Abstract
Chromanone or Chroman-4-one is the most important and interesting heterobicyclic compound and acts as a building block in medicinal chemistry for isolation, designing and synthesis of novel lead compounds. Structurally, absence of a double bond in chromanone between C-2 and C-3 shows a minor difference from chromone but exhibits significant variations in biological activities. In the present review, various studies published on synthesis, pharmacological evaluation on chroman-4-one analogues are addressed to signify the importance of chromanone as a versatile scaffold exhibiting a wide range of pharmacological activities. But, due to poor yield in the case of chemical synthesis and expensive isolation procedure from natural compounds, more studies are required to provide the most effective and cost-effective methods to synthesize novel chromanone analogs to give leads to chemistry community. Considering the versatility of chromanone, this review is designed to impart comprehensive, critical and authoritative information about chromanone template in drug designing and development.
Similar content being viewed by others

Avoid common mistakes on your manuscript.
1 Introduction
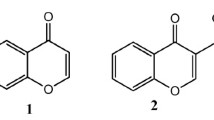
Chroman-4-one is one of the most important heterobicyclic moieties existing in natural compounds as polyphenols and as synthetic compounds like Taxifolin, also known as chromanone or benzo-dihydropyran or benzopyran. Structurally, chroman-4-one is a fusion of benzene nucleus (ring A) with dihydropyran (ring B) which relates to chromane, chromene, chromone and chromenone, but the absence of C2-C3 double bond of chroman-4-one skeleton makes a minor difference (Table 1) from chromone and associated with diverse biological activities [1].

Natural and synthetic chromanone analogs show various biological activities such as anticancer, tumor necrosis factor-α (TNF-α) inhibitors, antivascular, antidiabetic, antioxidant, antimicrobial, antifungal, antiviral, antileishmanial, insecticidal, spasmolytic, analgesic, anti-inflammatory, anticoagulant, estrogenic inhibitor, anti-acetylcholinesterase (AchE) inhibitor, antihuman immunodeficiency virus (HIV), anticonvulsant, antidepressants, anticoronal and antitubercular activity. Due to of these activities, several analogues of chromane are also available in market like tocopherols (vitamin E), taxifolin (antidiabetic), tetrazole (antidiabetic), troglitazone (antidiabetic), ormeloxifene (anticancer) and nebivolol (beta-blocker) [2]. Moreover, several isolated flavanones, flavonols, homoisoflavonoids like naringenin, naringin, myricetin, dihydroquercetin and kaempferol are also under clinical studies [3]. Therefore, among the diverse array of chromane, chroman-4-one/4-chromanone are of considerable interest to researchers due to potency of this clinically useful pharmacophore in the treatment of cancer and several other diseases [4]. Presently, a number of research groups are working in designing and development of more potent and significant chromanone analogues. So, in the present review, recent literature available up to 2020 about chroman-4-one derivative and their pharmacological activities is accrued. Present review article will offer a platform to the researchers in designing and development of novel potent chroman-4-one analogs.
2 Biological Activities of Chromanone
2.1 Anticancer Activity
Discovery and development of novel anticancer agents with potent cytotoxic activity is one of the top priorities in pharmaceutical research. Several limitations like variable efficacy, poor toxicity profile and adverse effects are associated with the usage of currently available chemotherapeutic agents [5]. Generally, cancer can be measured by assessing the degree of mitochondrial impairment and metabolic dysfunctions such as cellular energy supply, cell death signaling, irregulation of metabolic pathways, formation of reactive oxygen species (ROS), compromised enzyme actions, aerobic glycolysis augmented in tumor cells, alterations in lipid metabolism and unbalanced pH [6]. Therefore, to cure such metabolic dysfunctions, naturally occurring flavonoids, flavonols, flavanones (2-phenyl chroman-4-one derivatives) and homoisoflavanones exhibit good anticancer potential along with other pharmacological activities such as anti-inflammatory, antioxidative, antidiabetic, antibacterial, antimicrobial, antifungal, antimutagenic, antiparasitic and anti-HIV [7, 8]. In this regard, the following naturally occurring flavanones such as naringenin [9], naringin [9], sakuranetin [10], eriodictyol [11], calyxin G, deguelin [12] and sterubin [13] have been reported (Figs. 1, 2) to possess potent cytotoxic profile. Moreover, these naturally occurring flavanones can also regulate cellular metabolism, scavenge free radical and suppress proliferation of cancer cells [12, 13]. However, the precise molecular mechanisms of these flavonoids accountable for cytotoxic potential have not been completely elucidated till now. Moreover, poor yield is also a major hurdle to explore the potential of these compounds. So, synthesis of compounds containing chromanone with efficient cytotoxic action might be a privileged approach in searching new targets for cancer treatment [14]. A number of compounds are found to have significant anticancer activity. For instance, several compounds containing chroman-4-one and its natural and synthetic analogs possessing significant anticancer potential against cancer cell lines are enlisted in Table 2 [15].
2.2 Antioxidant
Excessive production of ROS, suppression of antioxidants as a result of normal biochemical processes due to the effect of several environmental and endogenous factors result in imbalance of oxidative-antioxidant. The excessive ROS higher than physiological concentrations cause oxidative stress [38, 39]. Oxidative stress affects all cellular functioning which may be responsible for generation of severe diseases like diabetes complications, neurological disease, atherosclerosis, skin lesions, inflammation, rheumatoid arthritis, aging, cardiovascular diseases and cancer [39, 40]. Generally, antioxidants react or debilitate excessive level of ROS [41].
There are a number of isolated flavanones and homoisoflavanones such as silymarin, pinobanksin, silybin, liquiritin, isointricatinol reported for their antioxidant activity in 1,1-diphenyl-2-picrylhydrazyl (DPPH) radical scavenging method. Along with isolated components, synthetic benzylidene chromanone derivatives also showed DPPH radical scavenging and ferric reducing antioxidant power (FRAP) [42, 43]. According to structure activity relationship (SAR) of chromanone analogs, C-2 and C-3 substitution with methoxyphenyl, amine derivatives, aromatic groups, benzylidene and cyclohexyl carbamoyl yields more potent antioxidant compounds which can produce equivalent antioxidant activity like vitamin E and Trolox [44] enlisted in Table 3. Therefore, more synthetic work is required to produce new chroman-4-one scaffolds as effective antioxidant compounds which can be used to manage the pathogenesis of diseases in which oxidative stress plays a significant role.
2.3 Anti-inflammatory Activity
Localized protective reaction to infection or injury well known as inflammatory reaction plays a significant role in the pathological mechanism of various inflammation-related diseases like cancer, Alzheimer, diabetes, atherosclerosis and cardiovascular disorders. Knowledge of etiology and mechanism of inflammation progression can prevent further development of diseases [59].
