Abstract
Aiming the ‘Bridge to Recovery’ course, we have developed a novel left ventricular assist device (LVAD) controlling system. It can change the rotational speed of the continuous flow LVAD, EVAHEART, synchronized with the cardiac beat. Employing this system, we have already demonstrated that myocardial oxygen consumption (MVO2), which is considered to be equivalent to native heart load, changes in the hearts of normal goats. Herein, we examined changes in goats with acute ischemic heart failure. We studied 14 goats (56.1 ± 6.9 kg) with acute ischemic heart failure due to coronary microsphere embolization. We installed the EVAHEART and drive in four modes: “circuit-clamp”, “continuous support”, “counter-pulse”, and “co-pulse”, with 50 and 100 % bypass. In comparison to the circuit-clamp mode, MVO2 was reduced to 70.4 ± 17.9 % in the counter-pulse mode and increased to 90.3 ± 14.5 % in the co-pulse mode, whereas it was 80.0 ± 14.5 % in the continuous mode, with 100 % bypass (p < 0.05). The same difference was confirmed with 50 % bypass. This means that we may have a chance to change the native heart load by controlling the LVAD rotation in synchrony with the cardiac rhythm, so we named our controller as the Native Heart Load Control System (NHLCS). Employing changeable MVO2 with NHLCS according to the patient’s condition may provide more opportunity for native heart recovery with LVAD, especially for patients with ischemic heart diseases.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Patients with severe heart failure are treated with a multidisciplinary approach involving conservative medical management, and prognoses have improved remarkably in recent years. And some patients who are resistant to current medical treatments are considered to have indications for left ventricular assist device (LVAD) implantation or heart transplantation. But, given the scarcity of donor hearts available in Japan, the number of patients with LVAD for severe chronic heart failure is rapidly increasing. Thus, there is an urgent need to establish methods which can improve the courses of patients with LVAD, especially aiming for the ‘Bridge to Recovery’ course (BTR) with the recovery of the native heart function [1–5].
Obviously, now is the time for continuous-flow LVAD. Due to its small size, this LVAD can be installed inside the body. Patients can be discharged from the hospital, and the risk of infection would thus presumably be reduced. The favorable clinical outcomes add impetus to this trend [6]. On the other hand, many reports comparing the systemic effects of pulsatile and continuous-flow LVAD have not described favorable results [6–27]. We consider the merit of our novel system to be its potential ability to provide physiological circulation.
Given this situation, we developed and introduced a power-control unit for centrifugal LVAD, the EVAHEART (Sun Medical, Nagano, Tokyo, Japan) [28, 29]. With this unit, we drive the EVAHEART in synchrony with the native rhythm of the heart, by adjusting the time span and the rotational speed (RS) in the systolic and diastolic phases [30–36]. Ours is the first trial to evaluate continuous-flow LVAD with the pulsatile driving technique. Apparently, the LVAD can decrease the native heart load by aiding native heart function, and decreasing left ventricular end-diastolic pressure and volume. The most suitable parameter for evaluating the native heart load is myocardial oxygen consumption (MVO2), because it reflects precisely the left ventricular pressure volume area (PVA), which is considered to be energetically equivalent to the sum of external energy and potential energy [37–41]. There are many reports showing the effect of reducing the native heart load employing LVAD [42–46]. In these reports, MVO2 was reduced by LVAD, regardless of whether it was continuous [42–44] or pulsatile [45, 46]. In addition, this trend does not depend on the condition of the native heart. This effect has been confirmed in both normal [42, 44, 46] and failing [43, 45] hearts.
Thus, we have recently focused on MVO2 in order to evaluate our novel system, and found that MVO2 can be altered according to the control mode in goats with normal hearts [31]. If we can achieve an appropriate amount of MVO2 by controlling the rotation of continuous-flow LVAD, in synchrony with the cardiac beat, we may have the opportunity to enhance the function of the native heart supported by LVAD. However, before application in a clinical situation, it is necessary to test this design in models of heart failure, which might be completely different from normal heart models. Therefore, in the present study, we examined changes in MVO2 in goats with acute ischemic heart failure. Herein, our aim is to show that MVO2 can be changed employing our novel control method in an acute ischemic heart failure model.
Materials and methods
Animals
We studied 14 goats with acute ischemic heart failure (56.1 ± 6.9 kg) due to coronary microsphere embolization. The animals used in this study were maintained in accordance with the guidelines of the Committee on Animal Studies at the National Cerebral and Cardiovascular Center. This study was approved by the National Cerebral and Cardiovascular Center Animal Investigation Committee. Institutional guidelines for the care and use of laboratory animals have been observed.
