Abstract
Purpose
The extracellular matrix (ECM) is a complicated milieu consisting of structural and functional molecules secreted by the resident cells that provides an optimal microenvironmental niche for enhanced cell adhesion, growth, differentiation, and tissue formation and maturation. For decades, ECM bio-scaffolds prepared from decellularized tissues have been used to promote skeletal muscle regeneration; however, it was recently discovered that these decellularized ECM (dECM) materials can be further processed into hydrogels, thus expanding the potential applications of dECM materials in skeletal muscle regenerative engineering (SMRE). This review article highlights the recent advances in dECM-derived hydrogels toward skeletal muscle regeneration and repair.
Method
We screened articles in PubMed and bibliographic search using a combination of keywords. Relevant and high-cited articles were chosen for inclusion in this narrative review.
Results
Here, we discuss the skeletal muscle ECM’s structure, function, and biochemical composition with emphasis on the role of the ECM during skeletal muscle embryogenesis, growth, development, and repair. Furthermore, we review various hydrogels used to promote skeletal muscle regeneration. We also review the current applications of dECM-derived hydrogels toward SMRE. Finally, we discuss the clinical translation potential of dECM-derived hydrogels for skeletal muscle regeneration and repair and their potential clinical considerations in the future.
Conclusion
Although much progress has been made in the field of dECM-derived hydrogels toward SMRE, it is still in its nascent stage. We believe improving and standardizing the methods of decellularization, lowering the immunogenicity of dECMs, and carrying out in vivo investigations in large animal models would advance their future clinical applications.
Lay Summary
Researchers have discovered an effective way to turn tissue materials into jelly-like substances known as extracellular matrix (ECM)-derived hydrogels. These ECM-derived hydrogels can help muscles heal better after serious injuries. They can be injected into gaps or used to guide muscle growth in the lab or body. This review article explains how these ECM-derived hydrogels are made and how they can be used to improve muscle healing. It also discusses their possible use in clinics and what needs to be considered before using them for medical treatments.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Of the many tissues found in the human body, the skeletal muscle constitutes the most, accounting for 40–45% of the total body weight [1]. The main function of the skeletal muscle is to facilitate body locomotion through moving bones and other body parts [2]. Additionally, the skeletal muscles is considered to be one of the major players in regulating the body temperature [3], metabolism [4], and peptides secretion which facilitates the interactions of skeletal muscle with other tissues [5]. Thus, promoting and maintaining skeletal muscle health are of extreme importance.
In cases of minor injuries, where muscle loss is less than 20%, the skeletal muscle displays a unique endogenous capacity for regeneration [1]. However, in cases of severe injuries—muscle loss greater than 20%, the skeletal muscle’s endogenous regenerative ability is impaired, resulting in extensive and irreversible fibrosis, scarring, and functional impairments—a condition known as volumetric muscle loss (VML) [6]. Such a condition can result from many events that include but are not limited to tumor resections, work/car accidents, or compact injuries [7,8,9]. Regardless of the cause, VML significantly impacts the patients’ movement and can lead to progressive muscle loss and degeneration if left untreated [9,10,11].
Currently, autologous muscle transfer, where muscle tissues from a healthy donor site are harvested and grafted into the injury site, is the gold standard for treating VML [12,13,14]. This treatment approach can effectively regenerate the lost muscle tissue and restore the impaired function [15, 16]. However, donor site morbidity, increased surgical times, and the limited availability of donor tissues, especially in severely injured individuals, are the significant drawbacks of this only viable clinical approach [1].
Since current clinical strategies cannot achieve satisfactory results for the treatment of VML, a paradigm shift is required to address the current challenges. Regenerative Engineering (RE) is a transdisciplinary approach founded on combining cutting-edge technologies available in disparate fields to develop highly translational technologies for the regeneration of complex tissues and organ systems [17,18,19,20,21,22,23,24,25]. Within this paradigm, advanced biomaterials are integral for developing highly regenerative scaffolding systems that can foster the regeneration process.
A scaffold is a temporary structure designed to guide cells’ three-dimensional (3D) growth during the tissue developmental stages [26]. To date, a substantial number of scaffolding systems have been developed and utilized for skeletal muscle RE (SMRE) [27] and of these, hydrogels are the most preferable [28]. Owing to their soft and viscoelastic biomimetic nature, hydrogels can regulate muscle cell behavior more efficiently, promote their myogenic differentiation, and allow for sufficient force production and contraction by the muscle tissue [28]. At present, several natural, synthetic, and composite hydrogel materials have been developed using the biochemical, mechanical, and structural information present in the native extracellular matrix (ECM) as a blueprint [29]. However, to date, the complexity of the native ECM could not be fully recapitulated by any natural, synthetic, or composite hydrogel material.
In recent years, there has been growing attention to biologic materials for synthesizing hydrogels, such as those derived from the ECM of native decellularized biological tissues [30]. In addition to their excellent interactions with the in vivo milieu, biocompatibility, soft and elastic consistency, decellularized ECM (dECM)-derived hydrogels are superior to natural, synthetic, and composite hydrogels since they contain the original ECM components and mimic the structural and mechanical complexity of the native tissues [31]. Hence, they provide an optimal niche with complex environmental cues for enhanced cell adhesion, growth, differentiation, and tissue formation and maturation [31, 32]. Owing to their excellent characteristics, dECM-derived hydrogels are currently considered the best candidates for regenerative engineering of different tissues [30], particularly skeletal muscles (Fig. 1).
This review article highlights the recent advances in dECM-derived hydrogels toward skeletal muscle regeneration and repair. First, we start by discussing the skeletal muscle ECM’s structure, function, and biochemical composition. We then mention the role of the ECM during skeletal muscle embryogenesis, growth, development, and repair. Next, we review the various hydrogels used to promote skeletal muscle regeneration, focusing more on dECM-derived hydrogels. In this context, a detailed discussion of the biological effects of tissue-specific vs. non-tissue-specific dECM-derived hydrogels on muscle cell behavior and regeneration is present. We also review the current applications of dECM-derived hydrogels for SMRE. Finally, we discuss the clinical translation potential of dECM-derived hydrogels toward skeletal muscle regeneration and repair and highlight some serious considerations to expedite their future presence in the clinic.
Skeletal Muscle Extracellular Matrix (smECM)
smECM Structure and Function
The ECM is the acellular element of any tissue or organ secreted by the resident cells [33]. The ECM represents approximately 10% of the dry weight in skeletal muscle [34]. The smECM acts as a mechanical support during muscle cell (myofibers) contraction, provides the tissue its elasticity, and facilitates transmitting the contractile forces down to the tendon [34]. In addition, the smECM houses the blood vessels and nerves found in the skeletal muscle tissue [35]. The smECM exhibits a well-defined structure distinct from other tissues and inherently correlates to its function [34] (Fig. 2). Briefly, each muscle is enclosed by a specialized fibrous tissue termed as the epimysium [36]. Its primary function is to protect muscles from friction against other neighboring muscles and bones and define the muscle volume [36]. Within a muscle, myofibers are arranged into bundles of skeletal muscle fibers, called fascicles [34]. Each fascicle is enclosed by an additional layer of fibrous tissue, named the perimysium [36]. The main function of the perimysium is to group myofibers into bundles and transmit lateral contractile movements [37]. Within each myofiber, uniaxially aligned myofibrils are present [37]. This myofibril alignment is crucial to facilitate muscle contraction in a single uniaxial direction. Each myofiber is enclosed within endomysium, which separates single myofibers from one another, allowing their autonomous gliding during the contraction process [38]. The endomysium also comprises the basement membrane, which is a layer of ECM, coating the skeletal muscle fibers [39]. The primary function of the basement membrane is to provide physical and biochemical cues to the overlying cells [40]. The basement membrane further consists of inner and outer layers. The inner portion of the basement membrane constitutes basal lamina, which is directly attached to the myofibers, whereas its outer portion constitutes the reticular lamina [41]. The basal lamina of the basement membrane functions by transmitting the contractile forces between myofibers and tendons, whereas its reticular lamina ensures a solid grip to the surrounding connective tissues to facilitate a full transmission of the produced forces [42].
Beyond its role as a mechanical support, the smECM’s topographical and elastic features can be sensed by muscle cells and are important regulators of their functions [43]. Integrins—proteins that muscle cells use to attach to the ECM, are known to be the major element in facilitating the connection between muscle cells and their ECM niche [43]. Besides their ability to enable the adhesion of muscle cells to the smECM, they also function by converting extracellular signals (e.g., matrix stiffness, tensile strain, topographical features, elasticity) to intracellular responses. This allows the muscle cells to sense their surrounding microenvironment intelligently and behave accordingly [44].
The smECM can also store various soluble proteins such as growth factors. During ECM remodeling and degradation, the stored growth factors can release into the ECM space, generating an active milieu, rich in signaling molecules that can trigger different cellular responses, including migration, proliferation, polarity, differentiation, and 3D spatial organization [45].
Overall, these properties depend on the matrix’s protein composition, which varies substantially among different tissues. For example, in skeletal muscle, the ECM constituents can roughly be divided into structural and non-structural ECM proteins. Tables 1 and 2 summarize all structural and non-structural ECM proteins found in the smECM and their corresponding functions.
smECM Biochemical Composition
Structural ECM Proteins
Structural ECM proteins are responsible for establishing the tissue’s physical support [32]. In skeletal muscle, the structural ECM proteins consist mainly of collagens, proteoglycans, glycoproteins, and elastin [46, 47]. Collagens are known to form a meshwork of skeletal muscle connective tissues (SMCTs), i.e., the epimysium, perimysium, and endomysium. Of the many collagen types constituting the SMCTs, types I and III are the most predominant, representing approximately 75% of the total collagen found in the SMCTs [34, 48,49,50]. Collagen type I arranges itself as densely packed parallel fibers, providing the muscle with a certain degree of rigidity and tensile strength, and is primarily found in the perimysium [51]. Collagen type III, on the other hand, makes up a loosely entangled network of fibers that confers the endomysium and epimysium resilience [51]. Other forms of collagens known to be present in the smECM, but in minor quantity, are collagens IV, V, VI, XII, XIII, XIV, XV, XVIII, XIX, and XXII [51] (Table 1).
Collagen type IV—a helical molecule, is the most abundant constituent of the inner portion of the basement membrane (i.e., basal lamina) that forms a network-like structure in the smECM [39]. It is the most essential structural collagen of the basement membrane, and it possesses a key signaling potential for various pathological and physiological functions [52]. Collagen V, a fibrillar molecular, can be found in all three SMCT layers, and it is essential for fibrillation of types I and III collagen, thereby ensuring optimal fibrillary formation [53]. Collagen VI has been found in all three layers of the SMCT, but it can mostly be found in the basal lamina of the basement membrane [54,55,56]. It is an integral part of the basement membrane and has been shown to influence satellite stem cells’ (SSCs) self-renewal together with fibronectin, another structural glycoprotein in the smECM [54,55,56]. Type XII, XIV, XIX, and XXII collagen are categorized to be fibril-associated collagens with interrupted triple helices (FACIT) [57]. The most crucial one of these is collagen type XXII, which can only be found at the myotendinous junction between skeletal muscle and tendon tissues [58]. It mainly functions by stabilizing the myotendinous junctions and strengthening skeletal muscle attachments during muscle contraction [58].
Proteoglycans in the smECM such as syndecan 3, syndecan 4, decorin, and perlecan have been identified as SSCs niche constituents [59]. Most of the proteoglycans in the smECM are known to be small leucine-rich, with the majority having dermatan sulfate and chondroitin sulfate GAG side chains [60]. Proteoglycans with a heparin sulfate GAG, such as agrin, perlecan, and collagen type XVIII, constitute nearly 30% of the proteoglycans in the smECM [61]. Agrin mainly supports the formation of neuromuscular junctions (NMJs) on the myofibers’ skeleton [61], thus promoting neurogenesis. The rest of these heparin sulfate proteoglycans mainly execute a bridging function, connecting the networks of collagens and laminin polymers in the muscle lamina. Laminin is a structural glycoprotein found in the smECM, with Laminin-211 being the most predominant isoform [41, 62]. Laminin forms polymers of a heterotrimeric complex that encompasses an α, β, and γ chain and is one of the main ligands of the dystrophin-associated glycoprotein complex and the α7β1 integrin [63]. These link laminin in the smECM to actin in the skeletal muscle fibers, allowing forces, upon myofiber contraction, to be transmitted across the membrane [63]. Altogether, these structural ECM proteins help stabilize muscle cells and maintain the smECM’s overall homeostasis by providing structural support and stimulating certain cellular processes.
The smECM is actively remodeled through degrading and building cycles. It is predominantly built and degraded by fibroblasts. However, evidence also suggests that other mononuclear cells, such as SSCs, and even multinucleated myofibers participate in secreting the smECM components and smECM-degrading enzymes, especially during muscle injury [64]. For instance, it has been shown that the degrading enzymes secreted by SSCs, such as matrix metalloproteinases, aid them in migrating toward the injury site to initiate the repair process [65, 66]. Moreover, the smECM is crucial for regulating the SSCs niche because substantial skeletal muscle regeneration depends highly on the interaction between SSCs and their micro-milieu [67].
Non-structural ECM Proteins
Non-structural ECM proteins are soluble factors secreted by the tissue’s resident cells, e.g., growth factors [82]. The smECM acts as a reservoir of the secreted growth factors, which can adhere to the ECM’s mesh by adhesion molecules that can bind and modulate growth factors [83]. The bound growth factors can later be released, free to diffuse through the tissue until they come into contact with their cognate growth factor receptors [84]. Specifically, when the cells secrete these factors, they travel in the ECM’s space and either bind to a specific transmembrane receptor on a target cell or onto the ECM’s mesh by the adhesion molecules [84]. Through this binding, the ECM can protect growth factors from degradation or help form a concentration gradient, which is important for stimulating site-specific cellular responses within the tissue [84]. Therefore, besides its role as a structural support, the smECM forms a storage place for various growth factors that can control the most fundamental behavior and characteristics of cells, such as proliferation, adhesion, migration, polarity, differentiation, and apoptosis [83]
Growth factors known to be vigorously present in the smECM include insulin-like growth factor 1 (IGF-1), insulin-like growth factor-binding protein 4 (IGFBP-4), transforming growth factor-beta1 (TGF-β1), transforming growth factor-beta3 (TGF-β3), platelet-derived growth factor (PDGF), vascular endothelial cell growth factor (VEGF), fibroblast growth factor (FGF), hepatocyte growth factor (HGF), and epidermal growth factor (EGF) [85, p. 3], [86,87,88, 88,89,90,91]. Generally, these growth factors work in tandem from first stimulating SSCs activation and proliferation until they gain a significant increase in cell mass to promoting their differentiation, fusion, and maturation [85, p. 3], [86,87,88, 88,89,90,91]. The specific functions of each of these smECM-related factors are reported in (Table 2).