For treatment of inflammation, diverse range of isolated homoisoflavanones, dihydroflavonols such as dihydromyricetin, hesperitin, stilbin and silybin containing chromanone pharmacophore are reported with vivid bioactivity and play an exclusive role in discovery and development of new anti-inflammatory drugs [60]. Among the isolated compounds from natural sources, chroman-4-one containing stilbin and silybin are used clinically as anti-inflammatory drugs. Moreover, these isolated compounds further pave the way for synthesis of new anti-inflammatory drugs via inhibition of cyclooxygenase 2 (COX-2). 4-Homisoflavonones enlisted in Table 4 developed as chroman-4-one analogs were found to have significant inhibition for COX 2 receptor binding in range of 0 ± 5.4 to 0 ± 21.3(% activity) [54, 61]. Likewise, a new chromanone, violacein A produced from Streptomyces violaceoruber emerged as potential therapeutic for treatment of inflammation-related disease by suppressing NF-kB (nuclear factor kappa light chain enhancer of activated B cells) signaling pathways. Moreover, it can be used for the treatment of other diseases like cancer [62]. Generally, SAR studies revealed that C-2 substitution with hydroxybenzylidine, arylidene, hydroxyphenyl, pyridine-3-yl and fluorophenyl displayed the best anti-inflammatory activity with significant inhibition. So, exploring the substitutions at C-3, 6, 7 and 8th of chromanone nucleus yields more efficient anti-inflammatory compounds which can compete isolated phytochemicals as well as existing anti-inflammatory drugs [60, 62]
2.4 Antidiabetic Activity
Diabetes mellitus represented as hyperglycemia is increasing globally at very significant pace and has become a complex chronic metabolic disease associated with diabetic complications like diabetic neuropathy and nephropathy [69]. A number of potent antidiabetic drugs like metformin, glucotrol, tolbutamide, acarbose, pioglitazone, gliptins, liraglutide are available in the market, but long-term use of the antidiabetic drugs may cause serious side effects, and moreover, these drugs are less effective in management of associated diabetic complications [70]. In this regard, a number of isolated compounds as natural flavonols, flavanones and homoisoflavanones containing chromanone pharmacophore such as naringenin, hesperitin, dihydroquercetin, eriodictyol, sakuranetin, aromadendrin, butin, pinocembrin, nymphaeol A, sophoraflavanone G and sterubin (Fig. 3) are already reported for their excellent potency in the treatment of diabetic mellitus and diabetes associated complications [2]. These chromanone containing flavanones treat diabetes through inhibition of receptors like glucagon-like peptide 1 (GLP-1), dipeptidyl peptidase-IV (DPP4), peroxisome proliferator receptor gamma (PPAR-γ), Alpha glucosidase (α-glucosidase), phosphatidylinositol 3-kinase (PI3K) in both in vivo and in vitro experimentation [71]. Moreover, several synthetic chromanone analogs are also reported (enlisted in Table 5) for antidiabetic potential via α-glucosidase inhibition and DPPH radical scavenging activity. Zhu et al. 2019 isolated various chromanone analogs from the seeds of Psoralea corylifolia and evaluated for diglyceride acyltransferase (DGAT), protein-tyrosine phosphatase 1B(PTP1B) and α-glucosidase activity. Out of theses isolated compounds, (2S)-7-methoxy-6-(2-hydroxy-3-methyl but-3-en-1-yl)-2-(4-hydroxyphenyl)chroman-4-one, (2S)-4′-hydroxyl-7-hydroxy methylene-6-(2′′,3′′-epoxy-3′′- methyl butyl) flavanone and bavachinone B exhibited good antidiabetic effect by inhibition of DGAT, PTP1B and α-glucosidase. Takao K. et al. introduced benzylidene-4-chromanone derivatives as the lead compound for the development of novel α-glucosidase inhibitors as well as significant antioxidative agent due to its potential in significantly inhibiting the level of DPPH. By the virtue of SAR studies, substitution at C-2, 3, 6 and 7th position of parent chromanone provide effective antidiabetic drugs which might help in the synthesis of more efficient novel compounds for the cure of diabetes and its complications [72, 73]. But, the research and published data about the synthetic chromanone containing medicinal compounds as antidiabetic are very less as compared to isolated flavanones, flavonols and homoisoflavanones. Therefore, a very effective synthetic approach is required for the synthesis and evaluation of more chromanone analogs as antidiabetics [72, 74].
2.5 Antibacterial and Antifungal Agents
Increased prevalence of pathogenic microbial infections necessitates the need for new antibacterial agents with distinct mechanism of action and broad spectrum of activity over a wide range of bacterial and fungal strains. Considering this viewpoint, several naturally occurring flavonoids and heterobicyclic compounds containing chromanone pharmacophore were found to have diverse activities ranging from antibacterial to antiviral activity. But these compounds were found to have two major limitations like poor yield and resistance against the general bacterial and fungal strains such as Staphylococcus aureus, Bacillus subtilis, Pseudomonas aeruginosa, Escherichia coli, Aspergillus niger, Fusarium oxysporum, Penicillium italicum, Pythium ultimum, Sclerotinia sclerotium, Phytophthora capsici, E. Faecalis, C. albicans, C. Krusei, C. Glabrata and A. fumigates [81]. Therefore, there is a need for new synthetic antibacterial and antifungal compounds containing chromanone moiety.
It was found that substitution at C-2 with pyrazol-4-yl derivatives, methoxyphenyl, alkyl, vinyl, hydroxyl methyl and chlorophenyl group displayed broad spectrum antibacterial activity against tested bacterial strains, whereas azolyl and benzylidene derivatization yields good antifungal agents as shown in Table 6 [82]. Moreover, C-2, 3, 4, 6 and 7 substitutions also predicted efficient antibacterial and antifungal compounds with broad spectrum activity as well as low or no resistance. Likewise, most important series of benzylidene derivatives C-3-substituted 3-(benzo[1,3]dioxol-5-ylmethylene)-7-hydroxychroman-4-one, a 3-benzylidene-4-chromanone exhibited significant antibacterial activity against gram positive and gram negative bacteria, while 3-azolyl-4-chromanone phenyl hydrazones exhibited antifungal potential against C. albicans, S. cerevisiae, A. niger and M. gypseum pathogens [83]. Hence, more exhaustive approach for the chromanone scaffold may lead medical chemist to synthesize more potent antibacterial and antifungal compounds with maximum efficacy and minimum resistance.
2.6 Anti TB Agents
Among the available treatments for tuberculosis, first-line and second-line antituberculosis (TB) drugs, DOTS (directly observed treatment short-course) is one of the most competent multidrug effective strategy developed by WHO against Mycobacterium tuberculosis. However, the success rate for cure of TB patients struggles to achieve 85% [99]. Therefore, more research is required for the development of novel potent antimycobacterial agents with promising efficacy and less side effects. From the series of heterobicyclic compounds, isolated as well as synthetic chroman-4-one analogs were reported to inhibit M. tuberculosis [100, 101]. Chromanone analogs having significant potential for the treatment of TB with greater efficacy and less side effects are enlisted in Table 7. This will provide us a new framework in designing and development of chromanone derivatives and their potent multidrug combinations like spiro chromanones as novel antiTB agents [102].
2.7 Antiviral Agents
A number of viral diseases in human ranging from mild upper respiratory infection to fatal cardiac and neurological illnesses are caused by numerous picornaviruses particularly rhinoviruses (HRVs) and enteroviruses (EVs). A diverse range of effective antipicornavirus agents are developed and some are in clinical phase study [111]. Literature evidence has established that natural and synthetic flavonoids containing chromane obstruct the replication step of picornavirus and further prevent the decapsidation of infected viral segments and release of corresponding RNA within cells [112]. Tait S et al. (2006) examined antiviral activity of homoisoflavonoids (3-benzylchroman-4-ones derivatives) on enteroviruses replication, and these chromanone analogs showed significant activity against coxsackie virus B1, B3, B4, A9, echovirus 30 and substitution at C-3 of parent chromanone exhibited good efficacy against HRVs growth. Moreover, introduction of thiopyran at C-2 by Hegab, MI et al. (2015) showed considerable antiviral activity against strained adenovirus type 7 with significant inhibition as shown in Table 8. So, following these substitutions, more antiviral chromanone analogs may be designed and synthesized with significant antiviral activity [35, 113].
2.8 Anti-HIV Compounds
Chromanone derivatives have attained much potential against treatment of Acquired Immune Deficiency Syndrome (AIDS) caused by HIV-1 [116]. In 1994, an isolated calanolide A and inophyllums from calophyllum genus reported as a novel anti-HIV chemotype inhibited HIV-1-specific nonnucleoside RT [117]. Furthermore, an enantioselective designing of 2, 3-dimethyl-4-chromanone ring attachment was attempted for the synthesis of potent calophyllum coumarins as anti-HIV 1 and it showed significant effect which can compete with currently available protease inhibitors (atazanavir, ritonavir, lopinavir, nelfinavir, saquinavir etc.) approved by Food and Drug Administration (FDA) [118, 119].
Dawood et al. (2005) tested a series of 3-benzylidene derivatives of chromanone and substitution of thio (S) group at the place of oxo(-O) at Ist position displayed impressive anti-HIV activity. Consequently, SAR studies revealed that C-2 and 6 substitution with oxo propionic acid and benzyl group exhibited anti-HIV activity by inhibition of HIV-1 IN strand transfer with good inhibitory values as shown in Table 9. So, more research and development on chromanone analogs can be a prominent approach for synthesis of novel HIV1 inhibitors against the treatment of AIDS [120].
