Abstract
The application of nanotechnology is significantly revolutionizing the domain of fisheries. Nanotechnology tools are applied to tackle many challenges pertaining to fish productivity, health, reproduction, prevention and treatment of diseases. Fish growth performance can be improved by adding essential minerals in the form of nano-feed supplements. Moreover, nanotechnology is currently assuming a pivotal position in the domain of fish reproduction, alongside its application in fish medicine, including antibacterial therapies, medication delivery mechanisms, and nano-vaccination. Also, there are significant evidences supporting the use of nanotechnology techniques for fish packing and water purification and remediation. In contrast, numerous nanoparticles possess harmful characteristics towards living organisms as a result of their tiny sizes, potent reactivity, and capacity to cross boundaries. They have the ability to modify several physiological activities and induce cytotoxicity, DNA damage, and histopathological alterations. Although nanotechnology has potential for enhancing growth performance and disease resistance in fish, there is ongoing debate about the potential toxicity of nanomaterials, their interactions with the environment, and their propensity to accumulate in animals. This review aims to clarify and analyze the different benefits and challenges associated with the application of nanotechnology in fish farming.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
The increasing world population and rapid economic growth have stressed up for the requirement for protein. Aquatic protein products are highly valued for their beneficial health impacts and significant food compositional features (Shah and Mraz 2020a). The consensus among nearly all nations globally is that fish is an essential component of a human diet (Mohanty 2015). In order to maintain livelihoods, guarantee nutrition, and improve food security, fish are essential. They provide vital fatty acids, amino acids, vitamins, and important nutrients like iodine and selenium that are frequently deficient in other foods or meats. They also function as a rich source of high-quality protein (Kwasek et al. 2020). Fish made up over 17% of all animal protein consumed worldwide in 2015; this percentage has been rising continuously (Action 2020). In fact, traditional fishing techniques in freshwater or marine habitats are unable to fulfil the growing demands of the increasing population. Fish farming must thus be given top priority, and deliberate measures must be made to achieve fish output self-sufficiency. The main elements essential to guaranteeing aquaculture's long-term survival and achieving a significant fish yield are optimal feeding, efficient disease control, and careful water quality management (Assefa and Abunna 2018; Khan et al. 2020).
Nanotechnology has become vital in its significance due to its explosive growth and development in a number of industries, including food, agriculture, medical, and environmental science (Subramani et al. 2019; He et al. 2019). International markets have over 300 nano-food items throughout the last decade (Narsale et al. 2024; Ramsden 2018). Nano-materials possess distinct physicochemical features due to their small size, enabling them to endure high pressure and temperature (Ilangovan et al. 2021). Nanotechnology is also being used in order to increase development and mitigate the problems regarding fish farming (Khosravi-Katuli et al. 2017; Luis et al. 2019).
Nanotechnology has both direct and indirect uses in fish aquaculture. Direct use of nanotechnology is increasing to improve fish growth, reproduction, and health (Sarkar et al. 2022). For example, the use of nanomaterials can make feed supplementation easier, which usually entails adding vitamins, minerals, and trace elements to animal diets (Ashouri et al. 2015; Khan et al. 2017; Xia et al. 2019). Fish that consume component nano-forms have easier absorption and easier passage across the intestinal barrier, which improves fish immune system, development, and reproduction (Bhattacharyya et al. 2015; Chris et al. 2018). In a study, iron nanoparticles (NPs) were found to enhance the growth rate of Carassius auratus and Acipenser gueldenstaedtii by 30% and 24% respectively (Srinivasan et al. 2016a). Nano selenium (Se) source supplemented diets improved the growth, antioxidant status and muscle Se concentration of Carassius auratus (Zhou et al. 2009). A study has demonstrated that the inclusion of nano Zinc in the food of Pangasius hypophthalmus resulted in enhanced survival and growth (Kumar et al. 2018). The effective management of viruses, bacteria, and fungus necessitates the timely identification and eradication of pathogens. Nanomaterials, which function on a comparable magnitude to particle-infecting viruses or diseases, have been identified as a potential solution for this purpose (Ismael et al. 2021; Nasr-Eldahan et al. 2021). The uses of nanomaterials in fish health involve the use of porous nanostructures and nanosensors to create antibacterial or antifungal surfaces. These surfaces are used in aquaculture systems to detect pathogens in water and to deliver fish medicines through fish meals (Abbas 2021; Nasr-Eldahan et al. 2021). Additionally, the utilization of chitosan-based wrapping as a nano encapsulation carrier for the purpose of effectively treating fish diseases caused by bacteria and viruses has been well-documented. This approach is advantageous due to the durability of nano encapsulated materials, which can withstand high temperatures and acidic environments (Abimbola et al. 2023). Moreover, nanomaterials are more effective than conventional antibiotics in treating fish diseases. Traditional therapies may cause bacteria to develop antibiotic resistance, which would eventually reduce its effectiveness. These treatments may also leave behind toxic chemical residues and contaminate water supplies, creating threats to the environment. Chitosan and polylactic-glycolic acid (PLGA) nanoparticles have a wide range of applications, including vaccines, medication delivery and hormone administration (Bhat et al. 2019a, b; Fenaroli et al. 2014; Mohd Ashraf Rather et al. 2013). Nano-vaccination is superior to conventional methods due to its capacity to provide sustained release, enhance stability, improve absorption, and prolong residence duration (Kitiyodom et al. 2019; Rajesh Kumar et al. 2008; Rivas-Aravena et al. 2015).
The indirect uses of nanotechnology in aquaculture generally focus on improving water quality. This includes sterilizing ponds, reducing the need for water exchange, decreasing the concentration of nitrogenous compounds, and treating chemical or biological pollutants in the water (Huang et al. 2015; Khosravi-Katuli et al. 2017; Tayel et al. 2019). Furthermore, the utilization of nanotechnology in water purification serves as an additional benefits by effectively inhibiting the advancement of infectious disorders caused by diverse microorganisms (Tayel et al. 2019; Yu et al. 2002). Additionally, the anti-fouling property of certain nanomaterials is a suitable application of nanotechnology, to attain high water quality in fish ponds, by lowering phosphate content and reducing the development of algae and other microbes (Ashraf and Edwin 2016).
Although nanotechnology has several benefits in fish farming. There are concerns for its safety for fish health, human health, and the environment. The existence of nanoparticles, specifically those made of metals, may harm a number of fish physiological functions. Reproductive hormone levels may be disrupted antioxidant and enzyme activity can change, the survival of embryonic developmental stages may be impacted, and histopathological changes are possible (Aruoja et al. 2009; Jovanović et al. 2015; Klingelfus et al. 2019; Kumar et al. 2017; Rajkumar et al. 2016; Ren et al. 2018; Sumi and Chitra 2019; Zhu et al. 2012). There are still a lot of outstanding concerns about the possible toxicity of using nanotechnology in aquaculture, causing a need for further study in this particular field. Further research is required to determine the minimum effective dosage, duration and specially lethal and sublethal concentrations and also need to observe bioaccumulation, residue concentration, and environmental destiny. To protect the environment, laws regulating the creation and useful uses of nanomaterials should be implemented. The purpose of this study is to investigate the different uses of nanotechnology in fish production, both directly and indirectly, and to look at the potential benefits and difficulties associated with such applications.
Direct applications of nanotechnology in fish culture
Feed supplements
Fish in their natural environment feed on phytoplankton, zooplankton, and other tiny creatures. Fish cultured in fish farming need extra nutrients to ensure rapid and healthy growth. Fish meal should have protein (32%), carbohydrate (20–35%), fat (4–6%), fiber (< 4%), and dietary energy (8.5–9.5%). These main requirements differ by fish species and age (Abbas 2021). Fish feed supplements with minute organic and inorganic concentrations boost growth and immunity. Nano-materials are rapidly absorbed in low dosages and pass through the gastro-intestinal system and small intestine to enter the circulatory system, facilitating more efficient distribution to vital organs compared to bulk materials (Bhuvaneshwari et al. 2015). Selenium (Se), Zinc(Zn), and iron (Fe) nano-metals as feed additives may improve fish survival, growth, and health (Chris et al. 2018). Chitosan also delivers micronutrients (Khosravi-Katuli et al. 2017). Nano-Se high bioavailability, strong reactivity, low toxicity, antioxidant defense, immuno-modulatory, and growth promoter effects have made it prevalent in aquaculture (Sonkusre et al. 2014; Xia et al. 2019). Studies has revealed that as compared to fish fed a basal diet, fish supplemented with nano-selenium (nano-Se) up to 0.68 mg n-Se/kg dry feed exhibited significantly higher weight gain, feed conversion efficiency, and specific growth rate specifically in masheer fish (Tor pulitora). It is important that fish provided the supplementary diet showed a significantly lower feed conversion ratio than fish fed the basal diet. Khan et al. (2017) conducted a 90-day feeding experiment to investigate the effects of zinc oxide (ZnO), zinc sulphate (ZnSO4), and zinc oxide nanoparticles (ZnO-NP) on the growth and haematological indicators of juvenile grass carp (Ctenopharyngodon idella). Fish given the ZnO-NP diet (30 mg Kg−1) had significantly greater weight increases, specific growth rate, and feed conversion ratio. Faiz et al. (2015) conducted a research and found that giving grass carp (C. idella) nanoparticles as a food supplement improved their growth and red blood cell count. Supplementing the meal of Cirrhuinus mrigala with ZnO nanoparticles enhanced metabolism, although resulting in a decrease in the count of white blood cells (Rajan et al. 2021).
