Abstract
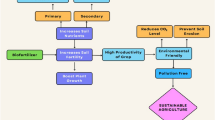
Due to intensive agricultural practices, agricultural lands are subject to continuous degradation. Agricultural productivity depends to a great degree on soil characteristics — soil is the substrate which when managed properly can support plant growth, but when abused requires additional inputs. Research is increasingly focused on the development of new non-synthetic and non-fossil-based products having less impact on the environment and health. In this context, the use of algae biomass (macro and micro) has been widely researched as a fertilizer for agricultural production as a “green economy” alternative product. Currently, the production of marine algae far outstrips the production of microalgae and its main use is for food consumption and some industrial applications. The cost of production is also variable, as marine algae are usually cultivated in integrated multitrophic aquaculture systems. The production of microalgae involves many variables, among them the species, the culture medium, obtaining process, final application, etc. and, based on these, its production cost is estimated. Some researchers point out that the use of simplified technology and increased production capacity tend to reduce operating costs within the ideal photosynthetic yield. This review sought to highlight the importance of algae and their extracts as natural biofertilizers and biostimulants, as well as the mechanisms of action and the possible relationship of these organisms with cultivated plants.
Highlights
• Macroalgae are potentially highly advantageous for sustainable agriculture.
• Microalgae can serve as biofertilizers or biostimulants.
• Algal extracts promote plant growth and nutrients uptake.
• The economic viability of biofertilizer production must be addressed.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
1 Introduction
1.1 Traditional and Contemporary Uses of Macro- and Microalgae
Macroalgae or (seaweeds) and microalgae, covered under the blanket term of “algae,” are a large and diverse group of organisms found preferentially in aquatic environments. With more than 156,000 described species, eukaryotic algae (both macro- and microalgae) are a rich source of biological diversity (Hughes et al. 2021). The macroalgae (seaweed) are multicellular organisms, present in both fresh- and saltwater, classified as green (Chlorophyta), brown (Phaeophyta), and red algae (Rhodophyta) (Overland et al. 2018). They are ecologically important because they help to provide oxygen to the sea and act as one of the main primary producers in the marine food chain (Chan et al. 2006). In recent times, seaweeds are increasingly gaining attention because they are perceived as being an environmentally friendly food, rich in nutrients, due to their contents of micronutrient minerals and vitamins (e.g., calcium, boron, zinc, potassium, phosphorus, magnesium, and several other trace elements) and the only plant-based sources of natural omega-3 long-chain fatty acids (Patel et al. 2021). Also, seaweeds can be used for feed additives, cosmetics, and dietary substitutes and additives, and other species are industrially processed to extract thickening agents such as alginate and agar (Chandía et al. 2004) or bioactive compounds such as lipids, proteins, minerals, carbohydrates, amino acids, antimicrobial compounds, osmoprotectors, and phytohormones (Patel et al. 2021), the latter of which are increasingly used in agriculture. Macroalgae (seaweed) are wild-collected as well as cultivated, and are primarily used as human food, being a traditional product in Asian food. Also, in coastal areas of Europe and the Americas, whether present on beaches or cultivated, seaweed is traditionally consumed and introduced in simple dishes as well as in the modernist kitchen and molecular gastronomy (Mouritsen et al. 2021).
Microalgae are photoautotrophic microorganisms with fast growth and good ability to adapt to different environmental condition (Ortiz-moreno et al. 2019). Microalgae, which comprise eukaryotic organisms and prokaryotic cyanobacteria (blue-green algae), are attracting growing interest because of their simple unicellular structure, high photosynthetic efficiency, ability for heterotrophic growth, adaptability to domestic and industrial wastewater, amenability to metabolic engineering, and potential for yielding valuable co-products (Chiaiese et al. 2018). Microalgae contain many compounds which serve at the biomolecular level for protection and survival in a marine environment. However, only a few microalgal species have been reported to have commercial value. This gap in knowledge of the chemistry produced by microalgae has generated interest in the biotechnology sector since microalgae have been reported to have applications in biofuels, cosmetics, nutraceuticals, and pharmaceuticals (Fernández et al. 2021; Khan et al. 2018; Porcelli et al. 2020). In addition, they can be used in the bioremediation of pollutants by purifying water and, removing heavy metals (Chatzissavvidis and Therios 2014) under pure or mixed strains purification. Algae play an increasingly important role in agriculture where they are used as biofertilizers (Grzesik et al. 2017; Haroun and Hussein 2003). In the literature, Algae play an increasingly important role in agriculture where they are used as biofertilizers (Grzesik et al. 2017; Haroun and Hussein 2003). Specialized metabolites, with promising biological activities, have been widely reported for seaweeds, and more recently for extracts from microalgae, because they maintain or regenerate agricultural soil fertility and also improve high productivity in agricultural crops (Hughes et al. 2021). These compounds are very interesting because they maintain or regenerate agricultural soil fertility and also improve high productivity in agricultural crops.
There is a changing paradigm in agriculture, shifting from focusing solely on direct nutrition to other functions which support plant growth in indirect ways, as well as protecting soil quality. Algae have a special potential in this regard, and since this is a novel field, this review aims to highlight the current state of knowledge to guide research and development. Due to the increasing use of algae-based biostimulants and biofertilizers, this review also highlights the role that algae (macro and micro) may play in maintaining soil fertility and stability, as well as the mechanisms of action and the possible relationship between these organisms and cultivated plants. In addition, the review provides a comprehensive overview of the global production of algae and the estimated cost of transforming algae biomass into biofertilizers.
1.2 Modern Applications of Algae in Agriculture: Focus on Microalgae
Due to intensive agricultural practices, agricultural land is subject to continuous degradation. Agricultural productivity depends essentially on the soil, which represents a substrate for plants and a fundamental resource that requires nutrients to be replenished. However, research is gradually more focused on developing new natural products from aquatic environments with the same efficacy as some synthetic products, but with less impact on the environment and health (Dineshkumar et al. 2018). The use of algae or its extracts as fertilizer can then meet the global challenges of using non-toxic and biodegradable products (Dhargalkar and Pereira 2005; Prakash and Nikhil 2014).
Though an age-old practice, the available information in the literature refers to the application of fresh or dried and chopped seaweed as a soil amendment to enrich plants with nutrients, hormones, and metabolites resulting in increased growth (Prakash and Nikhil 2014; Uysal et al. 2015; Vasileva et al. 2016). Records show that brown algae are generally richer in potassium and the most widely used in agriculture such as, Turbinaria spp., Laminaria spp., Fucus spp., and Sargassum spp. (Hong et al. 2007; Ugarte et al. 2006). Microalgae (such as Scenedesmus, Chlamydomonas, and Chlorococcum) and cyanobacteria have a better capacity to perform a high number of N-transforming reactions compared to other soil microbes (Xiao and Zheng 2016). They allow nitrogen fixation and the accumulation of inorganic phosphate and the production of polyphosphates (Mukherjee et al. 2015; Whitton et al. 1991; Xiao and Zheng 2016). Consequently, the positive effects of algae and their derivatives in agriculture include the resistance of plants to pathogens, tolerance to various stresses, improved seed germination, growth of root systems, and increased plant yield (Chatzissavvidis and Therios 2014; Eyras et al. 1998; Kuwada et al. 2006). The production of microalgal/cyanobacterial biomass can be quite advantageous, when compared to the production of other biological resources. Production systems pose little competition with food production since microalgae can be cultured in non-arable areas and have low requirements for freshwater and nutrients since microalgae can grow in low-quality waters such as wastewaters. Additionally, many species of microalgae and cyanobacteria have the ability to uptake CO2 from the atmosphere, thus reducing the carbon footprint of agricultural practices (Gonçalves 2021).
Uysal et al. (2015) revealed that microbial fertilizers obtained from microalgae increased soil organic matter and water retention capacity. Recently, Abinandan (et al. 2019) investigated the ability of these beneficial microorganisms in maintaining soil fertility and health. Nevertheless, microalgae and cyanobacteria are associated with the development of the top layer of the soil, called “biological soil crusts” and the formation of soil biofilms, which can be triggered by inoculation of soils with one or more organisms (Manjunath et al. 2016; Marks et al. 2017; Rossi et al. 2017). The potential of cyanobacteria and microalgae for stabilization and recovery of degraded soils have been well documented in a number of reviews, as they have been shown to be effective in increasing fertility, stability, and recovery of natural soils, agricultural, burnt areas, and desert areas (Chamizo et al. 2018; Chen et al. 2013; Chock et al. 2019; Wang et al. 2009; Wu et al. 2013; Zaady et al. 2017).
2 Macro- and Microalgae Fertilizing Products on the World Market
In agriculture, soil fertility and health soil are key for productivity, which varies according to soil properties in addition to precipitation and temperature (Cong and Brady 2012). The use of commercial fertilizers has generated impacts on the environment that are increasingly visible and, taking these into consideration, new forms of fertilization are under evaluation in order to minimize negative impacts while maintaining productivity. Among them, the use of algal biomass in the soil stands out.
According to the most recent data provided by the Food and Agriculture Organization of the United Nations (FAO), algae world aquaculture production in wet weight reached 32.4 million tons, which is in monetary terms equivalent to USD 13.3 billion (FAO 2020). Global production of farmed aquatic algae is dominated by marine macroalgae, in contrast to microalgae production which comprises a low portion (FAO 2018a).
Farmed seaweeds corresponded to 97.1% of the total volume produced in 2018, and that year the production lightly fell by 0.7%. Nonetheless, farmed seaweeds have tripled up from 10.6 million tons in 2000 to more than 31 million tons in 2015; subsequent years recorded slower growth until 2018, about 31.6 to 32.4 million tons per year. East and Southeast Asian countries are the largest producers, with major producers being China and Indonesia with 57.1 and 28.8% of the market share, respectively, followed by Republic of Korea, Philippines, and another ten countries summing to just 14.1% of the remaining production (FAO 2020).
