Abstract
Long noncoding RNAs (lncRNAs) are implicated in various biological processes, such as cell proliferation, differentiation, apoptosis, migration, and invasion. They are also key players in various biological pathways. LncRNA was considered as ‘translational noise’ before 1980s. It has been reported that lncRNAs are aberrantly expressed in different cancers, either as oncogene or tumor suppressor gene. Therefore, more and more lncRNAs are recognized as potential diagnostic biomarkers and/or therapeutic targets. As competitive endogenous RNA, lncRNAs can interact with microRNA to alter the expression of target genes, which may have extensive clinical implications in cancers, including diagnosis, treatment, prognosis, and chemoresistance. This review comprehensively summarizes the functions and clinical relevance of lncRNAs in digestive system cancers, especially as a potential tool to overcome chemoresistance.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Noncoding RNA is a type of RNA that is transcribed, but not translated into protein, such as transfer RNA, ribosomal RNA, and microRNA. It has been demonstrated that noncoding RNAs can act as key regulators of gene expression in diverse cellular systems, biological processes, and various pathways (Yu et al. 2019b; Ferre et al. 2016; Barangi et al. 2019; Lin and Yang 2018). Long noncoding RNA (lncRNA) is defined as the transcripts exceeding 200 nucleotides that are not translated into protein. In the late 1980s and early 1990s, lncRNA H19 and Xist were first found in mouse (Brannan et al. 1990; Brockdorff et al. 1991). Since then, lncRNA has attracted significant attention and becomes one of the hot topics of research interest. lncRNAs can be divided into several subclasses (Fig. 1A) according to their genomic position related to the neighboring genetic code gene: sense, antisense, bidirectional or divergent, intronic and intergenic lncRNA (Herrera-Solorio et al. 2017). Sense, antisense, bidirectional, or divergent lncRNA, respectively, represent the transcription direction of lncRNA and its neighboring protein-coding genes, either in the same, or opposite, or at different directions. Conversely, intronic and intergenic lncRNA represent whether a lncRNA is transcribed from the intron region of the gene or from the intergenic region. lncRNAs are generally, but not exclusively, spliced and transcribed by RNA polymerase II and were considered as ‘translational noise’ previously (Quinn and Chang 2016). However, increasing evidence has proven that lncRNAs perform many functions, such as mediating chromatin remodeling, acting as scaffold linking different proteins interaction and regulate transcription regulation (Chen and Carmichael 2010). At the transcriptional level, the production of mRNA can be affected by lncRNAs. Some lncRNAs gene are located at the promoter region, upstream of the coding gene, and can be transcribed into corresponding lncRNAs, which then acts as a cis-acting element to interfere with the transcription of downstream genes; thereby, affecting the production of mRNA (Martens et al. 2004). Previous studies have also found that lncRNA can form a complex with ribonucleoprotein to regulate gene expression (Ponting et al. 2009). At post-transcriptional level, lncRNA can form complex with pre-mRNA, therefore, regulate the gene expression at post-transcription stage (Beltran et al. 2008). Also, lncRNA can bind to miRNA and indirectly affect the downstream process.
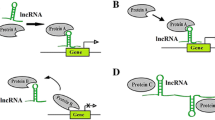
A The type of lncRNAs. Sense, antisense, bidirectional or divergent lncRNA respectively, represent the transcription direction of lncRNA and its neighboring protein-coding genes, either in the same, or opposite, or at different directions. Intronic and intergenic lncRNA represent whether a lncRNA is transcribed from the intron region of the gene or from the intergenic region. B The roles and functions of lncRNA. I LncRNA can mediate gene expression in different ways: lncRNA can form complex with chromatin, different proteins or ribonucleoprotein, lncRNA can acts as cis-acting element to interfere with the transcription of downstream mRNA production, target gene will be released from miRNA when lncRNA sponge to miRNA. II LncRNAs are involved in many biological processes which are investigated in cancer study such as cell proliferation, apoptosis, migration, invasion, and lymph node metastasis. Aberrant expression of lncRNAs is usually found in cancers, and it has been proved to play an important role in various pathways as well as in chemoresistance study
LncRNAs are involved in many biological processes, including cell proliferation, migration, and invasion (Liu et al. 2017), differentiation (Chen et al. 2017c), metastasis (Chen et al. 2017d), inflammation (Du et al. 2017b), angiogenesis (Li et al. 2017d), and metabolism (Fan et al. 2017), and so can regulate the pathophysiological processes in cancer and other human diseases. Aberrant expression of lncRNAs is usually found in cancers, which is associated with tumorigenesis by promoting malignant biological behaviors of tumor cells, such as proliferation, invasion, and metastasis (Xiao et al. 2017a; Weidle et al. 2017). Cancer-related lncRNAs can be oncogene or tumor suppressor gene depending on their dysregulated expression and corresponding function.
The number of lncRNA-related studies is increasing rapidly and new lncRNAs are identified continuously, but their functions have not been completely characterized. In this mini-review, we briefly summarize the lncRNAs implicated in digestive system cancers and their clinical relevance, with a particular focus on the multiple roles of lncRNAs in genetic alteration, molecular mechanisms and signaling pathways involved in tumor progression, metastasis, and chemoresistance.
lncRNAs in cancers
Numerous reports have showed aberrant expression, various functions as well as chemoresistance of lncRNAs in different cancers (the functions and roles of lncRNAs are displayed in Fig. 1B). Some lncRNAs are up-regulated, as oncogenes, while others are down-regulated, as tumor suppressors.
Some lncRNAs are involved in many biological processes such as cell proliferation, migration, invasion, apoptosis, lymph node metastasis, and pathological differentiation (Liu et al. 2018f), although the specific mechanisms remain unclear so far. Some lncRNAs are functional in different pathways such as Wnt/β-catenin signaling pathway (Zhou et al. 2019a), p53 pathway, and AKT/mTOR signaling pathway (Yu et al. 2017b). In addition, there are lncRNAs that are involved in more than one pathway such as lncRNA MEG3, NF-κB pathway, p53 signaling pathway, and PI3K/Akt pathway (Zhang et al. 2017a, 2018g; Zhu et al. 2019b). miRNA is a type of noncoding RNA with 19–25 nucleotides. miRNA is involved in many aspects of biological regulation, including regulation of cell cycle, differentiation, development, metabolism, and body aging. miRNA also serves as a potential tumor molecular marker. A change in miRNA expression is related to the progression of the tumor (Lee and Dutta 2009). lncRNAs may act as a regulator by targeting miRNA, functioning as a ‘sponge’ or competing endogenous RNA (ceRNA). Thus, it can diminish the regulatory effect of miRNAs on target mRNA. For instance, 3’-untranslated region of lncRNA FOXD2-AS1 could directly bind to miR-150-5p in breast cancer (Jiang et al. 2019a); NR2F2-AS1 functions as ceRNA that directly binds to miR-320b to regulate downstream target gene and promote tumorigenesis in nonsmall cell lung cancer (Zhang et al. 2018e); and lncRNA UCA1 regulates colorectal cancer through modulating miR-28-5p, where UCA1 binds to miR-28-5p, then targets HOXB3 to mediate cell proliferation and migration of colorectal cancer cells (Cui et al. 2019). These lncRNAs can be potential therapeutic targets because they play pivotal functions via miRNA to modulate the malignancy and tumorigenesis of diverse cancers.
lncRNAs in digestive system cancers
Colorectal cancer (CRC) and gastric cancer are among the most common malignant cancers associated with high morbidity and mortality. With the increasing cases of colorectal cancer, the development of diagnostic indicators for early diagnosis and treatment has become a common concern of many scientific and clinical researchers. It is currently known that the carcinoembryonic antigen level is a good tumor marker and can be used as a reference or an indicator in judging the disease development, treatment efficacy, as well as in monitoring and prognostic evaluation. However, the lack of high specificity and sensitivity is making it unsuitable and ineffective as an early diagnosis tool of colorectal cancer (Wang et al. 2014).
