Abstract
Applications of nanotechnology in fish cultures have participated in getting over various difficulties that hinder fish productivity. They can achieve growth performance after adding some important minerals and vitamins in the form of nano-feed supplements like selenium, zinc, iron, and vitamin C. Also, they have an important role in reproduction, and fish medicine as antimicrobial, drug delivery, nano-vaccination, and rapid disease diagnosis. Moreover, their roles in water remediation and purification, and fish packaging are documented. On the other hand, some nanoparticles exhibit toxic effects on living organisms, which return to their tiny size, high reactivity, and permeability. They can alter many physiological functions and cause cytotoxicity, DNA damage, and histopathological changes. Also, nanotechnology applications cause new secondary pollutants to be introduced into the environment that can negatively affect fish health and the surrounding living organisms. So, in spite of the promising applications of nanotechnology to fulfill high growth performance and pathogen-free fish, there are a lot of debates about the potential toxicity of nanomaterials, their reactivity with the surrounding environment, and bioaccumulation. The present review aims to elucidate and discuss various advantages and challenges of nanotechnology applications in fish cultures. Also, it points to green nanotechnology as a promising alternative to chemical ones.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
One of the great challenges facing humanity is the increase in food production to meet the ever-growing human population. Undoubtedly, fish production can lend a helping hand as a source of cheap, valuable, and easily digestible protein. In 2015, fish accounted for about 17% of the total world’s consumption of animal protein, and this percent continuous to grow annually (FAO 2018). Actually, catching fish either from natural freshwater or marine resources does not meet the tremendous and continuous increase in population, that is why there is a necessity to focus on fish cultures and to do the best effort to achieve the self-sufficiency of fish productivity. Good nutrition, control of fish pathogens, and maintaining good water quality are the most important ways to sustain aquaculture development and achieve high fish production (Assefa and Abunna 2018).
Nanotechnology has become one of the most important interdisciplinary sciences, and it integrates physics, chemistry, and biology (Subramani et al. 2019). Also, it gains public interest worldwide due to its rapidly evolving and expanding in many applications in engineering, electronics, agriculture, medicine, food industry, and environment (He et al. 2018). According to the nano-size of nano-materials, they have new unique physicochemical properties, overcome high pressure and temperature, and participate in many applications (Aruoja et al. 2009). The nanotechnology revolution is extended to include fish cultures, to participate in their growth, and to overcome their challenges (Khosravi-Katuli et al. 2017; Luis et al. 2019). Applications of nanotechnology in fish cultures include direct and indirect applications.
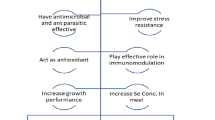
Direct applications of nanotechnology are dealing mainly with fish: growth, reproduction, and health. Nano-materials have a role in the efficient delivery of nutrients, trace elements, and vitamins as feed supplements like selenium (Se) (Ashouri et al. 2015; Khan et al. 2017; Xia et al. 2019), iron (Fe) (Akbary and Jahanbakhshi 2019; Srinivasan et al. 2016), zinc (Zn) (Awad et al. 2019; Faiz et al. 2015; Tawfik et al. 2017), and vitamin C (Alishahi et al. 2011; Jiménez-Fernández et al. 2014). Feeding of fish on nanoforms of these ingredients facilitates their absorption and passing through the intestinal wall, to enhance the growth performance, reproduction, and innate immunity of fish (Bhattacharyya et al. 2015; Chris et al. 2018). Also, using different nano-materials to control fish diseases is more effective than traditional antibiotics and chemicals that perform many side effects such as resistant bacteria strains, water pollution, and the accumulation of undesired chemical residues. Nano-materials are acting as antiviral (Ochoa-Meza et al. 2019), antibacterial (Nikapitiya et al. 2018; Raissy and Ansari 2011; Swain et al. 2014; Tello-Olea et al. 2019), antifungal (Kalatehjari et al. 2015; Shaalan et al. 2017), and anti-parasitic agents (Barakat et al. 2016; Saleh et al. 2017). Moreover, chitosan and poly lactic-glycolic acid (PLGA) nanoparticles play an important role in drug and hormone delivery, and vaccination (Bhat et al. 2019; Fenaroli et al. 2014; Rather et al. 2013). Undoubtedly, nano-vaccination has many advantages over the conventional methods, it assures sustain release, and increases stability, bioavailability, and residence time (Kitiyodom et al. 2019; Kumar et al. 2008; Rivas-Aravena et al. 2015). Furthermore, the most pronounced application of nanotechnology in fish culture is its use in rapid and effective diagnosis of different fish pathogens (Elsheshtawy et al. 2019; Saleh et al. 2011).
Indirect applications of nanotechnology in fish cultures are concerned mainly with water quality: sterilization of ponds, reducing the rate of water exchange, reducing nitrogenous compounds concentration, water treatment, and remediation of either chemical or biological contaminants (Huang et al. 2015; Khosravi-Katuli et al. 2017; Tayel et al. 2019). Several nano-materials have been applied to remove chemical pollutants from the water as metal oxide nanoparticles (Cai et al. 2019; Luo et al. 2018), carbon nanotubes (Li et al. 2003), and natural nano-adsorbents (Johari et al. 2016). Also, purification of water from various pathogens represents another desired effect of nanotechnology applications to restrict the progress of infectious diseases (Jimmy et al. 2002; Tayel et al. 2019). Furthermore, the anti-fouling effect of some nano-materials, through decreasing phosphate concentration and reduce the growth of algae and other microorganisms, is a good application of nanotechnology, to achieve good water quality in fish ponds (Ashraf and Edwin 2016).
Despite the abovementioned advantages of nanotechnology applications in fish cultures, some challenges raise several questions about their complete safety on fish health, human health, and the surrounding environment. Toxicity of nanoparticles especially metal nanoparticles can affect various fish physiological functions, antioxidant and enzyme activities, reproductive hormones, survivability of early developmental stages, and histopathological pictures (Aruoja et al. 2009; Jovanović et al. 2015; Klingelfus et al. 2019; Kumar et al. 2017; Rajkumar et al. 2016; Ren et al. 2018; Sumi and Chitra 2019; Zhu et al. 2012). Moreover, the environmental transformation of nanoparticles that is affected by many physical, chemical, or biological factors produces secondary pollutants in the environment that affect the untargeted living organisms, affect the biological diversity, and bio-accumulate within the food chain to finally affect human health (Cai et al. 2019; Gehrke et al. 2015; Mueller and Nowack 2008; Wang et al. 2016; Zhang et al. 2018).
Green nanotechnology or biosynthesis of nanoparticles represents an ecofriendly, low-cost, and effective solution to most challenges of metal nanomaterials. This includes using natural plants (Abdel-Tawwab et al. 2018), plant extracts (Kumar et al. 2014), microorganisms (Dawood et al. 2019b; Roy et al. 2019), or others like cellulose, chitosan, or tree gums (Iravani 2011). Furthermore, chitosan is a versatile natural polymer that represents a good alternative to metal nanoparticles (Abdel-Tawwab et al. 2019; Abdel-Wahhab et al. 2016; Ahmed et al. 2019; El-Naby et al. 2020; El-Naby et al. 2019).
Accordingly, there are many implications of nanotechnology applications in fish farms, and they need more efforts and studies to increase the knowledge about the possible toxicity on fish and untargeted organisms. Also, further investigations about the lowest effective dose, duration, lethal and sublethal concentrations, bioaccumulation, residues concentration, and environmental fate are required. Moreover, legislation and rules of nanomaterials synthesis and field applications should be triggered to ensure environmental safety.
The present review highlights the numerous direct and indirect applications of nanotechnology in fish cultures and discusses their possible benefits and challenges. Also, it points to the possible environmental hazard of metal nanoparticles and provides an update on the promising uses of natural alternatives for further advanced nano-applications in fish cultures.
Nanoparticles, preparation, and different forms
The prefix “nano” is a Greek word meaning dwarf and it is used to refer to the materials that have one (1–100) nm dimension and/or more in its structure. Nano-materials are adopted by the US FDA that they have new unique properties different from their bulk materials. These properties include small size, large surface area to volume ratio, and high surface activity (Martinez-Castanon et al. 2008; Moghimi et al. 2012). Two main strategies are used to prepare nanoparticles: top-down and bottom-up. The top-down strategy involves the breakdown of bulk material into the required nano-size, as in mechanical grinding, crushing, milling, and homogenization. After that, a stabilizing agent is added to ensure the persistence stability of the produced nanoparticle. The bottom-up strategy involves building up nanoparticles from atoms and molecules through crystallization, deposition, or microbial synthesis. Both strategies are used chemical, physical, or biological methods to be achieved (Mohanty et al. 2017; Shaalan et al. 2016).
There are many forms of nanoparticles: nano-spheres, which are nano-sized spherical particles used in tissue regeneration and drug delivery (Donbrow 1991; Ramalingam et al. 2013). Nano-capsules, which are composed of an outer nano-scale cover, protect an inner core that contains the drug dissolved in water or oil matrix (Torchilin 2006). Carbon nanotubes, which are tubes made of carbon with a nanometer-scale diameter, like microneedles, have a huge surface area and highly penetrate the cell membranes, so they are used in drug delivery, while some studies have reported their possible risk on blood vessels and causing of blood clotting (Gaffney et al. 2015; Reilly 2007). Dendrimers, which are three-dimensional highly branched nanostructures, are used in a gene, drug, and vaccine delivery, tissue regeneration, and antimicrobial (Gillies and Frechet 2005; Wu et al. 2015). Polymeric nanoparticles, which are polymers like chitosan, are used in delivery systems and tissue regeneration (Alishahi et al. 2014).
Application of nanotechnology in fish cultures
There are three main obstacles faced by fish cultures; poor nutrition, fish infectious diseases, and water pollution. Nanotechnology has many applications in fish cultures and can actually contribute to their development. Applications of nanotechnology in the fish cultures may be direct or indirect. Direct applications are mainly concerned with the fish: its feed, growth, reproduction, health, and disease control, while indirect applications are concerned with the water quality, wastewater treatment, remediation of chemical and biological contaminants, bio-fouling, and fish packaging (Khosravi-Katuli et al. 2017).
Direct applications of nanotechnology in fish cultures
Feed supplements
Fish in the natural environment can find their food from phytoplankton, zooplankton, and smaller animals, while in fish cultures, there is a need to choose complementary food ingredients to maintain fast and healthy growth. Fish food should mainly contain protein (32%), carbohydrate (20–35%), fat (4–6%), fiber (< 4%), and dietary energy (8.5–9.5%). However, these main requirements vary according to the species and age of fish (Robinson and Menghe 2006). Adding of minute concentrates of organic and/or inorganic materials have been used as fish feed supplements to stimulate growth and immunity. Feed supplement is one of the most important applications of nanotechnology in fish cultures. Nano-materials can be easily absorbed in low doses and passed across the gastrointestinal tract and small intestine to reach the blood and distributed into the different vital organs to do their function more efficiently than bulk materials (Bhattacharyya et al. 2015). Incorporation of some nano-metals like Se, Zn, and Fe as feed supplements may have many advantages on survivability, growth, and health of fish (Chris et al. 2018). Moreover, an organic polymer like chitosan has an important role in micronutrient delivery (Khosravi-Katuli et al. 2017).
Nano-selenium
Selenium is an essential trace element, and it is naturally found in the marine environment, and so marine fish have more Se level than fresh ones. Consequently, Se is an essential supplement that should be added to fish food especially in the case of replacement of fish meal by other plant ingredients (Sele et al. 2018). Selenium plays an important role in many physiological functions in fish like protein biosynthesis, and antioxidants and stimulates the growth and production of hormones. Nano-Se has been gained great attention in aquaculture due to its high bioavailability, strong reactivity, and lower toxicity, and it also has antioxidant defense properties, immuno-modulatory, and growth promoter effects (Sonkusre et al. 2014; Xia et al. 2019). Dietary supplementation with 0.5 and 2.5 mg/kg doses of nano-Se showed a significant increase in Nile tilapia (Oreochromis niloticus) growth rate (Deng and Chen 2003). Also, feeding the crucian carp (Carassius auratus) with nano-Se increased growth, muscle protein, and some antioxidant enzymes (Wang et al. 2013; Zhou et al. 2009). Dietary supplementation of common carp (Cyprinus carpio) with different concentrations of nano-Se showed an increase in growth performance parameters. Moreover, it caused higher Se contents in the muscle and liver, increase total protein, globulin, and antioxidant activities (Ashouri et al. 2015). Also, dietary supplementation of mahseer fish (Tor putitora) with nano-Se revealed improvement in weight gain, the specific growth rate, and feed conversion efficiency at a concentration up to 0.68 mg nano-Se/kg fish feed, compared to fish feed on a basal diet (Khan et al. 2017). Recently, dietary supplementation with nano-Se (1–2 mg/Kg diet) for 45 days revealed enhancement of immune response, and increased total protein and antioxidant activities in sea bream (Pagrus major) (Dawood et al. 2019a).
Nano-zinc
Zinc is another important essential element, and it is necessary for many metabolic pathways, protein synthesis, energy consumption, and animal growth. Also, Zn was used as an antimicrobial agent and improves both immune and reproductive systems, and its deficiency leads to many function disorders (Muralisankar et al. 2014). Dietary supplementation of grass carp (Ctepharyngodon idella) with nano-ZnO (optimal concentration was 30 mg/Kg fish feed) revealed an improvement in growth, immune response, and hematological parameters when compared with bulk ZnO and ZnSO4 (Faiz et al. 2015). Also, there was a significant increase in the final weight, protein content, and antioxidant activities of prawn: Macrobrachium rosenbergii (M. rosenbergii) after fed a diet supplemented with nano-ZnO for 3 months (Muralisankar et al. 2014). Recently, dietary supplementation of O. niloticus with nanoparticles of ZnO revealed the highest survival rate, increase in total protein, lysozyme, and total antioxidant capacity as well as upregulation in interleukins genes (IL 8, IL 1) at 30 mg/kg diet, while the higher dose of 60 mg/Kg diet caused impairment in immune and antioxidant systems and increased the inflammatory response. Therefore, using low concentrations of nano ZnO in fish farms was recommended to improve fish health (Awad et al. 2019).