2.9 Antileishmanial Agents
A group of parasites of genus Leishmania causes tropical disease known as Leishmaniasis, which spreads in living beings by the bite of infected sandflies [123]. These flies have a multifarious life cycle which exhibits an amastigote phase in the host cell as well as a promastigote phase in vector flies. A number of clinical representations, especially cutaneous, visceral and mucosal forms, attain the most serious grade for Leishmaniasis [124]. Although diverse treatment for Leishmaniasis or cutaneous leishmaniasis (CL) exists, less potency, toxicity and cost issues are some of the significant limitations associated with the present treatment options. So, development of more effective novel compounds against Leishmaniasis remains an important stratagem. A wide range of natural flavonoids having chromanone pharmacophore and synthetic oxygenated heterocyclic compounds such as hydrazones, thiosemicarbazones and semicarbazones have been reported to exhibit antileishmanial potential. Among these, semicarbazone and thiosemicarbazone derivatives of flavanone and thioflavanone could be considered a potential privileged structure [125]. Using this approach, derivatization of chroman-4-one and thiochroman-4-ones with acyl hydrazones resulted in significant enhancement of antileishmanial potential [126]. As thiochroman-4-one derivatives showed immense similarity with the chroman-4-one compounds so could be considered as potential scaffold with wide range of bioactivity like antiviral, antitumoral, antimalarial, antibacterial and antileishmanial [127]. Therefore, more research in thiochromanone derivatization is required for more novel antileishmanial compounds, and some relevant compounds are reported in Table 10.
2.10 Anti-Acetyl Cholinesterase (AchE) Agents for Alzheimer Treatment
Alzheimer is a complicated neurodegenerative disease that poses severe threat to human health and characterized by memory loss, behavioral abnormalities and cognitive deficits [131]. Alzheimer’s Association reported that the number of patients suffering from Alzheimer’s disease (AD) is now approximately 47 million and this figure is projected to increase around 100 million by 2050 entire over the world [132]. So, demand of a better treatment for AD around the world is a challenge for the medical science.
Polyphenolic compounds like flavonoids, flavones, isoflavone exhibit broad range of biological properties including inhibition of AchE enzymes and its catalytic steps [133]. In this review, we included a number of chroman-4-one containing flavonoids which exhibit AchE inhibition potential [134]. This study presents that presence of an amino group, piperidinyl of chromanone derivatives and novel dithiocarbamates show potent AchE inhibition in comparison with the standard drugs like Tacrine (Table 11). Therefore, designing and development of new effective chromanone hybrids as AchE inhibitors provide a new framework to the treatment of AD and other neurodegenerative disorders [135].
2.11 Anticonvulsants
Epilepsy is a chronic neurological disease in which patient suffers from seizures, transient attacks that affect at least 70 million people around the world. A number of therapeutic agents are available in the market as antiepileptic drugs such as phenytoin, phenobarbital, benzodiazepines, carbamazepine, ethosuximide and sodium valproate exhibit acceptable management on seizures, but resistance is observed in 30–40% epilepsy patients [140]. Therefore, MDT (multiple-drug therapies) is preferred for the control of seizures. But still no MDT or single drug can prevent the development or treat epilepsy for longer time interval without development of resistance [141]. Considering this viewpoint, more potent compounds which can cure or prevent epilepsy with high efficacy and less side effects are need of the hour.
A series of chroman-4-one derivatives such as azolylchromanones, azolylchromanone oximes and imidazolyl chromanone oxime ethers were tested for their anticonvulsant potential in lithium, pilocarpine induced seizure and pentylenetetrazol (PTZ) kindling model of epilepsy [142]. Among these tested compounds, some analogs (Enlisted in Table 12) showed good anticonvulsant activity with significant seizure latency and seizure duration. SAR studies showed that presence of azolyl ring at 3-postion, halogen (Chloro) group at the 7-position and/or an alkyl (especially methyl) group at the 2-position of the chromane ring resulted in an enhancement of anti-seizure efficiency in O-(2,4-dichlorobenzyl) oxime series and no significant difference in efficacy was observed for both (Z)- and (E)-isomers against the seizure durations and seizure latency [143]. Therefore, effective report of promising chroman-4-one analogs (Table 11) in epileptic models can predict potential clinical worth in treatment of adequate epileptic disorder.
2.12 Antidepressants
Depression, a mental disorder, seeks a major concern in medical practice, and it is more common in patient than reported. Its generation is associated with amino acid metabolism in which tryptophan, an essential amino acid metabolizes amines and releases various amine neurotransmitters like serotonin, epinephrine, norepinephrine (NE), melanin and dopamine (DA) controlling the autonomic nervous system (ANS) and central nervous system (CNS) functioning under the catalysis of flavoenzyme monoamine oxidases (MAOs). It exists as in two isomeric forms MAO-A and MAO-B, from which MAO-A selectively metabolizes NE, epinephrine, 5-HT, whereas MAO-B specifically deactivates β-phenethylamine, benzylamine and inactivated by the following inhibitors such as rasagiline and selegiline with many side effects and resistance [146, 147]. So, new MAOs inhibitors are required for developing new antidepressant drugs.
For the development of more effective novel MAO inhibitors, the chromanone scaffolds from natural and synthetic sources displayed a promising effect and exhibited their potency for MAO-B inhibition selectively [148]. In particular, chromanone derivatives substituted with benzyloxy group at C-7 are more potent MAO-B inhibitors and after searching of more chromanone derivatives, DSP-1053, a novel compound examined for serotonin reuptake in vivo with 5-HT1A partial agonistic activity and inhibited serotonin reuptake and considered as fast antidepressant drug in clinical practice (Table 13) [149]. Consequently, after reviewing the clinical data of DSP-1053, a probability comes out that more research in chromanone compounds might add a new class of antidepressant drugs.
2.13 Insecticides
As per the literature, it is reported that for the development and metamorphosis regulation in insect, mainly 20-hydroxy-ecdysone and juvenile hormones are necessary and play promoting roles in endocrine systems of insects as growth regulators [153]. In 1988, 1-tert-Butyl-1, 2-dibenzoyl hydrazine, first non-steroidal ecdysone agonist, was reported. Further, a variety of ecdysone agonists were identified among them diacyl hydrazine compounds produced significant response for the growth and regulation of the insect structure [154, 155]. In 2007, Zhao et al. have introduced new design of insecticidal agents as chromanone derivatives of diacylhydrazines in which chromafenozide group was attached on a chromanone moiety instead of chromane structure (Table 14). This study provided significant evidence that chromanone derivatives of diacylhydrazines exhibited more potency for insecticidal activity as compared to reference compound ANS-118 (commercial insecticide containing chroman moiety) [156].
2.14 Diagnostic agents
Ganand et al. synthesized a series of (E)-3-benzylidenechroman-4-one’s derivatives (homoisoflavonoids) and evaluated for their diagnostic imaging potential for pathogenesis of AD by targeting β-amyloid (Aβ) plaques. In vitro studies revealed that (E)-3-(4-methoxybenzylidene)-6-bromo-chroman-4-one and (E)-3-(4-dimethylamino-benzylidene)-6-bromo-chroman-4-one derivatives exhibited high binding affinities to Aβ plaques with 9.98 and 9.10 nM (Ki values), respectively, as compared to [125I] 2-(4′- dimethylaminophenyl)-6-iodoimidazo[1,2-α] pyridine (IMPY) (reference compound) [1, 158], whereas fluorescent staining test (applied on the brain sections of AD patients) and biodistribution data revealed that (E)-3-(4-dimethylamino-benzylidene) derivatives can selectively label Aβ plaques in brain sections and [125I]-radio labeled compound showed adequate brain uptake for brain scanning (Table 15) [1]. Chroman-4-one derivatives may be helpful diagnostic imaging agent for early finding of Aβ plaques in AD brain. Hence, further research and development of novel chromanone derivatives for their diagnostic potential creates a great interest in the field of medicinal chemistry.
3 Miscellaneous
3.1 Antidengue
M. M. V. Ramana et al. (2015) reported a docking study of isolated flavanones and designed chroman-4-one compounds against NS2B/NS3 protease (dengue virus protein). As per literature survey, naringenin and pinocembrin are known to have effective potential as antidengue agent with good dock score. Similarly, in-silco experimentation revealed the antidengue activity of isolated eriodictyol having Leu 149 and Asn 152 hydrogen bonding and 2-(2, 4-dihydroxy-6-methylphenyl)-5, 7-dihydroxychroman-4-one [designed compound] also showed interactions with Asp 75 and Asn 152 with a comparable glide score -7.31 that characterizes its antidengue activity [159].