Supplementing fish diets with iron is critical as most natural iron sources have limited solubility and bioavailability, making it difficult to meet dietary needs (Hilty et al. 2011). Iron improves the immune system and oxygen transport, respiration, and fat oxidation. Due to its reduced solubility and bioavailability, bulk iron (Fe) sources cannot provide dietary iron (Fe) needs (V. Srinivasan et al. 2016b).The addition of iron nanoparticles, specifically iron oxide or iron nanoparticles (NPs), at a dosage of 63.75 mg/kg, has been observed to have prominent advantageous impacts on (Oreochromis niloticus) when included in their supplementary meal. The effects include enhanced growth, immune response, phagocytosis activity against foreign particles such as bacteria, decreased mortality rate, elevated protein and lipid content, heightened muscle concentration, improved red and white blood cell counts, increased antioxidant capacity, and fortified disease resistance (El-Shenawy et al. 2019). Other studies on Nile Tilapia (O. niloticus) indicated that chitosan had positive effects on fish development. The study initiate that supplementing Nile tilapia feed with chitosan at a dosage of 0–8 g/kg dry food for 56 days resulted in the determination that 4 g/kg of chitosan was the most effective dose for promoting the greatest rates of body weight gain (BWG) and specific growth rate (SGR) (Wu 2020). Moreover, the addition of chitosan at a dosage of 5 g/kg to the diet of Nile tilapia for a duration of 60 days resulted in enhanced growth performance, body weight gain (BWG), specific growth rate (SGR), and feed conversion ratio (FCR) (Fadl et al. 2020).It has been shown that chromium nanoparticles added to the supplemental feed improve growth metrics, boost immunity, and lower insulin and cortisol levels. In particular, it has been shown that adding chromium nanoparticles to meals based on sunflower seeds at a dose of 1.5–2 mg/kg − 1 body weight increases the growth factor of C. mrigala(Akter et al. 2018).Additionally, adding chromium to Catla catla fingerlings at a dose of 2 mg/kg body weight enhanced their development, digestion, and hematological parameters (Ahmad et al. 2022).Supplementing fish feed with selenium nanoparticles may successfully treat selenium insufficiency in fish. Insufficient presence of selenium nanoparticles in the feed might result in fish developing abnormalities (Abd El-Kader et al. 2021). Research was carried out on Nile tilapia (O. niloticus), which showed that the addition of selenium nanoparticles (1 mg/kg) to their diet led to enhanced immune response, increased antioxidant capability, and better intestinal morphology(Dawood et al. 2020a, b).
Fish reproduction
Incomplete vitellogenesis in females is a frequent issue in artificial reproduction of commercial aquatic animals because it prevents the final oocyte from maturing and ovulating. In order to solve this issue, we need to find ways to regulate reproduction. The endogenous hormone may be carried by chitosan NPs and released in a regulated manner (Khosravi-Katuli et al. 2017). Nanotechnology facilitated the delivery of fadrozole, an inhibitor of estrogen synthesis, to Nile Tilapia (O. niloticus). Exposure to high concentrations of Fadrozole on PLGA nanoparticles, ranging from 50 to 500 parts per million (ppm), for a duration of one month, resulted in a complete male population at concentrations of 350 and 500 ppm (Joshi et al. 2019). In addition, (Clarias magur) fish were injected with chitosan nanoparticles and eurycomanone nanoparticles (extracted from Eurycoma longifolia plant, which had previously improved animal sexuality and fertility). The gonado-somatic index, calcium (Ca) and selenium (Se) concentrations, reproductive ability, and endocrine hormone gene expression increased after 7 days (Bhat et al. 2019a, b).
Selenium nanoparticles (Se-N) supplementation of a plant protein-rich diet (60% of fish meal was replaced with a mixture of alternative PP sources) for three months was tested on Arabian yellowfin sea bream (Acanthopagrus arabicus) males' sperm kinetics and fertilization capacity, and PP-rich diets received 0, 0.5, 1, 2, and 4 mg/kg Se-N. More sperm was detected in 2 mg Se-N/kg PP-fed fish. PP-rich foods improved Se-N, sperm motility, straight movement, lifespan, and fertilization. The maximum semen superoxide dismutase activity was found in fish given 4 mg Se-N/kg PP-rich diets, whereas the lowest was 2 mg. Semen glutathione peroxidase activity was highest in fish fed 4 mg Se-N/kg of PP-rich diet and lowest in those fed 0 and 0.5 mg. Fish given 1 or 4 mg Se-N/kg PP-rich diets have higher antioxidant capability. Fish given PP without Se-N had greater semen malondialdehyde levels. Arabian yellowfin seabream (A. arabicus) males had better sperm kinetics and fertility on a PP-rich diet with 2–4 mg Se-N/kg (Khademzade et al. 2022).
Fish medicine
Pathogens (viruses, bacteria, fungi, and parasites) cause substantial economic losses in fish aquaculture. The conventional fish treatment methods included antibiotics and chemicals, which created antibiotic-resistant microorganisms, water pollution, and chemical residues in fish tissues (Abbas 2021). One major problem in aquaculture was controlling infectious diseases produced by microbial pathogens. Antibiotic overuse in fish aquaculture has led to several disease-causing bacteria in fish developing resistance to frequently used antibiotics. This has necessitated the development of novel treatment strategies to address this difficulty. Nanoparticles are suggested as alternative antimicrobials to address the issue of bacteria resistance to antibiotics in aquaculture (Okeke et al. 2022). Metal nanoparticles have shown effective antimicrobial properties against bacterial, fungal, and viral infections by destroying the microbial cell membrane/cell wall, disrupting protein transports, inactivating key enzymes, and many other mechanisms (Nasr-Eldahan et al. 2021). The most common antimicrobial nanomaterials are metals and metal oxides: Silver (Ag), Gold(Au), Zincoxide (ZnO), Copper (Cu), and Titanium oxide (TiO2) (Abbas 2021).
The most documented antibacterial is Nano-Ag (Abbas 2021). The biologically synthesized nano-Ag particles derived from red algae (Portieria hornemannii) has shown antibacterial efficacy against four strains of fish pathogens (V. harveyi, V. anguillarum, V. vulnificus, and V. parahaemolyticus) (Fatima et al. 2020). The ZnO nanoparticles also have shown significant antibacterial activity against a variety of microbes (Raghunath and Perumal 2017; Sirelkhatim et al. 2015). In a study was conducted to evaluate the in vitro antibacterial efficacy of ZnO nanoparticles (ZnO NPs) against a pathogenic strain of Streptococcus parauberis. Zinc oxide nanoparticles (ZnO NPs) demonstrated significant suppression of proliferation in a pathogenic strain of Streptococcus parauberis at concentrations of 0.125 mg/ml and 0.250 mg/ml (Fadl et al. 2021). Nano-Au also affect microbial cell activities after interacting with proteins and lipopolysaccharides (Sumbayev et al. 2013). Nano-Au biologically produced with cashew nut shell liquid inhibited the growth of A. bestiarum and p. fluorescens in laboratory tests (Velmurugan et al. 2014). A study showed the antibacterial activity of chitosan nanoparticles (CNP) against the bacteria isolated from Nile tilapia (O. niloticus). The highest inhibitory zones were seen for Aspergillus flavus, Mucor sp., and Candida sp. at 80 μg/ml CNP dosages, although less impacted, as were Aspergillus niger, A. fumigatus, and Fusarium sp. The examined bacteria showed the greatest inhibitory zones with 20 μg/ml of CNP (Abdel-Razek 2019).
In a study the nano-ZnO exhibited antifungal properties against Aphanomyces invadans, the primary agent of red spot disease in fish (Shaalan et al. 2017). Instead of poisonous and carcinogenic malachite green, nano-Cu has shown antifungal activity against Saprolegnia sp. in vitro (Kalatehjari et al. 2015).
Metal nanoparticles have shown beneficial effects in the treatment of many parasitic diseases (Brahmchari et al. 2023). One treatment of nano-Ag in a concentration of 10 ng/ml of nano-Ag exhibited anti-parasitic effects against Ichthyophthirius multifiliis infection in rainbow trout (Oncoryhnchus mykiss) both in vitro and in vivo. It resulted in a 50% death rate of I. multifiliis after 30 min and a 100% mortality rate within 2 h (Abdel-Baki et al. 2017). A study was carried out to examine the anti-parasitic effects of biologically synthesized iron nanoparticles against Argulus siamensis in a controlled laboratory environment. The Fe-NPs had the highest argulocidal activity when used at a dose of 1.75 mg ml−1 for juveniles and 2.00 mg ml−1 for adult argulids. This resulted in a mortality rate of 100% for juveniles and 87% for adults over a period of 6 h (Brahmchari et al. 2023).
Producing efficient vaccinations and delivery systems to combat viral diseases is crucial to fish farming. Fish are vaccinated by injection, immersion, or oral means. The orally encapsulated vaccine is the best because it inhibits antigens from exiting food granules, protects them from acidic stomachs, decreases fish stress, and is appropriate for mass vaccination purposes (Vinay et al. 2018). Nanotechnology supports to eliminate harmful and carcinogenic chemical adjuvants in fish vaccination, and oral or immersion vaccinations are better than injections to avoid stress (Rivas-Aravena et al. 2015). Fish vaccinations against the infectious salmon anemia virus (ISAV) have been created with the use of chitosan nanoparticles. One such vaccine uses an adjuvant made of the DNA coding for ISAV replicas. In terms of ISAV protection, this immunization showed > 77% protection rates (Rivas-Aravena et al. 2015). Kole et al. (2018) vaccinated rohu fish (Labeo rohita) using chitosan nanoparticles combined with a bicistronic DNA plasmid containing the antigen Edwardsiella tarda glyceraldehyde 3-phosphate dehydrogenase and the immune adjuvant gene Labeo rohita IFN-γ. Rainbow trout (Oncorhynchus mykiss) that were immunized against bacterial infection (Lactococcus garvieae and Streptococcus iniae) using a chitosan-alginate coated vaccine showed improved outcomes compared to fish who received a non-coated vaccination. These improvements included greater survival rates, increased expression of immune-related genes, and higher antibody levels (Halimi et al. 2019). Olive flounder (Paralichthys olivaceus) that received a vaccine against inactivated viral haemorrhagic septicaemia virus, which was encapsulated with chitosan, demonstrated successful immunization in the head kidney, the primary organ responsible for initiating adaptive immunity in fish. Additionally, the vaccine was effective in stimulating immune responses in the skin and intestine, which are the primary sites for antigen uptake and mucosal immunity. In addition to the increased expression of IgM, IgT, pIgR, MHC-I, MHC-II, and IFN-γ in the three tissues, caspase 3 was also significantly upregulated 48 h after the challenge. This indicates the presence of cytotoxicity caused by a rapid T-cell response and inhibition of viral replication (Kole et al. 2019). Chitosan-based bivalent nano-vaccines, which included S. iniae and F. covae, were administered to Asian Seabass (Lates calcarifer) using immersion immunization at 30 and 40 days after hatching. The third vaccine was administered orally by food at 50 days after hatching. The results showed a significant rise in the levels of total IgM and specific IgM for both S. iniae and F. covae. Significantly greater levels of IgT, IgM, MHCIIα, and TCRα were seen in all vaccinated groups. Every group that received immunizations had higher survival rates when faced with the F. covae challenge (Meachasompop et al. 2024).
