Abstract
Rotary blood pumps have the potential to become a viable long-term treatment option for the heart failure patients to bridge to transplantation or destination therapy. However, these devices operate at a constant speed which may lead to long term complications or they may operate in an undesired support mode such as excessive pumping in the patients’ body. A possible solution to such physiological problems or using these devices as a destination therapy or maintaining the optimal support level according to changing conditions in the body is applying a varying speed rotary blood pump support. Over the years, different varying operating speed support algorithms have been proposed to alleviate the effect of the constant speed rotary blood pump support and improve the outcome of these devices. In this paper, it is aimed to compile and present proposed varying speed rotary blood pump support algorithms by classifying them according to the considered physiological problem in each study.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Heart failure has a complex structure at organ and cellular levels, and is the end result of several different disease processes that may be subject to a variety of therapies and it is conventionally treated with inotropic support, diuretics, and moderate exercise. When these methods fail, especially towards the final stage of the disease, heart transplantation is called for. With the current state of donor organ supply, many patients would not be treated, due to lack of a fitting donor organ. For these patients, to bridge the time between the decision to transplant and the actual transplantation, cardiac assist devices have been introduced into clinical practice [1]. Rotary blood pumps (RBPs) represent the new generation of the mechanical assist device treatment of the heart failure. There are a number of advantages of these devices over pulsatile mechanical circulatory support systems such as having smaller size, less moving parts, and smaller blood contact surface. Overall, the good durability of the devices has led to acceptation of RBP support as a viable alternative for total heart transplantation [2].
RBPs are designed to operate at a constant speed in patient’s body [3–5] and conventionally they operate at a constant speed in the body. Such an operating mode may cause problems such as gastrointestinal bleeding or aortic valve insufficiency [3] in long term support although the aim of long-term RBP support is to reduce patient dependence on clinical management of the implanted device, and improve their quality of life. Improvement in this RBP support mode requires implementation of an automatic, adaptive and robust control system aiming to adjust pump output automatically according to the changes in the patient’s state. Such a task requires identification of the physiological mechanisms in the heart failure patients to design a controller that is able to automatically adapt according to patient’s physiological demands.
Developed varying speed RBP algorithms may help to improve certain physiological complications due to complex interactions between the organ functions and mechanisms in the cardiovascular system. Also there is a dynamic change in the hemodynamical signals over a cardiac cycle due to contraction of the heart. Each phase of the cardiac cycle is dependent on the different mechanisms and parameters such as atrial kick, ventricular contraction and relaxation, preload and afterload of the ventricle etc. Therefore only one type of varying speed operating mode will not provide a complete solution in long term RBP support for different type of the problems. Furthermore, the developed varying speed RBP algorithms will be complicated due to complex physiological mechanisms and requires estimation techniques which make it more complicated due to lack of reliable long term blood pressure sensors.
This paper aims to summarize the varying speed RBP applications that have been done to improve the outcome of the RBP support or to assess the cardiovascular physiology under varying speed RBP support. In this paper, varying RBP speed is defined as any change that has been applied actively in the operating speed during the pump support. Also, varying speed RBP operating mode is considered as the changes in the constant RBP operating speed to avoid the harmful effects of pump support such as ventricular suction and the dynamic RBP speed support over a cardiac cycle to improve outcomes the RBP support such as arterial pulsatility or aortic valve function.
Papers reviewing different topics in RBP applications have been published. Although these papers mention the varying operating speed RBP applications, they only refer the studies which are related a specific topic in the published paper and do not cover physiological effects of the varying speed RBP support. For instance, Bertram [6] reviews with a brief look at the wider measurement context for the RBPs that propose to use these devices to provide circulatory support. He refers the studies that describe the estimation or measurement of the parameters such as pressure, flow or motor current which can be used to develop varying speed RBP control algorithms. However, the varying speed applications are not covered in this study. AlOmari et al. [7] review the developments in control systems for RBPs for the heart failure patients. That paper focuses on the control system design for the RBPs. Although varying speed applications part of the control studies they do not have to mention the control design. Therefore it does not cover the varying speed RBP operation for the different physiological scenarios and lacks of the studies focusing on the physiology under varying speed RBP support. Amacher et al. [8] summarize the proposed RBP driving methods to increase the pulsatility. They describe a series of in vitro measurements to test the constant speed and various cases of speed modulation. However, this study focuses on ventricular unloading and pulsatility under varying speed RBP support and does not cover other outcomes of the RBP support such as aortic valve function, sufficient perfusion or more physiological hemodynamic signals etc.
Rotary blood pump speed regulation
In this paper, varying speed RBP support algorithms are classified according to considered physiological case in each study. The studies proposing varying speed RBP support algorithms are summarized in Table 1 according to motivation in the cited paper and RBP speed regulation technique. In Table 1, RBP speed regulation technique does not include the utilized indicators to detect the physiological case but the feedback variable to regulate the RBP operating speed.
RBP speed regulation for generating sufficient blood flow and detecting ventricular suction
RBP characteristic response to alterations in hydrodynamic load is opposite to the requirement of the body. As a result of their pressure difference and flow rate characteristics, pressure generation decreases with increased pump flow rate at a constant speed while the body needs increased flow for increased pressure. Therefore, operating speed of an RBP should be adjusted according to changing preload and afterload to meet the physiological demand in a patient. Moreover, suction in the left ventricle may occur under relatively higher RBP operating speeds and it is associated with hemolysis, thrombus release and tissue damage at the inlet of the RBP [9]. In contrast, blood regurgitates through the pump under relatively lower operating speed RBP support [10]. Suction due to excessive pumping, reverse flow through the pump and level of the perfusion are related with flow and pressure generation by the RBP.
Direct measurement of the RBP flow rate and pressure difference across the RBP at the same time would provide optimum pump operating conditions while avoiding suction in the left ventricle and reverse pump flow. However, a reliable long term blood pressure sensor does not exist and only Micromed DeBakey RBP includes an attached flow sensor [11]. Therefore these variables were estimated by using pump motor current, operating speed or harmonic components in the proposed RBP control algorithms. For instance, increase of the higher power spectral density components indicates the occurrence of the suction in the left ventricle [12–15]. Power spectral density components of the RBP motor current and RBP flow rate for pump support under normal and suction conditions are given in Fig. 1.
Power spectral density components of the RBP motor current and RBP flow for normal support and suction conditions, i RBP motor current at 10,500 rpm operating speed, ii RBP flow at 10,500 rpm operating speed, iii power spectral density components of the RBP motor current at 10,500 rpm operating speed, iv Power spectral density components of the RBP flow at 10,500 rpm operating speed, v RBP motor current at 13500 rpm operating speed, vi RBP flow at 13,500 rpm operating speed, vii Power spectral density components of the RBP motor current at 13,500 rpm operating speed, viii Power spectral density components of the RBP flow at 13,500 rpm operating speed
Three peaks can be seen in the power spectral density graph for both RBP motor current and RBP flow under normal RBP support conditions at 10,500 rpm operating speed. When the RBP operating speed is increased to 13,500 rpm seven peaks become noticeable in the power spectral density graph. Moreover, shape of the RBP motor current and RBP flow signals change because of the suction resistance at the inlet of RBP. Such an algorithm provides the information about the occurrence of the suction in the left ventricle. Use of numerical models describing the electro–mechanical and hydraulic characteristics of the RBPs provide the information about instantaneous pump flow. Furthermore, contraction of the heart generates a pulsatile flow and differential pressure signals, thus pump motor current signals. However, pulsatility of these signals change with respect to RBP operating speed. This allows developing indicators for suction detection based on the pulsatility of the RBP flow rate and motor current signals [16–21]. RBP flow rate and differential pressure across the pump are given in Fig. 2 to show the change of the amplitude and pulsatility for the increased pump operating speed.