The most popular farmed seaweed specie is Japanese kelp (Laminaria japonica) with more than 11.4 million tons produced in 2018; this is consumed as food in dried or pickled form. The second most produced are the tropical seaweeds Eucheuma spp., with 9.2 million tons, used for food processing as well as an ingredient in cosmetics and as raw material for carrageenan extraction. Next are the Gracilaria seaweeds (Gracilaria spp.), Wakame (Undaria pinnatifida), Nori nei (Porphyra spp.), and Elkhorn sea moss (Kappaphycus alvarezii), used in traditional food and some of them in industrial applications, with an annual production between 4 and 1 million tons (FAO 2020).
Seaweed farming is gaining increasing attention to be promoted and monitored for climate and environmentally friendly bioeconomy development around the world (Duarte et al. 2017). However, in agriculture, the application of seaweeds as fertilizers has been very common since ancient times, especially in coastal areas (Mukherjee and Patel 2020). Their use involved the direct application of a liquid solution over the soil, then seaweeds were dried and powdered for easy application. Although the popularization of chemical fertilizer products in the middle of the last century stagnated their use, lately seaweed extracts have become an important alternative in sustainable agriculture (Shah et al. 2013). The fertilizers that are derived from seaweeds are known as new generation fertilizers, containing considerable amount of nutrients which are easily absorbed by plants, and the extracts are used in different ways like seed treatment, foliar spray, and soil application for plant protection and for plant growth promotion (Verma et al. 2020).
Some studies have estimated the cost of production of different seaweeds as part of an integrated multi-trophic aquaculture system, reporting a wide variety of results about its cost-effectiveness. In commercial terms, producers claim that production costs of Saccharina latissima are less than €10–38/kg N removed (Holdt and Edwards 2014). The global seaweed industry has produced more than US$ 6 billion per year and of this amount, 85% comes from its use for food production, and the rest proceed from other industrial applications such as cosmetics, animal feed additives, and fertilizers (FAO 2018b), whereas it is noted that the latter is a growing industry.
In 2018, the microalgae production recorded by FAO were about 87 thousand tons produced from some countries such as Australia, China, Czechia, France, Iceland, India, Israel, Italy, Japan, Malaysia, Myanmar, and the USA. Nonetheless, 86.6 thousand tons was reported from China alone; hence, the FAO data understate the real scale of world microalgae farming because of unavailable data from important producers (FAO 2020).
There are many variables involved in the production process to obtain microalgae biomass, among them is the algae species, wastewater or nutrient influent, culture medium, final application, obtaining process, current available technology, etc. (Acién Fernández et al. 2019). Thus, the production cost depends on all aspects of that process. Some studies carried out to find the most efficient process estimated that the production cost of high value microalgae biomass was 69 €/kg, from tubular photobioreactors operated in continuous mode, with an annual production capacity of 90 t/ha/year. Simplification of the technology and scale-up to a production capacity can allow to reduce the production cost up to 12.6 €/kg. Nonetheless, to achieve an economically feasible process requires a substantial reduction of the current costs and to operate them near the optimal photosynthetic yield (Acién et al. 2012).
Other authors affirm that the key is scaling up the microalgae production, which has a very large impact on the cost. Based on the state-of-the-art technologies to produce microalgae for aquaculture, a cost reduction of 92% could be achieved by changing the scale from 25 to 1500 m2, resulting in a cost of 43 €/kg, producing 3.9 t/year for tubular reactors in a greenhouse (Oostlander et al. 2020).
Regarding agriculture application, another study demonstrates that microalgae biomass production systems are viable technically and economically for the purification of wastewater effluents, producing fertilizer products and biomass for livestock feed. The analysis carried out shows that is more versatile and cost-friendly to reduce digestate turbidity through electro-flocculation method, which has a cost of 0.63 €L−1 (Miñón Martínez 2017).
One of the most expensive process steps is the biomass recovery from the culture medium — harvesting the microalgae may contribute to 20–30% of the total biomass production cost (Molina Grima et al. 2003). It is crucial to understand how to select the right algae species, create an optimal system, and build a cost-effective cultivation unit that can precisely deliver the formula to each individual algae cell, no matter the size of the facility, or its geographical location (Mata et al. 2010).
3 Role of Macroalgae (Seaweed) in Agriculture
Seaweeds are used since antiquity for agricultural applications and current demands of organic agriculture and organic food have considerably encouraged the use of organic treatments including seaweed extracts in agriculture (Nabti et al. 2016). Given its characteristics and the presence of several beneficial components in marine algae, research has been developed aiming at the potential contribution to crop growth and productivity through the modulation of metabolic pathways (Craigie 2011). Thus, increasing attention has been focused on marine-based sources.
The use of algae as a biofertilizers has many benefits, such as stimulating seed germination and enhancing plant health and growth, namely root and shoot elongation (González-Pérez et al. 2022). Moreover, other beneficial effects of seaweed extract applications on plants include better absorption of water and nutrients, resistance to frost and salt, enhancement of the growth of beneficial soil microbes, and enhancing abiotic stress tolerance and defense against pests, diseases, and microorganisms (Chai and Schachtman 2022).
Different fertilizers based on seaweeds are available on the market such as liquid fertilizers and a powder from seaweed manure (Patel et al. 2021). The use of liquid seaweed extracts as foliar applications has been shown to be potent in increasing the growth of many crops, including various herbs, grains, flowers, and vegetables (Kocira et al. 2018; Patel et al. 2021). The brown seaweed Ecklonia maxima collected from the Atlantic coast of South Africa is extensively used as a seaweed liquid additive (Stirk et al. 2014a). Currently, the fertilizers produced from seaweed extracts introduced in the 1950s have reached a wider use and market than seaweed and seaweed meal because of their easy utilization (Renaut et al. 2019), and is also likely related to ease of distribution, concentration of active substances, and factors such as shelf life.
Currently, the increasing number of studies evaluating the importance of seaweeds for their utilization in agriculture field confirms the potential of this organism in the preparation of the extracts used in plant health products and fertilizers.
4 Role of Microalgae in Agriculture
The importance of microalgae in soil formation, improvement of fertility, and increasing crop productivity cannot be underestimated in achieving world food security (Adesalu and Olugbemi 2015; Li et al. 2017; Liu et al. 2017). Microalgae act in the decomposition of organic matter, participating directly in the biogeochemical cycle of nutrients and, consequently, mediating their availability in the soil (Balota et al. 1998; Nayak et al. 2019), in addition to degrading toxic substances (Priya et al. 2014). The use of microalgae biomass can be advantageous because it is rich in nitrogen, as well as benefits of carbon sequestration, improved soil health, soil water retention, stability of soil aggregates, and prevention of nutrient losses (Solé-Bundó et al. 2017).
The application of microalgae biomass to the soil has been investigated and the results point to improvements in chemical characteristics, in the increase of the microbial community, respiration, and photosynthetic activity, in addition to the capacity for bioremediation of pollutants. Currently, research has focused on developing various techniques for introducing microalgae into agricultural production (Alvarez et al. 2021). As described above, microalgae have generated interest due to their great adaptability to various nutrient substrates and their ability to grow under different environmental conditions (Chiaiese et al. 2018; Safi et al. 2014). Due to their phototrophic nature, they contribute to carbon emissions reduction, and may even have a role in soil carbon sequestration since they increase soil organic matter (Jeong et al. 2003; Renuka et al. 2018; Sayre 2010). Microalgae, in addition to being found in aquatic environments, also occur in terrestrial environments and are encountered both on soil surface and within the soil profile (Mathushika and Gomes 2022). This interesting group of microorganisms has applications in modern agriculture since they maintain and release nutrients in plant-assimilable forms, increase soil organic matter levels, and indirectly enhance plant growth and crop yields through stimulation of soil microbial activity (Marks et al. 2019; Renuka et al. 2018).
4.1 Direct Fertilization vs. Indirect Effects
Microalgal biomass is effective for soil fertilization since it releases nutrients into the soil in a form that is adequate for roots and at an appropriate rate (Schreiber et al. 2018); in this sense, their potential nutrient use efficiency can be greater than many chemical fertilizers (Coppens et al. 2016; Mulbry et al. 2005; Renuka et al. 2016; Volf and Rosolem 2020), which can result in less environmental pollution and a higher rate of nutrient utilization. Research has shown that microalgae may be employed as an organic slow-release fertilizer (Coppens et al. 2016; Mulbry et al. 2005, 2007) through a gradual release of N and inorganic P from organic P-compounds in soil to meet plant demands (Mulbry et al. 2007; Ortiz-moreno et al. 2019). Many detailed studies examining the effects of eukaryotic microalgae on soil in terms of impact on some hydrolytic enzymes show that inorganic P concentrations are increased due to increased phytase activity (Zhu and Wakisaka 2020). Microalgae have the ability to fertilize soil through polysaccharides and mucilage’s secretions, which serve as C source (Karthikeyan et al. 2007; Kholssi et al. 2018; Ortiz-moreno et al. 2019). The application of microalgae leads to increased plant growth, reduces effects of saline stress on plants, and increases crop production (Chanda et al. 2019; Elarroussi et al. 2016).
Indirect effects and special applications are also of note: organic fertilization, with the application of microalgae biomass, can affect the biomass, activity, and structure of soil microbial communities through the modification of physical and chemical characteristics (Alvarez et al. 2021; Renuka et al. 2018; Shi et al. 2018). In addition to these characteristics (Ahmad et al. 2017; Cooke and Mouradov 2015; Priya et al. 2014), with microalgae comes the capacity for bioremediation of metallic and organic pollutants. Microalgae contain bioactive compounds and are capable of improving growth (Chanda et al. 2019; Chiaiese et al. 2018; Renuka et al. 2018) and increase the activity of enzymes that participate in the release of nutrients required by plants (De Caire et al. 2000; Rai et al. 2019).
4.2 Microalgae Applications Involving Whole or Viable Cells
Soil health is crucial for sustainable agriculture, and applications of biofertilizers increasing biomass and activities of the microbiota have indirect effects on the availability of soil nutrients and biological processes (Marks et al. 2019). Microalgae applications involving whole or viable cells are differentiated from extracts of algae (biostimulants, next section) since the methods of application may differ and can be used to promote certain outcomes. For horticultural and agronomic crops, their targeted applications have been adopted, depending greatly on the microalgal product and formulation. The application of microalgae as fertilizers includes (i) soil amendment with algal formulations using ideal carriers, (ii) soil amendment with algal dry biomass or suspended liquid culture, and (iii) foliar spray or substrate/soil drench with algal culture (Chiaiese et al. 2018; Coppens et al. 2016; Renuka et al. 2018). Chlorella vulgaris, Chlorella fusca, and Spirulina platensis have been used for various plants such as tomato, cucumber, onion, lettuce, and pepper cultivation with a view to promote their production with marketable quality (Elarroussi et al. 2016; Kim et al. 2018; Rachidi et al. 2020; Zhang et al. 2017).