Surgical procedures, implanted devices
The animals were tranquilized with ketamine hydrochloride (8–10 mg/kg intramuscularly) and then intubated and mechanically ventilated. The goats were anesthetized with isoflurane (1–3 vol/100 ml in oxygen), and then draped and prepped in the right lateral recumbent position. A left thoracotomy was performed and the fifth costal bone was resected. We retained the left intra-thoracic artery and vein to measure the aortic pressure (AoP) and the central vein pressure (CVP), and also to collect data for blood gas analysis. The blood flows in the ascending aorta, LVAD, and coronary artery (left main trunk, LMT) were measured using a flow meter. We used an electromagnetic flow meter (16–22 mm in diameter, EMF-1000, Nihon Kohden, Tokyo, Japan) for the aorta, an ultrasonic flow meter (3–4 mm, HQD3FSB, Transonic Systems, Ithaca, NY, USA) for the LMT, and another ultrasonic flow meter (16 mm, TS420, Transonic Systems) for the LVAD. After heparinization (200 U/kg), centrifugal LVAD, EVAHEART was installed. Inflow cannula was inserted from the left ventricular apex and the outflow conduit was to the descending aorta. A 6Fr conductance catheter (2S-RH-6DA-116, Taisho Biomed Instrument, Osaka, Japan) and a 4Fr micro-tip catheter pressure transducer (Millar Instruments, Houston, TX, USA) were inserted into the left ventricle from the anterior wall to collect data for the pressure volume curve of the left ventricle. Because the coronary sinus is connected to the accessory hemiazygos vein in goats, we inserted a Sarns retrograde cannula (10Fr, Terumo corp., Tokyo, Japan) into the coronary sinus from the accessory hemiazygos vein and thereby sampled coronary sinus vein blood. We calculated the MVO2 by multiplying the difference in oxygen saturation between the artery (ScaO2) and coronary sinus blood (ScvO2), the hemoglobin concentration of arterial blood (aHb), and the amount of coronary flow (CoF) (MVO2 = (ScaO2 − ScvO2) × 1.34 × aHb × CoF). The aforementioned vital data were recorded in Labchart5 (ADInstruments, Bella Vista NSW, Australia).
Making left ventricular dysfunction models
To create the acute ischemic heart failure model, we micro-embolized the left anterior descending coronary artery (LAD), as described in earlier reports [47–50]. A multipurpose Judkins catheter (4Fr, Create Medic Co., LTD, Japan) was introduced through a long sheath (4Fr × 17 cm) into the left carotid artery toward the LAD under fluoroscopic guidance. We then injected approximately 0.3 million (0.005 million/kg) microspheres (50 μm in diameter) into the LAD. Ten minutes after this injection, we observed the animal’s general condition, including aortic flow. If aortic flow exceeded 60 % of the baseline value, we arbitrarily added half the amount of microspheres (0.0025 million/kg), to achieve a total amount of 0.30 ± 0.14 million. After 30 min of further observation, we collected data to assure stable optimal cardiac function. We planned to reduce and then maintain cardiac output at approximately 60 % of the native heart function documented prior to creation of the acute ischemic heart failure model.
Study protocol, LVAD control method
We controlled the AoP and CVP to ensure stable conditions during the examination. This assured that there were no changes in the afterload or the preload of the heart. Heart rate was also controlled. We controlled these values by adjusting the volume of infusion and changing the depth of anesthesia, not by using either vasodilators or catecholamines. We used 2 % lidocaine (1 mg/kg/h) and Nifekalant hydrochloride (0.4 mg/kg/h) during the experiment to prevent ventricular arrhythmias.
We previously reported the details of our novel pump controller, which can change the RS of the EVAHEART in synchrony with the cardiac cycle [30–36]. We defined the systolic phase as 35 % of the RR interval and the diastolic phase as 65 % of the RR interval, and we input the duration of each phase according to the heart rate. Our controller can change the RS of each phase, detecting the R wave from an electrocardiogram (ECG). The bypass rate (BR) was calculated by dividing the pump flow rate (PF) by the sum of the PF and aortic flow (AoF) rates.
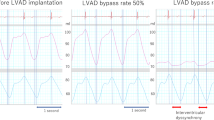
Using this controller, we compared four driving modes in this study. The first was the “circuit-clamp” (pump-off) mode, clamping the LVAD circuit so as to evaluate the conditions of the native heart. The next was the “continuous support” mode, driving the LVAD continuously at a stable RS. This is the mode we usually apply in clinical situations. The third was the “counter-pulse” mode, in which we set the RS of the systolic phase to approximately 700 rpm, the minimum speed of the LVAD system, and adjusted the RS of the diastolic phase to achieve the most appropriate BR, as needed. In this study, the BRs were set at 50 and 100 %. The latter was the “co-pulse” mode. This mode was defined in an opposite manner; we set the RS of the diastolic phase to approximately 1,000 rpm so as to avoid inducing a reverse flow inside the LVAD circuit, and adjusted the RS of the systolic phase to achieve BRs of 50 and 100 % (Fig. 1). We obtained the various data at 5 min after setting each mode. This was considered to be a sufficient period for the animal’s condition to stabilize. In this study, we mainly evaluated the MVO2. The comparison was performed by repeated analysis of variance followed by Tukey’s multiple comparison test, and a p value less than 0.05 was considered as statistically significant.