Amplitude of the RBP flow and differential pressure across the pump were higher than 11,500 rpm and 13,500 rpm at 9500 rpm pump operating speed. Amplitude of these signals becomes very low at 11,500 rpm. However, suction still does not occur in the left ventricle. Further increase in the RBP operating speed causes suction and amplitude of RBP flow rate signal increases considerably while differential pressure signal across the pump increased a little. Increased pulsatility in the RBP flow can be used as an indicator of suction in the left ventricle at the relatively higher pump operating speeds. Developed varying speed RBP support algorithms may require output of the patient’s native heart contractility and arterial pressure [17–20]. It should be noted that the proposed control algorithms using these indicators detect the suction in the in vivo experiments when these parameters are measured [22]. However, these parameters need to be estimated in a patient. Developed suction indexes namely pulsatility index algorithm, diminishing return algorithm and harmonic index algorithm help to assess the efficiency of different control methods regulating the RBP operating speed when the suction is detected. The main motivation to apply these different control algorithms is the parameter uncertainty in the RBP supported cardiovascular system [23]. Different control techniques such as fuzzy logic control also showed a good performance in RBP response to the suction when different pump flow rate pulsatility indexes were used [23–26]. These indexes were also used to develop RBP control algorithms for delivering sufficient blood to the arteries [27, 28]. It is possible to classify the severity of suction while regulating the RBP operating speed according to level of the patient’s activity by applying a ruled based control algorithm [29, 30]. Multi-objective RBP speed regulating algorithms were evaluated in the in vitro mock circulatory systems by using similar indicators to detect the suction and provide optimal perfusion [31]. However, these algorithms can detect suction occurrence point approximately while adjusting the RBP flow at an optimal rate in the in vivo models [32]. Detection of the exact suction occurrence point was presented in [33] when an in vivo model is used. Input of the RBP speed control algorithms is the motor current in a practical application. However, it is possible to use RBP operating speed as the input of the system independently in the simulation studies. Relation between the RBP motor current and the RBP operating speed and a control application based on RBP motor current to detect the suction are presented by Faragallah et al. [34]. A specialized suction detection algorithm for Micromed DeBakey RBP was presented in [35]. This algorithm evaluates the RBP motor signals and shows the change of the pump flow and motor current signals in occurrence of the suction. Since this device includes an attached flow sensor it is possible to measure and analyze the RBP flow signal to detect the suction. This system can also work autonomously besides the flow rate based suction algorithm [35].
A flow pattern classification algorithm including also suction detection indicators was presented in [36, 37]. This system utilizes a Lagrangian Support Vector Machine algorithm which can precisely classify pump flow patterns. However, it should be noted that this is not the first suction detection study while classifying the RBP flow patterns at the same time. Vollkron et al. [38–40] used seven different criteria to detect the suction in the left ventricle and eleven different algorithms to classify the RBP flow patterns. These algorithms classify the flow patterns by utilizing the history of the patient under RBP support, however, selection of the algorithms requires an expert to monitor the patient and apply them. A similar approach which requires the history of the patient under RBP support was applied by developing seven suction indicators according to the patient data [41]. However, it should be noted that such suction detection algorithms needing the patient data and history require an expert to select the suitable algorithm and it may not be always practical to apply because they are not automated. Another pump flow rate based suction detection and RBP control algorithm is presented in [42, 43]. Since the slope of minimum of the RBP flow rate increases continuously when the RBP operating speed also increases continuously, a negative change is seen in this slope at the onset of the suction. It is possible to detect the suction by utilizing the change of the slope of the RBP flow. However, such an RBP operating mode is not feasible to apply in a patient’s body because continuously increasing RBP speed inevitably will cause suction, so at a certain level the pump operating speed should be saturated.
RBP flow rate control is the simplest way of assuring the sufficient blood is delivered to provide optimum perfusion. However, RBP flow rate should be estimated since the heart pumps do not include a flow sensor. The studies focusing on the RBP flow rate control without suction detection algorithms show that it is possible to estimate the pump flow rate with a high accuracy [44, 45].
Selection of RBP differential pressure or aortic pressure as a control variable ensures a certain level of mean aortic pressure which is dependent on the pump pressure generation and strength of the contracting heart. Such an RBP speed regulation method is useful when the patient’s activity level changes and body needs a higher pressure for perfusion and organ functions. However, in these studies pressure should also be estimated due to lack of implantable long term pressure transducers on RBPs. An RBP differential pressure control algorithm was presented by Waters et al. [46]. In this study, the aim was to maintain the physiological pressure and flow levels in the body. The reference pressure difference was estimated by using motor voltage, motor current and RBP operating speed through the models that were developed from the experimental data. This study requires a particular attention because a complete numerical model that describes the RBP motor, mechanical and hydraulic component dynamics was presented and stability analyses were done using this model. In short a complete control method was given to drive the RBP at a varying speed. Physiological effects of differential RBP pressure control were investigated by Giridharan et al. [47–51] in a series of studies considering different pathological and physiological conditions such as normal heart to left heart asystole, and rest to strenuous exercise. An RBP driven by employing such a control application also generates a pump flow rate which is in the physiological range. Moreover, it is possible to avoid suction occurrence in the left ventricle. In these studies, need of sensors was eliminated by using an extended Kalman filter to predict the pressure difference across the RBP. Although this prediction method showed a good performance in the simulations and in vitro experiments, it was used in simplified numerical models or mock circulatory loops driven by these models. However, applicability of such an application in a real heart remains as a question mark. Because remodeling process in a failing and RBP supported heart is not very well known. Moreover, there is no such a study predicting RBP or cardiovascular system dynamics in animals or patients. Similar sensorless control algorithms were suggested by utilizing RBP operating speed and power in [52, 53]. Upper and lower limits of the pump operating speed were regulated while maintaining the average reference differential pressure across the RBP. Differential pressure across the pump was estimated by using an extended Kalman filter. The simplest way of maintaining the mean aortic pressure is to control the RBP outlet pressure by applying a constant pressure reference value in the control application. Such an application was proposed by Wu et al. [54–59] applying an optimal control to RBP output pressure and differential pressure. Differential pressure control ensures the prevention of the suction in the left ventricle while output pressure control maintains the arterial pressure in the physiological range. This application was also tested for resting and moderate exercise condition. However, an increase in the patient’s activity level requires an accurate estimation of the necessary physiological RBP output pressure level according to the body demands. Therefore it also needs an estimator for the contraction strength of the heart. Hsu et al. [60] suggested a driving method including two different control algorithms for the intra-aortic RBPs. The first control algorithm switches off the RBP if the upper body pressure falls below a certain threshold. In addition, if the blood pressure in the lower body rises beyond a healthy value the RBP is again switched off. The second control algorithm increases cardiac output by reducing afterload. However, when the limit is reached beyond which the failing heart cannot further increase cardiac output, further pump activity in the descending aorta would lead to a fall in upper body pressure.
A total artificial heart replaces both of the ventricles and it might cause suction in the atria due to excessive pumping. Olegario et al. [61] proposed a suction detection algorithm for the left atrium under support of a centrifugal total artificial heart. This control algorithm maintains the right pump output at a constant level to keep left atrial pressure constant as a part of an automatic control algorithm that can respond to changing physiological demand and at the same time prevent atrial wall suction. Keeping the left atrial pressure at a constant level prevents the atrial suction, however, such an operating mode always provides the same level of inlet pressure for the RBP that replacing the left ventricle. Therefore the outlet pressure of the RBP should be also controlled considering pump characteristics for the desired flow rate values.
Direct control of the RBP operating speed by synchronizing it according to duration of a heartbeat can also help to enhance the perfusion while avoiding the suction. A stepwise RBP operating speed change was studied by He et al. [62]. RBP operating speed was changed by applying an optimal control which takes into account one of the four membership functions namely stroke volume, mean left atrial pressure, aortic diastolic pressure and mean pump rotation speed. Since the only measurable parameter is pump rotation, the other parameters should be estimated again. Synchronization of the RBP operating speed over a cardiac cycle may increase the or decrease the myocardial oxygen consumption with respect to constant speed RBP operating speed depending on co-pulsation or counter-pulsation synchronization [63]. Synchronizing the RBP operating speed is not difficult to apply in the patients having an implantable cardioverter defibrillator. However, in the patients without an implantable cardioverter defibrillator such task again needs use of estimation techniques. Counterpulsative RBP support provides a better coronary perfusion with respect to copulsative and constant speed pump support modes [64] and it prevents regurgitant RBP flow [65]. It should be noted that an RBP reverse flow estimation algorithm was presented long before this study [66]. The relation between the heart function and RBP efficiency was used to detect the reverse flow through the pump. If the heart function and RBP efficiency are stable the ratio between RBP motor current at the systolic phase and diastolic phase becomes a fixed value. However, if they are not stable due to suction or RBP reverse flow then this ratio becomes variable. Again detection of the heart function becomes vital for such an application. It is also shown in [67] that RBP differential pressure control or RBP flow control performs better to avoid the reverse flow through the RBP. Comparison of the continuous and intermittent reduced RBP operating modes shows that renal and intestinal regional blood flows did not produce any significant changes compared with baseline values in either continuous or intermittent reduced RBP operating modes. However, the venous oxygen contents and carbon dioxide contents significantly increases in continuous and intermittent reduced RBP operating modes and speeds compared with baseline [68].