Al-Maliki and Breesam (2020), assessing, in corn cultivation, the interactions between arbuscular mycorrhizal fungi (Glomus mosseae), algae biomass (Chlamydomonas sp.), and subsequent effects on carbon mineralization, bacterial biomass carbon, soil pH, root density, among others, detected that combination of AM fungi with algae caused carbon decomposition and formation of soil aggregates, reducing pH and increasing bacterial biomass C (carbon), as well as root density. Liu et al. (2019) evaluated in rice fields whether it influences the composition of the biofilm community and whether it drives phosphorus immobilization at the soil–water interface and found, among other conclusions, that the increase in soil organic carbon (SOC) and total nitrogen (STN) is responsible for the great diversity of algae, with dominance of the production of chlorophytes and exopolysaccharides (EPS), and that this scenario allows the increase of phosphorus assimilation and adsorption by phototrophic biofilms in rice fields. Using microalgae in microcosms, Marks et al. (2017) found that in treatments with algae application, soil respiration was increased, and the development of a stable community of eukaryotic and prokaryotic microorganisms on the soil surface was accelerated. Moreover, studies have shown that the growth of several plants was enhanced where microalgae are used (Grzesik and Romanowska-Duda 2014; Kholssi et al. 2018; Mukherjee et al. 2015, 2016; Mulbry et al. 2005; Schreiber et al. 2018). Whether applied fresh as live cell suspensions (Alvarez et al. 2021) or even dried, studies have cited increases in root hair length when compared to the mineral fertilizer (Schreiber et al. 2018). Grzesik et al. (2017) indicated that foliar application with freshwater microalgae such Chlorella sp. produced an increase in growth and physiological activity of willow plants (Ortiz-moreno et al. 2019). Table 1 is an overview of studies carried out in the most important algal strains at laboratory or greenhouse scales, showing the effects of application of cell suspensions (fresh biomass) or dried algal biomass on plant growth.
Besides their effect of enriching soils with essential nutrients and improving plant growth, recent studies have shown that application of photosynthetic algal suspensions not only enhances plant growth but also helps in improving soil biological quality through increasing eukaryotic and prokaryotic biomass and the activities of heterotrophic microorganisms in the soil (Marks et al. 2017). Many other authors have already demonstrated modification of soil microbiota by some cyanobacteria, whose prolific production of extracellular polymeric substances serves as a carbon source for bacteria in the rhizosphere (Manjunath et al. 2016; Priya et al. 2014; Xiao and Zheng 2016). An improvement in the soil structure may be due to the number of polysaccharides such as alginates and fucoidan present in the algal extracts. These can join to metallic ions in soil and help in gel formation, which leads to a better water retention and aggregate structure (Khan et al. 2009). This also facilitates the development of a strong root system that is responsible for nutrient uptake. Increases in the soil indigenous microbial population using N2-fixing cyanophyte Nostoc muscorum were confirmed as well by Rogers (1994), with numbers of bacteria 500, fungi 16, and actinomycetes 48 times compared to non-inoculated soil on day 300. Renuka et al. (2017) evaluated the potential of wastewater-grown microalgal consortia in enhancing the soil micronutrient availability and uptake in wheat crop detected a strong positive correlation and was recorded between the availability of micronutrients in soil at mid-crop and grain yield.
5 Algae-Based Biostimulants
The European Biostimulant Industry Council (EBIC) defines a biostimulant as a material whose content function when applied to plant or soil is to stimulate natural processes to improve nutrient uptake or efficiency and improve abiotic stress tolerance and crop quality, regardless of its nutrient content (Ricci et al. 2019). Use of seaweeds as a fertilizer has been common historically (Mukherjee and Patel 2020), but their recently discovered role as a biostimulant is a new way to understand agricultural applications of these organisms. The growing environmental concern in agriculture, which entails that high production has to be sustainable and minimize its environmental impact, is the reason why the study of algal extracts as biostimulants products is one of the main directions of algae biotechnology (Shekhar et al. 2012). Macroalgae (seaweed) and microalgae have been considered a rich source of plant biostimulants, with macroalgae being more exploited, because for microalgae, although known to have positive effects on crop development, growth, and yield, their commercial implementation is limited due to lack of research and production cost (Kapoore et al. 2021). Even though algal extracts have lately been gained interest in crop production because of their stimulatory effects, the mechanisms underlying these benefits are still unclear (Tan et al. 2021). Seaweed concentrates are typically applied in low doses and show a growth-promoting effect due to their content in plant growth regulators, mainly auxins, brassinosteroids, cytokinins, gibberellins, and abscisic acid (Reed et al. 1998; Nguyen et al. 2020), but also some other purified compounds present in the commercial algae products are laminarin, alginates, carrageenan, polyanionic substances (Du Jardin 2015), abscisic acid, gibberellins, amino acids, betaines (Michalak and Chojnacka 2015), and brassinosteroids (Stirk et al. 2014b). They can be applied directly to plants as a foliar treatment or as a solution to soil. The resulting plant growth promotion could be the result of its synergistic effect (Santner et al. 2009). Table 2 details these main compounds and its application effect.
Several mechanisms have been proposed to explain the improvement shown in terms of nutrient content, plant growth, and higher yield by the application of algal extracts. These can be divided between mechanisms affecting the soil or the plant physiology. Applications targeting plant physiology and biochemistry will usually be applied as a foliar spray, but may also include inoculation of roots or seeds or soil, while applications for improving soil properties are applied as soil drenching.
5.1 Biostimulant Applications Targeting Plant Physiology
There are several studies carried out both in the field and in the greenhouse that show the plant biostimulant properties of algal extracts. Du Jardin (2015) reported that seaweed extract is considered as a natural and organic plant growth stimulant due to its effects on enhancing nutrition efficiency, abiotic stress tolerance (more in following section), and/or crop quality traits. In relation to the literature focused on studying nutrient uptake and plant growth, Almaroai and Eissa (2020) conducted a 2-year field experiment to study the effect of different doses of foliar application of marine algae (Amphora ovalis) extracts on onion. The results showed that the highest dose of the algal extract increased N (nitrogen), P (phosphorus), and K (potassium) uptake by the onions compared to the unsprayed plants. In relation to the literature focused on studying nutrient uptake and plant growth, Turan and Köse (2004) tested the foliar application effect of three algal extracts’ commercial products on grape nutrient uptake. These products showed a significant increase in the Cu uptake when the nutrient availability was low. When in optimal conditions, uptake of the following improved: N, P, K, Ca (calcium), Cu (copper), Fe (iron), Mg (magnesium), Mn (manganese), and Zn (zinc). Rathore et al. (2009) observed an increment of N, P, K, and S (sulfur) concentration in soybean when Kappaphycus alvarezii extracts were foliar applied. This improvement was positively correlated with the dose, with the largest nutrient concentration in the treatment that received the highest dose. Merwad (2020) applied an Ascophyllum extract on wheat under salinity stress and observed a higher straw and grain yield, plant height, and protein content compared to control. Crouch et al. (1990) measured the effect of a commercial algal extract on lettuce growth and Ca, Mg, and K uptake. They reported an enhanced plant growth and nutrient uptake when the plants were also receiving an optimal nutrient solution. Nelson and Van Staden (1984), using the same commercial product, showed a significant increase in root growth and higher N and P content compared to the control treatment. Several studies have reported that plants treated with seaweed extracts had higher mineral contents compared with non-treated plants (Di Filippo-Herrera et al. 2019; Mahmoud et al. 2019; Mikhailyuk et al. 2014).
El-Aziz Kasim et al. (2016) reported that priming of seeds with seaweed extracts enhanced the photosynthetic pigment content in leaves of radish plant. On the other hand, Mahmoud et al. (2019) demonstrated that the application of seaweed extracts led to an enhancement in leaf pigment contents of chlorophyll a, chlorophyll b, chlorophyll a + b, and carotenoids. Similarly, in other studies, the author observed that the application of seaweed Pterocladia capillacea liquid extract improved the concentrations of chlorophyll a, chlorophyll b, and carotene in Jew’s mallow during two successive seasons (Ashour et al. 2020).
Mahmoud et al. (2019) showed that the content of total flavonoids, total phenolics, and anthocyanins in leaves and roots of red radish plants was higher with brown seaweed extract-soaked seeds than water-soaked seeds during the two successive seasons. In previous studies, the authors found that the accumulations of bioactive compounds and antioxidant activity were improved when the brown seaweed was utilized of as biofertilizer, thus increasing the nutritional value of treated plants (El-Aziz Kasim et al. 2016; Uma Maheshwari 2017).
Rayirath et al. (2009) have tested the applicability of using Arabidopsis thaliana as a model to study the activity of two different extracts of algae from A. nodosum and the results of this study suggest that extracts of A. nodosum affect the root growth of Arabidopsis at very low concentrations (0.1 gL−1), whereas plant height and number of leaves were affected at concentrations of 1 gL−1over control plants. Mahmoud et al. (2019) observed the effect of brown seaweed extract of Sargassum vulgare as a natural plant growth stimulant used as a pre-sowing seed soaking. The results indicated that the application of Sargassum vulgare led to greater plant height, number of leaves or plantlets, root diameter, and fresh and dry weights of leaves and roots. In addition, authors reported a direct correlation between foliar spraying of seaweed extract and the enhancement of plant vegetative growth plants (Garai et al. 2021).