Driving modes of NHLCS. We compared MVO2 in 4 modes: “circuit-clamp”, “continuous support”, “counter-pulse” (to raise RS in diastole), and “co-pulse” (to raise RS in systole), with 50 and 100 % bypass. We defined the systolic phase as 35 % of the RR interval and the diastolic phase as 65 % of the RR interval, and set the upper rotational speed so as to achieve an appropriate bypass rate, as needed
Results
Sample waveforms of the ECG, AoP, CVP, left ventricular pressure (LVP), PF, AoF, CoF, RS, and the BR are shown in Fig. 2. In the continuous mode, the output of the native heart (AoF) was decreased by removing blood with the LVAD. The pulse pressure was also reduced as compared to that of the circuit-clamp mode. In the counter-pulse mode, increased RS in the diastolic phase produced much gentler AoP waveforms than those obtained with the continuous or the circuit-clamp mode. The gentle waveform resulted from a counter-pulse effect, much like that seen with intra-aortic balloon pumping, as we expected. Although the PF waveform became almost flat, there was no change in the total amount of PF among the three driving modes (except the circuit-clamp mode). The amount of CoF in the diastolic phase was higher than with the other modes due to the counter-pulse effect. By contrast, in the co-pulse mode, the waveforms of AoP and PF were more precipitous than with the other mode, due to the higher RS in the systolic phase. The amount of CoF in the diastolic phase was lower than with the other three modes.
Waveforms of pressure and flow. PF pump flow, AoF ascending aortic flow, CoF coronary flow, RS rotational speed, BR bypass rate. In the counter-pulse mode, increased RS resulted in gentler waveforms of AoP and PF in the diastolic phase. In the co-pulse mode, increased RS resulted in sharper waveforms of AoP and PF in the systolic phase
Hemodynamic parameters are presented in Table 1. Heart rate (HR), CVP, AoP, LVP, total flow (the sum of AoF and PF), and the BR are shown. The baseline data (before heart failure) are on the left, and those of acute ischemic heart failure models with each of the driving modes and BRs are on the right. In the right table, the total flow amount in the circuit-clamp mode is equivalent to native heart output with acute heart failure. The parameters shown are essentially 60 % of the baseline data. This means the amount of native heart output was reduced to approximately 60 %, as we expected. Considering the differences among driving modes with LVAD (other than the circuit-clamp mode), there were no significant differences in these hemodynamic parameters.
Figure 3 shows the MVO2 in each mode as a percentage of that in the circuit-clamp mode (=100 %). The MVO2 was decreased in continuous mode as compared to the circuit-clamp mode, regardless of the BR. This means that the LVAD would reduce the load on the native heart in the setting of acute heart failure. Comparing the MVO2 among each of the driving modes, it was lower in the counter-pulse mode and higher in the co-pulse mode than in the continuous mode (p < 0.05) at both the 50 and the 100 % BR. With the 50 % BR, the MVO2 was 83.9 ± 14.8 % in the counter-pulse mode and 100.8 ± 9.0 % in the co-pulse mode, whereas it was 92.2 ± 9.0 % in the continuous support mode (Fig. 3a). The same trend was detected with the 100 % BR, where the MVO2 was 70.4 ± 17.9 % in the counter-pulse mode and 90.3 ± 14.5 % in the co-pulse mode, whereas it was 80.0 ± 14.5 % in the continuous support mode (Fig. 3b).
Amount of myocardial oxygen consumption (circuit-clamp = 100). The amount of MVO2 was decreased by LVAD in acute ischemic heart failure models. MVO2 decreased in the counter-pulse mode and increased in the co-pulse mode relative to the continuous mode (p < 0.05) for both the 50 % (Fig. 3a) and the 100 % (Fig. 3b) bypass rates
Figure 4 shows the end-diastolic volume of the left ventricle (EDV) in each mode as a percentage of that in the circuit-clamp mode (=100 %). The EDV was lower in the counter-pulse mode and higher in the co-pulse mode than in the continuous mode (p < 0.05) for both the 50 and the 100 % BR. With the 50 % BR, the EDV was 94.8 ± 3.5 % in the counter-pulse mode and 100.3 ± 3.3 % in the co-pulse mode, whereas it was 97.3 ± 3.1 % in the continuous support mode. The same trend was detected with the 100 % BR, where the EDV was 83.2 ± 8.2 % in the counter-pulse mode and 93.4 ± 8.0 % in the co-pulse mode, whereas it was 88.3 ± 8.6 % in the continuous support mode.
Figure 5 shows the echocardiographic images of the left ventricle in the end-diastolic phase. The upper chamber was the left ventricle, and the inflow cannula was inserted from the apex. The size of the left ventricle was significantly decreased in the counter-pulse mode (Fig. 5b) and increased in the co-pulse mode (Fig. 5c) as compared with that in the continuous mode (Fig. 5a).