Adding pressure sensors to an RBP would provide accurate measurements and precise control of these devices by eliminating the need of pressure estimation techniques. Although a pressure sensor implanted RBP does not exist, researchers proposed control methods based on pressure measurements. For instance Bullister et al. [69] applied a hierarchical control to the inlet and outlet pressures of an RBP. Control of RBP inlet pressure prevents the suction in the left ventricle while outlet RBP pressure control maintains the aortic pressure at a physiological range. A limitation will be the pressure-flow characteristics of the pump when RBP flow is considered in such an application. A pressure sensor at the inlet of the RBP can only be used for preventing the suction [70, 71] and the outlet pressure thus the RBP flow will be determined by the pressure-flow characteristics of the pump and the contraction strength of the heart. A fully equipped RBP with pressure and flow sensors will help to adjust the RBP operating speed or operating mode according the patient’s activity level or demand of the body [72].
RBP speed regulation for improving ventricular unloading
Myocardial recovery due to reverse remodeling in the ventricle may occur under long term support with complete unloading of the left ventricle [73], although, exact mechanism of this reverse modeling is not known [74]. However, it is known that RBP removal may be feasible in the patients with dilated cardiomyopathy [75]. It is thought that a better ventricular unloading would contribute to myocardial recovery under RBP support. Comparison studies show that synchronous sinusoidal RBP speed change leads to maximization of stroke volume and minimization of ventricular pressure, thus optimally unloading the ventricle at the end of the systole [76]. In asynchronous pulsatile mode, the hemodynamic signals become highly non-physiological and may change slowly as a result of phase shifts. Moreover, counterpulsative RBP support mode yields the minimum left ventricular stroke work, left ventricular end diastolic volume, and aortic pulse pressure [77, 78]. Left ventricular pressure–volume loop and left ventricular volume for copulsative, counterpulsative and constant speed RBP support are given in Fig. 3.
Left ventricular pressure–volume loop under copulsative (i), counterpulsative (ii) and constant speed RBP support (iii) modes, left ventricular volume under copulsative (iv), counterpulsative (v) and constant speed RBP support (vi) modes. Sinusoidal speed variation applied to RBP to simulate copulsative and counterpulsative pump support. Applied constant operating speed was 9500 rpm and mean operating speed over a cardiac cycle was 9500 rpm for copulsative and counterpulsative pump support
Left ventricular pressure–volume loop area becomes larger under copulsative RBP support while left ventricular volume is lower than the counterpulsative and constant speed RBP support at the end of the systole. Counterpulsative RBP support yields the smaller pressure–volume loop area while left ventricular volume is higher than copulsative and constant speed RBP support at the end of the systole.
Evaluation of these variables using animal models also validated these findings [79–82]. Furthermore, myocardial oxygen consumption is reduced with the decrease of the left ventricular stroke work [82] while coronary blood flow increased under counterpulsative RBP support [77]. A detailed investigation of the phasic RBP support is given in [83]. Synchronization of the RBP systole and the ECG signal with a prefixed delay shows that intra-ventricular pressure during RBP systole was minimized with delays of around 40-80 per cent of one cycle. It should be noted that these studies do not propose any solution or method that can be used as tool to left ventricular recovery. Such a study was presented in [84] by aiming to provide an explicitly definable loading condition for the failing ventricle. The proposed control strategy provides a defined and adjustable load to the failing ventricle by an automatic regulation of the RBP speed and allows a controlled training of the myocardium which may help as a tool for the cardiac recovery. The left ventricular pressure was estimated to calculate the reference flow rate for the RBP using an extended Kalman Filter. Another possible method for ventricular recovery is presented in [85]. The applied control adapts the hydraulic power output of the VAD to the end-diastolic volume of the left ventricle. However, it was not addressed the issue of the left ventricular volume measurement under RBP support in a real application.
RBP speed regulation for increasing arterial pulsatility
Continuous operating speed of RBP support reduces the arterial pulsatility because of generated non-pulsatile blood flow by the RBP. Reduced arterial pulsatility is associated with long term complications such as gastrointestinal bleeding, functional changes in the vascular system, aortic wall remodeling, worse auto regulatory function, increased inflammatory response etc. in the patient’s body [86–91]. A higher arterial pulsatility can be achieved under copulsative RBP support. However, the generated flow signals may be different than the physiological signals because of generated non-physiological flow and pressure signals by RBP. For instance Shiose et al. [92] suggested a speed modulation algorithm for a continuous flow total artificial heart to generate physiologic arterial waveform. They generated three different speed profiles namely sinusoidal, rectangular and optimized (physiological) speed profiles and assessed them using a mock circulatory system. Arterial pulse pressure was higher under rectangular speed profile of RBP with respect to other speed profiles. A stepwise RBP operating speed change over a cardiac cycle was suggested to increase the arterial pulsatility in [93] by changing the pump speed from a low value to a high value at the peak systole to keep the arterial pressure increasing although relaxation of the left ventricle started. RBP operating speed was reduced to a low value again at the end of the systole to reduce the RBP output during the diastolic phase. Such an RBP driving mode increases the arterial pulsatility, however, the implementation of the method is disregarded in this particular study. A similar method to adjust the RBP operating speed at a high level and at a low level over a cardiac cycle was presented in [94]. Unlike the preceding application the high RBP operating speed was applied over entire systolic phase in this study. A method to synchronize the RBP speed and systolic phase is presented in [95]. A pacing lead was used to detect the ventricular electrocardiogram for the synchronization of the RBP speed change. As a results energy equivalent pressure was 9 per cent higher than the mean aortic pressure under pulsatile speed RBP support when the pump was operated at copulsative mode while these two parameters were almost at the same level under constant speed RBP support. Shi et al. [96, 97] evaluated the cardiovascular response under pulsatile operating RBP support using a numerical model by applying different pulsation ratio and the phase shift values to the RBP motion profile. An optimization algorithm was used to balance the importance of the characteristic cardiovascular variables by introducing a cost function.
Using different RBP flow rate signals rather than direct speed control would generate more physiological blood flow in the arteries. Such an application presented in [98, 99] by using different RBP flow rate signal profiles to modulate the operating speed in order to increase the pulsatility. The pulse width and amplitude of the flow signal through the pump was changed to assess the effects of the suggested RBP flow profiles. Thus, it was shown the possibility of increasing pulsatility through pump control, although the control method itself was disregarded in the numerical simulations. Therefore, this study remains unclear in terms of control system design and whether such an operating mode for RBPs is achievable for the flow rate signals having relatively higher amplitude. Bozkurt et al. [100] suggested a control algorithm to increase the arterial pulsatility while obtaining more physiological arterial pressure signals in a following study. This control application includes a reference model to describe the flow rate signal through the aortic valve over a cardiac cycle. Thus the RBP will generate a physiological blood flow in each cardiac cycle. This method was also evaluated in ex vivo experiment using a similar reference model in the control application [101]. However, RBP did not respond fast enough to follow the reference signal although it was possible to obtain more physiological blood flow and higher pulsatility in the arteries. Comparison of the arterial pressure signals for a healthy cardiovascular system model and RBP support modes for stepwise change in the pump operating speed, constant pump speed and RBP speed regulation by utilizing a physiological reference model for the pump flow as in [100] is given in Fig. 4. The mean pump operating speed was 9500 rpm for the RBP supported cardiovascular system models.
Arterial pressure signal for a healthy cardiovascular system model (i), constant speed RBP support mode (ii), stepwise RBP speed support mode (iii) and RBP speed regulation by using a physiological reference model (iv). The mean pump operating speed was 9500 rpm for the RBP supported cardiovascular system models
Amplitude of the arterial pressure signals was the highest in the healthy cardiovascular system model. RBP support reduced the amplitude of the arterial pressure signals in all pump support modes. Constant speed RBP support reduced the amplitude of the arterial pressure signals more with respect to other RBP support modes. Although arterial pressure signal amplitude was the highest in stepwise RBP speed support, control application utilizing a physiological model to regulate the RBP speed generated more physiological arterial blood pressure signal shape.