Nano-iron
Iron compensates in many physiological processes, especially oxygen transport, respiration, and lipid oxidation, and enhances the immune system against any infection. Dietary requirements of Fe are naturally difficult to be achieved through bulk Fe sources according to the lower solubility and bioavailability of Fe. Dietary supplementation of freshwater prawn (M. rosenbergii) with nano-Fe2O3 at a concentration of 10–20 mg nano-Fe2O3/Kg diet revealed significant improvement in survivability, growth, activities of the digestive enzymes, and biochemical and hematological parameters of fish compared to the basal diet (Srinivasan et al. 2016). Also, feeding of juvenile goldfish (C. auratus) on nano-Fe2O3 for 2 months positively affected the physiology of fish at a concentration of 0.5 g/kg diet. It caused an increase in the specific growth rate, alkaline phosphatase activity, and upregulated the growth factor genes (Akbary and Jahanbakhshi 2019). On the other hand, higher doses of nano-Fe2O3 (30–50 mg/kg diet) caused various negative stress responses in freshwater prawns (Srinivasan et al. 2016). Also, O. mossambicus showed negative hematological and physiological changes, and oxidative stress after exposure to 0.5, 5, and 10 μg/ml of biologically synthesized α-nano-Fe2O3 (Karthikeyeni et al. 2013). Similarly, negative changes were noticed in the major carp; Labeo rohita (L. rohita) after 25 days of exposure to 500 mg nano-Fe2O3/l (Remya et al. 2015).
Nano-chitosan
Chitosan is a polysaccharide, which can be obtained from the chitin of shrimp or other crustacean shells after treatment with an alkaline substance. Many researchers have studied chitosan as a carrier for many vitamins or essential elements and for growth, and immunity stimulation in fish. Feeding of rainbow trout (Oncorhynchus mykiss) with nano-chitosan for 20 days showed an increase in vitamin C absorption (Alishahi et al. 2014). Also, Jiménez-Fernández et al. (2014) used nano-chitosan to deliver ascorbic acid to zebrafish. This may be returned to the high ability of nano-chitosan to penetrate the intestinal wall of the digestive system, which consequently increases the ascorbic acid level in fish. Also, nano-chitosan can be used in encapsulation of the fine nutrient ingredients, to prevent their loss in the water (Ji et al. 2015). Moreover, its role in modulating the effect of some mycotoxins in catfish was estimated by Abdel-Wahhab et al. (2016), since oral exposure to nano-chitosan activated various fish antioxidants when accompanied by gallic acid. Recently, enhancement of growth and immune response of O. niloticus after exposure to chitosan as a natural and safe nanomaterial were ensured (Abdel-Tawwab et al. 2019; El-Naby et al. 2020; El-Naby et al. 2019). However, the optimum dose that increased the O. niloticus growth performance was a 1.0 g/kg diet for 45 days as estimated by Abdel-Tawwab et al. (2019).
Fish reproduction
Production of monosex tilapia is a popular aim in Oreochromis sp. fish cultures to avoid the un-controlled spawning, pond overcrowding, and production of tiny fish. Controlling of fish reproduction can be achieved through the application of steroid hormones, which has an undesired effect in-between fish consumers (El-Greisy and El-Gamal 2012). Nanotechnology has participated, like a good drug carrier in the delivery of fadrozole (inhibitor of estrogen synthesis) to O. niloticus. Feeding with fadrozole overloaded on PLGA nanoparticles at 50–500 ppm for 1 month caused 100% male at both 350 and 500 ppm (Joshi et al. 2019). Nanotechnology has an important role in enhancement reproduction in other fish species. Particularly, nano-chitosan has been used to carry and release reproductive hormones. A composite of chitosan-nano-gold prolonged the presence of reproductive hormones in salmon blood and increased the fertilization rate of eggs (Rather et al. 2013). Furthermore, nanoparticles of eurycomanone (extracted from Eurycoma longifolia plant, and had formerly achieved desired sexual prowess and fertility in animals) were combined with chitosan nanoparticles and injected into Clarias magur fish. After 7 days, there was an increase in the gonado-somatic index, concentrations of Ca and Se, reproductive capacity, and gene expression levels of endocrine hormones (Bhat et al. 2019).
Fish medicine
Fish cultures are subjected to many pathogens (virus, bacteria, fungi, and parasites) that lead to huge economic losses. The conventional strategies for fish treatment include using antibiotics and chemicals that caused many side effects as the production of antibiotic-resistant bacteria, water pollution, and chemical residues in fish tissues.
Antibiotic-resistant bacteria in fish cultures
Antibiotics are used all over the time to control microbial infections. The excessive and unregulated use of antibiotics can lead to the production of new bacteria-resistant strain that is not sensitive to antibiotics. Some previous studies have reported that the tetracycline antibiotic is the most used in fish farms to control different microbial infections (Tuševljak et al. 2013). For example, Aeromonas hydrophila (A. hydrophila) isolated from tilapia fish were found to be resistant to many antibiotics: tetracycline, erythromycin, and streptomycin. Also, other species of Pseudomonas, Vibrio, Edwardsiella, and Listeria have shown antibiotic resistance (Atyah et al. 2010; Sørum 2008; Tuševljak et al. 2013). With the progress of nanotechnology in many fields, it inevitably spilled to intensive aquaculture to overcome the bacterial antibiotic resistance and participate in rapid detection and control of fish microbial diseases (Julio et al. 2018). Nano-medicine is the science that using nanoparticles and/or nano-devices for biomedical applications (Cavalieri et al. 2014). Nanoparticles have different roles in fish medicine, and they act as antimicrobial agents, drug and vaccine carriers, and in the diagnosis of fish pathogens.
Antimicrobial
Some nanomaterials have been reported to have high antimicrobial activity against viral, fungal, bacterial, and protozoa infections in aquatic animals (Swain et al. 2014). The most used antimicrobial nano-materials are the metal and metal oxides: silver (Ag), gold (Au), zinc oxide (ZnO), copper (Cu), and titanium oxide (TiO2).
Silver nanoparticles
Nano-Ag is the most reported nano-antibacterial agent. Treatment of juvenile shrimp Feneropenaeus indicus infected by Vibrio harveyi (V. harveyi) with 10 μg/kg diet nano-Ag for 5 days revealed a decrease in mortality by 71% (Vaseeharan et al. 2010). Also, nano-Ag proved to have in vitro antimicrobial effect in 24 h, against Lactococcus garvieae and Streptococcus iniae isolated from diseased rainbow trout, the minimum inhibitory nano-Ag concentrations were 1.12–5.0 and 1.12–2.5 μg/ml for L. garvieae and S. iniae, respectively (Raissy and Ansari 2011). Moreover, 10 ng/ml of nano-Ag showed in vitro and in vivo anti-parasitic effect against Ichthyophthirius multifiliis infection in rainbow trout (Oncoryhnchus mykiss). It caused 50% mortality of I. multifiliis theronts in half an hour, and 100% mortality in 2 h (Saleh et al. 2017). Furthermore, Ochoa-Meza et al. (2019) reported that only one dose of nano-Ag (12 ng/ml for 4 days) was capable to enhance the immunity of shrimp (Penaeus vannamei) against the white spot syndrome virus disease without the appearance of any toxic effects within healthy shrimps.
Although the application of nano-Ag may have some toxic effects, the biological synthetized nano-Ag showed better results in fish medicine and without physiological changes or toxic effects. For example, nano-Ag synthesized by the natural extract of the tea plant Camellia sinensis (Vaseeharan et al. 2010) or by Bacillus subtilis (B. subtilis) bacteria (Sivaramasamy et al. 2016). Also, the immersion of goldfish (C. auratus) in a bath contained 10 ng of nano-Ag encapsulated with starch for 20 min revealed anti-parasitic and antifungal effects against white spot and red spot diseases, respectively, without side effects (Daniel et al. 2016). Nanocomposites of chitosan and Ag are another cheap and effective nano-materials that prevent diseases in fish, the positively charged chitosan can adhere the negatively charged bacterial cells, while Ag nanoparticles make pores on their wall, causing rapid alteration of membranes and cell death. Also, composite of nano-Ag and chitosan had no toxic effects on both zebrafish and rock bream fish (Dananjaya et al. 2016; López-Carballo et al. 2012). Recently, Fatima et al. (2019) reported that nano-Ag particles that biologically synthesized by using Red algae (Portieria hornemannii) have a high antibacterial effect against four strains of fish pathogens (V. harveyi, V. anguillarum, V. vulnificus, and V. parahaemolyticus).
Gold nanoparticles
Nano-Au may alter the biological functions of the microbial cells after interact with proteins and lipopolysaccharides (Sumbayev et al. 2013). Biological preparation of nano-Au by using cashew nut shell liquid showed an antibacterial effect against A. bestiarum, and P. fluorescens in vitro (Velmurugan et al. 2014). Also, oral administration with 0.2, 2, and 20 μg nano Au/g diet in shrimp (Litopenaeus vannamei) for 6 days revealed upregulation in immune genes, the high survival rate after bacterial challenged with V. parahaemolyticus, and no hepato-pancreas toxicity was recorded at a dose of 2 μg/g (Tello-Olea et al. 2019).
Zinc oxide nanoparticles
Nano-ZnO has both antibacterial and antifungal effects. Previous studies have reported the antibacterial role of nano-ZnO in fish culture against many bacterial species; V. harveyi, A. hydrophila, F. branchiophilum, E. tarda, S. aureus, and P. aeruginosa (Gunalan et al. 2012; Ramamoorthy et al. 2013; Swain et al. 2014). Also, nano-ZnO had an antifungal effect against Aphanomyces invadans, the main cause of red spot disease in fish at 3.15 μg/ml for 24 h (Shaalan et al. 2017).
Titanium dioxide nanoparticles
Nano-TiO2 are effective photo-catalyst nanoparticles that have the ability to kill many microorganisms. They have an antibacterial effect against E. tarda, S. iniae, and Photobacterium damselae infections in fish after activation by light (Cheng et al. 2009; Cheng et al. 2011). Also, fish pathogens can actively bind to a composite composed of nano-TiO2 and magnetic nano-Fe3O4, then extracted from water by using a regular magnet (Cheng et al. 2009). Moreover, they have an antibacterial role against E. coli (Alhadrami and Al-Hazmi 2017).
Copper nanoparticles
Nano-Cu has been proved to have in vitro antifungal effect against Saprolegnia sp., isolated from eggs of the Caspian white fish (Rutilus kutum), as an alternative to the toxic and carcinogenic malachite green. This antifungal effect depended on nano-Cu concentration and duration of exposure, and the minimum effective concentration was 10 ppm for 3 days (Kalatehjari et al. 2015).
Mechanism of action
Metal nanoparticles have multiple mechanisms to do their antimicrobial role and to avoid the possible bacterial resistance (Knetsch and Koole 2011; Lara et al. 2010; Prakash et al. 2013). Most of these mechanisms are achieved after releasing metal ions (Fig. 1), such as the production of reactive oxygen species that stimulate the activity of antioxidant enzymes (Matsumura et al. 2003). Interaction with DNA (phosphorus-containing structures), which prevents its replication and inhibits bacterial cell division (Rai et al. 2009). Interaction with the thiol group (an organosulphur compound R-SH) that is included in many enzymes and the protein of the cell wall, causing inactivation of them (Lara et al. 2010). Destabilize and disrupt the protein of the bacterial outer cell membrane, breakdown its potential, and minimize the intracellular ATP concentration (Lok et al. 2006). Also, the membrane damage causes leaking of cytoplasmic contents (Liu et al. 2009b). Penetrate the thick wall of bacterial cells, aggregate inside it, and disrupt the cell function (Mohamed et al. 2017).
Mechanisms of action of silver nanoparticles for antimicrobial effect (Roy et al. 2019)
Furthermore, other non-metallic nanoparticles can be used as antibacterial in fish, for example, chitosan nanoparticles (5 μg/ml) had effective immune-modulatory characteristics, and caused a strong antibacterial effect against A. hydrophila infection in zebrafish larvae (Nikapitiya et al. 2018). Also, feeding O. niloticus with chitosan nanoparticles (1.0 g/kg fish diet) for 21 days before bacterial challenge with A. sobria, A. hydrophila, and S. agalactiae, increased survivability of the infected fish (Abdel-Razek 2019).
Nanoparticles as drug and hormone delivery vehicles
Nanoparticles can easily pass through biological barriers like the blood-brain barrier due to their small sizes. They also possess high reactivity due to their high surface area (De Jong and Borm 2008; Lockman et al. 2002; Wang et al. 2011). Nanoparticles of chitosan and PLGA are the most used nanoparticles for drug delivery in fish medicine.
Chitosan nanoparticles
Chitosan is a non-toxic, easily excreted, and biodegradable polymer (De Jong &Borm 2008). It achieves slow and sustainable drug release because of its muco-adhesive properties (Costa et al. 2015). Vitamin C was conjugated with nano-chitosan to ensure its delivery to rainbow trout (O. mykiss), and the results indicated its continuous release up for 2 days after oral administration, also an immune system stimulation was reported (Alishahi et al. 2011). Moreover, there was an increase in the egg fertilization rates after injection of C. carpio fish with luteinizing hormone (only one dose) conjugated with nano-chitosan, and this was explained by the sustained release of a hormone (Rather et al. 2013).
PLGA (poly lactic-glycolic acid) nanoparticles
PLGA is a co-polymer composed of poly lactic acid and poly glycolic acid. It was approved as biodegradable and non-toxic material by the Food and Drug Administration (Lü et al. 2009; Makadia and Siegel 2011). Injection of zebrafish embryos with PLGA nanoparticles loaded with an anti-mycobacterial agent (rifampicin) showed an increase in the therapeutic effect against Mycobacterium marinum infection. Also, there was an increase in the embryo survival rate compared to injection with rifampicin alone (Fenaroli et al. 2014).
