treatment via feedback regulation. In combination with EPSPS gene amplification, the EPSPS promoter containing specific cis-elements or increased transcription factor activity may increase EPSPS expression and confer glyphosate tolerance in C. arvensis. Further study will be necessary to detect the function of these cis-elements in the glyphosate feedback regulatory mechanism.
Conclusion
We have demonstrated that C. arvensis is naturally tolerant to glyphosate at a much higher dose than glyphosate-susceptible C. hederacea. C. arvensis accumulated less shikimic acids when treated with glyphosate. The EPSPS of C. arvensis shares high similarity with that of C. hederacea, with six different conserved amino acids; however, the response to glyphosate in EPSPS transgenic Arabidopsis assays showed that these plants shared similar glyphosate tolerance levels. We also observed that the EPSPS copy number in C. arvensis was approximately 2 times higher than that of C. hederacea, and the EPSPS mRNA in C. arvensis could be highly induced by glyphosate. We conclude that the underlying basis for the glyphosate tolerance of C. arvensis is primarily due to high EPSPS gene copy numbers and specific promoter-mediated EPSPS overexpression after glyphosate treatment. This study could be of increased importance in weed management if the weeds share a similar glyphosate tolerance mechanism. Our future studies will focus on identifying the putative cis-elements of CaEPSPS-P in the glyphosate feedback regulatory mechanism.
References
Bradshaw, L. D., Padgette, S. R., Kimball, S. L. & Wells, B. H. Perspectives on glyphosate resistance. Weed Technology 111, 89–198 (1997).
Dinelli, G. et al. Physiological and molecular insight on the mechanisms of resistance to glyphosate in Conyza canadensis (L.) Cronq. biotypes. Pesticide Biochemistry and Physiology 86, 30–41 (2006).
Steinrücken, H. C. & Amrhein, N. The herbicide glyphosate is a potent inhibitor of 5-enolpyruvylshikimic acid-3-phosphate synthase. Biochem Bioph Res Co 94, 1207–1212 (1980).
Duke, S. O. & Powles, S. B. Glyphosate: a once-in-a-century herbicide. Pest Manag Sci 64, 319–325 (2008).
Nandula, V. K., Reddy, K. N., Duke, S. O. & Poston, D. H. Glyphosate-resistant weeds: current status and future outlook. Outlooks on Pest Management 16(4), 183–187 (2005).
Powles, S. B. & Christopher, P. Evolved glyphosate resistance in plants: biochemical and genetic basis of resistance. Weed Technology 20(2), 282–289 (2006).
Powles, S. B., Lorraine-Colwell, D. F., Dellow, J. J. & Preston, C. Evolved resistance to glyphosate in rigid ryegrass (Lolium rigidum) in Australia. Weed Science 46, 604–607 (1998).
Heap I. The international survey of herbicide resistant weeds. Available on-line, www.weedscience.com. Accessed Dec 20 (2018).
Baerson, S. R. et al. Glyphosate-resistant goosegrass. Identification of a mutation in the target enzyme 5-enolpyruvylshikimate-3-phosphate synthase. Plant Physiol 129, 1265–1275 (2002).
Wakelin, A. M. & Preston, C. A target-site mutation is present in a glyphosate-resistant Lolium rigidum population. Weed Research 46, 432–440 (2006).
Kaundun, S. S. et al. A novel P106L mutation in EPSPS and an unknown mechanism(s) act additively to confer resistance to glyphosate in a South African Lolium rigidum population. J Agric Food Chem 59, 3227–3233 (2011).
Perez-Jones, A., Park, K. W., Polge, N., Colquhoun, J. & Mallory-Smith, C. A. Investigating the mechanisms of glyphosate resistance in Lolium multiflorum. Planta 226, 395–404 (2007).
Alarcón-Reverte, R. & Fischer, A. J. Resistance to glyphosate in junglerice (Echinochloa colona) from California. Weed Science 61(1), 48–54 (2013).
Bell, M. S., Hager, A. G. & Tranel, P. J. Multiple resistance to herbicides from four site-of-action groups in waterhemp (Amaranthus tuberculatus). Weed Science 61, 460–468 (2013).
Nandula, V. K., Ray, J. D., Ribeiro, D. N., Pan, Z. & Reddy, K. N. Glyphosate resistance in tall waterhemp (Amaranthus tuberculatus) from Mississippi is due to both altered target-site and nontarget-site mechanisms. Weed Science 61, 374–383 (2013).
Chen, J. et al. Mutations and amplification of EPSPS gene confer resistance to glyphosate in goosegrass (Eleusine indica). Planta 242(4), 859–68 (2015).
Yu, Q. et al. Evolution of a double amino acid substitution in the 5-enolpyruvylshikimate-3-phosphate synthase in Eleusine indica conferring high-level glyphosate resistance. Plant Physiology 167(4), 1440–1447 (2015).