González-González et al. (2020) also found out an increased root length and weight along with higher protein concentration in tomato when a seaweed extract was applied. They also reported a synergistic positive effect in the plant when an arbuscular mycorrhizal fungus and the seaweed extract were applied together. Supraja et al. (2020) tested different doses of a mixed consortium of microalgae consisting mainly of Chlorella sp., Scenedesmus sp., Spirulina sp., and Synechocystis sp. This mix was characterized in terms of carbohydrates, proteins, and lipids and was applied as a foliar spray to tomato plants and as a seed primer to tomato seeds. Treated seeds showed a faster germination rate and a faster plant growth. Both applications resulted in a higher total plant height, root length, and biomass content compared to control. Kumar and Sahoo (2011) tested a seaweed liquid extract of Sargassum wightii on the germination rate, growth, and yield of Triticum aestivum. They observed an enhanced growth, yield, and seed germination when a low concentration of the extract was applied. Michalak et al. (2015) used microwave-assisted extraction to acquire liquid extracts from Baltic seaweeds. Results showed higher height, weight, chlorophyll, and micro- and microelement content in treated plants in comparison to the control group. Yao et al. (2020) showed the ability of seaweed extracts to improve yields, leaf photosynthesis, ripening time, and net returns of tomato (Solanum lycopersicum Mill). Spinelli et al. (2010) noted that a commercial extract from Ascophyllum nodosum could be used to replace sequestrene, a standard iron chelate.
An increase in the root shoot ratio has been observed in several crops, regardless of a foliar or a soil application of the extracts. This effect has been reported in cucumber, tomato, barley, raspberry, and bean (Nelson and van Staden 1984; Spinelli et al. 2010; Steveni et al. 1992; Stirk and Van Staden, 1997). Lastly, also reported is promotion of the symbiotic relationships between mycorrhizal fungi and plant roots. This type of fungi plays a very important role in nutrient uptake. The application of algal extracts has shown an increased growth and infection rates of these fungi (Khan et al. 2009). Kuwada et al. (2006) reported that extracts from Gracilaria verrucosa, Gelidium amansii, Eucheuma cottonii, and Chlorella pyrenoidosa successfully stimulated Gigaspora margarita and Glomus caledonium growth in papaya and passion fruit. Other proposed mechanisms regarding the plant physiology include the modulation of root exudates to benefit plant growth–promoting rhizobacteria (PGPR) and growth of other friendly microbes (Chatzissavvidis and Therios 2014).
5.2 Biostimulant Applications to Soil
Application of seaweeds and seaweed extracts improves soil moisture-holding capacity by promoting the growth of beneficial soil microbes which triggers the secretion of soil-conditioning substances by these microbes (Khan et al. 2009). In addition, it may be possible that the algal extract application increases micronutrient availability due to chelation phenomena by the largest organic molecules (Halpern et al. 2015). Research by Ashour et al. (2020) with the brown seaweed M. pyrifera and E. arborea has shown that liquid seaweed extracts can increase soil nutrient content and crop yields for some cultivated plants as they contain micro- and macro-elements which are beneficial for growing plants. Barone et al. (2019) evaluated the effect of Chlorella vulgaris, Scenedesmus quadricauda, or their extracts applied directly into the soil, by monitoring the complex soil-microorganism-plant system showed that both positively affected soil biological activity by increasing values of the biochemical index of potential soil fertility both in cultivated and uncultivated soils.
5.3 Biostimulants and Stress Tolerance
In relation to the role of algal extracts in the enhancement of stress tolerance, several studies have been carried out aiming to observe the effect of its application to different crops (Van Oosten et al. 2017), since algal extracts are known to protect the plant against a biotic/abiotic stress (Michalak et al. 2017). Mansori et al. (2015) attribute this protection against stress to a higher phenolic content, while Shukla et al. (2018) attribute it to an increase in antioxidant activity or even a regulatory role in responsive gene expression. Most of the literature has focused on the study of these products against drought, salinity, or high temperature conditions, but little is known about these effects facing nutrient unbalance and the mechanisms underlying (Carrasco-Gil et al. 2018).
Rayirath et al. (2009) tested the ability of different organic sub-fractions from an A. nodosum extract to promote freezing tolerance of Arabidopsis thaliana, reporting that the lipophilic sub-fractions were the ones increasing this tolerance. Mansori et al. (2015) studied the effect of the red algae Ulva rigida and brown algae Fucus spiralis on drought stress tolerance in green bean plants (Phaseolus vulgaris L.). The results revealed that treatment with seaweed extract improved plant growth with and without drought stress conditions in bean plants. Bradáčová et al. (2016) used extracts containing Zn and Mn which were shown to enhance cold stress tolerance, and the authors concluded that their seaweed extract was effective thanks to the increase in those microelements. Carrasco-Gil et al. (2021) investigated tolerance to an Fe deficiency stress in tomato when different compounds from algal extracts were applied in different doses. They reported that the lowest dosage of phenolics, laminarin, and fucose compounds helped to alleviate this stress. Regarding salinity stress, Bonomelli et al. (2018) found that the application of A. nodosum extracts could alleviate the plant growth suppression caused by saline stress in avocado plants, but only at an early stage. Hussein et al. (2021) tested the impact of seaweed liquid extracts on seed germination of Zea mays and Vigna sinensis, finding a positive correlation between the application of these extracts and higher germination indices, higher growth of seedlings, and reduce the effect of seawater supplementation.
Finally, a considerable number of recent studies have demonstrated that algal extracts can be used to lessen the effects of drought on crops. Martynenko et al. (2016) measured the survival of soybean during 5 days in a drought assay. Leaves from plants treated with a commercial seaweed extract did not wilt (as was the case in the control treatment) and had a better survival rate after rewatering on the fifth day. Santaniello et al. (2017) applied A. nodosum extracts as a pre-treatment to Arabidopsis plants, helping the plant to cope with the stress conditions and improve its water use efficiency.
5.4 Commercial Biostimulants
“Maxicrop” was the first liquid seaweed extract derived from fresh Norwegian kelp (Ascophyllum nodosum) for agricultural use. It was developed in the late 1940s and used for years by organic farmers for its many plant health benefits (Craigie 2011). Currently, there are many other liquid and powdered seaweed extracts based primarily from brown seaweed that are sold as plant biostimulants (Khan et al. 2009). In addition, another commercial biostimulant used in agriculture made from the kelp Ecklonia maxima was reported to promote different responses including greater resistance to abiotic and biotic stressors, enhanced root and shoot growth, and higher yields (Stirk et al. 2014a). The polyamines (putrescine and spermine) and ethylene precursor 1-aminocyclopropane-1-carboxylic acid have also been detected in Kelpak® (Nelson and van Staden 1984). Papenfus et al. (2013) demonstrated that Kelpak® improved root growth and thus the higher seedling vigor of nutrient-stressed okra seedlings. In another work, it was found that other groups of plant growth regulators like abscisic acid, gibberellins, and brassinosteroids were present in seaweed extract Kelpak® (Stirk et al. 2014b). Until now, many seaweed extracts are available on the market as commercial products for their utilization for plants: Ekologik R (Chile), Kelpak 66 (South Africa), Maxicrop (UK), Seasol (Australia), Goemill (France), Seamac Ultra Plus Liquid and Turfcomplex (UK), SeaCrop16 (USA), Algamino Plant (Poland), Actiwave R (Italy), and Acadian (Canada) (Khan et al. 2009).
6 Conclusions
Macro- and microalgae contain an incredible wealth of bioactive compounds, some of which are already available in commercial products, and are already playing a role as environmentally friendly bio-based fertilizers. There are a number of key points and topics covered in this review relevant to the application of algae in sustainable agriculture.
Historically, where permitting, algae have been used a source of nutrients for sustaining indigenous or traditional agricultural practices, serving as a testament to the actual nutrient contents of algae. While both micro- and macroalgae have direct fertilizing potential (nutrients), indirect fertilizing product potential (biostimulant) and the exploitation of physiological responses via different biochemical pathways are the focus of current research. Current research is exploring how to incorporate macroalgae and microalgae as part of fertilization plans to improve the health and fertility of agricultural soils. The approach is to stimulate the internal metabolism of plants and protect crops from stress and agents causing damage in the field, strengthening cultivated plants, improving productivity, and optimizing agricultural yields. Also notable is how microalgae can be used to improve soil quality and protection, including the maintenance of soil organic matter.
Though macroalgae have historically dominated algae applications in agriculture due to ease of harvest and preparation (of fertilizers or extracts), microalgae have a number of advantages for large-scale production and application as biofertilizers, in part because their production is decoupled from marine waters and can be linked with other industrial processes. Nonetheless, microalgae production for agriculture or industrial applications is hampered by technological hurdles (harvesting, among others) and sufficient market value.
What has been highlighted is the positioning of microalgae for the production of biostimulants, use as biofertilizers, and potential for alleviating stress. Their contents of active biochemical or inorganic compounds which intervene in plant growth — particularly hormones but also other compounds such as antioxidants, micronutrients, or nutrient supplements — are now of high value in the fertilizing product market.
The use of biofertilizers is rapidly gaining ground in agriculture throughout the world, offering alternatives or products which are complementary to chemical fertilizers, with the aim of maintaining agricultural production and soil fertility while reducing soil degradation and environmental contamination. The beneficial effects of algae and particularly microalgae have been documented, although the prospects for large-scale production and the feasibility of producing inexpensive formulations require more attention. New initiatives should address production methods and applications to increase the cost–benefit ratios and improve market acceptance.