A study has shown that after a 5-year follow-up, around 70% patients with gastric cancer (GC) died from this disease globally (Verdecchia et al. 2007). Smoking, alcohol drinking, eating habits, and Helicobacter pylori infection are significant risks of GC. The interaction between host-related factors and environmental factors is a key factor in the high mortality rate of GC. The patients were often diagnosed as GC in late stages, when the metastasis has occurred, and the prognosis is quite poor while the palliative chemotherapy is the main treatment method (Digklia and Wagner 2016). Pancreatic cancer (PC) is not common, but aggressive at high growth rate, often diagnosed at late stages. Owing to the poor early diagnosis, most patients have already lost the surgery opportunities when diagnosed. Although the median survival period after diagnosis is around 2–8 months, the 5-year survival rate is only 5% which is largely contributed to the poor prognosis of the disease. The etiology of pancreatic cancer is unclear so far, and its occurrence and development are an extremely complex process. Epidemiological investigation results have shown that the occurrence and development of pancreatic cancer are related to a variety of risk factors. Long-term smoking, high-fat diet, and chronic pancreatitis or concomitant diabetes are nongenetic factors in the onset of pancreatic cancer (Rebelo et al. 2017).
In the recent years, more studies have shown that lncRNA participates in the regulation of tumors at both molecular and cellular levels, rendering their potential as important indicators for the diagnosis, treatment, and prognosis of digestive system cancer (Lv and Huang 2019; Deng et al. 2020; Gao et al. 2020). In addition, miRNAs have been proven to be involved in tumorigenesis and development, angiogenesis, metastasis, invasion, and apoptosis by inhibiting/promoting the expression of oncogene. The ceRNA regulatory network composed of lncRNA–miRNA–mRNA network has been revealed in digestive system cancer study (Gong et al. 2018; Lv and Huang 2019; Chen et al. 2017b). lncRNAs can bind to miRNAs as ceRNA, suppress the expression of miRNA, and negatively regulate the downstream target gene. Thus, by interfering the lncRNA expression, the proliferation, and invasion of cancer cells can be inhibited, and cancer progression can be prevented. Some lncRNAs are shared by different digestive system cancers, but function via different mechanisms. For example, lncRNA CRNDE acts as a regulator by targeting miR-217, miR-136, miR-181a-5p (Han et al. 2017) in CRC, miR-384 in PC (Wang et al. 2017a), and miR-145 in GC (Hu et al. 2017). Several new lncRNAs are found only in a single cancer and implicated in some basic biological processes. Table 1 summarizes the functions and clinical relevance of a wide range of lncRNAs in terms of digestive system cancer type.
lncRNA and tumor chemoresistance in digestive system cancers
Even though chemotherapy is currently an effective treatment in cancer, chemoresistance is still one of the major barriers that leads to cancer relapse and eventually treatment failure (Zheng 2017). One of the underlying mechanisms of chemotherapy is to induce apoptosis, however, cancer cells may improve survival via autophagy and so show poor sensitivity to chemotherapy (Yang and Klionsky 2010). Some biological processes, such as epithelial–mesenchymal transition (EMT) process could confer drug resistance (Mitra et al. 2015). Prominent activation of some signaling pathways (e.g., Erk/MAPK and p38/MAPK pathways) has been reported in case of chemoresistance (Chung et al. 2012). Interestingly, the regulation of lncRNA can induce drug resistance by interfering with the drug efflux system, drug metabolism, DNA repair, cell cycle, EMT, and others. Table 2 summarizes the involvement and critical roles played by lncRNA in chemoresistance to several commonly used chemotherapy drugs in the treatment of digestive system cancers.
Chemoresistance to 5-fluorouracil (5-Fu)
Among all the chemotherapeutic drugs, 5-Fu is the most commonly used in digestive cancers. LncRNA SLC25A25-AS1 expression was significantly decreased in both serum and tumor tissues of CRC patients. In addition, it was reported that SLC25A25-AS1 was associated with EMT process. Upregulation of SLC25A25-AS1 led to declined mesenchymal characteristics such as mesenchymal marker vimentin and snail expression. Elevated levels of Erk phosphorylation and p38 downregulation were found in cell line which suggests that SLC25A25-AS1 affects the activation of these pathways. Downregulation of SLC25A25-AS1 apparently increased chemoresistance, whereas overexpression increased the sensitivity to 5-Fu and DOX in CRC cell line (Li et al. 2016).
Another example of EMT-related lncRNA was LEIGC in GC. The overexpression of LEIGC promoted the sensitivity of GC cells to 5-Fu by inhibiting EMT (Han et al. 2014). lncRNA CRNDE was upregulated in CRC tissue sample and miR-181a-5p was identified as the inhibitory target. An increasing serial of concentrations of 5-Fu was applied to make lncRNA CRNDE knockdown or overexpression in CRC cells. The results indicated that CRNDE knockdown and miR-181a-5p overexpression increased the sensitivity of CRC cells to 5-Fu therapy, but the sensitivity was decreased in CRNDE overexpression and miR-181a-5p knockdown group (Han et al. 2017).
Another lncRNA HOTAIR also contributes to 5-Fu resistance by inhibiting miR-218 and promoting NF-κB signaling pathway in CRC (Yu et al. 2017b), by inhibiting miR-203a-3p and activating Wnt/β-catenin signaling pathway (Xiao et al. 2018). The expression of miR-31 was up-regulated in 5-Fu-resistant cell line, while lncRNA ENST00000547547 could bind to miR-31 and suppress its expression, indicating that ENST00000547547 diminished the chemoresistance to 5-Fu via competitively binding to miR-31 (LI et al. 2017b).
Thymidylate synthase (TYMS) was thought to be a critical target when 5-Fu exerts its anticancer effect (Marquez-Jurado et al. 2018). The expression of lncRNA XIST was reported to be increased in 5-Fu resistant CRC cell lines and knockdown of XIST could boost the sensitivity through regulating TYMS expression (Xiao et al. 2017b). Another research showed that TYMS was the direct downstream target of lncRNA TUG1, knockdown of which could re-sensitize the cells to 5-Fu and cause CRC cell apoptosis (Wang et al. 2019c).