Nanoparticles as hormone and drug delivery vehicles
Significant progress has been made recently in the use of nanoparticles to deliver medicinal medications to their target sites(Obeid et al. 2017).The use of nanotechnology in drug delivery allows for unique characteristics such as controlled release, precise control over the size, shape, and surface charge of targeted materials, location-specific and multi-route delivery methods, and regulated degradation of the nanocarrier (Patra et al. 2018). In fish, chitosan and polylactic acid (PLGA) nanoparticles have been studied for their potential as medication delivery vehicles (Shaalan et al. 2016). Polylactic-glycolic acid nanoparticles are a copolymer. It is FDA-approved as biodegradable and non -toxic (Abbas 2021).
Chitosan nanoparticles have been used for medication delivery in research focused on promoting appropriate gonadal development in aquaculture. Bhat et al. (2016) injected walking catfish (Clarias batrachus) with chitosan conjugated with salmon luteinizing hormone-releasing hormone (sLHRH) to stimulate gonadal development. Chitosan-conjugated sLHRH and naked sLHRH had comparable effects, causing a rise in Sox9 expression in the gonads and elevating levels of testosterone and 11-ketotestosterone in males, and testosterone and 17β-estradiol in females. Conjugating sLHRH with chitosan resulted in a continuous and regulated release of hormones, reaching maximum levels after 36 h. In contrast, administering naked sLHRH led to peak levels of circulating steroid hormones after 12 h (Bhat et al. 2016). Compared to administering naked kisspeptin-10, injecting chitosan-encapsulated kisspeptin-10 into immature female Catla catla caused a delayed but more significant increase in gonadotropin-releasing hormone, luteinizing hormone, and follicle-stimulating hormone expression, as well as circulating levels of 11-ketotestosterone and 17β-estradiol (Rather et al. 2016). Chitosan was tested for gene delivery to influence gonadal development in fish. Chitosan nanoparticles conjugated with a plasmid encoding steroidogenic acute regulatory protein (StAR) administered intramuscularly in walking catfish (Clarias batrachus) showed longer-lasting stimulatory effects on the expression of key genes involved in reproduction, such as cytochrome P450 (CYP) 11A1, CYP17A1, CYP19A1, 3β-hydroxysteroid dehydrogenase, and 173β-hydroxysteroid dehydrogenase, compared to administration of the naked plasmid construct (Rathor et al. 2017). To encourage goldfish oogenesis, research suggested oral delivery of chitosan nanoparticles along with a GnRH analog. Chitosan, 50 μg GnRHa/kg b.w., 100 μg, chitosan + 50 μg, and chitosan + 100 μg were administered to adult female goldfish. The width of the follicular layer (Fl), the thickness of the zona radiata (Zr), the diameter of the oocyte (OD), and the gonadosomatic index (GSI) were measured. The metrics consistently rose in the group that received a dosage of 100 μg of GnRH or a combination of chitosan nanoparticles and 100 μg of GnRHa. The investigation discovered that the use of oral chitosan in conjunction with a dosage of 100 μg GnRHa/kg b.w. significantly enhanced the maturation and expansion of ovarian oocytes (Kookaram et al. 2021). Direct applications of nanotechnology in fish culture are summarized in Table 1.
Indirect applications of nanotechnology in fish culture
Water purification
Due to their unique features, greater surface area, and numerous absorption sites, nanomaterials immobilize and adsorb metals well, allowing them to remediate contaminated water and sediments. A variety of nanomaterials, including metal oxide nanoparticles, nano zero-valent iron, carbon nanotubes, and natural adsorbents, have been utilized in the remediation of heavy metals (Cai et al. 2019).
Metal oxide nanoparticles (MON) are Fe2O3, Al2O3, MnO, MgO, and TiO2. MON also catalyzes the degradation of non-degradable pesticides including PCBs and organochlorines (Abbas 2021). Nano zero-valent iron allows metal adsorption with its metallic iron core and iron oxide shell (Yirsaw et al. 2016). Bentonite, kaolinite, and montmorillonite can remove heavy metals, hence several research mixed them with chitosan nanoparticles to improve their adsorption (Abbas 2021).
Pathogens in the water supply are known to multiply in fish farms owing to the high population density and leftover food particles. Many infectious disorders in fish are caused by pathogens such viruses, fungus, bacteria, and protozoa. Risks to pathogen resistance, user health, aquatic wildlife, and the environment are only some of the consequences of using traditional anti-pathogen chemicals (Abbas 2021). Nanotechnology could solve water sterilization and disinfection issues (Tayel et al. 2019).
Moreover, the presence of heavy metals and microbes in these waterways results in fish mortality and growth retardation, which causes a significant economic loss for the fishing sector. Nanotechnology is widely used in aquaculture to clean water to provide a safe and favorable environment for fish to spawn. From this angle, the scientific community supports photocatalysis and adsorption as the most cost-effective and efficient methods of water filtration. Furthermore, Fig. 1 demonstrates the practical functioning of several nanoparticle-based photocatalytic adsorbents and hydrogel biofilms in water purification. It demonstrates the recommended approach by showcasing the removal of nitrate (NO3−), fluoride (F−), and coliforms (E. Coli) from contaminated water (Shah and Mraz 2020b).
Bio-fouling control
Nanoparticles that function as the major oxides of the metals may combat biofouling caused by bacterial assault. When additional contaminants, including poisonous metals, build up in the water, biofouling may seem much more perilous and cause the deaths of many fish and other aquatic species. Aquaculture production and prawn culture may both benefit from the advancements in disease management, feeding formulation, and biofouling control that nanotechnology has made possible. To keep an eye out for undesired bacteria (as biofilm), nanostructures may be painted or coated with metal oxide nanoparticles like zinc oxide (ZnO), copper oxide (CuO), and silicon dioxide (SiO2), allowing for the monitoring of invertebrates like mussels and barnacles and algae like seaweeds and diatoms. Since nanotechnology is the primary scientific technique by which such severe environmental contamination may be managed, this is the case (Munawar et al. 2021).Anti-fouling agents made from lanthanides (La) oxides nanoparticles have been shown to inhibit algal and microbial development by absorbing phosphate from the surrounding water (Ashraf et al. 2011; Gerber et al. 2012). One of the commercial solutions that deal with the management of fish culture is called Nano-Check. Its structure is built on 40 nm nano La, which has the ability to absorb phosphate from the water and, as a result, limit the production of algae (Ashraf et al. 2011). In addition, because of nano-Ag's antibacterial properties, biofouling may be prevented and controlled (Hassan and Abd El-latif 2018; Vijayan et al. 2014a).
Fish packaging
Plastics, such as polyolefins, polyesters, and polyamides are gaining popularity among the wide variety of fish packaging materials due to the fact that they are readily available in large quantities at a low cost and have favorable functionality characteristics. These characteristics include good tensile and tear strength, good barrier properties to O2 and aroma compounds, and the ability to be heat sealed. Unfortunately, with the exception of cellophane, the majority of polymers are not biodegradable, and as a result, they contribute to environmental contamination and the subsequent severe ecological difficulties. In order to solve these problems, its use in any way, shape, or form ought to be limited and ought to be phased out gradually (Agarwal 2020).
Finally, nanotechnology can delay microbiological and enzymatic degradation to extend fish packaging and marketing shelf life. Nano-materials in biodegradable food packaging act as antibacterial and antifungal agents, eliminate oxygen, limit biological deterioration, and immobilize enzymes to promote product stability, according to several studies (Jiang et al. 2015; Kuswandi 2017; Rhim et al. 2013). Furthermore, nano-composites made from proteins, lipids, or polysaccharides are considered healthier packaging materials than petrochemical-based plastics (Can et al. 2011). Overview of indirect applications of nanotechnology in fish culture are given in Table 2 (Fig. 2).
Challenges of nanotechnology applications in fish culture
Environmental risk
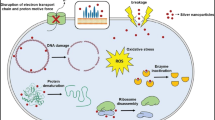
Environmental concerns have been raised over the widespread use of nanoparticles in several industries. However, nanoparticles and other foreign contaminants eventually find their way into the aquatic environment. In water, NPs can undergo chemical, physical, and biological changes through aggregation, adsorption, dissolution, and redox reaction. Because of this, NPs are modified to generate materials with novel attributes and reactivity (Odzak et al. 2017; Yin et al. 2015). The interaction of new materials with the biological system, both inorganic and organic components, and live organisms that are not specifically targeted causes damage to all of these entities. Cytotoxicity, malfunction of various cell organelles (including the cell membrane, mitochondria, and ribosomes), DNA damage and genetic toxicity meant that toxic effects might be passed to the fetus and increased while the fetus was developing (Fig. 3) (Zhang et al. 2018). Nanomaterials can be coated and interacted with organic compounds like humic acid and fulvic acid to change their physicochemical properties, behavior, and harmful impacts on living beings (Cai et al. 2019; Tang et al. 2014). Additionally, nanomaterials like apatite and biochar supported by Fe-phosphate increase water phosphorus levels, causing eutrophication, a major issue in fish culture (Qiao et al. 2017).