Pressure volume curves for the left ventricle in the each of the modes with acute heart failure are shown in Fig. 6. Inferior vena cava occlusion was performed to make the loop. The vertical axis is the left ventricular pressure, and the horizontal axis is the left ventricular volume. The left ventricular EDV was increased in the co-pulse mode (rightward shift), and was decreased in the counter-pulse mode (leftward shift). External work is equivalent to the area enclosed by the pressure volume curve. This is also decreased in the counter-pulse mode, and increased in the co-pulse mode as compared to the continuous mode.
Discussion
With the goal of improving the function of the native heart employing LVAD, and allowing patients a BTR course, we developed a novel driving system, the Native Heart Load Control System (NHLCS), for continuous-flow LVAD. The first concern at the development was the effect of LVAD pulsatility. Many reports have compared clinical outcomes or effects on circulatory dynamics between pulsatile and continuous-flow LVAD [6–27]. The norepinephrine level, which is higher in the non-pulsatile than in the pulsatile LVAD, may affect oxygen metabolic conditions and worsen systemic oxygen uptake in the acute phase [10–13]. The systemic vascular resistance response to norepinephrine decreased markedly with non-pulsatile flow. From the viewpoint of hemodynamic change, pulsatile flow generates more energy, which may be beneficial for vital organ perfusion [14], with the amount depending on the timing of mechanical cardiac beating [15, 16]. Pulsatile flow may be beneficial for the end-organ micro-circulation [17] and for coronary flow [18], but the effect on brain metabolism remains a source of controversy [20–22]. The vascular system is also affected by pulsatility [23]. Structural change in the aortic wall, caused by atrophy of aortic smooth muscle cells, occurs with non-pulsatile flow [24–27]. In summary, the overall systemic effects of long-term support with continuous LVAD have yet to be determined. We simply do not know which effects, if any, are beneficial or harmful. However, as to clinical outcomes, unfavorable results with pulsatile LVAD have been reported [6]. These may, however, arise from the higher risks of complications with pulsatile LVAD. In our view, the ability of pulsatile LVAD to provide physiological circulation is a benefit that cannot be denied.
Thus, we developed the NHLCS, with which we can drive the continuous-flow LVAD synchronized with the actual cardiac rhythm. Our recent report was the first description of such a pulsatile driving technique [30–36]. Herein, we aimed to confirm the effect of this system on the native heart load from the aspect of MVO2, which we consider to be the most important factor in evaluating native heart load changes. The pressure–volume area (PVA) is defined as the area framed by the lines of the end-diastolic and systolic pressure volume relationship and the pressure–volume curve of the systolic phase. The rectangular area within the pressure volume loop represents the external work performed, and the triangular area under the Emax line (end-systolic slope of the pressure volume relationship as maximum elastance) represents the elastic potential energy stored in the left ventricle. Therefore, PVA is considered to be equivalent to the sum of external work and potential energy. Furthermore, PVA is known to show a strong positive correlation with MVO2. [37–41] Thus, the MVO2, the amount of oxygen used by the left ventricle, is closely related to cardiac energy dynamics, and is the most useful factor for evaluating the native heart load. The MVO2 is the optimal parameter to represent oxygen utilization and energy use by the myocardium under ischemic conditions. This is why we chose MVO2 as the parameter for assessing the NHLCS effect on the native heart load. In addition, in this study, we aimed to evaluate the effect of NHLCS on acute heart failure models. In these models, the myocardial oxygen demand is inevitably higher than in normal heart models, due to the suppression of myocardial perfusion. High end-diastolic pressure and sympathetic hyperactivity with high catecholamine concentrations may greatly exacerbate these demands. However, in response to the increased oxygen requirements, low oxygen delivery may occur due to insufficient myocardial perfusion, caused by stenosis of the coronary artery itself, high intra-myocardial pressure, low blood pressure and so on. It is quite interesting to evaluate changes in the myocardial oxygen dynamic state with NHLCS under ischemic conditions.
In such a situation, we have demonstrated the possibility of changing the MVO2 intentionally with our NHLCS. First, when comparing the circuit-clamp and continuous modes, MVO2 was decreased with LVAD whether the myocardium was ischemic or not. This result is consistent with the outcomes of other reports [42–46]. Simply put, this observation means that the native heart load is reduced with LVAD because it carries part of the burden on the native heart (external workload). This may partially be explained by the decrease in EDV with LVAD [41]. According to the Frank-Starling law of the heart, the external work applied against afterload relies upon the EDV. This means that cardiac energy metabolism is greatly influenced by EDV. Theoretically, it is easy to understand that LVAD can decrease the native heart load and MVO2 via its volume unloading effect.