ABCC1 was highly linked to the emergence of chemoresistance in cancer cells (Gottesman et al. 2002). High level of lncRNA ANRIL was demonstrated in CRC tissues and cells. Knockdown of ANRIL enhanced the sensitivity to 5-Fu in HCT116 and SW480. Further study revealed that ANRIL could affect the expression of ABCC1 by regulating Let-7a (Zhang et al. 2018h).
Autophagy also played an important role in chemotherapy. SIRT1-mediated autophagy could be upregulated by lncRNA H19 via modulating miR-194-5p to confer 5-Fu resistance in CRC (Wang et al. 2018d). Another lncRNA, i.e., SNHG6, also regulates autophagy to induce 5-Fu chemoresistance by sponging miR-26a-5p both in vitro and in vivo, where the cell lines with knockdown of SNHG6 could be more sensitive to 5-Fu, which improved 5-Fu therapy in mouse tumor model(Wang et al. 2019e). Knockdown of lncRNA NEAT1 also increased 5-Fu sensitivity by targeting miR-34a and consequently attenuating autophagy (Liu et al. 2020).
There are some other lncRNAs examples related to 5-Fu resistance. SCARNA2 expression increased in CRC tissue, which induced the resistance to 5-Fu by inhibiting miR-342-3p signaling pathway (Zhang et al. 2019b). MiR-204-mediated HMGA2/PI3K signaling pathway was inhibited by lncRNA PCAT6 to enhance the 5-Fu-based therapy, which was confirmed in CRC cells (Wu et al. 2019). Linc00467/miR-133b/ferritin light chain (FTL) formed an axis in the chemoresistance to 5-Fu in CRC, where linc00467 regulates FTL expression through miR-133b to promote metastasis and chemoresistance in CRC (Li et al. 2019c). HAND2-AS1/miR-20a/PDCD axis was also identified to inhibit 5-Fu resistance in CRC both in vitro and in vivo (Jiang et al. 2020). Furthermore, LINC00152 was implicated in conferring 5-Fu resistance (Bian et al. 2017; Chen et al. 2018a). Linc01296 upregulation advocated tumorigenesis and chemoresistance of CRC both in vitro and in vivo (Liu et al. 2018a). Chemoresistance response to 5-Fu was suppressed in GC by downregulating lncRNA FGD5-AS1, which showed significant antitumor effect on GC proliferation both in vitro and in vivo (Gao et al. 2020).
Chemoresistance to oxaliplatin (OXA)
OXA is a platinum compound which is often used to treat CRC and GC (Dy et al. 2009). Similar obstacle as 5-Fu, large proportion of patients turned into chemoresistant and metastatic (Goldberg et al. 2004). LncRNA MEG3 was downregulated in OXA resistant CRC cell lines, while overexpression of MEG3 could partially reverse the chemoresistance to OXA in CRC. Investigation also reported that MEG3 could improve OXA-induced apoptosis in CRC cells (Li et al. 2017c). The mechanism lies in that MEG3 as a ceRNA regulated OXA sensitivity by modulating miR-141/PDCD4 axis, where MEG3 bound and suppressed miR-141 directly through binding site. As the target of miR-141, PDCD4 contained a binding site of miR-141, and MEG3 could increase PDCD4 expression by binding miR-141 as ceRNA. MEG3 overcame OXA resistance by regulating miR-141/PDCD4 axis (Wang et al. 2018b).
MALAT1 is another lncRNA associated with OXA and overexpressed in CRC patients as an oncogene, linked to poor response to OXA treatment. The 3’ end region of MALAT1 interacted with EZH2 to inhibit E-cadherin expression. Such a negative regulation of E-cadherin expression in CRC indicates that MALAT1 is involved in OXA-induced EMT process (Sha et al. 2017).
As mentioned in 5-Fu chemoresistance, ABCC1 is also a critical factor in OXA resistance, which was positively regulated by lncRNA CACS15 via sponging miR-145. The silencing of CASC15 was proved to overcome OXA resistance of CRC in vivo (Gao et al. 2019). It is elucidated that LncRNA KCNQ1OT1 can promote the protective autophagy of CRC cells by increasing the expression of Atg4B via regulating miR-34a, so that the chemoresistance to OXA was enhanced in vitro and in vivo (Li et al. 2019b). Another lncRNA, linc00152, was mentioned above in 5-Fu chemoresistance, which also reported that functioned as ceRNA through sponging miR-193a-3p to confer OXA resistance in CRC both in vitro and in vivo (Yue et al. 2016).
LncRNA CCAL was associated with apoptosis and lower OXA chemoresistance in CRC cells, which could be a potential target to reverse the chemoresistance (Deng et al. 2020). LncRNA BLACAT1 was upregulated in OXA-resistant GC tissue and cells. Knockdown of BLACAT1 could inhibit ABCB1 expression and invasion in vitro and in vivo as well as OXA resistance with higher apoptosis (Wu et al. 2018).
Stemness is a significant factor in cancer stemness maintenance and chemoresistance. For example, lncRNA H19 not only overcame 5-Fu resistance, but also confer OXA resistance, in terms of carcinoma-associated fibroblast (CAF). OXA resistance in CRC was markedly promoted by overexpression of H19, while knocking down of H19 suppressed the tumor growth in xenograft model. The study indicated that CAF-derived exosomes increase the expression of H19, stemness and OXA resistance of CRC cells both in vitro and in vivo (Ren et al. 2018b). Other two lncRNAs, name as lnc273–31 and lnc273–34 were reported to be upregulated by p53-R273H then enhancing CRC stem cell stemness and chemoresistance of OXA both in vitro and in vivo (Zhao et al. 2019). The combination use of 5-Fu and OXA is also common seen in treatment. The mesenchymal stem cells (MSCs) play a critical role in tumor progression and anticancer drug resistance (Houthuijzen et al. 2012). LncRNA MACC1-AS1 was overexpressed in GC, and the study revealed that MSC boosted MACC1-AS1 expression which subsequently positively regulated fatty acid oxidation-dependent stemness and 5-Fu/OXA resistance were verified both in vitro and in vivo (He et al. 2019a).
Chemoresistance to cisplatin (DDP)
DDP-based chemotherapy is the backbone of GC treatment. Yet, cisplatin resistance may lead to tumor recurrence (Amable 2016). Body of evidence suggests that cancer-derived exosomes can advocate tumor progression and metastasis (Kahlert and Kalluri 2013), and the exosomes related to chemosensitive or resistant cells might influence the therapeutic response through transferring specific lncRNAs (Xu et al. 2016; Qu et al. 2016). In DDP-resistant GC cells, EMT and higher level of lncRNA HOTTIP were observed. Downregulation of HOTTIP could decrease cisplatin sensitivity. Exosomal HOTTIP activated HMGA1 to induce DDP-resistance in GC cells (Wang et al. 2019b).