RBP speed regulation for assessing aortic valve function
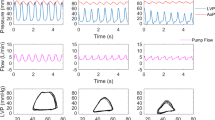
Native aortic valve is subject to cyclic load under normal physiological conditions. Constant speed RBP support alters the load on the aortic valve and aortic valve does not fully open or remains closed over a cardiac cycle [10, 102]. As a result aortic insufficiency develops and causes reverse flow through the valve which increases the pump work and decreases systemic perfusion [103]. Applied varying speed RBP support mode would not improve the support level when moderate or worse aortic insufficiency occurs [104]. Therefore, a varying speed RBP support should aim to prevent the aortic insufficiency rather than aiming to improve the support in the insufficient aortic valve. Synchronization of the RBP operating speed by considering different phases over a cardiac cycle may ensure the aortic valve opening while maintain the RBP output at a physiological level [105–108]. However, it should be noted that opening of the aortic valve may not alleviate the aortic valve problems because of altered loads on it under RBP support. Therefore more elaborated methods should be used to improve the opening and closing behavior of the aortic valve. Comparison studies shows that it is possible to increase the aortic valve mean orifice area and duty cycle over a heartbeat when a counterpulsative RBP support is applied [109]. Comparison of flow through the aortic valve and pump flow rate in constant speed, copulsative and counterpulsative RBP support modes is given in Fig. 5. RBP was operated at 9500 rpm speed and at copulsative and counterpulsative pump support modes. Flow rate through the RBP was the same for all pump operating modes (3.65 L/min).
Aortic valve flow over the ejection phase for 9500 rpm RBP operating speed (i) and 3.65 L/min mean RBP flow for the copulsative (ii) and counterpulsative (iii) pump support. RBP flow rate at 9500 rpm operating speed (iv) and 3.65 L/min mean pump flow for the copulsative (v) and counterpulsative (vi) support modes
Aortic valve opening time becomes the highest in counterpulsative RBP support mode while it is the lowest in copulsative RBP support mode. However, RBP flow pulsatility is the highest in copulsative RBP support mode and it becomes the lowest under counterpulsative RBP support mode.
Physiological control of RBPs
A physiological control system is defined as the control theory and its application to physiological systems [110]. However, in this paper it will be used as a control system designed to operate RBPs to imitate a physiological mechanism. Because in any applications including the constant speed RBP support mode as well, control theory has to be applied to regulate the RBP operating speed because of changing load on the impeller. Therefore physiological control can be defined as a control system application developed to imitate a physiological mechanism. According to this definition the studies cited in this subsection are classified as physiological control applications. Imitation of the Frank–Starling mechanism can be considered such a control design for the RBPs. Since the Frank–Starling mechanism is expressed as the change of stroke volume in the ventricles is a response to the volume of blood filling the heart [111] the supporting RBP should adjust its output accordingly. A control algorithm imitating the Frank–Starling mechanism is presented in [112] by describing the RBP flow as a function of the left atrial pressure. It is reported that the applied control strategy regulated pump speed in response to a decrease and following increase in venous return while a constant speed RBP caused ventricular suction due to low venous return and higher ventricular volumes because of high venous return. Theoretical foundations of this control design are presented in [113] by employing numerical simulations and in vitro evaluation of this study presents the experimental validation [114]. Findings show that RBP flow pulsatility corresponds with changes in left ventricular stroke work before and after opening of the aortic valve and was least affected by contraction strength of the heart, blood volume, peripheral vascular resistance, and heart rate. Furthermore, patient studies investigating cause, effects, and implications for starling-like-control of changes in RBP flow showed that increasing pump flows causes significant improvements in maximal exercise capacity [115]. Application of such a control in bi-ventricular RBPs shows a better performance with respect to constant speed pump support in terms of suction occurrence and unloading the ventricles at the end of diastole [116]. However, it is also reported that the inlet cannula resistance of RBPs would affect the performance of such a control design [117]. Different control methods such as sliding mode control may improve the performance of such control systems [118, 119].
RBP speed regulation for different purposes
Except the cited studies in the previous sub sections, researchers suggested different pump operating speed regulation algorithms aiming to achieve different physiological objectives for the RBP support. An automatic detection algorithm for irregular heartbeats and the feedback control of the phase shift between the systolic phase and the assist device is suggested in [120]. An RBP driving method to prevent right ventricular failure induced by RBP support by monitoring the septal shift to detect the right ventricular deformation is presented in [121]. A varying speed RBP support algorithm changing the pump operating speed to regulate heart rate of a patient is given in [122] by including a baroreflex model [123] which describes the heart rate in the simulations. A control algorithm is developed by defining a blood assist index which includes power term of the RBP and the cardiovascular system for left ventricular recovery in [124]. An investigation on the influence of an electrocardiogram synchronized rotational speed change system of an RBP on the hemolytic performance is done in [125]. It can be told that these are the initial efforts to achieve these tasks and needs time and more investigation and research to develop efficient varying speed RBP support algorithms.
It is also possible to improve different physiological conditions by applying the same RBP driving mode. For instance, an optimal control strategy to maximize the flow through the aortic valve and minimize the left-ventricular stroke work by synchronizing the RBP speed profile to a heartbeat is given in [126]. Different control strategies are also combined in one algorithm to improve different physiological aspects under RBP support. For instance Arntd et al. [127] suggested a control method for RBPs with selectable therapeutic options: maximum support with the highest feasible flow rate versus medium support with maximum ventricular washout and controlled opening of the aortic valve. They used a pulsatility indicator which was calculated by utilizing the pressure difference across the RBP as the control variable. In the following study they assessed different control strategies using only one control input (pressure difference across the RBP) [128]. Such a control application can be useful in the RBP implanted patients suffering from different problems or switching between the operating modes according to the task having the priority.
Discussion
Aims in the initial studies on developing varying speed RBP support algorithms were generating adequate blood flow according to the physiological demand and detection of the ventricular suction and regurgitant RBP flow. In these studies different indicators such as power spectral density of harmonics of the RBP motor current, pulsatility of the RBP motor current, change of RBP flow rate with respect to RBP operating speed, pulsatility ratio of the RBP flow rate or numerical models were used to detect the ventricular suction under RBP support. Furthermore, methods such as pressure difference across the RBP control or synchronization of the RBP operating speed with respect to beginning of the cardiac cycle were used provide the sufficient blood flow in the cardiovascular system. Lack of reliable long term pressure sensors motivated researchers to develop sensorless estimation techniques to drive the RBP at a varying speed. Extended Kalman filter was used to estimate the RBP flow and pressure. However, suggested varying speed RBP algorithms were assessed either numerically or using mock circulatory systems driven by simplified reference models in their control algorithm. However, in a failing heart, the mechanism of the heart contraction is likely to change [1] and it is not known very well for the RBP recipient patients. This can be considered as a limitation for the RBPs driven by the control algorithms using these estimation methods. As mentioned in “Rotary blood pump speed regulation” section there are proposed studies utilizing sensors. However, reliability of these sensors is not known for long term RBP support and a pressure sensor integrated RBP does not exist in the market. It should be noted that it is an ongoing research to develop reliable pressure sensors for long term RBP support [129, 130]. Furthermore, only Micromed DeBakey RBP includes an additional flow sensor to measure the flow rate through the pump [11].