Vaccination
Production of effective vaccines and delivery systems to control infectious diseases is an important goal to sustain fish culture. Generally, there are three routes of vaccination in fish: injection, immersion, and oral. The oral encapsulation vaccine is the most recommended because it prevents the escaping of antigens from the food pellets and protecting the antigen against the acidic stomach; also it reduces fish stress and is suitable for mass immunization (Vinay et al. 2018). Application of polymeric nanoparticles in fish cultures as adjuvants or vaccine releasing vehicles have many advantages (Myhr and Myskja 2011; Soliman et al. 2019; Vinay et al. 2018; Zhao et al. 2014): act as efficient adjuvants instead of carcinogenic chemical ones, grasped by endocytosis that ensures its cellular uptake, perform vaccine stability against enzyme degradation, increase bioavailability and residence time, maintaining the immunogenicity and sustainable release of vaccine. Many degradable and safe polymeric nanoparticles are used in fish vaccine delivery, chitosan and PLGA are again the most appropriate of them, and they either carry dead vaccine (inactivated antigen or recombinant DNA) or live vaccine (attenuated, gene or DNA). An inactivated nano-vaccine against Flavobacterium columnare, that infected red tilapia, was prepared from formalin-killed bacteria by sonication and homogenization before coated with muco-adhesive chitosan to ensure the delivery and binding of nano-vaccine to fish mucosal membrane. Vaccination was carried by immersion in 1 ml/l of nano-vaccine emulsion for 30 min before bacterial challenged. The vaccinated fish showed more than 60% survivability compared to 100% mortality in non-vaccinated ones (Kitiyodom et al. 2019). Also, oral administration of Atlantic salmon (Salmo salar) with nano-chitosan overloaded with inactivated infectious salmon anemia virus (ISAV) showed a 77% protection rate against the virus infection (Rivas-Aravena et al. 2015). Moreover, oral administration of nano-chitosan encapsulated DNA or overloaded with either DNA or recombinant DNA vaccine positively stimulated the immune response against some fish pathogens, for example, the European sea bass (Dicentrarchus labrax) against nodavirus (Valero et al. 2016), Asian sea bass (Lates calcarifer) against V. anguillarum (Kumar et al. 2008), black seabream (Acanthopagrus schlegelii) against V. parahemolyticus (Li et al. 2013), and Shrimp (P. monodon) against the white spot syndrome virus (Rajeshkumar et al. 2009). In the same manner, nano-PLGA was used in oral DNA vaccines of Japanese flounder (Paralichthys olivaceus) against lymphocytic disease virus (Tian et al. 2008) and of rainbow trout (O. mykiss) against infectious hematopoietic necrosis virus (Adomako et al. 2012). Accordingly, the application of nanotechnology in the vaccination of fish plays an important role in limiting and/or stopping the use of toxic and carcinogenic chemical adjuvants, besides using oral vaccination or immersion ones are more preferable than injection to avoid stress (Rivas-Aravena et al. 2015).
Diagnosis and nano-sensors
Rapid diagnosis and control of different pathogens are very important in fish cultures to avoid economic losses and to sustain high fish productivity. Nanotechnology can be applied in the rapid, specific, and sensitive diagnosis of low concentrations of pathogens including viruses, bacteria, and different parasites. Nano-Au is the most widely used nano-material in pathogen diagnosis. Saleh et al. (2011) coated nano-Au with specific antibodies of A. salmonicida and could detect the presence of bacterial infection in fish tissues in about 45 min. It is worth to mention that, the antibodies that coated nano-Au were stable at 4 °C for about 60 days. Also, Saleh et al. (2012) developed a highly specific and rapid colorimetric assay, by using the virus probe and Au nanoparticles, to detect the spring viremia virus of carp without amplification of the viral nucleic acids. In the same way, Elsheshtawy et al. (2019) developed a rapid and sensitive assay to detect A. hydrophila infection in fish, and this assay can detect the bacterial DNA without prior PCR amplification. They conjugated a specific oligonucleotide probe, which is based on a certainly known gene of A. hydrophila, with Au nanoparticles. This assay was visually detected A. hydrophila in only half an hour and gave negative results with other bacterial strains.
Nano-sensors are nano-devices, based in their structures on different nano-materials, such as carbon nanotubes, nano-Ag, and nano-Au, and they have been applied as labels in the biological assays. Nano-biosensors are portable, highly sensitive, and specific on-site tools for fish pathogens diagnosis (Amaro et al. 2012; Chunglok et al. 2011). Gold nanoparticles are the most used because of their numerous characteristics like high surface area, high surface immobilization, and easy detection through various analytical techniques and so improve the signal response and the DNA biosensors sensitivity (Cui et al. 2012; Jain 2003; Lima et al. 2013). Kuan et al. (2013) investigated the fungal (A. invadans) infection of fish by using nano-Au-based DNA biosensors, in which the electro-chemical genosensor was coated by nano-Au. Similarly, yellow head virus in shrimp (Jaroenram et al. 2012) and white spot syndrome virus in shrimp (Seetang-Nun et al. 2013) were detected.
Indirect applications of nanotechnology in fish cultures
Actually poor water quality in fish cultures leads to many fish disorders that finally negatively affect the fish culture productivity. Fish cultures are suffering from several types of pollutants including the ordinary pollutants found in the natural water and wastewater (heavy metals, pesticides, ammonia, and phosphate), the specific pollutants of the culture (food residues, excretory products, and feces), the chemical disinfectants (hydrogen peroxide and malachite green), antibiotics residues (tetracycline and sulfonamides), and anthelmintic (Julio et al. 2018; Khosravi-Katuli et al. 2017). The ordinary and conventional used way to improve the water quality was to renew the water and continuously changed it, which is hardly achieved in respect to the present water shortage. Nanotechnology can be applied in fish cultures to detect and remove various toxic pollutants and refine the water to sustain the imperative good water quality for healthy fish (Gehrke et al. 2015). Various nanoparticles can be applied to remediate chemical and biological pollutants, whether inside the fish cultures or in the treatment of its effluents to sustain the safe environment around it. Nanoparticles may be used individually or incorporated in filter membranes (Pradeep 2009).
Remediation of chemical contaminants
Due to the unique properties of nano-materials, their large surface area, and the presence of more sorption sites, they have an excellent capacity of immobilizing and adsorbing metals, which allowed to use in the remediation of contaminated water and sediments. Many nanomaterials have been applied to remediate heavy metals such as metal oxide nanoparticles, nano zero-valent iron, carbon nanotubes, and natural adsorbents (Cai et al. 2019).
Metal oxide nanoparticles (MON)
They include nanoparticles of Fe2O3, Al2O3, MnO, MgO, and TiO2, which have high adsorption affinity for different heavy metals from an aqueous environment. Also, MON is used as a catalyst for degrading various non-degradable pesticides as polychlorinated biphenyls (PCBs) and organochlorine pesticides (Zhang et al. 1998). Nano-TiO2 is the most used photo-catalyst nanomaterial in water and wastewater treatments, and it has low toxicity, low cost, high chemical stability, and raw material abundance. A previous study which compared the toxic effects of TiO2, Cu, Al, Co, Ni, and Ag on some aquatic organisms revealed that TiO2 was the only one that had no toxic effects (Griffitt et al. 2008), while photo-catalysis is an oxidation process that enhanced the bio-degradability of most persistent toxic pollutants (Tayel et al. 2019). Moreover, TiO2 has high adsorption capacity and precipitates in the sediment causing changes in the mobility, accumulation as well as decrease the toxicity of many pollutants (Li et al. 2018b; Luo et al. 2018).
Nano zero-valent iron (n ZVI)
It is consisting of metallic iron (electron donor core) and iron oxide layer (electron acceptor shell) that permits the metal adsorption (Yirsaw et al. 2016). Nano-ZVI actively decreased the availability and mobility of various heavy metals and so decreased their toxic effects in the surrounding environment as in the case of Pb and Zn (Fajardo et al. 2012), Cr (Su et al. 2016) and Cu, Pb, Cd, and Cr (Chen et al. 2016).
Carbon nano-tubes
They are composed of nano-graphene sheets that are rolled up like a tube, and they represent promising materials for removing and adsorption different organic and inorganic contaminants (especially heavy metals) (Dai 2002; Gong et al. 2009). The adsorption mechanism depends on the surface area, functional groups, adsorption sites, porosity, and purity of the carbon nano-tubes (Ren et al. 2011). The mechanisms of metal absorption include many chemical and physical operations such as electrostatic attraction, precipitation, physical adsorption, and chemical interaction between the metal and the functional groups of carbon nano-tubes (Li et al. 2003; Ren et al. 2011).
Magnetic engineered NPs
Application of nano-materials to purify water and wastewater may have dangerous effects on the living organisms. Accordingly, removing nano-materials from the ponds after doing its remediation effect represents an imperative goal, and this can be achieved by producing magnetic engineered nanoparticles that are composed of magnetic core covered by actively adsorbent materiel like alumina and TiO2 (Chiavola et al. 2017). A nano-compound of Fe and manganese oxide represented an effective sorbent material for the removal of some metals like arsenic from the water of fish cultures (Bhattacharyya et al. 2015).
Natural adsorbents
Clays such as bentonite, kaolinite, and montmorillonite have a high ability to remove heavy metals, some studies combined clays and chitosan nanoparticles in a composite to increase its affinity for adsorption of heavy metals (Futalan et al. 2011; Pandey and Mishra 2011). Also, chitosan-magnetite nano-composites were used for heavy metal removal from an aqueous solution (Fan et al. 2017; Namdeo and Bajpai 2008). Zeolite is another natural microporous crystalline hydrated aluminosilicate stone, which has unique adsorption–desorption properties. It has various applications in fish culture, the most important of them is its ability to remove ammonia and heavy metal contaminants from the water (Ghasemi et al. 2018). Some nano metals, like nano-Ag, can be attracted to zeolite pores and reacted in a synergetic effect on reducing ammonia and nitrate concentrations in fish farms (Johari et al. 2016). Also, some nano-materials of activated carbon or alumina can be combined with zeolite and Fe to be used in aquaculture for removing ammonia, nitrates, and nitrites. Moreover, the nano-Fe powder was used as an effective nanoparticle in converting some chemical pollutants like carbon tetrachloride, polychlorinated biphenyls, and dioxins to simpler and less toxic carbon compounds (Rather et al. 2011). Apatite is a phosphate mineral, nano-composites that contain apatite mineral—whether natural or synthetic—and can decrease the mobility and bioavailability of various metals in water and sediment (Mohammad et al. 2017; Qian et al. 2009). Recently, Abdou et al. (2018) have proved that adding nano-Ag to some agriculture by-products (rice husk and sawdust) improved their absorbance efficiency. The study was performed on private fish culture and the results revealed that the use of nano-Ag composites with natural adsorbents improved water quality parameters (pH, alkalinity, electrical conductivity, nitrite, nitrate, ammonia, and total hardness) and removed heavy metal (Pb and Cd) from pond's water.
Remediation of biological contaminants
Sterilization and disinfection of water
The proliferation of water pathogens in fish cultures is one of the known problems due to the accumulation of food residues and the presence of a huge number of organisms. These pathogens include viruses, fungi, bacteria, and protozoa, and propagate to cause many fish infectious diseases. Ordinary conventional chemicals that are used as anti-pathogens may result in many risks on the pathogen resistance, users’ health, aquatic animals, and the environment (Twiddy et al. 1995). Applications of nanotechnology in water sterilization and disinfection can be considered as a promising solution to those problems (Tayel et al. 2019). Application of 0.1 g/l nano-TiO2 for 2 h could achieve about 98% sterilization on A. hydrophila, V. anguillarum, and E. coli in UV or sunlight (Huang et al. 2010). Other nano-materials such as Ag, ZnO, and a composite of nano-montmorillonite and Cu showed bactericidal effects (Liu et al. 2009a; Mühling et al. 2009; Pati et al. 2014). Using nano-Ag coated zeolite, as a water filter, reduced Saprolegnia infection in the fertilized eggs of rainbow trout, while adding of activated carbon increase the egg survival rate more than nano-Ag coated filters (Johari et al. 2016). The sterilization efficiency is depended on the pollutant type and its concentration, nano-material concentration, temperature, dissolved oxygen, pH, and light intensity (Chong et al. 2010). Recently, Hamad (2019) used chitosan-Ag nanoparticles (2 g/l) for 3 h to improve water quality collected from El-Gharbia drain to be suitable for fish farming or irrigation. He showed that the total count of P. aeruginosa, coliform, fecal streptococci, fecal coliform, and S. aureus was decreased from 5 × 103, 198 × 105, 4 × 103, 84 × 105, and 16 × 103 to zero CFU/ml, respectively.
Bio-fouling control
Bio-fouling is another challenge faced by fish cultures. It is the accumulation of microorganisms, algae, and plants on wet surfaces. Bio-fouling resulted in a reduction in water flow, oxygen level, and food availability, while it causes weight increase, corrosion, and distortion of mariculture cages (Champ 2003). Application of some nanomaterials (nano-ZnO and nano-CuO) in fish cultures can participate in getting rid of fouling organisms. Antifouling nanoparticles serve as barriers to fouling constituents due to their unique high surface area (Rather et al. 2011). A significant reduction in bio-fouling after application of nano-CuO for 3 months was reported by Ashraf and Edwin (2016). Lanthanides (La) oxides nanoparticles are another anti-fouling, and they can absorb phosphate from the surrounding water and hence reduce algal and other microorganisms growth (Ashraf et al. 2011; Gerber et al. 2012). NanoCheck is one of the commercial products that deal with the management of fish cultures; it is based on 40 nm nano La in its structure that can absorb phosphate from the water and consequently reduce algal formation (Ashraf et al. 2011). Also, nano-Ag has the ability to control bio-fouling through its antibacterial effect (Hassan and El-latif 2018; Vijayan et al. 2014).
Fish packaging
Finally, nanotechnology can also have a role in fish packaging and marketing by delaying microbial and enzymatic spoilage to extend the product shelf-life. Several studies have reported that the incorporation of nano-materials in the packaging of biodegradable food has many advantages: they act as antimicrobial and antifungal agents, eliminate oxygen, prevent biological degradation, and immobilize enzymes to increase the stability of the final products (Jiang et al. 2015; Kuswandi 2017; Rhim et al. 2013). Also, some nano-composites that are obtained from natural biopolymers like proteins, lipids, or polysaccharides are accepted as healthy packaging materials instead of toxic petro-chemical based plastics (Can et al. 2011).