Autophagy increased in DDP-resistant GC cells but could be suppressed by MALAT1 via binding with miR-30e to coordinate the expression of ATG5. Silencing of MALAT1 could prohibit chemo-induced autophagy, thus overcome chemoresistance in GC cell lines as well as in GC xenograft mice model (Zhang et al. 2020a). Another investigation stated that MALAT1 sequestered miR-30b from ATG5 to increase its expression and potentiated autophagy-related DDP resistance (Xi et al. 2019). When MALAT1 sequestered miR-23b-3p, then the expression of its target ATG12 increased, which contributed to autophagy-related chemoresistance to DDP and the drug-sensitivity assay were performed both in vitro and in vivo (YiRen et al. 2017). Previous studies found that MALAT1 was a promising target for DDP resistance in GC. Another lncRNA ARHGAP5-AS1 can also promote DDP resistance in GC by autophagy and adds more evidence that autophagy was a critical process in chemoresistance (Zhu et al. 2019a).
The mechanism underlying DDP resistance conferred by lncRNA HOXD-AS1 may be epigenetically silencing of PDCD4 via recruiting EZH2 in GC (Ye et al. 2019b). EZH2 also can be recruited by lncRNA PCAT-1 via epigenetically silencing of PTEN. Downregulation of PCAT-1 could promote sensitivity of DDP-resistant GC cells to DDP (Li et al. 2020a). Hence, EZH2-related lncRNA provided a novel therapeutic strategy targeting DDP chemoresistance in GC. DPP resistance is also conferred by PCAT-1 via another axis (i.e., miR-128/ZEB1). PCAT-1 acted as a sponge of miR-128 and the target was ZEB1. Knockdown of PCAT-1 could improve DDP sensitivity in GC tumors in vivo (Guo et al. 2019). The resistance of GC cells to DDP was promoted by FOXD1-AS1, so that depletion of FOXD1-AS1 reversed DDP resistance both in vitro and in vivo by targeting PI3K/AKT/mTOR pathway (Wu et al. 2020b).
DDP was also used in CRC treatment, even not commonly as 5-Fu and OXA, additional example about autophagy in chemoresistance is lncRNA SNHG14 in CRC, which stimulated CRC cell autophagy via miR-186/ATG14 axis (Han et al. 2020). There are some other lncRNAs examples related to DDP resistance. For example, silencing of lncRNA FGF9 could reverse DDP resistance via regulation of Wnt/β-catenin signaling pathway in CRC (Zhang et al. 2020b). Silencing of PVT1 could inhibit DDP resistance in CRC cells (Ping et al. 2018) and GC cells both in vitro and in vivo (Wu et al. 2020a). The overexpression of CASC2 could overcome DDP resistance in GC by binding to miR-19a (Li et al. 2018b). LncRNA DANCR could promoted DDP resistance through miR-125b-5p/HK2 axis both in vitro and in vivo (Shi et al. 2020).
Chemoresistance to gemcitabine
Gemcitabine-based chemotherapy is the first-line treatment for PC. Just as in CRC and GC, gemcitabine resistance has been major barrier in treating PC (Ju et al. 2015). LncRNA SLC7A11-AS1 was overexpressed in PC tissues and gemcitabine-resistant cell lines. Knockdown of SLC7A11-AS1 can boost pancreatic cancer cell sensitivity to gemcitabine. This implies that SLC7A11-AS1 is a promising target for stemming gemcitabine resistance in PC (Yang et al. 2020). The knockdown of lncRNA HOTTIP could promote the chemosensitivity of PC cells to gemcitabine by modulating HOXA13 both in vitro and in vivo was reported in previous paper (Li et al. 2015).
GSTM3TV2 is a lncRNA associated with higher chemoresistance to gemcitabine in pancreatic cancer in vitro and in vivo by acting as a ceRNA to sponge let-7 and regulate the expression of its direct targets LAT2 and OLR1 (Xiong et al. 2019). Linc-DYNC2H1-4 is upregulated in gemcitabine-resistant PC cells and knockdown of Linc-DYNC2H1-4 could suppress EMT via sponging miR-145, which targeted EMT markers (Gao et al. 2017b). SNHG14 is also a potential autophagy-related target in PC. It interreacted with miR-101 to stimulate autophagy and increase gemcitabine resistance (Zhang et al. 2019d). Downregulation of lncRNA HOST2 could improve the sensitivity to gemcitabine in PC, but the detailed mechanism of which is still unknown (An and Cheng 2020). Inhibition of lncRNA PVT1 led to higher efficacy of gemcitabine by targeting miR-1207 (You et al. 2018). The recent study indicated PVT1 could promote gemcitabine resistance of PC both in vitro and in vivo. The study demonstrated that PVT1 induced gemcitabine resistance was associated with elevated increased Wnt/β-catenin signaling pathway and autophagic activity. MiR-619-5p was directly targeted by PVT1, and the gemcitabine resistance was reversed by miR-619-5p in PC (Zhou et al. 2020). In addition, many other lncRNAs, such as TUG1, HOTAIR, GAS5, and HOTTIP, are also implicated in regulating gemcitabine resistance in PC (Yang et al. 2018a; Wang et al. 2017b; Gao et al. 2018; Li et al. 2015).
LncRNA AGAP2-AS1 worked as a ceRNA of miR-497 which targeted on fibroblast growth factor receptor 1. Gemcitabine resistance could be diminished by silencing AGAP2-AS1, which also cause G1/M phase cell cycle arrest in CRC cells (Hong et al. 2020).
Chemoresistance to other chemotherapy
Besides the commonly used drugs mentioned above, there are other chemotherapy drugs used in digestive system cancer treatment. Doxorubicin (DOX) is an anthracycline drug used to treat many malignancies and chemoresistance is the major treatment challenge. XIST is upregulated in CRC tissues and cells while knockdown of XIST could curb DOX resistance via interacting with miR-124, thereby positively regulate SGK1 expression in DOX-resistant CRC cells. The antitumor effect of DOX was improved further both in vitro and in vivo (Zhu et al. 2018a). lncRNAs D63785 and NEAT1 were also reported to regulate DOX resistance in GC (Zhou et al. 2018b; Zhang et al. 2018a).
Oxymatrine plays a role in anti-arrhythmia, antifibrosis, anti-inflammation, and antitumor in CRC and PC (Zhang and Huang 2004; Liang and Huang 2016; Chen et al. 2013). In oxymatrine-resistant CRC cells, lncRNA MALAT1 was upregulated, while knockdown of MALAT1 could partially reverse EMT. MALAT1 is a stimulator for oxymatrine resistance in CRC, which can inform better therapy treatment of CRC patients (Xiong et al. 2018). Carboplatin chemotherapy also face the challenge of chemoresistance. The expression of lncRNA BORG could enhance the viability of CRC cells by downregulating p53 so that downregulation of BORG could be a novel clue to overcoming the chemoresistance (Li et al. 2020b).
