Abstract
Norovirus is an important etiologic agent of acute gastroenteritis and has become even more relevant in Brazil after the implementation of the monovalent rotavirus vaccine in 2006 through the public health system, now representing a significant portion of the etiology of acute diarrheal diseases. Although diagnosing acute gastroenteritis caused by norovirus is a relatively simple process, and the infection tends to be self-limited, the virus can be considerably harmful to vulnerable populations, such as children, the elderly, and immunocompromised individuals. The spread of norovirus is also particularly favorable among such groups due to its mode of transmission, favored by cluttered environments such as in hospitals and densely populated regions. Additionally, norovirus’ ability to spread through water and food creates the need for measures to ensure adequate sanitation and the development of effective measures to prevent outbreaks and severe manifestations of the disease. This review aims to address the main reports of human norovirus detected in Brazil over the years, focusing on clinical-hospital, food-related, and urban conglomerate contexts, including the circulating strains.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Gastroenteritis represents a significant issue worldwide, constituting a debilitating condition that can lead to fatality in various circumstances (mainly because of hydroelectrolytic losses). The etiology of gastroenteritis is diverse, as it can have an infectious origin, caused by viruses, bacteria, protozoa, and so forth, as well as non-infectious causes of diarrhea. Among viral gastroenteritis, norovirus (NoV) plays a substantial role in the promotion of acute gastroenteritis (AGE) outbreaks, alongside rotavirus, adenovirus, and astrovirus [1].
NoV, belonging to the Caliciviridae family, is a non-enveloped, positive-sense, single-stranded RNA virus with a size of ≈ 7.5 kb. It contains three open reading frames (ORFs): ORF1 encodes non-structural proteins, including RNA-dependent RNA polymerase (RdRp); ORF2 is responsible for coding the VP1 capsid protein, while ORF3 encodes a smaller protein, VP2 [2]. Phylogenetic studies of complete amino acid sequences of VP1 have classified NoV into 10 genogroups (GI - GX), including genogroups GI, GII, GIV, GVIII, and GIX, which have been identified in humans (called HuNoV) and can be further divided into 48 genotypes [3].
NoV infection has an average incubation period of 12 to 48 h and usually occurs in early childhood, with most cases of AGE occurring in children under 5 years of age [4, 5]. These infections can often be asymptomatic, especially in the pediatric age group [6]. Due to the generally self-limiting nature of the infection, which rarely lasts more than seven days, hospitalization is necessary only in cases of low tolerance to oral hydration and high fluid losses [7]. Although less common, seizures with generalized or complex partial tonic-clonic patterns can occur in the early symptomatic stage of gastroenteritis and can be a cause for concern [8].
In Brazil, infectious gastroenteritis and diarrhea represent a serious issue. In this regard, it is possible to observe that in the year 2020, the expenses incurred for hospitalizations to treat these conditions amounted to R$ 54,441,885.97, accumulated across all regions of the country [1]. Under this perspective, gastroenteritis represented the primary cause of hospitalization for conditions sensitive to primary care in the northern and northeastern regions of the country from 2008 to 2019 [9]. In these regions, some epidemiological characterization studies highlight a wide range of living conditions and cultural contexts, including communities residing in indigenous reserves, and indicate the prevalence of certain viruses causing AGE (with the highest prevalence of NoV and human adenovirus, followed by rotavirus) [10].
In this context, in 2006, the rotavirus vaccine was introduced into the Brazilian national vaccination schedule, leading to a variation in the presentation of different viral etiologies of AGE, as vaccination rates against rotavirus had been consistently high since the initiation of the immunobiological in the Brazilian national calendar (82% during the 2008–2009 period) [11]. Going back to the years before the introduction of the rotavirus vaccine in Brazil, a study conducted in the Brazilian state of Rio de Janeiro was responsible for showing the presence of three viral pathogens in children with AGE (rotavirus, norovirus, and human astrovirus), which showed again the importance of rotavirus and norovirus as causative agents of AGE (rotavirus was detected in 41.9% of 136 children in the study and NoV got the second place with 30.3%) [12]. Subsequently, some studies observed that the influence of rotavirus as a cause of AGE decreased in various regions of Brazil, with NoV emerging as one of the most prominent causes in the period following the introduction of this vaccine [10, 11, 13].
This transition is relevant, especially considering the challenge represented by the emergence of recombinant variants of NoV in the ORF1/ORF2 junction region, which complicates the identification and characterization of gastroenteritis outbreaks caused by this virus [14]. Genetic recombination among circulating strains can result in a wide range of genotypes, complicating the precise determination of strains responsible for outbreaks. To effectively address this diversity generated by recombinant strains, a dual typing system has been developed. This system considers both the complete amino acid sequences of the VP1 protein and sequences from the RdRp region: (ORF1-RdRp = type P, ORF2 = genotype). As a result, the RdRp designation can be assigned independently of the VP1 genotype from the ORF2 region. For example, a strain may be designated as GII.6 [GII.P7], allowing for a more precise classification of possible recombinant strains [3].
Under this perspective, NoV is a significant contributor to this issue, as it can be transmitted via the oral-fecal route, posing a concern in settings with a high concentration of people, such as hospitals. Furthermore, transmission through water and food also represents a crucial means of virus dissemination [15]. Thus, considering the foodborne transmission of this agent, it can be observed that NoV is the primary pathogen present in this type of infection, emphasizing its significance associated with the consumption of contaminated condiments and in specific contexts such as hospitals, cruises, and other closed environments with a high human congregation [16].
Thus, this review aims to address NoV in the Brazilian context by providing compiled information on reports and outbreaks that have occurred, its behavior in urban environments in Brazil, the impact of NoV on hospitalized and outpatient patients, and its association with foodborne transmission. Furthermore, it seeks to provide information about the genotypes and genetic variants that have spread in Brazilian territory over the years.
Human norovirus detection in the Context of Outpatient clinics and hospitals in Brazil
The significance of HuNoV in the hospital environment arises from its mode of transmission, primarily through the oral-fecal route, which is facilitated in crowded settings with intense interaction among exposed individuals. Furthermore, among the virus’s impacts, the considerable role of noroviruses as an etiological factor in AGE is evident.
Nosocomial HuNoV infections pose a risk to patients, particularly to immunocompromised individuals, the elderly, and children [17]. In this context, an observational study conducted across four countries (Brazil, Chile, Philippines, and Thailand) highlights the severity of nosocomial HuNoV infections in children, indicating that 73% of positive cases in all these countries would be classified as severe using Vesikari score [18]. Besides that, Brazil exhibited the highest proportion of positive symptomatic cases in outpatients and hospitalized treatment (31% in ambulatory treatment, 33% with community-acquired infections requiring hospitalization, and 42% of nosocomial infections) than the Philippines (16% in outpatients, 15% in community-acquired infections requiring hospitalization) and Thailand (18% in outpatients, 9% in community-acquired infections requiring hospitalization) which did not present nosocomial infection [18].
HuNoV infections do not exhibit a seasonal pattern, as described in a study that analyzed epidemiological patterns through data analysis of 1,546 fecal samples from symptomatic outpatients in 10 states across the Northeast (24.3% positive for HuNoV), Southeast (34.3% positive for HuNoV), and South (38.5% positive for HuNoV) regions of Brazil between 2017 and 2018 [19]. Additionally, in the same study, it was possible to observe the age distribution of virus infections, with a higher prevalence among children in the age range of 6 to 24 months, along with the distribution of genogroups (GI with 10.9%, GII with 89.1%, and coinfections with 1.4%) [19].