Changing our frame of reference to the difference in MVO2 between the two experimental driving modes, the MVO2 amount was decreased with the counter-pulse mode and increased with the co-pulse mode, as compared with the continuous mode. This trend was apparent regardless of the cardiac state or the BR [31], and the same trend was recognized for the difference in EDV [36]. The EDV was confirmed to be one of the determinants of MVO2, based on our present results. Considering the equivalence between MVO2 and the native heart load, the significance of this result is that we may have a chance to produce a desirable load suiting the condition of the native heart by choosing our novel driving mode of continuous-flow LVAD. With the counter-pulse mode, we can reduce the native heart load by unloading the ventricular volume for patients with acute ischemic heart failure in the counter-pulse mode. However, when full LVAD support is needed for a severely failing heart, it may be more favorable to choose the counter-pulse mode with its heart load reduction effect. Furthermore, raising RS in the diastolic phase may induce left ventricular unloading and provide an AoP boost-up effect, which would especially benefit patients with ischemia via its so-called counter-pulse effect mimicking that of intra-aortic balloon pumping. On the other hand, in the co-pulse mode, the load is increased to the level of the circuit-clamp mode, even though the heart has an installed LVAD. We can create more strain on the native heart by loading it with excess volume when we want to train the native heart, or to assess the possibility of weaning from the LVAD in the recovery stage. The strain level is almost the same as if the LVAD is detached. Therefore, if we choose the appropriate mode of NHLCS according to the various clinical situations encountered on the road to recovery, we may have a chance to create the ideal conditions for the native heart by providing the most suitable load.
These results may be mistakenly understood to mean that the LVAD has the potential only to provide a small amount of oxygen. It may well be assumed that it would be natural for the native heart to use more oxygen than in the condition without LVAD, after attaching the LVAD to the acute ischemic heart, and establishing the system to provide more CoF and oxygen to myocardium. However, this would simply be due to the decrease in myocardial oxygen demand, secondary to the reduced native heart load and the sufficient CoF. Especially with the counter-pulse mode, the oxygen demand may be decreased with the EDV and native heart load reductions. However, conversely, with the co-pulse mode, the demand may be increased.
Goldstein implicated various indices of energy metabolism, external work, PVA, the tension-time index, the integral of systolic force, and intra-cardiac pressure, as major determinants of MVO2, in a report on the effects of a low BR driving the LVAD [43]. On the other hand, there were no correlations between MVO2 and parameters associated with the oxygen delivery potential (AoP in diastole, the amount of CoF, and myocardial oxygen delivery [CoF × O2 content of arterial blood]). They also noted that the severely failing myocardium was quite sensitive to even the slight relief provided by LVAD, not because the oxygen supply was augmented, but because oxygen requirements were diminished. When we install the LVAD in an ischemic heart, MVO2 depends on the native heart load, not on the oxygen delivery condition. It may be smaller than that without LVAD, because of the substantial reduction in the native heart load with diminished oxygen demand, rather than because of an insufficient oxygen supply. These results are in good agreement with our observations.
This study has limitations. First of all, our results are based on an acute ischemic heart failure model, such that it is unclear whether the same results would apply to the chronically failing heart in clinical situations. However, the change in MVO2 depends on the ventricular hemodynamic state (EDV, etc.), as detailed above. Therefore, we believe the same trend would be seen in the chronically failing heart. We have already begun to evaluate the effects of NHLCS on the native heart, by creating chronic heart failure models. We aim to ascertain the chronic effects of NHLCS, not only on the hemodynamic state and energy metabolism, but also on tissues and genetic expressions including the reverse remodeling effect. The second limitation involves the mechanical factor. Herein, we defined the spans of the systolic and diastolic phases as 35 and 65 % of the RR interval based on our experience with goats. However, there may well be marked alterations in these parameters among individuals or according to the state of the native heart. In addition, the timing of synchronization is problematic, because the continuous-flow LVAD is characterized by the RS changing gradually, i.e., within minutes rather than seconds. If we incorporate a signal to change the RS, it will still take some time to obtain the ideal RS. Therefore, we must confirm whether the timings of the maximum or minimum RS are attuned to the cardiac cycle. For this study, we employed data with which the LVAD rotation was in synchrony with the heartbeat, in terms of span and timing. We are now endeavoring to adjust the system for NHLCS, which has the capacity to change the span and timing of synchronization according to the native heartbeat. The third limitation may be the site of the outflow graft. Usually, we place the outflow graft of the LVAD at the ascending aorta. In this study, however, we placed it in the descending aorta, because of the shortness of the ascending aorta in goats. These animals have a bovine carotid artery which diverges from the ascending aorta near the base of the heart. Furthermore, all of the carotid and subclavian arteries bifurcate from this bovine artery. We will need to examine outflow grafts placed at the ascending aorta in the future. Although these differences may have impacted the cardiac after-load, many reports suggest equivalence between these outflow graft sites [51, 52].
Herein, we demonstrated the possibility of changing the native heart load employing NHLCS, based on changes in MVO2. This means that a desirable native heart load can be produced by choosing our novel driving mode of continuous-flow LVAD. We may thus be able to create the appropriate conditions for BTR. We can reduce the native heart load for patients with severe heart failure in the counter-pulse mode. On the other hand, we can create more strain on the native heart in the co-pulse mode when assessing the possibility of weaning from the LVAD in the recovery stage. Further studies are underway, including analyses of cardiac energy metabolism and myocardial perfusion using chronic heart failure models.