Change history
17 October 2022
A Correction to this paper has been published: https://doi.org/10.1007/s42729-022-01037-x
References
Abinandan S, Subashchandrabose SR, Megharaj M (2019) Critical Reviews in Biotechnology Soil microalgae and cyanobacteria: the biotechnological potential in the maintenance of soil fertility and health. Crit Rev Biotechnol 39:981–998. https://doi.org/10.1080/07388551.2019.1654972
Acién FG, Fernández JM, Magán JJ, Molina E (2012) Production cost of a real microalgae production plant and strategies to reduce it. Biotechnol Adv 30:1344–1353. https://doi.org/10.1016/j.biotechadv.2012.02.005
Acién Fernández FG, Fernández Sevilla JM, Molina Grima E (2019) Costs analysis of microalgae production. in Biofuels from Algae. Editors Pandey A, Chang JS, Soccol CR, Lee DJ, and Chisti Y(Elsevier), 551–566. https://doi.org/10.1016/b978-0-444-64192-2.00021-4
Adesalu TA, Olugbemi OM (2015) Soil algae: a case study of two vegetable farmlands in Lagos and Ogun states, southwest Nigeria. Ife J Sci 17:765–772
Agwa OK, Ogugbue CJ, Williams EE, Agwa OK, Ogugbue CJ, Williams EE (2018) Growth promotion of Telfairia occidentalis by application of Chlorella vulgaris (bioinoculant) colonized seeds and soil under tropical field conditions. Am J Plant Sci 9:403–415. https://doi.org/10.4236/AJPS.2018.93031
Ahmad I, Khan MS, Altaf MM, Qais FA, Ansari FA, Rumbaugh KP (2017) Biofilms: an overview of their significance in plant and soil health. In: Ahmad I, Husain, FM(Eds.), Biofilms in Plant and Soil Health. John Wiley & Sons, Ltd, Chichester, UK, pp. 1–25. https://doi.org/10.1002/9781119246329.ch1
Al-Maliki S, Breesam H (2020) Changes in soil carbon mineralization, soil microbes, roots density and soil structure following the application of the arbuscular mycorrhizal fungi and green algae in the arid saline soil. Rhizosphere 14:100203. https://doi.org/10.1016/j.rhisph.2020.100203
Almaroai YA, Eissa MA (2020) Role of marine algae extracts in water stress resistance of onion under semiarid conditions. J Soil Sci Plant Nutr 20:1092–1101. https://doi.org/10.1007/s42729-020-00195-0
Alvarez AL, Weyers SL, Goemann HM, Peyton BM, Gardner RD (2021) Microalgae, soil and plants: a critical review of microalgae as renewable resources for agriculture. Algal Res 54:102200. https://doi.org/10.1016/j.algal.2021.102200
Ashour M, El-Shafei AA, Khairy HM, Abd-Elkader DY, Mattar MA, Alataway A, Hassan SM (2020) Effect of Pterocladia capillacea seaweed extracts on growth parameters and biochemical constituents of Jew’s Mallow Agron 10:420. https://doi.org/10.3390/agronomy10030420
Balota ELL, Colozzi-Filho A, Andrade DSS, Hungria M (1998) Microbial biomass and its activity in soils under different tillage and crop rotation systems. (In portuguese: Biomassa Microbiana e sua atividade em solos sob diferentes sistemas de preparo e sucessão de culturas). Congr Bras Ciência Do Solo 22:641–649. https://doi.org/10.1590/S0100-06831998000400009
Barone V, Puglisi I, Fragalà F, Stevanato P, Baglieri A (2019) Effect of living cells of microalgae or their extracts on soil enzyme activities. Arch Agron Soil Sci 65:712–726. https://doi.org/10.1080/03650340.2018.1521513
Bonomelli C, Celis V, Lombardi G, Mártiz J (2018) Salt stress effects on avocado (Persea americana Mill.) plants with and without seaweed extract (Ascophyllum nodosum) application. Agron. 8:64. https://doi.org/10.3390/agronomy8050064
Bose SK, Yadav RK, Mishra S, Sangwan RS, Singh AK, Mishra B, Srivastava AK, Sangwan NS (2013) Effect of gibberellic acid and calliterpenone on plant growth attributes, trichomes, essential oil biosynthesis and pathway gene expression in differential manner in Mentha arvensis L. Plant Physiol Biochem 66:150–158. https://doi.org/10.1016/j.plaphy.2013.02.011
Bradáčová K, Weber NF, Morad-Talab N, Asim M, Imran M, Weinmann M, Neumann G (2016) Micronutrients (Zn/Mn), seaweed extracts, and plant growth-promoting bacteria as cold-stress protectants in maize. Chem Biol Technol Agric 3:1–11. https://doi.org/10.1186/s40538-016-0069-1
Carrasco-Gil S, Hernandez-Apaolaza L, Lucena JJ (2018) Effect of several commercial seaweed extracts in the mitigation of iron chlorosis of tomato plants (Solanum lycopersicum L.). Plant Growth Regul 86:401–411. https://doi.org/10.1007/s10725-018-0438-9
Carrasco-Gil S, Allende-Montalbán R, Hernández-Apaolaza L, Lucena JJ (2021) Application of seaweed organic components increases tolerance to fe deficiency in tomato plants. Agronomy 11:507. https://doi.org/10.3390/agronomy11030507
Chai YN, Schachtman DP (2022) Root exudates impact plant performance under abiotic stress. Trends Plant Sci 27:80–91. https://doi.org/10.1016/J.TPLANTS.2021.08.003
Chamizo S, Mugnai G, Rossi F, Certini G, De Philippis R (2018) Cyanobacteria inoculation improves soil stability and fertility on different textured soils: gaining insights for applicability in soil restoration. Front Environ Sci 6:49 https://doi.org/10.3389/fenvs.2018.00049
Chan KMA, Shaw MR, Cameron DR, Underwood EC, Daily GC (2006) Conservation planning for ecosystem services. PLoS Biol 4:e379. https://doi.org/10.1371/journal.pbio.0040379
Chanda MJ, Merghoub N, Arroussi HE (2019) Microalgae polysaccharides: the new sustainable bioactive products for the development of plant bio-stimulants? World J Microbiol Biotechnol 35:1–10. https://doi.org/10.1007/s11274-019-2745-3
Chandía NP, Matsuhiro B, Mejías E (2004) Moenne A (2004) Alginic acids in Lessonia vadosa: partial hydrolysis and elicitor properties of the polymannuronic acid fraction. J Appl Phycol 162(16):127–133. https://doi.org/10.1023/B:JAPH.0000044778.44193.A8
Chatzissavvidis C, Therios I (2014) Role of algae in agriculture. In: Seaweeds. Vitor H. Pomin (Ed.). Nova Science Publishers, Inc. ISBN: 978–1–63117–571–8
Chen L, Deng S, De Philippis R, Tian W, Wu H, Wang J (2013) UV-B resistance as a criterion for the selection of desert microalgae to be utilized for inoculating desert soils. J Appl Phycol 25:1009–1015. https://doi.org/10.1007/s10811-012-9906-1
Chiaiese P, Corrado G, Colla G, Kyriacou MC, Rouphael Y (2018) Renewable sources of plant biostimulation: microalgae as a sustainable means to improve crop performance. Front Plant Sci 871:1–6. https://doi.org/10.3389/fpls.2018.01782
Chock T, Antoninka AJ, Faist AM, Bowker MA, Belnap J, Barger NN (2019) Responses of biological soil crusts to rehabilitation strategies. J Arid Environ 163:77–85. https://doi.org/10.1016/j.jaridenv.2018.10.007
Cong R-G, Brady M (2012) The cientificWorldJOURNAL The interdependence between rainfall and temperature: copula analyses. Sci World J 2012:11. https://doi.org/10.1100/2012/405675
Cooke B, Mouradov A (2015) Microalgae as bioremediators of polluted ecosystems. Curr Biotechnol 4:1–10
Coppens J, Grunert O, Van Den HS, Vanhoutte I, Boon N, Haesaert G, De GL (2016) The use of microalgae as a high-value organic slow-release fertilizer results in tomatoes with increased carotenoid and sugar levels. J Appl Phycol 28:2367–2377. https://doi.org/10.1007/s10811-015-0775-2
Craigie JS (2011) Seaweed extract stimuli in plant science and agriculture. J Appl Phycol 23:371–393. https://doi.org/10.1007/s10811-010-9560-4
Crouch IJ, Beckett RP, van Staden J (1990) Effect of seaweed concentrate on the growth and mineral nutrition of nutrient-stressed lettuce. J Appl Phycol 2:269–272. https://doi.org/10.1007/BF02179784
De Caire GZ, De Cano MS, Palma RM, De Mulé CZ (2000) Short communication: changes in soil enzyme activities following additions of cyanobacterial biomass and exopolysaccharide. Soil Biol Biochem 32:1985–1987. https://doi.org/10.1016/S0038-0717(00)00174-7
Dhargalkar VK, Pereira N (2005) Seaweed: promising plant of the millennium. Sci Cult 71:60–66
Di Filippo-Herrera DA, Muñoz-Ochoa M, Hernández-Herrera RM, Hernández-Carmona G (2019) Biostimulant activity of individual and blended seaweed extracts on the germination and growth of the mung bean. J Appl Phycol 31:2025–2037. https://doi.org/10.1007/S10811-018-1680-2/FIGURES/6
Dineshkumar R, Kumaravel R, Gopalsamy J, Sikder MNA, Sampathkumar P (2018) Microalgae as bio-fertilizers for rice growth and seed yield productivity. Waste Biomass Valorization 9:793–800. https://doi.org/10.1007/s12649-017-9873-5
du Jardin P (2015) Plant biostimulants: definition, concept, main categories and regulation. Sci Hortic (amsterdam) 196:3–14. https://doi.org/10.1016/j.scienta.2015.09.021
