Abstract
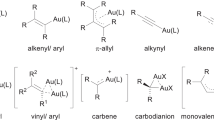
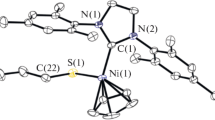
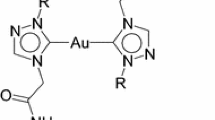
N-heterocyclic carbene gold(I) chloride and hydroxide complexes are regularly used as synthons to access various oxygen-, nitrogen- or carbon-bound gold complexes. They are also widely employed as efficient catalysts in addition reactions of hydroelements to unsaturated bonds and in several rearrangement and decarboxylation protocols. Here we describe the multigram synthesis of the most common mononuclear N-heterocyclic carbene gold(I) chloride complexes bearing the N,N′-bis-(2,4,6-trimethylphenyl)imidazol-2-ylidene (IMes), N,N′-bis(2,6-diisopropylphenyl)imidazol-2-ylidene (IPr) and N,N′-bis(2,6-bis(diphenylmethyl)-4-methylphenyl)imidazol-2-ylidene (IPr*) ligands. Their synthesis is achieved through the straightforward and practical weak base approach in a total time of 4–5 h. This straightforward methodology is conducted under air and possesses considerable advantages over alternative routes, such as the use of a sustainable reaction solvent, minimal amounts of a mild base and commercially available or easily obtained starting materials. Additionally, we describe the synthesis of the mononuclear gold(I) hydroxide complex bearing the IPr ligand, using the state-of-the-art method requiring 24 h. Finally, the improved synthesis of the dinuclear gold(I) hydroxide complex [{Au(IPr)}2(μ-OH)][BF4] is described (~3 h). All procedures can be performed by researchers with standard training and lead to high yields (76–99%) of microanalytically pure bench-stable materials.





Similar content being viewed by others
Data availability
The authors confirm that the data supporting the findings of this study are available within the article, its Supplementary Information and its Source Data files, as well as the primary supporting research papers.
References
Hashmi, A. S. K. The catalysis gold rush: new claims. Angew. Chem. Int. Ed 44, 6990–6993 (2005).
Hashmi, A. S. K. Gold-catalyzed organic reactions. Chem. Rev. 107, 3180–3211 (2007).
Gorin, D. & Toste, F. D. Relativistic effects in homogeneous gold catalysis. Nature 446, 395–403 (2007).
Li, Z., Brouwer, C. & He, C. Gold-catalyzed organic transformations. Chem. Rev. 108, 3239–3265 (2008).
Arcadi, A. Alternative synthetic methods through new developments in catalysis by gold. Chem. Rev. 108, 3266–3325 (2008).
Joost, M., Amgoune, A. & Bourissou, D. Reactivity of gold complexes towards elementary organometallic reactions. Angew. Chem. Int. Ed. 54, 15022–15045 (2015).
Ciriminna, R., Falletta, E., Della Pina, C., Teles, J. H. & Pagliaro, M. Industrial applications of gold catalysis. Angew. Chem. Int. Ed. 55, 14210–14217 (2016).
Schmidbaur, H. & Schier, A. Aurophilic interactions as a subject of current research: an update. Chem. Soc. Rev. 41, 370–412 (2012).
Smith, C. A. et al. N-Heterocyclic carbenes in materials chemistry. Chem. Rev. 119, 4986–5056 (2019).
Pujadas, M. & Rodríguez, L. Luminescent phosphine gold(I) alkynyl complexes. Highlights from 2010 to 2018. Coord. Chem. Rev. 408, 213179 (2020).
Dominelli, B., Correia, J. D. G. & Kühn, F. E. Medicinal applications of gold(I/III)-based complexes bearing N-heterocyclic carbene and phosphine ligands. J. Organomet. Chem. 866, 153–164 (2018).
Qian, D. & Zhang, J. Gold-catalyzed cyclopropanation reactions using a carbenoid precursor toolbox. Chem. Soc. Rev. 44, 677–698 (2015).
Praveen, C. Carbophilic activation of π-systems via gold coordination: towards regioselective access of intermolecular addition products. Coord. Chem. Rev. 392, 1–34 (2019).
Nijamudheen, A. & Datta, A. Gold‐catalyzed cross‐coupling reactions: an overview of design strategies, mechanistic studies, and applications. Chem. Eur. J. 26, 1442–1487 (2019).