Conclusion
Based on our experimental results in goats with acute ischemic heart failure, LVAD can change MVO2. The MVO2 was reduced with the counter-pulse mode and increased with the co-pulse mode, relative to the continuous support mode, which we usually apply in clinical practice. This means that we can change the native heart load by controlling the LVAD rotation in synchrony with the cardiac rhythm. The changeable native heart load made by NHLCS may enhance the chance of BTR, especially for patients with ischemic heart diseases.
References
Frazier OH, Benedict CR, Radovancevic B, Bick RJ, Capek P, Springer WE, Macris MP, Delgado R, Buja LM. Improved left ventricular function after chronic left ventricular unloading. Ann Thorac Surg. 1996;62:675–81. discussion 681-672.
Birks EJ, Tansley PD, Hardy J, George RS, Bowles CT, Burke M, Banner NR, Khaghani A, Yacoub MH. Left ventricular assist device and drug therapy for the reversal of heart failure. N Engl J Med. 2006;355:1873–84.
Dandel M, Weng Y, Siniawski H, Potapov E, Lehmkuhl HB, Hetzer R. Long-term results in patients with idiopathic dilated cardiomyopathy after weaning from left ventricular assist devices. Circulation. 2005;112:I37–45.
Saito S, Nishinaka T, Yamazaki K. Long-term circulatory support with a left ventricular assist device therapy in Japan. Circ J. 2010;74:624–5.
Matsumiya G, Saitoh S, Sakata Y, Sawa Y. Myocardial recovery by mechanical unloading with left ventricular assist system. Circ J. 2009;73:1386–92.
Slaughter MS, Rogers JG, Milano CA, Russell SD, Conte JV, Feldman D, Sun B, Tatooles AJ, Delgado RM, Long JW, Wozniak TC, Ghumman W, Farrar DJ, Frazier OH, HeartMate II Investigators. Advanced heart failure treated with continuous-flow left ventricular assist device. N Engl J Med. 2009;361:2241–51.
Ji B, Ündar A. An evaluation of the benefits of pulsatile versus nonpulsatile perfusion during cardiopulmonary bypass procedures in pediatric and adult cardiac patients. ASAIO J. 2006;52:357–61.
Loebe M, Koster A, Sänger S, Potapov EV, Kuppe H, Noon GP, Hetzer R. Inflammatory response after implantation of a left ventricular assist device: comparison between the axial flow MicroMed DeBakey VAD and the pulsatile Novacor device. ASAIO J. 2001;47:272–4.
Nishinaka T, Tatsumi E, Taenaka Y, Takano H, Koyanagi H. Influence of pulsatile and nonpulsatile left heart bypass on the hormonal circadian rhythm. ASAIO J. 2000;46:582–6.
Toda K, Tatsumi E, Taenaka Y, Masuzawa T, Miyazaki K, Wakisaka Y, Nakatani T, Baba Y, Eya K, Takano H. How does the sympathetic nervous system behave during non pulsatile circulation? ASAIO J. 1995;41:M465–8.
Tatsumi E, Toda K, Taenaka Y, Miyazaki K, Masuzawa T, Nakatani T, Baba Y, Yagura A, Eya K, Wakisaka Y. Acute phase responses of vasoactive hormones to non pulsatile systemic circulation. ASAIO J. 1995;41:M460–5.
Tatsumi E, Miyazaki K, Toda K, Taenaka Y, Nakatani T, Baba Y, Masuzawa T, Wakisaka Y, Eya K, Nishimura T, Takewa Y, Ohno T, Takano H. Influence of non pulsatile systemic circulation on tissue blood flow and oxygen metabolism. ASAIO J. 1996;42:M757–62.
Tatsumi E, Miyazaki K, Toda K, Taenaka Y, Nakatani T, Baba Y, Masuzawa T, Eya K, Wakisaka Y, Nishimura T, Takewa Y, Ohno T, Takano H. Altered oxygen metabolic conditions associated with increased norepinephrine levels in a nonpulsatile systemic circulation. ASAIO J. 1996;42:M854–7.
Undar A, Masai T, Frazier OH, Fraser CDJ. Pulsatile and nonpulsatile flows can be quantified in terms of energy equivalent pressure during cardiopulmonary bypass for direct comparisons. ASAIO J. 1999;45:610–4.
Vandenberghe S, Segers P, Antaki JF, Meyns B, Verdonck PR. Hemodynamic modes of ventricular assist with a rotary blood pump: continuous, pulsatile, and failure. ASAIO J. 2005;51:711–8.
Bartoli CR, Giridharan GAS, Litwak KNP, Sobieski M, Prabhu SD, Slaughter MS, Koenig SCS. Hemodynamic responses to continuous versus pulsatile mechanical unloading of the failing left ventricle. ASAIO J. 2010;56:410–6.