3.2 Antiparasitic
Chroman-4-one analogs 6-hydroxy-2-(3-hydroxyphenyl) chroman-4-one, 6-hydroxy-2-(4-hydroxyphenyl) chroman-4-one and 2-(3,4-dihydroxyphenyl)-6-hydroxychroman-4-one displayed antiparasitic activity by targeting pteridine reductase-1 and showed significant inhibition against T. brucei and L. infantum at 10 µM and 50 µM. Therefore, more research on chroman-4-one derivatives as antiparasitic may provide a new platform for the development of new antiparasitic agents. [160].

3.3 Anti-aging
Chroman-4-one derivatives were used as active compound in cosmetic preparations for care, improvement and refreshment of texture of the skin and hairs, for treatment of skin as well as hair-related defects like inflammation, allergies or wound healing process. Furthermore, cosmetic formulations containing vitamin A and its derivatives like retinol esters, retinoic acid have shown their action on the differentiation process of epithelial cells and therefore used against psoriasis, acne, skin spotting, wrinkles and discoloration. Therefore, chroman-4-one scaffolds have significant cosmetic value along with pharmacological activities [160].

3.4 Anticoronal
Kwon Dur-Han, et al. proposed inhibitory potential of an isolated flavanone, quercetin-7-rhamnoside against viral propagation. Furthermore, experimentation showed effective results against inhibition of coronavirus via specific inhibition of PEDV (porcine epidemic diarrhea virus) proliferation in Vero cells with good inhibitory activity = -7.143 CC50/IC50 = > (100 g/mL) as compared to standard antiviral drug (ribavirin). Accordingly, in vitro inhibiting activity of quercetin-7-rhamnoside against PEDV, a number of flavonoids such as quercetin, luteolin, apigenin showed effective inhibition in vero cells. In spite of PEDV inhibition, quercetin-7-rhamnoside also exhibited antiviral potential against porcine transmissible gastroenteritis virus (TGEV) with inhibition value > 1.58 [CC50/IC50 = > (100 g/mL/63.3 µg/ml)] and porcine respiratory coronavirus (PRCV) with inhibition value > 1.67 [CC50/IC50 = > (100 g/mL/59.8 µg/ml)]. Therefore, quercetin-7-rhamnoside displayed better antiviral inhibition potential against three types of coronaviruses like TGEV, PEDV and PRCV than reference drug (ribavirin) in vitro. In conclusion, more experimentation on chromanone containing flavonoids as anticoronal for the inhibition of other classes of corona virus might be led a new frame for drug discovery and development [161]

4 Conclusion
Chroman-4-one/chromanone pharmacophore is a privileged scaffold in medicinal research, consisting of two rings in which 2, 3-dihydro-γ-pyranone fused with an aromatic benzene nucleus and derivatization at 2, 3 and 4-positions of chromanone skeleton yields more effective families of flavonoids like 3-benzylidene-chromanones, spirochromanone, hydrazones, oximes, flavanones, homisoflavonones and isoflavanones. This structural diversification of chromanone occupied an important role in pharmaceutical field as they owe numerous biological activities like anticancer, antioxidant, antidiabetic, anti-inflammatory, antiviral, antitubercular, antibacterial, antifungal, antiparasitic, anti-AchE, anticonvulsant, anti-HIV and antileishmanial properties. As chromanone scaffolds exhibit numerous potent biological activities, even some derivatives are novel like DSP-1053 (novel serotonin reuptake inhibitor and fast antidepressant), calanolide A (anti-HIV), Silibinin and chrysin(anticancer), taxifolin (antidiabetic), tetrazole (antidiabetic), troglitazone (antidiabetic), ormeloxifene (anticancer) and nebivolol (beta-blockers); nevertheless, the market proportion of potent chromanone analogs is fewer. Therefore, for future prospective, more consideration is required for designing and developing potent synthetic chromanone analogs which may provide better therapeutic value.
References
Emami, S.; Ghanbarimasir, Z.: Recent advances of chroman-4-one derivatives: synthetic approaches and bioactivities. Eur. J. Med. Chem. 93, 539–563 (2015)
Panche, A.N.; Diwan, A.D.; Chandra, R.: Flavonoids: an overview. J. Nutr. Sci. 5(47), 1–15 (2016)
Christophe Carola H., Ralf Rosskopf M.: Use of chroman-4-one derivatives.: United States Patent application publication., US 2010/0028278 A1,1, 1–24 (2010).
Cotelle, N.: Role of flavonoids in oxidative stress. Curr. Top. Med. Chem. I(2001), 569–590 (2001)
Jaracz, S.; Chen, J.; Kuznetsova, L.V., et al.: Recent advances in tumor-targeting anticancer drug conjugates. Biomed. Pharmacother. 13, 5043–5054 (2005)
Schindler, R.; Mentlein, R.: Flavonoids and vitamin E reduce the release of the angiogenic peptide vascular endothelial growth factor from human tumor cells. J. Nutr. 136, 1477–1482 (2006)
Piyush, K.; Kuldeep, S.; Azizur, R.M., et al.: A review of benzopyran derivatives in pharmacotherapy of breast cancer. Asian J. Pharm. Clin. Res. 11(7), 43–46 (2018)
Raj, V.; Lee, J.: 2H/4H-Chromenes-A versatile biologically attractive scaffold. Front. Chem. 8, 623 (2020)
Felgines, C.; Texier, O.; Morand, C., et al.: Bioavailability of the flavanone naringenin and its glycosides in rats. Am. J. Physiol. Gastrointest. Liver Physiol. 279(6), 1148–1154 (2000)
Kodama, O.: Sakuranetin, a flavanone ultraviolet-irradiated phytoalexin from rice leaves. Phytochemistry 31(11), 3807–3809 (1992)
Ley, J.P.: Evaluation of bitter masking flavanones from herba santa (Eriodictyon californicum (H. & A.) Torr., Hydrophyllaceae ). J. Agric. Food Chem. 53, 6061–6066 (2005)
Kumar, D.; Sharma, P.; Singh, H., et al.: The value of pyrans as anticancer scaffolds in medicinal chemistry. RSC Adv. 7(59), 36977–36999 (2017)
Gordo, J.; Cabrita, E.; Oliva, A., et al.: Thymus mastichina: chemical constituents and their anti-cancer activity. Nat. Prod. Commun. 7(11), 1491–1494 (2012)
Lopez-Lazaro, M.: Flavonoids as anticancer agents: structure-activity relationship study. Curr. Med. Chem. - Anti-Cancer Agents. 2(6), 691–714 (2002)
Kopustinskiene, D.M.; Jakstas, V.; Savickas, A., et al.: Flavonoids as anticancer agents. Nutrients 12(2), 1–24 (2020)
Noushini, S.; Alipour, E.; Emami, S., et al.: Synthesis and cytotoxic properties of novel (E)-3-benzylidene-7- methoxychroman-4-one derivatives. DARU J. Pharm. Sci. 21(1), 1–10 (2013)
Ivanova, L.; Varinska, L.; Pilatova, M., et al.: Cyclic chalcone analogue KRP6 as a potent modulator of cell proliferation: An in vitro study in HUVECs. Mol. Bio. Rep. 40(7), 4571–4580 (2013)
Alipour, E.; Mousavi, Z.; Safaei, Z., et al.: Synthesis and cytotoxic evaluation of some new[1,3]dioxolo[4,5-g]chromen-8-one derivatives. DARU J. Pharm. Sci. 22, 41 (2014)
Bahram, L.: Synthesis and in vitro cytotoxic activity of novel chalcone-like agents. Iran J. Basic Med. Sci. 16, 1155–1162 (2013)
Shen, S.; Ko, H.; Tseng, S., et al.: Structurally related antitumor effects of flavanones in vitro and in vivo: involvement of caspase 3 activation, p21 gene expression, and reactive oxygen species production. Toxicol. Appl. Pharmacol. 197, 84–95 (2004)
Choi, E.J.; Lee, J.I.; Kim, G.: Effects of 4 ’, 7-Dimethoxyflavanone on cell cycle arrest and apoptosis in human breast cancer MCF-7 Cells. Arch. Pharm. Res. 34(12), 2125–2130 (2011)