Marion, N. & Nolan, S. P. N-Heterocyclic carbenes in gold catalysis. Chem. Soc. Rev. 37, 1776–1782 (2008).
Nolan, S. P. The development and catalytic uses of N-heterocyclic carbene gold complexes. Acc. Chem. Res. 44, 91–100 (2010).
Gaillard, S., Cazin, C. S. J. & Nolan, S. P. N-Heterocyclic carbene gold(I) and copper(I) complexes in C–H bond activation. Acc. Chem. Res. 45, 778–787 (2011).
Xu, Y., Hu, X., Zhang, S., Xi, X. & Wu, Y. Room-temperature hydration of alkynes catalyzed by different carbene gold complexes and their precursors. ChemCatChem 8, 262–267 (2016).
Trinchillo, M. et al. Extensive experimental and computational study of counterion effect in the reaction mechanism of NHC–gold(I)-catalyzed alkoxylation of alkynes. Organometallics 35, 641–654 (2016).
Sarmiento, J. T., Suárez-Pantiga, S., Olmos, A., Varea, T. & Asensio, G. Silica-immobilized NHC–gold(I) complexes: versatile catalysts for the functionalization of alkynes under batch and continuous flow conditions. ACS Catal. 7, 7146–7155 (2017).
Mora, M., Gimeno, M. C. & Visbal, R. Recent advances in gold–NHC complexes with biological properties. Chem. Soc. Rev. 48, 447–462 (2019).
Sak, H., Mawick, M. & Krause, N. Sustainable gold catalysis in water using cyclodextrin‐tagged NHC–gold complexes. ChemCatChem 11, 5821–5829 (2019).
Tugny, C. et al. β-Cyclodextrin–NHC–gold(I) complex (β-ICyD)AuCl: a chiral nanoreactor for enantioselective and substrate-selective alkoxycyclization reactions. ACS Catal. 10, 5964–5972 (2020).
Michalak, M. & Kośnik, W. Chiral N-heterocyclic carbene gold complexes: synthesis and applications in catalysis. Catalysts 9, 890 (2019).
Tang, X.-T. et al. Recent progress in N-heterocyclic carbene gold-catalyzed reactions of alkynes involving oxidation/amination/cycloaddition. Catalysts 10, 350 (2020).
Wang, W., Hammond, G. B. & Xu, B. Ligand effects and ligand design in homogeneous gold(I) catalysis. J. Am. Chem. Soc. 134, 5697–5705 (2012).
Gatineau, D., Goddard, J., Mouriès-Mansuy, V. & Fensterbank, L. When NHC ligands make a difference in gold catalysis. Isr. J. Chem. 53, 892–900 (2013).
Ranieri, B., Escofet, I. & Echavarren, A. M. Anatomy of gold catalysts: facts and myths. Org. Biomol. Chem. 13, 7103–7118 (2015).
de Frémont, P., Scott, N. M., Stevens, E. D. & Nolan, S. P. Synthesis and structural characterization of N-heterocyclic carbene gold(I) complexes. Organometallics 24, 2411–2418 (2005).
Díez-González, S., Marion, N. & Nolan, S. P. N-Heterocyclic carbenes in late transition metal catalysis. Chem. Rev. 109, 3612–3676 (2009).
Scattolin, T. & Nolan, S. P. Synthetic routes to late transition metal–NHC complexes. Trends Chem. 2, 721–736 (2020).
Garrison, J. C. & Youngs, W. J. Ag(I) N-heterocyclic carbene complexes: synthesis, structure, and application. Chem. Rev. 105, 3978–4008 (2005).
Nahra, F., Gómez-Herrera, A. & Cazin, C. S. J. Copper(I)–NHC complexes as NHC transfer agents. Dalton Trans. 46, 628–631 (2017).
Fèvre, M. et al. Imidazol(in)ium hydrogen carbonates as a genuine source of N-heterocyclic carbenes (NHCs): applications to the facile preparation of NHC metal complexes and to NHC-organocatalyzed molecular and macromolecular syntheses. J. Am. Chem. Soc. 134, 6776–6784 (2012).
Zhu, S. et al. A direct and practical approach for the synthesis of Au(I)–NHC complexes from commercially available imidazolium salts and Au(III) salts. Tetrahedron Lett. 53, 815–818 (2012).