Number of the studies aiming to drive the RBPs for achieving physiological aspects rather than optimal perfusion, prevention of the suction and regurgitant pump is considerably less. One of the explanations of this situation can be that the main concern to develop a control strategy for the RBPs was delivering optimal blood flow without any significant pumping effect because of the RBP operating speed in the cardiovascular system in the early RBP support studies. Initially, RBP operating speed control utilizing indicators developed in frequency and time domains was suggested to detect suction in the left ventricle and provide sufficient blood flow to the arteries. RBP flow rate control and pressure control techniques were started to being suggested in 2000s to deliver sufficient blood flow to the arteries and detect the suction in the left ventricle. These control techniques also utilize RBP flow rate or pressure based suction indicators. Once the suction in the left ventricle is detected the speed controller reduces the RBP operating speed to a lower value. However, RBP flow rate or pressure controller may regulate the pump operating speed continuously over a cardiac cycle depending on the control reference signal. Delivering adequate blood flow to arteries can be achieved by using all these control methods. Therefore, developed suction indicators become more important to detect and classify the suction in the left ventricle accurately. Frequency based suction detection algorithms provide information about occurrence of suction in the left ventricle while suggested suction indicators in time domain provide information about hemodynamical signals as a whole and they allow to describe critical points over a cardiac cycle. The studies focusing on the other physiological aspects of the varying speed RBP support are relatively newer topics and recently started get attention. RBP speed control and RBP flow rate control were applied to investigate unloading of the left ventricle under varying speed pump support. Applied pump speed waveforms were sinusoidal or square waveforms. Arterial pulsatility was also investigated under speed controlled and flow rate controlled RBP. However, it should be noted that applied sinusoidal or stepwise speed profiles do not generate physiological RBP pressure or flow rate signals although they improve ventricular unloading and arterial pulsatility. Moreover, physiological control algorithms utilize one of these variables to imitate the Frank–Starling mechanism in the body. Furthermore, varying speed RBP support algorithms which aim improving the physiological aspects rather than providing sufficient blood flow are expected to utilize RBP flow rate or pressure. Such an operating mode will also allow improving outcome of the RBP support. For instance, although the efficient ventricular unloading is being studied, there are not too many studies aiming to drive the RBP to recover the left ventricle by controlled training. It is possible to increase the arterial pulsatility, however, the shape of the hemodynamical signals will remain non-physiological if a proper reference model describing the cardiovascular physiology accurately is not applied in the control algorithm. Furthermore an increase in the aortic valve area or duty cycle may not be enough to alleviate the aortic valve problems under RBP support. Opening and closing behavior of the aortic valve leaflets should be as close as possible to the unassisted aortic valve behavior. To do so vortex formation in the aortic valve sinuses should be investigated in an RBP supported heart to develop an efficiently working control strategy to achieve this task.
Rhythm disorders are one of the serious problems for RBP supported circulation [131, 132] and there are not many studies proposing a solution for this problem. Although most of the RBP patients have an implantable cardioverter defibrillator along with RBP [133] to obtain regular heartbeats in these patients the remaining will still have a problem. Cardiac arrhythmia detection algorithms should be implemented to RBPs to overcome the effects of the cardiac arrhythmias. However, this also requires identification of exact mechanism of the cardiac arrhythmias under RBP support and remains as another challenge in the RBP patients.
One of the potential adverse effects of the applied varying speed RBP support modes is the blood damage due to increased shear stress caused by the accelerating impeller. However, this requires the measurement of hemolysis. Although it was reported in [125] there is not a significant difference between hemolysis levels under constant speed and varying speed RBP support, this can be valid only for this study. Therefore the risk of hemolysis and alleviation of it remains as another challenge and should be investigated for the varying speed RBP support.
The developed varying speed RBP support algorithms aiming to render a better ventricular unloading, increase the arterial pulsatility, improve the aortic valve function and applying a physiological control require a continuous change in the operating speed over a cardiac cycle. However, it is more difficult to achieve such a driving mode with respect to RBP support mode which keeps the operating speed constant over a cardiac cycle and changing this constant speed according to the changing conditions in the patient’s body. Because the design of the RBPs does not allow an immediate response to the control inputs [101]. Furthermore, such an operating mode requires more elaborated control algorithms to drive the RBP. This task also remains as a challenge in the control problems of RBPs. It should also be noted that designing more elaborated control algorithms require identification of the exact mechanisms of the physiological case. Improvement in the sensor technology and change in the RBP design to have a better response to the control input signals will help to achieve these objectives as well.
It should also be noted that power consumption of the varying speed RBP support will be higher than the constant speed RBP support [134]. This is one of the problems that should be solved due to short battery life. Use of longer lasting batteries in the RBP will also make these algorithms more feasible to apply in a patient.
Conclusion
Different varying speed RBP driving algorithms were proposed for avoiding the harmful effects of the constant speed RBP support. However, each varying speed RBP algorithm is developed for a specific objective and they have limitations. An optimal solution would be combining different algorithms to achieve multi task RBP support mode by driving it at a dynamic speed over a cardiac cycle alongside identification of the physiological mechanism for the heart failure and problems occurring due to continuous speed RBP support.
References
Libby P, Bonow RO, Mann D, Zipes DP (2007) Braunwald’s heart disease: a textbook of cardiovascular medicine, 8th edn. Saunders, Philadelphia
Lund LH, Matthews J, Aaronson K (2010) Patient selection for left ventricular assist devices. Eur J Heart Fail 12:434–443. doi:10.1093/eurjhf/hfq006
Slaughter MS, Pagani FD, Rogers JG et al (2010) Clinical management of continuous-flow left ventricular assist devices in advanced heart failure. J Heart Lung Transpl 29:S1–S39. doi:10.1016/j.healun.2010.01.011
Bourque K, Gernes DB, Loree HM et al (2001) HeartMate III: pump design for a centrifugal LVAD with a magnetically levitated rotor. ASAIO J 47:401–405
Bozkurt S, Safak KK (2013) Evaluating the hemodynamical response of a cardiovascular system under support of a continuous flow left ventricular assist device via numerical modeling and simulations. Comput Math Methods Med 2013:e986430. doi:10.1155/2013/986430
Bertram CD (2005) Measurement for implantable rotary blood pumps. Physiol Meas 26:R99–R117. doi:10.1088/0967-3334/26/4/R01
AlOmari A-HH, Savkin AV, Stevens M et al (2013) Developments in control systems for rotary left ventricular assist devices for heart failure patients: a review. Physiol Meas 34:R1–R27. doi:10.1088/0967-3334/34/1/R1
Amacher R, Ochsner G, Schmid Daners M (2014) Synchronized pulsatile speed control of turbodynamic left ventricular assist devices: review and prospects. Artif Organs 38:867–875. doi:10.1111/aor.12253