Some lncRNAs are implicated in multiple chemoresistance, such as XIST. It can modify the resistance to 5-FU, mitomycin, DDP, and DOX by collaborating with miR-30a in CRC cells (Zhang et al. 2019c). LncRNA GIHCG is associated with chemoresistance to 5-Fu and OXA and CRC progression (Jiang et al. 2019b). Apart from in 5-Fu chemoresistance, knockdown of CRNDE can increase sensitivity to chemotherapeutic drugs. This is also confirmed for OXA (Han et al. 2017). Similar sensitivity recovery was found in 5-Fu and OXA when ANRIL was knocked down (Zhang et al. 2018h). Overexpression of SLC25A25-AS1 not only increased the sensitivity to 5-Fu, but also to DOX in CRC cell line (Li et al. 2016). Knockdown of HULC contributed to the sensitivity of GC cells to DDP, DOX and 5-Fu (Zhang et al. 2016a). Knockdown of CASC9 significantly reduced the resistance to paclitaxel and DOX in GC cells (Shang et al. 2017).
Future perspectives
LncRNAs have attracted great attention in the past decade with increasing number of studies reporting on novel lncRNAs involved in various digestive system cancers. Due to its huge potential in modulating cancer development, it is very motivating to elucidate the lncRNAs regulatory mechanisms, especially those controlling the gene expression responsible for carcinogenesis or overcoming chemoresistance. It is also encouraging to note that several clinical trials involving lncRNAs and cancers, specifically thyroid cancer and breast cancer, have already completed their studies while others are still recruiting (https://clinicaltrials.gov/ct2/home). Although the results of these trials have not been published yet, their promising roles have been verified both in vitro and in vivo by numerous studies, and thus, signifies the potential of lncRNAs as therapeutic targets and/or biomarkers in cancer diagnosis and therapy.
Availability of data and material
Not applicable.
Code availability
Not applicable.
References
Amable L (2016) Cisplatin resistance and opportunities for precision medicine. Pharmacol Res 106:27–36. https://doi.org/10.1016/j.phrs.2016.01.001
An N, Cheng D (2020) The long noncoding RNA HOST2 promotes gemcitabine resistance in human pancreatic cancer cells. Pathol Oncol Res 26(1):425–431. https://doi.org/10.1007/s12253-018-0486-5
Barangi S, Hayes AW, Reiter R et al (2019) The therapeutic role of long non-coding RNAs in human diseases: a focus on the recent insights into autophagy. Pharmacol Res 142:22–29. https://doi.org/10.1016/j.phrs.2019.02.010
Beltran M, Puig I, Peña C et al (2008) A natural antisense transcript regulates Zeb2/Sip1 gene expression during Snail1-induced epithelial-mesenchymal transition. Genes Dev 22(6):756–769. https://doi.org/10.1101/gad.455708
Bian Z, Zhang J, Li M et al (2017) Long non-coding RNA LINC00152 promotes cell proliferation, metastasis, and confers 5-FU resistance in colorectal cancer by inhibiting miR-139-5p. Oncogenesis 6(11):395. https://doi.org/10.1038/s41389-017-0008-4
Brannan CI, Dees EC, Ingram RS et al (1990) The product of the H19 gene may function as an RNA. Mol Cell Biol 10(1):28–36. https://doi.org/10.1128/mcb.10.1.28
Brockdorff N, Ashworth A, Kay GF et al (1991) Conservation of position and exclusive expression of mouse Xist from the inactive X chromosome. Nature 351(6324):329–331. https://doi.org/10.1038/351329a0
Cao J, Zhang Y, Yang J et al (2016) NEAT1 regulates pancreatic cancer cell growth, invasion and migration though mircroRNA-335-5p/c-met axis. Am J Cancer Res 6(10):2361–2374
Chen LL, Carmichael GG (2010) Decoding the function of nuclear long non-coding RNAs. Curr Opin Cell Biol 22(3):357–364. https://doi.org/10.1016/j.ceb.2010.03.003
Chen H, Zhang J, Luo J et al (2013) Antiangiogenic effects of oxymatrine on pancreatic cancer by inhibition of the NF-kappaB-mediated VEGF signaling pathway. Oncol Rep 30(2):589–595. https://doi.org/10.3892/or.2013.2529
Chen DL, Chen LZ, Lu YX et al (2017a) Long noncoding RNA XIST expedites metastasis and modulates epithelial-mesenchymal transition in colorectal cancer. Cell Death Dis 8(8):e3011. https://doi.org/10.1038/cddis.2017.421
Chen DL, Lu YX, Zhang JX et al (2017b) Long non-coding RNA UICLM promotes colorectal cancer liver metastasis by acting as a ceRNA for microRNA-215 to regulate ZEB2 expression. Theranostics 7(19):4836–4849. https://doi.org/10.7150/thno.20942
Chen X, Jiang C, Qin B et al (2017c) LncRNA ZNF503-AS1 promotes RPE differentiation by downregulating ZNF503 expression. Cell Death Dis 8(9):e3046. https://doi.org/10.1038/cddis.2017.382
Chen Y, Yu X, Xu Y et al (2017d) Identification of dysregulated lncRNAs profiling and metastasis-associated lncRNAs in colorectal cancer by genome-wide analysis. Cancer Med 6(10):2321–2330. https://doi.org/10.1002/cam4.1168
Chen Z, Cai X, Chang L et al (2018a) LINC00152 is a potential biomarker involved in the modulation of biological characteristics of residual colorectal cancer cells following chemoradiotherapy. Oncol Lett 15(4):4177–4184. https://doi.org/10.3892/ol.2018.7833
Chen ZP, Wei JC, Wang Q et al (2018b) Long noncoding RNA 00152 functions as a competing endogenous RNA to regulate NRP1 expression by sponging with miRNA206 in colorectal cancer. Int J Oncol 53(3):1227–1236. https://doi.org/10.3892/ijo.2018.4451
Cheng K, Zhao Z, Wang G et al (2018) lncRNA GAS5 inhibits colorectal cancer cell proliferation via the miR1825p/FOXO3a axis. Oncol Rep 40(4):2371–2380. https://doi.org/10.3892/or.2018.6584
Chung LY, Tang SJ, Sun GH et al (2012) Galectin-1 promotes lung cancer progression and chemoresistance by upregulating p38 MAPK, ERK, and cyclooxygenase-2. Clin Cancer Res 18(15):4037–4047. https://doi.org/10.1158/1078-0432.Ccr-11-3348
Cui M, Chen M, Shen Z et al (2019) LncRNA-UCA1 modulates progression of colon cancer through regulating the miR-28-5p/HOXB3 axis. J Cell Biochem. https://doi.org/10.1002/jcb.27630