The Role of ECM During Skeletal Muscle Embryogenesis, Growth, Development, and Repair
The Role of ECM During Skeletal Muscle Embryogenesis, Growth, and Development
Apart from providing the structural support and acting as a reservoir for growth factors, the smECM plays a vital role in skeletal muscle embryogenesis, growth, and development through a myriad of interactions with muscle progenitors and myofibers. The cell phenotype that makes up the collagenous component of the smECM are believed to be myofibroblasts [95,96,97]. Previous works have provided clear evidence that not only the SMCT myofibroblasts are involved in making up the collagenous component of the smECM, but they, along with the connective tissues produced by them also play a crucial role in both myogenesis and muscle morphogenesis. Therefore, they are an essential regulator during the developmental stages of the skeletal muscle. This cascade encompasses multiple phases. Briefly, the SMCT guides the migration of muscle precursors to their respective regions through a combination of attractive and repulsive signals [98, 99]. Once muscle progenitors arrive at their target regions, the SMCT acts as a bioactive milieu that promotes their proliferation, survival, and differentiation into mature myofibers [100,101,102,103]. Therefore, it can be concluded that the SMCT plays an essential role during skeletal muscle morphogenesis during its embryonic developmental stages [100,101,102,103].
Unlike many other tissues, the skeletal muscle displays a unique endogenous capacity for adaptation and regeneration throughout life [1]. This inherent ability of the skeletal muscle to adapt and regenerate depends mainly on SSCs. However, while solid evidence confirms that SSCs represent a key determinant for skeletal muscle regeneration, a “qualified” environment is no doubt necessary to ensure and achieve functional outcomes [104]. SSCs reside in specialized niches within the basement membrane (i.e., on the periphery of myofibers) [105]. Their fate (e.g., maintenance, activation, proliferation, differentiation, and self-renewal) is largely controlled via the interactions with their ECM niche [105]. For instance, the SSCs’ niche is a dynamic environment that constantly transmits mechanical and chemical signals to the resident SSCs [105]. Consequently, these signals modulate the behavior of SSCs through maintaining their quiescent state or inducing their activation, proliferation, and subsequent fusion into multinucleated myofibers [105].
In their quiescent state, SSCs continuously express paired box protein 7 (PAX7) – a canonical cell regulator [106]. Previous studies have shown that a greater portion of isolated SSCs could maintain their expression to PAX7 when cultured on or within hydrogels containing a mixture of multiple ECM proteins and growth factors resembling those found in their original niche, such as Matrigel [107, 108]. However, when these cells were cultured on standard polystyrene surfaces, they instantly entered the cell cycle and lost their myogenic regenerative capacity, further supporting the vital role of the ECM in maintaining their quiescent state [109]. Astonishingly, SSCs can sense and respond to different ECM mechanical properties. Culturing SSCs on or within hydrogels that mimic the physiological stiffness of the muscle tissue (10–30 kPa) maintained SSCs’ survival and PAX7 expression [109]. Additionally, SSCs cultured on or within hydrogels with biomimetic stiffness displayed a greater functional capacity post-transplantation in vivo [110].
Besides the role of the ECM’s mechanical and chemical properties in modulating SSCs behavior, the ECM’s biochemical composition has also been shown to influence SSCs functions. For instance, the proteins fibronectin [111], collagen type VI [112], and various proteoglycans such as syndecan 3, syndecan 4, decorin, and perlecan [59, 113] have been identified as the major constituents of the SSCs’ niche. These proteins work together to control the balance between SSCs’ self-renewal and differentiation, thereby, maintaining the regenerative capacity of the skeletal muscle.
In the steady-state, SSCs are quiescent [114]. However, in response to injuries, they get activated at the injury site by local signals, and become proliferating myoblasts, which ultimately differentiate and fuse to form myofibers that can aid in the process of muscle repair [114]. Numerous studies have indicated that the ECM influences each of these steps by timely controlling the expression of many myogenic transcription factors including myogenic 5 (MYF5) and myogenin (MyoG) [105]. In addition, while the contribution of individual proteins are largely unknown, previous studies have suggested that the concomitant presence of laminin and poly-d-lysine [115], glycodsminoglycans [116], and heparin sulfate proteoglycans [117] within the smECM plays a crucial role in modulating SSCs’ behavior, further adding to the notion that the ECM’s proteins work together to control the balance between SSCs’ activation, proliferation, differentiation, and the self-renewal.
Upon activation of SSCs, local remodeling of the ECM occurs, followed by depositing laminin into the inner portion of the basement membrane (i.e., basal lamina) [118]. In addition, a previous study observed that SSCs maintain their inactivated quiescent state by synthesizing certain types of ECM collagens, and collagen type V has been identified as an important constituent for maintaining the SSCs quiescence, since its elimination was found to play a major role in reducing the ability of SSCs to maintain their quiescence [119]. Altogether, these data provide evidence of the strong role that the smECM plays during skeletal muscle embryogenesis, growth, and development.
The Role of the ECM During Skeletal Muscle Repair
Two centuries ago, it was thought that the skeletal muscle did not exhibit an endogenous capacity for regeneration [227]. However, the research efforts from the current era have provided significant evidence that the skeletal muscle exhibits an endogenous regenerative capacity, and, therefore, it has become well-known [104, 227]. The skeletal muscle’s inherent capacity to regenerate encompasses multiple stages that can be divided into five interrelated and time-dependent phases (Fig. 3). First, an initial phase of inflammatory immune response is elicited upon muscle injury to clear the damaged muscle debris [104, 227]. Next, a regenerative phase is triggered, which involves SSCs activation, ECM deposition, and myoblast fusion into myofibers [227]. Thereafter, a remodeling phase takes place, followed by a maturation phase until a full muscle restoration is achieved [227]. During these stages, resident muscle cells and infiltrating immune cells from the circulatory system work together to restore muscle structure, function, and hemostasis [227]. The smECM plays a vital role during several phases of the repair process, which will be discussed in the following subsections [227].
Degenerative and Inflammatory Phase
Upon muscle injury, all layers of the SMCT undergo physical damage, which triggers myofibers degeneration, resulting in increased myofiber permeability and, thereby, myofiber necrosis and death [120, 227]. Due to the presence of SMCT and degenerated myofibers debris at the site of the injury, resident immune cells are activated, which start producing chemotactic signals to recruit other immune cells from the circulatory system to the site of the injury [121, 227]. Neutrophils being the first responders to these signals, contribute to clearing the site of injury by releasing degrading enzymes [227]. The presence of neutrophils recruits even more immune cells to the injury site, such as macrophages, which trigger both pro-inflammatory and pro-remodeling responses [122, 227]. First, the polarized pro-inflammatory M1 macrophages aid neutrophiles in clearing the tissue debris in the injury site by producing pro-inflammatory cytokines and reactive oxygen species [123, 227]. Following the removal of the tissue debris, the number of M1 macrophages drops, and the polarized pro-remodeling M2 macrophages become the predominant subtype in preparation for initiating the regeneration process [227].
Regenerative Phase
In response to the cytokines and growth factors released at the injury site during the inflammatory phase, SSCs get activated, and become proliferating myoblasts, which ultimately differentiate and fuse to form myofibers that can aid in the regenerative process [124, 227]. However, a small portion of the SSCs retain their quiescence to maintain a pool of SSCs in case any repair needed in the future [114, 227]. Once the injury site is filled with proliferating myoblasts, they terminally differentiate and subsequently fuse to form myofibers [125, 227]. During this stage, SMCT fibroblasts simultaneously deposit a temporary ECM, which acts as a scaffold for the newly forming myofibers [126, 227]. In addition, immune cells such as regulatory T-cells and eosinophils contribute to myogenesis during this regenerative phase by enhancing SSCs’ myogenic differentiation and fusion [127, 227].
Remodeling and Maturation Phase
Post-regenerative phase, the temporary ECM deposited by the SMCT fibroblasts gets remodeled, thus, marking the beginning of the remodeling phase [128, 227]. Tissue remodeling can broadly be defined as reorganizing or renovating an existing tissue’s ECM by simultaneously releasing degrading enzymes and ECM building proteins by the resident cells [129, 227]. During this remodeling phase, endothelial cells migrate into the newly regenerated site and rapidly proliferate to form new blood vessels and capillaries to feed the newly regenerated tissue with oxygen, nutrients, and other major elements [130]. In addition, the secreted factors by endothelial cells such as angiopoietin-1 [131], IGF-I [132], HGF [93, 133], and VEGF [130] help recruit more endothelial cells to facilitate more vascularization and further support the ongoing regenerative process by influencing SSCs’ proliferation, differentiation, and fusion [227].
Lastly, the re-establishment of the lost motor neurons takes place to facilitate functional recovery, accompanied by the formation of new NMJs [134, 227]. This innervation process is known to take place a few weeks after injury, after which newly terminally differentiated myofibers are regenerated [134, 227]. However, the speed and extent of reinnervation largely depend on the severity of the injury. In severely damaged muscles, the reinnervation is less effective and takes much longer because the basal lamina, which serves as structural support to the newly regenerating motor neurons, is compromised [134, 227]. Upon full reinnervation, the nerve activity further promotes the maturation of myofibers, which can be characterized by their ability to contract, and their expression to adult fast and slow myosin heavy chain (MHC) isoforms [134, 227]. At the end of the regeneration process, full structural and functional muscle restoration is achieved [227].
Hydrogel Biomaterials for Skeletal Muscle Regenerative Engineering (SMRE)
Based on the above discussion, we can speculate that the smECM holds a pivotal role during the regeneration process. Hence, much efforts have been devoted to developing biomimetic approaches, using the information present in the native smECM as a blueprint, to achieve a successful functional and structural muscle regeneration in post-traumatic injuries [27]. These efforts have resulted in the emergence of multiple biomaterial systems. Of these, hydrogels are the most preferable owing to their soft and viscoelastic biomimetic nature, which can more efficiently regulate muscle cell behavior, promote their myogenic differentiation, and allow for sufficient force production and contraction by the muscle tissue [28]. Hydrogels are 3D hydrophilic networks capable of retaining large contents of water or physiological fluids. Hence, they mimic to a greater degree the muscle tissue [135]. The advantages of hydrogels in comparison to other biomaterial systems are that besides their biocompatibility, their structure and mechanical properties are highly tunable, can be pre-shaped to match almost any desired geometry, and can easily be functionalized with various growth factors and other molecules to render them bioactive [136]. Currently, different classes of hydrogel biomaterials, including (1) conventional hydrogels, which can further be subcategorized into natural, synthetic, and composite hydrogels; and (2) dECM-derived hydrogels, have been developed to promote the regeneration of skeletal muscle (Fig. 4). The role of these hydrogels in SMRE will be discussed in greater detail in the following sections.
Conventional Hydrogels for SMRE
Natural Hydrogels
Natural hydrogels are one of the most popular choices for SMRE because they are primarily composed of ECM derivates. Thus, they possess the ability to induce skeletal muscle regeneration [137]. The most extensively investigated natural hydrogels for SMRE include collagen, fibrin, alginate, and agarose [137].
Collagen is a fibrous protein and the most abundant constituent in the smECM [137,138,139]. Collagen type I is the most predominantly utilized collagen form for SMRE, and can be derived from many tissues, such as bone, cartilage, ligament, and skin [139]. Collagen type I hydrogels can easily be fabricated under physiological conditions (e.g., pH of ~ 7.4, balanced salts, and 37 °C) via the self-assembly of solubilized type I collagen fibrils into a 3D mesh network [139]. Fibrin is a microfibrillar, branched polymer of fibrinogen formed by thrombin-catalyzed enzymatic polymerization, and it is the major component of blood clots [140, 140]. Furthermore, fibrinogen can directly be obtained from the patient’s blood plasma via precipitation methods [141], thus, lowering the risk of immunogenicity [142]. Alginate is a copolymer of (1,4)-linked β-d-mannuronic (block M unit) and α-l-guluronic (block G unit), and is often derived from brown seaweeds species [143]. A main advantage of alginate is its ability to rapidly turn into a gel state when positively charged divalent cations such as Ca2+, Ba2+, and Mg2+ are present [144]. Agarose is a natural polysaccharide derived from red seaweed and can also be found in the cell wall of certain marine algae [145]. The composition of agarose constitutes 1,3-linked β-d-galactopyranose and 1,4-linked 3,6-anhydro-α-l-galactopyranose [146]. Agarose-based hydrogels are obtained by dissolving agarose in hot water. Once the solution is allowed to cool down below 45 °C, a hydrogel can instantly be formed due to extensive hydrogen bonding between the agarose chains [147].
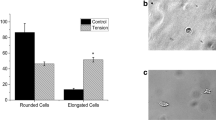
For SMRE, hydrogels should ideally be able to maintain their mechanical integrity (to prevent mechanical failure during myofiber contraction) while also supporting myogenesis. Pollet et al. evaluated the tensile strength and myogenic potential of collagen, fibrin, alginate, agarose, and collagen-chitosan hydrogels in vitro to estimate the best candidate for SMRE applications [137]. Mechanical evaluation studies revealed that collagen, collagen-chitosan, and fibrin had the highest elasticity as they could elongate at least 2-folds without failing (100% stretch). In contrast, agarose was the most brittle (20% maximum stretch), while alginate demonstrated poor handleability; thus, its mechanical characteristics could not be assessed. The different hydrogels were further evaluated in vitro for their myogenic potential using primary rat SSCs. Gene expression evaluations against MyoD-(early marker), MyoG-(mid-marker), and MHC-(late marker) showed that the expression of MyoD declined during the 14-day study period in all groups while MyoG expressions elevated on day 7 before falling again by day 14 of culture. Out of all the tested hydrogels, only fibrin could support higher expression of MHC on both timepoints. Based on the mechanical characteristics and gene expression patterns, it was concluded that fibrin provides a better hydrogel material for SMRE, followed by collagen. Based on these findings, several other studies investigated the myogenic potential of fibrin hydrogels toward SMRE in vitro and in vivo with promising results [148, 149]. These studies either utilized fibrin alone or with the addition of other ECM derivatives[150], such as laminin, to improve its biological performance with myoblasts.
In addition to the hydrogels mentioned above, gelatin is another widely used natural hydrogel for SMRE. Gelatin is the denatured form of collagen, which is obtained through a hydrolysis process [138, 151]. Gelatin retains the cell-binding arginylglycylaspartic acid (RGD) sites as a collagen derivative. Also, gelatin exhibits much lower immunogenicity potential than collagen because it contains less aromatic groups [152, 153]. Moreover, gelatin has unique thermo-responsive properties characterized by its ability to form structurally and mechanically stable hydrogels at temperatures below 30 °C but liquefies as single-coils at physiological temperatures. Various crosslinking agents could be used to retain/maintain the structural and mechanical stability of gelatin hydrogels, including microbial transglutaminase (mTG) or genipin [154,155,156]. Gupta et al. fabricated micro-molded gelatin hydrogels crosslinked with mTG and were incubated under physiological conditions (in culture media at 37 °C and 5% CO2), during which their mechanical characteristics were examined [157]. It was found that the hydrogels displayed stable elastic modulus and structure during the 2-week incubation period, suggesting that crosslinking with mTG is an efficient method to obtain mechanically and structurally stable gelatin hydrogels. In addition, C2C12 or primary chick myoblasts seeded on the micro-molded gelatin hydrogels could fuse into aligned and multinucleated myotubes with relatively mature sarcomeres [157]. These data suggest that gelatin hydrogels could be used as structurally and mechanically stable hydrogels that possess a myogenic potential for SMRE.