Challenges of nanotechnology applications in fish culture
Many advantages can be achieved, as mentioned before, by introducing nano-materials in fish cultures, whether as a feed supplement, antimicrobial, drug, vaccine, or in water treatment and remediation of different pollutants. The most common nanoparticles used in fish cultures and their advantages were tabulated in Table 1. However, the application of nanotechnology in fish cultures is faced with many challenges (Gregory et al. 2013; Mishra et al. 2019), that can be summarized in the following aspects:
Toxicity
Nano-material toxicity is returned to its high reactivity and new physical and chemical properties which increased the production of reactive oxygen species. Nanoparticles easily penetrate cell membranes due to their unique small size and react with the intracellular organelles causing many dysfunctions (Kumar et al. 2017). Generally, comparing the physiological effects of metals and nano metals proved that nano metals caused more toxic effects (Shaw and Handy 2011). Some toxic doses of both nano metals and bulk forms and their effects on various aquatic organisms are tabulated in Table 2. It is worth to mention that metal nanoparticles are the most toxic particles to living organisms than other nanoparticles. For example, exposure of Medaka fish (Oryzias latipes) to nano-Se (3.2 mg/l) for only 2 days caused 100% mortality, while exposure to sodium selenite caused only 10% mortality at 2.0 mg/l and 80% mortality at 8.0 mg/l. Also, Nano-Se revealed a 6-fold increase of accumulation in fish liver than selenite (Li et al. 2008). Some nano-materials that are used in water remediation, like nZVI, were reported to cause toxicity to microorganisms, crustaceans, fish larvae, and other aquatic and soil living organisms (Stefaniuk et al. 2016). Also, Li et al. (2018b) have reported that the co-exposure of zebrafish to nanoparticles of TiO2 and cypermethrin insecticide caused an increase in the accumulation of cypermethrin, and consequently its neurotoxic effects on fish and decreased the locomotion of fish larvae. Similarly, nano-TiO2 increased the up-take, adsorption, and bio-concentration of an organophosphate compound inside tissues of adults of zebrafish that causing reduction in reproductive hormones as testosterone, estradiol, follicle-stimulating hormone, and luteinizing hormone, and so inhibited the reproduction process (Ren et al. 2018). This may point to the possible risk of interaction between newly introduced nano-materials and the presence of organic toxicants in the external environment. Furthermore, some literature reported the toxic effect of nano TiO2-NPs injection (2 ng/g and 10 μg/g fish) of fathead minnow fish (Pimephales promelas), and it inhibited the fish immune system and increased its mortality rate during the bacterial challenge by A. hydrophila and Edwardsiella ictaluri (Jovanović et al. 2011; Jovanović et al. 2015). Other toxic effects of different nanoparticles on various fish species are tabulated in Table 3.
Mechanisms of nano-metal uptake and cytotoxicity
The uptake of nano-metals is affected mainly by the specific properties of each nano-metal. Diffusion, phagocytosis, and endocytosis are the main known strategies to enter the cell. Inside the cell, metal ions are aggregated, induce antioxidants and cytokines, bind to DNA causing its damage, mitochondria dysfunction, impair cell proliferative, and finally cause cell death (Aruoja et al. 2009).
Green nanotechnology
The recent fast-growing research dealing with the green synthesis of metal nanoparticles (green nanotechnology) may act as a solution to toxicity problems of metal nanoparticles and can minimize their adverse effects on living organisms. It involves using natural sources, which are not toxic, biodegradable, and biocompatible materials such as plant extracts, bacteria, fungi, cellulose, dextran, chitosan, or tree gums (Akter et al. 2018; Cinelli et al. 2015; Iravani 2011; Vinod et al. 2011). Green synthesis of metal nanoparticles is a very cost-effective approach since it can be used as an ecofriendly, economic, and valuable alternative for the traditional chemical and physical synthesis. It was the first time in 2002 when Ag and Au-nanoparticles were biologically synthesized inside alfalfa plants. Actually, the alfalfa plant was grown in soil enriched with AuCl4 and the complete follow up of the metal uptake and nanoparticle formation was shown by the X-ray absorption spectroscopy and transmission electron microscopy (Gardea-Torresdey et al. 2002; Gardea-Torresdey et al. 2003). Later on, Kumar et al. (2014) biosynthesized Ag nanoparticles by using the plant extract of Boerhaavia diffusa, as a reducing agent, and they reported its antibacterial effect against some fish bacterial strains: P. fluorescens, A. hydrophila, and F. branchiophilum. Furthermore, Ishwarya et al. (2018) biosynthesized nanoparticles of ZnO by using the marine seaweed Sargassum wightii and studied its effect on the green tiger shrimp (P. semisulcatus); they reported a high anti-biofilm activity against both Gram-positive (B. subtilis, S. aureus) and Gram-negative bacteria (Shigella sonnei, P. aeruginosa). Recently, a fermentation technology to produce nano-Se from lactic acid bacteria was achieved and evaluated as a fish feed supplement. Dietary supplementation of O. niloticus on bio-synthesized nano-Se at a concentration of 1–2 mg/Kg diet for 8 weeks, caused an increase in growth, antioxidant activities, total protein, lysozyme, phagocytic activities, and upregulated IL 1-beta gene expression (Dawood et al. 2019b).
Although using plant extracts and microorganisms represent the most effective biosynthesis approach of nanoparticles (Poguberović et al. 2016; Shaik et al. 2013), other natural sources can be used to produce nanoparticles. Fish gelatin nanoparticles can be produced from tilapia skin and act as a drug carrier (Subara et al. 2018). Also, hemocyanin can be extracted from shrimp hemolymph to produce ZnO nanoparticles (Ishwarya et al. 2019). Moreover, some rare studies were dealing with the nanoparticles of some raw natural plants. Adding cinnamon nanoparticles to O. niloticus diet (3.0 g/Kg) enhanced the growth through activation of digestion enzymes and nutrient digestibility. Furthermore, nanoparticles of cinnamon enhanced fish immunity against A. hydrophila infection and activated some antioxidant enzymes: catalase, superoxide dismutase, and malondialdehyde (Abdel-Tawwab et al. 2018). They also stated that the nano form of cinnamon was more efficient than the bulk form studied formerly by Ahmad et al. (2011). In the same way, feeding C. carpio on nanoparticles of ginger (1 g/kg fish diet) for 30 days increased growth, protein, albumin, and lysozyme activity (Korni and Khalil 2017). The more effectiveness of nano cinnamon and ginger are returned to the unique properties of the nanoparticles, they can remain in the blood for a long period that facilitate their good bioavailability. Nanoparticles of another natural product, clove oil, were evaluated as a cheap, safe, and effective anesthetic, and it was reported that nanoparticles of clove oil have an antibacterial effect against S. agalactiae and F. columnare infection in fish farms (Yostawonkul et al. 2019).
Environmental risk
Many environmental risks can be occurred due to the wide application of nanoparticles in various fields. Unfortunately, the aquatic environment is the main recipient of any introduced materials, including nanoparticles. In water, many chemical, physical, and/or biological transformations of NPs can take place through aggregation, adsorption, dissolution, and redox reactions. So, NPs are ultimately transformed to produce more or less different materials with new features and reactivity (Odzak et al. 2017; Yin et al. 2015). Such new materials interact with the biological system, organic and inorganic constituents in the surrounding environment, and negatively affect the non-targeted living organisms. They caused cytotoxicity, dysfunction of many cell organelles (cell membrane, mitochondria, and ribosomes), DNA damage, and hereditary toxicity that mean the possibility of transfer of toxic effects during the different fetal developmental stages and magnifying their toxic effect (Fig. 2) (Zhang et al. 2018). Many researchers have denoted the toxic effects after application of nano-materials, on non-targeted organisms. Wang et al. (2016) have stated that the application of carbon nano-tubes caused secondary pollution and enhanced the toxic effect of cadmium on Daphnia magna. Also, the negative impacts of MON on various phytoplanktons, zooplanktons, and early developmental stages of fish have been reported (Aruoja et al. 2009; Regier et al. 2015). Bhuvaneshwari et al. (2015) revealed a decrease in cell viability and increase in ROS production in algae (Scenedesmus obliquus) after exposure to nano-ZnO under light conditions, which may be attributed to the more release of Zn ion in the presence of light than dark conditions. Recently, Borase et al. (2019) have reported that exposure to sublethal doses (1.4 and 2.9 mg/l) of Au nanoparticles had some toxic effects on Moina macrocopa crustacean as a non-target organism, they reduced acetylcholinesterase, trypsin, and amylase activities, increased oxidative activity and affect its swimming activity. Due to the uniquely small size properties and high reactivity of nanoparticles, they are agglomerated in the aquatic environment, losing their nanoscale properties, and reacted with various organic and inorganic constituents (Gehrke et al. 2015; Zhang et al. 2018). Also, nano-materials may be coated and reacted with some organic substances such as humic acid and fulvic acid that changed their physicochemical properties, behavior, and toxic effects to living organisms (Cai et al. 2019; Tang et al. 2014). Besides that, some nano-materials like apatite and biochar-supported by Fe-phosphate contribute to increasing phosphorus level in the surrounding water that inevitably leading to eutrophication, which represents a serious problem in fish cultures (Qiao et al. 2017).
Toxicity of the environmental transformed metal nanoparticles (Zhang et al. 2018)
High cost
Until this moment, it is expensive to introduce some applications of nanotechnology such as nano-filters, nano-membranes, or nano-sensors in the fish culture because of its high cost. Nevertheless, its applications in egg and hatchery ponds and in case of valuable broad-stocks (Bhattacharyya et al. 2015).
Technical producing difficulties
It is difficult in producing nanoparticles with stable properties and sustains their properties without changing, since they re-aggregate, transformed, and react with various environmental factors (Zhang et al. 2018). Furthermore, a lot of high-cost techniques must be used for the characterization of nanoparticles, and to identify their size, shape, and morphology (Patil and Kim 2017). Table 4 summarized the instruments that can be used in the characterizations of nanoparticles.
Lack of information
Despite the rapid progress in nanotechnology applications all over the world in the last decade, there is scarce data about their assimilation, distribution, accumulation through the food chain, and excretion from living organisms (Tripathi et al. 2017). Also, most of the studies that deal with nanoparticles and their effects on fish are restricted to in vitro and lab studies. I think it will be a great challenge to take the step of field application without more knowledge and more research.
Environmental fate and residue determination
Data about the uptake, absorption, distribution, metabolism, and excretion of nano-materials is limited. It was stated that the biological uptake of nanoparticles is performed through endocytosis, diffusion across the cell membrane, or phagocytosis, not via ion transporters like metal bulk form (Akter et al. 2018; Shaw and Handy 2011). Regarding the metabolism and excretion of nanoparticles, the liver is the main organ responsible for the excretion of nanoparticles in fish, since these particles cannot pass through the glomeruli filtration. Nano-metals deposit in the liver, but if they will stay as inert deposits or be excreted through the bile is still unknown (Handy et al. 2008; Shaw and Handy 2011).
After using nano-materials in an application, their residues in fish tissue should be identified and quantified to determine their hazardous effect. The routine methods cannot be used for the direct measurement of nanoparticles in fish tissues, while other possible approaches may involve: chromatography, electron microscopy, Atomic fluorescence spectrophotometer, light scattering, auto-fluorescence techniques, and use of radiolabelled metal nanoparticles (Bandyopadhyay et al. 2013; Li et al. 2008). Also, using ICP-MS (Inductively coupled plasma mass spectrometry) to detect nano-metals in different fish tissues was recommended (Ramsden et al. 2009). However, nano-metals do not behave the same way as their bulk forms and the ordinary standard protocols for metal determination, such as digestion with concentrated nitric acid before the analysis, may not work for nano-metals. So there is a need to modify such protocols (Shaw and Handy 2011).
Conclusion
From the above scenario, we can conclude that nanotechnology, like many new technologies, represents a double-edged weapon. The special unique characteristics of nanoparticles are the same that causing both positive and negative effects. Applications of nanotechnology in fish cultures have the ability to revolutionize them and solve many aquaculture problems more efficiently than conventional methods. They can participate in enhancing the fish growth performance and productivity, control and diagnosis of fish diseases, water purification and remediation of the pollutants, and finally extend the shelf life time in fish packaging. At the same time, applications of nanotechnology in fish cultures have some challenges; the most serious of them is the possible toxicity of nanoparticles, their hazardous effects on fish and the non-targeted organisms, besides their environmental transformation. However, green nanotechnology may act as eco-friendly and effective alternatives to toxic metal nanoparticles. Nevertheless, the complete safety of nanoparticles application in fish cultures should be assessed through the fulfillment of the present shortage in data concerning the environmental fate of nanoparticles, and their accumulation through the food chain to sustain safety for the fish, environment, and human consumption.
Recommendations
Many toxicological studies should be performed on a case-by-case basis to determine the potential hazards of nanoparticles, determine the least effective concentration, duration, and possible recovery time before field applications.
Make of cost-benefit balance study, especially in case of using new nano-devices in water purification, and use of high-cost devices in only egg hatching ponds and for valuable broad stocks.
Increase the knowledge about environmental fate; uptake, bioaccumulation, transformation, excretion, and analysis.
Guidelines of nanoparticles type, size, form, and concentration for fish health and human consumption should be created.
Regulations and legislations are required to control its synthesis, field application, and safely get rid of its residues.
In my point of view, I recommend the functional use of nano-materials in fish cultures, i.e., only use the nanomaterial to overcome a certain problem in your pond for a definite time and under specific concentration. Whereas using nanotechnology without the presence of an urgent problem may be a cause of introducing new problems.
The integration between nanotechnology and natural products like plants, and using biodegradable nano natural polymers, like chitosan instead of metal nano-materials are also highly encouraged to achieve progress in fish productivity.