Technical producing difficulties
Nanoparticles are especially difficult to manufacture because they are prone to re-aggregation, transformation, and reaction with an extensive range of environmental variables (Zhang et al. 2018). Nanoparticles are difficult to characterize because of the high expense of the procedures required to determine their shape, size, and morphology (Patil and Kim 2017).
High cost
Until now, the high price of nanotechnology has prevented its widespread use in fish farming. This is especially true of nano-filters, nano-membranes, and nano-sensors. But its uses in egg and hatching ponds, as well as in the case of prized broad-stocks, have been documented (Bhattacharyya et al. 2015).
Lack of information
There is a lack of information regarding how nanoparticles are absorbed, distributed, accumulate in the food chain, and excreted from living creatures, despite the widespread use of nanotechnology over the past decade (Tripathi et al. 2017). Most research on nanoparticles and their impact on fish have only been conducted in the laboratory or in vitro. Without further information and more study, it will be very difficult to move forward with field application.
Toxicity
Since nanoparticles are being used in so many different applications, scientists will inevitably look into their potential effects on humans as well as the environment. Overuse and incorrect disposal of nanoparticles lead to harmful consequences and negative impacts on the environment, which in turn have detrimental effects on the health of living beings (Hu et al. 2016; Samrot et al. 2019).
Nanoparticles are so tiny that they may cross cell membranes and induce genotoxicity within the cell. One of the primary toxicity mechanisms of nanoparticles is the increased synthesis of free radicals and reactive oxygen species (ROS). It is possible that this will not only cause oxidative stress and inflammation, but it may also cause problems with DNA and proteins. Evidence shows that exposure to nanomaterials may cause mutations in DNA and severe damage to mitochondrial structure, which may ultimately lead to cell death (Majumder and Dash 2017; Meghani et al. 2020; Vicari et al. 2018). Studies in nanotoxicology have been performed to better understand the regulatory framework and bio-distribution of these substances while also assessing the possible molecular hazards they pose to ecosystems (Bello and Leong 2017).
It has been shown that several nano-materials employed in water remediation, such as nanoscale metallic iron (nZVI), are harmful to bacteria, crustaceans, fish larvae, and other aquatic and soil dwelling creatures(Stefaniuk et al. 2016).After being exposed to Al2O3-NPs, Nile tilapia (Oreochromis niloticus) displayed symptoms of oxidative stress, as shown by a decrease in antioxidant enzyme (SOD, CAT, and GPx) activity (Temiz and Kargın 2022). Nile tilapia (Oreochromis niloticus) cytokine transcription was affected by copper oxide nanoparticles (Abdel-Latif et al. 2021a, b). Cells exposed to Cu-NPs or CuSO4 may cause an elevation in malondialdehyde and reactive oxygen species levels. This can lead to a decline in mitochondrial bioenergetics and impair physiological functions. After then, caspase-3 and caspase-9 were activated due to the release of Cyt-c from mitochondria into the cytosol, which prompted apoptosis. Cu-induced apoptosis in adolescent E. coioides was mediated via the mitochondrial route (Wang et al. 2015). The inability of cells to repair the damage to their membranes that had been caused by lipid peroxidation as a result of being exposed to AgNP was what ultimately led to the death of the cells. The autophagy process was impacted by the nanoparticles that were internalized, which resulted in the discharge of nanoparticles into the cytosol. Because of this, the lysosomes were unable to function properly, and the mitochondrial membrane became more permeable (Quevedo et al. 2021). Toxicity NPs in fish culture using different parameters are also illustrated in Table 3.
Conclusion
The preceding situation suggests that, similar to other developing technologies, nanotechnology possesses positive as well as negative aspects. The use of nanotechnology in fish culture has the potential to completely transform them and address several fish culture challenges with more efficiency compared to traditional approaches. They have the ability to contribute to improving fish development performance and production, managing fish illnesses, purifying water, remediating contaminants, and ultimately prolonging the shelf-life of fish in packaging. Simultaneously, the use of nanotechnology in fish farming encounters several obstacles, with the most significant being the potential toxicity of nanoparticles and their harmful effects on fish and other unintended creatures. However, it is essential to evaluate the absolute safety of using nanoparticles in fish farming and to determine their potential accumulation in the food chain to ensure the health of the fish, the environment, and human consumption.
References
Abbas WT (2021) Advantages and prospective challenges of nanotechnology applications in fish cultures: a comparative review. Environ Sci Pollut Res 28(7):7669–7690. https://doi.org/10.1007/s11356-020-12166-0
Abd El-Kader MF, Fath El-Bab AF, Abd-Elghany MF, Abdel-Warith A-WA, Younis EM, Dawood MAO (2021) Selenium nanoparticles act potentially on the growth performance, hemato-biochemical indices, antioxidative, and immune-related genes of European seabass (Dicentrarchus labrax). Biol Trace Elem Res 199(8):3126–3134. https://doi.org/10.1007/s12011-020-02431-1
Abd El-Naby FS, Naiel MAE, Al-Sagheer AA, Negm SS (2019) Dietary chitosan nanoparticles enhance the growth, production performance, and immunity in Oreochromis niloticus. Aquaculture 501:82–89. https://doi.org/10.1016/j.aquaculture.2018.11.014
Abd El-Naby AS, Al-Sagheer AA, Negm SS, Naiel MAE (2020) Dietary combination of chitosan nanoparticle and thymol affects feed utilization, digestive enzymes, antioxidant status, and intestinal morphology of Oreochromis niloticus. Aquaculture 515:734577. https://doi.org/10.1016/j.aquaculture.2019.734577
Abdel-Baki A-A, Al-Quraishy S, Dkhil MA, El-Matbouli M, Saleh M (2017) Antiprotozoal effects of metal nanoparticles against Ichthyophthirius multifiliis. Parasitology 144(13):1802–1810. https://doi.org/10.1017/S0031182017001184
Abdel-Latif HM, Dawood MA, Mahmoud SF, Shukry M, Noreldin AE, Ghetas HA, Khallaf MA (2021a) Copper oxide nanoparticles alter serum biochemical indices, induce histopathological alterations, and modulate transcription of cytokines, HSP70, and oxidative stress genes in Oreochromis niloticus. Animals 11(3):652
Abdel-Latif HMR, Dawood MAO, Mahmoud SF, Shukry M, Noreldin AE, Ghetas HA, Khallaf MA (2021b) Copper oxide nanoparticles alter serum biochemical indices, induce histopathological alterations, and modulate transcription of cytokines, HSP70, and oxidative stress genes in Oreochromis niloticus. Animals. https://doi.org/10.3390/ani11030652
Abdel-Razek N (2019) Antimicrobial activities of chitosan nanoparticles against pathogenic microorganisms in Nile tilapia Oreochromis niloticus. Aquac Int 27(5):1315–1330. https://doi.org/10.1007/s10499-019-00388-0
Abdel-Tawwab M, Razek NA, Abdel-Rahman AM (2019) Immunostimulatory effect of dietary chitosan nanoparticles on the performance of Nile tilapia, Oreochromis niloticus (L.). Fish Shellfish Immunol 88:254–258. https://doi.org/10.1016/j.fsi.2019.02.063
Abimbola OF, Olotu TM, Adetunji CO, Alabetutu A (2023) Chapter 16: applications of nanochitosan in the detection and control of aquatic diseases. In: Adetunji C, Hefft D, Jeevanandam J, Danquah M (eds) Next generation nanochitosan. Academic Press, pp 221–234
Action S (2020) World fisheries and aquaculture. Food Agric Organ 2020:1–244
Agarwal S (2020) Biodegradable polymers: present opportunities and challenges in providing a microplastic-free environment. Macromol Chem Phys 221(6):2000017. https://doi.org/10.1002/macp.202000017
Ahmad N, Hussain SM, Azam SM, Shahzad MM, Noureen A, Yaqoob R, Ahmad S (2022) Effects of Se nanoparticles supplementation on growth performance, hematological parameters and nutrient digestibility of Labeo rohita fingerling fed sunflower meal based diet. Braz J Biol 84:e253555
Akter N, Alam MJ, Jewel MAS, Ayenuddin M, Haque SK, Akter S (2018) Evaluation of dietary metallic iron nanoparticles as feed additive for growth and physiology of Bagridae catfish Clarias batrachus (Linnaeus, 1758). Int J Fish Aquatic Stud 6(3):371–377
Alhadrami HA, Al-Hazmi F (2017) Antibacterial activities of titanium oxide nanoparticles. J Bioelectron Nanotechnol2(5)
Aruoja V, Dubourguier H-C, Kasemets K, Kahru A (2009) Toxicity of nanoparticles of CuO, ZnO and TiO2 to microalgae Pseudokirchneriella subcapitata. Sci Total Environ 407(4):1461–1468. https://doi.org/10.1016/j.scitotenv.2008.10.053
Ashouri S, Keyvanshokooh S, Salati AP, Johari SA, Pasha-Zanoosi H (2015) Effects of different levels of dietary selenium nanoparticles on growth performance, muscle composition, blood biochemical profiles and antioxidant status of common carp (Cyprinus carpio). Aquaculture 446:25–29. https://doi.org/10.1016/j.aquaculture.2015.04.021
Ashraf PM, Edwin L (2016) Nano copper oxide incorporated polyethylene glycol hydrogel: an efficient antifouling coating for cage fishing net. Int Biodeterior Biodegradation 115:39–48. https://doi.org/10.1016/j.ibiod.2016.07.015
Ashraf M, Aklakur M, Sharma R, Ahmad SKM (2011) Nanotechnology as a novel tool in fisheries and aquaculture development: a review. Iran J Energy Environ 2(3):258–261
Assefa A, Abunna F (2018) Maintenance of fish health in aquaculture: review of epidemiological approaches for prevention and control of infectious disease of fish. Veterin Med Int 2018:5432497. https://doi.org/10.1155/2018/5432497
Aziz S, Abdullah S (2023) Evaluation of toxicity induced by engineered CuO nanoparticles in freshwater fish, labeo rohita. Turk J Fish Aquat Sci 23(8)
Baranowska-Wójcik E, Szwajgier D, Oleszczuk P, Winiarska-Mieczan A (2020) Effects of titanium dioxide nanoparticles exposure on human health—a review. Biol Trace Elem Res 193(1):118–129. https://doi.org/10.1007/s12011-019-01706-6
Bello, D., & Leong, D. (2017). A decade of nanotoxicology: Assessing the impact on human health and the environment! NanoImpact, 7. https://doi.org/10.1016/j.impact.2017.04.001
Bhat IA, Rather MA, Saha R, Pathakota G-B, Pavan-Kumar A, Sharma R (2016) Expression analysis of Sox9 genes during annual reproductive cycles in gonads and after nanodelivery of LHRH in Clarias batrachus. Res Vet Sci 106:100–106. https://doi.org/10.1016/j.rvsc.2016.03.022
Bhat IA, Ahmad I, Mir IN, Bhat RAH, Gireesh-Babu P, Goswami M (2019a) … Biology, M. Chitosan-Eurycomanone Nanoformulation Acts on Steroidogenesis Pathway Genes to Increase the Reproduction Rate in Fish 185:237–247
Bhat IA, Ahmad I, Mir IN, Bhat RAH, P, G.-B., Goswami, M., … Sharma, R. (2019b) Chitosan-eurycomanone nanoformulation acts on steroidogenesis pathway genes to increase the reproduction rate in fish. J Steroid Biochem Mol Biol 185:237–247. https://doi.org/10.1016/j.jsbmb.2018.09.011
Bhattacharyya, A., Reddy, S., Hasan, M., Adeyemi, M., Marye, R., & Naika, H. J. I. J. o. B. (2015). Nanotechnology-a unique future technology in aquaculture for the food security. 4(7), 4115–4126.