In the northern region of Brazil, a 2016 study first described the GII.P22/GII.5 and GII.Pg/GII.1 strains in South America and detected the GII.P7/GII.6 strain in northern Brazil from fecal samples obtained during sporadic cases of diarrhea from inpatients who attended public health facilities in Manaus [20]. Another study obtained information from the National Viral Gastroenteritis Surveillance Program, which includes information used in this study from inpatients attended in states from the Northern region of Brazil (Amazonas, Pará, Roraima, Amapá, and Tocantins) between 2015 and 2016. This study includes fecal samples from children under 5 years of age (n = 645) with 208 testing positive for NoV and 95 genotyped. The genotype GII.17_2014 emerged in 2015 with a frequency of 12.6%. The GII.4[P31] genotype was the most prevalent in the period of the study, present in 48/95 of the genotyped samples [21].
Another study analyzing 534 stool samples of people from 6 states (Amazonas, Pará, Roraima, Amapá, Tocantins, and Piauí) treated in the public health system has described a large number of positive cases (n = 170), and children aged 6–12 months were the most affected age group (45%). Moreover, nine genotypes of the HuNoV (GII.P31/GII.4, GII.P16/GII.4, GII.P17/GII.17, GII.P7/GII.6, GII.P33/GII.1, GII.P21/GII.13, GI.P7/GI.7, GII.P4/NT, GII.7/NT) were detected in 74 samples in the years of 2015 (n = 30) and 2016 (n = 44). In both years, GII.P31/GII.4 was the most frequent genotype detected (63.3% − 19/30 in 2015 and 56.8% − 25/44 in 2016) followed by GII⋅P16/GII.4 [22].
Circulation of HuNoV in the Brazilian Amazon has been well established since the 1980s when researches were started involving longitudinal, cross-sectional, randomized clinical trials, and case-control studies, especially in Pará State, a strategic entry route into the Amazon, from other regions of Brazil. In this context, NoV was detected in hospitalized children between 1998 and 2000 (6.8%), 2003 (9.8%), and 2008–2011 (35.4%), including a nosocomial study conducted between 1992 and 1994 at a pediatric hospital showing 14.6% of prevalence to this agent. Outpatients were also evaluated between 1990 and 1992 (15.7%), 2001–2002 (10.2%), and 2004–2005 (26%), showing a broad spectrum of genotypes (more than 30 different types) including a high prevalence of genetic recombinant events [23].
In 2016, the Asian variant GII.17_2014 was described for the first time in Brazil in patients admitted to a large pediatric hospital in Belém (Pará State) with 38.5% of the samples sequenced [24]. In the same year, the GII.P16/GII.4 strain emerged in the Amazon region and was detected in the same city with 16.9% of AGE cases [25].
Other epidemiological studies involving NoV have already been carried out in other more remote regions of the Amazon, such as in a pediatric hospital in Boa Vista (Roraima State) between 2016 and 2017, where the presence of HuNoV in 38% of the cases of AGE in children under 5 years of age was identified [26]. In Rio Branco (Acre State), an overall NoV prevalence of 15.8% was observed in symptomatic patients treated in Emergency Care Units and ambulatory care centers during the year 2012 [27].
In the Amazonas, the largest state in territorial extension in Brazil, it was possible to find HuNoV in 35.2% of children hospitalized with sporadic cases of AGE of all age groups, showing the highest positivity among the 1- to 2-year-old group, between 2010 and 2011 [28]. Also in Amazonas, molecular epidemiology and temporal evolution of AGE-associated NoV in hospitalized children showed high rates of recombination, with rapid changing of their antigenic epitopes in samples collected between 2010 and 2016 [25, 29].
Also in the northern region, a more recent study analyzed stool samples (n = 695) collected from patients aged 0 to 70 years in sentinel units of the Viral Gastroenteritis Surveillance Network (Ministry of Health) from 2018 to 2022 in the Brazilian Amazon. In this study, the majority of sequenced samples belonged to genogroup GII, accounting for 98.1% (52/53) of the cases analyzed. Genogroup GI was identified in 1.9% (1/53) of the cases. The identified genotypes were as follows: GII.P16/GII.4 (56.6% − 30/53); GII.P16/GII.12 (7.5% − 4/53); GII.P7/GII.6 (5.7% − 3/53); GII.P4/GII.4 (3.8% − 2/53); GII.P7/GII.7, GII.P30/GII.3, GII.P17/GII.17, and GII.P13/GI.3 (1.9% − 1/53). Among the samples genotyped by the ORF2 region only, GII.4 was detected in 17% (9/53) and GII.6 in 1.9% (1/53) [30].
The frequency of HuNoV as the causative agent of AGE in a hospital setting is described in Brazil through studies that aim to identify the prevalence rate in these environments, such as the study conducted in Sergipe that aimed to rank the causes of AGE cases in pediatric patients observed in a public hospital, revealing a detection of NoV in 49% of observed cases, with the GII genogroup being particularly predominant (98% prevalence) [31]. Another older study also carried out in the Northeast region of Brazil, had already reported for the first time the incidence of NoV in children hospitalized with AGE in São Luís, Maranhão, with a 32.6% prevalence in diarrheal patients between 1997 and 1999 [32]. A recent study in the Northeast, analyzing fecal samples collected between 2014 and 2017 from Pernambuco state, detected four norovirus GII genotypes: GII.Pe-GII.4, GII.P16-GII.3, GII.P16-GII.4, and GII.P4-GII.4 [33].
Contributing to the manifestation of viral gastroenteritis, the HuNoV is also found in association with other enteropathogens, as described in a study that analyzed fecal samples from hospitalized pediatric patients (134 patients with symptoms of AGE up to 12 years of age) in two different health units in the city of Vitória, in the Brazilian state of Espírito Santo. This finding came with the observation of a prevalence of mixed infections in more than half of the observed cases (51.5% of the detected viral infections) and a strong association with HuNoV GII.4 and rotavirus A (RVA) (9.7%), as well as a significant correlation with human adenovirus (HadV) (6.7%), and even a triple infection with HuNoV GII.4, RVA and HadV (21.6%) [34].
Another study in a pediatric unit, following an AGE outbreak, was also conducted in a hospital in the southeast of Brazil. This time, it was documented that the initial case occurred in a member of the hospital’s professional staff. After this case, the onset of symptoms in the first child occurred three days later, leading to speculation about transmission from this staff member to the patients. In this outbreak, the observed variant is once again GII.4 Sydney, particularly about the ORF1 variation, GII.P31, which is one of the primary variants of GII.4 (GII.4 Sydney[P31]) [35].
A study observed infections with RVA and HuNoV GII.4 Sydney variant in a pediatric hospital in Rio de Janeiro. It was possible to ascertain the high viral load of the HuNoV, a determining factor in its transmissibility to healthcare professionals and patients’ relatives from this hospital. With this in mind, a high viral load of HuNoV GII.4 Sydney (8.4 × 108 genome copies per gram (gc/g) being the highest detected) was observed in the patients with AGE symptoms in the infirmary of pediatrics, which could potentially contribute to an extended length of hospital stay and, consequently, the development of severe AGE cases. Additionally, this could result in a considerable duration of viral shedding and infection [36].
GII is the most prevalent genogroup detected in the context of HuNoV infections in Brazil. However, in another study also conducted in the Brazilian state of Rio de Janeiro it was detected the GIV genogroup by analyzing 316 stool samples collected from inpatients and outpatients from hospitals and central laboratories in the Brazilian public health system. In this study, it was possible to observe, for the first time in a clinical study in Brazil, the presence of the GIV genogroup in three stool samples. Still, it was not possible to say that this genogroup was the cause of the AGE in the affected patients because it was found in association with the GI and GII genogroups [37].
There is a study conducted in Brazil between January 2015 and July 2016 that aimed to analyze 190 fecal specimens from children up to 11 years old and quantify the norovirus and rotavirus A frequencies. These samples were obtained in the Brazilian state of Espírito Santo in a pediatric hospital and a public health facility in the capital of the state. Besides that, three norovirus-positive samples were included and they were collected in the state of Rio de Janeiro, located in the same region of the country. In light of this, 226.8% (n = 51) of samples were positive for GII norovirus genogroup and 37 of them were sequenced, in which 22 were characterized as GII.4[P16] Sydney variant. The GII.P16 was the most prevalent polymerase genotype detected in this context as well (n = 22) [38].