Despite being natural, however, the above hydrogels are not without limitations. For instance, collagen type I hydrogels were shown to prevent myofiber contraction in vitro and induce fibrosis upon transplantation in vivo [158,159,160]. On the other hand, cell-loaded fibrin hydrogels possess a poor degradation profile. They can undergo a rapid enzymatic degradation within the first few days of culture unless some enzymatic inhibitors, such as aprotinin or 6-aminocaproic acid, are supplemented in the culture media [161]. This suggests that fibrin hydrogels could undergo rapid degradation once administered in vivo, thus, masking their function to act as structural supports for muscle cell migration, growth, and differentiation during the initial regenerative phase. Alginate and agarose exhibit several limitations, including the lack of cell-supporting binding sites, their complete inertness, and poor biodegradation in mammalian systems due to their plant-based source [162].
Moreover, crosslinking of gelatin with either mTG or genipin is time-consuming and can take up to several hours until a complete crosslinking is achieved [157, 163]. This can potentially result in an uneven cell distribution within the hydrogel due to gravity-driven sedimentation, which can force most of the encapsulated cells to settle down due to the delayed formation of a 3D mesh network. Other general limitations of natural hydrogels include low viscosity and poor mechanical and biodegradation properties, limiting their utility in various applications such as in 3D bioprinting [142].
Synthetic Hydrogels
The advantages of synthetic hydrogels over their naturally-derived counterparts are their tunable mechanical and biodegradation properties, ease of production, high availability, and lower cost [164]. In addition, synthetic hydrogels exhibit low immunogenicity and avoid patch-patch variation [164]. Despite these advantages, synthetic hydrogels are a less popular choice for SMRE than natural polymeric hydrogels for several reasons. First, synthetic hydrogels lack the ability to promote cell functions unless they are functionalized with bioactive cues [165]. Additionally, synthetic hydrogels are hydrophobic, often resulting in suboptimal cell attachment, leading to poor cell growth and differentiation, which can thus result in inferior tissue development and maturation[166]. Yet, many synthetic hydrogels have been investigated for SMRE, including poly(ethylene glycol) diacrylate (PEGDA), N-isopropyl acrylamide (NIPAAm), acrylic acid (AA), polyacrylamide (PAAm), polyvinyl alcohol (PVA), polylactic acid (PLA), polyethylene glycol (PEG), and polyurethane (PU) [167,168,169,170,171,172].
To discuss a few, Gilbert et al. showed that SSCs seeded on soft PEG hydrogels exhibiting a comparable muscle elasticity of 12 kPa were able to maintain their stemness compared to seeding them on polystyrene surfaces (~ 106 GPa). Furthermore, when these cells were detached and subsequently transplanted into a mouse model of muscle injury, they were able to show acceptable engraftment, dynamic proliferation, and self-renewal capacity [109].
In a different study, Vannozzi et al. developed a hydrogel-based skeletal muscle construct using low molecular weight PEGDA. The mechanical properties of the hydrogel were highly tunable; however, since PEGDA does not support cell attachment, the construct’s surface was coated with Irgacure 2959, gelatin methacrylate, laminin, and fibronectin before photo-crosslinking to allow for C2C12 attachment. Three-day post-seeding, C2C12 displayed high viability and enhanced attachment, characterized by their spindle-like morphology. Twenty-day post-seeding, C2C12 were found to have spread and covered the entire construct [170].
Browe et al. developed a biocompatible and electroactive hydrogel-based construct using PEGDA combined with AA for use in actuation while endorsing muscle tissue maturation [168]. By controlling the PEGDA:AA ratio, they could obtain similar values of elastic modules to those previously reported for mammals limb muscles [173]. During the 10-day culture period, good viability and metabolic activities in C2C12 were observed on most of the PEGDA: AA ratios; however, they showed the best viability and metabolic activity on the 1:4 hydrogel. While the strength of contraction was observed to be similar in all the PEGDA-based constructs, data revealed that the addition of AA was needed for the constructs to actuate. Also, further analysis showed that increasing the ratio of AA resulted in increasing the strength of contraction.
Although the above hydrogels were shown to be highly tunable and support muscle cell growth, differentiation, and maturation to some extent, other comparative studies confirmed that muscle cells behaved significantly much better within naturally-derived hydrogels [29].
Composite Hydrogels
To merge the best attributes of both synthetic and natural hydrogels for SMRE, research efforts over the past two decades have led to the emergence of composite hydrogels. Composite hydrogels are mostly made when one synthetic and one natural material are mixed together to produce a hydrogel [29]. One of the most popular choice for the synthetic component of composite hydrogels is PEG due to its unique biocompatibility and the ability to easily functionalize it with various bioactive functional groups and growth factors [174]. For instance, in one study, bFGF and human adipose-derived stem cells (h-ADSCs) were encapsulated within a tyramine functionalized gelatin-PEG (GPT) hydrogel. The hydrogel showed high biocompatibility and a controlled bFGF release profile. The hydrogel was further evaluated in vivo in a gastrocnemius muscle injury model for its ability to promote muscle regeneration and growth. It was demonstrated that treatment with a combination of GPT, h-ADSCs, and bFGF attenuated fibrosis and restored muscle mass and function 4-week post-transplantation [175].
In a different study, protein-PEG hybrid hydrogel was fabricated and used to co-deliver human-induced pluripotent stem cells-derived endothelial cells (hiPSCs-ECs) and VEGF in a gastrocnemius ischemia mouse model [176]. The results showed that the co-delivery of hiPSCs-ECs and VEGF significantly reduced inflammation and promoted de novo muscle regeneration 2-week post-delivery compared to the hydrogel alone or saline controls. Fuoco et al. developed a PEG-fibrinogen hydrogel to promote skeletal muscle regeneration [177]. The hydrogel was able to support the in vitro differentiation of muscle-derived pericytes into MHC + myofibers, as evidenced by immunofluorescent and western blotting analysis. The cell-ladened hydrogel was further evaluated in vivo in an immunodeficient subcutaneous mouse model for 30 days. Histological examination revealed that the cell-ladened hydrogel supported robust myofiber formation and blood vessel infiltration. In a different study, the same author transplanted the same cell-ladened hydrogel in vivo in a (tibialis anterior) TA VML model, resulting in the regeneration of an indistinguishable muscle to the native one [178].
Several other synthetic hydrogels have also been combined with natural materials for SMRE. In one study, methacrylated alginate was mixed with PEGDA [179]. The composite hydrogel was then subjected to lyophilization to introduce micro-pores within the hydrogel. Since alginate and PEGDA lack cell-binding sites, the resultant composite hydrogel was blended with RGD peptide to facilitate myoblasts’ attachment. The results from the in vitro studies showed that the composite hydrogel supported myoblasts’ attachment and infiltration into the hydrogel’s matrix and improved their viability and muscle-related gene expression,
The results from these studies show that by mixing natural and synthetic hydrogels, the best attributes of both could be obtained in one system, resulting in an enhanced microenvironmental niche with tunable properties for SMRE in comparison to using either one alone.
dECM-Derived Hydrogels for SMRE
Although, based on the above discussion that natural or composite hydrogels are superior in maintaining skeletal muscle cells’ viability, regulating their responses, and promoting their myogenic differentiation compared to their synthetic counterparts, they still lack many important instructive cues that are necessary for skeletal muscle regeneration, function, and homeostasis [104]. Due to the compositional, structural, and mechanical complexity of the smECM, these conventional hydrogels are unable to recapitulate all characteristics found in its ECM [2].
Since the native ECM provides a bioactive environment and contains all the essential components needed by the cells to properly function, the use of ECM without cells can offer an optimal cellular niche for tissue regeneration [180]. Decellularization can broadly be defined as the process of removing the cellular compartment of living tissues while retaining the bioactivity of the ECM [181]. Therefore, decellularization represents a valuable tool for obtaining acellular bioactive scaffolds that can support enhanced cellular functions in vitro and in vivo [181]. To date, ECM bio-scaffolds prepared from various decellularized tissues have been used to promote skeletal muscle regeneration post-traumatic injuries [180]. However, compared to ECM bio-scaffolds, an injectable form of the dECM can easily adapt to any defect’s 3D geometry, and it can be transported to the injury site with minimally invasive approaches [182]. In this context, gradual advancement of the decellularization technology has led, over the years, to the discovery that dECM bio-scaffolds could be converted into hydrogels, which greatly extended their potential applications toward skeletal muscle tissue regeneration and repair [30]. After Badylak et al. reported the formation of a dECM-derived hydrogel a decade and a half ago [182], several groups attempted to form hydrogels from various decellularized tissues for SMRE, as reviewed in greater detail in the next section.
The process of synthesizing dECM-derived hydrogels generally involves several steps (Fig. 5). First, the tissue is decellularized to remove the cellular component to reduce its immunogenicity. Post-decellularization, the ECM is lyophilized and grounded into a powder, followed by digestion and solubilization in an acidic-based pepsin solution under vortexing to prepare a homogeneous ECM solution [183]. Finally, the solution’s salt ion concentration, pH, and temperature are adjusted to induce crosslinking to form a hydrogel. In theory, dECM hydrogels are formed via a self-assembly process of collagen molecules; and glycol proteoglycans, proteoglycans, and other essential tissue components that are inherently found in the dECM were shown to be important regulators of this process [31, 183]. It is noteworthy, although it is beyond the scope of the current review, that the protocol followed for decellularization can remarkably alter the biochemical composition of the ECM, which can in turn alter the hydrogel’s self-assembly gelation process [183]. For instance, Fu et al. evaluated the effect of the decellularization protocol on the gelation kinetics of ECM-derived hydrogels [184]. Out of the five tested protocols, tissues decellularized with sodium dodecyl sulfate (SDS) could not form a hydrogel. In contrast, tissues decellularized with sodium deoxycholate (SDC) and Triton X-100 successfully formed 3D and stable hydrogels. Since the tissue source was the same, these differences were attributed to the harsh effect of SDS on the ECM, while SDC and Triton X-100 were considered to be milder decellularization detergents although this is still questionable. Fernandez-Perez et al. recently demonstrated that the decellularization protocol not only could affect the biochemical composition and gelation kinetics of the formed hydrogel but also the behavior of the encapsulated cells [185]. It was reported that hydrogels derived from cornea ECM post-decellularization with SDS were completely cytotoxic, whereas hydrogels derived post-decellularization with Triton X-100 exhibited better cytocompatibility. Therefore, optimizing a decellularization protocol that can achieve maximal cell removal with minimal ECM damage and cytotoxic effects is crucial to attaining stable and cytocompatible hydrogels. For a detailed discussion on the impact of the decellularized protocol on the overall properties of the finally produced hydrogel, the readers are directed to the following references [185, 186].
The Biological Effects of Tissue-Specific Vs. Non-tissue-Specific dECM-Derived Hydrogels on Muscle Cell Behavior and Regeneration
For SMRE, dECM-derived hydrogels have been synthesized from various tissue sources, including lung [187], skin [188], blood vessels [189], urinary bladder [188], and skeletal muscles [190]. Wolf et al. developed dECM hydrogels derived from dermal and urinary bladders of pigs [188]. The hydrogels were encapsulated with C2C12, and their in vitro and in vivo myogenic potential were evaluated. In vitro, dermal ECM hydrogels supported greater C2C12 myoblast fusion compared to hydrogels derived from urinary bladders. In vivo, both hydrogels were degraded by day 35 of injection in a rat abdominal wall defect model, but the urinary bladder-derived hydrogels degraded much quicker and showed better myogenesis compared to dermal ECM-derived hydrogels. It was concluded that the ECM hydrogel properties may vary by the tissue source and that these properties can remarkably affect the behavior of the encapsulated cells.
Although the decellularized dermal and urinary bladder ECM-derived hydrogels were shown to provide biological cues that could promote myogenesis both in vitro and in vivo, it is likely that muscle-specific dECM-derived hydrogels would further improve this process. DeQuach et al. developed hydrogels derived from decellularized cardiac and skeletal muscle tissues [191]. The author could demonstrate that the biochemical composition of the decellularized matrices was highly unique. Mass spectrometry revealed a distinct biochemical profile of the respective decellularized cardiac and skeletal muscle tissues based on the differences observed in the collagen types, proteoglycanase, and glycoproteins. Therefore, due to the variations in the ECM’s components among the different tissues, it can be speculated that tissue-specific dECM-derived hydrogels can direct better physiologically relevant cellular responses over non-matched tissue sources [192]. In fact, this has been evidenced by a recent comparative study conducted by Ungerleider et al., where the myogenic potential of decellularized lung ECM (dlECM) and decellularized smECM (dsmECM)-derived hydrogels were evaluated in vivo [192]. Both hydrogels were decellularized using the same protocol and were obtained from the same donor to eliminate any variables in the results other than the sole material. Accordingly, it was hypothesized that the dsmECM would provide a better microenvironmental niche for enhanced muscle precursor cell growth and differentiation due to its tissue specificity. Both hydrogels were intramuscularly injected in a mouse muscle injury model, after which the injected materials were harvested for examination. On day 5, it was found that the density of pax7 + SSCs was dramatically elevated in muscles receiving the dsmECM-derived hydrogel. Moreover, muscle-related gene expression was found to be higher in the dsmECM-derived hydrogel group. On day 14, the dsmECM-derived hydrogels significantly enhanced muscle formation and maturation compared to dlECM-derived hydrogels.
The importance of utilizing tissue-specific dECM-derived hydrogels was further demonstrated in a recent study by Zhang et al. [193]. In this study, cells isolated from skin, skeletal muscle, and liver tissues were grown within dECM derived from skin, skeletal muscle, and liver tissues, with collagen type I serving as a control group. In vitro proliferation and differentiation, assessments revealed that each cell phenotype showed the best proliferation, phenotypic maintenance, and expression of tissue-specific proteins when grown within their native ECM. In a different study, Choi et al. developed dsmECM and decellularized vascular ECM-derived (dvECM) hydrogels [194]. Both hydrogels were encapsulated with human muscle progenitor cells (hMPCs) or human umbilical vein endothelial cells (HUVECs), and the behavior of both cells in both hydrogels was evaluated in vitro. It was found that both cell phenotypes exhibited the highest proliferation and tissue-specific gene expression when they were cultured in their tissue-specific dECM-derived hydrogels.