Landers, B. & Navarro, O. Microwave-assisted synthesis of (N-heterocyclic carbene)MCl complexes of group 11 metals. Eur. J. Inorg. Chem. 2012, 2980–2982 (2012).
Collado, A., Gómez-Suárez, A., Martin, A. R., Slawin, A. M. Z. & Nolan, S. P. Straightforward synthesis of [Au(NHC)X] (NHC = N-heterocyclic carbene, X = Cl, Br, I) complexes. Chem. Commun. 49, 5541–5543 (2013).
Visbal, R., Laguna, A. & Gimeno, M. C. Simple and efficient synthesis of [MCI(NHC)] (M = Au, Ag) complexes. Chem. Commun. 49, 5642–5644 (2013).
Johnson, A. & Gimeno, M. C. An efficient and sustainable synthesis of NHC gold complexes. Chem. Commun. 52, 9664–9667 (2016).
Santoro, O., Collado, A., Slawin, A. M. Z., Nolan, S. P. & Cazin, C. S. J. A general synthetic route to [Cu(X)(NHC)] (NHC = N-heterocyclic carbene, X = Cl, Br, I) complexes. Chem. Commun. 49, 10483–10485 (2013).
Savka, R. & Plenio, H. Facile synthesis of [(NHC)MX(cod)] and [(NHC)MCl(CO)2] (M = Rh, Ir; X = Cl, I) complexes. Dalton Trans. 44, 891–893 (2015).
Zinser, C. et al. A simple synthetic entryway into palladium cross-coupling catalysis. Chem. Commun. 53, 7990–7993 (2017).
Guillet, S. G. et al. Understanding existing and designing novel synthetic routes to Pd-PEPPSI-NHC and Pd-PEPPSI-PR3 pre-catalysts. Chem. Commun. 56, 5953–5956 (2020).
Tzouras, N. V. et al. A mechanistically and operationally simple route to metal–N‐heterocyclic carbene (NHC) complexes. Chem. Eur. J. 26, 4515–4519 (2020).
Gaillard, S., Slawin, A. M. Z. & Nolan, S. P. A N-heterocyclic carbene gold hydroxide complex: a golden synthon. Chem. Commun. 46, 2742–2744 (2010).
Gasperini, D. et al. Gold-acetonyl complexes: from side-products to valuable synthons. Chem. Eur. J. 21, 5403–5412 (2015).
Tzouras, N. V. et al. Simple synthetic routes to N‐heterocyclic carbene gold(I)–aryl complexes: expanded scope and reactivity. Chem. Eur. J. 26, 5541–5551 (2020).
Ramón, R. S., Veenboer, R. M. P., Nahra, F. & Nolan, S. P. N,N′-Bis(2,6-diisopropylphenyl)imidazol-2-ylidene gold(I) chloride. in Encyclopedia of Reagents for Organic Synthesis (Wiley, 2019).
Obradors, C. & Echavarren, A. M. Chloro[1,3‐dihydro‐1,3‐bis(2,4,6‐trimethylphenyl)‐2H‐imidazol‐2‐ylidene]gold. in Encyclopedia of Reagents for Organic Synthesis (Wiley, 2011).
Gómez-Suárez, A. et al. Influence of a very bulky N-heterocyclic carbene in gold-mediated catalysis. Organometallics 30, 5463–5470 (2011).
Nahra, F. et al. Hydrofluorination of alkynes catalysed by gold bifluorides. ChemCatChem 7, 240–244 (2014).
Hussong, M. W., Rominger, F., Krämer, P. & Straub, B. F. Isolation of a non-heteroatom-stabilized gold–carbene complex. Angew. Chem. Int. Ed. 53, 9372–9375 (2014).
de Frémont, P., Singh, R., Stevens, E. D., Petersen, J. L. & Nolan, S. P. Synthesis, characterization and reactivity of N-heterocyclic carbene gold(III) complexes. Organometallics 26, 1376–1385 (2007).
Nahra, F. & Nolan, S. P. [1,3-bis[2,6-bis(1-methylethyl)phenyl]-1,3-dihydro-2H-imidazol-2-ylidene]hydroxy gold. in Encyclopedia of Reagents for Organic Synthesis (Wiley, 2017).