A general overview of the different genotypes identified in infections in the context of Brazilian outpatient clinics and hospitals can be seen better in Table 1.
Norovirus reports and outbreaks Associated with contaminated Food Consumption in Brazil
Foodborne transmission can take place through in situ contamination of food-by-food handlers or during the initial stages of food production [39]. Unlike bacteria, viruses cannot grow in food, thus, the viral presence in fresh food does not affect its degradation, nor does it compromise the appearance, color, texture, flavor, or smell and, therefore, are undetectable for consumers and pose a health risk [40, 41].
In this context, outbreaks of HuNoV have been documented globally due to the consumption of contaminated oysters and other shellfish [42, 43]. The risk of contracting illness from the consumption of bivalve molluscan shellfish, particularly oysters and clams, can be heightened when these shellfish are ingested raw or only lightly cooked [44]. In Brazil, Moresco et al. (2012) investigated the presence of HuNoV in three shellfish-producing regions in southern Brazil: Sambaqui, Santo Antônio de Lisboa, and Ribeirão da Ilha. These regions are situated in Santa Catarina, a state responsible for over 95% of the national mollusk production. Despite HAdV being the most prevalent virus, detected in 61% of the samples, HuNoV GI was identified in 5% of the samples, while HuNoV GII was not detected in these three areas [45].
In other regions of Brazil, HuNoV GII has also been detected in shellfish, such as Rio de Janeiro (42,3%) [46] and Espírito Santo (27%) [47]. In another study that aimed to evaluate the presence of astrovirus and HuNoV GII in mussels (Mytella falcata) and oysters (Crassostrea brasiliana) collected from the Cananéia Lagoon Complex, São Paulo State, HuNoV GII was detected in 21 samples (14%), with 8 mussel samples (38%) and 13 oyster samples (62%). Considering that Cananéia is one of the most important oyster-producing areas in São Paulo, this finding underscores the imperative need for virological surveillance initiatives to ensure the quality and safety of shellfish intended for human consumption [48, 49].
Furthermore, in southern Brazil, outbreaks of HuNoV associated with ice pop consumption have been documented. Through the investigation conducted by Fumian et al. (2021), HuNoV contamination in ice pops was ascertained via the collection of ice pops and fecal samples from symptomatic patients. Five clinical samples tested positive for HuNoV GII. This outbreak affected over 200 individuals across nine municipalities in 2020. Additionally, this study marked the first identification of the recombinant form of norovirus GII.12 [P16] in Brazil [50].
Andrade et al. (2018) investigated an AGE outbreak that occurred after a celebration lunch carried out at Belo Horizonte. Five stool samples of individuals who attended a celebration party and who presented symptoms the day after lunch were collected. Analysis revealed the presence of HuNoV in all of the collected stool samples, with a specific strain, the Asian variant GII.17, linked to the outbreak. All affected individuals exhibited more than two episodes of vomiting and two episodes of diarrhea per day [51].
The occurrence of HuNoV outbreaks associated with cruise ships and the provided food is also important. Morillo et al. (2017) analyzed food and fecal samples collected during an outbreak on a cruise ship that traveled along the Brazilian coast in February 2014. NoV was detected in orange mousse samples. Among the 12 clinical samples analyzed, HuNoV GII.Pe (ORF 1) was identified in five, with the genotype matching that of the contaminated food [52].
Another cruise ship-associated case was reported by Gabbay et al. [53], who described an outbreak of NoV among passengers and crew members aboard a cruise ship that sailed from Rio de Janeiro with stops in the cities of Belém and Manaus, located in the Amazon region, in March 2011. In this case, all seven rectal swabs collected were positive for HuNoV GII. Affected passengers exhibited symptoms of mild to moderate AGE, and only one patient required intravenous rehydration. Seven passengers experienced 10 to 20 episodes of daily evacuations, while two of them had 10 to 20 vomiting episodes [53]. These findings underscore the importance of careful monitoring for NoV outbreaks on cruise ships.
In Brazil, studies have been conducted to evaluate viral and bacterial contamination in typical Brazilian cheeses. Regarding HuNoV, Melgaço et al. (2018) analyzed 90 cheese samples (Minas, Prato, and sliced Prato) commercially obtained in the Greater Metropolitan Region of the State of Rio de Janeiro. These cheeses were randomly acquired in primary packaging from various commercial establishments. HuNoV GI and GII were detected in one sample each of Prato and Minas cheeses at concentrations of 1.5 × 10^6 gc/g and 7.3 × 10^5 gc/g, respectively [41]. Silva et al. (2021) sought to estimate the prevalence of HuNoV and HAdV in 100 samples of Brazilian artisanal raw milk cheeses (Minas and Coalho), each obtained from different producers in four regions in the states of Minas Gerais and one in Piauí. They found 26 strains of HuNoV GI, while HuNoV GII strains were not detected in any cheese samples [54].
Nevertheless, some studies have been produced showcasing the absence of HuNoV in shellfish, such as the study that analyzed 233 mollusks samples at 9 coast cities in Pernambuco (Goiana, Itapissuma, Itamaracá, Sirinhaém, Recife, Cabo de Santo Agostinho, Paulista, Ipojuca, Tamandaré). After the testing (GI and GII), they discovered that no samples were contaminated with HuNoV [55].
In 2022, a study that evaluated commercial mussels and oysters purchased from a producer and a few local markets in the state of Rio de Janeiro for the presence of enteric viruses found that norovirus was the most frequent contaminant throughout the 12-month period during which the samples were collected. Out of 134 samples, 54 (40.3%) tested positive for NoV, of which 4 were identified as belonging to the GI genotype (2.9% out of the 134 collected), 27 samples (20.1%) to the GII genotype, and 23 samples (17.2%) tested positive for both GI and GII genotypes. Further genetic analysis was attempted on the 27 samples that contained only GII viruses, of which 13 (48.1%) were successfully sequenced [56].
The most prevalent genotype was GII.17, identified in 58.3% (7 out of 13) of the samples, followed by GII.2 and GII.6, both found in 4 samples each and lastly GII.9 and GII.27, found in a single sample each. This study concluded that bivalves can be an important source of contamination, allowing not only human infections but also the introduction of new variants into human communities due to the high variety of strains that tend to accumulate inside oysters and mussels, as evidenced by the rise in outbreaks caused by GII.17 variants, which was the most common genotype in bivalves from Rio de Janeiro [56].
An overview of the different norovirus genotypes identified in reports and outbreaks associated with the consumption of contaminated food in Brazil can be better seen in Table 2.
Norovirus Detection in the context of Brazilian Urban communities
HuNoV outbreaks have been reported, by several studies, in crowded environments, such as daycare centers [57, 58], nursing homes [59], prisons [60], and cruise ships [53]. Viral spread can be facilitated in such environments due to the virus’ transmission modes which include person-to-person contact, fecal-oral route, fomites, or ingestion of contaminated water and food [60]. Consequently, urban communities are particularly vulnerable to the spread of pathogens due to their great population density and the fact that a significant portion of the inhabitants rely on the same sources of water and food. Especially when considering the phylogenetic variety of the circulating strains detected in the northern territory of Brazil, particularly with the emergence of new variants such as GII.2[P16]. The detection of this emerging strain in the northern region of the country is a warning sign for potential spread to other areas [61]. Therefore, investigating the presence of HuNoV samples in urban environments and its role in AGE outbreaks is paramount to the development of effective strategies to combat infections.