Reesink K, Dekker A, Van der Nagel T et al (2007) Suction due to left ventricular assist: implications for device control and management. Artif Organs 31:542–549. doi:10.1111/j.1525-1594.2007.00420.x
Karantonis DM, Cloherty SL, Mason DG, et al. (2006) Automated non-invasive detection of pumping states in an implantable rotary blood pump. In: 28th annual international conference of the IEEE engineering in medicine and biology society. EMBS 06, pp 5386–5389
Pennings KAMA, Martina JR, Rodermans BFM et al (2013) Pump flow estimation from pressure head and power uptake for the HeartAssist5, HeartMate II, and HeartWare VADs. ASAIO J 59:420–426. doi:10.1097/MAT.0b013e3182937a3a
Konishi H, Antaki JF, Amin DV et al (1996) Controller for an axial flow blood pump. Artif Organs 20:618–620
Iijima T, Inamoto T, Nogawa M, Takatani S (1997) Control of centrifugal blood pump based on the motor current. Artif Organs 21:655–660
Yuhki A, Hatoh E, Nogawa M et al (1999) Detection of suction and regurgitation of the implantable centrifugal pump based on the motor current waveform analysis and its application to optimization of pump flow. Artif Organs 23:532–537. doi:10.1046/j.1525-1594.1999.06397.x
Ohuchi K, Kikugawa D, Takahashi K et al (2001) Control strategy for rotary blood pumps. Artif Organs 25:366–370
Oshikawa M, Araki K, Endo G et al (2000) Sensorless controlling method for a continuous flow left ventricular assist device. Artif Organs 24:600–605
Boston JR, Simaan MA, Antaki JF, et al. (1998) Intelligent control design for heart assist devices. In: Proceedings of the Intelligent control ISIC 1998 held jointly IEEE international symposium on computational intelligence in robotics and automation, CIRA intelligent systems and semiotics ISAS, pp 497–502
Boston JR, Simaan MA, Antaki JF, Yu Y-C (2000) Control issues in rotary heart assist devices. In: Proceedings of the 2000 American control conference, vol 5, pp 3473–3477
Boston JR, Baloa L, Liu D, et al. (2000) Combination of data approaches to heuristic control and fault detection [heart assist devices]. In: Proceedings of the 2000 IEEE international conference on control applications, pp 98–103
Boston JR, Antaki JF, Simaan MA (2003) Hierarchical control of heart-assist devices. IEEE Robot Autom Mag 10:54–64. doi:10.1109/MRA.2003.1191711
Tanaka A, Yoshizawa M, Olegario P et al (2005) Detection and avoiding ventricular suction of ventricular assist devices. IEEE Int Conf Eng Med Biol Soc 1:402–405. doi:10.1109/IEMBS.2005.1616430
Baloa LA, Liu D, Boston JR, et al. (2000) Control of rotary heart assist devices. In: Proceedings of the 2000 American control conference, vol 5, pp 2982–2986
Choi S, Antaki JF, Boston JR, Thomas D (2001) A sensorless approach to control of a turbodynamic left ventricular assist system. IEEE Trans Control Syst Technol 9:473–482. doi:10.1109/87.918900
Choi S (2003) Suction detection in left ventricular assist system: data fusion approach. Int J Control Autom Syst 1:368–375
Choi S, Boston JR, Antaki JF (2005) An investigation of the pump operating characteristics as a novel control index for LVAD control. Int J Control Autom Syst 3:100–108
Choi S, Boston JR, Antaki JF (2007) Hemodynamic controller for left ventricular assist device based on pulsatility ratio. Artif Organs 31:114–125. doi:10.1111/j.1525-1594.2007.00350.x
Fu M, Xu L (2000) Computer simulation of sensorless fuzzy control of a rotary blood pump to assure normal physiology. ASAIO J 46:273–278
Chen S, Antaki JF, Simaan MA, Boston JR (2005) Physiological control of left ventricular assist devices based on gradient of flow. In: Proceedings of the 2005 American control conference, vol 6, pp 3829–3834
Ferreira A, Boston JR, Antaki JF (2007) A rule-based controller based on suction detection for rotary blood pumps. IEEE Int Conf Eng Med Biol Soc 2007:3978–3981. doi:10.1109/IEMBS.2007.4353205
Ferreira A, Boston JR, Antaki JF (2009) A control system for rotary blood pumps based on suction detection. IEEE Trans Biomed Eng 56:656–665. doi:10.1109/TBME.2008.2005960
Gwak K-W, Ricci M, Snyder S et al (2005) In vitro evaluation of multiobjective hemodynamic control of a heart-assist pump. ASAIO J 51:329–335
Gwak K-W (2007) Application of extremum seeking control to turbodynamic blood pumps. ASAIO J 53:403–409. doi:10.1097/MAT.0b013e31806ada0a
Gwak K-W, Antaki JF, Paden BE, Kang B (2011) Safety-enhanced optimal control of turbodynamic blood pumps. Artif Organs 35:725–732. doi:10.1111/j.1525-1594.2011.01230.x
Faragallah G, Wang Y, Divo E, Simaan MA (2011) A new current-based control model of the combined cardiovascular and rotary left ventricular assist device. Am Control Conf 2011:4775–4780
Voigt O, Benkowski RJ, Morello GF (2005) Suction detection for the MicroMed DeBakey left ventricular assist device. ASAIO J 51:321–328
Wang Y, Faragallah G, Divo E, Simaan MA (2012) Feedback control of a rotary left ventricular assist device supporting a failing cardiovascular system. Am Control Conf 2012:1137–1142
Wang Y, Simaan MA (2013) A suction detection system for rotary blood pumps based on the Lagrangian support vector machine algorithm. IEEE J Biomed Health Inform 17:654–663. doi:10.1109/TITB.2012.2228877
Vollkron M, Schima H, Huber L et al (2004) Development of a suction detection system for axial blood pumps. Artif Organs 28:709–716. doi:10.1111/j.1525-1594.2004.00011.x
Vollkron M, Schima H, Huber L et al (2005) Development of a reliable automatic speed control system for rotary blood pumps. J Heart Lung Transpl 24:1878–1885. doi:10.1016/j.healun.2005.02.004
Vollkron M, Schima H, Huber L et al (2006) Advanced suction detection for an axial flow pump. Artif Organs 30:665–670. doi:10.1111/j.1525-1594.2006.00282.x
Mason DG, Hilton AK, Salamonsen RF (2008) Reliable suction detection for patients with rotary blood pumps. ASAIO J 54:359–366. doi:10.1097/MAT.0b013e31817b5b0e
Simaan MA (2008) Modeling and control of the heart left ventricle supported with a rotary assist device. In: 47th IEEE conference on decision and control 2008, pp 2656–2661
Simaan MA, Ferreira A, Chen S et al (2009) A dynamical state space representation and performance analysis of a feedback-controlled rotary left ventricular assist device. IEEE Trans Control Syst Technol 17:15–28. doi:10.1109/TCST.2008.912123
Casas F, Orozco A, Smith WA et al (2004) A fuzzy system cardio pulmonary bypass rotary blood pump controller. Expert Syst Appl 26:357–361. doi:10.1016/j.eswa.2003.09.006
Casas F, Ahmed N, Reeves A (2007) Minimal sensor count approach to fuzzy logic rotary blood pump flow control. ASAIO J 53:140–146. doi:10.1097/01.mat.0000250786.56697.f2
Waters T, Allaire P, Tao G et al (1999) Motor feedback physiological control for a continuous flow ventricular assist device. Artif Organs 23:480–486
Giridharan GA, Skliar M (2002) Nonlinear controller for ventricular assist devices. Artif Organs 26:980–984. doi:10.1046/j.1525-1594.2002.07136.x
Giridharan GA, Skliar M (2003) Physiological control of blood pumps without implantable sensors. In: Proceedings of the 2003 American control conference, pp 471–476
Giridharan GA, Skliar M (2003) Control strategy for maintaining physiological perfusion with rotary blood pumps. Artif Organs 27:639–648
Giridharan G, Pantalos G, Koenig S, et al. (2005) Achieving physiologic perfusion with ventricular assist devices: comparison of control strategies. Proceedings of the 2005 American control conference, vol 6, pp 3823–3828
Giridharan GA, Skliar M (2006) Physiological control of blood pumps using intrinsic pump parameters: a computer simulation study. Artif Organs 30:301–307. doi:10.1111/j.1525-1594.2006.00217.x
Wang Y, Koenig SC, Slaughter MS, Giridharan GA (2015) Rotary blood pump control strategy for preventing left ventricular suction. ASAIO J 61:21–30. doi:10.1097/MAT.0000000000000152
Wang Y, Koenig SC, Slaughter MS, Giridharan GA (2015) Suction prevention and physiologic control of continuous flow left ventricular assist devices using intrinsic pump parameters. ASAIO J 61:170–177. doi:10.1097/MAT.0000000000000168
Wu Y, Allaire P, Tao G et al (2003) An advanced physiological controller design for a left ventricular assist device to prevent left ventricular collapse. Artif Organs 27:926–930