Ricardo, A.-delaC. et al. Target and non-target Site mechanisms developed by glyphosate-resistant Hairy beggarticks (Bidens pilosa L.) populations from Mexico. Frontiers in Plant Science 7, 1492 (2016).
Gaines, T. A. et al. Gene amplification confers glyphosate resistance in Amaranthus palmeri. Proceedings of the National Academy of Sciences of the United States of America 107(3), 1029–1034 (2010).
Salas, R. A. et al. EPSPS gene amplification in glyphosate‐resistant Italian ryegrass (Lolium perenne ssp. multiflorum) from Arkansas. Pest Management Science 68(9), 1223–1230 (2012).
Nandula, V. K. et al. EPSPS amplification in glyphosate-resistant spiny amaranth (Amaranthus spinosus): a case of gene transfer via interspecific hybridization from glyphosate-resistant palmer amaranth (Amaranthus palmeri). Pest Management Science 70(12), 1902 (2014).
Tranel, P. J., Riggins, C. W., Bell, M. S. & Hager, A. G. Herbicide resistances in Amaranthus tuberculatus: a call for new options. J Agric Food Chem 59, 5808–5812 (2011).
Lorentz, L. et al. Characterization of glyphosate resistance in Amaranthus tuberculatus populations. J Agric Food Chem 62, 8134–8142 (2014).
Yuan, C. I., Chaing, M. Y. & Chen, Y. M. Triple mechanisms of glyphosate-resistance in a naturally occurring glyphosate-resistant plant Dicliptera chinensis. Plant Science 163, 543–554 (2002).
Mao, C., Xie, H., Chen, S., Valverde, B. E. & Qiang, S. Multiple mechanism confers natural tolerance of three lilyturf species to glyphosate. Planta 243(2), 321–335 (2016).
Sammons, R. D. & Gaines, T. A. Glyphosate resistance: state of knowledge. Pest Manag. Sci. 70, 1367–1377 (2014).
Vila-Aiub, M. M. et al. Glyphosate resistance in perennial Sorghum halepense (Johnsongrass), endowed by reduced glyphosate translocation and leaf uptake. Pest Manag. Sci. 68, 430–436 (2012).
Ge, X. et al. Vacuolar glyphosate-sequestration correlates with glyphosate resistance in ryegrass (Lolium spp.) from Australia, South America, and Europe: a 31P NMR investigation. J Agric Food Chem 60, 1243–1250 (2012).
Preston, C. & Wakelin, A. M. Resistance to glyphosate from altered herbicide translocation patterns. Pest Manag Sci 64, 372–376 (2008).
Yu, Q., Abdallah, I., Han, H. P., Owen, M. & Powles, S. B. Distinct nontarget site mechanisms endow resistance to glyphosate, ACCase and ALS-inhibiting herbicides in multiple herbicide-resistant Lolium rigidum. Planta 230, 713–723 (2009).
Ge, X. et al. Glyphosate-resistant horseweed made sensitive to glyphosate: low-temperature suppression of glyphosate vacuolar sequestration revealed by 31P NMR. Pest Manag Sci 67, 1215–1221 (2011).
Nol, N., Tsikou, D., Eid, M., Livieratos, I. C. & Giannopolitis, C. N. Shikimate leaf disc assay for early detection of glyphosate resistance in Conyza canadensis and relative transcript levels of EPSPS and ABC transporter genes. Weed Research 52, 233–241 (2012).
González-Torralva, F., Brown, A. P. & Chivasa, S. Comparative proteomic analysis of horseweed (Conyza canadensis) biotypes identifies candidate proteins for glyphosate resistance. Scientific Reports 7, 42565 (2017).
Huang, Z. F. et al. Molecular cloning and characterization of 5-enolpyruvylshikimate-3-phosphate synthase gene from Convolvulus arvensis L. Mol Biol Rep 41, 2077–2084 (2014).
DeGennaro, F. P. & Weller, S. C. Differential sensitivity of field bindweed (Convolvulus arvensis) biotypes to glyphosate. Weed Science 32, 472–476 (1984).
Westwood, J. H. & Weller, S. C. Absorption and translocation of glyphosate in tolerant and susceptible biotypes of field bindweed (Convolvulus arvensis L). Weed Science 45(4), 658–663 (1997).
Westwood, J. H. & Weller, S. C. Cellular mechanisms influence differential glyphosate sensitivity in field bindweed (Convolvulus arvensis). Weed Science 45(1), 2–11 (1997).
Zhang, M., Liu, Y., Zhang, C. X., Wei, S. H. & Huang, H. J. Glyphosate-tolerant mechanisms in field bindweed Convolvulus arvensis. Acta Phytophylacica Sinica, 38 ( 6 ), 551–556 (in Chinese) (2011).