Corroborating the possibility that urbanization favors HuNoV dissemination, a study analyzing the presence of enteric viruses on Engenho Novo River`s water alongside its course throughout the transition zone between native forest and urbanized regions in the metropolitan region of Rio de Janeiro city has found that, although there was no observed change in the physicochemical properties of the samples (except for a slight increase in turbidity in the urban areas), only those collected in points adjacent to inhabited areas were found to be contaminated with HuNoV [62].
The important role that waters play as a transmission mean for HuNoV in urban environments is reiterated by the finding of HuNoV in 8% of the rainwater samples collected after contact with rooftops in low-income densely populated communities in Rio de Janeiro [63]. In the same city, a similar study conducted in high-income neighborhoods did not find either HuNoV GI or GII strains in any of the 48 samples analyzed, which were obtained from faucets that dispensed rainwater from a collection system that gathered water after it had been in contact with rooftops, gutters, balconies, and indoor floors [64].
Another study conducted in Rio de Janeiro´s Wastewater Treatment Plant between 2013 and 2014 analyzed sewage samples and characterized the HuNoV presentation in this environment. The specimens (156 samples) were collected before and after water treatment and tested for HuNoV. In total, GI genogroups were found in 42 samples, and GII in 103. Though samples from all stages of treatment tested positive for NoV, in final effluent water samples (water that had undergone the complete treatment process) NoV was only found in 1 out of 52 (GI) and 3 out of 52 (GII) samples. The study analyzed raw sewage samples for GII genetic diversity and detected 13 different GII capsid genotypes of which were the most prevalent in the following 6 genotypes, listed from most recurrent to least so: GII.4, GII.17 (Kawasaki variant), GII.5, GII.2, GII.3 and GII.1 [65].
In São Paulo, the biggest city in Brazil, HuNoV cases were found in children under 5 years of age in the north, south, east, west, and southwest zones, as well as in neighboring towns [66].
In the context of urbanization and its impact on the frequency of HuNoV in populations, it is possible to observe the presence of these agents even in semi-isolated communities. A study carried out with children under 10 years of age, with and without AGE, reveals the presence of HuNoV only in the diarrheal group, with a frequency of 19.7%, providing epidemiological and molecular information on the circulation of this agent in Afro-descendant communities (quilombola population) in Northern Brazil, between 2008 and 2010 [67]. This research showed different genotypes from those previously detected in studies carried out in the urban area of the metropolitan region of Belém, probably due to the dynamics of entry and exit from the community, which is naturally different from what is observed in easily accessible urban centers.
Although a clear seasonal pattern in the incidence of HuNoV cases has not been established, human behavior must be considered in the epidemiology of this agent. In a study carried out in the warmer months in Rio Branco, Acre, AGE was associated with an increase in the incidence of NoV cases due to a drought that forced inhabitants to hoard their consumption water, often stored in inadequate containers [27].
Other studies in the region reinforce the importance of different types of water for HuNoV dissemination. About 9.4% of the raw sewage and water samples collected at different points such as a river, a bay, or a stream, in Belém, Pará tested positive for HuNoV GIV.1 in a study published in 2015 [68]. These aquatic environments can be used for water consumption, but also for recreation, as demonstrated in the study by Deus et al. (2019), with recreational waters from four estuarine beaches located on Mosqueiro Island, also in the city of Belém, during two years of monitoring (2012 and 2013), showing HuNoV in 37.5% of surface waters [69].
Still in the Amazon Region, a study carried out in 2017 and 2018 on water samples from three rivers located in a mangrove forest, during periods of greater and lesser precipitation, showed no presence of human enteric viruses, including HuNoV [70]. These rivers are known for the community’s aquaculture activities along the mangrove coast, including oyster farming. The absence of pathogenic viruses reveals the small impact of local communities on the region’s rivers, demonstrating a directly proportional correlation between low population density and little environmental impact in river pollution, highlighting the context of urban impact.
In another context of population density, in the city of Vitória, southeastern Brazil, researchers found that 49% of the 134 children diagnosed with AGE in two public health unities between September 2011 and August 2011, who resided in low-income communities in which mangrove waters were used for recreation and fishing, tested positive for HuNoV GII.4 [34].
Moreover, researchers conducted a study in the Brazilian state of Sergipe, in which they analyzed 1432 stool samples collected from children presenting AGE during two periods of time (October 2006 to September 2009 and July 2011 to January 2013). In this study, they detected the HuNoV in 280 of the samples, especially from July 2011 to 2013 (23.9% of the samples collected in this period). They also identified the infants (< 12 months) as the group of children more likely to be infected with HuNoV in this study (167/190 of them). At last, they successfully sequenced 188 of the positive samples and detected GII as the most frequent genogroup (n = 176) followed by GI (n = 12) and GII.4 and GII.2 as the most prevalent genotypes (149/188 and 6/188 respectively) [71].
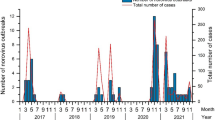
A study conducted in the country showed the behavior of the HuNoV infections during the COVID-19 pandemic. This study was conducted in three Brazilian regions (southern, southeastern, and northeastern) and analyzed 2913 stool samples collected from January 2019 to December 2022 from outpatients presenting AGE symptoms. The norovirus was detected in 37.2% of the samples and they successfully sequenced the HuNov in 602 of the positive samples. Moreover, the most affected age group was the 12 to 24-month-old patients (48.5% of the samples) [72].
The same study exhibited the lowest rate of detection in the same period of the interventional measures to control the spread of SARS-CoV-2, followed by a peak of infection and AGE cases in the second half of 2021 with a HuNoV detection rate of 72.9% in September 2021, which is the year with the highest detection rate (55.4%). In addition, the GII genogroup was the most predominant (92.9%), followed by GI (6%) and GII.4 Sydney[P16] (57.3%) and GII.2[P16] (14.8%) as the most prevalent genotypes. Besides that, they detected six GI polymerase types (GI.P2, GI.P3, GI.P4, GI.P5, GI.P7, and GI.P13) and eight GII polymerase types (GII.P4, GII.P7, GII.P12, GII.P15, GII.P16, GII.P17, GII.P21 and GII.P31) [72].
During another study investigating gastroenteritis outbreaks occurring between 2004 and 2011 in the southern region of Brazil, specifically in the state of Rio Grande do Sul, partial phylogenetic analysis of the ORF1 and ORF2 regions revealed that about 85% of the 27 non-GII.4 samples exhibited recombinant NoV genotypes. The identified genotypes included GIIP.g/GII.12, GII.P16/GII.3, GII.Pe/GII.17, GII.P7/GII.14, GII.P13/GII.17, GII.P21/GII.3, and GII.P21/GII.13. In contrast, the analyzed GII.4 strains did not show recombination, with only the GII.4 variants Den Haag_2006b and New Orleans_2009 being observed [73].
The effect of weather changes on norovirus’ spread can be attributed to several individual variables, such as temperature, humidity, and human behavior [74], the latest being well illustrated by previous examples of contamination through flood waters and poorly stored water during a drought. Furthermore, weather changes can also affect the amount of time people tend to spend inside and in contact with others which can in turn influence norovirus circulation and host immunocompetence [75].
In Brazil, the occurrence of floods may exacerbate outbreaks of norovirus. A study conducted in 2023 investigated an outbreak in Santa Catarina, Brazil, which was linked to storm events that likely increased the discharge of contaminated wastewater and stormwater into bodies of water, such as rivers and beaches. The study identified several uncommon norovirus genotypes among the GI strains, GI.1[P1], GI.4[P4], GI.7[P7], and GI.3[P13] were detected. For GII, the study detected GII.17[P17], GII.4[P16], GII.2[P16], GII.3[P12], GII.7[P7], and GII.8[P8]. The co-circulation of various norovirus genotypes among acute diarrheal disease cases possibly suggests multiple sources of viral infection potentially related to sewage and contaminated water transmission. This demonstrates how norovirus outbreaks are influenced by heavy rainfall and frequent flooding, which can undermine healthcare systems and sanitation infrastructure [74].