Data availability
Not applicable
Abbreviations
- Abb:
-
Full name
- A. hydrophila :
-
Aeromonas hydrophila
- A. bestiarum :
-
Aeromonas bestiarum
- A. invadans :
-
Aphanomyces invadans
- Ag:
-
silver
- Al2O3 :
-
aluminum oxide
- ATP:
-
adenosine triphosphate
- Au:
-
gold
- AuCl4 :
-
tetra chloro aurate
- B. subtilis :
-
Bacillus subtilis
- C. auratus :
-
Carassius auratus
- C. carpio :
-
Cyprinus carpio
- Ca:
-
calcium
- Cd:
-
cadmium
- Cr:
-
chromium
- Cu:
-
copper
- DLS:
-
dynamic light scattering
- DNA:
-
deoxyribonucleic acid
- E. coli :
-
Escherichia coli
- E. tarda :
-
Edwardsiella tarda
- EC50 :
-
effective concentration
- EDX:
-
energy-dispersive x-ray spectroscopy
- F. branchiophilum :
-
Flavobacterium branchiophilum
- F. columnare :
-
Flavobacterium columnare
- FDA:
-
Food and Drug Administration
- Fe:
-
iron
- Fe2O3 :
-
ferric oxide
- FT-IR:
-
Fourier transform infrared spectroscopy
- I. multifiliis :
-
Ichthyophthirius multifiliis
- ICP-MS:
-
inductively coupled plasma mass spectrometry
- ISAV:
-
infectious Salmon Anemia Virus
- L. garvieae :
-
Lactococcus garvieae
- L. rohita :
-
Labeo rohita
- La:
-
lanthanides
- LC50 :
-
half lethal concentration that caused 50% mortality of organisms
- M. rosenbergii :
-
Macrobrachium rosenbergii
- MgO:
-
magnesium oxide
- MnO:
-
manganese oxide
- MON:
-
metal oxide nanoparticles
- NPs:
-
nanoparticles
- nZVI:
-
nano zero-valent iron
- O. latipes :
-
Oryzias latipes
- O. mykiss :
-
Oncorhynchus mykiss
- O. niloticus :
-
Oreochromis niloticus
- P. monodon :
-
Penaeus monodon
- P. semisulcatus :
-
Penaeus semisulcatus
- P. aeruginosa :
-
Pseudomonas aeruginosa
- P. fluorescens :
-
Pseudomonas fluorescens
- P. olivaceus :
-
Paralichthys olivaceus
- Pb:
-
lead
- PCBs:
-
polychlorinated biphenyls
- PLGA:
-
poly lactic-glycolic acid
- ROS:
-
reactive oxygen species
- S. aureus :
-
Staphylococcus aureus
- S. iniae :
-
Streptococcus iniae
- Se:
-
selenium
- SEM:
-
scanning electron microscopy
- SiO2 :
-
silicon dioxide
- TiO2 :
-
titanium dioxide
- UV-vis:
-
ultraviolet-visible spectrophotometer
- V. anguillarum :
-
Vibrio anguillarum
- V. harveyi :
-
Vibrio harveyi
- V. parahaemolyticus :
-
Vibrio parahaemolyticus
- V. vulnificus :
-
Vibrio vulnificus
- XRD:
-
X-ray powder diffraction
- Zn:
-
zinc
- ZP:
-
zeta potential measurement
References
Abdel-Khalek AA, Badran SR, Marie M-AS (2016a) Toxicity evaluation of copper oxide bulk and nanoparticles in Nile tilapia, Oreochromis niloticus, using hematological, bioaccumulation and histological biomarkers. Fish Physiol Biochem 42:1225–1236
Abdel-Khalek AA, Hamed A, Marie M-A (2016b) The Accumulation Potency of Bulk and Nano Zinc Metal and Their Impacts on the Hematological and Histological Perturbations of Oreochromis niloticus. Water Air Soil Pollut 227:206
Abdel-Razek N (2019): Antimicrobial activities of chitosan nanoparticles against pathogenic microorganisms in Nile tilapia, Oreochromis niloticus. Aquac Int, 1-16
Abdel-Tawwab M, Samir F, El-Naby ASA, Monier MN (2018) Antioxidative and immunostimulatory effect of dietary cinnamon nanoparticles on the performance of Nile tilapia, Oreochromis niloticus (L.) and its susceptibility to hypoxia stress and Aeromonas hydrophila infection. Fish Shellfish Immunol 74:19–25
Abdel-Tawwab M, Razek NA, Abdel-Rahman AM (2019) Immunostimulatory effect of dietary chitosan nanoparticles on the performance of Nile tilapia, Oreochromis niloticus (L.). Fish Shellfish Immunol 88:254–258
Abdel-Wahhab MA, Aljawish A, Kenawy AM, El-Nekeety AA, Hamed HS, Abdel-Aziem SH (2016) Grafting of gallic acid onto chitosan nano particles enhances antioxidant activities in vitro and protects against ochratoxin A toxicity in catfish (Clarias gariepinus). Environ Toxicol Pharmacol 41:279–288
Abdou KA, Mohammed AN, Moselhy WAER, Farghali AA (2018) Assessment of Modified Rice Husk and Sawdust as Bio-adsorbent for Heavy Metals Removal Using Nano Particles in Fish Farm. Asian J Anim Vet Adv 13:180–188
Abu-Elala NM, AbuBakr HO, Khattab MS, Mohamed SH, El-hady MA, Ghandour RA, Morsi RE (2018a) Aquatic environmental risk assessment of chitosan/silver, copper and carbon nanotube nanocomposites as antimicrobial agents. Int J Biol Macromol 113:1105–1115
Abu-Elala NM, Attia MM, Abd-Elsalam RM (2018b) Chitosan-silver nanocomposites in goldfish aquaria: A new perspective in Lernaea cyprinacea control. Int J Biol Macromol 111:614–622
Adomako M, St-Hilaire S, Zheng Y, Eley J, Marcum R, Sealey W, Donahower B, LaPatra S, Sheridan P (2012) Oral DNA vaccination of rainbow trout, Oncorhynchus mykiss (Walbaum), against infectious haematopoietic necrosis virus using PLGA [Poly (D, L-Lactic-Co-Glycolic Acid)] nanoparticles. J Fish Dis 35:203–214
Afifi M, Zinada OAA, Ali H, Couderchet M (2016) Zinc nanoparticles induced brain lesions and behavioral changes in two Tilapia species. Jpn J Vet Res 64:S161–S166
Ahmad MH, El Mesallamy AM, Samir F, Zahran F (2011) Effect of cinnamon (Cinnamomum zeylanicum) on growth performance, feed utilization, whole-body composition, and resistance to Aeromonas hydrophila in nile tilapia. J Appl Aquac 23:289–298
Ahmed F, Soliman FM, Adly MA, Soliman HA, El-Matbouli M, Saleh M (2019) Recent progress in biomedical applications of chitosan and its nanocomposites in aquaculture: A review. Res Vet Sci
Akbary P, Jahanbakhshi A (2019) Nano and macro iron oxide (Fe2O3) as feed additives: Effects on growth, biochemical, activity of hepatic enzymes, liver histopathology and appetite-related gene transcript in goldfish (Carassius auratus). Aquaculture 510:191–197
Akter M, Sikder MT, Rahman MM, Ullah AA, Hossain KFB, Banik S, Hosokawa T, Saito T, Kurasaki M (2018) A systematic review on silver nanoparticles-induced cytotoxicity: Physicochemical properties and perspectives. J Adv Res 9:1–16
Alhadrami H, Al-Hazmi F (2017) Antibacterial activities of titanium oxide nanoparticles. J Bioelectron Nanotechnol 2(1):5
Alishahi A, Mirvaghefi A, Tehrani M, Farahmand H, Koshio S, Dorkoosh F, Elsabee MZ (2011) Chitosan nanoparticle to carry vitamin C through the gastrointestinal tract and induce the non-specific immunity system of rainbow trout (Oncorhynchus mykiss). Carbohydr Polym 86:142–146
Alishahi A, Proulx J, Aider M (2014): Chitosan as Biobased Nanocomposite in Seafood Industry and Aquaculture. Seafood Sci: Advances in Chemistry, Technology and Applications 211
Amaro M, Oaew S, Surareungchai W (2012) Scano-magneto immunoassay based on carbon nanotubes/gold nanoparticles nanocomposite for Salmonella enterica serovar Typhimurium detection. Biosens Bioelectron 38:157–162
Aruoja V, Dubourguier H-C, Kasemets K, Kahru A (2009) Toxicity of nanoparticles of CuO, ZnO and TiO2 to microalgae Pseudokirchneriella subcapitata. Sci Total Environ 407:1461–1468
Ashouri S, Keyvanshokooh S, Salati AP, Johari SA, Pasha-Zanoosi H (2015) Effects of different levels of dietary selenium nanoparticles on growth performance, muscle composition, blood biochemical profiles and antioxidant status of common carp (Cyprinus carpio). Aquaculture 446:25–29
Ashraf M, Aklakur M, Sharma R, Ahmad S, Khan M (2011) Nanotechnology as a novel tool in fisheries and aquaculture development: a review. Iran J Energy Environ 2:258–261
Ashraf PM, Edwin L (2016) Nano copper oxide incorporated polyethylene glycol hydrogel: an efficient antifouling coating for cage fishing net. Int Biodeterior Biodegradation 115:39–48
Assefa A, Abunna F (2018) Maintenance of fish health in aquaculture: review of epidemiological approaches for prevention and control of infectious disease of fish. Vet Med Int 2018:5432497
Atyah M, Zamri-Saad M, Siti-Zahrah A (2010) First report of methicillin-resistant Staphylococcus aureus from cage-cultured tilapia (Oreochromis niloticus). Vet Microbiol 144:502–504
Awad A, Zaglool AW, Ahmed SA, Khalil SR (2019) Transcriptomic profile change, immunological response and disease resistance of Oreochromis niloticus fed with conventional and Nano-Zinc oxide dietary supplements. Fish Shellfish Immunol 93:336–343
Bandyopadhyay S, Peralta-Videa JR, Gardea-Torresdey JL (2013) Advanced analytical techniques for the measurement of nanomaterials in food and agricultural samples: a review. Environ Eng Sci 30:118–125
Barakat KM, El-Sayed HS, Gohar YM (2016) Protective effect of squilla chitosan–silver nanoparticles for Dicentrarchus labrax larvae infected with Vibrio anguillarum. Int Aquat Res 8:179–189
Bhat IA, Ahmad I, Mir IN, Bhat RAH, Gireesh-Babu P, Goswami M, Sundaray J, Sharma R (2019) Chitosan-eurycomanone nanoformulation acts on steroidogenesis pathway genes to increase the reproduction rate in fish. J Steroid Biochem Mol Biol 185:237–247
Bhattacharyya A, Reddy J, Hasan MM, Adeyemi M, Marye RR (2015) Nanotechnology-a unique future technology in aquaculture for the food security. Int J Bioassays 4:4115–4126
Bhuvaneshwari M, Iswarya V, Archanaa S, Madhu G, Kumar GS, Nagarajan R, Chandrasekaran N, Mukherjee A (2015) Cytotoxicity of ZnO NPs towards fresh water algae Scenedesmus obliquus at low exposure concentrations in UV-C, visible and dark conditions. Aquat Toxicol 162:29–38
Borase HP, Muley AB, Patil SV, Singhal RS (2019) Nano-eco toxicity study of gold nanoparticles on aquatic organism Moina macrocopa: As new versatile ecotoxicity testing model. Environ Toxicol Pharmacol 68:4–12
Buffet P-E, Tankoua OF, Pan J-F, Berhanu D, Herrenknecht C, Poirier L, Amiard-Triquet C, Amiard J-C, Bérard J-B, Risso C (2011) Behavioural and biochemical responses of two marine invertebrates Scrobicularia plana and Hediste diversicolor to copper oxide nanoparticles. Chemosphere 84:166–174
Cai C, Zhao M, Yu Z, Rong H, Zhang C (2019) Utilization of nanomaterials for in-situ remediation of heavy metal (loid) contaminated sediments: A review. Sci Total Environ 662:205–217
Can E, Kizak V, Kayim M, Can SS, Kutlu B, Ates M, Kocabas M, Demirtas N (2011) Nanotechnological applications in aquaculture-seafood industries and adverse effects of nanoparticles on environment. J Mater Sci Eng 5:605–609
Cavalieri F, Tortora M, Stringaro A, Colone M, Baldassarri L (2014) Nanomedicines for antimicrobial interventions. J Hosp Infect 88:183–190
Champ MA (2003) Economic and environmental impacts on ports and harbors from the convention to ban harmful marine anti-fouling systems. Mar Pollut Bull 46:935–940
Chen P-J, Su C-H, Tseng C-Y, Tan S-W, Cheng C-H (2011) Toxicity assessments of nanoscale zerovalent iron and its oxidation products in medaka (Oryzias latipes) fish. Mar Pollut Bull 63:339–346
Chen W-F, Zhang J, Zhang X, Wang W, Li Y (2016) Investigation of heavy metal (Cu, Pb, Cd, and Cr) stabilization in river sediment by nano-zero-valent iron/activated carbon composite. Environ Sci Pollut Res 23:1460–1470
Cheng T, Yao K, Yeh N, Chang C, Hsu H, Chien Y, Chang C (2009) Visible light activated bactericidal effect of TiO2/Fe3O4 magnetic particles on fish pathogens. Surf Coat Technol 204:1141–1144
Cheng T, Yao K, Yeh N, Chang C, Hsu H, Gonzalez F, Chang C (2011) Bactericidal effect of blue LED light irradiated TiO2/Fe3O4 particles on fish pathogen in seawater. Thin Solid Films 519:5002–5006
Chiavola A, Stoller M, Di Palma L, Boni MR (2017) Magnetic core nanoparticles coated by titania and alumina for water and wastewater remediation from metal contaminants. Chem Eng Trans 60:205–210
Chong MN, Jin B, Chow CW, Saint C (2010) Recent developments in photocatalytic water treatment technology: a review. Water Res 44:2997–3027
Chris UO, Singh N, Agarwal A (2018) Nanoparticles as feed supplement on Growth behaviour of Cultured Catfish (Clarias gariepinus) fingerlings. Mater Today: Proceedings 5:9076–9081
Chunglok W, Wuragil DK, Oaew S, Somasundrum M, Surareungchai W (2011) Immunoassay based on carbon nanotubes-enhanced ELISA for Salmonella enterica serovar Typhimurium. Biosens Bioelectron 26:3584–3589
Chupani L, Zusková E, Niksirat H, Panáček A, Lünsmann V, Haange S-B, von Bergen M, Jehmlich N (2017) Effects of chronic dietary exposure of zinc oxide nanoparticles on the serum protein profile of juvenile common carp (Cyprinus carpio L.). Sci Total Environ 579:1504–1511
Cinelli M, Coles SR, Nadagouda MN, Błaszczyński J, Słowiński R, Varma RS, Kirwan K (2015) A green chemistry-based classification model for the synthesis of silver nanoparticles. Green Chem 17:2825–2839
Connolly M, Fernández M, Conde E, Torrent F, Navas JM, Fernández-Cruz ML (2016) Tissue distribution of zinc and subtle oxidative stress effects after dietary administration of ZnO nanoparticles to rainbow trout. Sci Total Environ 551:334–343