Bhuvaneshwari M, Iswarya V, Archanaa S, Madhu GM, Kumar GS, Nagarajan R, Mukherjee A (2015) Cytotoxicity of ZnO NPs towards fresh water algae Scenedesmus obliquus at low exposure concentrations in UV-C, visible and dark conditions. Aquatic Toxicol 162:29–38
Cai C, Zhao M, Yu Z, Rong H, Zhang C (2019) Utilization of nanomaterials for in-situ remediation of heavy metal(loid) contaminated sediments: a review. Sci Total Environ 662:205–217. https://doi.org/10.1016/j.scitotenv.2019.01.180
Can E, Kizak V, Kayim M, Can SS, Kutlu B, Ates M (2011). Nanotechnological applications in aquaculture-seafood industries and adverse effects of nanoparticles on environment. J Mater Sci Eng 5(5)
Cheng TC, Yao KS, Yeh N, Chang CI, Hsu HC, Chien YT, Chang CY (2009) Visible light activated bactericidal effect of TiO2/Fe3O4 magnetic particles on fish pathogens. Surf Coat Technol 204(6):1141–1144. https://doi.org/10.1016/j.surfcoat.2009.06.050
Cheng TC, Yao KS, Yeh N, Chang CI, Hsu HC, Gonzalez F, Chang CY (2011) Bactericidal effect of blue LED light irradiated TiO2/Fe3O4 particles on fish pathogen in seawater. Thin Solid Films 519(15):5002–5006. https://doi.org/10.1016/j.tsf.2011.01.069
Chris UO, Singh NB, Agarwal A (2018) Nanoparticles as feed supplement on growth behaviour of cultured catfish (Clarias gariepinus) fingerlings. Mater Today Proc 5(3):9076–9081. https://doi.org/10.1016/j.matpr.2017.10.023
Dawood MAO, Koshio S, Zaineldin AI, Van Doan H, Moustafa EM, Abdel-Daim MM, Hassaan MS (2019) Dietary supplementation of selenium nanoparticles modulated systemic and mucosal immune status and stress resistance of red sea bream (Pagrus major). Fish Physiol Biochem 45(1):219–230. https://doi.org/10.1007/s10695-018-0556-3
Dawood MAO, Eweedah NM, Moustafa EM, El-Sharawy ME, Soliman AA, Amer AA, Atia MH (2020a) Copper nanoparticles mitigate the growth, immunity, and oxidation resistance in common carp (Cyprinus carpio). Biol Trace Elem Res 198(1):283–292. https://doi.org/10.1007/s12011-020-02068-0
Dawood MAO, Zommara M, Eweedah NM, Helal AI (2020b) The evaluation of growth performance, blood health, oxidative status and immune-related gene expression in Nile tilapia (Oreochromis niloticus) fed dietary nanoselenium spheres produced by lactic acid bacteria. Aquaculture 515:734571. https://doi.org/10.1016/j.aquaculture.2019.734571
Dubey S, Avadhani K, Mutalik S, Sivadasan SM, Maiti B, Paul J, Munangandu HM (2016) Aeromonas hydrophila OmpW PLGA nanoparticle oral vaccine shows a dose-dependent protective immunity in rohu (Labeo rohita). Vaccines 4(2):21
El-Shenawy AM, Gad DM, Yassin S (2019) Effect of iron nanoparticles on the development of fish farm feeds. Alexand J Veterin Sci 60(1)
Fadl SE, El-Gammal GA, Abdo WS, Barakat M, Sakr OA, Nassef E, El-Sheshtawy HS (2020) Evaluation of dietary chitosan effects on growth performance, immunity, body composition and histopathology of Nile tilapia (Oreochromis niloticus) as well as the resistance to Streptococcus agalactiae infection. Aquac Res 51(3):1120–1132. https://doi.org/10.1111/are.14458
Faiz H, Zuberi A, Nazir S, Rauf, M, Younus N (2015) Zinc oxide, zinc sulfate and zinc oxide nanoparticles as source of dietary zinc: comparative effects on growth and hematological indices of juvenile grass carp (Ctenopharyngodon idella). Int J Agric Biol 17(3)
Fajardo C, Ortíz LT, Rodríguez-Membibre ML, Nande M, Lobo MC, Martin M (2012) Assessing the impact of zero-valent iron (ZVI) nanotechnology on soil microbial structure and functionality: a molecular approach. Chemosphere 86(8):802–808
Fenaroli F, Westmoreland D, Benjaminsen J, Kolstad T, Skjeldal FM, Meijer AH, Griffiths G (2014) Nanoparticles as drug delivery system against tuberculosis in zebrafish embryos: direct visualization and treatment. ACS Nano 8(7):7014–7026. https://doi.org/10.1021/nn5019126
Gerber LC, Moser N, Luechinger NA, Stark WJ, Grass RN (2012) Phosphate starvation as an antimicrobial strategy: the controllable toxicity of lanthanum oxide nanoparticles. Chem Commun 48(32):3869–3871
Ghafarifarsani H, Hedayati SA, Yousefi M, Hoseinifar SH, Yarahmadi P, Mahmoudi SS, Van Doan H (2023) Toxic and bioaccumulative effects of zinc nanoparticle exposure to goldfish, Carassius auratus (Linnaeus, 1758). Drug Chem Toxicol 46(5):984–994. https://doi.org/10.1080/01480545.2022.2115509
Gunalan S, Sivaraj R, Rajendran V (2012) Green synthesized ZnO nanoparticles against bacterial and fungal pathogens. Progress Nat Sci Mater Int 22(6):693–700. https://doi.org/10.1016/j.pnsc.2012.11.015
Hajji S, Hamdi M, Boufi S, Li S, Nasri M (2019) Suitability of chitosan nanoparticles as cryoprotectant on shelf life of restructured fish surimi during chilled storage. Cellulose 26(11):6825–6847. https://doi.org/10.1007/s10570-019-02555-1
Halimi M, Alishahi M, Abbaspour MR, Ghorbanpoor M, Tabandeh MR (2019) Valuable method for production of oral vaccine by using alginate and chitosan against Lactococcus garvieae/Streptococcus iniae in rainbow trout (Oncorhynchus mykiss). Fish Shellfish Immunol 90:431–439. https://doi.org/10.1016/j.fsi.2019.05.020
Hassan SW, Abd El-latif HH (2018) Characterization and applications of the biosynthesized silver nanoparticles by marine Pseudomonas sp. H64. J Pure Appl Microbiol 12(3):1289–1299
He X, Deng H, Hwang H-M (2019) The current application of nanotechnology in food and agriculture. J Food Drug Anal 27(1):1–21. https://doi.org/10.1016/j.jfda.2018.12.002
Hilty FM, Knijnenburg JTN, Teleki A, Krumeich F, Hurrell RF, Pratsinis SE, Zimmermann MB (2011) Incorporation of Mg and Ca into nanostructured Fe2O3 improves fe solubility in dilute acid and sensory characteristics in foods. J Food Sci 76(1):N2–N10. https://doi.org/10.1111/j.1750-3841.2010.01885.x
Hu X, Li D, Gao Y, Mu L, Zhou Q (2016) Knowledge gaps between nanotoxicological research and nanomaterial safety. Environ Int 94:8–23. https://doi.org/10.1016/j.envint.2016.05.001
Huang S, Wang L, Liu L, Hou Y, Li L (2015) Nanotechnology in agriculture, livestock, and aquaculture in China: a review. Agron Sustain Dev 35(2):369–400. https://doi.org/10.1007/s13593-014-0274-x
Ibrahim ATA (2020) Toxicological impact of green synthesized silver nanoparticles and protective role of different selenium type on Oreochromis niloticus: hematological and biochemical response. J Trace Elem Med Biol 61:126507. https://doi.org/10.1016/j.jtemb.2020.126507
Ilangovan R, Subha V, Ravindran RE, Kirubanandan S, Renganathan S (2021) Nanomaterials: synthesis, physicochemical characterization, and biopharmaceutical applications. In Nanoscale processing, pp 33–70. Elsevier
Ismael NE, Abd El-hameed SA, Salama AM, Naiel MA, Abdel-Latif HM (2021) The effects of dietary clinoptilolite and chitosan nanoparticles on growth, body composition, haemato-biochemical parameters, immune responses, and antioxidative status of Nile tilapia exposed to imidacloprid. Environ Sci Pollut Res 28:29535–29550
Jiang X, Valdeperez D, Nazarenus M, Wang Z, Stellacci F, Parak WJ, del Pino P (2015) Future perspectives towards the use of nanomaterials for smart food packaging and quality control. Part Part Syst Charact 32(4):408–416. https://doi.org/10.1002/ppsc.201400192
Johari SA, Kalbassi MR, Soltani M, Yu IJ (2016) Application of nanosilver-coated zeolite as water filter media for fungal disinfection of rainbow trout (Oncorhynchus mykiss) eggs. Aquac Int 24:23–38
Joshi HD, Tiwari VK, Gupta S, Sharma R, Lakra WS, Sahoo U (2019) Application of nanotechnology for the production of masculinized Tilapia, Oreochromis niloticus (Linnaeus, 1758). Aquaculture 511:734206