In conclusion, despite the lack of concrete evidence for norovirus seasonality in equatorial regions [75], the changes brought up by global warming on its circulation inside national territory should not be discarded, as extreme weather events and changes in climatic factors are major conditioning factors for both human behavior and the pathogen’s interaction with the environment.
An overview of the different norovirus genotypes detected in infections in the context of Brazilian urban communities can be better seen in Table 3.
Conclusion
HuNoV is a highly relevant viral pathogenic agent in Brazil. For years, it caused (and still causes) outbreaks in the country, and its mode of transmission is crucial, especially in crowded environments. Despite this, community transmission through contaminated water and food also plays a significant role in sustaining this problem. These transmission routes can have a wide reach, especially affecting low-income populations in urban and semi-isolated environments. In the context of outbreaks, the importance of the GII genogroup in these episodes is evident. However, other genogroups (GI and GIV) were also detected, showcasing even more genogroups that further complicate global control of the virus. The significance of this pathogen in the described hospital environments is evident, particularly among children and immunocompromised populations. Furthermore, foodborne transmission is an important route for the spread of HuNoV in Brazil and can involve a variety of foods, including shellfish, popsicles, cheese, and foods served on cruise ships. Therefore, the production of new studies on HuNoV is necessary to fully understand the behavior of this agent in different regions of Brazil, which can be achieved by improving surveillance systems across the country.
References
Veras LDL, Soares LWF, d. Silva Neto MR, d. S. Rodrigues MG, d. Silva ACF, Galdino MRL, Pureza SVB, d. Mendonça MHR (2022) Diarrhea and gastroenteritis of presumed infectious origin: analysis of the epidemiological profile in the regions of Brazil from 2012 to 2021. Res Soc Dev 11(7):Artigoe52711730295. https://doi.org/10.33448/rsd-v11i7.30295
Campillay-Véliz CP, Carvajal JJ, Avellaneda AM, Escobar D, Covián C, Kalergis AM, Lay MK (2020) Human norovirus proteins: implications in the replicative cycle, Pathogenesis, and the host Immune Response. Front Immunol 11:961. https://doi.org/10.3389/fimmu.2020.00961
Chhabra P, de Graaf M, Parra GI, Chan MC, Green K, Martella V, Wang Q, White PA, Katayama K, Vennema H, Koopmans MPG, Vinjé J (2019) Updated classification of norovirus genogroups and genotypes. J Gen Virol 100(10):1393–1406. https://doi.org/10.1099/jgv.0.001318
Hossian ME, Islam MM, Miah M, Haque W, Vinjé J, Rahman MZ, Faruque ASG, Khan AI, Ahmed T, Rahman M (2021) Viral etiology of Acute Gastroenteritis among forcibly displaced Myanmar nationals and adjacent host Population in Bangladesh. J Infect Dis 224(12 Suppl 2):S864–S872. https://doi.org/10.1093/infdis/jiab466
Chai SJ, Gu W, O’Connor KA, Richardson LC, Tauxe RV (2019) Incubation periods of enteric illnesses in foodborne outbreaks, United States, 1998–2013. Epidemiol Infect 147:e285. https://doi.org/10.1017/S0950268819001651
Phattanawiboon B, Nonthabenjawan N, Boonyos P, Jetsukontorn C, Towayunanta W, Chuntrakool K, Ngaopravet K, Ruchusatsawat K, Uppapong B, Sangkitporn S, Mekada E, Matsuura Y, Tatsumi M, Mizushima H (2020) Norovirus transmission mediated by asymptomatic family members in households. PLoS ONE 15(7):e0236502. https://doi.org/10.1371/journal.pone.0236502
Cervantes-Izaguirre AP, Rendón-Macías ME, Martina-Luna M, Iglesias-Leboreiro J, Bernárdez-Zapata I, Martínez-Hernández A (2019) Gastroenteritis aguda por norovirus en pacientes con atención hospitalaria: estudio transversal. Boletín Médico Del Hosp Infantil De México 76(4). https://doi.org/10.24875/bmhim.19000006
Hu M-H, Lin K-L, Wu C-T, Chen S-Y, Huang G-S (2017) Clinical characteristics and risk factors for seizures Associated with Norovirus Gastroenteritis in Childhood. J Child Neurol 32(9):810–814. https://doi.org/10.1177/0883073817707302
Santos AdS, Castro LR, Freitas JLG, Cavalcante DFB, Pereira PPdS, Oliveira TMCd, Alves JC (2023) Hospitalizations for ambulatory care-sensitive conditions in children, Rondônia, Brazil, 2008–2019. Ciênc saúde Coletiva 28(4):1003–1010. https://doi.org/10.1590/1413-81232023284.07902022
Olivares AIO, Leitão GAA, Pimenta YC, Cantelli CP, Fumian TM, Fialho AM, da Silva e Mouta S, Delgado IF, Nordgren J, Svensson L, Miagostovich MP, Leite JPG, de Moraes MTB (2021) Epidemiology of enteric virus infections in children living in the Amazon region. Int J Infect Dis 108:494–502. https://doi.org/10.1016/j.ijid.2021.05.060
Sá ACC, Gómez MM, Lima IFN, Quetz JS, Havt A, Oriá RB, Lima AA, Leite JPG (2015) Group a rotavirus and norovirus genotypes circulating in the northeastern Brazil in the post-monovalent vaccination era. J Med Virol 87(9):1480–1490. https://doi.org/10.1002/jmv.24144
Ferreira MSR, Cubel Garcia RdC, Xavier MPTP, Ribeiro RL, Assis RM, Mota MCM, Leite JPG, Miagostovich MP, Oliveira SA (2012) d. Genotipagem de vírus gastroentéricos em crianças hospitalizadas: primeiro relato do norovírus GII.21 no Brasil. Memórias do Instituto Oswald Cross, 107 (8), 1064–1067. https://doi.org/10.1590/s0074-02762012000800017
Razera A, Semaan LM, Ambrosini VA, Santos JR, Carraro E (2022) Acute norovirus gastroenteritis in up to five years children highly rotavirus-vaccinated population in southern Brazil. Res Soc Dev 11(4) Artigo e24611427301. https://doi.org/10.33448/rsd-v11i4.27301
Ludwig-Begall LF, Mauroy A, Thiry E (2018) Norovirus recombinants: recurrent in the field, recalcitrant in the lab– a scoping review of recombination and recombinant types of noroviruses. J Gen Virol 99(8):970–988. https://doi.org/10.1099/jgv.0.001103
Chard AN, Trinies V, Moss DM, Chang HH, Doumbia S, Lammie PJ, Freeman MC (2018) The impact of school water, sanitation, and hygiene improvements on infectious disease using serum antibody detection. PLoS Negl Trop Dis 12(4) Artigo e0006418. https://doi.org/10.1371/journal.pntd.0006418
World Health Organization (2015) WHO estimates of the global burden of foodborne diseases. Geneva: World Health Organization. Retrieved from https://apps.who.int/iris/bitstream/handle/10665/199350/9789241565165_eng.pdf?sequence