Taken together, these data indicate the tremendous regenerative capacity and myogenic potential of dsmECM hydrogels and suggest enormous benefits from using tissue-specific dECM hydrogels for each tissue targeted for repair.
The Current Applications of dECM-Derived Hydrogels for SMRE
As mentioned above, the ability to transform dECMs into hydrogels has dramatically widened their applications both in vitro and in vivo. However, as for SMRE, hydrogels derived from dsmECM have mostly been the primary focus in recent studies due to the developed knowledge on the effect of utilizing tissue-specific dECM-derived hydrogels over non-matched tissue sources on muscle cell behavior and muscle regeneration. Therefore, this section will only review the current applications of hydrogels derived from dsmECM toward skeletal muscle regeneration and repair.
dECM-Derived Hydrogels as Injectable Systems for Skeletal Muscle Regeneration Following Ischemia and VML
Regeneration strategies involving minimally invasive techniques offer several benefits, including a reduced infection risk at the site of surgery and a quicker recovery [223]. An injectable hydrogel delivers biomaterials through a syringe to fill in irregular defects [223]. In order to reduce the risks associated with invasive heart surgeries, injectable systems have mainly been developed to treat cardiac muscle [195,196,197, 223]. Although injectable systems for skeletal muscle regeneration, particularly injectable dsmECM-derived hydrogels, have received less attention, they still exist and, therefore, are worth investigating. In general, injectable dsmECM-derived hydrogels have either been used as a stand-alone biomaterial therapy or as vehicles for delivering therapeutic and/or regenerative cells to further enhance the regeneration process.
DeQuach et al. developed an injectable hydrogel derived from dsmECM and examined its potential for treating peripheral artery disease (PAD) in a rat hindlimb ischemia model [198]. At 3-, 5-, 7-, or 14-day post-injection, the muscles were harvested and examined for neovascularization, cell infiltration, and neo-muscle regeneration. Immunohistochemistry analysis demonstrated the positive effect of the injectable hydrogel on neovascularization. Additionally, significantly higher muscle progenitor cells were recruited and infiltrated into the dsmECM hydrogel compared to the control collagen type I hydrogel, suggesting its potential for treating patients with PAD. Based on these promising preliminary results, the same group conducted a follow-up study using the same biomaterial and animal model but for a more extended timepoint of 35 days to further validate the potential of the dsmECM hydrogel toward skeletal muscle regeneration and repair following ischemia [199]. Based on Laser Speckle Contrast Imaging, significant improvement in the hindlimb tissue perfusion and prefusion kinetics was observed on day 35 post-injection. In addition, histological assessments indicated that the injection of the dsmECM hydrogel resulted in arteriogenesis and improved the recruitment of muscle precursor cells to the injury site. Morphological assessments revealed that treatment with dsmECM hydrogels restored the healthy tissue morphology. Also, further assessments showed that the dsmECM hydrogel positively modulated the immune response, attenuated cell apoptosis, and enhanced angiogenesis and muscle formation. Based on these findings, it was concluded that the dsmECM hydrogel holds a great potential as an injectable biomaterial for treating PAD and restoring muscle mass after ischemia.
Besides using the dsmECM hydrogel as a stand-alone biomaterial therapy, it can also be used as a vehicle for the delivery of therapeutic and regenerative cells to further enhance its therapeutic effects. Rao et al. examined the potential of the dsmECM hydrogel as a cell delivery vehicle in a mouse hindlimb ischemia model [200]. It was demonstrated that the hydrogel system could improve the survival of the delivered skeletal muscle fibroblasts and myoblasts and their engraftment post-injection, resulting in enhanced perfusion and increased vascularization at early timepoints. In addition, further improvement in perfusion was observed at day 35 post-injection. These results suggest that the dsmECM-derived hydrogel can protect the cells in the initial ischemic environment post-delivery. In a separate study by Quarta et al., a dsmECM scaffold was loaded with a cell-ladened hydrogel composed of muscle progenitor cells encapsulated within a dsmECM-derived hydrogel, followed by transplantation in a mouse acute VML injury model [201]. Imaging of the bioconstruct indicated the survival and growth of the delivered cells, and ex vivo functional analysis revealed force restoration of treated muscles. Furthermore, histological assessments confirmed myofiber formation and neovascularization at the transplantation site. Further transplantation of the bioconstruct in a mouse chronic VML injury model resulted in enhanced neovascularization, restored biomechanical force production and muscle regeneration. Taken together, these results indicated that recapitulating the tissue environment in the delivery vehicle can enhance the delivery of cells to the skeletal muscle, induce neovascularization and promote muscle regeneration following ischemia and VML injuries.
dECM-Derived Hydrogels as Bioinks for 3D Bioprinting
As discussed previously, injectable hydrogels have several advantages. However, control over the architecture, porosity, and anisotropic cell organization can be difficult to modulate in injectable systems. Skeletal muscle is anisotropic and comprises tightly bundled and highly aligned structures of densely-packed myofibers [202]. In SMRE, inducing the differentiation of muscle cells toward a uniaxial direction is vital for the successful engineering of myo-mimetic skeletal muscles [203, 204]. Furthermore, the alignment-mediated cellular response has been shown to be determinantal in regulating myogenesis and functional myofiber development [205]. In this context, many engineering-based approaches have been developed to better control these features within hydrogels to produce myo-mimetic skeletal muscle tissues [27]. Of these, 3D bioprinting is emerging as a tool to produce bioengineered constructs with pre-defined architecture and pore volume, a high level of anisotropic cellular organization, and the possibility of combining different cell-laden hydrogels, the so-called bioinks [206]. Although synthetic and natural materials are frequently being used to develop bioinks for SMRE, they lack the complexity found in the native smECM and are not tissue-specific.
The exceptional biochemical composition of dECMs and possibility of transforming them to a liquid form, render them as attractive candidates to serve as bioinks for tissue bioprinting. Thus, it can be speculated that the use of skeletal muscle-specific bioinks in 3D bioprinting technology will provide tissue-specific biochemical and topographical cues. In this regard, impressive effort has been accomplished in 3D bioprinting of dECMs in the past decade toward SMRE. Choi et al. fabricated a 3D-bioprinted construct made entirely from dsmECM-derived bioink [207]. The bioprinted construct supported C2C12 viability, alignment along the longitudinal printing axis, and myotube formation. Of particular interest, the dsmECM-based bioprinted constructs supported greater formation of AChRs within the formed myotubes in comparison to the collagen-based bioprinted constructs. Finally, the mechanical properties of the dsmECM-based 3D-bioprinted construct at day 14 of culture exhibited a similar stiffness to the native muscle. Encouraged by their findings, the same group further examined the potential of their dsmECM-based bioprinted construct to support the functional and structural regeneration of skeletal muscle in a rat TA VML model [194]. In this study, the 3D-bioprinted constructs were produced using hMPCs to increase the clinical relevance. The author compared the bioprinted construct with a non-printed cell-ladened dsmECM construct. Four-week post-transplantation, de novo muscle formation was seen in the non-printed and 3D-bioprinted construct groups. However, the 3D-bioprinted construct supported improved structural and functional muscle regeneration. While improved functional recovery was observed in the 3D-bioprinted group, only 71% of the contractile function was restored. In the latter study, the author employed a co-axial bioprinting approach to produce a composite structure comprising a dsmECM cell-laden core enveloped by a dvECM cell-laden as a shell. The dsmECM cell-laden core contained hMPCs, whereas the dvECM cell-laden shell contained HUVECs. Surprisingly, when the composite bioprinted constructed was transplanted into the same rat TA VML model, the functional recovery was further increased to 85% compared to the uninjured sham control group. The results of these studies highlight the beneficial effects of combining 3D bioprinting technology with dECM hydrogels derived from the tissue of origin toward efficient skeletal muscle regeneration [223]. Even though utilizing dsmECM hydrogels in SMRE is still a new area of research, these early findings provided evidence of their therapeutic and regenerative potential [223]. Yet, many hurdles and challenges are still to overcome before their presence in the clinic can be a reality, which will be discussed in the next section in greater details.
Future Prospective Toward the Clinical Translation of dECM-Derived Hydrogels
Decellularization Method
This review article aims to comprehensively summarize the current progress of a novel class of biomaterial, dECM-derived hydrogels, toward SMRE. While the utility of dECM-derived hydrogels paved the way to numerous breakthrough findings, much work needs to be conducted prior to their utilization in clinical practices. The goal of decellularization is to eliminate all the cellular content within the ECM while fully retaining its biochemical composition to obtain the desired benefits from using such materials [224]. Based on the current literature, it seems that an optimal decellularization protocol, particularly for skeletal muscles, that achieves a balance between these two factors does not exist. None of the decellularization protocols currently used to derive hydrogels from skeletal muscle have led to their clinical translation, in contrast to other dECM-derived hydrogels such as decellularized cardiac muscle ECM-derived hydrogels [208]. Skeletal muscle is a thick and robust tissue; therefore, using harsh detergents such as SDS alone or in combination with Triton X-100 is often desired for an efficient decellularization [207, 209]. While all studies utilizing these detergents have achieved complete removal of the cellular component, none has reported full preservation of the biochemical properties when compared to the native non-decellularized tissue [209]. Perhaps, a combination of grinding the tissue into smaller fragments using an off-the-shelf meat grinder and milder decellularization detergents or detergent-free decellularization methods (e.g., mechanical disruption through freeze-thaw cycles) could be enough to completely release the cells out of the ECM while greatly preserving its inherent biochemical components. Another potential method, although it may not be practical for mass production of dECM-derived hydrogels, is embedding the tissue in frozen media, followed by sectioning it into thin sheets prior to decellularization with mild methods. In this case, harsh decellularization detergents may not be required for efficient cellular removal since the thickness of the tissue has significantly been reduced, thus, ensuring full cellular removal while preserving the ECM’s biochemical blueprint.
Immunogenicity
Besides the lack of an optimal decellularization protocol, there is an unmet need to standardize decellularization protocols. The high variability in decellularization detergents, detergent concentrations, incubation times, and conditions (at room temperature vs. at 4 °C) makes deriving general conclusions extremely difficult. In addition, the variability in the origin of the harvested muscle, the species used, animal strain, and whether the animals used are obtained from slaughterhouses or certified animal institutions renders difficulty in comparing the results among the studies and can often lead to contradicting results. Hence, comparative studies that consider the factors mentioned above are immensely needed. Although criteria to assess the efficiency of cellular removal post-decellularization do exist [210], no criteria exist for other factors, for instance, the effect of the decellularization protocol on the biochemical, physical, mechanical, and biological characteristics of the ECM [225]. Since each tissue possesses its own unique characteristics, it is crucial to establish specific guidelines that meet each tissue’s needs during decellularization. Therefore, a thorough understanding of the relationship between tissue-specific characteristics and decellularization protocols would greatly advance the field and bring dECM-based materials one step closer to clinical translation. Also, as dECM-derived hydrogels continue to be evaluated for various applications, the differences between laboratory and manufacturing-scale batches should be carefully considered to avoid costly and timely optimization during scale-up.
An emerging issue is that most of the current studies, if not all, focus on assessing the characteristics of the ECM post-decellularization rather than assessing the properties of the finally-formed hydrogels. A common lack of knowledge in the field is that the additional steps following decellularization, such as grinding, solubilization, and neutralization, can have additional effects on the properties of the dECM. For example, the sGAG and collagen content were lower in dECM-derived hydrogels compared to the intact dECMs [211]. These data indicate that the initial characteristics revealed from dECMs do not necessarily replicate in the finally-formed hydrogel. Therefore, future research should focus more on characterizing the finally-formed hydrogel rather than the dECM since the hydrogel is the final product.
Based on the current literature, deriving hydrogels from allogenic sources is not always a viable option due to the limited supply of donor tissues. Therefore, most of the existing studies have utilized tissues from animal sources. In these studies, much focus is paid to removing the cellular component from the tissue, with less emphasis on removing other immunogenic elements. Although decellularization can efficiently remove the cellular component from the tissue, it is unable to remove other components that are also known to elicit adverse immunological responses in human systems, such as xeno-antigens [212]. The α-Gal (α1,3-Gal-epitopes) is the primary xeno-antigen that induces hyperacute rejection of animal tissue-derived materials in humans [213,214,215, 226]. Furthermore, the remnant α-Gal antigen in animal tissue-derived materials has been found to elicit adverse immunologic responses in humans, which can negatively disturb the outcome of tissue remodeling [216, 226]. All non-primate mammals, including pigs, rodents, bovine, rabbits, horses, prosimians, and New World monkeys, carry the α-Gal antigen in their tissues [217, 226]. Therefore, to facilitate the clinical translation of hydrogels derived from decellularized animal matrices, decellularization should be accompanied by the removal of the α-Gal epitopes to reduce the overall immunogenicity of such materials. In this context, previous works have demonstrated several protocols for removing the α-Gal antigen from xeno-tissues, which mainly focus on treating the ECM post-decellularization with α-Galactosidase. This enzyme specifically cleaves the α-Gal antigen from tissues without masking their regenerative potential [218, 219]. In these studies, the M86 antibody has been used as a qualitative method to assess the efficiency of the α-Gal antigen removal via immunohistochemistry [218, 219]. Although not widely used due to their novelty, few studies have recently reported quantitative methods for determining the remnant content of the α-Gal antigen in the xeno-tissues post-treatment with α-Galactosidase [218, 219]. These recommendations could also be applied to other tissues, including skeletal muscles, to further reduce their overall immunogenicity post-decellularization for potential clinical acceptance.
It is important to note that while the methods described above can provide preliminary confirmation on the efficacy of the α-Gal antigen removal from xeno-tissues, in vivo assessments are no doubt necessary to further confirm these findings. A valid method to predict the biocompatibility and immunogenicity of such materials in the human system would be to administer them in xenotransplantation animal models, followed by a comprehensive assessment of the immune response to the administered biomaterials [220]. Non-human primate, and α-Gal Knockout pig and mice models have widely been used in xenotransplantation studies to predict the biocompatibility and immunogenicity of xeno-grafts. The non-human primate model, for instance, was successfully utilized to evaluate the host immune response to decellularized lungs obtained from wild-type and α-Gal Knockout pigs. A favorable immune response was revealed to the decellularized lungs obtained from the α-Gal Knockout pigs compared to the wild-type pigs, suggesting that removing the α-Gal antigen from xenogenic tissues can significantly reduce their overall immunogenicity [221].