Duarte CM, Wu J, Xiao X, Bruhn A, Krause-Jensen D (2017) Can seaweed farming play a role in climate change mitigation and adaptation? Front Mar Sci 4:8. https://doi.org/10.3389/fmars.2017.00100
Elarroussi H, Elmernissi N, Benhima R, Meftah El Kadmiri I, Bendaou N, Smouni A (2016) Microalgae polysaccharides a promising plant growth biostimulant. J Algal Biomass Utln 7:55–63
El-Aziz Kasim WA, Saad-Allah KM, Hamouda M (2016) Seed priming with extracts of two seaweeds alleviates the physiological and molecular impacts of salinity stress on radish (Raphanus sativus). Int J Agric Biol 18:653–660. https://doi.org/10.17957/IJAB/15.0152
Eyras MC, Rostagno CM, Defossé GE (1998) Biological evaluation of seaweed composting. Compost Sci Util 6:74–81. https://doi.org/10.1080/1065657X.1998.10701943
Faheed FA, Fattah ZA-E (2008) Effect of Chlorella vulgaris as bio-fertilizer on growth parameters and metabolic aspects of lettuce plant. J Agric Soc Sci 4:165–169
Food and Agriculture Organization (FAO)(2018a) The state of world fisheries and aquaculture - meeting the sustainable development goals. Rome
Food and Agriculture Organization (FAO) (2018b) The future of food and agriculture - alternative pathways to 2050. Rome
FAO (2020) The State of World Fisheries and Aquaculture 2020. Sustainability in action, Rome
Fernández FGA, Reis A, Wijffels RH, Barbosa M, Verdelho V, Llamas B (2021) The role of microalgae in the bioeconomy. N Biotechnol 61:99–107. https://doi.org/10.1016/j.nbt.2020.11.011
Garai S, Brahmachari K, Sarkar S, Mondal M, Banerjee H, Nanda MK, Chakravarty K (2021) Impact of seaweed sap foliar application on growth, yield, and tuber quality of potato (Solanum tuberosum L.). J Appl Phycol 33:1893–1904. https://doi.org/10.1007/S10811-021-02386-3/TABLES/7
Garcia-gonzalez J, Sommerfeld M (2016) Biofertilizer and biostimulant properties of the microalga Acutodesmus dimorphus. J Appl Phycol 28:1051–1061. https://doi.org/10.1007/s10811-015-0625-2
Gonçalves AL (2021) The use of microalgae and cyanobacteria in the improvement of agricultural practices: a review on their biofertilising, biostimulating and biopesticide roles. Appl Sci 11:1–21. https://doi.org/10.3390/app11020871
González-González MF, Ocampo-Alvarez H, Santacruz-Ruvalcaba F, Sánchez-Hernández CV, Casarrubias-Castillo K, Becerril-Espinosa A, Castañeda-Nava JJ, Hernández-Herrera RM (2020) Physiological, ecological, and biochemical implications in tomato plants of two plant biostimulants: arbuscular mycorrhizal fungi and seaweed extract. Front Plant Sci 11:1–18. https://doi.org/10.3389/fpls.2020.00999
González-Pérez BK, Rivas-Castillo AM, Valdez-Calderón A, Gayosso-Morales MA (2022) Microalgae as biostimulants: a new approach in agriculture. World J Microbiol Biotechnol 38:1–12. https://doi.org/10.1007/S11274-021-03192-2/TABLES/1
Grzesik M, Romanowska-Duda Z (2014) Improvements in germination, growth, and metabolic activity of corn seedlings by grain conditioning and root application with Cyanobacteria and microalgae. Polish J Environ Stud 23:1147–1153
Grzesik M, Romanowska-Duda Z, Kalaji HM (2017) Effectiveness of cyanobacteria and green algae in enhancing the photosynthetic performance and growth of willow (Salix viminalis L.) plants under limited synthetic fertilizers application. Photosynthetica 55:510–521. https://doi.org/10.1007/s11099-017-0716-1
Halpern M, Bar-Tal A, Ofek M, Minz D, Muller T, Yermiyahu U (2015) The use of biostimulants for enhancing nutrient uptake
Haroun SA, Hussein MH (2003) The promotive effect of algal biofertilizers on growth, protein pattern and some metabolic activities of Lupinus termis plants grown in siliceous soil. Asian J Plant Sci 13:944–951
Holdt SL, Edwards MD (2014) Cost-effective IMTA: a comparison of the production efficiencies of mussels and seaweed. J Appl Phycol 26:933–945. https://doi.org/10.1007/s10811-014-0273-y
Hong DD, Hien HM, Son PN (2007) Seaweeds from Vietnam used for functional food, medicine and biofertilizer. J Appl Phycol 19:817–826. https://doi.org/10.1007/s10811-007-9228-x
Hughes AH, Magot F, Tawfike AF, Rad-Menéndez C, Thomas N, Young LC, Stucchi L, Carettoni D, Stanley MS, Edrada-Ebel R, Duncan KR (2021) Exploring the chemical space of macro-and micro-algae using comparative metabolomics. Microorganisms 9:1–16. https://doi.org/10.3390/microorganisms9020311
Hussein MH, Eltanahy E, Al Bakry AF, Elsafty N, Elshamy MM (2021) Seaweed extracts as prospective plant growth bio-stimulant and salinity stress alleviator for Vigna sinensis and Zea mays. J Appl Phycol 33:1273–1291. https://doi.org/10.1007/s10811-020-02330-x
Jeong ML, Gillis JM, Hwang JY (2003) Carbon dioxide mitigation by microalgal photosynthesis. Bull Korean Chem Soc 24:1763–1766. https://doi.org/10.5012/bkcs.2003.24.12.1763
Kapoore RV, Wood EE, Llewellyn CA (2021) Algae biostimulants: a critical look at microalgal biostimulants for sustainable agricultural practices. Biotechnol Adv 49:107754. https://doi.org/10.1016/j.biotechadv.2021.107754
Karthikeyan N, Prasanna R, Nain L, Kaushik BD (2007) Evaluating the potential of plant growth promoting cyanobacteria as inoculants for wheat. Eur J Soil Biol 43:23–30. https://doi.org/10.1016/j.ejsobi.2006.11.001
Khan W, Rayirath UP, Subramanian S, Jithesh MN, Rayorath P, Hodges DM, Critchley AT, Craigie JS, Norrie J, Prithiviraj B (2009) Seaweed extracts as biostimulants of plant growth and development. J Plant Growth Regul 28:386–399. https://doi.org/10.1007/s00344-009-9103-x
Khan MI, Shin JH, Kim JD (2018) The promising future of microalgae: current status, challenges, and optimization of a sustainable and renewable industry for biofuels, feed, and other products. Microb Cell Fact 17:1–21. https://doi.org/10.1186/s12934-018-0879-x
Kholssi R, Marks EAN, Montero O, Maté AP, Debdoubi A, Rad C (2018) The growth of filamentous microalgae is increased on biochar solid supports. Biocatal Agric Biotechnol 13:182–185. https://doi.org/10.1016/j.bcab.2017.12.011
Kim M, Shim CK, Kim YK, Ko BG, Park JH, Hwang SG, Kim BH (2018) Effect of biostimulator Chlorella fusca on improving growth and qualities of Chinese chives and spinach in organic farm. Plant Pathol J 34:567–574. https://doi.org/10.5423/PPJ.FT.11.2018.0254
Kocira A, Świeca M, Kocira S, Złotek U, Jakubczyk A (2018) Enhancement of yield, nutritional and nutraceutical properties of two common bean cultivars following the application of seaweed extract (Ecklonia maxima). Saudi J Biol Sci 25:563–571. https://doi.org/10.1016/J.SJBS.2016.01.039
Kumar G, Sahoo D (2011) Effect of seaweed liquid extract on growth and yield of Triticum aestivum var. Pusa Gold J Appl Phycol 23:251–255. https://doi.org/10.1007/s10811-011-9660-9
Kuwada K, Wamocho LS, Utamura M, Matsushita I, Ishii T (2006) Effect of red and green algal extracts on hyphal growth of arbuscular mycorrhizal fungi, and on mycorrhizal development and growth of papaya and passionfruit. Agron J 98:1340–1344. https://doi.org/10.2134/agronj2005.0354
Lee JY, Rahman A, Behrens J, Brennan C, Ham B, Kim HS, Nho CW, Yun ST, Azam H, Kwon MJ (2018) Nutrient removal from hydroponic wastewater by a microbial consortium and a culture of Paracercomonas saepenatans. N Biotechnol 41:15–24. https://doi.org/10.1016/j.nbt.2017.11.003
Li R, Tao R, Ling N, Chu G (2017) Chemical, organic and bio-fertilizer management practices effect on soil physicochemical property and antagonistic bacteria abundance of a cotton field: implications for soil biological quality. Soil Tillage Res 167:30–38. https://doi.org/10.1016/j.still.2016.11.001
Liu P, Wen H, Harich FK et al (2017) Conflict between conservation and development: cash forest encroachment in Asian elephant distributions. Sci Rep 7:1–10. https://doi.org/10.1038/s41598-017-06751-6
Liu J, Sun P, Sun R, Wang S, Gao B, Tang J, Wu Y, Dolfing J (2019) Carbon-nutrient stoichiometry drives phosphorus immobilization in phototrophic biofilms at the soil-water interface in paddy fields. Water Res 167:115129. https://doi.org/10.1016/j.watres.2019.115129
Maheshwari Uma (2017) Utilization of seaweed extract to enhance growth and biochemical parameters of RAPHANUS SATIVUS V AR. PUSA CHETKI. World J Pharm Pharm Sci 6:1177–1183. https://doi.org/10.20959/wjpps20173-8825
Mahmoud SH, Salama DM, El-Tanahy AMM, Abd El-Samad EH (2019) Utilization of seaweed (Sargassum vulgare) extract to enhance growth, yield and nutritional quality of red radish plants. Ann Agric Sci 64:167–175. https://doi.org/10.1016/J.AOAS.2019.11.002