Orime Y, Shiono M, Nakata K, Hata M, Sezai A, Yamada H, Iida M, Kashiwazaki S, Nemoto M, Kinoshita J, Kojima T, Saito T, Sezai Y. The role of pulsatility in end-organ microcirculation after cardiogenic shock. ASAIO J. 1996;42:M724–8.
Jung JS, Son HS, Lim CH, Sun K. Pulsatile versus nonpulsatile flow to maintain the equivalent coronary blood flow in the fibrillating heart. ASAIO J. 2007;53:785–90.
Satoh H, Miyamoto Y, Shimazaki Y, Kadoba K, Masai T, Yagura A, Matsuda H. Comparison between pulsatile and nonpulsatile circulatory assist for the recovery of shock liver. ASAIO J. 1995;41:M596–600.
Anstadt MP, Tedder M, Hegde SS, Perez-Tamayo RA, Crain BJ, Khian Ha VL, Abdel-Aleem S, White WD, Lowe JE. Pulsatile versus nonpulsatile reperfusion improves cerebral blood flow after cardiac arrest. Ann Thorac Surg. 1993;56:453–61.
Henze T, Stephan H, Sonntag H. Cerebral dysfunction following extracorporeal circulation for aortocoronary bypass surgery: no differences in neuropsychological outcome after pulsatile versus nonpulsatile flow. Thorac Cardiovasc Surg. 1990;38:65–8.
Nishinaka T, Tatsumi E, Nishimura T, Taenaka Y, Imada K, Takano H, Koyanagi H. Effects of reduced pulse pressure to the cerebral metabolism during prolonged nonpulsatile left heart bypass. Artif Organs. 2000;24:676–9.
Wilson E, Mai Q, Sudhir K, Weiss RH, Ives HE. Mechanical strain induces growth of vascular smooth muscle cells via autocrine action of PDGF. J Cell Biol. 1993;123:741–7.
Nishimura T, Tatsumi E, Takaichi S, Taenaka Y, Wakisaka Y, Nakatani T, Masuzawa T, Takewa Y, Nakamura M, Endo S, Nakata M, Takano H. Prolonged nonpulsatile left heart bypass with reduced systemic pulse pressure causes morphological changes in the aortic wall. Artif Organs. 1998;22:405–10.
Nishimura T, Tatsumi E, Nishinaka T, Taenaka Y, Nakata M, Takano H. Prolonged nonpulsatile left heart bypass diminishes vascular contractility. Int J Artif Organs. 1999;22:492–8.
Nishimura T, Tatsumi E, Taenaka Y, Nishinaka T, Nakatani T, Masuzawa T, Nakata M, Nakamura M, Endo S, Takano H. Effects of long-term nonpulsatile left heart bypass on the mechanical properties of the aortic wall. ASAIO J. 1999;45:455–9.
Nishimura T, Tatsumi E, Takaichi S, Taenaka Y, Wakisaka Y, Nakatani T, Masuzawa T, Takewa Y, Nakamura M, Endo S, Sohn YS, Takano H. Morphologic changes of the aortic wall due to reduced systemic pulse pressure in prolonged non pulsatile left heart bypass. ASAIO J. 1997;43:M691–5.
Yamazaki K, Kihara S, Akimoto T, Tagusari O, Kawai A, Umezu M, Tomioka J, Kormos RL, Griffith BP, Kurosawa H. EVAHEART: an implantable centrifugal blood pump for long-term circulatory support. Jpn J Thorac Cardiovasc Surg. 2002;50:461–5.
Yamazaki K, Saito S, Kihara S, Tagusari O, Kurosawa H. Completely pulsatile high flow circulatory support with a constant-speed centrifugal blood pump: mechanisms and early clinical observations. Gen Thorac Cardiovasc Surg. 2007;55:158–62.
Ando M, Takewa Y, Nishimura T, Yamazaki K, Kyo S, Ono M, Tsukiya T, Mizuno T, Taenaka Y, Tatsumi E. A novel counterpulsation mode of rotary left ventricular assist devices can enhance myocardial perfusion. J Artif Organs. 2011;14:185–91.
Ando M, Takewa Y, Nishimura T, Yamazaki K, Kyo S, Ono M, Tsukiya T, Mizuno T, Taenaka Y, Tatsumi E. Coronary vascular resistance increases under full bypass support of centrifugal pumps-relation between myocardial perfusion and ventricular workload during pump support. Artif Organs. 2012;36(1):105–10.
Ando M, Nishimura T, Takewa Y, Yamazaki K, Kyo S, Ono M, Tsukiya T, Mizuno T, Taenaka Y, Tatsumi E. Electrocardiogram-synchronized rotational speed change mode in rotary pumps could improve pulsatility. Artif Organs. 2011;35:941–7.
Ando M, Nishimura T, Takewa Y, Kyo S, Ono M, Taenaka Y, Tatsumi E. Creating an ideal “off-test mode” for rotary left ventricular assist devices: establishing a safe and appropriate weaning protocol after myocardial recovery. J Thorac Cardiovasc Surg. 2012;143(5):1176–82.