While initial promising in vivo results were demonstrated in terms of the myogenic potential of the dsmECM-derived hydrogels, no study has so far been attempted to remove the α-Gal antigen from such hydrogels, nor thoroughly evaluated their immunogenicity in vivo (Fig. 6). In addition, most of the in vivo studies to assess the regenerative potential of the dsmECM-derived hydrogel have currently been conducted in immunodeficient animal models. Therefore, immunogenicity data of this hydrogel is presently lacking. While results from these studies could provide initial information about the myogenic potential of the dsmECM hydrogel, they do not necessarily reveal its true potential for regeneration, as the immune response plays a major role during the regeneration process. Therefore, future studies should focus on implementing a step for the removal of the α-Gal antigen from the smECM during decellularization and prior to hydrogel formation, followed by thorough immunological evaluations. Additionally, future studies should consider utilizing immunocompetent animal models when evaluating the myogenic potential of the dsmECM hydrogel. We believe that carrying out such investigations would greatly accelerate the clinical presence of dECM-derived hydrogels, particularly those derived from the skeletal muscle tissue.
Publication trend on A the removal of α-Gal antigen from decellularized tissues and skeletal muscle tissues; and B the immunogenicity of dECM-derived hydrogels and dsmECM-derived hydrogels. (data obtained from PubMed; search strings for A “removal of α-Gal antigen from” and “decellularized tissues” or “decellularized skeletal muscle tissues”, search strings for B “immunogenicity of” and “decellularized ECM-derived hydrogels” or “decellularized skeletal muscle ECM-derived hydrogels”)
Sterilization
Another crucial factor for clinical applications is sterilization. Sterilization can broadly be defined as the process of killing all microorganisms in a matter [222]. Although often neglected, previous works have demonstrated several sterilization methods with well-defined advantages and disadvantages [222]. These sterilization methods include gamma irradiation, ethylene oxide (ETO), and ultraviolet ray (UV). Gamma irradiation has no cytotoxic effects and exhibits a strong penetrability, making it one of the most preferred methods for sterilizing dECM-derived products. However, Gamma irradiation can cause alteration to the physical and chemical properties and bioactivity of the ECM. ETO is the second most preferred sterilization method due to its strong penetrability and less damaging effects on the ECM. Yet, ETO has cytotoxic effects and has been shown to react with water to produce toxic substances. UV is a simpler and easier method to disinfect surfaces but has weak penetrability [222]. A possible approach to avoid the drawbacks of the current sterilization methods is to carry out the entire process of decellularization and preparation of the ECM-derived products under aseptic conditions (e.g., utilizing sterile tools and detergents).
When it comes to the diversity in applications, dECM-derived hydrogels make an ideal example. For instance, the ability to use them as injectable systems to fill irregular defect geometries remarkably overcame the limitation of using a whole intact decellularized muscle. An injectable hydrogel derived from decellularized cardiac-ECM has already reached the clinical settings, with a published first-in-human study demonstrating this [208]. It will be crucial to investigate whether the dsmECM-derived hydrogels can also be used in patients. We anticipate that if future research efforts consider all the aforementioned factors, the clinical translation of the dsmECM-derived hydrogels can one day become a reality.
Extended Applications
Besides the ability to use dECM-derived hydrogels as injectable systems, they could also serve as bioinks for 3D bioprinting, as discussed above. With this technology, it might be possible to create analogs of human tissues that can subsequently be transplanted, thus, overcoming the issue of the limited human tissue supply. However, the poor mechanical properties and structural stability of dECM-derived hydrogels may be a major hurdle to achieving this goal. To overcome this limitation, dECM-derived hydrogels could mechanically be reinforced with electrospun fibers derived from decellularized skeletal muscles or chemically functionalized to render them photo- or enzymatically cross-linkable without masking their regenerative potential. Further research in this direction will be necessary to enable using them for the production of large-volume and biomimetic skeletal muscle constructs for clinical practices.
In conclusion, although much progress has been made in the field of dECM-derived hydrogels toward SMRE, it is still in its infant stage. Nevertheless, we believe that improving and standardizing the methods of decellularization, further lowering the immunogenicity of dECMs through paying more attention to the removal of xenogeneic antigens and carrying out the in vivo investigations in immunocompetent as well as larger animal models should advance their clinical utility.
References
Mulbauer GD, Matthew HWT. Biomimetic scaffolds in skeletal muscle regeneration. Discoveries. 7(1):e90, https://doi.org/10.15190/d.2019.3 (Craiova).
Csapo R, Gumpenberger M, Wessner B. Skeletal muscle extracellular matrix - what do we know about its composition, regulation, and physiological roles? A Narrative Review. Front Physiol. 2020;11:253. https://doi.org/10.3389/fphys.2020.00253.
Rowland LA, Bal NC, Periasamy M. The role of skeletal-muscle-based thermogenic mechanisms in vertebrate endothermy. Biol Rev Camb Philos Soc. 2015;90(4):1279–97. https://doi.org/10.1111/brv.12157.
Baskin KK, Winders BR, Olson EN. Muscle as a “mediator“ of systemic metabolism. Cell Metab. 2015;21(2):237–48. https://doi.org/10.1016/j.cmet.2014.12.021.
Pedersen BK, Febbraio MA. Muscles, exercise and obesity: skeletal muscle as a secretory organ. Nat Rev Endocrinol. 2012;8(8). https://doi.org/10.1038/nrendo.2012.49.
Kasukonis B, et al. Codelivery of infusion decellularized skeletal muscle with minced muscle autografts improved recovery from volumetric muscle loss injury in a rat model. Tissue Eng Part A. 2016;22(19–20):1151–63. https://doi.org/10.1089/ten.TEA.2016.0134.
Corona BT, Rivera JC, Owens JG, Wenke JC, Rathbone CR. Volumetric muscle loss leads to permanent disability following extremity trauma. J Rehabil Res Dev. 2015;52(7):785–92. https://doi.org/10.1682/JRRD.2014.07.0165.
Grogan BF, Hsu JR, STR Consortium. Volumetric muscle loss. JAAOS - J Am Acad Orthop Surg. 2011;19:S35.
Liu J, Saul D, Böker KO, Ernst J, Lehman W, Schilling AF. Current methods for skeletal muscle tissue repair and regeneration. BioMed Res Int. 2018. https://www.hindawi.com/journals/bmri/2018/1984879/. Accessed 04 Oct 2018.
Kalyani RR, Corriere M, Ferrucci L. Age-related and disease-related muscle loss: the effect of diabetes, obesity, and other diseases. Lancet Diabetes Endocrinol. 2014;2(10):819–29. https://doi.org/10.1016/S2213-8587(14)70034-8.
West SL, Lok CE, Jamal SA. Fracture risk assessment in chronic kidney disease, prospective testing under real world environments (FRACTURE): a prospective study. BMC Nephrol. 2010;11:17. https://doi.org/10.1186/1471-2369-11-17.
Bianchi B, Copelli C, Ferrari S, Ferri A, Sesenna E. Free flaps: outcomes and complications in head and neck reconstructions. J Craniomaxillofac Surg. 2009;37(8):438–42. https://doi.org/10.1016/j.jcms.2009.05.003.
Aguilar CA, et al. Multiscale analysis of a regenerative therapy for treatment of volumetric muscle loss injury. Cell Death Discov. 2018;4:33. https://doi.org/10.1038/s41420-018-0027-8.
Eckardt A, Fokas K. Microsurgical reconstruction in the head and neck region: an 18-year experience with 500 consecutive cases. J Craniomaxillofac Surg. 2003;31(4):197–201. https://doi.org/10.1016/s1010-5182(03)00039-8.
Stevanovic MV, Cuéllar VG, Ghiassi A, Sharpe F. Single-stage reconstruction of elbow flexion associated with massive soft-tissue defect using the latissimus dorsi muscle bipolar rotational transfer. Plast Reconstr Surg Glob Open. 2016;4(9):e1066. https://doi.org/10.1097/GOX.0000000000001066.
Barrera-Ochoa S, Collado-Delfa JM, Sallent A, Lluch A, Velez R. Free neurovascular latissimus dorsi muscle transplantation for reconstruction of hip abductors. Plast Reconstr Surg Glob Open. 2017;5(9):e1498. https://doi.org/10.1097/GOX.0000000000001498.
Laurencin CT, Khan Y. Regenerative engineering. Sci Transl Med. 2012;4(160):160ed9. https://doi.org/10.1126/scitranslmed.3004467.
Laurencin, C.T., & Khan, Y. (Eds.). (2013) Regenerative Engineering (1st ed.). CRC Press. https://doi.org/10.1201/b14925
Laurencin CT, Nair LS. The Quest toward limb regeneration: a regenerative engineering approach. Regen Biomater. 2016;3(2):123–5. https://doi.org/10.1093/rb/rbw002.
Mengsteab PY, Freeman J, Barajaa MA, Nair LS, Laurencin CT. Ligament regenerative engineering: braiding scalable and tunable bioengineered ligaments using a bench-top braiding machine. Regen Eng Transl Med. 2020. https://doi.org/10.1007/s40883-020-00178-8.
Barajaa MA, Nair LS, Laurencin CT. Bioinspired scaffold designs for regenerating musculoskeletal tissue interfaces. Regen Eng Transl Med. 2019. https://doi.org/10.1007/s40883-019-00132-3.
Daneshmandi L, Barajaa M, Tahmasbi Rad A, Sydlik SA, Laurencin CT. Graphene-based biomaterials for bone regenerative engineering: a comprehensive review of the field and considerations regarding biocompatibility and biodegradation. Adv Healthc Mater. 2021;10(1):2001414. https://doi.org/10.1002/adhm.202001414.
Barajaa MA, Nair LS, Laurencin CT. Robust phenotypic maintenance of limb cells during heterogeneous culture in a physiologically relevant polymeric-based constructed graft system. Sci Rep. 2020;10(1):11739. https://doi.org/10.1038/s41598-020-68658-z.
Ogueri KS, et al. In vivo evaluation of the regenerative capability of glycylglycine ethyl ester-substituted polyphosphazene and poly(lactic-co-glycolic acid) blends: a rabbit critical-sized bone defect model. ACS Biomater Sci Eng. 2021;7(4):1564–72. https://doi.org/10.1021/acsbiomaterials.0c01650.
Seyedsalehi A, Daneshmandi L, Barajaa M, Riordan J, Laurencin CT. Fabrication and characterization of mechanically competent 3D printed polycaprolactone-reduced graphene oxide scaffolds. Sci Rep. 2020;10(1):22210. https://doi.org/10.1038/s41598-020-78977-w.
Chan BP, Leong KW. Scaffolding in tissue engineering: general approaches and tissue-specific considerations. Eur Spine J. 2008;17(Suppl 4):467–79. https://doi.org/10.1007/s00586-008-0745-3.
Tang X, Daneshmandi L, Awale G, Nair LS, Laurencin CT. Skeletal muscle regenerative engineering. Regen Eng Transl Med. 2019;5(3):233–51. https://doi.org/10.1007/s40883-019-00102-9.
Lev R, Seliktar D. Hydrogel biomaterials and their therapeutic potential for muscle injuries and muscular dystrophies. J R Soc Interface. 2018;15(138):20170380. https://doi.org/10.1098/rsif.2017.0380.
Fischer KM, et al. Hydrogels for skeletal muscle regeneration. Regen Eng Transl Med. 2021;7(3):353–61. https://doi.org/10.1007/s40883-019-00146-x.
Saldin LT, Cramer MC, Velankar SS, White LJ, Badylak SF. Extracellular matrix hydrogels from decellularized tissues: structure and function. Acta Biomater. 2017;49:1–15. https://doi.org/10.1016/j.actbio.2016.11.068.
Boso D, Maghin E, Carraro E, Giagante M, Pavan P, Piccoli M. Extracellular matrix-derived hydrogels as biomaterial for different skeletal muscle tissue replacements. Materials. 2020;13(11):2483. https://doi.org/10.3390/ma13112483. (Basel).
Yue B. Biology of the extracellular matrix: an overview. J Glaucoma. 2014;23(8 Suppl 1):S20-23. https://doi.org/10.1097/IJG.0000000000000108.
Brown BN, Badylak SF. Extracellular matrix as an inductive scaffold for functional tissue reconstruction. Transl Res. 2014;163(4):268–85. https://doi.org/10.1016/j.trsl.2013.11.003.
Gillies AR, Lieber RL. Structure and function of the skeletal muscle extracellular matrix. Muscle Nerve. 2011;44(3):318–31. https://doi.org/10.1002/mus.22094.
The importance of extracellular matrix in skeletal muscle development and function | IntechOpen. https://www.intechopen.com/chapters/49985. Accessed 06 Jun 2022.
Purslow PP. The structure and role of intramuscular connective tissue in muscle function. Front Physiol. 2020;11:495. https://doi.org/10.3389/fphys.2020.00495.
Passerieux E, et al. Structural organization of the perimysium in bovine skeletal muscle: Junctional plates and associated intracellular subdomains. J Struct Biol. 2006;154(2):206–16. https://doi.org/10.1016/j.jsb.2006.01.002.
Stecco C, Hammer W, Vleeming A, De Caro R. 3 - Deep Fasciae. In Functional Atlas of the Human Fascial System, C. Stecco, W. Hammer, A. Vleeming, and R. De Caro, Eds., Churchill Livingstone; 2015. pp. 51–102. https://doi.org/10.1016/B978-0-7020-4430-4.00003-8.
Sanes JR. The basement membrane/basal lamina of skeletal muscle. J Biol Chem. 2003;278(15):12601–4. https://doi.org/10.1074/jbc.R200027200.
Khalilgharibi N, Mao Y. To form and function: on the role of basement membrane mechanics in tissue development, homeostasis and disease. Open Biol. 2021;11(2):200360. https://doi.org/10.1098/rsob.200360.
Holmberg J, Durbeej M. Laminin-211 in skeletal muscle function. Cell Adh Migr. 2013;7(1):111–21. https://doi.org/10.4161/cam.22618.
Kjær M. Role of extracellular matrix in adaptation of tendon and skeletal muscle to mechanical loading. Physiol Rev. 2004;84(2):649–98. https://doi.org/10.1152/physrev.00031.2003.
Janson IA, Putnam AJ. Extracellular matrix elasticity and topography: material-based cues that affect cell function via conserved mechanisms. J Biomed Mater Res A. 2015;103(3):1246–58. https://doi.org/10.1002/jbm.a.35254.
Grzelkowska-Kowalczyk K. The importance of extracellular matrix in skeletal muscle development and function. IntechOpen. 2016. https://doi.org/10.5772/62230.
Thorsteinsdóttir S, Deries M, Cachaço AS, Bajanca F. The extracellular matrix dimension of skeletal muscle development. Dev Biol. 2011;354(2):191–207. https://doi.org/10.1016/j.ydbio.2011.03.015.
Takala TE, Virtanen P. Biochemical composition of muscle extracellular matrix: the effect of loading. Scand J Med Sci Sports. 2000;10(6):321–5. https://doi.org/10.1034/j.1600-0838.2000.010006321.x.
Halper J, Kjaer M. Basic components of connective tissues and extracellular matrix: elastin, fibrillin, fibulins, fibrinogen, fibronectin, laminin, tenascins and thrombospondins. Adv Exp Med Biol. 2014;802:31–47. https://doi.org/10.1007/978-94-007-7893-1_3.