Tkatchouk, E., Mankad, N. P., Benitez, D., Goddard, W. A. & Toste, F. D. Two metals are better than one in the gold catalyzed oxidative heteroarylation of alkenes. J. Am. Chem. Soc. 133, 14293–14300 (2011).
Ibrahim, N., Vilhelmsen, M. H., Pernpointner, M., Rominger, F. & Hashmi, A. S. K. Gold phenolate complexes: synthesis, structure, and reactivity. Organometallics 32, 2576–2583 (2013).
Nun, P., Ramón, R. S., Gaillard, S. & Nolan, S. P. Efficient silver-free gold(I)-catalyzed hydration of alkynes at low catalyst loading. J. Organomet. Chem. 696, 7–11 (2011).
Ramón, R. S. et al. Gold-catalyzed Meyer−Schuster rearrangement: application to the synthesis of prostaglandins. Organometallics 29, 3665–3668 (2010).
Boogaerts, I. I. F. & Nolan, S. P. Carboxylation of C−H bonds using N-heterocyclic carbene gold(I) complexes. J. Am. Chem. Soc. 132, 8858–8859 (2010).
Nun, P., Egbert, J. D., Oliva-Madrid, M.-J. & Nolan, S. P. Gold(I)-catalyzed stereoselective synthesis of alkenyl phosphates by hydrophosphoryloxylation. Chem. Eur. J. 18, 1064–1067 (2011).
Patrick, S. R., Gómez-Suárez, A., Slawin, A. M. Z. & Nolan, S. P. New [Au(NHC)(OH)] complexes for silver-free protocols. Organometallics 33, 421–424 (2013).
Patrick, S. R., Collado, A., Meiries, S., Slawin, A. M. Z. & Nolan, S. P. Synthesis and characterisation of Au(I)-(ITent) complexes. J. Organomet. Chem. 775, 152–154 (2015).
Nahra, F., Patrick, S. R., Collado, A. & Nolan, S. P. A novel route for large-scale synthesis of [Au(NHC)(OH)] complexes. Polyhedron 84, 59–62 (2014).
Gómez-Suárez, A. & Nolan, S. P. Dinuclear gold catalysis: are two gold centers better than one? Angew. Chem. Int. Ed. 51, 8156–8159 (2013).
Hashmi, A. S. K. Dual gold catalysis. Acc. Chem. Res. 47, 864–876 (2014).
Ramón, R. S. et al. [{Au(IPr)}2(μ-OH)]X Complexes: synthetic, structural and catalytic studies. Chem. Eur. J. 17, 1238–1246 (2010).
Gómez-Suárez, A., Oonishi, Y., Meiries, S. & Nolan, S. P. [{Au(NHC)}2(μ-OH)][BF4]: silver-free and acid-free catalysts for water-inclusive gold-mediated organic transformations. Organometallics 32, 1106–6993 (2013).
Oonishi, Y., Gómez-Suárez, A., Martin, A. & Nolan, S. P. Hydrophenoxylation of alkynes by cooperative gold catalysis. Angew. Chem. Int. Ed. 52, 9767–9771 (2013).
Dupuy, S., Gasperini, D. & Nolan, S. P. Highly efficient gold(I)-catalyzed regio- and stereoselective hydrocarboxylation of internal alkynes. ACS Catal. 5, 6918–6921 (2015).
Veenboer, R. M. P., Dupuy, S. & Nolan, S. P. Stereoselective gold(I)-catalyzed intermolecular hydroalkoxlation of alkynes. ACS Catal. 5, 1330–1334 (2015).
Gasperini, D. et al. Gold(I)-catalysed cyclisation of alkynoic acids: towards an efficient and eco-friendly synthesis of γ-, δ- and ϵ-lactones. Adv. Synth. Catal. 358, 3857–3862 (2016).
Gómez-Suárez, A., Dupuy, S., Slawin, A. M. Z. & Nolan, S. P. Straightforward synthetic access to gem-diaurated and digold σ,π-acetylide species. Angew. Chem. Int. Ed. 52, 938–942 (2012).
Gagosz, F. Gold vinylidenes as useful intermediates in synthetic organic chemistry. Synthesis 51, 1087–1099 (2019).
Ferrer, S. & Echavarren, A. M. Role of σ,π-digold(I) alkyne complexes in reactions of enynes. Organometallics 37, 781–786 (2018).