Safavi, M.; Esmati, N.; Kabudanian, S., et al.: Halogenated flavanones as potential apoptosis-inducing agents: synthesis and biological activity evaluation. Eur. J. Med. Chem. 58, 573–580 (2012)
Orlikova, B.: Methylenedioxy flavonoids: assessment of cytotoxic and anti-cancer potential in human leukemia cells. Eur. J. Med. Chem. 84, 173–180 (2014)
Shi, L.; Feng, X.E.; Rong, J., et al.: Synthesis and biological activity of flavanone derivatives. Bioorg. Med. Chem. Lett. 20(18), 5466–5468 (2010)
Kanagalakshmi, K.; Premanathan, M.; Priyanka, R., et al.: Synthesis, anticancer and antioxidant activities of 7-methoxyisoflavanone. Eur. J. Med. Chem. 45(6), 2447–2452 (2010)
Kupcewicz, B.; Balcerowska-Czerniak, G.; Małecka, M., et al.: Structure-cytotoxic activity relationship of 3-arylideneflavanone and chromanone (E, Z isomers) and 3-arylflavones. Bioorganic Med. Chem. Lett. 23(14), 4102–4106 (2013)
Simon, L.; Abdul Salam, A.A.; Kumar, S., et al.: Synthesis, anticancer, structural, and computational docking studies of 3-benzylchroman-4-one derivatives. Bioorg. Med. Chem. Lett. 27(23), 5284–5290 (2017)
Holshouser, M.H.; Loeffler, J.L.: Synthesis and antitumor testing of 3-methenylthiochroman-4-one-l,1 -dioxide. J. Pharm. Sci. 71(6), 715–717 (1982)
Ei-fotooh, A.: Synthesis of novel tricyclic heterocyclic compounds as potential anticancer agents using chromanone and thiochromanone as synthons. Indian J. Chem. -Section B. 42(august), 1985–1993 (2003)
Abdelatef, S.A.; El-Saadi, M.T.; Amin, N.H., et al.: Synthesis and anticancer screening of novel spiro[chroman-2,4’- piperidin]-4-one derivatives with apoptosis-inducing activity. J. Appl. Pharm. Sci. 8(1), 009–016 (2018)
Maasomi, Z.J.; Soltanahmadi, Y.P.; Dadashpour, M., et al.: Synergistic anticancer effects of silibinin and chrysin in T47D breast cancer cells. Asian Pacific J. Cancer Prev. 18(5), 1283–1287 (2017)
Murti, Y.; Mishra, P.: Synthesis and evaluation of flavanones as anticancer agents. Indian J. Pharm. Sci. 76(2), 163–166 (2014)
Gaspar, A.; Mohabbati, M.; Cagide, F., et al.: Searching for new cytotoxic agents based on chromen-4-one and chromane-2,4-dione scaffolds. Res. Pharm. Sci. 14(1), 74–83 (2019)
Demirayak, S.; Yurttas, L.; Gundogdu-Karaburun, N., et al.: New chroman-4-one/thiochroman-4-one derivatives as potential anticancer agents. Saudi Pharm. J. 25(7), 1063–1072 (2017)
Hegab, M.I., Morsy. E, Mageed. A.E. et al.: Synthesis and characterization of new 3′′,5′′-diaryl-3′′H,4′H-dispiropyran/thiopyran[4,2′-chroman-3′,2′′-[1,3,4-thiadiazol]-4′-one derivatives and related compounds as anticancer and antiviral agents, Phosphorus, Sulfur, and Silicon and the Related Elements. 190(11):00–00 (2015).
Siddiqui, Z.N.; Farooq, F.A.: Practical one pot synthesis of novel 2-hydroxy-4-chromanone derivatives from 3-formylchromone. J. Chem. Sci. 124(5), 1097–1105 (2012)
Pontius, A.; Krick, A.; Kehraus, S., et al.: Noduliprevenone: A novel heterodimeric chromanone with cancer chemopreventive potential. Chem. Eur. J 14, 9860–9863 (2008)
Bolwell, G.P.; Wojtaszek, P.: Mechanisms for the generation of reactive oxygen species in plant defence: a broad perspective. Physiol. Mol. Plant Pathol. 51(6), 347–366 (1997)
Chen, Q.; Vazquez, E.J.; Moghaddas, S., et al.: Production of reactive oxygen species by mitochondria: central role of complex III. J. Biol. Chem. 278(38), 36027–36031 (2003)
Miyake, Y.; Yamamoto, K.; Tsujihara, N., et al.: Protective effects of lemon flavonoids on oxidative stress in diabetic rats. Lipids 33(7), 689–695 (1998)
Gholamian-Dehkordi, N.; Luther, T.; Asadi-Samani, M., et al.: An overview on natural antioxidants for oxidative stress reduction in cancers; a systematic review. Immunopathol. Persa. 3(2), 12 (2017)
Karak, P.: Biological activities of flavonoids: an overview. Int. J. Pharm. Sci. Res. 10(4), 1567–1574 (2019)
Chen, X.; Mukwaya, E.; Wong, M.S., et al.: A systematic review on biological activities of prenylated flavonoids. Pharm. Biol. 52, 655–660 (2014)
Huyut, Z.; Beydemir, S.; Gulcin, I.: Antioxidant and antiradical properties of selected flavonoids and phenolic compounds. Biochem. Res. Int. 2017, 7616791 (2017)
Foroumadi, A.; Samzadeh-Kermani, A.; Emami, S., et al.: Synthesis and antioxidant properties of substituted 3-benzylidene-7-alkoxychroman-4-ones. Bioorganic Med. Chem. Lett. 17(24), 6764–6769 (2007)
Lee, H.; Lee, K.; Jung, J.K., et al.: Synthesis and evaluation of 6-hydroxy-7-methoxy-4-chromanone- and chroman-2-carboxamides as antioxidants. Bioorganic Med. Chem. Lett. 15(11), 2745–2748 (2005)
Ayunda, R.D.; Prasetyastuti, H.P.: Effect of 7-hydroxy-2-(4-hydroxy-3-methoxyphenyl)-chroman-4-one on level of mangan-superoxide dismutase (Mn-sod) and superoxide dismutase 2 (SOD2) gene expression in hyperlipidemia rats. Indones. J. Pharm. 30(3), 180–186 (2019)
Wang, D.; Zeng, L.; Li, D., et al.: Antioxidant activities of different extracts and homoisoflavanones isolated from the Polygonatum odoratum. Nat. Prod. Res. 27(12), 1111–1114 (2013)
Mistry, B.; Patel, R.V.; Keum, Y.: Access to the substituted benzyl-1, 2, 3-triazolyl hesperetin derivatives expressing antioxidant and anticancer effects. Arab. J. Chem. 10(2), 157–166 (2017)
Calvo, M.I.: Homoisoflavanones from Ledebouria floribunda. Fitoterapia 80(2), 96–101 (2009)
Hu, H.; Chen, X.; Sun, K., et al.: Silver-catalyzed radical cascade cyclization toward 1,5-/1,3-dicarbonyl heterocycles: an atom-/step-economical strategy leading to chromenopyridines and isoxazole-/pyrazole-containing chroman-4-ones. Org. Lett. 20(19), 6157–6160 (2018)
Roy, S.K.; Agrahari, U.C.; Gautam, R., et al.: Isointricatinol, a new antioxidant homoisoflavonoid from the roots of Caesalpinia digyna Rottler. Nat. Prod. Res. 26(8), 690–695 (2012)
Sun, Y.X.; Tang, Y.; Wu, A.L., et al.: Neuroprotective effect of liquiritin against focal cerebral ischemia/reperfusion in mice via its antioxidant and antiapoptosis properties. J. Asian Nat. Prod. Res. 12(12), 1051–1060 (2010)
Gazak, R.; Svobodova, A.; Psotova, J., et al.: Oxidised derivatives of silybin and their antiradical and antioxidant activity. Bioorganic Med. Chem. 12(21), 5677–5687 (2004)
Peng, Z.Y.; Liu, X.Y.; Yang, Y.L., et al.: 7-Methoxy-3-(4-methoxyphenyl) chroman-4-one. Acta Cryst. E68, 250 (2012)
Zheng, Y.Z.; Deng, G.; Chen, D.F., et al.: Theoretical studies on the antioxidant activity of pinobanksin and its ester derivatives: effects of the chain length and solvent. Food Chem. 240, 323–329 (2018)
Siddaiah, V.; Maheswara, M.; Venkata Rao, C., et al.: Synthesis, structural revision, and antioxidant activities of antimutagenic homoisoflavonoids from Hoffmanosseggia intricata. Bioorg. Med. Chem. Lett. 17(5), 1288–1290 (2007)
Hadaruga, D.I.; Hadaruga, N.G.: Antioxidant activity of hepatoprotective silymarin and silybum marianum L. extract. Chem. Bull. 54(68), 104–107 (2009)
Zhao, J.W.; Chen, D.S.; Deng, C.S., et al.: Evaluation of anti-inflammatory activity of compounds isolated from the rhizome of Ophiopogon japonicas. BMC Complement Altern. Med. 17(1), 1–12 (2017)
Ali, K.A.; Abdelhafez, N.A.A.; Ragab, E.A., et al.: Design and synthesis of novel fused heterocycles using 4-chromanone as synthon. Russ. J. Gen. Chem. 85(12), 2853–2860 (2015)