Costa AS, Brandao HDM, da Silva S, Bentes-Sousa A, Diniz Junior J, Pinheiro J, Melo M, Silva Junior J, Matos E, Ribeiro-Costa R (2015) Mucoadhesive nanoparticles: a new perspective for fish drug application. Embrapa Gado de Leite-Nota Técnica/Nota Científica (ALICE) 39:503–506
Cui Y, Zhao Y, Tian Y, Zhang W, Lü X, Jiang X (2012) The molecular mechanism of action of bactericidal gold nanoparticles on Escherichia coli. Biomaterials 33:2327–2333
Dai H (2002) Carbon nanotubes: opportunities and challenges. Surf Sci 500:218–241
Dananjaya S, Godahewa G, Jayasooriya R, Lee J, De Zoysa M (2016) Antimicrobial effects of chitosan silver nano composites (CAgNCs) on fish pathogenic Aliivibrio (Vibrio) salmonicida. Aquaculture 450:422–430
Daniel S, Sironmani TA, Dinakaran S (2016) Nano formulations as curative and protective agent for fish diseases: studies on red spot and white spot diseases of ornamental gold fish Carassius auratus. Int J Fish Aquat Stud 4:255–261
Dawood MA, Koshio S, Zaineldin AI, Van Doan H, Moustafa EM, Abdel-Daim MM, Esteban MA, Hassaan MS (2019a) Dietary supplementation of selenium nanoparticles modulated systemic and mucosal immune status and stress resistance of red sea bream (Pagrus major). Fish Physiol Biochem 45:219–230
Dawood MA, Zommara M, Eweedah NM, Helal AI (2019b) The evaluation of growth performance, blood health, oxidative status and immune-related gene expression in Nile tilapia (Oreochromis niloticus) fed dietary nanoselenium spheres produced by lactic acid bacteria. Aquaculture 515:734571
De Jong WH, Borm PJ (2008) Drug delivery and nanoparticles: applications and hazards. Int J Nanomedicine 3:133
Deng Y, Chen Q (2003) Affects of nano-selenium on the growth of nile tilapia (Oreochromis niloticus). Inland Aquat Prod 6:28–30
Donbrow M (1991): Microcapsules and nanoparticles in medicine and pharmacy. CRC press
El-Greisy Z, El-Gamal A (2012) Monosex production of tilapia, Oreochromis niloticus using different doses of 17α-methyltestosterone with respect to the degree of sex stability after one year of treatment. Egypt J Aquat Res 38:59–66
El-Naby ASA, Al-Sagheer AA, Negm SS, Naiel MA (2020) Dietary combination of chitosan nanoparticle and thymol affects feed utilization, digestive enzymes, antioxidant status, and intestinal morphology of Oreochromis niloticus. Aquaculture 515:734577
El-Naby FSA, Naiel MA, Al-Sagheer AA, Negm SS (2019) Dietary chitosan nanoparticles enhance the growth, production performance, and immunity in Oreochromis niloticus. Aquaculture 501:82–89
Elsheshtawy A, Yehia N, Elkemary M, Soliman H (2019) Direct detection of unamplified Aeromonas hydrophila DNA in clinical fish samples using gold nanoparticle probe-based assay. Aquaculture 500:451–457
Faiz H, Zuberi A, Nazir S, Rauf M, Younus N (2015) Zinc oxide, zinc sulfate and zinc oxide nanoparticles as source of dietary zinc: Comparative effects on growth and hematological indices of juvenile grass carp (Ctenopharyngodon idella). Int J Agric Biol 17:568–574
Fajardo C, Ortíz L, Rodríguez-Membibre M, Nande M, Lobo M, Martin M (2012) Assessing the impact of zero-valent iron (ZVI) nanotechnology on soil microbial structure and functionality: a molecular approach. Chemosphere 86:802–808
Fan H-L, Zhou S-F, Jiao W-Z, Qi G-S, Liu Y-Z (2017) Removal of heavy metal ions by magnetic chitosan nanoparticles prepared continuously via high-gravity reactive precipitation method. Carbohydr Polym 174:1192–1200
FAO (2018) The State of World Fisheries and Aquaculture 2018-Meeting the sustainable development goals. FAO Rome, Italy
Fatima R, Priya M, Indurthi L, Radhakrishnan V, Sudhakaran R (2019) Biosynthesis of silver nanoparticles using red algae Portieria hornemannii and its antibacterial activity against fish pathogens. Microb Pathog 138:103780
Federici G, Shaw BJ, Handy RD (2007) Toxicity of titanium dioxide nanoparticles to rainbow trout (Oncorhynchus mykiss): Gill injury, oxidative stress, and other physiological effects. Aquat Toxicol 84:415–430
Fenaroli F, Westmoreland D, Benjaminsen J, Kolstad T, Skjeldal FM, Meijer AH, van der Vaart M, Ulanova L, Roos N, Nyström B (2014) Nanoparticles as drug delivery system against tuberculosis in zebrafish embryos: direct visualization and treatment. ACS Nano 8:7014–7026
Futalan CM, Kan C-C, Dalida ML, Pascua C, Wan M-W (2011) Fixed-bed column studies on the removal of copper using chitosan immobilized on bentonite. Carbohydr Polym 83:697–704
Gaffney AM, Santos-Martinez MJ, Satti A, Major TC, Wynne KJ, Gun'ko YK, Annich GM, Elia G, Radomski MW (2015) Blood biocompatibility of surface-bound multi-walled carbon nanotubes. Nanomedicine 11:39–46
Gardea-Torresdey J, Parsons J, Gomez E, Peralta-Videa J, Troiani H, Santiago P, Yacaman MJ (2002) Formation and growth of Au nanoparticles inside live alfalfa plants. Nano Lett 2:397–401
Gardea-Torresdey JL, Gomez E, Peralta-Videa JR, Parsons JG, Troiani H, Jose-Yacaman M (2003) Alfalfa sprouts: a natural source for the synthesis of silver nanoparticles. Langmuir 19:1357–1361
Gehrke I, Geiser A, Somborn-Schulz A (2015) Innovations in nanotechnology for water treatment. Nanotechnol Sci Appl 8:1–17
Gerber LC, Moser N, Luechinger NA, Stark WJ, Grass RN (2012) Phosphate starvation as an antimicrobial strategy: the controllable toxicity of lanthanum oxide nanoparticles. Chem Commun 48:3869–3871
Ghasemi Z, Sourinejad I, Kazemian H, Rohani S (2018) Application of zeolites in aquaculture industry: a review. Rev Aquac 10:75–95
Gillies ER, Frechet JM (2005) Dendrimers and dendritic polymers in drug delivery. Drug Discov Today 10:35–43
Gong J-L, Wang B, Zeng G-M, Yang C-P, Niu C-G, Niu Q-Y, Zhou W-J, Liang Y (2009) Removal of cationic dyes from aqueous solution using magnetic multi-wall carbon nanotube nanocomposite as adsorbent. J Hazard Mater 164:1517–1522
Gregory AE, Williamson D, Titball R (2013) Vaccine delivery using nanoparticles. Front Cell Infect Microbiol 3:13
Griffitt RJ, Weil R, Hyndman KA, Denslow ND, Powers K, Taylor D, Barber DS (2007) Exposure to copper nanoparticles causes gill injury and acute lethality in zebrafish (Danio rerio). Environ Sci Technol 41:8178–8186
Griffitt RJ, Luo J, Gao J, Bonzongo JC, Barber DS (2008) Effects of particle composition and species on toxicity of metallic nanomaterials in aquatic organisms. Environ Toxicol Chem: An International Journal 27:1972–1978
Gunalan S, Sivaraj R, Rajendran V (2012) Green synthesized ZnO nanoparticles against bacterial and fungal pathogens. Progress Nat Sci: Materials International 22:693–700
Hamad MTMH (2019) Disinfection of El-Gharbia drain from pathogenic bacteria using chitosan–silver nanoparticles. Int J Environ Sci Technol 16:8267–8282
Handy RD, Henry TB, Scown TM, Johnston BD, Tyler CR (2008) Manufactured nanoparticles: their uptake and effects on fish—a mechanistic analysis. Ecotoxicology 17:396–409
Hao L, Chen L, Hao J, Zhong N (2013) Bioaccumulation and sub-acute toxicity of zinc oxide nanoparticles in juvenile carp (Cyprinus carpio): a comparative study with its bulk counterparts. Ecotoxicol Environ Saf 91:52–60
Hassan SW, El-latif HHA (2018) Characterization and Applications of the Biosynthesized Silver Nanoparticles by Marine Pseudomonas sp. H64. J Pure Appl Microbiol 12:1289–1300
He X, Deng H, Hwang H-M (2018) The current application of nanotechnology in food and agriculture. J Food Drug Anal 27:1–21
Hu Y-L, Qi W, Han F, Shao J-Z, Gao J-Q (2011) Toxicity evaluation of biodegradable chitosan nanoparticles using a zebrafish embryo model. Int J Nanomedicine 6:3351
Huang L, Xu Y, Guo S (2010) Disinfecting aquatic pathogenic bacteria by photocatalytic activity with nanometer TiO2. J Jimei Univ (Nat Sci) 4:54–57
Huang S, Wang L, Liu L, Hou Y, Li L (2015) Nanotechnology in agriculture, livestock, and aquaculture in China. A review. Agron Sustain Dev 35:369–400
Huang X, Lan Y, Liu Z, Huang W, Guo Q, Liu L, Hu M, Sui Y, Wu F, Lu W (2018) Salinity mediates the toxic effect of nano-TiO2 on the juvenile olive flounder Paralichthys olivaceus. Sci Total Environ 640:726–735
Iravani S (2011) Green synthesis of metal nanoparticles using plants. Green Chem 13:2638–2650
Ishwarya R, Vaseeharan B, Subbaiah S, Nazar AK, Govindarajan M, Alharbi NS, Kadaikunnan S, Khaled JM, Al-anbr MN (2018) Sargassum wightii-synthesized ZnO nanoparticles–from antibacterial and insecticidal activity to immunostimulatory effects on the green tiger shrimp Penaeus semisulcatus. J Photochem Photobiol B Biol 183:318–330
Ishwarya R, Jayakumar R, Abinaya M, Govindarajan M, Alharbi NS, Kadaikunnan S, Khaled JM, Al-Anbr MN, Vaseeharan B (2019) Facile synthesis of haemocyanin-capped zinc oxide nanoparticles: Effect on growth performance, digestive-enzyme activity, and immune responses of Penaeus semisulcatus. Int J Biol Macromol 139:688–696
Jain KK (2003) Nanodiagnostics: application of nanotechnology in molecular diagnostics. Expert Rev Mol Diagn 3:153–161
Jaroenram W, Arunrut N, Kiatpathomchai W (2012) Rapid and sensitive detection of shrimp yellow head virus using loop-mediated isothermal amplification and a colorogenic nanogold hybridization probe. J Virol Methods 186:36–42
Ji J, Torrealba D, Ruyra À, Roher N (2015) Nanodelivery systems as new tools for immunostimulant or vaccine administration: targeting the fish immune system. Biology 4:664–696
Jiang X, Valdeperez D, Nazarenus M, Wang Z, Stellacci F, Parak WJ, Del Pino P (2015) Future perspectives towards the use of nanomaterials for smart food packaging and quality control. Part Part Syst Charact 32:408–416
Jiménez-Fernández E, Ruyra A, Roher N, Zuasti E, Infante C, Fernández-Díaz C (2014) Nanoparticles as a novel delivery system for vitamin C administration in aquaculture. Aquaculture 432:426–433
Jimmy CY, Tang HY, Yu J, Chan H, Zhang L, Xie Y, Wang H, Wong S (2002) Bactericidal and photocatalytic activities of TiO2 thin films prepared by sol–gel and reverse micelle methods. J Photochem Photobiol A Chem 153:211–219
Johari S, Kalbassi M, Soltani M, Yu I (2013) Toxicity comparison of colloidal silver nanoparticles in various life stages of rainbow trout (Oncorhynchus mykiss). Iran J Fish Sci 12:76–95
Johari SA, Kalbassi MR, Soltani M, Yu IJ (2016) Application of nanosilver-coated zeolite as water filter media for fungal disinfection of rainbow trout (Oncorhynchus mykiss) eggs. Aquac Int 24:23–38
Joshi HD, Tiwari V, Gupta S, Sharma R, Lakra W, Sahoo U (2019) Application of nanotechnology for the production of masculinized Tilapia, Oreochromis niloticus (Linnaeus, 1758). Aquaculture 511:734206
Jovanović B, Anastasova L, Rowe EW, Zhang Y, Clapp AR, Palić D (2011) Effects of nanosized titanium dioxide on innate immune system of fathead minnow (Pimephales promelas Rafinesque, 1820). Ecotoxicol Environ Saf 74:675–683
Jovanović B, Whitley EM, Kimura K, Crumpton A, Palić D (2015) Titanium dioxide nanoparticles enhance mortality of fish exposed to bacterial pathogens. Environ Pollut 203:153–164
Julio C, Aida H, María D, Jorge C, Jaime A (2018) Silver nanoparticles applications (AgNPS) in aquaculture. Int J Fish Aquat Stud 6:05–11
Kalatehjari P, Yousefian M, Khalilzadeh MA (2015) Assessment of antifungal effects of copper nanoparticles on the growth of the fungus Saprolegnia sp. on white fish (Rutilus frisii kutum) eggs. Egypt J Aquat Res 41:303–306
Karthikeyeni S, Siva Vijayakumar T, Vasanth S, Arul Ganesh MM, Subramanian P (2013) Biosynthesis of Iron oxide nanoparticles and its haematological effects on fresh water fish Oreochromis mossambicus. J Acad Indus Res 10:645–649
Kaya H, Aydın F, Gürkan M, Yılmaz S, Ates M, Demir V, Arslan Z (2015) Effects of zinc oxide nanoparticles on bioaccumulation and oxidative stress in different organs of tilapia (Oreochromis niloticus). Environ Toxicol Pharmacol 40:936–947
Khan KU, Zuberi A, Nazir S, Fernandes JBK, Jamil Z, Sarwar H (2016) Effects of dietary selenium nanoparticles on physiological and biochemical aspects of juvenile Tor putitora. Turk J Zool 40:704–712
Khan KU, Zuberi A, Nazir S, Ullah I, Jamil Z, Sarwar H (2017) Synergistic effects of dietary nano selenium and vitamin C on growth, feeding, and physiological parameters of mahseer fish (Tor putitora). Aquaculture Rep 5:70–75
Khosravi-Katuli K, Prato E, Lofrano G, Guida M, Vale G, Libralato G (2017) Effects of nanoparticles in species of aquaculture interest. Environ Sci Pollut Res 24:17326–17346