Reymão TKA, Fumian TM, Justino MCA, Hernandez JM, Bandeira RS, Lucena MSS, Teixeira DM, Farias FP, Silva LD, Linhares AC, Gabbay YB (2018) Norovirus RNA in serum associated with increased fecal viral load in children: detection, quantification and molecular analysis. PLoS ONE 13(7) Artigo e0199763. https://doi.org/10.1371/journal.pone.0199763
Safadi MA, Riera-Montes M, Bravo L, Tangsathapornpong A, Lagos R, Thisyakorn U, Linhares AC, Capeding R, Prommalikit O, Verstraeten T, O’Ryan M (2021) The burden of norovirus disease in children: a multi-country study in Chile, Brazil, Thailand and the Philippines. Int J Infect Dis 109:77–84. https://doi.org/10.1016/j.ijid.2021.06.037
Sarmento SK, de Andrade JdSR, Miagostovich MP, Fumian TM (2021) Virological and Epidemiological Features of Norovirus Infections in Brazil, 2017–2018. Viruses 13(9):1724. https://doi.org/10.3390/v13091724
Hernandez JDM, Silva LDD, Sousa EC, Junior, Lucena MSS, Soares LDS, Mascarenhas JDP, Gabbay YB (2016) Analysis of uncommon norovirus recombinants from Manaus, Amazon region, Brazil: GII.P22/GII.5, GII.P7/GII.6 and GII.Pg/GII.1. Infection, genetics and evolution: journal of molecular epidemiology and evolutionary genetics in infectious diseases. 39:365–371. https://doi.org/10.1016/j.meegid.2016.02.007
Costa LCP, das N, Teixeira DM, Portela ACR, Lima ICGde, Bandeira RdaS, Júnior ECS, Siqueira JAM, Resque HR, Silva LDda, Gabbay YB (2019) Molecular and evolutionary characterization of norovirus GII.17 in the northern region of Brazil. BMC Infect Dis 19(1). https://doi.org/10.1186/s12879-019-4628-5
Portela AR, Hernandez JM, Bandeira RS, Junior ECS, de Melo TC, Lucena MSS, Teixeira DM, Siqueira JAM, Gabbay YB, Silva LD (2021) Retrospective molecular analysis of norovirus recombinant strains in the amazon region, Brazil. Infect Genet Evol 96:105130. https://doi.org/10.1016/j.meegid.2021.105130
Siqueira JA, Bandeira RS, Oliveira DS, dos Santos LFP, Gabbay YB (2017) Genotype diversity and molecular evolution of noroviruses: a 30-year (1982–2011) comprehensive study with children from Northern Brazil. PLoS ONE 12(6) Article e0178909. https://doi.org/10.1371/journal.pone.0178909
Silva LD, Bandeira RdaS, Junior ECS, de Lima ICG, da Penha Júnior ET, Teixeira DM, Siqueira JAM, Resque HR, de Abreu Campos EMN, Justino MCA, Linhares AC, Gabbay YB (2017) Detection and genetic characterization of the emergent GII.17_2014 norovirus genotype among children with gastroenteritis from Northern Brazil. Infect Genet Evol 48:1–3. https://doi.org/10.1016/j.meegid.2016.11.027
Hernandez JM, Silva LD, Junior ECS, Cardoso JF, Reymão TKA, Portela ACR, Lima CPS, Teixeira DM, Lucena MSS, Nunes MRT, Gabbay YB (2020) Evolutionary and Molecular Analysis of Complete Genome Sequences of Norovirus from Brazil: emerging recombinant strain GII.P16/GII.4. Front Microbiol 11. https://doi.org/10.3389/fmicb.2020.01870
Olivares AIO (2020) Estudo epidemiológico da diarreia aguda, correlação com etiologia viral e antígenos do grupo histo-sanguíneo em crianças ≤ 5 anos atendidas no Hospital da Criança de Santo Antônio em Boa Vista, Roraima, 2016–2017. [PhD Thesis]. https://www.arca.fiocruz.br/handle/icict/44939
Bitencurt ELR, Siqueira JAM, Medeiros TB, Bandeira RS, Oliveira DS, Guimarães RJPS, Soares LS, Mascarenhas JDP, Teixeira DM, Silva RSU, Loureiro ECB, Silva MCM, Silva LD, Gabbay YB (2019) Epidemiological and molecular investigation of norovirus and astrovirus infections in Rio Branco, Acre, Northern Brazil: a retrospective study. J Med Virol 91(6):997–1007. https://doi.org/10.1002/jmv.25395
Costa STP, Fumian TM, Lima ICG, Siqueira JAM, Silva LD, Hernández JM, Lucena MSS, Reymão TKA, Soares LS, Mascarenhas JDP, Gabbay YB (2017) High prevalence of norovirus in children with sporadic acute gastroenteritis in Manaus, Amazon Region, northern Brazil. Mem Inst Oswaldo Cruz 112(6):391–395. https://doi.org/10.1590/0074-02760160357
Hernandez JM, Silva LD, Junior ECS, Bandeira RS, Rodrigues EAM, Lucena MSS, Costa STP, Gabbay YB (2018) Molecular epidemiology and temporal evolution of norovirus associated with acute gastroenteritis in Amazonas state, Brazil. BMC Infect Dis 18(1):147. https://doi.org/10.1186/s12879-018-3068-y
Costa JND, Siqueira JAM, Teixeira DM, Lobo PDS, Guerra SFDS, Souza IM, Cardoso BTM, Farias LSS, Resque HR, Gabbay YB, Silva LDD (2024) Epidemiological and molecular surveillance of norovirus in the Brazilian Amazon: description of recombinant genotypes and improvement of evolutionary analysis, vol 66. Revista do Instituto de Medicina Tropical de Sao Paulo, p e22. https://doi.org/10.1590/S1678-9946202466022
Vicente A (2017) Proporção de rotavírus, norovírus e Cryptosporidium ssp. em crianças com diarreia aguda atendidas no Hospital de Urgências de Sergipe. Ri.ufs.br. http://ri.ufs.br/jspui/handle/riufs/9782
Portal TM, Siqueira JAM, Costa LCPN, Lima ICG, Lucena MSS, Bandeira RS, Linhares AC, Luz CRNE, Gabbay YB, Resque HR (2016) Caliciviruses in hospitalized children, São Luís, Maranhão, 1997–1999: detection of norovirus GII.12. Brazilian J Microbiol 47(3):724–730. https://doi.org/10.1016/j.bjm.2016.04.008
Guarines KM, Mendes RPG, de Magalhães JJF, Pena L (2020) Partial genome sequences of human norovirus strains from Northeast Brazil. Microbiol Resource Announcements 9(1):e01135–e01119. https://doi.org/10.1128/MRA.01135-19
Pratte-Santos R, Miagostovich MP, Fumian TM, Maciel EL, Martins SA, Cassini ST, Keller R (2019) High prevalence of enteric viruses associated with acute gastroenteritis in pediatric patients in a low-income area in Vitória, Southeastern Brazil. J Med Virol 91(5):744–750. https://doi.org/10.1002/jmv.25392
Volpini LPB, Barreira DMPG, Almeida PLDS, Spano LC (2020) An outbreak due to a norovirus GII.Pe-GII.4 Sydney_2012 recombinant in neonatal and pediatric intensive care units. J Infect Public Health 13(1):89–93. https://doi.org/10.1016/j.jiph.2019.06.012
Gutierrez MB, de Figueiredo MR, Fialho AM, Cantelli CP, Miagostovich MP, Fumian TM (2021) Nosocomial acute gastroenteritis outbreak caused by an equine-like G3P[8] DS-1-like rotavirus and GII.4 Sydney[P16] norovirus at a pediatric hospital in Rio De Janeiro, Brazil, 2019. Hum Vaccines Immunotherapeutics 17(11):4654–4660. https://doi.org/10.1080/21645515.2021.1963169
Fioretti JM, Fumian TM, Rocha MS, Dos Santos IAL, Carvalho-Costa FA, de Assis MR, Rodrigues JS, Leite JPG, Miagostovich MP (2018) Surveillance of Noroviruses in Rio De Janeiro, Brazil: occurrence of New GIV genotype in Clinical and Wastewater samples. Food Environ Virol 10(1):1–6. https://doi.org/10.1007/s12560-017-9308-2