Waller, C.P.; Thumser, A.E.; Langat, M.K., et al.: COX-2 inhibitory activity of homoisoflavanones and xanthones from the bulbs of the southern african ledebouria socialis and ledebouria ovatifolia (Hyacinthaceae: Hyacinthoideae). Phytochemistry 95(6), 284–290 (2013)
Ma, J.; Cao, B.; Chen, X., et al.: Violacin A, a new chromanone produced by Streptomyces violaceoruber and its anti-inflammatory activity. Bioorganic Med. Chem. Lett. 28(5), 947–951 (2018)
Wang, Q.Q.; Shi, J.B.; Chen, C., et al.: Hesperetin derivatives: Synthesis and anti-inflammatory activity. Bioorganic Med. Chem. Lett. 26(5), 1460–1465 (2016)
Shaikh, M.M.; Kruger, H.G.; Bodenstein, J., et al.: Anti-inflammatory activities of selected synthetic homoisoflavanones. Nat. Prod. Res. 26(16), 1473–1482 (2012)
Hu, C.; Zhou, Z.; Xiang, Y., et al.: Design, synthesis and anti-inflammatory activity of dihydroflavonol derivatives. Med. Chem. Res. 27(1), 194–205 (2018)
Zheng, C.; Wang, L.; Han, T., et al.: Pruinosanones A-C, anti-inflammatory isoflavone derivatives from Caragana pruinosa. Sci Rep. 6, 2–9 (2016)
Winekenstadde, D.; Angelis, A.; Waltenberger, B., et al.: Phytochemical profile of the aerial parts of sedum sediforme and anti-inflammatory activity of myricitrin. Nat. Prod. Commun. 10(1), 83–88 (2015)
Wright, George C., Goldenberg, Marvin M.: 7-Dimethylamino-4-chromanoneUnited StatesMorton-Norwich Products, Inc. (Norwich, NY), 4108872 (1978).
Edwards, J.L.; Vincent, A.M.; Cheng, H.T., et al.: Diabetic neuropathy: Mechanisms to management. Pharmacol. Therap. 2008, 1–34 (2008)
Unnikrishnan, M.K.; Veerapur, V.; Nayak, Y., et al.: Antidiabetic, antihyperlipidemic and antioxidant effects of the flavonoids. Polyphenols Hum. Health Dis 2013, 143–161 (2013)
Adams, G.L.; Velazquez, F.; Jayne, C., et al.: Discovery of chromane propionic acid analogues as selective agonists of GPR120 with in vivo activity in rodents. ACS Med. Chem. Lett. 8(1), 96–101 (2017)
Takao, K.; Yamashita, M.; Yashiro, A.; Sugita, Y.: Synthesis and biological evaluation of 3-Benzylidene-4-chromanone derivatives as free radical scavengers and α-Glucosidase inhibitors. Chem. Pharm. Bull. 64(8), 1203–1207 (2016)
Zhu, G.; Luo, Y.; Xu, X., et al.: Anti-diabetic compounds from the seeds of Psoralea corylifolia. Fitoterapia 139,(2019)
Kocevar, N.; Glavac, I.; Kreft, S.: Flavonoidi. Farm Vestn. 58(4), 145–148 (2007)
Lotulung, P.D.N.; Fajriah, S.; Sundowo, A., et al.: Anti diabetic flavanone compound from the leaves of Artocarpus communis. Indones. J. Chem. 9(3), 505–508 (2010)
Sadewa, A.H.: Effect of 7-hydroxy-2- (4-hydroxy-3-methoxy-phenyl ) - chroman-4-one ( swietenia macrophylla king seed ) on retinol binding protein-4 and phosphoenolpyruvate carboxykinase gene expression in type 2 diabetic rats. Rom. J. Diabetes Nutr. Metab. Dis. 23(3), 255–265 (2016)
Sneha Jose, E.; Philip, J.E.; Shanty, A.A.: Novel class of mononuclear 2-methoxy-4-chromanones ligated Cu (II), Zn (II), Ni (II) complexes: synthesis, characterisation and biological studies. Inorganica Chim. Acta. 478(I), 155–165 (2018)
Park, J.; Seo, Y.; Han, J.: in vitro and alleviates postprandial hyperglycemia in diabetic mice. Eur. J. Pharmacol. 863(September),(2019)
Ravi, S.; Sadashiva, C.T.; Tamizmani, T.: In vitro glucose uptake by isolated rat hemi-diaphragm study of Aegle marmelos correa root. Bangladesh J. Pharmacol. 4(1), 65–68 (2009)
Raju, B.C.; Tiwari, A.K.; Kumar, J.A., et al.: α-Glucosidase inhibitory antihyperglycemic activity of substituted chromenone derivatives. Bioorg. Med. Chem. 18(1), 358–365 (2010)
Shaikh, M.M.; Kruger, H.G.; Smith, P., et al.: Crystal structure and potent antifungal activity of synthetic homoisoflavanone analogues. J. Pharm. Res. 6(1), 1–5 (2013)
Tiratsuyan, S.G.; Hovhannisyan, A.A.; Karapetyan, A.V., et al.: Synthesis and biological activities of novel pyridazine derivatives. Mor. J. Chemi. 63(5), 698–704 (2016)
Ayati, A.; Falahati, M.; Irannejad, H., et al.: Synthesis, in vitro antifungal evaluation and in silico study of 3-azolyl-4-chromanone phenylhydrazones. DARU J. Pharm. Sci. 20(46), 1–7 (2012)
Dongamanti, A.; Naji, H.H.; Bommidi, V.L., et al.: Microwave-assisted one-pot synthesis and antimicrobial evaluation of 2-(1-phenyl-3-(2-thienyl)-1H-pyrazol-4-yl)chroman-4-one derivatives. Heterocycl. Commun. 22(5), 259–264 (2016)
Ashok, D.; Kumar, R.S.: Solvent-free microwave-assisted synthesis and biological evaluation of aurones and flavanones based on 2, 2-dimethylchroman-4-one. Chem. Heterocycl. Compd. 52(7), 453–459 (2016)
Hoettecke, N.; Rotzoll, S.; Albrecht, U.: Synthesis and antimicrobial activity of 2-alkenylchroman-4-ones, 2-alkenylthiochroman-4-ones and 2-alkenylquinol-4-ones. Bioorg. Med. Chem. 16(24), 10319–10325 (2008)
Kang, J.G.; Shin, S.Y.; Kim, M.J., et al.: Isolation and anti-fungal activities of 2-hydroxymethyl-chroman-4-one produced by Burkholderia sp. MSSP. J. Antibiot. 57(11), 726–731 (2004)
Albrecht, U.; Lalk, M.; Langer, P.: Synthesis and structure-activity relationships of 2-vinylchroman-4-ones as potent antibiotic agents. Bioorg. Med. Chem. 13(5), 1531–1536 (2005)
Sriram, D.; Srinivasan, S.; Santhosh, K.C.: 3-phenylsulfonyl-3-(2-propenyl)chroman-4-one. Acta Crystallogr. Cryst Struct. Commun. 53(6), 793–794 (1997)
Sharma, S.; Patial, V.; Singh, D., et al.: Antimicrobial homoisoflavonoids from the Rhizomes of Polygonatum verticillatum. Chem. Biodivers. 15(12), 1800430 (2018)
Feng, L.; Maddox, M.M.; Alam, M.Z., et al.: Synthesis, structure-activity relationship studies, and antibacterial evaluation of 4-chromanones and chalcones, as well as olympicin A and derivatives. J. Med. Chem. 57(20), 8398–8420 (2014)
Albogami, A.S.; Alkhathlan, H.Z.; Saleh, T.S., et al.: Microwave-assisted synthesis of potent antimicrobial agents of flavanone derivatives. Orient J. Chem. 30(2), 435–443 (2014)
Pawar, M.J.; Burungale, B.; Karale, B.K.: Synthesis and antimicrobial activity of spiro [chromeno [4, 3- thiadiazoline compounds. ARKIVOC: Online. J. Org. Chem. 2009, 97–107 (2009)
Ramana Kishore, N.; Ashok, D.; Sarasija, M., et al.: Microwave-assisted synthesis of novel spirochromanone–aurone hybrids and their antimicrobial activity. Russ. J. Gen. Chem. 88(5), 1015–1019 (2018)
Alsarahni, A.; Eldeen, Z.M.; Alkaissi, E., et al.: Synthesis and structural elucidation of amino acetylenic and thiocarbamates derivatives for 2-mercaptobenzothiazole as antimicrobial agents. Int. J. Pharm. Sci. 9(2), 192 (2017)
Qing-hui, X.U.; Ji-zhen, L.I.; Jiang-hua, H.E., et al.: Synthesis of novel flavanone derivatives and their anti Staphylococcus aureus evaluation. Chem. Res. Chin. Univ. 29(4), 695–698 (2013)
Thinagar, S.; Velmurugan, D.; Amalraj, R.R.: Crystal structure of spiro [2-benzoyl-cyclohexyl-4, 5-diphenylpyrrolidine-3,3’-chroman-4-one]. Cryst Res. Technol. 35(8), 979–986 (2000)
Agnes Kenez, L.J., et al.: A simple synthesis of selinone, an antifungal component of monotes engleri. Heterocycl. Commun. 8(6), 543–548 (2002)
Kaufmann, S. H. E., van Helden P.: Handbook of tuberculosis: Clinics, Diagnostics. Wiley-VCH, therapy and epidemiology. Weinheim (2008).