Jovanović B, Whitley EM, Kimura K, Crumpton A, Palić D (2015) Titanium dioxide nanoparticles enhance mortality of fish exposed to bacterial pathogens. Environ Pollut 203:153–164. https://doi.org/10.1016/j.envpol.2015.04.003
Kakakhel MA, Wu F, Sajjad W, Zhang Q, Khan I, Ullah K, Wang W (2021) Long-term exposure to high-concentration silver nanoparticles induced toxicity, fatality, bioaccumulation, and histological alteration in fish (Cyprinus carpio). Environ Sci Eur 33(1):14. https://doi.org/10.1186/s12302-021-00453-7
Kalatehjari P, Yousefian M, Khalilzadeh MA (2015) Assessment of antifungal effects of copper nanoparticles on the growth of the fungus Saprolegnia sp. on white fish (Rutilus frisii kutum) eggs. Egypt J Aquat Res 41(4):303–306. https://doi.org/10.1016/j.ejar.2015.07.004
Khademzade O, Kochanian P, Zakeri M, Alavi SMH, Mozanzadeh MT (2022) Oxidative stress-related semen quality and fertility in the male Arabian Yellowfin Sea Bream (Acanthopagrus arabicus) fed a selenium nanoparticle-supplemented plant protein-rich diet. Aquac Nutr 2022:3979203. https://doi.org/10.1155/2022/3979203
Khan KU, Zuberi A, Nazir S, Ullah I, Jamil Z, Sarwar H (2017) Synergistic effects of dietary nano selenium and vitamin C on growth, feeding, and physiological parameters of mahseer fish (Tor putitora). Aquac Rep 5:70–75. https://doi.org/10.1016/j.aqrep.2017.01.002
Khan MZH, Hossain MMM, Khan M, Ali MS, Aktar S, Moniruzzaman M, Khan M (2020) Influence of nanoparticle-based nano-nutrients on the growth performance and physiological parameters in tilapia (Oreochromis niloticus). RSC Adv 10(50):29918–29922
Khoei AJ (2021) Evaluation of potential immunotoxic effects of iron oxide nanoparticles (IONPs) on antioxidant capacity, immune responses and tissue bioaccumulation in common carp (Cyprinus carpio). Comp Biochem Physiol C: Toxicol Pharmacol 244:109005. https://doi.org/10.1016/j.cbpc.2021.109005
Khosravi-Katuli K, Prato E, Lofrano G, Guida M, Vale G, Libralato G (2017) Effects of nanoparticles in species of aquaculture interest. Environ Sci Pollut Res 24(21):17326–17346. https://doi.org/10.1007/s11356-017-9360-3
Kitiyodom S, Yata T, Yostawornkul J, Kaewmalun S, Nittayasut N, Suktham K, Pirarat N (2019) Enhanced efficacy of immersion vaccination in tilapia against columnaris disease by chitosan-coated “pathogen-like” mucoadhesive nanovaccines. Fish Shellfish Immunol 95:213–219. https://doi.org/10.1016/j.fsi.2019.09.064
Klingelfus T, Disner GR, Voigt CL, Alle LF, Cestari MM, Leme DM (2019) Nanomaterials induce DNA-protein crosslink and DNA oxidation: a mechanistic study with RTG-2 fish cell line and Comet assay modifications. Chemosphere 215:703–709. https://doi.org/10.1016/j.chemosphere.2018.10.118
Kole S, Qadiri SSN, Shin S-M, Kim W-S, Lee J, Jung S-J (2019) Nanoencapsulation of inactivated-viral vaccine using chitosan nanoparticles: evaluation of its protective efficacy and immune modulatory effects in olive flounder (Paralichthys olivaceus) against viral haemorrhagic septicaemia virus (VHSV) infection. Fish Shellfish Immunol 91:136–147. https://doi.org/10.1016/j.fsi.2019.05.017
Kookaram K, Mojazi Amiri B, Dorkoosh FA, Nematollahi MA, Mortazavian E, Abed Elmdoust A (2021) Effect of oral administration of GnRHa+nanoparticles of chitosan in oogenesis acceleration of goldfish Carassius auratus. Fish Physiol Biochem 47(2):477–486. https://doi.org/10.1007/s10695-021-00926-9
Kumar V, Sharma N, Maitra SS (2017) In vitro and in vivo toxicity assessment of nanoparticles. Int Nano Lett 7(4):243–256. https://doi.org/10.1007/s40089-017-0221-3
Kumar N, Krishnani KK, Singh NP (2018) Effect of dietary zinc-nanoparticles on growth performance, anti-oxidative and immunological status of fish reared under multiple stressors. Biol Trace Elem Res 186(1):267–278
Kuswandi B (2017) Environmental friendly food nano-packaging. Environ Chem Lett 15(2):205–221. https://doi.org/10.1007/s10311-017-0613-7
Kwasek K, Thorne-Lyman AL, Phillips M (2020) Can human nutrition be improved through better fish feeding practices? a review paper. Crit Rev Food Sci Nutr 60(22):3822–3835. https://doi.org/10.1080/10408398.2019.1708698
Li M, Wu Q, Wang Q, Xiang D, Zhu G (2018) Effect of titanium dioxide nanoparticles on the bioavailability and neurotoxicity of cypermethrin in zebrafish larvae. Aquat Toxicol 199:212–219
López-Barrera EA, Grotzner SR, Esquivel L, Voigt CL, Campos SX, de Oliveira Ribeiro CA (2021) Histopathological effects of silver nanoparticles in Rhamdia quelen after oral exposure. Ecotoxicol Environ Contam 16(1):83–89
Luis AIS, Campos EVR, de Oliveira JL, Fraceto LF (2019) Trends in aquaculture sciences: from now to use of nanotechnology for disease control. Rev Aquac 11(1):119–132. https://doi.org/10.1111/raq.12229
Luo Z, Li M, Wang Z, Li J, Guo J, Rosenfeldt RR, Yan C (2018) Effect of titanium dioxide nanoparticles on the accumulation and distribution of arsenate in Daphnia magna in the presence of an algal food. Environ Sci Pollut Res 25:20911–20919
Majumder D, Dash G (2017) Application of nanotechnology in fisheries and aquaculture. Everyman Sci 51(6):358–364
Meachasompop P, Bunnoy A, Keaswejjareansuk W, Dechbumroong P, Namdee K, Srisapoome P (2024) Development of immersion and oral bivalent nanovaccines for streptococcosis and columnaris disease prevention in fry and fingerling asian seabass (Lates calcarifer) nursery farms. Vaccines. https://doi.org/10.3390/vaccines12010017
Meghani N, Dave S, Kumar A (2020) Introduction to nanofood. In: Hebbar U, Ranjan S, Dasgupta N, Kumar-Mishra R (eds) Nano-food engineering, vol 1. Springer International Publishing, Cham, pp 1–23
Mizielińska M, Kowalska U, Jarosz M, Sumińska P (2018) A comparison of the effects of packaging containing nano ZnO or polylysine on the microbial purity and texture of cod (Gadus morhua) Fillets. Nanomaterials. https://doi.org/10.3390/nano8030158
Mohanty BP (2015) Nutritional value of food fish. Conspect Inland Fish Manag 4:15–21
Munawar N, Aleem M, Abru S, Laraib T, Zainab SN, Manzoor S, Ali A (2021) Interaction and applications of nanoparticles in fishes and aquaculture. Sch Bull 7(6):150–155
Naguib M, Mahmoud UM, Mekkawy IA, Sayed AE-DH (2020) Hepatotoxic effects of silver nanoparticles on Clarias gariepinus; Biochemical, histopathological, and histochemical studies. Toxicol Rep 7:133–141. https://doi.org/10.1016/j.toxrep.2020.01.002
Narsale SA, Debbarma S, Prakash P, Sheikh S, Shinde S, Baidya S (2024) Exploring the potential of nanotechnology in fisheries and aquaculture: opportunities and implications. J Adv Biol Biotechnol 27(3):1–17
Nasr-Eldahan S, Nabil-Adam A, Shreadah MA, Maher AM, El-Sayed Ali T (2021) A review article on nanotechnology in aquaculture sustainability as a novel tool in fish disease control. Aquacult Int 29(4):1459–1480. https://doi.org/10.1007/s10499-021-00677-7
Obeid MA, Al Qaraghuli MM, Alsaadi M, Alzahrani AR, Niwasabutra K, Ferro VA (2017) Delivering natural products and biotherapeutics to improve drug efficacy. Ther Deliv 8(11):947–956. https://doi.org/10.4155/tde-2017-0060
Odzak N, Kistler D, Sigg L (2017) Influence of daylight on the fate of silver and zinc oxide nanoparticles in natural aquatic environments. Environ Pollut 226:1–11. https://doi.org/10.1016/j.envpol.2017.04.006
Okeke ES, Chukwudozie KI, Nyaruaba R, Ita RE, Oladipo A, Ejeromedoghene O, Okoye CO (2022) Antibiotic resistance in aquaculture and aquatic organisms: a review of current nanotechnology applications for sustainable management. Environ Sci Pollut Res 29(46):69241–69274. https://doi.org/10.1007/s11356-022-22319-y
Patil MP, Kim G-D (2017) Eco-friendly approach for nanoparticles synthesis and mechanism behind antibacterial activity of silver and anticancer activity of gold nanoparticles. Appl Microbiol Biotechnol 101(1):79–92. https://doi.org/10.1007/s00253-016-8012-8