Barreira DMPG, Fumian TM, Tonini MAL, Volpini LPB, Santos RP, Ribeiro ALC, Leite JPG, Souza MTB de M. e, Brasil P, da Cunha DC, Miagostovich MP, Spano LC (2017) Detection and molecular characterization of the novel recombinant norovirus GII.P16-GII.4 Sydney in southeastern Brazil in 2016. PLOS ONE, 12(12), e0189504–e0189504. https://doi.org/10.1371/journal.pone.0189504
Rodríguez-Lázaro, D., Cook, N., Ruggeri, F. M., Sellwood, J., Nasser, A., Nascimento,M. S., D’Agostino, M., Santos, R., Saiz, J. C., Rzeżutka, A., Bosch, A., Gironés,R., Carducci, A., Muscillo, M., Kovač, K., Diez-Valcarce, M., Vantarakis, A., von Bonsdorff, C. H., de Roda Husman, A. M., Hernández, M.,… van der Poel, W. H. (2012).Virus hazards from food, water and other contaminated environments. FEMS microbiology reviews, 36(4), 786–814. https://doi.org/10.1111/j.1574-6976.2011.00306.x]
Atreya CD (2004) Major foodborne illness causing viruses and current status of vaccines against the diseases. Foodborne Pathog Dis 1(2):89–96. https://doi.org/10.1089/153531404323143602
Melgaço FG, Luz IS, Assis MRS, Caldas MS, Maranhão AG, Silva DAF, Brandão MLL, Medeiros VM, Rosas CO, Reis SML, Miagostovich MP (2018) Assessment of viral and bacterial contamination of fresh and ripened semi-hard cheeses. FEMS Microbiol Lett 365(20). https://doi.org/10.1093/femsle/fny225
Fouillet A, Fournet N, Forgeot C, Jones G, Septfons A, Franconeri L, Ambert-Balay K, Schmidt J, Guérin P, de Valk H, Caserio-Schönemann C (2020) Large concomitant outbreaks of acute gastroenteritis emergency visits in adults and food-borne events suspected to be linked to raw shellfish, France, December 2019 to January 2020. Euro surveillance: bulletin Europeen sur les maladies transmissibles = European communicable disease bulletin. 25(7):2000060. https://doi.org/10.2807/1560-7917.ES.2020.25.7.2000060
Woods JW, Calci KR, Marchant-Tambone JG, Burkhardt W 3rd (2016) Detection and molecular characterization of norovirus from oysters implicated in outbreaks in the US. Food Microbiol 59:76–84. https://doi.org/10.1016/j.fm.2016.05.009
Pouillot R, Smith M, Van Doren JM, Catford A, Holtzman J, Calci KR, Edwards R, Goblick G, Roberts C, Stobo J, White J, Woods J, DePaola A, Buenaventura E, Burkhardt W (2022) Risk Assessment of Norovirus Illness from Consumption of Raw oysters in the United States and in Canada. Risk Analysis: Official Publication Soc Risk Anal 42(2):344–369. https://doi.org/10.1111/risa.13755
Moresco V, Viancelli A, Nascimento MA, Souza DSM, Ramos APD, Garcia LAT, Simões CMO, Barardi CRM (2012) Microbiological and physicochemical analysis of the coastal waters of southern Brazil. Mar Pollut Bull 64(1):40–48. https://doi.org/10.1016/j.marpolbul.2011.10.026
do Nascimento LG, Sarmento SK, Leonardo R, Gutierrez MB, Malta FC, de Oliveira JM, Guerra CR, Coutinho R, Miagostovich MP, Fumian TM (2022) Detection and molecular characterization of enteric viruses in Bivalve mollusks Collected in Arraial Do Cabo, Rio De Janeiro, Brazil. Viruses 14(11):2359. https://doi.org/10.3390/v14112359
Keller R, Pratte-Santos R, Scarpati K, Martins SA, Loss SM, Fumian TM, Miagostovich MP, Cassini ST (2019) Surveillance of enteric viruses and thermotolerant coliforms in Surface Water and bivalves from a Mangrove Estuary in Southeastern Brazil. Food Environ Virol 11(3):288–296. https://doi.org/10.1007/s12560-019-09391-3
Vasquez-García A, Mejia-Ballesteros JE, de Godoy SHS, Barbieri E, de Sousa RLM, Fernandes AM (2022) Norovirus GII and Astrovirus in shellfish from a mangrove region in Cananéia, Brazil: molecular detection and characterization. Brazilian J Microbiology: [publication Brazilian Soc Microbiology] 53(1):317–326. https://doi.org/10.1007/s42770-021-00631-y
Leal DAG, Souza DSM, Caumo KS, Fongaro G, Panatieri LF, Durigan M, Rott MB, Barardi CRM, Franco RMB (2018) Genotypic characterization and assessment of infectivity of human waterborne pathogens recovered from oysters and estuarine waters in Brazil. Water Res 137:273–280. https://doi.org/10.1016/j.watres.2018.03.024
Fumian TM, Ferreira FC, de Andrade JDSR, Canal N, Silva Gomes G, Teixeira LB, Miagostovich MP (2021) Norovirus Foodborne Outbreak Associated with the consumption of ice pop, Southern Brazil, 2020. Food Environ Virol 13(4):553–559. https://doi.org/10.1007/s12560-021-09495-9
Andrade JSR, Fumian TM, Leite JPG, de Assis MR, Fialho AM, Mouta S, Santiago CMP, Miagostovich MP (2018) Norovirus GII.17 Associated with a foodborne Acute Gastroenteritis Outbreak in Brazil, 2016. Food Environ Virol 10(2):212–216. https://doi.org/10.1007/s12560-017-9326-0
Morillo SG, Luchs A, Cilli A, Ribeiro CD (2017) de Cássia Compagnoli Carmona, R., & do Carmo Sampaio Tavares Timenetsky, M. Norovirus GII.Pe Genotype: Tracking a Foodborne Outbreak on a Cruise Ship Through Molecular Epidemiology, Brazil, 2014. Food and environmental virology, 9(2), 142–148. https://doi.org/10.1007/s12560-016-9272-2
Gabbay YB, Monteiro A, Carlos I, Dielle Monteiro Teixeira D, Gisele C, Daniele, D’Arc J, Alexandre (2014) Norovirus outbreak in a cruise ship along the Brazilian coast, March 2011. Revista Pan-Amazônica de Saúde, 5(1), 43–51. https://doi.org/10.5123/s2176-62232014000100005
Silva MR, Ferreira FC, Maranhão AG, Lanzarini NM, de Carvalho Castro KN, Miagostovich MP (2021) Assessment of viral contamination of five Brazilian Artisanal cheese produced from raw milk: a Randomized Survey. Food Environ Virol 13(4):528–534. https://doi.org/10.1007/s12560-021-09491-z
Guarines KM, Mendes RPG, Cordeiro MT, Miagostovich MP, Gil LHVG, Green KY, Pena LJ (2020) Absence of norovirus contamination in shellfish harvested and commercialized in the Northeast coast of Brazil. Braz J Med Biol Res 53(11). https://doi.org/10.1590/1414-431X20209529
Cantelli CP, Tavares GCL, Sarmento SK, Burlandy FM, Fumian TM, Maranhão AG, Silva ESRF, Horta MAP, Miagostovich MP, Yang Z, Leite JPG (2024) Assessment of Gastroenteric viruses in Marketed Bivalve mollusks in the Tourist cities of Rio De Janeiro, Brazil, 2022. Viruses 16(3):317–317. https://doi.org/10.3390/v16030317
Spano LC, Guerrieri CG, Volpini LPB et al (2021) EHEC O111:H8 strain and norovirus GII.4 Sydney [P16] causing an outbreak in a daycare center, Brazil, 2019. BMC Microbiol 21:95. https://doi.org/10.1186/s12866-021-02161-x