Duance VC, Restall DJ, Beard H, Bourne FJ, Bailey AJ. The location of three collagen types in skeletal muscle. FEBS Lett. 1977;79(2):248–52. https://doi.org/10.1016/0014-5793(77)80797-7.
Light N, Champion AE. Characterization of muscle epimysium, perimysium and endomysium collagens. Biochem J. 1984;219(3):1017–26. https://doi.org/10.1042/bj2191017.
McKee TJ, Perlman G, Morris M, Komarova SV. Extracellular matrix composition of connective tissues: a systematic review and meta-analysis. Sci Rep. 2019;9(1):10542. https://doi.org/10.1038/s41598-019-46896-0.
Kovanen V. Intramuscular extracellular matrix: complex environment of muscle cells. Exerc Sport Sci Rev. 2002;30(1):20–5. https://doi.org/10.1097/00003677-200201000-00005.
Sand JMB, Genovese F, Gudmann NS, Karsdal MA. Chapter 4 - Type IV collagen. In Biochemistry of Collagens, Laminins and Elastin (Second Edition), M. A. Karsdal, Ed., Academic Press; 2019. pp. 37–49. https://doi.org/10.1016/B978-0-12-817068-7.00004-5.
Leeming DJ, Karsdal MA. Chapter 5 - Type V collagen. In Biochemistry of Collagens, Laminins and Elastin (Second Edition), M. A. Karsdal, Ed., Academic Press; 2019. pp. 51–57. https://doi.org/10.1016/B978-0-12-817068-7.00005-7.
Bönnemann CG. The collagen VI-related myopathies: muscle meets its matrix. Nat Rev Neurol. 2011;7(7):379–90. https://doi.org/10.1038/nrneurol.2011.81.
Sabatelli P, et al. Expression of collagen VI α5 and α6 chains in human muscle and in Duchenne muscular dystrophy-related muscle fibrosis. Matrix Biol. 2012;31(3):187–96. https://doi.org/10.1016/j.matbio.2011.12.003.
Cescon M, Gattazzo F, Chen P, Bonaldo P. Collagen VI at a glance. J Cell Sci. 2015;128(19):3525–31. https://doi.org/10.1242/jcs.169748.
Chiquet M, Birk DE, Bönnemann CG, Koch M. Collagen XII: protecting bone and muscle integrity by organizing collagen fibrils. Int J Biochem Cell Biol. 2014;53:51–4. https://doi.org/10.1016/j.biocel.2014.04.020.
Koch M, et al. A novel marker of tissue junctions, collagen XXII. J Biol Chem. 2004;279(21):22514–21. https://doi.org/10.1074/jbc.M400536200.
Cornelison DD, Filla MS, Stanley HM, Rapraeger AC, Olwin BB. Syndecan-3 and syndecan-4 specifically mark skeletal muscle satellite cells and are implicated in satellite cell maintenance and muscle regeneration. Dev Biol. 2001;239(1):79–94. https://doi.org/10.1006/dbio.2001.0416.
Handley CJ, Samiric T, Ilic MZ. Structure, metabolism, and tissue roles of chondroitin sulfate proteoglycans. Adv Pharmacol. 2006;53:219–32. https://doi.org/10.1016/S1054-3589(05)53010-2.
Brandan E. Proteoglycans in skeletal muscle. Braz J Med Biol Res. 1994;27(9):2109–16.
Kölbel H, Hathazi D, Jennings M, Horvath R, Roos A, Schara U. Identification of candidate protein markers in skeletal muscle of laminin-211-deficient CMD type 1A-patients. Front Neurol. 2019;10. [Online]. Available: https://www.frontiersin.org/article/https://doi.org/10.3389/fneur.2019.00470. Accessed 06 Jun 2022.
Boppart MD, Mahmassani ZS. Integrin signaling: linking mechanical stimulation to skeletal muscle hypertrophy. Am J Physiol Cell Physiol. 2019;317(4):C629–41. https://doi.org/10.1152/ajpcell.00009.2019.
Franchi MV, Reeves ND, Narici MV. Skeletal muscle remodeling in response to eccentric vs. concentric loading: morphological, molecular, and metabolic adaptations. Front Physiol. 2017;8. [Online]. Available: https://www.frontiersin.org/article/https://doi.org/10.3389/fphys.2017.00447. Accessed 23 Jun 2022.
Guérin CW, Holland PC. Synthesis and secretion of matrix-degrading metalloproteases by human skeletal muscle satellite cells. Dev Dyn. 1995;202(1):91–9. https://doi.org/10.1002/aja.1002020109.
Nishimura T, Nakamura K, Kishioka Y, Kato-Mori Y, Wakamatsu J, Hattori A. Inhibition of matrix metalloproteinases suppresses the migration of skeletal muscle cells. J Muscle Res Cell Motil. 2008;29(1):37–44. https://doi.org/10.1007/s10974-008-9140-2.
Thomas K, Engler AJ, Meyer GA. Extracellular matrix regulation in the muscle satellite cell niche. Connect Tissue Res. 2015;56(1):1–8. https://doi.org/10.3109/03008207.2014.947369.
Jakobsen JR, Mackey AL, Knudsen AB, Koch M, Kjaer M, Krogsgaard MR. Composition and adaptation of human myotendinous junction and neighboring muscle fibers to heavy resistance training. Scand J Med Sci Sports. 2017;27(12):1547–59. https://doi.org/10.1111/sms.12794.
Härönen H, et al. Collagen XIII secures pre- and postsynaptic integrity of the neuromuscular synapse. Hum Mol Genet. 2017;26(11):2076–90. https://doi.org/10.1093/hmg/ddx101.
Heikkinen A, Härönen H, Norman O, Pihlajaniemi T. Collagen XIII and other ECM components in the assembly and disease of the neuromuscular junction. Anat Rec. 2020;303(6):1653–63. https://doi.org/10.1002/ar.24092. (Hoboken).
Eklund L, et al. Lack of type XV collagen causes a skeletal myopathy and cardiovascular defects in mice. Proc Natl Acad Sci. 2001;98(3):1194–9. https://doi.org/10.1073/pnas.98.3.1194.
Guillon E, Bretaud S, Ruggiero F. Slow muscle precursors lay down a collagen XV matrix fingerprint to guide motor axon navigation. J Neurosci. 2016;36(9):2663–76. https://doi.org/10.1523/JNEUROSCI.2847-15.2016.
Heljasvaara R, Aikio M, Ruotsalainen H, Pihlajaniemi T. Collagen XVIII in tissue homeostasis and dysregulation - lessons learned from model organisms and human patients. Matrix Biol. 2017;57–58:55–75. https://doi.org/10.1016/j.matbio.2016.10.002.
Khaleduzzaman M, Sumiyoshi H, Ueki Y, Inoguchi K, Ninomiya Y, Yoshioka H. Structure of the human type XIX collagen (COL19A1) gene, which suggests it has arisen from an ancestor gene of the FACIT family. Genomics. 1997;45(2):304–12. https://doi.org/10.1006/geno.1997.4921.
Sumiyoshi H, Laub F, Yoshioka H, Ramirez F. Embryonic expression of type XIX collagen is transient and confined to muscle cells. Dev Dyn. 2001;220(2):155–62. https://doi.org/10.1002/1097-0177(2000)9999:9999%3c::AID-DVDY1099%3e3.0.CO;2-W.
Charvet B, et al. Knockdown of col22a1 gene in zebrafish induces a muscular dystrophy by disruption of the myotendinous junction. Development. 2013;140(22):4602–13. https://doi.org/10.1242/dev.096024.
Pisconti A, Bernet JD, Olwin BB. Syndecans in skeletal muscle development, regeneration and homeostasis. Muscles Ligaments Tendons J. 2012;2(1):1–9.
Decorin, a growth hormone-regulated protein in humans in: European Journal of Endocrinology. 2018;178(2). https://eje.bioscientifica.com/view/journals/eje/178/2/EJE-17-0844.xml. Accessed 06 Jun 2022.
Yamashita Y, et al. Perlecan, a heparan sulfate proteoglycan, regulates systemic metabolism with dynamic changes in adipose tissue and skeletal muscle. Sci Rep. 2018;8:7766. https://doi.org/10.1038/s41598-018-25635-x.
Nastase MV, Young MF, Schaefer L. Biglycan. J Histochem Cytochem. 2012;60(12):963–75. https://doi.org/10.1369/0022155412456380.
Werle MJ. Neuromuscular junction (NMJ): postsynaptic basal lamina. In Encyclopedia of Neuroscience, L. R. Squire, Ed., Oxford: Academic Press; 2009. pp 595–600. https://doi.org/10.1016/B978-008045046-9.01285-7.
Ren X, Zhao M, Lash B, Martino MM, Julier Z. Growth factor engineering strategies for regenerative medicine applications. Front Bioeng Biotechnol. 2020;7. [Online]. Available: https://www.frontiersin.org/article/https://doi.org/10.3389/fbioe.2019.00469. Accessed 23 Jun 2022.
Syverud BC, VanDusen KW, Larkin LM. Growth factors for skeletal muscle tissue engineering. Cells Tissues Organs. 2016;202(3–4):169–79. https://doi.org/10.1159/000444671.
Wilgus TA. Growth factor–extracellular matrix interactions regulate wound repair. Adv Wound Care. 2012;1(6):249–54. https://doi.org/10.1089/wound.2011.0344. (New Rochelle).
Liu D, Black BL, Derynck R. TGF-β inhibits muscle differentiation through functional repression of myogenic transcription factors by Smad3. Genes Dev. 2001;15(22):2950–66. https://doi.org/10.1101/gad.925901.
TGFB3 gene: MedlinePlus Genetics. https://medlineplus.gov/genetics/gene/tgfb3/. Accessed 05 Jun 2022.
Arsic N, et al. Vascular endothelial growth factor stimulates skeletal muscle regeneration in Vivo. Mol Ther. 2004;10(5):844–54. https://doi.org/10.1016/j.ymthe.2004.08.007.
Piñol-Jurado P, et al. Platelet-derived growth factor bb influences muscle regeneration in Duchenne muscle dystrophy. Am J Pathol. 2017;187(8):1814–27. https://doi.org/10.1016/j.ajpath.2017.04.011.
Barclay RD, Burd NA, Tyler C, Tillin NA, Mackenzie RW. The role of the IGF-1 signaling cascade in muscle protein synthesis and anabolic resistance in aging skeletal muscle. Front Nutr. 2019;6. [Online]. Available: https://www.frontiersin.org/article/https://doi.org/10.3389/fnut.2019.00146. Accessed 05 Jun 2022.
Schiaffino S, Mammucari C. Regulation of skeletal muscle growth by the IGF1-Akt/PKB pathway: insights from genetic models. Skelet Muscle. 2011;1(1):4. https://doi.org/10.1186/2044-5040-1-4.
D’Andrea P, Sciancalepore M, Veltruska K, Lorenzon P, Bandiera A. Epidermal growth factor – based adhesion substrates elicit myoblast scattering, proliferation, differentiation and promote satellite cell myogenic activation. Biochim Biophys Acta (BBA) – Mol Cell Res. 2019;1866(3):504–517. https://doi.org/10.1016/j.bbamcr.2018.10.012.
Jia W, et al. Effects of fasting on the expression pattern of FGFs in different skeletal muscle fibre types and sexes in mice. Biol Sex Differ. 2020;11(1):9. https://doi.org/10.1186/s13293-020-00287-7.
Choi W, Lee J, Lee J, Lee SH, Kim S. Hepatocyte growth factor regulates macrophage transition to the M2 phenotype and promotes murine skeletal muscle regeneration. Front Physiol. 2019;10. [Online]. Available: https://www.frontiersin.org/article/https://doi.org/10.3389/fphys.2019.00914. Accessed 05 Jun 2022.
Yamaguchi A, Sakuma K, Fujikawa T, Morita I. Expression of specific IGFBPs is associated with those of the proliferating and differentiating markers in regenerating rat plantaris muscle. J Physiol Sci. 2013;63(1). https://doi.org/10.1007/s12576-012-0227-6.
Pearse RV, Scherz PJ, Campbell JK, Tabin CJ. A cellular lineage analysis of the chick limb bud. Dev Biol. 2007;310(2):388–400. https://doi.org/10.1016/j.ydbio.2007.08.002.
Nowicki JL, Takimoto R, Burke AC. The lateral somitic frontier: dorso-ventral aspects of anterio-posterior regionalization in avian embryos. Mech Dev. 2003;120(2):227–40. https://doi.org/10.1016/s0925-4773(02)00415-x.
Olsson L, Falck P, Lopez K, Cobb J, Hanken J. Cranial neural crest cells contribute to connective tissue in cranial muscles in the anuran amphibian, Bombina orientalis. Dev Biol. 2001;237(2):354–67. https://doi.org/10.1006/dbio.2001.0377.
Dietrich S, et al. The role of SF/HGF and c-Met in the development of skeletal muscle. Development. 1999;126(8):1621–9. https://doi.org/10.1242/dev.126.8.1621.
Swartz ME, Eberhart J, Pasquale EB, Krull CE. EphA4/ephrin-A5 interactions in muscle precursor cell migration in the avian forelimb. Development. 2001;128(23):4669–80. https://doi.org/10.1242/dev.128.23.4669.
Kardon G, Harfe BD, Tabin CJ. A Tcf4-positive mesodermal population provides a prepattern for vertebrate limb muscle patterning. Dev Cell. 2003;5(6):937–44. https://doi.org/10.1016/s1534-5807(03)00360-5.
Hasson P, et al. Tbx4 and tbx5 acting in connective tissue are required for limb muscle and tendon patterning. Dev Cell. 2010;18(1):148–56. https://doi.org/10.1016/j.devcel.2009.11.013.
Iwata J, Suzuki A, Pelikan RC, Ho T-V, Chai Y. Noncanonical transforming growth factor β (TGFβ) signaling in cranial neural crest cells causes tongue muscle developmental defects. J Biol Chem. 2013;288(41):29760–70. https://doi.org/10.1074/jbc.M113.493551.
Vallecillo-García P, et al. Odd skipped-related 1 identifies a population of embryonic fibro-adipogenic progenitors regulating myogenesis during limb development. Nat Commun. 2017;8(1):1218. https://doi.org/10.1038/s41467-017-01120-3.
Musarò A. The basis of muscle regeneration. Advances in Biology. 2014;2014:e612471. https://doi.org/10.1155/2014/612471.
Almada AE, Wagers AJ. Molecular circuitry of stem cell fate in skeletal muscle regeneration, ageing and disease. Nat Rev Mol Cell Biol. 2016;17(5):267–79. https://doi.org/10.1038/nrm.2016.7.
Olguin HC, Olwin BB. Pax-7 up-regulation inhibits myogenesis and cell cycle progression in satellite cells: a potential mechanism for self-renewal. Dev Biol. 2004;275(2):375–88. https://doi.org/10.1016/j.ydbio.2004.08.015.