Patra JK, Das G, Fraceto LF, Campos EVR, Rodriguez-Torres MP, Acosta-Torres LS, Shin H-S (2018) Nano based drug delivery systems: recent developments and future prospects. J Nanobiotechnol 16(1):71. https://doi.org/10.1186/s12951-018-0392-8
Poland C, Larsen P, Read S, Varet J, Hankin S, Lam H (2016) Assessment if nano-enabled technologies in. Allergy 9:62
Qiao Y, Wu J, Xu Y, Fang Z, Zheng L, Cheng W, Zhao D (2017) Remediation of cadmium in soil by biochar-supported iron phosphate nanoparticles. Ecol Eng 106:515–522. https://doi.org/10.1016/j.ecoleng.2017.06.023
Quevedo AC, Lynch I, Valsami-Jones E (2021) Silver nanoparticle induced toxicity and cell death mechanisms in embryonic zebrafish cells. Nanoscale 13(12):6142–6161
Rajan MR, Rohini R (2021) Impact of different quantity of Zinc oxide nanoparticles on growth and hematology of Mrigal Cirrhinus mrigala. J Water Environ Nanotechnol 6(1):62–71
Rajesh Kumar S, Ishaq Ahmed VP, Parameswaran V, Sudhakaran R, Sarath Babu V, Sahul Hameed AS (2008) Potential use of chitosan nanoparticles for oral delivery of DNA vaccine in Asian sea bass (Lates calcarifer) to protect from Vibrio (Listonella) anguillarum. Fish Shellfish Immunol 25(1):47–56. https://doi.org/10.1016/j.fsi.2007.12.004
Rajkumar KS, Kanipandian N, Thirumurugan R (2016) Toxicity assessment on haemotology, biochemical and histopathological alterations of silver nanoparticles-exposed freshwater fish Labeo rohita. Appl Nanosci 6(1):19–29. https://doi.org/10.1007/s13204-015-0417-7
Rajkumar KS, Sivagaami P, Ramkumar A, Murugadas A, Srinivasan V, Arun S, Thirumurugan R (2022) Bio-functionalized zinc oxide nanoparticles: potential toxicity impact on freshwater fish Cyprinus carpio. Chemosphere 290:133220. https://doi.org/10.1016/j.chemosphere.2021.133220
Ramamoorthy S, Kannaiyan P, Moturi M, Devadas T, Muthuramalingam J, Natarajan L, Ponniah AG (2013) Antibacterial activity of zinc oxide nanoparticles against Vibrio harveyi. Indian J Fish 60:107–112
Ramsden J (2018) Applied nanotechnology: the conversion of research results to products. William Andrew, Norwich
Rather MA, Sharma R, Gupta S, Ferosekhan S, Ramya VL, Jadhao SB (2013) Chitosan-nanoconjugated hormone nanoparticles for sustained surge of gonadotropins and enhanced reproductive output in female fish. PLoS ONE 8(2):e57094. https://doi.org/10.1371/journal.pone.0057094
Rather MA, Bhat IA, Gireesh-Babu P, Chaudhari A, Sundaray JK, Sharma R (2016) Molecular characterization of kisspeptin gene and effect of nano–encapsulted kisspeptin-10 on reproductive maturation in Catla catla. Domest Anim Endocrinol 56:36–47. https://doi.org/10.1016/j.domaniend.2016.01.005
Rathor PK, Bhat IA, Rather MA, Gireesh-Babu P, Kumar K, Purayil SBP, Sharma R (2017) Steroidogenic acute regulatory protein (StAR) gene expression construct: development, nanodelivery and effect on reproduction in air-breathing catfish, Clarias batrachus. Int J Biol Macromol 104:1082–1090. https://doi.org/10.1016/j.ijbiomac.2017.06.104
Raza MA, Kanwal Z, Shahid A, Fatima S, Sajjad A, Riaz S, Naseem S (2021) Toxicity evaluation of arsenic nanoparticles on growth, biochemical, hematological, and physiological parameters of labeo rohita juveniles. Adv Mater Sci Eng 2021:1–10
Ren X, Zhao X, Duan X, Fang Z (2018) Enhanced bio-concentration of tris(1,3-dichloro-2-propyl) phosphate in the presence of nano-TiO2 can lead to adverse reproductive outcomes in zebrafish. Environ Pollut 233:612–622. https://doi.org/10.1016/j.envpol.2017.10.101
Rhim J-W, Park H-M, Ha C-S (2013) Bio-nanocomposites for food packaging applications. Prog Polym Sci 38(10):1629–1652. https://doi.org/10.1016/j.progpolymsci.2013.05.008
Rivas-Aravena A, Fuentes Y, Cartagena J, Brito T, Poggio V, La Torre J, Spencer E (2015) Development of a nanoparticle-based oral vaccine for Atlantic salmon against ISAV using an alphavirus replicon as adjuvant. Fish Shellfish Immunol 45(1):157–166. https://doi.org/10.1016/j.fsi.2015.03.033
Saleh M, Abdel-Baki AA, Dkhil MA, El-Matbouli M, Al-Quraishy S (2017) Antiprotozoal effects of metal nanoparticles against Ichthyophthirius multifiliis. Parasitology 144(13):1802–1810
Samim AR, Vaseem H (2023) Exposure to nickel oxide nanoparticles induces alterations in antioxidant system, metabolic enzymes and nutritional composition in muscles of Heteropneustes fossilis. Bull Environ Contam Toxicol 110(4):79. https://doi.org/10.1007/s00128-023-03714-8
Samrot AV, Angalene JLA, Roshini SM, Raji P, Stefi SM, Preethi R, Madankumar A (2019) Bioactivity and heavy metal removal using plant gum mediated green synthesized silver nanoparticles. J Cluster Sci 30(6):1599–1610. https://doi.org/10.1007/s10876-019-01602-y
Sarkar B, Mahanty A, Gupta SK, Choudhury AR, Daware A, Bhattacharjee S (2022) Nanotechnology: a next-generation tool for sustainable aquaculture. Aquaculture 546:737330
Sayed AE-DH, Mekkawy IA, Mahmoud UM, Nagiub M (2020) Histopathological and histochemical effects of silver nanoparticles on the gills and muscles of African catfish (Clarias garepinus). Scientific African 7:e00230. https://doi.org/10.1016/j.sciaf.2019.e00230
Shaalan M, Saleh M, El-Mahdy M, El-Matbouli M (2016) Recent progress in applications of nanoparticles in fish medicine:a review. Nanomed Nanotechnol Biol Med 12(3):701–710. https://doi.org/10.1016/j.nano.2015.11.005
Shaalan MI, El-Mahdy MM, Theiner S, El-Matbouli M, Saleh M (2017) In vitro assessment of the antimicrobial activity of silver and zinc oxide nanoparticles against fish pathogens. Acta Vet Scand 59(1):49. https://doi.org/10.1186/s13028-017-0317-9
Shah BR, Mraz J (2020a) Advances in nanotechnology for sustainable aquaculture and fisheries. Rev Aquac 12(2):925–942. https://doi.org/10.1111/raq.12356
Shah BR, Mraz J (2020b) Advances in nanotechnology for sustainable aquaculture and fisheries. Rev Aquac 12(2):925–942. https://doi.org/10.1111/raq.12356
Shahzad K, Khan MN, Jabeen F, Chaudhry AS, Khan MKA, Ara C, Khan MS (2022) Study of some toxicological aspects of titanium dioxide nanoparticles through oxidative stress, genotoxicity, and histopathology in tilapia. Oreochromis Mossambicus Bionanoscience 12(4):1116–1124. https://doi.org/10.1007/s12668-022-01024-7
Sibiya A, Gopi N, Jeyavani J, Mahboob S, Al-Ghanim KA, Sultana S, Vaseeharan B (2022) Comparative toxicity of silver nanoparticles and silver nitrate in freshwater fish Oreochromis mossambicus: a multi-biomarker approach. Comparat Biochem Physiol Part C Toxicol Pharmacol 259:109391. https://doi.org/10.1016/j.cbpc.2022.109391
Sonkusre P, Nanduri R, Gupta P, Cameotra SS (2014) Improved extraction of intracellular biogenic selenium nanoparticles and their specificity for cancer chemoprevention. J Nanomed Nanotechnol 5(2):1
Srinivasan V, Bhavan PS, Rajkumar G, Satgurunathan T, Muralisankar T (2016a) Effects of dietary iron oxide nanoparticles on the growth performance, biochemical constituents and physiological stress responses of the giant freshwater prawn Macrobrachium rosenbergii post-larvae. Int J Fish Aquat Stud 4(2):170–182
Srinivasan V, Bhavan PS, Rajkumar G, Satgurunathan T, Muralisankar T (2016b) Effects of dietary iron oxide nanoparticles on the growth performance, biochemical constituents and physiological stress responses of the giant freshwater prawn Macrobrachium rosenbergii post-larvae. Int J Fish Aquat Stud 4(2):170–182
Stefaniuk M, Oleszczuk P, Ok YS (2016) Review on nano zerovalent iron (nZVI): from synthesis to environmental applications. Chem Eng J 287:618–632. https://doi.org/10.1016/j.cej.2015.11.046