Deng X, Ruan H, Zhang X et al (2020) Long noncoding RNA CCAL transferred from fibroblasts by exosomes promotes chemoresistance of colorectal cancer cells. Int J Cancer 146(6):1700–1716. https://doi.org/10.1002/ijc.32608
Digklia A, Wagner AD (2016) Advanced gastric cancer: current treatment landscape and future perspectives. World J Gastroenterol 22(8):2403–2414. https://doi.org/10.3748/wjg.v22.i8.2403
Ding J, Li J, Wang H et al (2017) Long noncoding RNA CRNDE promotes colorectal cancer cell proliferation via epigenetically silencing DUSP5/CDKN1A expression. Cell Death Dis 8(8):e2997. https://doi.org/10.1038/cddis.2017.328
Dong H, Hu J, Zou K et al (2019a) Activation of LncRNA TINCR by H3K27 acetylation promotes Trastuzumab resistance and epithelial-mesenchymal transition by targeting MicroRNA-125b in breast Cancer. Mol Cancer 18(1):3. https://doi.org/10.1186/s12943-018-0931-9
Dong S, Zhang X, Liu D (2019b) Overexpression of long noncoding RNA GAS5 suppresses tumorigenesis and development of gastric cancer by sponging miR-106a-5p through the Akt/mTOR pathway. Biol Open. https://doi.org/10.1242/bio.041343
Du DX, Lian DB, Amin BH et al (2017a) Long non-coding RNA CRNDE is a novel tumor promoter by modulating PI3K/AKT signal pathways in human gastric cancer. Eur Rev Med Pharmacol Sci 21(23):5392–5398. https://doi.org/10.26355/eurrev_201712_13925
Du M, Yuan L, Tan X et al (2017b) The LPS-inducible lncRNA Mirt2 is a negative regulator of inflammation. Nat Commun 8(1):2049. https://doi.org/10.1038/s41467-017-02229-1
Dy GK, Hobday TJ, Nelson G et al (2009) Long-term survivors of metastatic colorectal cancer treated with systemic chemotherapy alone: a North Central Cancer Treatment Group review of 3811 patients, N0144. Clin Colorectal Cancer 8(2):88–93
Fan C, Tang Y, Wang J et al (2017) Role of long non-coding RNAs in glucose metabolism in cancer. Mol Cancer 16(1):130. https://doi.org/10.1186/s12943-017-0699-3
Ferre F, Colantoni A, Helmer-Citterich M (2016) Revealing protein-lncRNA interaction. Brief Bioinform 17(1):106–116. https://doi.org/10.1093/bib/bbv031
Gao H, Song X, Kang T et al (2017a) Long noncoding RNA CRNDE functions as a competing endogenous RNA to promote metastasis and oxaliplatin resistance by sponging miR-136 in colorectal cancer. Onco Targets Ther 10:205–216. https://doi.org/10.2147/ott.s116178
Gao Y, Zhang Z, Li K et al (2017b) Linc-DYNC2H1-4 promotes EMT and CSC phenotypes by acting as a sponge of miR-145 in pancreatic cancer cells. Cell Death Dis 8(7):e2924. https://doi.org/10.1038/cddis.2017.311
Gao ZQ, Wang JF, Chen DH et al (2017c) Long non-coding RNA GAS5 suppresses pancreatic cancer metastasis through modulating miR-32-5p/PTEN axis. Cell Biosci 7:66. https://doi.org/10.1186/s13578-017-0192-0
Gao ZQ, Wang JF, Chen DH et al (2018) Long non-coding RNA GAS5 antagonizes the chemoresistance of pancreatic cancer cells through down-regulation of miR-181c-5p. Biomed Pharmacother 97:809–817. https://doi.org/10.1016/j.biopha.2017.10.157
Gao R, Fang C, Xu J et al (2019) LncRNA CACS15 contributes to oxaliplatin resistance in colorectal cancer by positively regulating ABCC1 through sponging miR-145. Arch Biochem Biophys 663:183–191. https://doi.org/10.1016/j.abb.2019.01.005
Gao Y, Xie M, Guo Y et al (2020) Long non-coding RNA FGD5-AS1 regulates cancer cell proliferation and chemoresistance in gastric cancer through miR-153-3p/CITED2 axis. Front Genet 11:715. https://doi.org/10.3389/fgene.2020.00715
Goldberg RM, Sargent DJ, Morton RF et al (2004) A randomized controlled trial of fluorouracil plus leucovorin, irinotecan, and oxaliplatin combinations in patients with previously untreated metastatic colorectal cancer. J Clin Oncol 22(1):23–30. https://doi.org/10.1200/jco.2004.09.046
Gong P, Qiao F, Wu H et al (2018) LncRNA UCA1 promotes tumor metastasis by inducing miR-203/ZEB2 axis in gastric cancer. Cell Death Dis 9(12):1158. https://doi.org/10.1038/s41419-018-1170-0
Gong J, Lu X, Xu J et al (2019) Coexpression of UCA1 and ITGA2 in pancreatic cancer cells target the expression of miR-107 through focal adhesion pathway. J Cell Physiol 234(8):12884–12896. https://doi.org/10.1002/jcp.27953
Gottesman MM, Fojo T, Bates SE (2002) Multidrug resistance in cancer: role of ATP-dependent transporters. Nat Rev Cancer 2(1):48–58. https://doi.org/10.1038/nrc706
Gu L, Lu L, Zhou D et al (2018a) Long noncoding RNA BCYRN1 promotes the proliferation of colorectal cancer cells via up-regulating NPR3 expression. Cell Physiol Biochem 48(6):2337–2349. https://doi.org/10.1159/000492649
Gu L, Lu LS, Zhou DL et al (2018b) UCA1 promotes cell proliferation and invasion of gastric cancer by targeting CREB1 sponging to miR-590-3p. Cancer Med 7(4):1253–1263. https://doi.org/10.1002/cam4.1310
Guo Y, Yue P, Wang Y et al (2019) PCAT-1 contributes to cisplatin resistance in gastric cancer through miR-128/ZEB1 axis. Biomed Pharmacother 118:109255. https://doi.org/10.1016/j.biopha.2019.109255
Han Y, Ye J, Wu D et al (2014) LEIGC long non-coding RNA acts as a tumor suppressor in gastric carcinoma by inhibiting the epithelial-to-mesenchymal transition. BMC Cancer 14:932. https://doi.org/10.1186/1471-2407-14-932
Han P, Li JW, Zhang BM et al (2017) The lncRNA CRNDE promotes colorectal cancer cell proliferation and chemoresistance via miR-181a-5p-mediated regulation of Wnt/beta-catenin signaling. Mol Cancer 16(1):9. https://doi.org/10.1186/s12943-017-0583-1
Han Y, Zhou S, Wang X et al (2020) SNHG14 stimulates cell autophagy to facilitate cisplatin resistance of colorectal cancer by regulating miR-186/ATG14 axis. Biomed Pharmacother 121:109580. https://doi.org/10.1016/j.biopha.2019.109580
He W, Liang B, Wang C et al (2019a) MSC-regulated lncRNA MACC1-AS1 promotes stemness and chemoresistance through fatty acid oxidation in gastric cancer. Oncogene 38(23):4637–4654. https://doi.org/10.1038/s41388-019-0747-0