Saengchantara, S.T.; Wallace, T.W.: Chromanols, chromanones, and chromones. Nat. Prod. Rep. 3, 465–475 (1986)
Alvey, L.; Prado, S.; Saint-joanis, B., et al.: Diversity-oriented synthesis of furo [3, 2- f ] chromanes with antimycobacterial activity. Eur. J. Med. Chem. 44, 2497–2505 (2009)
Wu, M.; Peng, C.; Chen, I., et al.: Antitubercular chromones and flavonoids from Pisonia aculeata. J. Nat. Prod. 5(74), 976–982 (2011)
Pini, E.; Poli, G.; Tuccinardi, T., et al.: New chromane-based derivatives as inhibitors of mycobacterium tuberculosis salicylate synthase: Preliminary biological evaluation and molecular modeling studies. Molecules 23(7), 1–14 (2018)
Yempala, T.; Sriram, D.; Yogeeswari, P., et al.: Molecular hybridization of bioactives: synthesis and antitubercular evaluation of novel dibenzofuran embodied homoisoflavonoids via Baylis-Hillman reaction. Bioorganic Med. Chem. Lett. 22(24), 7426–7430 (2012)
Asif, M.A.: Brief overview on recent advances in the development of anti-tubercular compounds containing different heterocyclic ring systems. Appl. Microbiol. Open Access. 02(04), 1–14 (2016)
Roy, S.K.; Kumari, N.; Gupta, S., et al.: 7-Hydroxy-(E)-3-phenylmethylene-chroman-4-one analogues as efflux pump inhibitors against Mycobacterium smegmatis mc2 155. Eur. J. Med. Chem. 66, 499–507 (2013)
Anand, N.; Singh, P.; Sharma, A., et al.: Synthesis and evaluation of small libraries of triazolylmethoxy chalcones, flavanones and 2-aminopyrimidines as inhibitors of mycobacterial FAS-II and PknG. Bioorg. Med. Chem. 20(17), 5150–5163 (2012)
Mujahid, M.; Gonnade, R.G.; Yogeeswari, P., et al.: Synthesis and antitubercular activity of amino alcohol fused spirochromone conjugates. Bioorg. Med. Chem. Lett. 23(5), 1416–1419 (2013)
Donnell, G.; Bucar, F.; Gibbons, S.: Phytochemistry and antimycobacterial activity of Chlorophytum inornatum. Phytochemistry 67, 178–182 (2006)
Dongamanti, A.; Aamate, V.K.; Devulapally, M.G., et al.: Bis-spirochromanones as potent inhibitors of Mycobacterium tuberculosis: synthesis and biological evaluation. Mol. Divers. 21(4), 999–1010 (2017)
Lee H, Kr D, Kim JH.: ( 12 ) United States Patent. Vol. 2. 1–5(2011).
Gaikwad, M.S.; Mane, A.S.; Hangarge, R.V., et al.: Synthesis of newer selenadiazoles and thiadiazoles from their chroman-4-one precursors. Indian J. Chem. Sect. B. 42(January), 189–191 (2003)
Tait, S.; Laura, A.; Desideri, N.; Fiore, L.: Antiviral activity of substituted homoisoflavonoids on enteroviruses. Antiviral Res. 72, 252–255 (2006)
Conti, C.; Proietti, L.; Desideri, N.: Design, synthesis and in-vitro evaluation of novel chroman-4-one, chroman, and 2 H-chromene derivatives as human rhinovirus capsid-binding inhibitors. Bioorg Med Chem. 19(24), 7357–7364 (2011)
Quaglia, M.G.; Desideri, N.; Bossu, E., et al.: Enantioseparation and anti-rhinovirus activity of 3-Benzylchroman-4-ones. Chirality 500, 495–500 (1999)
Kang, Y.; Mei, Y.; Du, Y.; Jin, Z.: Total synthesis of the highly potent anti-HIV natural product daurichromenic acid along with its two chromane derivatives, rhododaurichromanic acids A and B. Organic Lett. 5(23), 4481–4484 (2003)
Rama Rao, A.V.; Galtonde, A.S., et al.: A conclse selective of chlral of the chromanol wolety of anti-HIV agent calanolide A. Tetrahedron Lett. 35(34), 6347–6350 (1994)
Ishikawa, T.; Oku, Y.; Tanaka, T., et al.: An approach to anti-HIV-1 active Calophyllum coumarin synthesis: an enantioselective construction of 2,3-dimethyl-4-chromanone ring by quinineassisted intramolecular Michael-type addition. Tetrahedron Lett. 40(19), 3777–3780 (1999)
Pokorna, J.; Machala, L.; Rezacova, P.; Konvalinka, J.: Current and novel inhibitors of HIV protease. Viruses 1, 1209–1239 (2009)
Dawood, K.M.: Regio and stereoselective synthesis of bis-spiropyrazoline-5,3′- chroman(thiochroman)-4-one derivatives via bis-nitrilimines. Tetrahedron 61(22), 5229–5233 (2005)
Sekino, E.; Kumamoto, T.; Tanaka, T., et al.: Concise synthesis of anti-HIV-1 Active (+)-Inophyllum B and (+)-Calanolide A by application of (-)-quinine-catalyzed intramolecular oxo-michael addition. J. Org. Chem. 69(8), 2760–2767 (2004)
Park, J.H.; Lee, S.U.; Kim, S.H., et al.: Chromone and chromanone derivatives as strand transfer inhibitors of HIV-1 integrase. Arch. Pharm. Res. 31(1), 1–5 (2008)
Nagle, A.S.; Khare, S.; Kumar, A.B., et al.: Recent developments in drug discovery for leishmaniasis and human African trypanosomiasis. Chem. Rev. 114, 11305–11347 (2014)
Kaye, P.; Scott, P.: Leishmaniasis: complexity at the host-pathogen interface. Nat. Publ. Gr. 9(8), 604–615 (2011)
Boer, M.D.; Argaw, D.; Jannin, J., et al.: Leishmaniasis impact and treatment access. Clin Microbiol. Infect. 17(10), 1471–1477 (2011)
Cywin, C.L.; Firestone, R.A.; Mcneil, D.W., et al.: The design of potent hydrazones and disulfides as cathepsin S inhibitors. Bioorg. Med. Chem. 11, 733–740 (2003)
Das, P.; De, T.; Chakraborti, T.: Leishmania donovani secretory serine protease alters macrophage inflammatory response via COX-2 mediated PGE-2 production. Indian J. Biochem. Biophys. 51(December), 542–551 (2014)
Id, E.V.; Echeverri, F.; Upegui, Y.A., et al.: Hydrazone derivatives enhance antileishmanial activity of thiochroman-4-ones. Molecules 23(70), 1–12 (2018)
Upegui, Y.; Rios, K.; Quinones, W., et al.: Chroman-4-one hydrazones derivatives: synthesis, characterization, and in vitro and in vivo antileishmanial effects. Med. Chem. Res. 28(12), 2184–2199 (2019)
Castro, H.; Aguiar, P.D.; Cardoso, S., et al.: Synthesis and evaluation of novel chromanone and quinolinone analogues of uniflorol as anti-leishmanial agents. Heliyon. 6(March), 1–11 (2020)
Roberson, E.D.; Mucke, L.: 100 years and counting: Prospects for defeating Alzheimer’s disease. Science 80(314), 781–784 (2006)