Subramani K, Elhissi A, Subbiah U, Ahmed W (2019) Chapter 1 - Introduction to nanotechnology. In: Subramani K, Ahmed W (eds) Nanobiomaterials in clinical dentistry, 2nd edn. Elsevier, pp 3–18
Sudhabose S, Sooryakanth B, Rajan MR (2023) Acute toxicity, hematological profile, and histopathological effects of MgO nanoparticles on gills, muscle, liver of mrigal Cirrhinus mrigala. Biol Trace Element Res 202(2):736–742. https://doi.org/10.1007/s12011-023-03704-1
Sumbayev VV, Yasinska IM, Gibbs BF (2013) Biomedical applications of gold nanoparticles. WSEAS Press, Athens, pp 342–348
Sumi N, Chitra KC (2019) Fullerene C60 nanomaterial induced oxidative imbalance in gonads of the freshwater fish, Anabas testudineus (Bloch, 1792). Aquat Toxicol 210:196–206. https://doi.org/10.1016/j.aquatox.2019.03.003
Sungur Ş, Kaya P, Koroglu M (2020) Determination of titanium dioxide nanoparticles used in various foods. Food Addit Contam Part B 13(4):260–267. https://doi.org/10.1080/19393210.2020.1769193
Swain P, Nayak SK, Sasmal A, Behera T, Barik SK, Swain SK, Jayasankar P (2014) Antimicrobial activity of metal based nanoparticles against microbes associated with diseases in aquaculture. World J Microbiol Biotechnol 30(9):2491–2502. https://doi.org/10.1007/s11274-014-1674-4
Tang W-W, Zeng G-M, Gong J-L, Liang J, Xu P, Zhang C, Huang B-B (2014) Impact of humic/fulvic acid on the removal of heavy metals from aqueous solutions using nanomaterials: a review. Sci Total Environ 468–469:1014–1027. https://doi.org/10.1016/j.scitotenv.2013.09.044
Tang Z-P, Chen C-W, Xie J (2018) Development of antimicrobial active films based on poly(vinyl alcohol) containing nano-TiO2 and its application in macrobrachium rosenbergii packaging. J Food Process Preserv 42(8):e13702. https://doi.org/10.1111/jfpp.13702
Tayel AA, Elsaied BE, Diab AM (2019a) Nanotechnology for aquaculture. In: Pudake RN, Chauhan N, Kole C (eds) Nanoscience for sustainable agriculture. Springer International Publishing, Cham, pp 479–544
Tello-Olea M, Rosales-Mendoza S, Campa-Córdova AI, Palestino G, Luna-González A, Reyes-Becerril M, Angulo C (2019) Gold nanoparticles (AuNP) exert immunostimulatory and protective effects in shrimp (Litopenaeus vannamei) against Vibrio parahaemolyticus. Fish Shellfish Immunol 84:756–767. https://doi.org/10.1016/j.fsi.2018.10.056
Temiz Ö, Kargın F (2022) Toxicological impacts on antioxidant responses, stress protein, and genotoxicity parameters of aluminum oxide nanoparticles in the liver of Oreochromis niloticus. Biol Trace Element Res 200(3):1339–1346
Tripathi DK, Tripathi A, Shweta, Singh S, Singh Y, Vishwakarma K, Chauhan DK (2017) Uptake, accumulation and toxicity of silver nanoparticle in autotrophic plants, and heterotrophic microbes: a concentric review. Front Microbiol 8:07
Velmurugan P, Iydroose M, Lee SM, Cho M, Park JH, Balachandar V, Oh BT (2014) Synthesis of silver and gold nanoparticles using cashew nut shell liquid and its antibacterial activity against fish pathogens. Indian J Microbiol 54:196–202
Vicari T, Dagostim AC, Klingelfus T, Galvan GL, Monteiro PS, da Silva Pereira L, Cestari MM (2018) Co-exposure to titanium dioxide nanoparticles (NpTiO2) and lead at environmentally relevant concentrations in the Neotropical fish species Hoplias intermedius. Toxicol Rep 5:1032–1043. https://doi.org/10.1016/j.toxrep.2018.09.001
Vijayan SR, Santhiyagu P, Singamuthu M, Kumari Ahila N, Jayaraman R, Ethiraj K (2014a) Synthesis and characterization of silver and gold nanoparticles using aqueous extract of seaweed, Turbinaria conoides and their antimicrofouling activity. Sci World J 2014:938272. https://doi.org/10.1155/2014/938272
Vijayan SR, Santhiyagu P, Singamuthu M, Kumari Ahila N, Jayaraman R, Ethiraj K (2014) Synthesis and characterization of silver and gold nanoparticles using aqueous extract of seaweed, Turbinaria conoides, and their antimicrofouling activity. Sci World J
Vinay TN, Bhat S, Gon Choudhury T, Paria A, Jung MH, Shivani Kallappa G, Jung SJ (2018) Recent advances in application of nanoparticles in fish vaccine delivery. Rev Fish Sci Aquac 26(1):29–41
Waheed N, Saeed S, Ghayur S, Nawaz U, Ahmad Z, Waheed A, Shah A (2023) Assesement of toxic impacts of greenly synthesized silver nanoparticles (AgNPs) on behavioural, hemato-biochemical and histopathological profile of mahseer fish (tor putitora). A comprehensive study. https://doi.org/10.2139/ssrn.4489934
Wang T, Long X, Liu Z, Cheng Y, Yan S (2015) Effect of copper nanoparticles and copper sulphate on oxidation stress, cell apoptosis and immune responses in the intestines of juvenile Epinephelus coioides. Fish Shellfish Immunol 44(2):674–682
Wu S (2020) The growth performance, body composition and nonspecific immunity of Tilapia (Oreochromis niloticus) affected by chitosan. Int J Biol Macromol 145:682–685. https://doi.org/10.1016/j.ijbiomac.2019.12.235
Xia IF, Cheung JS, Wu M, Wong KS, Kong HK, Zheng XT, Kwok KW (2019) Dietary chitosan-selenium nanoparticle (CTS-SeNP) enhance immunity and disease resistance in zebrafish. Fish Shellfish Immunol 87:449–459. https://doi.org/10.1016/j.fsi.2019.01.042
Yallappa S, Kaveri K, Vijaykumar K (2020) Toxic effect of green synthesized silver nanoparticles on freshwater fish tilapia, Oreochromis mossambicus (Peters)
Yin Y, Yang X, Zhou X, Wang W, Yu S, Liu J, Jiang G (2015) Water chemistry controlled aggregation and photo-transformation of silver nanoparticles in environmental waters. J Environ Sci 34:116–125. https://doi.org/10.1016/j.jes.2015.04.005
Yirsaw BD, Megharaj M, Chen Z, Naidu R (2016) Environmental application and ecological significance of nano-zero valent iron. J Environ Sci 44:88–98
Yu JC, Tang HY, Yu J, Chan HC, Zhang L, Xie Y, Wong SP (2002) Bactericidal and photocatalytic activities of TiO2 thin films prepared by sol–gel and reverse micelle methods. J Photochem Photobiol A Chem 153(1):211–219. https://doi.org/10.1016/S1010-6030(02)00275-7
Zhang J, Guo W, Li Q, Wang Z, Liu S (2018) The effects and the potential mechanism of environmental transformation of metal nanoparticles on their toxicity in organisms. Environ Sci Nano 5(11):2482–2499
Zhou X, Wang Y, Gu Q, Li W (2009) Effects of different dietary selenium sources (selenium nanoparticle and selenomethionine) on growth performance, muscle composition and glutathione peroxidase enzyme activity of crucian carp (Carassius auratus gibelio). Aquaculture 291(1–2):78–81
Zhu X, Tian S, Cai Z (2012) Toxicity assessment of iron oxide nanoparticles in zebrafish (Danio rerio) early life stages. PLoS ONE 7(9):e46286. https://doi.org/10.1371/journal.pone.0046286
Acknowledgements
None.
Funding
None.
Author information
Authors and Affiliations
Contributions
Zunaira Faiz: Conceptualization, original draft writing; Muhammad Tayyab: Original draft writing; Investigation, Shakeela Parveen: Investigation, Supervision, Review and editing; Muhammad Hussain: Original draft writing; Saba Saeed: Original draft writing, Review and editing; Zainab Riaz: Original draft writing, Urwah Ishaque: Original draft writing. Muhammad Farhan Khan revised the manuscript. All authors approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interests
Authors do not have any financial or non-financial interests that are directly or indirectly related to the work submitted for publication.
Additional information
Editorial responsibility: Maryam Shabani.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Faiz, Z., Tayyab, M., Parveen, S. et al. Nanotechnology: applications and regulatory challenges in fish culture—a review. Int. J. Environ. Sci. Technol. (2024). https://doi.org/10.1007/s13762-024-05753-y
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s13762-024-05753-y