Wilschut KJ, Haagsman HP, Roelen BAJ. Extracellular matrix components direct porcine muscle stem cell behavior. Exp Cell Res. 2010;316(3):341–52. https://doi.org/10.1016/j.yexcr.2009.10.014.
Grefte S, Vullinghs S, Kuijpers-Jagtman AM, Torensma R, Von den Hoff JW. Matrigel, but not collagen I, maintains the differentiation capacity of muscle derived cells in vitro. Biomed Mater. 2012;7(5):055004. https://doi.org/10.1088/1748-6041/7/5/055004.
Gilbert PM, et al. Substrate elasticity regulates skeletal muscle stem cell self-renewal in culture. Science. 2010;329(5995):1078–81. https://doi.org/10.1126/science.1191035.
Cosgrove BD, et al. Rejuvenation of the muscle stem cell population restores strength to injured aged muscles. Nat Med. 2014;20(3):255–64. https://doi.org/10.1038/nm.3464.
Bentzinger CF, Wang YX, von Maltzahn J, Soleimani VD, Yin H, Rudnicki MA. Fibronectin regulates Wnt7a signaling and satellite cell expansion. Cell Stem Cell. 2013;12(1):75–87. https://doi.org/10.1016/j.stem.2012.09.015.
Urciuolo A, et al. Collagen VI regulates satellite cell self-renewal and muscle regeneration. Nat Commun. 2013;4:1964. https://doi.org/10.1038/ncomms2964.
Brack AS, Conboy IM, Conboy MJ, Shen J, Rando TA. A temporal switch from notch to Wnt signaling in muscle stem cells is necessary for normal adult myogenesis. Cell Stem Cell. 2008;2(1):50–9. https://doi.org/10.1016/j.stem.2007.10.006.
Zammit PS, Partridge TA, Yablonka-Reuveni Z. The skeletal muscle satellite cell: the stem cell that came in from the cold. J Histochem Cytochem. 2006;54(11):1177–91. https://doi.org/10.1369/jhc.6R6995.2006.
Boonen KJM, Rosaria-Chak KY, Baaijens FPT, van der Schaft DWJ, Post MJ. Essential environmental cues from the satellite cell niche: optimizing proliferation and differentiation. Am J Physiol Cell Physiol. 2009;296(6):C1338-1345. https://doi.org/10.1152/ajpcell.00015.2009.
Rønning SB, Pedersen ME, Andersen PV, Hollung K. The combination of glycosaminoglycans and fibrous proteins improves cell proliferation and early differentiation of bovine primary skeletal muscle cells. Differentiation. 2013;86(1–2):13–22. https://doi.org/10.1016/j.diff.2013.06.006.
Gutiérrez J, Brandan E. A novel mechanism of sequestering fibroblast growth factor 2 by glypican in lipid rafts, allowing skeletal muscle differentiation. Mol Cell Biol. 2010;30(7):1634–49. https://doi.org/10.1128/MCB.01164-09.
Rayagiri SS, et al. Basal lamina remodeling at the skeletal muscle stem cell niche mediates stem cell self-renewal. Nat Commun. 2018;9(1):1075. https://doi.org/10.1038/s41467-018-03425-3.
Baghdadi MB, et al. Reciprocal signalling by Notch-Collagen V-CALCR retains muscle stem cells in their niche. Nature. 2018;557(7707):714–8. https://doi.org/10.1038/s41586-018-0144-9.
Chargé SBP, Rudnicki MA. Cellular and molecular regulation of muscle regeneration. Physiol Rev. 2004;84(1):209–38. https://doi.org/10.1152/physrev.00019.2003.
Tidball JG. Inflammatory cell response to acute muscle injury. Med Sci Sports Exerc. 1995;27(7):1022–32. https://doi.org/10.1249/00005768-199507000-00011.
Butterfield TA, Best TM, Merrick MA. The dual roles of neutrophils and macrophages in inflammation: a critical balance between tissue damage and repair. J Athl Train. 2006;41(4):457–65.
Oishi Y, Manabe I. Macrophages in inflammation, repair and regeneration. Int Immunol. 2018;30(11):511–28. https://doi.org/10.1093/intimm/dxy054.
Mauro A. Satellite cell of skeletal muscle fibers. J Biophys Biochem Cytol. 1961;9:493–5. https://doi.org/10.1083/jcb.9.2.493.
Skuk D, Goulet M, Tremblay JP. Transplanted myoblasts can migrate several millimeters to fuse with damaged myofibers in nonhuman primate skeletal muscle. J Neuropathol Exp Neurol. 2011;70(9):770–8. https://doi.org/10.1097/NEN.0b013e31822a6baa.
Hawke TJ, Garry DJ. Myogenic satellite cells: physiology to molecular biology. J Appl Physiol (1985). 2001;91(2):534–51. https://doi.org/10.1152/jappl.2001.91.2.534.
Kääriäinen M, Järvinen T, Järvinen M, Rantanen J, Kalimo H. Relation between myofibers and connective tissue during muscle injury repair. Scand J Med Sci Sports. 2000;10(6):332–7. https://doi.org/10.1034/j.1600-0838.2000.010006332.x.
Mann CJ, et al. Aberrant repair and fibrosis development in skeletal muscle. Skelet Muscle. 2011;1(1):21. https://doi.org/10.1186/2044-5040-1-21.
Ambrosi D, et al. Growth and remodelling of living tissues: perspectives, challenges and opportunities. J R Soc Interface. 2019;16(157):20190233. https://doi.org/10.1098/rsif.2019.0233.
Bryan BA, et al. Coordinated vascular endothelial growth factor expression and signaling during skeletal myogenic differentiation. MBoC. 2008;19(3):994–1006. https://doi.org/10.1091/mbc.e07-09-0856.
Mofarrahi M, et al. Angiopoietin-1 enhances skeletal muscle regeneration in mice. Am J Physiol Regul Integr Comp Physiol. 2015;308(7):R576-589. https://doi.org/10.1152/ajpregu.00267.2014.
Christov C, et al. Muscle satellite cells and endothelial cells: close neighbors and privileged partners. Mol Biol Cell. 2007;18(4):1397–409. https://doi.org/10.1091/mbc.e06-08-0693.
Tatsumi R, Anderson JE, Nevoret CJ, Halevy O, Allen RE. HGF/SF is present in normal adult skeletal muscle and is capable of activating satellite cells. Dev Biol. 1998;194(1):114–28. https://doi.org/10.1006/dbio.1997.8803.
Forcina L, Miano C, Pelosi L, Musarò A. An overview about the biology of skeletal muscle satellite cells. Curr Genomics. 2019;20(1):24–37. https://doi.org/10.2174/1389202920666190116094736.
Bahram M, Mohseni N, Moghtader M. An introduction to hydrogels and some recent applications. IntechOpen. 2016. https://doi.org/10.5772/64301.
Mantha S, et al. Smart hydrogels in tissue engineering and regenerative medicine. Materials. 2019;12(20):3323. https://doi.org/10.3390/ma12203323. (Basel).
Pollot BE, Rathbone CR, Wenke JC, Guda T. Natural polymeric hydrogel evaluation for skeletal muscle tissue engineering. J Biomed Mater Res B Appl Biomater. 2018;106(2):672–9. https://doi.org/10.1002/jbm.b.33859.
Saroia J, Yanen W, Wei Q, Zhang K, Lu T, Zhang B. A review on biocompatibility nature of hydrogels with 3D printing techniques, tissue engineering application and its future prospective. Bio-Des Manuf. 2018;1(4):265–79. https://doi.org/10.1007/s42242-018-0029-7.
Lin K, Zhang D, Macedo MH, Cui W, Sarmento B, Shen G. Advanced collagen-based biomaterials for regenerative biomedicine. Adv Func Mater. 2019;29(3):1804943. https://doi.org/10.1002/adfm.201804943.
Kim JK, Kim HJ, Chung J-Y, Lee J-H, Young S-B, Kim Y-H. Natural and synthetic biomaterials for controlled drug delivery. Arch Pharm Res. 2014;37(1):60–8. https://doi.org/10.1007/s12272-013-0280-6.
Dietrich M, et al. Fibrin-based tissue engineering: comparison of different methods of autologous fibrinogen isolation. Tissue Eng Part C Methods. 2013;19(3):216–26. https://doi.org/10.1089/ten.TEC.2011.0473.
Catoira MC, Fusaro L, Di Francesco D, Ramella M, Boccafoschi F. Overview of natural hydrogels for regenerative medicine applications. J Mater Sci: Mater Med. 2019;30(10):115. https://doi.org/10.1007/s10856-019-6318-7.
Rastogi P, Kandasubramanian B. Review of alginate-based hydrogel bioprinting for application in tissue engineering. Biofabrication. 2019;11(4):042001. https://doi.org/10.1088/1758-5090/ab331e.
Li H, Tan C, Li L. Review of 3D printable hydrogels and constructs. Mater Des. 2018;159:20–38. https://doi.org/10.1016/j.matdes.2018.08.023.
Bitas D, Samanidou V, Kabir A, Lucena R, Cárdenas S. 9 - Membrane sorptive phases. In Analytical Sample Preparation With Nano- and Other High-Performance Materials, R. Lucena and S. Cárdenas, Eds., Elsevier; 2021. pp. 199–228. https://doi.org/10.1016/B978-0-12-822139-6.00015-8.
Roberts JJ, Martens PJ. 9 - Engineering biosynthetic cell encapsulation systems. In Biosynthetic Polymers for Medical Applications, L. Poole-Warren, P. Martens, and R. Green, Eds., in Woodhead Publishing Series in Biomaterials. Woodhead Publishing; 2016. pp. 205–239. https://doi.org/10.1016/B978-1-78242-105-4.00009-2.
Stringer R. ELECTROPHORESIS | Overview. In Encyclopedia of Analytical Science (Second Edition), P. Worsfold, A. Townshend, and C. Poole, Eds., Oxford: Elsevier; 2005. pp. 356–363. https://doi.org/10.1016/B0-12-369397-7/00120-5.
Matthias N, et al. Volumetric muscle loss injury repair using in situ fibrin gel cast seeded with muscle-derived stem cells (MDSCs). Stem Cell Res. 2018;27:65–73. https://doi.org/10.1016/j.scr.2018.01.008.
Thorrez L, DiSano K, Shansky J, Vandenburgh H. Engineering of human skeletal muscle with an autologous deposited extracellular matrix. Front Physiol. 2018;9. [Online]. Available: https://www.frontiersin.org/article/https://doi.org/10.3389/fphys.2018.01076. Accessed 24 Jun 2022.
Marcinczyk M, Elmashhady H, Talovic M, Dunn A, Bugis F, Garg K. Laminin-111 enriched fibrin hydrogels for skeletal muscle regeneration. Biomaterials. 2017;141:233–42. https://doi.org/10.1016/j.biomaterials.2017.07.003.
Cappello V, et al. Ultrastructural characterization of the lower motor system in a mouse model of Krabbe disease. Sci Rep. 2016;6(1):1. https://doi.org/10.1038/s41598-016-0001-8.
Jaipan P, Nguyen A, Narayan RJ. Gelatin-based hydrogels for biomedical applications. MRS Commun. 2017;7(3):416–26. https://doi.org/10.1557/mrc.2017.92.
Gyles DA, Castro LD, Silva JOC, Ribeiro-Costa RM. A review of the designs and prominent biomedical advances of natural and synthetic hydrogel formulations. Eur Polymer J. 2017;88:373–92. https://doi.org/10.1016/j.eurpolymj.2017.01.027.
Akhtar MF, Hanif M, Ranjha NM. Methods of synthesis of hydrogels … a review. Saudi Pharmac J. 2016;24(5):554–9. https://doi.org/10.1016/j.jsps.2015.03.022.
Yang G, et al. Enzymatically crosslinked gelatin hydrogel promotes the proliferation of adipose tissue-derived stromal cells. PeerJ. 2016;4:e2497. https://doi.org/10.7717/peerj.2497.
Kirchmajer DM, Watson CA, Ranson M, Panhuis M. Gelapin, a degradable genipin cross-linked gelatin hydrogel. RSC Adv. 2012;3(4):1073–81. https://doi.org/10.1039/C2RA22859A.
Gupta D, Santoso JW, McCain ML. Characterization of gelatin hydrogels cross-linked with microbial transglutaminase as engineered skeletal muscle substrates. Bioengineering. 2021;8(1):6. https://doi.org/10.3390/bioengineering8010006. (Basel).
Powell CA, Smiley BL, Mills J, Vandenburgh HH. Mechanical stimulation improves tissue-engineered human skeletal muscle. Am J Physiol Cell Physiol. 2002;283(5):C1557–65. https://doi.org/10.1152/ajpcell.00595.2001.
Chiron S, et al. Complex interactions between human myoblasts and the surrounding 3D fibrin-based matrix. PLoS ONE. 2012;7(4):e36173. https://doi.org/10.1371/journal.pone.0036173.
Mahdy MAA. Skeletal muscle fibrosis: an overview. Cell Tissue Res. 2019;375(3):575–88. https://doi.org/10.1007/s00441-018-2955-2.
Brown AC, Barker TH. Fibrin-based biomaterials: modulation of macroscopic properties through rational design at the molecular level. Acta Biomater. 2014;10(4):1502–14. https://doi.org/10.1016/j.actbio.2013.09.008.
Volpi M, Paradiso A, Costantini M, Świȩszkowski W. Hydrogel-based fiber biofabrication techniques for skeletal muscle tissue engineering. ACS Biomater Sci Eng. 2022;8(2):379–405. https://doi.org/10.1021/acsbiomaterials.1c01145.
Solorio L, Zwolinski C, Lund AW, Farrell MJ, Stegemann JP. Gelatin microspheres crosslinked with genipin for local delivery of growth factors. J Tissue Eng Regen Med. 2010;4(7):514–23. https://doi.org/10.1002/term.267.
Zhu J, Marchant RE. Design properties of hydrogel tissue-engineering scaffolds. Expert Rev Med Devices. 2011;8(5):607–26. https://doi.org/10.1586/erd.11.27.
Jia J, et al. Development of peptide-functionalized synthetic hydrogel microarrays for stem cell and tissue engineering applications. Acta Biomater. 2016;45:110–20. https://doi.org/10.1016/j.actbio.2016.09.006.
Oh SH, Lee JH. Hydrophilization of synthetic biodegradable polymer scaffolds for improved cell/tissue compatibility. Biomed Mater. 2013;8(1):014101. https://doi.org/10.1088/1748-6041/8/1/014101.
Villa C, et al. P(NIPAAM-co-HEMA) thermoresponsive hydrogels: an alternative approach for muscle cell sheet engineering. J Tissue Eng Regen Med. 2017;11(1):187–96. https://doi.org/10.1002/term.1898.
Browe DP, et al. Characterization and optimization of actuating poly(ethylene glycol) diacrylate/acrylic acid hydrogels as artificial muscles. Polymer. 2017;117:331–41. https://doi.org/10.1016/j.polymer.2017.04.044. (Guildf).