Hashmi, A. S. K. et al. Dual gold catalysis: σ,π-propyne acetylide and hydroxyl-bridged digold complexes as easy-to-prepare and easy-to-handle precatalysts. Chem. Eur. J. 19, 1058–1065 (2013).
Collado, A. et al. Trapping atmospheric CO2 with gold. Chem. Commun. 50, 11321–11324 (2014).
Nahra, F. & Nolan, S. P. Bis{1,3‐bis[2,6‐bis(1‐methylethyl)phenyl]‐1,3‐dihydro‐2H‐imidazol‐2‐ylidene}‐μ‐hydroxydigold(I) Tetrafluoroborate. in Encyclopedia of Reagents for Organic Synthesis (Wiley, 2017).
Bantreil, X. & Nolan, S. P. Synthesis of N-heterocyclic carbene ligands and derived ruthenium olefin metathesis catalysts. Nat. Protoc. 6, 69–77 (2010).
Chartoire, A. et al. Recyclable NHC catalyst for the development of a generalized approach to continuous Buchwald–Hartwig reaction and workup. Org. Proc. Res. Dev. 20, 551–557 (2016).
Dash, K. C. & Schmidbaur, H. Organogold-Chemie, XII. Komplexe von Gold(I)- und Gold (III)-halogeniden mit Thioäthern. Chem. Ber. 106, 1221–1225 (1973).
Marion, N., Ramón, R. S. & Nolan, S. P. [(NHC)AuI]-catalyzed acid-free alkyne hydration at part-per-million catalyst loadings. J. Am. Chem. Soc. 131, 448–449 (2009).
Gaillard, S. et al. Development of versatile and silver-free protocols for gold(I) catalysis. Chem. Eur. J. 16, 13729–13740 (2010).
Acknowledgements
We are grateful to VLAIO (SBO project CO2PERATE). The Special Research Fund of Ghent University is acknowledged for a doctoral scholarship (01D14919) to N.V.T., as well as starting and project grants to S.P.N. The Research Foundation–Flanders is also gratefully acknowledged for a Fundamental Research PhD fellowship to N.V.T. (11I6921N).
Author information
Authors and Affiliations
Contributions
F.N., N.V.T., A.C. and S.P.N. were involved in the design and optimization of the procedures described here. The manuscript was assembled and edited by all authors.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Peer review information Nature Protocols thanks Carlo Santini and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Related links
Key references using this protocol
Collado, A. et al. Chem. Commun. 49, 5541–5543 (2013): https://doi.org/10.1039/C3CC43076F
Nahra, F. et al. Polyhedron 84, 59–62 (2014): https://doi.org/10.1016/j.poly.2014.06.017
Gaillard, S. et al. Chem. Commun. 46, 2742–2744 (2010): https://doi.org/10.1039/C0CC00018C
Gaillard, S. et al. Chem. Eur. J. 16, 13729–13740 (2010): https://doi.org/10.1002/chem.201001688
Key data used in this protocol
Collado, A. et al. Chem. Commun. 49, 5541–5543 (2013): https://doi.org/10.1039/C3CC43076F
Nahra, F. et al. Polyhedron 84, 59–62 (2014): https://doi.org/10.1016/j.poly.2014.06.017
Gaillard, S. et al. Chem. Eur. J. 16, 13729–13740 (2010): https://doi.org/10.1002/chem.201001688
Gómez-Suárez, A. et al. Organometallics 32, 1106–1111 (2013): https://doi.org/10.1021/om301249r
Marion, N. et al. J. Am. Chem. Soc. 131, 448–449 (2009): https://doi.org/10.1021/ja809403e
Nun, P. et al. J. Organomet. Chem. 696, 7–11 (2011): https://doi.org/10.1016/j.jorganchem.2010.08.052
Supplementary information
Supplementary Information
General considerations and Supplementary Figs. 1–5.
Supplementary Data 1
Raw NMR data for Complex 8 and IR data for Complexes 7 and 8
Rights and permissions
About this article
Cite this article
Nahra, F., Tzouras, N.V., Collado, A. et al. Synthesis of N-heterocyclic carbene gold(I) complexes. Nat Protoc 16, 1476–1493 (2021). https://doi.org/10.1038/s41596-020-00461-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41596-020-00461-6
- Springer Nature Limited