Wu Y, Allaire PE, Tao G et al (2004) A bridge from short-term to long-term left ventricular assist device–experimental verification of a physiological controller. Artif Organs 28:927–932. doi:10.1111/j.1525-1594.2004.07381.x
Wu Y, Allaire P, Tao G, Olsen D (2005) Modeling, estimation and control of cardiovascular systems with a left ventricular assist device. In: Proceedings of the American control conference, vol 6, pp 3841–3846
Wu Y, Allaire PE, Tao G, Olsen D (2007) Modeling, estimation, and control of human circulatory system with a left ventricular assist device. IEEE Trans Control Syst Technol 15:754–767. doi:10.1109/TCST.2006.890288
Yi W (2007) Physiological control of rotary left ventricular assist device. In: Chinese control conference, pp 469–474
Wu Y (2009) Adaptive physiological speed/flow control of rotary blood pumps in permanent implantation using intrinsic pump parameters. ASAIO J 55:335–339. doi:10.1097/MAT.0b013e3181aa2554
Hsu P-L, Bruch J, McMahon R (2011) A controller for a miniature intra-aortic ventricular assist device. Artif Organs 35:282–287. doi:10.1111/j.1525-1594.2010.01060.x
Olegario PS, Yoshizawa M, Tanaka A et al (2003) Outflow control for avoiding atrial suction in a continuous flow total artificial heart. Artif Organs 27:92–98
He P, Bai J, Xia DD (2005) Optimum control of the Hemopump as a left-ventricular assist device. Med Biol Eng Comput 43:136–141
Umeki A, Nishimura T, Takewa Y et al (2013) Change in myocardial oxygen consumption employing continuous-flow LVAD with cardiac beat synchronizing system, in acute ischemic heart failure models. J Artif Organs 16:119–128. doi:10.1007/s10047-012-0682-0
Ando M, Takewa Y, Nishimura T et al (2011) A novel counterpulsation mode of rotary left ventricular assist devices can enhance myocardial perfusion. J Artif Organs 14:185–191. doi:10.1007/s10047-011-0573-9
Ando M, Nishimura T, Takewa Y et al (2011) A novel counterpulse drive mode of continuous-flow left ventricular assist devices can minimize intracircuit backward flow during pump weaning. J Artif Organs 14:74–79. doi:10.1007/s10047-010-0544-6
Nakata K, Ohtsuka G, Yoshikawa M et al (1999) A new control method that estimates the backflow in a centrifugal pump. Artif Organs 23:538–541
Bozkurt S, Safak KK, Sorguven E, et al. (2009) Control strategies for the left ventricular assist devices. In: Biomedical engineering meeting, BIYOMUT 2009 14th National, pp 1–4
Tuzun E, Chorpenning K, Liu MQ et al (2014) The effects of continuous and intermittent reduced speed modes on renal and intestinal perfusion in an ovine model. ASAIO J 60:19–24. doi:10.1097/MAT.0000000000000024
Bullister E, Reich S, Sluetz J (2002) Physiologic control algorithms for rotary blood pumps using pressure sensor input. Artif Organs 26:931–938
Cysyk J, Jhun C-S, Newswanger R et al (2011) Rotary blood pump control using integrated inlet pressure sensor. IEEE Int Conf Eng Med Biol Soc 2011:373–376. doi:10.1109/IEMBS.2011.6090121
Ishii K, Saito I, Isoyama T et al (2012) Development of normal-suction boundary control method based on inflow cannula pressure waveform for the undulation pump ventricular assist device. Artif Organs 36:812–816. doi:10.1111/j.1525-1594.2012.01451.x
Verbeni A, Fontana R, Silvestri M et al (2014) An innovative adaptive control strategy for sensorized left ventricular assist devices. IEEE Trans Biomed Circuits Syst 8:660–668. doi:10.1109/TBCAS.2014.2346015
Simon MA, Kormos RL, Murali S et al (2005) Myocardial recovery using ventricular assist devices: prevalence, clinical characteristics, and outcomes. Circulation 112:I32–I36. doi:10.1161/CIRCULATIONAHA.104.524124
Burkhoff D, Klotz S, Mancini DM (2006) LVAD-induced reverse remodeling: basic and clinical implications for myocardial recovery. J Card Fail 12:227–239. doi:10.1016/j.cardfail.2005.10.012
Dandel M, Weng Y, Siniawski H et al (2011) Heart failure reversal by ventricular unloading in patients with chronic cardiomyopathy: criteria for weaning from ventricular assist devices. Eur Heart J 32:1148–1160. doi:10.1093/eurheartj/ehq353
Vandenberghe S, Segers P, Antaki JF et al (2005) Hemodynamic modes of ventricular assist with a rotary blood pump: continuous, pulsatile, and failure. ASAIO J 51:711–718
Cox LGE, Loerakker S, Rutten MCM et al (2009) A mathematical model to evaluate control strategies for mechanical circulatory support. Artif Organs 33:593–603. doi:10.1111/j.1525-1594.2009.00755.x
Lim E, Alomari A-HH, Savkin AV et al (2011) A method for control of an implantable rotary blood pump for heart failure patients using noninvasive measurements. Artif Organs 35:E174–E180. doi:10.1111/j.1525-1594.2011.01268.x
Pirbodaghi T, Axiak S, Weber A et al (2012) Pulsatile control of rotary blood pumps: does the modulation waveform matter? J Thorac Cardiovasc Surg 144:970–977. doi:10.1016/j.jtcvs.2012.02.015
Pirbodaghi T, Weber A, Axiak S et al (2013) Asymmetric speed modulation of a rotary blood pump affects ventricular unloading. Eur J Cardio-Thorac Surg 43:383–388. doi:10.1093/ejcts/ezs299
Umeki A, Nishimura T, Ando M et al (2012) Alteration of LV end-diastolic volume by controlling the power of the continuous-flow LVAD, so it is synchronized with cardiac beat: development of a native heart load control system (NHLCS). J Artif Organs 15:128–133. doi:10.1007/s10047-011-0615-3
Soucy KG, Giridharan GA, Choi Y et al (2015) Rotary pump speed modulation for generating pulsatile flow and phasic left ventricular volume unloading in a bovine model of chronic ischemic heart failure. J Heart Lung Transpl 34:122–131. doi:10.1016/j.healun.2014.09.017
Heredero A, Perez-Caballero R, Otero J et al (2012) Synchrony relationships between the left ventricle and a left ventricular assist device: an experimental study in pigs. Int J Artif Organs 35:272–278. doi:10.5301/ijao.5000086
Moscato F, Arabia M, Colacino FM et al (2010) Left ventricle afterload impedance control by an axial flow ventricular assist device: a potential tool for ventricular recovery. Artif Organs 34:736–744. doi:10.1111/j.1525-1594.2010.01066.x
Ochsner G, Amacher R, Wilhelm MJ et al (2014) A physiological controller for turbodynamic ventricular assist devices based on a measurement of the left ventricular volume. Artif Organs 38:527–538. doi:10.1111/aor.12225
Ventura PA, Alharethi R, Budge D et al (2011) Differential impact on post-transplant outcomes between pulsatile- and continuous-flow left ventricular assist devices. Clin Transpl 25:E390–E395. doi:10.1111/j.1399-0012.2011.01433.x
Crow S, John R, Boyle A et al (2009) Gastrointestinal bleeding rates in recipients of nonpulsatile and pulsatile left ventricular assist devices. J Thorac Cardiovasc Surg 137:208–215. doi:10.1016/j.jtcvs.2008.07.032
Nishimura T, Tatsumi E, Takaichi S et al (1998) Prolonged nonpulsatile left heart bypass with reduced systemic pulse pressure causes morphological changes in the aortic wall. Artif Organs 22:405–410
Gokce N, Keaney JF, Hunter LM et al (2003) Predictive value of noninvasively determined endothelial dysfunction for long-term cardiovascular events in patients with peripheral vascular disease. J Am Coll Cardiol 41:1769–1775
Sandner SE, Zimpfer D, Zrunek P et al (2008) Renal function after implantation of continuous versus pulsatile flow left ventricular assist devices. J Heart Lung Transpl 27:469–473. doi:10.1016/j.healun.2007.12.012
Ji B, Undar A (2006) An evaluation of the benefits of pulsatile versus nonpulsatile perfusion during cardiopulmonary bypass procedures in pediatric and adult cardiac patients. ASAIO J 52:357–361. doi:10.1097/01.mat.0000225266.80021.9b
Shiose A, Nowak K, Horvath DJ et al (2010) Speed modulation of the continuous-flow total artificial heart to simulate a physiologic arterial pressure waveform. ASAIO J 56:403–409. doi:10.1097/MAT.0b013e3181e650f8
Bozkurt S, Pennings KAMA, Schampaert S et al (2011) A method to increase the pulsatility in hemodynamic variables in an LVAD supported human circulation system. In: Vlad S, Ciupa RV (eds) International conference on advanced medicine and health care technology. Berlin, Berlin, pp 328–331
Huang F, Ruan X, Fu X (2014) Pulse-pressure-enhancing controller for better physiologic perfusion of rotary blood pumps based on speed modulation. ASAIO J 60:269–279. doi:10.1097/MAT.0000000000000059