Ando M, Nishimura T, Takewa Y, Ogawa D, Yamazaki K, Kashiwa K, Kyo S, Ono M, Taenaka Y, Tatsumi E. A novel counterpulse drive mode of continuous-flow left ventricular assist devices can minimize intracircuit backward flow during pump weaning. J Artif Organs. 2011;14:74–9.
Ando M, Nishimura T, Takewa Y, Ogawa D, Yamazaki K, Kashiwa K, Kyo S, Ono M, Taenaka Y, Tatsumi E. What is the ideal off-test trial for continuous-flow ventricular-assist-device explantation? Intracircuit back-flow analysis in a mock circulation model. J Artif Organs. 2011;14:70–3.
Umeki A, Nishimura T, Ando M, Takewa Y, Yamazaki K, Kyo S, Ono M, Tsukiya T, Mizuno T, Taenaka Y, Tatsumi E. Alteration of LV end-diastolic volume by controlling the power of the continuous-flow LVAD, so it is synchronized with cardiac beat: development of a native heart load control system (NHLCS). J Artif Organs. 2012;15(2):128–33.
Suga H, Goto Y, Futaki S, Kawaguchi O, Yaku H, Hata K, Takasago T. Systolic pressure-volume area (PVA) as the energy of contraction in Starling’s law of the heart. Heart Vessels. 1991;6:65–70.
Suga H, Yasumura Y, Nozawa T, Futaki S, Tanaka N, Uenishi M. Ventricular systolic pressure-volume area (PVA) and contractile state (Emax) determine myocardial oxygen demand. Adv Exp Med Biol. 1988;222:421–30.
Suga H, Igarashi Y, Yamada O, Goto Y. Cardiac oxygen consumption and systolic pressure volume area. Basic Res Cardiol. 1986;81:39–50.
Suga H. Total mechanical energy of a ventricle model and cardiac oxygen consumption. Am J Physiol. 1979;236:H498–505.
Suga H. Ventricular energetics. Physiol Rev. 1990;70:247–77.
Tuzun E, Eya K, Chee HK, Conger JL, Bruno NK, Frazier OH, Kadipasaoglu KA. Myocardial hemodynamics, physiology, and perfusion with an axial flow left ventricular assist device in the calf. ASAIO J. 2004;50:47–53.
Goldstein AH, Monreal G, Kambara A, Spiwak AJ, Schlossberg ML, Abrishamchian AR, Gerhardt MA. Partial support with a centrifugal left ventricular assist device reduces myocardial oxygen consumption in chronic, ischemic heart failure. J Card Fail. 2005;11:142–51.
Voitl P, Vollkron M, Bergmeister H, Wieselthaler G, Schima H. Coronary hemodynamics and myocardial oxygen consumption during support with rotary blood pumps. Artif Organs. 2009;33:77–80.
Kawaguchi O, Pae WE, Daily WB, Sapirstein JS, Pierce WS. Left ventricular mechanoenergetics during asynchronous left atrial-to-aortic bypass. Effects of pumping rate on cardiac workload and myocardial oxygen consumption. J Thorac Cardiovasc Surg. 1995;110:793–9.
Pantalos GM, Marks JD, Riebman JB, Everett SD, Burns GL, Burton NA, DePaulis R. Left ventricular oxygen consumption and organ blood flow distribution during pulsatile ventricular assist. ASAIO Trans. 1988;34:356–60.
Klocke R, Tian W, Kuhlmann MT, Nikol S. Surgical animal models of heart failure related to coronary heart disease. Cardiovasc Res. 2007;74:29–38.
Dixon JA, Spinale FG. Large animal models of heart failure: a critical link in the translation of basic science to clinical practice. Circ Heart Fail. 2009;2:262–71.
Gill RM, Jones BD, Corbly AK, Wang J, Braz JC, Sandusky GE, Shen W. Cardiac diastolic dysfunction in conscious dogs with heart failure induced by chronic coronary microembolization. Am J Physiol Heart Circ Physiol. 2006;291:H3154–8.
Hayashi Y, Hamada M, Hiwada K. Characterization of left ventricular opacification using sonicated serum albumin in patients with dilated cardiomyopathy and myocardial infarction. Jpn Circ J. 1998;62:91–6.
Tuzun E, Narin C, Gregoric ID, Cohn WE, Frazier OH. Ventricular assist device outflow-graft site: effect on myocardial blood flow. J Surg Res. 2011;171:71–5.
May-Newman K, Hillen B, Dembitsky W. Effect of left ventricular assist device outflow conduit anastomosis location on flow patterns in the native aorta. ASAIO J. 2006;52:132–9.
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Umeki, A., Nishimura, T., Takewa, Y. et al. Change in myocardial oxygen consumption employing continuous-flow LVAD with cardiac beat synchronizing system, in acute ischemic heart failure models. J Artif Organs 16, 119–128 (2013). https://doi.org/10.1007/s10047-012-0682-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10047-012-0682-0