Manjunath M, Kanchan A, Ranjan K, Venkatachalam S, Prasanna R, Ramakrishnan B, Hossain F, Nain L, Shivay YS, Rai AB, Singh B (2016) Beneficial cyanobacteria and eubacteria synergistically enhance bioavailability of soil nutrients and yield of okra. Heliyon 2:e00066. https://doi.org/10.1016/j.heliyon.2016.e00066
Mansori M, Chernane H, Latique S, Benaliat A, Hsissou D, El Kaoua M (2015) Seaweed extract effect on water deficit and antioxidative mechanisms in bean plants (Phaseolus vulgaris L.). J Appl Phycol 27:1689–1698. https://doi.org/10.1007/s10811-014-0455-7
Marks EAN, Miñón J, Pascual A, Montero O, Navas LM, Rad C (2017) Application of a microalgal slurry to soil stimulates heterotrophic activity and promotes bacterial growth. Sci Total Environ 605–606:610–617. https://doi.org/10.1016/j.scitotenv.2017.06.169
Marks EAN, Montero O, Rad C (2019) The biostimulating effects of viable microalgal cells applied to a calcareous soil: increases in bacterial biomass, phosphorus scavenging, and precipitation of carbonates. Sci Total Environ 692:784–790. https://doi.org/10.1016/j.scitotenv.2019.07.289
Martynenko A, Shotton K, Astatkie T, Petrash G, Fowler C, Neily W, Critchley AT (2016) Thermal imaging of soybean response to drought stress: the effect of Ascophyllum nodosum seaweed extract. Springerplus 5:1393. https://doi.org/10.1186/s40064-016-3019-2
Mata TM, Martins AA, Caetano NS (2010) Microalgae for biodiesel production and other applications: a review. Renew Sustain Energy Rev 14:217–232. https://doi.org/10.1016/j.rser.2009.07.020
Mathushika J, Gomes C (2022) Development of microalgae-based biofuels as a viable green energy source: challenges and future perspectives. Review 12:3849–3882. https://doi.org/10.33263/BRIAC123.38493882
Merwad ARMA (2020) Mitigation of salinity stress effects on growth, yield and nutrient uptake of wheat by application of organic extracts. Commun Soil Sci Plant Anal 51:1150–1160. https://doi.org/10.1080/00103624.2020.1751188
Michalak I, Chojnacka K (2015) Algae as production systems of bioactive compounds. Eng Life Sci 15:160–176. https://doi.org/10.1002/elsc.201400191
Michalak I, Tuhy Ł, Chojnacka K (2015) Seaweed extract by microwave assisted extraction as plant growth biostimulant. Open Chem 13:1183–1195. https://doi.org/10.1515/chem-2015-0132
Michalak I, Chojnacka K, Saeid A (2017) Plant growth biostimulants, dietary feed supplements and cosmetics formulated with supercritical CO2 Algal Extracts. Molecules 22:1–17. https://doi.org/10.3390/molecules22010066
Mikhailyuk T, Holzinger A, Massalski A, Karsten U (2014) Morphology and ultrastructure of Interfilum and Klebsormidium (Klebsormidiales, Streptophyta) with special reference to cell division and thallus formation. Eur J Phycol 49:395–412. https://doi.org/10.1080/09670262.2014.949308
Miñón Martínez J (2017) Development and technical-economic analysis of the management of residual nutrients in the production of biomass from algae for agricultural and livestock purposes (In Spanish: Desarrollo y análisis técnico-económico de la gestión de nutrientes residuales en la producción de biomasa de algas para fines agrícolas y ganaderos). Universidad de Valladolid
Molina Grima E, Belarbi E-H, Fernández AF, Robles Medina A, Chisti Y (2003) Recovery of microalgal biomass and metabolites: process options and economics. Biotechnol Adv 20:491–515
Mouritsen OG, Rhatigan P, Cornish ML, Critchley AT, Pérez-Lloréns JL (2021) Saved by seaweeds: phyconomic contributions in times of crises. J Appl Phycol 33:443–458. https://doi.org/10.1007/S10811-020-02256-4/FIGURES/12
Mukherjee A, Patel JS (2020) Seaweed extract: biostimulator of plant defense and plant productivity. Int J Environ Sci Technol 17:553–558. https://doi.org/10.1007/s13762-019-02442-z
Mukherjee C, Chowdhury R, Ray K (2015) Phosphorus recycling from an unexplored source by polyphosphate accumulating microalgae and cyanobacteria — a step to phosphorus security in agriculture. Front Microbiol 6:1–7. https://doi.org/10.3389/fmicb.2015.01421
Mukherjee C, Chowdhury R, Sutradhar T, Begam M, Ghosh SM, Basak SK, Ray K (2016) Parboiled rice effluent: a wastewater niche for microalgae and cyanobacteria with growth coupled to comprehensive remediation and phosphorus biofertilization. Algal Res 19:225–236. https://doi.org/10.1016/j.algal.2016.09.009
Mulbry W, Westhead EK, Pizarro C, Sikora L (2005) Recycling of manure nutrients: use of algal biomass from dairy manure treatment as a slow release fertilizer. Bioresour Technol 96:451–458. https://doi.org/10.1016/j.biortech.2004.05.026
Mulbry W, Kondrad S, Pizarro C (2007) Biofertilizers from algal treatment of dairy and swine manure effluents. J Veg Sci 12:107–125. https://doi.org/10.1300/J484v12n04
Nabti E, Jha B, Hartmann A (2016) Impact of seaweeds on agricultural crop production as biofertilizer. Int J Environ Sci Technol. https://doi.org/10.1007/s13762-016-1202-1
Nayak M, Swain DK, Sen R (2019) Strategic valorization of de-oiled microalgal biomass waste as biofertilizer for sustainable and improved agriculture of rice (Oryza sativa L.)crop. Sci Total Environ 682:475–484. https://doi.org/10.1016/j.scitotenv.2019.05.123
Nelson WR, van Staden J (1984) The effect of seaweed concentrate on wheat culms. J Plant Physiol 115:433–437. https://doi.org/10.1016/S0176-1617(84)80042-5
Nelson WR, Van Staden J (1994) The effect of seaweed concentrate on the growth of Pinus pinea seedlings. HortScience 19:81–82. https://doi.org/10.1007/BF00025373
Nguyen TQ, Sesin V, Kisiala A, Emery RJN (2020) The role of phytohormones in enhancing metal remediation capacity of algae. Bull Environ Contam Toxicol 105:671–678. https://doi.org/10.1007/s00128-020-02880-3
Oostlander PC, van Houcke J, Wijffels RH, Barbosa MJ (2020) Microalgae production cost in aquaculture hatcheries. Aquaculture 525:735310. https://doi.org/10.1016/j.aquaculture.2020.735310
Ortiz-moreno ML, Sandoval-parra KX, Solarte-murillo LV (2019) Chlorella, a potential biofertilizer? (In Spanish: Chlorella, ¿ un potencial biofertilizante ?). Orinoquia 23:71–78. https://doi.org/10.22579/20112629.582
Overland M, Mydland LT, Skrede A (2018) Marine macroalgae as sources of protein and bioactive compounds in feed for monogastric animals _ Enhanced Reader.pdf. Jounal Sci food Agric 99:13–24. https://doi.org/10.1002/jsfa.9143
Panda D, Pramanik K, Nayak BR (2012) Use of sea weed extracts as plant growth regulators for sustainable agriculture. Int J Bio-Resource Stress Manag 3:404–411
Papenfus HB, Kulkarni MG, Stirk WA, Finnie JF, Van Staden J (2013) Effect of a commercial seaweed extract (Kelpak®) and polyamines on nutrient-deprived (N, P and K) okra seedlings. Sci Hortic (amsterdam) 151:142–146. https://doi.org/10.1016/J.SCIENTA.2012.12.022
Patel A, Sarkar O, Rova U, Christakopoulos P, Matsakas L (2021) Valorization of volatile fatty acids derived from low-cost organic waste for lipogenesis in oleaginous microorganisms - a review. Bioresour Technol 321:124457. https://doi.org/10.1016/J.BIORTECH.2020.124457
Popko M, Michalak I, Wilk R, Gramza M, Chojnacka K, Górecki H (2018) Effect of the new plant growth biostimulants based on amino acids on yield and grain quality of winter wheat. Molecules 23:470. https://doi.org/10.3390/molecules23020470
Porcelli R, Dotto F, Pezzolesi L, Marazza D, Greggio N, Righi S (2020) Comparative life cycle assessment of microalgae cultivation for non-energy purposes using different carbon dioxide sources. Sci Total Environ 721:137714. https://doi.org/10.1016/j.scitotenv.2020.137714
Prakash S, Nikhil K (2014) Algal as a soil conditioner. Int J Eng Tech Res 2:68–70
Priya M, Gurung N, Mukherjee K, Bose S (2014) Microalgae in removal of heavy metal and organic pollutants from soil. in Microbial Biodegradation and Bioremediation, ed. Das S (Oxford: Elsevier), 519–537. https://doi.org/10.1016/b978-0-12-800021-2.00023-6
Rachidi F, Benhima R, Sbabou L, El Arroussi H (2020) Microalgae polysaccharides bio-stimulating effect on tomato plants: growth and metabolic distribution. Biotechnol Reports 25:e00426. https://doi.org/10.1016/J.BTRE.2020.E00426
Rai AN, Singh AK, Syiem MB (2019) Chapter 23 - plant growth-promoting abilities in cyanobacteria. Editor(s): A.K. Mishra, D.N. Tiwari, A.N. Rai, Cyanobacteria, Academic Press. Pages 459–476, ISBN 9780128146675, https://doi.org/10.1016/B978-0-12-814667-5.00023-4.