Meinke, D. W., Cherry, J. M., Dean, C., Rounsley, S. D. & Koornneef, M. Arabidopsis thaliana: A Model Plant for Genome Analysis. Science 282, 662–668 (1998).
Chen, J. C., Huang, H. J., Wei, S. H., Zhang, C. X. & Huang, Z. F. Characterization of glyphosate-resistant goosegrass (Eleusine indica) populations in china. Journal of Integrative Agriculture 14(5), 919–925 (2015).
Clough, S. J. & Bent, A. F. Floral dip: a simplified method for Agrobacterium-mediated transformation of Arabidopsis thaliana. Plant J 16, 735–743 (1998).
Huang, Z. F. et al. Isolation and functional analysis of Convolvulus arvensis EPSPS promoter. Plant Molecular Biology Reporter 33, 1650–1658 (2015).
Boerboom, C. M., Wyse, D. L. & Somers, D. A. Mechanism of glyphosate tolerance in birdsfoot trefoil (Lotus corniculatus). Weed Science 38, 463–467 (1990).
Culpepper, A. S. Glyphosate-induced weed shifts. Weed Technol 20, 277–281 (2006).
Owen, M. D. K. & Zelaya, I. A. Herbicide-resistant crops and weed resistance to herbicides. Pest Manag Sci 61, 301–311 (2005).
Westhoven, A. M. et al. Characterization of selected common lambsquarters (Chenopodium album) biotypes with tolerance to glyphosate. Weed Sci 56, 685–691 (2008).
Yuan, C. I., Chen, Y. M. & Chaing, M. Y. Responses of Dicliptera chinensis to glyphosate. Plant Prot Bull Taiwan 43, 29–38 (2001).
Carvalho, L. B. et al. Pool of resistance mechanisms to glyphosate in Digitaria insularis. J. Agric. Food Chem. 60, 615–622 (2012).
Cross, R. et al. A Pro106 to Ala substitution is associated with resistance to glyphosate in annual bluegrass (Poa annua). Weed Sci. 63, 613–622 (2015).
Ngo, T. D., Krishnan, M., Boutsalis, P., Gill, G. & Preston, C. Target-site mutations conferring resistance to glyphosate in feathertop rhodes grass (Chloris virgata) populations in Australia. Pest Manag. Sci. 74, 1094–1100 (2018).
Singh, B. K. & Shaner, D. L. Rapid determination of glyphosate injury to plants and identification of glyphosate-tolerant plants. Weed Technol 12, 527–530 (1998).
Ribeiro, D. N. et al. Involvement of facultative apomixis in inheritance of EPSPS gene amplification in glyphosate-resistant Amaranthus palmeri. Planta 239, 199–212 (2014).
Chandi, A. et al. Inheritance of evolved glyphosate resistance in a North Carolina Palmer amaranth (Amaranthus palmeri) biotype. Int J Agron. 2012 (3) (2012).
Gaines, T. A. et al. Mechanism of resistance of evolved glyphosate resistant Palmer amaranth (Amaranthus palmeri). J Agric Food Chem 59, 5886–5889 (2011).
Dinelli, G. et al. Physiological and molecular insight on the mechanisms of resistance to glyphosate in Conyza canadensis L. Cronq Biotypes Pestic Biochem Physiol 86, 30–41 (2006).
Dinelli, G. et al. Physiological and molecular bases of glyphosate resistance in Conyza bonariensis biotypes from Spain. Weed Res 48, 257–265 (2008).
Li, Y. et al. Cloning and function analysis of an alfalfa (Medicago sativa L.) zinc finger protein promoter MsZPP. Mol Biol Rep 39(8), 8559–8569 (2012).
Acknowledgements
This work was supported by the National Natural Science Foundation of China (31501659), National Key Research and Development Program of China (2018YFD0200602), the Key R&D program of Special Funds for Construction Corps (2018AA006), and Key Laboratory of Integrated Pest Management on Crops in Northwestern Oasis, Ministry of Agriculture (KFJJ20180104).
Author information
Authors and Affiliations
Contributions
C.Z. and Z.H. designed the experiments; Z.H., H.H., C.J. and S.W. did the experiments; Z.H., L.J., Y.L. and S.W. analyzed data and wrote the manuscript.
Corresponding authors
Ethics declarations
Competing Interests
The authors declare no competing interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Huang, Z., Liu, Y., Zhang, C. et al. Molecular basis of natural tolerance to glyphosate in Convolvulus arvensis. Sci Rep 9, 8133 (2019). https://doi.org/10.1038/s41598-019-44583-8
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-019-44583-8
- Springer Nature Limited
This article is cited by
-
Effect of glyphosate and P on the growth and nutrition of Coffea arabica cultivars and on weed control
Scientific Reports (2021)