Kitiyodom S, Yata T, Yostawornkul J, Kaewmalun S, Nittayasut N, Suktham K, Surassmo S, Namdee K, Rodkhum C, Pirarat N (2019) Enhanced efficacy of immersion vaccination in tilapia against columnaris disease by chitosan-coated “pathogen-like” mucoadhesive nanovaccines. Fish Shellfish Immunol 95:213–219
Klingelfus T, Disner G, Voigt C, Alle L, Cestari M, Leme D (2019) Nanomaterials induce DNA-protein crosslink and DNA oxidation: A mechanistic study with RTG-2 fish cell line and Comet assay modifications. Chemosphere 215:703–709
Knetsch ML, Koole LH (2011) New strategies in the development of antimicrobial coatings: the example of increasing usage of silver and silver nanoparticles. Polymers 3:340–366
Korni F, Khalil F (2017) Effect of ginger and its nanoparticles on growth performance, cognition capability, immunity and prevention of motile Aeromonas septicaemia in Cyprinus carpio fingerlings. Aquac Nutr 23:1492–1499
Kuan GC, Sheng LP, Rijiravanich P, Marimuthu K, Ravichandran M, Yin LS, Lertanantawong B, Surareungchai W (2013) Gold-nanoparticle based electrochemical DNA sensor for the detection of fish pathogen Aphanomyces invadans. Talanta 117:312–317
Kumar PV, Pammi S, Kollu P, Satyanarayana K, Shameem U (2014) Green synthesis and characterization of silver nanoparticles using Boerhaavia diffusa plant extract and their anti bacterial activity. Ind Crop Prod 52:562–566
Kumar SR, Ahmed VI, Parameswaran V, Sudhakaran R, Babu VS, Hameed AS (2008) Potential use of chitosan nanoparticles for oral delivery of DNA vaccine in Asian sea bass (Lates calcarifer) to protect from Vibrio (Listonella) anguillarum. Fish Shellfish Immunol 25:47–56
Kumar V, Sharma N, Maitra S (2017) In vitro and in vivo toxicity assessment of nanoparticles. Int Nano Lett 7:243–256
Kuswandi B (2017) Environmental friendly food nano-packaging. Environ Chem Lett 15:205–221
Lara HH, Ayala-Núñez NV, Turrent LCI, Padilla CR (2010) Bactericidal effect of silver nanoparticles against multidrug-resistant bacteria. World J Microbiol Biotechnol 26:615–621
Li H, Zhang J, Wang T, Luo W, Zhou Q, Jiang G (2008) Elemental selenium particles at nano-size (Nano-Se) are more toxic to Medaka (Oryzias latipes) as a consequence of hyper-accumulation of selenium: a comparison with sodium selenite. Aquat Toxicol 89:251–256
Li J, Chen Z, Huang R, Miao Z, Cai L, Du Q (2018a) Toxicity assessment and histopathological analysis of nano-ZnO against marine fish (Mugilogobius chulae) embryos. J Environ Sci 73:78–88
Li L, Lin SL, Deng L, Liu ZG (2013) Potential use of chitosan nanoparticles for oral delivery of DNA vaccine in black seabream Acanthopagrus schlegelii Bleeker to protect from Vibrio parahaemolyticus. J Fish Dis 36:987–995
Li M, Wu Q, Wang Q, Xiang D, Zhu G (2018b) Effect of titanium dioxide nanoparticles on the bioavailability and neurotoxicity of cypermethrin in zebrafish larvae. Aquat Toxicol 199:212–219
Li Y-H, Ding J, Luan Z, Di Z, Zhu Y, Xu C, Wu D, Wei B (2003) Competitive adsorption of Pb2+, Cu2+ and Cd2+ ions from aqueous solutions by multiwalled carbon nanotubes. Carbon 41:2787–2792
Lima E, Guerra R, Lara V, Guzmán A (2013) Gold nanoparticles as efficient antimicrobial agents for Escherichia coli and Salmonella typhi. Chem Central J 7:11
Liu P, Guo T, Wei H (2009a) Bactericidal effects of copper-bearing nano-montmorillonite on three aquatic pathogenic bacteria and two intestinal available bacteria in vitro. J Shanghai Ocean Univ 18:520–526
Liu Y, He L, Mustapha A, Li H, Hu Z, Lin M (2009b) Antibacterial activities of zinc oxide nanoparticles against Escherichia coli O157: H7. J Appl Microbiol 107:1193–1201
Lockman P, Mumper R, Khan M, Allen D (2002) Nanoparticle technology for drug delivery across the blood-brain barrier. Drug Dev Ind Pharm 28:1–13
Lok C-N, Ho C-M, Chen R, He Q-Y, Yu W-Y, Sun H, Tam PK-H, Chiu J-F, Che C-M (2006) Proteomic analysis of the mode of antibacterial action of silver nanoparticles. J Proteome Res 5:916–924
López-Carballo G, Higueras L, Gavara R, Hernández-Muñoz P (2012) Silver ions release from antibacterial chitosan films containing in situ generated silver nanoparticles. J Agric Food Chem 61:260–267
Lü J-M, Wang X, Marin-Muller C, Wang H, Lin PH, Yao Q, Chen C (2009) Current advances in research and clinical applications of PLGA-based nanotechnology. Expert Rev Mol Diagn 9:325–341
Luis AIS, Campos EVR, de Oliveira JL, Fraceto LF (2019) Trends in aquaculture sciences: from now to use of nanotechnology for disease control. Rev Aquac 11:119–132
Luo Z, Li M, Wang Z, Li J, Guo J, Rosenfeldt RR, Seitz F, Yan C (2018) Effect of titanium dioxide nanoparticles on the accumulation and distribution of arsenate in Daphnia magna in the presence of an algal food. Environ Sci Pollut Res 25:20911–20919
Mahmoud UM, Mekkawy IA, Naguib M, Sayed AE-DH (2019) Silver nanoparticle–induced nephrotoxicity in Clarias gariepinus: physio-histological biomarkers. Fish Physiol Biochem 45:1895–1905
Makadia HK, Siegel SJ (2011) Poly lactic-co-glycolic acid (PLGA) as biodegradable controlled drug delivery carrier. Polymers 3:1377–1397
Martinez-Castanon G, Nino-Martinez N, Martinez-Gutierrez F, Martinez-Mendoza J, Ruiz F (2008) Synthesis and antibacterial activity of silver nanoparticles with different sizes. J Nanopart Res 10:1343–1348
Matsumura Y, Yoshikata K, S-i K, Tsuchido T (2003) Mode of bactericidal action of silver zeolite and its comparison with that of silver nitrate. Appl Environ Microbiol 69:4278–4281
Mekkawy IA, Mahmoud UM, Hana MN, Sayed AE-DH (2019) Cytotoxic and hemotoxic effects of silver nanoparticles on the African Catfish, Clarias gariepinus (Burchell, 1822). Ecotoxicol Environ Saf 171:638–646
Mishra M, Dashora K, Srivastava A, Fasake VD, Nag RH (2019) Prospects, challenges and need for regulation of nanotechnology with special reference to India. Ecotoxicol Environ Saf 171:677–682
Moghimi SM, Hunter A, Andresen T (2012) Factors controlling nanoparticle pharmacokinetics: an integrated analysis and perspective. Annu Rev Pharmacol Toxicol 52:481–503
Mohamed MM, Fouad SA, Elshoky HA, Mohammed GM, Salaheldin TA (2017) Antibacterial effect of gold nanoparticles against Corynebacterium pseudotuberculosis. Int J Vet Sci Med 5:23–29
Mohammad AM, Eldin TAS, Hassan MA, El-Anadouli BE (2017) Efficient treatment of lead-containing wastewater by hydroxyapatite/chitosan nanostructures. Arab J Chem 10:683–690
Mohanty B, Samanta S, Hassan M, Behera B, Pandit A, Manna R, Roy A, Meena D, Raman R, Sudhesan D (2017) Book of Abstracts: National Seminar on “Priorities in Fisheries and Aquaculture”(PFA-2017). ICAR–Central Inland Fisheries Research Institute, Barrackpore, pp 01–227
Mueller NC, Nowack B (2008) Exposure modeling of engineered nanoparticles in the environment. Environ Sci Technol 42:4447–4453
Mühling M, Bradford A, Readman JW, Somerfield PJ, Handy RD (2009) An investigation into the effects of silver nanoparticles on antibiotic resistance of naturally occurring bacteria in an estuarine sediment. Mar Environ Res 68:278–283
Muralisankar T, Bhavan PS, Radhakrishnan S, Seenivasan C, Manickam N, Srinivasan V (2014) Dietary supplementation of zinc nanoparticles and its influence on biology, physiology and immune responses of the freshwater prawn, Macrobrachium rosenbergii. Biol Trace Elem Res 160:56–66
Myhr AI, Myskja BK (2011) Precaution or integrated responsibility approach to nanovaccines in fish farming? A critical appraisal of the UNESCO precautionary principle. Nanoethics 5:73–86
Namdeo M, Bajpai S (2008) Chitosan–magnetite nanocomposites (CMNs) as magnetic carrier particles for removal of Fe (III) from aqueous solutions. Colloids Surf A Physicochem Eng Asp 320:161–168
Nikapitiya C, Dananjaya S, De Silva B, Heo G-J, Oh C, De Zoysa M, Lee J (2018) Chitosan nanoparticles: a positive immune response modulator as display in zebrafish larvae against Aeromonas hydrophila infection. Fish Shellfish Immunol 76:240–246
Ochoa-Meza AR, Álvarez-Sánchez AR, Romo-Quiñonez CR, Barraza A, Magallón-Barajas FJ, Chávez-Sánchez A, García-Ramos JC, Toledano-Magaña Y, Bogdanchikova N, Pestryakov A (2019) Silver nanoparticles enhance survival of white spot syndrome virus infected Penaeus vannamei shrimps by activation of its immunological system. Fish Shellfish Immunol 84:1083–1089
Odzak N, Kistler D, Sigg L (2017) Influence of daylight on the fate of silver and zinc oxide nanoparticles in natural aquatic environments. Environ Pollut 226:1–11
Pandey S, Mishra SB (2011) Organic–inorganic hybrid of chitosan/organoclay bionanocomposites for hexavalent chromium uptake. J Colloid Interface Sci 361:509–520
Pati R, Mehta RK, Mohanty S, Padhi A, Sengupta M, Vaseeharan B, Goswami C, Sonawane A (2014) Topical application of zinc oxide nanoparticles reduces bacterial skin infection in mice and exhibits antibacterial activity by inducing oxidative stress response and cell membrane disintegration in macrophages. Nanomedicine 10:1195–1208
Patil MP, Kim G-D (2017) Eco-friendly approach for nanoparticles synthesis and mechanism behind antibacterial activity of silver and anticancer activity of gold nanoparticles. Appl Microbiol Biotechnol 101:79–92
Poguberović SS, Krčmar DM, Maletić SP, Kónya Z, Pilipović DDT, Kerkez DV, Rončević SD (2016) Removal of As (III) and Cr (VI) from aqueous solutions using “green” zero-valent iron nanoparticles produced by oak, mulberry and cherry leaf extracts. Ecol Eng 90:42–49
Pradeep T (2009) Noble metal nanoparticles for water purification: a critical review. Thin Solid Films 517:6441–6478
Prakash P, Gnanaprakasam P, Emmanuel R, Arokiyaraj S, Saravanan M (2013) Green synthesis of silver nanoparticles from leaf extract of Mimusops elengi, Linn. for enhanced antibacterial activity against multi drug resistant clinical isolates. Colloids Surf B: Biointerfaces 108:255–259
Qian G, Chen W, Lim TT, Chui P (2009) In-situ stabilization of Pb, Zn, Cu, Cd and Ni in the multi-contaminated sediments with ferrihydrite and apatite composite additives. J Hazard Mater 170:1093–1100
Qiao Y, Wu J, Xu Y, Fang Z, Zheng L, Cheng W, Tsang EP, Fang J, Zhao D (2017) Remediation of cadmium in soil by biochar-supported iron phosphate nanoparticles. Ecol Eng 106:515–522
Rai M, Yadav A, Gade A (2009) Silver nanoparticles as a new generation of antimicrobials. Biotechnol Adv 27:76–83
Raissy M, Ansari M (2011) In vitro antimicrobial effect of silver nanoparticles on Lactococcus garvieae and Streptococcus iniae. Afr J Microbiol Res 5:4442–4445
Rajeshkumar S, Venkatesan C, Sarathi M, Sarathbabu V, Thomas J, Basha KA, Hameed AS (2009) Oral delivery of DNA construct using chitosan nanoparticles to protect the shrimp from white spot syndrome virus (WSSV). Fish Shellfish Immunol 26:429–437
Rajkumar K, Kanipandian N, Thirumurugan R (2016) Toxicity assessment on haemotology, biochemical and histopathological alterations of silver nanoparticles-exposed freshwater fish Labeo rohita. Appl Nanosci 6:19–29
Ramalingam M, Jabbari E, Ramakrishna S, Khademhosseini A (2013): Micro and Nanotechnologies in Engineering Stem Cells and Tissues, 39. John Wiley & Sons
Ramamoorthy S, Kannaiyan P, Moturi M, Devadas T, Muthuramalingam J, Natarajan L, Arunachalam N, Ponniah AG (2013) Antibacterial activity of zinc oxide nanoparticles against Vibrio harveyi. Indian J Fish 60:107–112
Ramsden CS, Smith TJ, Shaw BJ, Handy RD (2009) Dietary exposure to titanium dioxide nanoparticles in rainbow trout,(Oncorhynchus mykiss): no effect on growth, but subtle biochemical disturbances in the brain. Ecotoxicology 18:939–951
Rather M, Sharma R, Aklakur M, Ahmad S, Kumar N, Khan M, Ramya V (2011) Nanotechnology: a novel tool for aquaculture and fisheries development. A prospective mini-review. Fish Aquac J 16:3
Rather MA, Sharma R, Gupta S, Ferosekhan S, Ramya V, Jadhao SB (2013) Chitosan-nanoconjugated hormone nanoparticles for sustained surge of gonadotropins and enhanced reproductive output in female fish. PLoS One 8:e57094
Regier N, Cosio C, Von Moos N, Slaveykova VI (2015) Effects of copper-oxide nanoparticles, dissolved copper and ultraviolet radiation on copper bioaccumulation, photosynthesis and oxidative stress in the aquatic macrophyte Elodea nuttallii. Chemosphere 128:56–61
Reilly RM (2007) Carbon nanotubes: potential benefits and risks of nanotechnology in nuclear medicine. J Nucl Med 48:1039–1042
Remya AS, Ramesh M, Saravanan M, Poopal RK, Bharathi S, Nataraj D (2015) Iron oxide nanoparticles to an Indian major carp, Labeo rohita: Impacts on hematology, iono regulation and gill Na+/K+ ATPase activity. J King Saud Univ-Sci 27:151–160