He Z, Dang J, Song A et al (2019b) NEAT1 promotes colon cancer progression through sponging miR-495-3p and activating CDK6 in vitro and in vivo. J Cell Physiol. https://doi.org/10.1002/jcp.28557
Herrera-Solorio AM, Armas-Lopez L, Arrieta O et al (2017) Histone code and long non-coding RNAs (lncRNAs) aberrations in lung cancer: implications in the therapy response. Clin Epigenetics 9:98. https://doi.org/10.1186/s13148-017-0398-3
Hong S, Yan Z, Song Y et al (2020) LncRNA AGAP2-AS1 augments cell viability and mobility, and confers gemcitabine resistance by inhibiting miR-497 in colorectal cancer. Aging (albany NY) 12(6):5183–5194. https://doi.org/10.18632/aging.102940
Horita K, Kurosaki H, Nakatake M et al (2019) Long noncoding RNA UCA1 enhances sensitivity to oncolytic vaccinia virus by sponging miR-18a/miR-182 and modulating the Cdc42/filopodia axis in colorectal cancer. Biochem Biophys Res Commun 516(3):831–838. https://doi.org/10.1016/j.bbrc.2019.06.125
Houthuijzen JM, Daenen LG, Roodhart JM et al (2012) The role of mesenchymal stem cells in anti-cancer drug resistance and tumour progression. Br J Cancer 106(12):1901–1906. https://doi.org/10.1038/bjc.2012.201
Hu CE, Du PZ, Zhang HD et al (2017) Long noncoding RNA CRNDE promotes proliferation of gastric cancer cells by targeting miR-145. Cell Physiol Biochem 42(1):13–21. https://doi.org/10.1159/000477107
Huang B, Liu C, Wu Q et al (2017) Long non-coding RNA NEAT1 facilitates pancreatic cancer progression through negative modulation of miR-506-3p. Biochem Biophys Res Commun 482(4):828–834. https://doi.org/10.1016/j.bbrc.2016.11.120
Huang F, Chen W, Peng J et al (2018a) LncRNA PVT1 triggers Cyto-protective autophagy and promotes pancreatic ductal adenocarcinoma development via the miR-20a-5p/ULK1 Axis. Mol Cancer 17(1):98. https://doi.org/10.1186/s12943-018-0845-6
Huang Y, Luo H, Li F et al (2018b) LINC00152 down-regulated miR-193a-3p to enhance MCL1 expression and promote gastric cancer cells proliferation. Biosci Rep. https://doi.org/10.1042/bsr20171607
Jiang H, Wang Y, Ai M et al (2017a) Long noncoding RNA CRNDE stabilized by hnRNPUL2 accelerates cell proliferation and migration in colorectal carcinoma via activating Ras/MAPK signaling pathways. Cell Death Dis 8(6):e2862. https://doi.org/10.1038/cddis.2017.258
Jiang Y, Feng E, Sun L et al (2017b) An increased expression of long non-coding RNA PANDAR promotes cell proliferation and inhibits cell apoptosis in pancreatic ductal adenocarcinoma. Biomed Pharmacother 95:685–691. https://doi.org/10.1016/j.biopha.2017.08.124
Jiang M, Qiu N, Xia H et al (2019a) Long non-coding RNA FOXD2-AS1/miR-150-5p/PFN2 axis regulates breast cancer malignancy and tumorigenesis. Int J Oncol 54(3):1043–1052. https://doi.org/10.3892/ijo.2019.4671
Jiang X, Li Q, Zhang S et al (2019b) Long noncoding RNA GIHCG induces cancer progression and chemoresistance and indicates poor prognosis in colorectal cancer. Onco Targets Ther 12:1059–1070. https://doi.org/10.2147/ott.S192290
Jiang Z, Li L, Hou Z et al (2020) LncRNA HAND2-AS1 inhibits 5-fluorouracil resistance by modulating miR-20a/PDCD4 axis in colorectal cancer. Cell Signal 66:109483. https://doi.org/10.1016/j.cellsig.2019.109483
Ju HQ, Gocho T, Aguilar M et al (2015) Mechanisms of overcoming intrinsic resistance to gemcitabine in pancreatic ductal adenocarcinoma through the redox modulation. Mol Cancer Ther 14(3):788–798. https://doi.org/10.1158/1535-7163.Mct-14-0420
Kahlert C, Kalluri R (2013) Exosomes in tumor microenvironment influence cancer progression and metastasis. J Mol Med (berl) 91(4):431–437. https://doi.org/10.1007/s00109-013-1020-6
Lee YS, Dutta A (2009) MicroRNAs in cancer. Annu Rev Pathol 4:199–227. https://doi.org/10.1146/annurev.pathol.4.110807.092222
Li Z, Zhao X, Zhou Y et al (2015) The long non-coding RNA HOTTIP promotes progression and gemcitabine resistance by regulating HOXA13 in pancreatic cancer. J Transl Med 13:84. https://doi.org/10.1186/s12967-015-0442-z
Li Y, Huang S, Li Y et al (2016) Decreased expression of LncRNA SLC25A25-AS1 promotes proliferation, chemoresistance, and EMT in colorectal cancer cells. Tumour Biol 37(10):14205–14215. https://doi.org/10.1007/s13277-016-5254-0
Li C, Liang G, Yang S et al (2017a) Dysregulated lncRNA-UCA1 contributes to the progression of gastric cancer through regulation of the PI3K-Akt-mTOR signaling pathway. Oncotarget 8(55):93476–93491. https://doi.org/10.18632/oncotarget.19281
Li J, Li X, Cen C et al (2017b) The long non-coding RNA ENST00000547547 reduces 5-fluorouracil resistance of colorectal cancer cells via competitive binding to microRNA-31. Oncol Rep 39(1):217
Li L, Shang J, Zhang Y et al (2017c) MEG3 is a prognostic factor for CRC and promotes chemosensitivity by enhancing oxaliplatin-induced cell apoptosis. Oncol Rep 38(3):1383–1392. https://doi.org/10.3892/or.2017.5828
Li L, Wang M, Mei Z et al (2017d) lncRNAs HIF1A-AS2 facilitates the up-regulation of HIF-1alpha by sponging to miR-153-3p, whereby promoting angiogenesis in HUVECs in hypoxia. Biomed Pharmacother 96:165–172. https://doi.org/10.1016/j.biopha.2017.09.113
Li P, Zhang X, Wang H et al (2017e) MALAT1 Is associated with poor response to oxaliplatin-based chemotherapy in colorectal cancer patients and promotes chemoresistance through EZH2. Mol Cancer Ther 16(4):739–751. https://doi.org/10.1158/1535-7163.mct-16-0591
Li T, Meng XL, Yang WQ (2017f) Long noncoding RNA PVT1 acts as a “Sponge” to inhibit microRNA-152 in gastric cancer cells. Dig Dis Sci 62(11):3021–3028. https://doi.org/10.1007/s10620-017-4508-z
Li X, Wang F, Sun Y et al (2017g) Expression of long non-coding RNA PANDAR and its prognostic value in colorectal cancer patients. Int J Biol Markers 32(2):e218–e223. https://doi.org/10.5301/jbm.5000249