Cha SH, Lee HJ, Koh W-G. Study of myoblast differentiation using multi-dimensional scaffolds consisting of nano and micropatterns. Biomater Res. 2017;21(1):1. https://doi.org/10.1186/s40824-016-0087-x.
Vannozzi L, Yasa IC, Ceylan H, Menciassi A, Ricotti L, Sitti M. Self-folded hydrogel tubes for implantable muscular tissue scaffolds. Macromol Biosci. 2018;18(4):e1700377. https://doi.org/10.1002/mabi.201700377.
Xu Y, et al. Regulating myogenic differentiation of mesenchymal stem cells using thermosensitive hydrogels. Acta Biomater. 2015;26:23–33. https://doi.org/10.1016/j.actbio.2015.08.010.
Hosseinzadeh S, Rezayat SM, Giaseddin A, Aliyan A, Soleimani M. Microfluidic system for synthesis of nanofibrous conductive hydrogel and muscle differentiation. J Biomater Appl. 2018;32(7):853–61. https://doi.org/10.1177/0885328217744377.
McKeon-Fischer KD, Flagg DH, Freeman JW. Coaxial electrospun poly(ε-caprolactone), multiwalled carbon nanotubes, and polyacrylic acid/polyvinyl alcohol scaffold for skeletal muscle tissue engineering. J Biomed Mater Res A. 2011;99(3):493–9. https://doi.org/10.1002/jbm.a.33116.
Gaharwar AK, Peppas NA, Khademhosseini A. Nanocomposite hydrogels for biomedical applications. Biotechnol Bioeng. 2014;111(3):441–53. https://doi.org/10.1002/bit.25160.
Hwang JH, et al. Combination therapy of human adipose-derived stem cells and basic fibroblast growth factor hydrogel in muscle regeneration. Biomaterials. 2013;34(25):6037–45. https://doi.org/10.1016/j.biomaterials.2013.04.049.
Mulyasasmita W, et al. Avidity-controlled hydrogels for injectable co-delivery of induced pluripotent stem cell-derived endothelial cells and growth factors. J Control Release. 2014;191:71–81. https://doi.org/10.1016/j.jconrel.2014.05.015.
Fuoco C, et al. 3D hydrogel environment rejuvenates aged pericytes for skeletal muscle tissue engineering. Front Physiol. 2014;5:203. https://doi.org/10.3389/fphys.2014.00203.
In vivo generation of a mature and functional artificial skeletal muscle. EMBO Mol Med. 2015;7(4):411–422. https://doi.org/10.15252/emmm.201404062.
Rich MH, et al. Water–hydrogel binding affinity modulates freeze-drying-induced micropore architecture and skeletal myotube formation. Biomacromol. 2015;16(8):2255–64. https://doi.org/10.1021/acs.biomac.5b00652.
Urciuolo A, De Coppi P. Decellularized tissue for muscle regeneration. Int J Mol Sci. 2018;19(8):2392. https://doi.org/10.3390/ijms19082392.
Fu R-H, et al. Decellularization and recellularization technologies in tissue engineering. Cell Transplant. 2014;23(4–5):621–30. https://doi.org/10.3727/096368914X678382.
Freytes DO, Martin J, Velankar SS, Lee AS, Badylak SF. Preparation and rheological characterization of a gel form of the porcine urinary bladder matrix. Biomaterials. 2008;29(11):1630–7. https://doi.org/10.1016/j.biomaterials.2007.12.014.
Zhang W, Du A, Liu S, Lv M, Chen S. Research progress in decellularized extracellular matrix-derived hydrogels. Regen Ther. 2021;18:88–96. https://doi.org/10.1016/j.reth.2021.04.002.
Fu Y, et al. Decellularization of porcine skeletal muscle extracellular matrix for the formulation of a matrix hydrogel: a preliminary study. J Cell Mol Med. 2016;20(4):740–9. https://doi.org/10.1111/jcmm.12776.
Fernández-Pérez J, Ahearne M. The impact of decellularization methods on extracellular matrix derived hydrogels. Sci Rep. 2019;9(1). https://doi.org/10.1038/s41598-019-49575-2.
Sackett SD, et al. Extracellular matrix scaffold and hydrogel derived from decellularized and delipidized human pancreas. Sci Rep. 2018;8(1):10452. https://doi.org/10.1038/s41598-018-28857-1.
Pouliot RA, et al. Development and characterization of a naturally derived lung extracellular matrix hydrogel. J Biomed Mater Res A. 2016;104(8):1922–35. https://doi.org/10.1002/jbm.a.35726.
Wolf MT, et al. A hydrogel derived from decellularized dermal extracellular matrix. Biomaterials. 2012;33(29):7028–38. https://doi.org/10.1016/j.biomaterials.2012.06.051.
Liguori GR, et al. Abstract 14119: Decellularized arterial extracellular matrix-based hydrogel supports 3D bioprinting of the media layer of small-caliber blood vessels. Circulation. 2019;140(Suppl_1):A14119–A14119. https://doi.org/10.1161/circ.140.suppl_1.14119.
Stern MM, et al. The influence of extracellular matrix derived from skeletal muscle tissue on the proliferation and differentiation of myogenic progenitor cells ex vivo. Biomaterials. 2009;30(12):2393–9. https://doi.org/10.1016/j.biomaterials.2008.12.069.
DeQuach JA, et al. Simple and high yielding method for preparing tissue specific extracellular matrix coatings for cell culture. PLoS ONE. 2010;5(9):e13039. https://doi.org/10.1371/journal.pone.0013039.
Ungerleider JL, Dzieciatkowska M, Hansen KC, Christman KL. Tissue specific muscle extracellular matrix hydrogel improves skeletal muscle regeneration in vivo over non-matched tissue source. 2020. https://doi.org/10.1101/2020.06.30.181164.
Zhang Y, et al. Tissue-specific extracellular matrix coatings for the promotion of cell proliferation and maintenance of cell phenotype. Biomaterials. 2009;30(23–24):4021–8. https://doi.org/10.1016/j.biomaterials.2009.04.005.
Choi Y-J, et al. A 3D cell printed muscle construct with tissue-derived bioink for the treatment of volumetric muscle loss. Biomaterials. 2019;206:160–9. https://doi.org/10.1016/j.biomaterials.2019.03.036.
Nguyen MM, Gianneschi NC, Christman KL. Developing injectable nanomaterials to repair the heart. Curr Opin Biotechnol. 2015;34:225–31. https://doi.org/10.1016/j.copbio.2015.03.016.
Hernandez MJ, Christman KL. Designing acellular injectable biomaterial therapeutics for treating myocardial infarction and peripheral artery disease. JACC Basic Transl Sci. 2017;2(2):212–26. https://doi.org/10.1016/j.jacbts.2016.11.008.
Gaetani R, Ungerleider J, Christman KL. Chapter 25 - Acellular injectable biomaterials for treating cardiovascular disease. In Stem Cell and Gene Therapy for Cardiovascular Disease, E. C. Perin, L. W. Miller, D. A. Taylor, and J. T. Willerson, Eds., Boston: Academic Press; 2016. pp. 309–325. https://doi.org/10.1016/B978-0-12-801888-0.00025-4.
DeQuach JA, et al. Injectable skeletal muscle matrix hydrogel promotes neovascularization and muscle cell infiltration in a hindlimb ischemia model. Eur Cell Mater. 2012;23:400–412; discussion 412. https://doi.org/10.22203/ecm.v023a31.
Ungerleider JL, et al. Extracellular matrix hydrogel promotes tissue remodeling, arteriogenesis, and perfusion in a rat hindlimb ischemia model. JACC: Basic Transl Sci. 2016;1(1):32–44. https://doi.org/10.1016/j.jacbts.2016.01.009.
Rao N, et al. Engineering an injectable muscle-specific microenvironment for improved cell delivery using a nanofibrous extracellular matrix hydrogel. ACS Nano. 2017;11(4):3851–9. https://doi.org/10.1021/acsnano.7b00093.
Quarta M, et al. Bioengineered constructs combined with exercise enhance stem cell-mediated treatment of volumetric muscle loss. Nat Commun. 2017;8:15613. https://doi.org/10.1038/ncomms15613.
Jana S, Levengood SKL, Zhang M. Anisotropic materials for skeletal-muscle-tissue engineering. Adv Mater. 2016;28(48):10588–612. https://doi.org/10.1002/adma.201600240.
Nakayama KH, et al. Rehabilitative exercise and spatially patterned nanofibrillar scaffolds enhance vascularization and innervation following volumetric muscle loss. npj Regen Med. 2018;3(1). https://doi.org/10.1038/s41536-018-0054-3.
Nakayama KH, et al. Treatment of volumetric muscle loss in mice using nanofibrillar scaffolds enhances vascular organization and integration. Commun Biol. 2019;2(1). https://doi.org/10.1038/s42003-019-0416-4.
Ostrovidov S, et al. Skeletal muscle tissue engineering: methods to form skeletal myotubes and their applications. Tissue Eng Part B Rev. 2014;20(5):403–36. https://doi.org/10.1089/ten.TEB.2013.0534.
Ostrovidov S, et al. Three dimensional bioprinting in skeletal muscle tissue engineering. Small. 2019;15(24):e1805530. https://doi.org/10.1002/smll.201805530.
Choi Y-J, et al. 3D cell printing of functional skeletal muscle constructs using skeletal muscle-derived bioink. Adv Healthc Mater. 2016;5(20):2636–45. https://doi.org/10.1002/adhm.201600483.
Traverse JH, et al. First-in-man study of a cardiac extracellular matrix hydrogel in early and late myocardial infarction patients. JACC Basic Transl Sci. 2019;4(6):659–69. https://doi.org/10.1016/j.jacbts.2019.07.012.
Urciuolo A, et al. Decellularised skeletal muscles allow functional muscle regeneration by promoting host cell migration. Sci Rep. 2018;8(1):8398. https://doi.org/10.1038/s41598-018-26371-y.
Crapo PM, Gilbert TW, Badylak SF. An overview of tissue and whole organ decellularization processes. Biomaterials. 2011;32(12):3233–43. https://doi.org/10.1016/j.biomaterials.2011.01.057.
Bordbar S, LotfiBakhshaiesh N, Khanmohammadi M, Sayahpour FA, Alini M, BaghabanEslaminejad M. Production and evaluation of decellularized extracellular matrix hydrogel for cartilage regeneration derived from knee cartilage. J Biomed Mater Res A. 2020;108(4):938–46. https://doi.org/10.1002/jbm.a.36871.
Wong ML, Griffiths LG. Immunogenicity in xenogeneic scaffold generation: antigen removal vs. decellularization. Acta Biomater. 2014;10(5):1806–16. https://doi.org/10.1016/j.actbio.2014.01.028.
Galili U, Shohet SB, Kobrin E, Stults CL, Macher BA. Man, apes, and Old World monkeys differ from other mammals in the expression of alpha-galactosyl epitopes on nucleated cells. J Biol Chem. 1988;263(33):17755–62. https://doi.org/10.1016/S0021-9258(19)77900-9.
Sandrin MS, McKenzie IF. Gal alpha (1,3)Gal, the major xenoantigen(s) recognised in pigs by human natural antibodies. Immunol Rev. 1994;141:169–90. https://doi.org/10.1111/j.1600-065x.1994.tb00877.x.
Joziasse DH, Oriol R. Xenotransplantation: the importance of the Galα1,3Gal epitope in hyperacute vascular rejection. Biochim Biophys Acta (BBA) - Mol Basis Dis. 1999;1455(2):403–418. https://doi.org/10.1016/S0925-4439(99)00056-3.
Badylak SF, Gilbert TW. Immune response to biologic scaffold materials. Semin Immunol. 2008;20(2):109–16. https://doi.org/10.1016/j.smim.2007.11.003.
Galili U. Acceleration of wound healing by -gal nanoparticles interacting with the natural anti-gal antibody. J Immunol Res. 2015;2015:e589648. https://doi.org/10.1155/2015/589648.
Lu Y, et al. A standardized quantitative method for detecting remnant alpha-Gal antigen in animal tissues or animal tissue-derived biomaterials and its application. Sci Rep. 2018;8(1). https://doi.org/10.1038/s41598-018-32959-1.
Gao H-W, et al. Quantification of α-Gal Antigen Removal in the Porcine Dermal Tissue by α-Galactosidase. Tissue Eng Part C Methods. 2015;21(11):1197–204. https://doi.org/10.1089/ten.TEC.2015.0129.
Wang RM, et al. Humanized mouse model for assessing the human immune response to xenogeneic and allogeneic decellularized biomaterials. Biomaterials. 2017;129:98–110. https://doi.org/10.1016/j.biomaterials.2017.03.016.
Stahl E, et al. Evaluation of the host immune response to decellularized lung scaffolds derived from α-Gal knockout pigs in a non-human primate model. Biomaterials. 2018;87. https://doi.org/10.1016/j.biomaterials.2018.09.038.
Tao M, et al. Sterilization and disinfection methods for decellularized matrix materials: Review, consideration and proposal. Bioact Mater. 2021;6(9):2927–45. https://doi.org/10.1016/j.bioactmat.2021.02.010.
Smoak MM, Mikos AG. Advances in biomaterials for skeletal muscle engineering and obstacles still to overcome. Materials Today Bio. 2020;7:100069.
Anthony A, Robert L, Tony M, Robert N. Principles of Regenerative Medicine. 3rd Edition. 2019. Elsevier. https://doi.org/10.1016/C2015-0-02433-9
Zhuang P, An J, Chua CK, Tan LP. Bioprinting of 3D in vitro skeletal muscle models: A review. Mater Des. 2020;193:108794.
Lu Y, et al. A standardized quantitative method for detecting remnant alpha-Gal antigen in animal tissues or animal tissue-derived biomaterials and its application. Sci Rep. 2018;8:15424.
Philips C, Terrie L, Thorrez L. Decellularized skeletal muscle: a versatile biomaterial in tissue engineering and regenerative medicine. Biomaterials. 2022;283:121436.
Funding
This work was supported by funding from NIH T32 AR079114/AR/NIAMS and 1332329/EFRI/NSF. Mohammed A. Barajaa was funded by Imam Abdulrahman Bin Faisal University, Dammam, 34212, Saudi Arabia.
Author information
Authors and Affiliations
Contributions
The manuscript was conceptualized and written through the contributions of all authors. All authors have approved the final version of the manuscript.
Corresponding author
Ethics declarations
Competing Interests
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Barajaa, M.A., Ghosh, D. & Laurencin, C.T. Decellularized Extracellular Matrix-Derived Hydrogels: a Powerful Class of Biomaterials for Skeletal Muscle Regenerative Engineering Applications. Regen. Eng. Transl. Med. (2023). https://doi.org/10.1007/s40883-023-00328-8
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s40883-023-00328-8