Rodrigues EAM, Teixeira DM, Bandeira RS, Siqueira JAM, Júnior ECS, Lucena MSS, Soares LS, Gabbay YB, Silva LD (2021) Norovirus and Adenovirus among children from Public Day Care Centers in Brazil.Int. J Curr Microbiol App Sci 10(9):431–440. https://doi.org/10.20546/ijcmas.2021.1009.050
Carlos F, de Maria V, Ribeiro G, Fátima MO, Morillo MSG, Rita, Maria, Correa L, Pasternak J (2010) An outbreak of norovirus infection in a long-term care facility in Brazil. 8(4):410–413. https://doi.org/10.1590/S1679-45082010AO1779
Luchs A, Morillo SG, Ribeiro CD, Vilanova BC, Calux SJ, Carmona RdeC, Timenetsky MdoC (2011) Gastroenteritis outbreak due to G2P[4] rotavirus and GII norovirus at two correctional facilities in Brazil, 2010. Journal of clinical virology: the official publication of the Pan American Society for Clinical Virology. 51(3):213–214. https://doi.org/10.1016/j.jcv.2011.04.006
Tinker RJ, da Costa AC, Tahmasebi R, Milagres FAP, Dos Santos Morais V, Pandey RP, José-Abrego A, Brustulin R, Rodrigues Teles MDA, Cunha MS, Araújo ELL, Gómez MM, Deng X, Delwart E, Sabino EC, Leal E, Luchs A (2021) Norovirus strains in patients with acute gastroenteritis in rural and low-income urban areas in northern Brazil. Arch Virol 166(3):905–913. https://doi.org/10.1007/s00705-020-04944-5
Miagostovich MP, Guimarães FR, Vieira CB, Fumian TM, da Gama NP, Victoria M, de Oliveira JM, Mendes AC, Gaspar AM, Leite JP (2014) Assessment of water quality in a border region between the Atlantic forest and an urbanised area in Rio De Janeiro, Brazil. Food Environ Virol 6(2):110–115. https://doi.org/10.1007/s12560-014-9146-4
Shubo T, Maranhão A, Ferreira FC, Fumian TM, Pimenta MMA, do Rosário Vaz Morgado, Toze C, Ahmed S, Sidhu W, J., Miagostovich MP (2021) Virological Characterization of Roof-Harvested Rainwater of Densely Urbanized Low-Income Region. Food and environmental virology, 13(3), 412–420. https://doi.org/10.1007/s12560-021-09484-y
Shubo T, Maranhão AG, Ferreira FC, de Silva EM, Júnior S, de Pedrosa Macena LDG, do Rosário Vaz Morgado, Warish C, Sidhu A, J. P. S., Miagostovich MP (2022) Microbiological characterization of stormwater in a high-income neighborhood in Rio de Janeiro, Brazil. Environmental monitoring and assessment, 194(2), 51. https://doi.org/10.1007/s10661-021-09677-9
Fumian TM, Fioretti JM, Lun JH, Santos D, White IAL, P. A., Miagostovich MP (2019) Detection of norovirus epidemic genotypes in raw sewage using next generation sequencing. Environ Int 123:282–291. https://doi.org/10.1016/j.envint.2018.11.054
Kamioka GA, Madalosso G, Pavanello EI, De Sousa S, Bassit NP, Sato APS (2019) Norovirus in São Paulo city, Brazil, 2010–2016: a cross-sectional study of the leading cause of gastroenteritis in children*. Epidemiol Serv Saude 28(2). https://doi.org/10.5123/s1679-49742019000200016
Aragão GC, Mascarenhas JD, Kaiano JH, de Lucena MS, Siqueira JA, Fumian TM, Hernandez Jd, de Oliveira CS, Oliveira DdeS, Araújo EdaC, Soares LdaS, Linhares AC, Gabbay YB (2013) Norovirus diversity in diarrheic children from an african-descendant settlement in Belém, Northern Brazil. PLoS ONE 8(2):e56608. https://doi.org/10.1371/journal.pone.0056608
Teixeira DM, Hernandez JM, Silva LD, Oliveira DdeS, Spada PK, Gurjão TC, Mascarenhas JD, Linhares AC, Morais LL, Gabbay YB (2016) Occurrence of Norovirus GIV in Environmental Water Samples from Belém City, Amazon Region, Brazil. Food Environ Virol 8(1):101–104. https://doi.org/10.1007/s12560-015-9220-6
Deus DR, Teixeira DM, Dos Santos Alves JC, Smith VC, da Silva Bandeira R, Siqueira JAM, de Sá Morais LLC, Resque HR, Gabbay YB (2019) Occurrence of norovirus genogroups I and II in recreational water from four beaches in Belém City, Brazilian Amazon region. J Water Health 17(3):442–454. https://doi.org/10.2166/wh.2019.304
Teixeira DM, Junior ECS, da Silva LD, de Souza IPP, Passinho FSL, Silva MCM, Nunes MRT, Siqueira JAM, Resque HR, Gabbay YB (2022) Viral composition in metagenomes of rivers located in the amazon mangrove coast, northeast of Pará. Brazil Int J Cu rr Microbiol App Sci. 11(3):1–16. https://doi.org/10.20546/ijcmas.2022.1103.00
Santos VS, Gurgel RQ, Cavalcante SM, Kirby A, Café LP, Souto MJ, Dolabella SS, de Assis MR, Fumian TM, Miagostovich MP, Cunliffe NA, Cuevas LE (2017) Acute norovirus gastroenteritis in children in a highly rotavirus-vaccinated population in Northeast Brazil. Journal of clinical virology: the official publication of the Pan American Society for Clinical Virology. 88:33–38. https://doi.org/10.1016/j.jcv.2016.10.015
Sarmento SK, de Andrade JdaSR, Malta FC, Fialho AM, Mello M, de Burlandy S, F. M., Fumian TM (2023) Norovirus Epidemiology and genotype circulation during the COVID-19 pandemic in Brazil, 2019–2022. Pathogens 13(1):3–3. https://doi.org/10.3390/pathogens13010003
Fumian TM, de Andrade JdaSR, Leite JPG, Miagostovich MP (2016) Norovirus recombinant strains isolated from Gastroenteritis outbreaks in Southern Brazil, 2004–2011. PLoS ONE 11(4):e0145391. https://doi.org/10.1371/journal.pone.0145391
Fumian TM, Malta FC, Sarmento SK, Fernandes SB, Negri CM, Belettini SAA, Machado MH, Guimarães MAAM, de Assis RMS, Baduy GA, Fialho AM, Burlandy FM (2023) Acute gastroenteritis outbreak associated with multiple and rare norovirus genotypes after storm events in Santa Catarina, Brazil. J Med Virol 95(11):e29205. https://doi.org/10.1002/jmv.29205
Rohayem J (2009) Norovirus seasonality and the potential impact of climate change. Clin Microbiol Infect 15(6):524–527. https://doi.org/10.1111/j.1469-0691.2009.02846.x
Funding
The authors have no relevant financial or non-financial interests to disclose.
Author information
Authors and Affiliations
Contributions
Conceptualization: [Caio Augusto Martins Aires]; literature search and data analysis: [João Gabriel dos Santos Oliveira], [Antônio Samuel da Silva Costa], [Igor Vincenzi Ferreira], [Mateus de Oliveira Carvalho]; Writing - original draft preparation: [João Gabriel dos Santos Oliveira], [Antônio Samuel da Silva Costa], [Igor Vincenzi Ferreira], [Mateus de Oliveira Carvalho]; Critical review: [Caio Augusto Martins Aires], [Jones Anderson Monteiro Siqueira].
Corresponding author
Additional information
Responsible Editor: Luis Augusto Nero.
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Oliveira, J.G.d.S., Costa, A.S.d.S., Ferreira, I.V. et al. Human norovirus in Brazil: an update of reports in different settings. Braz J Microbiol 55, 2767–2782 (2024). https://doi.org/10.1007/s42770-024-01444-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s42770-024-01444-5