Ren X, Chen C, Nagatsu M, Wang X (2011) Carbon nanotubes as adsorbents in environmental pollution management: a review. Chem Eng J 170:395–410
Ren X, Zhao X, Duan X, Fang Z (2018) Enhanced bio-concentration of tris (1, 3-dichloro-2-propyl) phosphate in the presence of nano-TiO2 can lead to adverse reproductive outcomes in zebrafish. Environ Pollut 233:612–622
Rhim J-W, Park H-M, Ha C-S (2013) Bio-nanocomposites for food packaging applications. Prog Polym Sci 38:1629–1652
Rivas-Aravena A, Fuentes Y, Cartagena J, Brito T, Poggio V, La Torre J, Mendoza H, Gonzalez-Nilo F, Sandino AM, Spencer E (2015) Development of a nanoparticle-based oral vaccine for Atlantic salmon against ISAV using an alphavirus replicon as adjuvant. Fish Shellfish Immunol 45:157–166
Robinson E, Menghe H (2006) Catfish nutrition: Feeds. Mississippi State Univ Ext Serv Publ 2413:03–06
Roy A, Bulut O, Some S, Mandal AK, Yilmaz MD (2019) Green synthesis of silver nanoparticles: biomolecule-nanoparticle organizations targeting antimicrobial activity. RSC Adv 9:2673–2702
Saleh M, Soliman H, Haenen O, El-Matbouli M (2011) Antibody-coated gold nanoparticles immunoassay for direct detection of Aeromonas salmonicida in fish tissues. J Fish Dis 34:845–852
Saleh M, Soliman H, Schachner O, El-Matbouli M (2012) Direct detection of unamplified spring viraemia of carp virus RNA using unmodified gold nanoparticles. Dis Aquat Org 100:3–10
Saleh M, Kumar G, Abdel-Baki A-A, Al-Quraishy S, El-Matbouli M (2016) In vitro antimicrosporidial activity of gold nanoparticles against Heterosporis saurida. BMC Vet Res 12:44
Saleh M, Abdel-Baki A-A, Dkhil MA, El-Matbouli M, Al-Quraishy S (2017) Antiprotozoal effects of metal nanoparticles against Ichthyophthirius multifiliis. Parasitology 144:1802–1810
Sayed AE-DH (2016) Genotoxicity detection following exposure to silver nanoparticles in African catfish (Clarias gariepinus). Int J Nanoparticles 9:41–53
Seetang-Nun Y, Jaroenram W, Sriurairatana S, Suebsing R, Kiatpathomchai W (2013) Visual detection of white spot syndrome virus using DNA-functionalized gold nanoparticles as probes combined with loop-mediated isothermal amplification. Mol Cell Probes 27:71–79
Sele V, Ørnsrud R, Sloth JJ, Berntssen MH, Amlund H (2018) Selenium and selenium species in feeds and muscle tissue of Atlantic salmon. J Trace Elem Med Biol 47:124–133
Shaalan M, Saleh M, El-Mahdy M, El-Matbouli M (2016) Recent progress in applications of nanoparticles in fish medicine: a review. Nanomedicine 12:701–710
Shaalan MI, El-Mahdy MM, Theiner S, El-Matbouli M, Saleh M (2017) In vitro assessment of the antimicrobial activity of silver and zinc oxide nanoparticles against fish pathogens. Acta Vet Scand 59:49
Shaik S, Kummara MR, Poluru S, Allu C, Gooty JM, Kashayi CR, Subha MCS (2013) A green approach to synthesize silver nanoparticles in starch-co-poly (acrylamide) hydrogels by tridax procumbens leaf extract and their antibacterial activity. Int J Carbohydr Chem 2013:1–10
Shaw BJ, Handy RD (2011) Physiological effects of nanoparticles on fish: a comparison of nanometals versus metal ions. Environ Int 37:1083–1097
Sivaramasamy E, Zhiwei W, Li F, Xiang J (2016) Enhancement of vibriosis resistance in Litopenaeus vannamei by supplementation of biomastered silver nanoparticles by Bacillus subtilis. J Nanomed Nanotechnol 7:2
Soliman WS, Shaapan RM, Mohamed LA, Gayed SS (2019) Recent biocontrol measures for fish bacterial diseases, in particular to probiotics, bio-encapsulated vaccines, and phage therapy. Open Vet J 9:190–195
Song L, Vijver MG, Peijnenburg WJ, Galloway TS, Tyler CR (2015) A comparative analysis on the in vivo toxicity of copper nanoparticles in three species of freshwater fish. Chemosphere 139:181–189
Sonkusre P, Nanduri R, Gupta P, Cameotra SS (2014) Improved extraction of intracellular biogenic selenium nanoparticles and their specificity for cancer chemoprevention. J Nanomed Nanotechnol 5:1
Sørum H (2008): Antibiotic resistance associated with veterinary drug use in fish farms, Improving farmed fish quality and safety. Elsevier, pp. 157-182
Srinivasan V, Bhavan PS, Rajkumar G, Satgurunathan T, Muralisankar T (2016) Effects of dietary iron oxide nanoparticles on the growth performance, biochemical constituents and physiological stress responses of the giant freshwater prawn Macrobrachium rosenbergii post-larvae. Int J Fish Aquat Stud 4:170–182
Stefaniuk M, Oleszczuk P, Ok YS (2016) Review on nano zerovalent iron (nZVI): from synthesis to environmental applications. Chem Eng J 287:618–632
Su H, Fang Z, Tsang PE, Fang J, Zhao D (2016) Stabilisation of nanoscale zero-valent iron with biochar for enhanced transport and in-situ remediation of hexavalent chromium in soil. Environ Pollut 214:94–100
Subara D, Jaswir I, Alkhatib MFR, Noorbatcha IA (2018) Synthesis of fish gelatin nanoparticles and their application for the drug delivery based on response surface methodology. Adv Nat Sci Nanosci Nanotechnol 9:045014
Subramani K, Elhissi A, Subbiah U, Ahmed W (2019): Introduction to nanotechnology, Nanobiomaterials in Clinical Dentistry. Elsevier, pp. 3-18
Sumbayev VV, Yasinska IM, Gibbs BF (2013) Biomedical applications of gold nanoparticles. In: Recent advances in circuits, communications and signal processing. WSEAS Press, Athens, pp 342–348
Sumi N, Chitra K (2019) Fullerene C60 nanomaterial induced oxidative imbalance in gonads of the freshwater fish, Anabas testudineus (Bloch, 1792). Aquat Toxicol 210:196–206
Swain P, Nayak S, Sasmal A, Behera T, Barik S, Swain S, Mishra S, Sen A, Das J, Jayasankar P (2014) Antimicrobial activity of metal based nanoparticles against microbes associated with diseases in aquaculture. World J Microbiol Biotechnol 30:2491–2502
Tang W-W, Zeng G-M, Gong J-L, Liang J, Xu P, Zhang C, Huang B-B (2014) Impact of humic/fulvic acid on the removal of heavy metals from aqueous solutions using nanomaterials: a review. Sci Total Environ 468:1014–1027
Tawfik M, Moustafa M, Abumourad I, El-Meliegy E, Refai M (2017) Evaluation of Nano Zinc Oxide feed additive on tilapia Growth and Immunity, 15th International Conference on Environmental Science and Technology. Rhodes, Greece
Tayel AA, Elsaied BE, Diab AM (2019): Nanotechnology for Aquaculture, Nanoscience for Sustainable Agriculture. Springer, pp 479-544
Tello-Olea M, Rosales-Mendoza S, Campa-Córdova A, Palestino G, Luna-González A, Reyes-Becerril M, Velazquez E, Hernandez-Adame L, Angulo C (2019) Gold nanoparticles (AuNP) exert immunostimulatory and protective effects in shrimp (Litopenaeus vannamei) against Vibrio parahaemolyticus. Fish Shellfish Immunol 84:756–767
Tian J, Sun X, Chen X, Yu J, Qu L, Wang L (2008) The formulation and immunisation of oral poly (DL-lactide-co-glycolide) microcapsules containing a plasmid vaccine against lymphocystis disease virus in Japanese flounder (Paralichthys olivaceus). Int Immunopharmacol 8:900–908
Torchilin VP (2006): Nanoparticulates as drug carriers. Imperial college press
Tripathi DK, Tripathi A, Singh S, Singh Y, Vishwakarma K, Yadav G, Sharma S, Singh VK, Mishra RK, Upadhyay R (2017) Uptake, accumulation and toxicity of silver nanoparticle in autotrophic plants, and heterotrophic microbes: a concentric review. Front Microbiol 8:7
Tuševljak N, Dutil L, Rajić A, Uhland F, McClure C, St-Hilaire S, Reid-Smith R, McEwen S (2013) Antimicrobial use and resistance in aquaculture: findings of a globally administered survey of aquaculture-allied professionals. Zoonoses Public Health 60:426–436
Twiddy D, Reilly P, Chatham K (1995): Occurrence of antibiotic-resistant human-pathogens in integrated fish farms. FAO Fish Rep 23-37
Valero Y, Awad E, Buonocore F, Arizcun M, Esteban MÁ, Meseguer J, Chaves-Pozo E, Cuesta A (2016) An oral chitosan DNA vaccine against nodavirus improves transcription of cell-mediated cytotoxicity and interferon genes in the European sea bass juveniles gut and survival upon infection. Dev Comp Immunol 65:64–72
Vaseeharan B, Ramasamy P, Chen J (2010) Antibacterial activity of silver nanoparticles (AgNps) synthesized by tea leaf extracts against pathogenic Vibrio harveyi and its protective efficacy on juvenile Feneropenaeus indicus. Lett Appl Microbiol 50:352–356
Velmurugan P, Iydroose M, Lee S-M, Cho M, Park J-H, Balachandar V, Oh B-T (2014) Synthesis of silver and gold nanoparticles using cashew nut shell liquid and its antibacterial activity against fish pathogens. Indian J Microbiol 54:196–202
Vijayan SR, Santhiyagu P, Singamuthu M, Kumari Ahila N, Jayaraman R, Ethiraj K (2014) Synthesis and characterization of silver and gold nanoparticles using aqueous extract of seaweed, Turbinaria conoides, and their antimicrofouling activity. Sci World J 2014:938272
Vinay TN, Bhat S, Gon Choudhury T, Paria A, Jung M-H, Shivani Kallappa G, Jung S-J (2018) Recent advances in application of nanoparticles in fish vaccine delivery. Rev Fish Sci Aquac 26:29–41
Vinod V, Saravanan P, Sreedhar B, Devi DK, Sashidhar R (2011) A facile synthesis and characterization of Ag, Au and Pt nanoparticles using a natural hydrocolloid gum kondagogu (Cochlospermum gossypium). Colloids Surf B: Biointerfaces 83:291–298
Wang JJ, Zeng ZW, Xiao RZ, Xie T, Zhou GL, Zhan XR, Wang SL (2011) Recent advances of chitosan nanoparticles as drug carriers. Int J Nanomedicine 6:765
Wang X, Qu R, Liu J, Wei Z, Wang L, Yang S, Huang Q, Wang Z (2016) Effect of different carbon nanotubes on cadmium toxicity to Daphnia magna: The role of catalyst impurities and adsorption capacity. Environ Pollut 208:732–738
Wang Y, Yan X, Fu L (2013) Effect of selenium nanoparticles with different sizes in primary cultured intestinal epithelial cells of crucian carp, Carassius auratus gibelio. Int J Nanomedicine 8:4007
Wu L-p, Ficker M, Christensen JB, Trohopoulos PN, Moghimi SM (2015) Dendrimers in medicine: therapeutic concepts and pharmaceutical challenges. Bioconjug Chem 26:1198–1211
Xia IF, Cheung JS, Wu M, Wong K-S, Kong H-K, Zheng X-T, Wong K-H, Kwok KW (2019) Dietary chitosan-selenium nanoparticle (CTS-SeNP) enhance immunity and disease resistance in zebrafish. Fish Shellfish Immunol 87:449–459
Yin Y, Yang X, Zhou X, Wang W, Yu S, Liu J, Jiang G (2015) Water chemistry controlled aggregation and photo-transformation of silver nanoparticles in environmental waters. J Environ Sci 34:116–125
Yirsaw BD, Megharaj M, Chen Z, Naidu R (2016) Environmental application and ecological significance of nano-zero valent iron. J Environ Sci 44:88–98
Yostawonkul J, Kitiyodom S, Kaewmalun S, Suktham K, Nittayasut N, Khongkow M, Namdee K, Ruktanonchai UR, Rodkhum C, Pirarat N (2019) Bifunctional clove oil nanoparticles for anesthesia and anti-bacterial activity in Nile tilapia (Oreochromis niloticus). Aquaculture 503:589–595
Zhang J, Guo W, Li Q, Wang Z, Liu S (2018) The effects and the potential mechanism of environmental transformation of metal nanoparticles on their toxicity in organisms. Environ Sci: Nano 5:2482–2499
Zhang W-X, Wang C-B, Lien H-L (1998) Treatment of chlorinated organic contaminants with nanoscale bimetallic particles. Catal Today 40:387–395
Zhao D, Wang J, Sun B, Sun B, Gao J, Xu R (2000) Development and application of TiO2 photocatalysis as antimicrobialagent. J Liaoning Univ (Nat Sci Ed) 2:173–174
Zhao L, Seth A, Wibowo N, Zhao C-X, Mitter N, Yu C, Middelberg AP (2014) Nanoparticle vaccines. Vaccine 32:327–337
Zhou X, Wang Y, Gu Q, Li W (2009) Effects of different dietary selenium sources (selenium nanoparticle and selenomethionine) on growth performance, muscle composition and glutathione peroxidase enzyme activity of crucian carp (Carassius auratus gibelio). Aquaculture 291:78–81
Zhu X, Tian S, Cai Z (2012) Toxicity assessment of iron oxide nanoparticles in zebrafish (Danio rerio) early life stages. PLoS One 7:e46286
Author information
Authors and Affiliations
Contributions
Wafaa Abbas is the only author who wrote and revised this review article.
Corresponding author
Ethics declarations
Competing interests
The authors declare that they have no competing interests.
Ethics approval and consent to participate
Not applicable
Consent for publication
Not applicable
Additional information
Responsible editor: Philippe Garrigues
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Abbas, W.T. Advantages and prospective challenges of nanotechnology applications in fish cultures: a comparative review. Environ Sci Pollut Res 28, 7669–7690 (2021). https://doi.org/10.1007/s11356-020-12166-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-020-12166-0