Association, A.: Alzheimer ’ s disease facts and figures. Alzheimer’s and Dementia. 15(3), 321–387 (2019)
Jung, M.; Park, M.: Acetylcholinesterase inhibition by flavonoids from agrimonia pilosa. Molecules 12, 2130–2139 (2007)
Shen, Y.; Zhang, J.; Sheng, R., et al.: Synthesis and biological evaluation of novel flavonoid derivatives as dual binding acetylcholinesterase inhibitors. J. Enzyme Inhib. Med. Chem. 24(April), 372–380 (2009)
Parveen, M.; Malla, A.M.; Yaseen, Z., et al.: Synthesis, characterization, DNA-binding studies and acetylcholinesterase inhibition activity of new 3-formyl chromone derivatives. J. Photochem. Photobiol. B Biol. 130, 179–187 (2014)
Anand, P.; Singh, B.: A review on cholinesterase inhibitors for Alzheimer’s disease. Arch. Pharm. Res. 36(4), 375–399 (2013)
Shamsimeymandi, R.; Pourshojaei, Y.; Eskandari, K., et al.: Design, synthesis, biological evaluation, and molecular dynamics of novel cholinesterase inhibitors as anti-Alzheimer’s agents. Arch. Pharm. 352, 7 (2019)
Jiang, N.; Ding, J.; Liu, J., et al.: Novel chromanone-dithiocarbamate hybrids as multifunctional AchE inhibitors with β-amyloid anti-aggregation properties for the treatment of Alzheimer’s disease. Bioorg. Chem. 89(May),(2019)
Li, Y.; Qiang, X.; Luo, L., et al.: Multitarget drug design strategy against Alzheimer’s disease: Homoisoflavonoid mannich base derivatives serve as acetylcholinesterase and monoamine oxidase B dual inhibitors with multifunctional properties. Bioorg. Med. Chem. 25(2), 714–726 (2017)
Kwan, P.; Brodie, M.J.: Early identification of refractory epilepsy. N. Engl. J. Med. 342(5), 314–319 (2000)
Sabers, A.; Gram, L.: Newer anticonvulsants comparative review of drug interactions and adverse effects. Drugs 60(1), 23–33 (2000)
Gentry, J.R.; Hill, C.; Malcolm, R.: New Anticonvulsants: a review of applications for the management of substance abuse disorders. Ann. Clin. Psychiatry. 14(4), 233–245 (2002)
Emami, S.; Kebriaeezadeh, A.; Ahangar, N., et al.: Imidazolylchromanone oxime ethers as potential anticonvulsant agents : Anticonvulsive evaluation in PTZ-kindling model of epilepsy and SAR study. Bioorg. Med. Chem. Lett. 21(2), 655–659 (2011)
Kebriaeezadeh, A.; Emami, S.; Ebrahimi, M.: Anticonvulsant and antiepileptogenic effects of azolylchromanones on lithium pilocarpine induced seizures and pentylenetetrazole-kindling model of epilepsy. Biomed. Pharmacother. 62, 208–211 (2008)
Emami, S.; Kebriaeezadeh, A.; Zamani, J.; Shafiee, A.: Azolylchromans as a novel scaffold for anticonvulsant activity. Bioorganic Med. Chem. Lett. 16, 1803–1806 (2006)
Tang, S.W.; Helmeste, D.M.; Leonard, B.E.: Antidepressant compounds: a critical review. Depression Psychopathol. Pharmacother. 27, 1–19 (2010)
Wang, Y.; Sun, Y.; Guo, Y., et al.: Dual functional cholinesterase and MAO inhibitors for the treatment of Alzheimer ’ s disease : synthesis, pharmacological analysis and molecular modeling of homoisoflavonoid derivatives Dual functional cholinesterase and MAO inhibitors for the treatment. J. Enzyme Inhib. Med. Chem. march 2015, 1475–6374 (2015)
Dupuy, J.M.; Ostacher, M.J.; Huffman, J., et al.: A critical review of pharmacotherapy for major depressive disorder. Int. J. Neuropsychopharmacol. 14, 1417–1431 (2011)
Kato, T.; Matsumoto, Y.; Yamamoto, M., et al.: DSP-1053, a novel serotonin reuptake inhibitor with 5-HT 1A partial agonistic activity, displays fast antidepressant effect with minimal undesirable effects in juvenile rats. Pharmacol. Res. Perspect. 3(3), 1–17 (2015)
Desideri, N.; Fioravanti, R.; Biava, M., et al.: (E)-3-Heteroarylidenechroman-4-ones as potent and selective monoamine oxidase-B inhibitors Nicoletta. Eur. J. Med. Chem. 3, 1–30 (2016)
Seifert, T.; Malo, M.; Kokkola, T., et al.: Chroman-4-one- and chromone-based sirtuin 2 inhibitors with antiproliferative properties in cancer cells. J. Med. Chem. 57, 9870–9888 (2014)
Lan, J.S., Xie, S.S., Huang, M., et al.: Chromanones: selective and reversible Monoamine Oxidase B inhibitors with nanomolar potency. Med. chem. comm.1–33(2015).
Toya, T.; Fukasawa, H.; Masui, A.; Endo, Y.: Potent and selective partial Ecdysone agonist activity of chromafenozide in Sf9 Cells. Biochem. Biophys. Res. Commun. 292, 1087–1091 (2002)
Wing, K.D.: 5849, a nonsteroidal Ecdysone agonist: effects on a drosophila cell line. Science 241(4864), 467–469 (1988)
Michaell, G.: Recent advances in our knowledge of ecdysteroid biosynthesis in insects and crustaceans. Insect. Biochem. Molec. Biol. 24(2), 115–132 (1994)
Zhao, P.; Li, J.; Yang, G.: Synthesis and insecticidal activity of chromanone and chromone analogues of diacylhydrazines. Bioorg. Med. Chem. 15, 1888–1895 (2007)
Dev, S.; Koul, O.: Insecticides of natural origin. Harwood Academic Publishers, The Netherlands (1997)
Gan, C.; Zhao, Z.; Nan, D., et al.: Homoisoflavonoids as potential imaging agents for b -amyloid plaques in Alzheimer ’ s disease. Eur. J. Med. Chem. 76, 125–131 (2014)
Ramana, M.M.V.; Ranade, P.B.; Betkar, R.R., et al.: Flavanones : Potential antidengue targets in silico approach. J. Chem. Pharm Res. 7(9), 469–474 (2015)
Pisa, F.D.; Landi, G.; Iacono, L., et al.: Chroman-4-one derivatives targeting pteridine reductase 1 and showing anti-parasitic activity. Molecules 22(426), 1–16 (2017)
Kwon, D.H., Choi, W.J., Lee, C.H., Kim, J.H., Kim, M.B.: Flavonoid compound having an antiviral activity.2. United States Patent, 1–5 (2011).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kamboj, S., Singh, R. Chromanone-A Prerogative Therapeutic Scaffold: An Overview. Arab J Sci Eng 47, 75–111 (2022). https://doi.org/10.1007/s13369-021-05858-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13369-021-05858-3