Ando M, Nishimura T, Takewa Y et al (2011) Electrocardiogram-synchronized rotational speed change mode in rotary pumps could improve pulsatility. Artif Organs 35:941–947. doi:10.1111/j.1525-1594.2011.01205.x
Shi Y, Lawford PV, Hose DR (2010) Numerical modeling of hemodynamics with pulsatile impeller pump support. Ann Biomed Eng 38:2621–2634. doi:10.1007/s10439-010-0001-y
Shi Y, Brown AG, Lawford PV et al (2011) Computational modelling and evaluation of cardiovascular response under pulsatile impeller pump support. Interface Focus 1:320–337. doi:10.1098/rsfs.2010.0039
Ising M, Warren S, Sobieski MA et al (2011) Flow modulation algorithms for continuous flow left ventricular assist devices to increase vascular pulsatility: a computer simulation study. Cardiovasc Eng Technol 2:90–100. doi:10.1007/s13239-011-0042-x
Ising MS, Koenig SC, Sobieski MA et al (2013) Flow modulation algorithms for intra-aortic rotary blood pumps to minimize coronary steal. ASAIO J 59:261–268. doi:10.1097/MAT.0b013e31828fd6c8
Bozkurt S, van de Vosse FN, Rutten MCM (2014) Improving arterial pulsatility by feedback control of a continuous flow left ventricular assist device via in silico modeling. Int J Artif Organs 37:773–785. doi:10.5301/ijao.5000328
Bozkurt S, van Tuijl S, Schampaert S et al (2014) Arterial pulsatility improvement in a feedback-controlled continuous flow left ventricular assist device: an ex vivo experimental study. Med Eng Phys 36:1288–1295. doi:10.1016/j.medengphy.2014.07.005
Tuzun E, Pennings K, van Tuijl S et al (2014) Assessment of aortic valve pressure overload and leaflet functions in an ex vivo beating heart loaded with a continuous flow cardiac assist device. Eur J Cardio-Thorac Surg 45:377–383. doi:10.1093/ejcts/ezt355
John R, Mantz K, Eckman P et al (2010) Aortic valve pathophysiology during left ventricular assist device support. J Heart Lung Transpl 29:1321–1329. doi:10.1016/j.healun.2010.06.006
Gregory SD, Stevens MC, Wu E et al (2013) In vitro evaluation of aortic insufficiency with a rotary left ventricular assist device. Artif Organs 37:802–809. doi:10.1111/aor.12143
Tuzun E, Gregoric ID, Conger JL et al (2005) The effect of intermittent low speed mode upon aortic valve opening in calves supported with a Jarvik 2000 axial flow device. ASAIO J 51:139–143
Kishimoto Y, Takewa Y, Arakawa M et al (2013) Development of a novel drive mode to prevent aortic insufficiency during continuous-flow LVAD support by synchronizing rotational speed with heartbeat. J Artif Organs 16:129–137. doi:10.1007/s10047-012-0685-x
Leao T, Bock E, Silva MB (2013) study of speed control of the implantable centrifugal blood pump to avoid aortic valve stenosis. In: 2014 5th IEEE RAS & EMBS international conference on biomedical robotics and biomechatronics, pp 6133–6138
Leao T, Fonseca J, Bock E, et al. (2014) Speed control of the implantable centrifugal blood pump to avoid aortic valve stenosis: simulation and implementation. In: 2014 5th IEEE RAS & EMBS international conference on biomedical robotics and biomechatronics, pp 82–86
Bozkurt S, van de Vosse FN, Rutten MCM (2014) Aortic valve function under support of a left ventricular assist device: continuous vs dynamic speed support. Ann Biomed 43:1727–1737. doi:10.1007/s10439-014-1204-4
Khoo MCK (1999) Physiological control systems: analysis, simulation, and estimation, 1st edn. Wiley, New York
Hall JE (2015) Guyton and hall textbook of medical physiology, 13th edn. Saunders, Philadelphia
Stevens MC, Gaddum NR, Pearcy M, et al. (2011) Frank–starling control of a left ventricular assist device. In: Annual International Conference of the IEEE engineering in medicine and biology society (EMBC), pp 1335–1338
Salamonsen RF, Lim E, Gaddum N et al (2012) Theoretical foundations of a Starling-like controller for rotary blood pumps. Artif Organs 36:787–796. doi:10.1111/j.1525-1594.2012.01457.x
Gaddum NR, Stevens M, Lim E et al (2014) Starling-like flow control of a left ventricular assist device. In vitro validation. Artif Organs 38:E46–E56. doi:10.1111/aor.12221
Salamonsen RF, Pellegrino V, Fraser JF et al (2013) Exercise studies in patients with rotary blood pumps: cause, effects, and implications for starling-like control of changes in pump flow. Artif Organs 37:695–703. doi:10.1111/aor.12070
Stevens MC, Wilson S, Bradley A et al (2014) Physiological control of dual rotary pumps as a biventricular assist device using a master/slave approach. Artif Organs 38:766–774. doi:10.1111/aor.12303
Gaddum NR, Timms DL, Stevens M et al (2012) Comparison of preload-sensitive pressure and flow controller strategies for a dual device biventricular support system. Artif Organs 36:256–265. doi:10.1111/j.1525-1594.2011.01344.x
Bakouri MA, Salamonsen RF, Savkin AV, et al. (2013) Physiological control of implantable rotary blood pumps for heart failure patients. In: 2013 35th annual international conference of the IEEE engineering in medicine and biology society (EMBC), pp 675–678
Bakouri MA, Salamonsen RF, Savkin AV et al (2014) A sliding mode-based starling-like controller for implantable rotary blood pumps. Artif Organs 38:587–593. doi:10.1111/aor.12223
Amacher R, Ochsner G, Ferreira A et al (2013) A robust reference signal generator for synchronized ventricular assist devices. IEEE Trans Biomed Eng 60:2174–2183. doi:10.1109/TBME.2013.2251634
Arakawa M, Nishimura T, Takewa Y et al (2014) Novel control system to prevent right ventricular failure induced by rotary blood pump. J Artif Organs 17:135–141. doi:10.1007/s10047-014-0757-1
Chang Y, Gao B, Gu K (2011) A model-free adaptive control to a blood pump based on heart rate. ASAIO J 57:262–267. doi:10.1097/MAT.0b013e31821798aa
Ursino M (1998) Interaction between carotid baroregulation and the pulsating heart: a mathematical model. Am J Physiol 275:H1733–H1747
Gao B, Gu K, Zeng Y et al (2011) A blood assist index control by intraaorta pump: a control strategy for ventricular recovery. ASAIO J 57:358–362. doi:10.1097/MAT.0b013e3182257fac
Kishimoto S, Date K, Arakawa M et al (2014) Influence of a novel electrocardiogram-synchronized rotational-speed-change system of an implantable continuous-flow left ventricular assist device (EVAHEART) on hemolytic performance. J Artif Organs 17:373–377. doi:10.1007/s10047-014-0787-8
Amacher R, Asprion J, Ochsner G et al (2013) Numerical optimal control of turbo dynamic ventricular assist devices. Bioengineering 1:22–46. doi:10.3390/bioengineering1010022
Arndt A, Nüsser P, Graichen K et al (2008) Physiological control of a rotary blood pump with selectable therapeutic options: control of pulsatility gradient. Artif Organs 32:761–771. doi:10.1111/j.1525-1594.2008.00628.x
Arndt A, Nüsser P, Lampe B (2010) Fully autonomous preload-sensitive control of implantable rotary blood pumps. Artif Organs 34:726–735. doi:10.1111/j.1525-1594.2010.01092.x
Fritz B, Cysyk J, Newswanger R et al (2010) Development of an inlet pressure sensor for control in a left ventricular assist device. ASAIO J 56:180–185. doi:10.1097/MAT.0b013e3181d2a56e
Zhou M-D, Yang C, Liu Z et al (2012) An implantable Fabry-Pérot pressure sensor fabricated on left ventricular assist device for heart failure. Biomed Microdevices 14:235–245. doi:10.1007/s10544-011-9601-z
Refaat M, Chemaly E, Lebeche D et al (2008) Ventricular arrhythmias after left ventricular assist device implantation. Pacing Clin Electrophysiol 31:1246–1252. doi:10.1111/j.1540-8159.2008.01173.x
Nakahara S, Chien C, Gelow J et al (2013) Ventricular arrhythmias after left ventricular assist device. Circ Arrhythm Electrophysiol. doi:10.1161/CIRCEP.113.000113
Pettit SJ, Petrie MC, Connelly DT et al (2012) Use of implantable cardioverter defibrillators in patients with left ventricular assist devices. Eur J Heart Fail 14:696–702. doi:10.1093/eurjhf/hfs062
Pirbodaghi T, Cotter C, Bourque K (2014) Power consumption of rotary blood pumps: pulsatile versus constant-speed mode. Artif Organs 38:1024–1028. doi:10.1111/aor.12323
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Bozkurt, S. Physiologic outcome of varying speed rotary blood pump support algorithms: a review study. Australas Phys Eng Sci Med 39, 13–28 (2016). https://doi.org/10.1007/s13246-015-0405-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13246-015-0405-y