Rathore SS, Chaudhary DR, Boricha GN, Ghosh A, Bhatt BP, Zodape ST, Patolia JS (2009) Effect of seaweed extract on the growth, yield and nutrient uptake of soybean (Glycine max) under rainfed conditions. South African J Bot 75:351–355. https://doi.org/10.1016/j.sajb.2008.10.009
Rayirath P, Benkel B, Mark Hodges D, Allan-Wojtas P, MacKinnon S, Critchley AT, Prithiviraj B (2009) Lipophilic components of the brown seaweed, Ascophyllum nodosum, enhance freezing tolerance in Arabidopsis thaliana. Planta 230:135–147. https://doi.org/10.1007/s00425-009-0920-8
Reed RC, Brady SR, Muday GK (1998) Inhibition of auxin movement from the shoot into the root inhibits lateral root development in arabidopsis. Plant Physiol 118:1369–1378. https://doi.org/10.1104/pp.118.4.1369
Renaut S, Masse J, Norrie JP, Blal B, Hijri M (2019) A commercial seaweed extract structured microbial communities associated with tomato and pepper roots and significantly increased crop yield. Microb Biotechnol 12:1346–1358. https://doi.org/10.1111/1751-7915.13473
Renuka N, Prasanna R, Sood A, Ahluwalia AS, Bansal R, Babu S, Singh R, Shivay YS, Nain L (2016) Exploring the efficacy of wastewater-grown microalgal biomass as a biofertilizer for wheat. Environ Sci Pollut Res 23:6608–6620. https://doi.org/10.1007/s11356-015-5884-6
Renuka N, Prasanna R, Sood A, Bansal R, Bidyarani N, Singh R, Shivay YS, Nain L, Ahluwalia AS (2017) Wastewater grown microalgal biomass as inoculants for improving micronutrient availability in wheat. Rhizosphere 3:150–159. https://doi.org/10.1016/j.rhisph.2017.04.005
Renuka N, Guldhe A, Prasanna R, Singh P, Bux F (2018) Microalgae as multi-functional options in modern agriculture: current trends, prospects and challenges. Biotechnol Adv 36:1255–1273. https://doi.org/10.1016/j.biotechadv.2018.04.004
Ricci M, Tilbury L, Daridon B, Sukalac K (2019) General principles to justify plant biostimulant claims. Front Plant Sci 10:1–8. https://doi.org/10.3389/fpls.2019.00494
Rogers SL (1994) Burns RG (1994) Changes in aggregate stability, nutrient status, indigenous microbial populations, and seedling emergence, following inoculation of soil with Nostoc muscorum. Biol Fertil Soils 183(18):209–215. https://doi.org/10.1007/BF00647668
Ronga D, Biazzi E, Parati K, Carminati D, Carminati E, Tava A (2019) Microalgal biostimulants and biofertilisers in crop productions. Agronomy 9:1–22. https://doi.org/10.3390/agronomy9040192
Rossi F, Li H, Liu Y, De Philippis R (2017) Cyanobacterial inoculation (cyanobacterisation): perspectives for the development of a standardized multifunctional technology for soil fertilization and desertification reversal. Earth-Science Rev 171:28–43. https://doi.org/10.1016/j.earscirev.2017.05.006
Safi C, Ursu AV, Laroche C, Zebib B, Merah O, Pontalier PY, Vaca-Garcia C (2014) Aqueous extraction of proteins from microalgae: effect of different cell disruption methods. Algal Res 3:61–65. https://doi.org/10.1016/j.algal.2013.12.004
Santaniello A, Scartazza A, Gresta F, Loreti E, Biasone A, Di Tommaso D, Piaggesi A, Perata P (2017) Ascophyllum nodosum seaweed extract alleviates drought stress in Arabidopsis by affecting photosynthetic performance and related gene expression. Front Plant Sci 8:1–15. https://doi.org/10.3389/fpls.2017.01362
Santner A, Calderon-Villalobos LIA, Estelle M (2009) Plant hormones are versatile chemical regulators of plant growth. Nat Chem Biol 5:301–307. https://doi.org/10.1038/nchembio.165
Sayre R (2010) Microalgae: the potential for carbon capture. Bioscience 60:722–727. https://doi.org/10.1525/bio.2010.60.9.9
Schreiber C, Schiedung H, Harrison L et al (2018) Evaluating potential of green alga Chlorella vulgaris to accumulate phosphorus and to fertilize nutrient-poor soil substrates for crop plants. J Appl Phycol 30:2827–2836. https://doi.org/10.1007/s10811-018-1390-9
Shah MT, Zodape ST, Chaudhary DR, Eswaran K, Chikara J (2013) Seaweed sap as an alternative liquid fertilizer for yield and quality improvement of wheat. J Plant Nutr 36:192–200. https://doi.org/10.1080/01904167.2012.737886
Shekhar SHS, Lyons G, McRoberts C, McCall D, Carmichael E, Andrews F, McCormack R (2012) Brown seaweed species from Strangford Lough: compositional analyses of seaweed species and biostimulant formulations by rapid instrumental methods. J Appl Phycol 24:1141–1157. https://doi.org/10.1007/s10811-011-9744-6
Shi Y, Ziadi N, Hamel C, Bittman S, Hunt D, Lalande R, Shang J (2018) Soil microbial biomass, activity, and community composition as affected by dairy manure slurry applications in grassland production. Appl Soil Ecol 125:97–107. https://doi.org/10.1016/j.apsoil.2017.12.022
Shukla PS, Shotton K, Norman E, Neily W, Critchley AT, Prithiviraj B (2018) Seaweed extract improve drought tolerance of soybean by regulating stress-response genes. AoB Plants 10:1–8. https://doi.org/10.1093/aobpla/plx051
Solé-Bundó M, Cucina M, Folch M, Tàpias J, Gigliotti G, Garfí M, Ferrer I (2017) Assessing the agricultural reuse of the digestate from microalgae anaerobic digestion and co-digestion with sewage sludge. Sci Total Environ 586:1–9. https://doi.org/10.1016/j.scitotenv.2017.02.006
Spinelli F, Fiori G, Noferini M, Sprocatti M, Costa G (2010) A novel type of seaweed extract as a natural alternative to the use of iron chelates in strawberry production. Sci Hortic (amsterdam) 125:263–269. https://doi.org/10.1016/j.scienta.2010.03.011
Steveni CM, Norrington-Davies J, Hankins SD (1992) Effect of seaweed concentrate on hydroponically grown spring barley. J Appl Phycol 4:173–180. https://doi.org/10.1007/BF02442466
Stirk WA, Van Staden J (1997) Comparison of cytokinin- and auxin-like activity in some commercially used seaweed extracts. J Appl Phycol 8:503–508. https://doi.org/10.1007/BF02186328
Stirk WA, Bálint P, Tarkowská D, Novák O, Maróti G, Ljung K, Turecková V, Strnad M, Ördög V, van Staden J (2014a) Effect of light on growth and endogenous hormones in Chlorella minutissima (Trebouxiophyceae). Plant Physiol Biochem 79:66–76
Stirk WA, Tarkowská D, Turečová V, Strnad M, van Staden J (2014b) Abscisic acid, gibberellins and brassinosteroids in Kelpak®, a commercial seaweed extract made from Ecklonia maxima. J Appl Phycol 26:561–567. https://doi.org/10.1007/s10811-013-0062-z
Supraja KV, Bunushree B, Balasubramanian P (2020) Efficacy of microalgal extracts as biostimulants through seed treatment and foliar spray for tomato cultivation. Ind Crops Prod 151:112453. https://doi.org/10.1016/j.indcrop.2020.112453
Tan BC, Schwartz SH, Zeevaart JAD, Mccarty DR (1997) Genetic control of abscisic acid biosynthesis in maize. Proc Natl Acad Sci 94:12235–12240
Tan CY, Dodd IC, Chen JE, Phang SM, Chin CF, Yow YY, Ratnayeke S (2021) Regulation of algal and cyanobacterial auxin production, physiology, and application in agriculture: an overview. J Appl Phycol. https://doi.org/10.1007/s10811-021-02475-3
Turan M, Köse C (2004) Seaweed extracts improve copper uptake of grapevine. Acta Agric Scand Sect B Soil Plant Sci 54:213–220. https://doi.org/10.1080/09064710410030311
Ugarte RA, Sharp G, Moore B (2006) Changes in the brown seaweed Ascophyllum nodosum (L.) Le Jol. plant morphology and biomass produced by cutter rake harvests in southern New Brunswick. Canada J Appl Phycol 18:351–359. https://doi.org/10.1007/S10811-006-9044-8
Uysal O, Uysal FO, Ekinci K (2015) Evaluation of microalgae as microbial fertilizer. Eur J Sustain Dev 4:77–82. https://doi.org/10.14207/ejsd.2015.v4n2p77
Van Oosten MJ, Pepe O, De Pascale S, Silletti S, Maggio A (2017) The role of biostimulants and bioeffectors as alleviators of abiotic stress in crop plants. Chem Biol Technol Agric 4:1–12. https://doi.org/10.1186/s40538-017-0089-5
Vasileva I, Ivanova J, Paunov M, Angelova L (2016) Urea from waste waters – perspective nitrogen and carbon source for green algae Scenedesmus Sp. cultivation. J Int Sci Publ 10:1314–7234
Verma N, Sehrawat AR, Pandey D, Pandey BK (2020) Seaweed: a novel organic biomaterial. Curr J Appl Sci Technol 39:1–8. https://doi.org/10.9734/cjast/2020/v39i1430690
Volf MR, Rosolem CA (2020) Soil P diffusion and availability modified by controlled-release P fertilizers. J Soil Sci Plant Nutr. https://doi.org/10.1007/s42729-020-00350-7
Wang W, Liu Y, Li D, Hu C, Rao B (2009) Feasibility of cyanobacterial inoculation for biological soil crusts formation in desert area. Soil Biol Biochem 41:926–929. https://doi.org/10.1016/j.soilbio.2008.07.001
Whitton BA, Grainger SLJ, Hawley GRW, Simon JW (1991) Cell-bound and extracellular phosphatase activities of cyanobacterial isolates. Microb Ecol 21:85–98
Wu Y, Rao B, Wu P, Liu Y, Li G, Li D (2013) Development of artificially induced biological soil crusts in fields and their effects on top soil. Plant Soil 370:115–124. https://doi.org/10.1007/s11104-013-1611-6
Xiao R, Zheng Y (2016) Overview of microalgal extracellular polymeric substances (EPS) and their applications. Biotechnol Adv. https://doi.org/10.1016/j.biotechadv.2016.08.004
Yao Y, Wang X, Chen B, Zhang M, Ma J (2020) Seaweed extract improved yields, leaf photosynthesis, ripening time, and net returns of tomato (Solanum lycopersicum Mill.). ACS Omega 5:4242–4249. https://doi.org/10.1021/ACSOMEGA.9B04155/ASSET/IMAGES/MEDIUM/AO9B04155_M002.GIF
Zaady E, Katra I, Barkai D, Knoll Y, Sarig S (2017) The coupling effects of using coal fly-ash and bio-inoculant for rehabilitation of disturbed biocrusts in active sand dunes. L Degrad Dev 28:1228–1236. https://doi.org/10.1002/ldr.2510
Zhang X, Wang K, Ervin EH (2010) Optimizing dosages of seaweed extract-based cytokinins and zeatin riboside for improving creeping bentgrass heat tolerance. Crop Sci 50:316–320. https://doi.org/10.2135/cropsci2009.02.0090
Zhang J, Wang X, Zhou Q (2017) Co-cultivation of Chlorella spp and tomato in a hydroponic system. Biomass Bioenerg 97:132–138. https://doi.org/10.1016/j.biombioe.2016.12.024
Zhu J, Wakisaka M (2020) Finding of phytase: understanding growth promotion mechanism of phytic acid to freshwater microalga Euglena gracilis. Bioresour Technol 296:122343. https://doi.org/10.1016/j.biortech.2019.122343
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
The original version of this article was revised: The affiliation for coauthor Omar Castaño-Sánchez has been corrected since this article’s original publication.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Kholssi, R., Lougraimzi, H., Grina, F. et al. Green Agriculture: a Review of the Application of Micro- and Macroalgae and Their Impact on Crop Production on Soil Quality. J Soil Sci Plant Nutr 22, 4627–4641 (2022). https://doi.org/10.1007/s42729-022-00944-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s42729-022-00944-3