Li Y, Gu J, Lu H (2017h) The GAS5/miR-222 axis regulates proliferation of gastric cancer cells through the PTEN/Akt/mTOR pathway. Dig Dis Sci 62(12):3426–3437. https://doi.org/10.1007/s10620-017-4831-4
Li Y, Li D, Zhao M et al (2018a) Long noncoding RNA SNHG6 regulates p21 expression via activation of the JNK pathway and regulation of EZH2 in gastric cancer cells. Life Sci 208:295–304. https://doi.org/10.1016/j.lfs.2018.07.032
Li Y, Lv S, Ning H et al (2018b) Down-regulation of CASC2 contributes to cisplatin resistance in gastric cancer by sponging miR-19a. Biomed Pharmacother 108:1775–1782. https://doi.org/10.1016/j.biopha.2018.09.181
Li M, Wang Q, Xue F et al (2019a) lncRNA-CYTOR works as an oncogene through the CYTOR/miR-3679-5p/MACC1 axis in colorectal cancer. DNA Cell Biol. https://doi.org/10.1089/dna.2018.4548
Li Y, Li C, Li D et al (2019b) lncRNA KCNQ1OT1 enhances the chemoresistance of oxaliplatin in colon cancer by targeting the miR-34a/ATG4B pathway. Onco Targets Ther 12:2649–2660. https://doi.org/10.2147/ott.S188054
Li Z, Liu J, Chen H et al (2019c) Ferritin light chain (FTL) competes with long noncoding RNA Linc00467 for miR-133b binding site to regulate chemoresistance and metastasis of colorectal cancer. Carcinogenesis. https://doi.org/10.1093/carcin/bgz181
Li H, Ma X, Yang D et al (2020a) PCAT-1 contributes to cisplatin resistance in gastric cancer through epigenetically silencing PTEN via recruiting EZH2. J Cell Biochem 121(2):1353–1361. https://doi.org/10.1002/jcb.29370
Li J, Ma J, Zhang X et al (2020b) Long non-coding RNA (lncRNA) BMP/OP-responsive gene (BORG) promotes development of chemoresistance of colorectal cancer cells to carboplatin. Med Sci Monit 26:e919103. https://doi.org/10.12659/msm.919103
Liang L, Huang J (2016) Oxymatrine inhibits epithelial–mesenchymal transition through regulation of NF-kappaB signaling in colorectal cancer cells. Oncol Rep 36(3):1333–1338. https://doi.org/10.3892/or.2016.4927
Lin C, Yang L (2018) Long noncoding RNA in cancer: wiring signaling circuitry. Trends Cell Biol 28(4):287–301. https://doi.org/10.1016/j.tcb.2017.11.008
Liu X, Jiao T, Wang Y et al (2016) Long non-coding RNA GAS5 acts as a molecular sponge to regulate miR-23a in gastric cancer. Minerva Med
Liu K, Hou Y, Liu Y et al (2017) LncRNA SNHG15 contributes to proliferation, invasion and autophagy in osteosarcoma cells by sponging miR-141. J Biomed Sci 24(1):46. https://doi.org/10.1186/s12929-017-0353-9
Liu B, Pan S, Xiao Y et al (2018a) LINC01296/miR-26a/GALNT3 axis contributes to colorectal cancer progression by regulating O-glycosylated MUC1 via PI3K/AKT pathway. J Exp Clin Cancer Res 37(1):316. https://doi.org/10.1186/s13046-018-0994-x
Liu B, Wu S, Ma J et al (2018b) lncRNA GAS5 reverses EMT and tumor stem cell-mediated gemcitabine resistance and metastasis by targeting miR-221/SOCS3 in pancreatic cancer. Mol Ther Nucleic Acids 13:472–482. https://doi.org/10.1016/j.omtn.2018.09.026
Liu J, Ben Q, Lu E et al (2018c) Long noncoding RNA PANDAR blocks CDKN1A gene transcription by competitive interaction with p53 protein in gastric cancer. Cell Death Dis 9(2):168. https://doi.org/10.1038/s41419-017-0246-6
Liu L, Meng T, Yang XH et al (2018d) Prognostic and predictive value of long non-coding RNA GAS5 and mircoRNA-221 in colorectal cancer and their effects on colorectal cancer cell proliferation, migration and invasion. Cancer Biomark 22(2):283–299. https://doi.org/10.3233/cbm-171011
Liu X, Cui L, Hua D (2018e) Long noncoding RNA XIST regulates miR-137-EZH2 axis to promote tumor metastasis in colorectal cancer. Oncol Res 27(1):99–106. https://doi.org/10.3727/096504018x15195193936573
Liu X, Yang B, Han J (2018f) Increased long noncoding RNA LINP1 expression and its prognostic significance in human breast cancer. Eur Rev Med Pharmacol Sci 22(24):8749–8754
Liu A, Liu L, Lu H (2019a) LncRNA XIST facilitates proliferation and epithelial–mesenchymal transition of colorectal cancer cells through targeting miR-486-5p and promoting neuropilin-2. J Cell Physiol. https://doi.org/10.1002/jcp.28054
Liu A, Liu L, Lu H (2019b) LncRNA XIST facilitates proliferation and epithelial–mesenchymal transition of colorectal cancer cells through targeting miR-486-5p and promoting neuropilin-2. J Cell Physiol 234(8):13747–13761. https://doi.org/10.1002/jcp.28054
Liu F, Ai FY, Zhang DC et al (2020) LncRNA NEAT1 knockdown attenuates autophagy to elevate 5-FU sensitivity in colorectal cancer via targeting miR-34a. Cancer Med 9(3):1079–1091. https://doi.org/10.1002/cam4.2746
Lu M, Liu Z, Li B et al (2017) The high expression of long non-coding RNA PANDAR indicates a poor prognosis for colorectal cancer and promotes metastasis by EMT pathway. J Cancer Res Clin Oncol 143(1):71–81. https://doi.org/10.1007/s00432-016-2252-y
Luo Y, Chen JJ, Lv Q et al (2019a) Long non-coding RNA NEAT1 promotes colorectal cancer progression by competitively binding miR-34a with SIRT1 and enhancing the Wnt/beta-catenin signaling pathway. Cancer Lett 440–441:11–22. https://doi.org/10.1016/j.canlet.2018.10.002
Luo Z, Yi ZJ, Ou ZL et al (2019b) RELA/NEAT1/miR-302a-3p/RELA feedback loop modulates pancreatic ductal adenocarcinoma cell proliferation and migration. J Cell Physiol 234(4):3583–3597. https://doi.org/10.1002/jcp.27039
Lv Y, Huang S (2019) Role of non-coding RNA in pancreatic cancer. Oncol Lett 18(4):3963–3973. https://doi.org/10.3892/ol.2019.10758
Ma P, Xu T, Huang M et al (2016) Increased expression of LncRNA PANDAR predicts a poor prognosis in gastric cancer. Biomed Pharmacother 78:172–176. https://doi.org/10.1016/j.biopha.2016.01.025
Ma L, Zhou Y, Luo X et al (2017) Long non-coding RNA XIST promotes cell growth and invasion through regulating miR-497/MACC1 axis in gastric cancer. Oncotarget 8(3):4125–4135. https://doi.org/10.18632/oncotarget.13670
Mao Y, Tie Y, Du J et al (2019) LINC00152 promotes the proliferation of gastric cancer cells by regulating B-cell lymphoma-2. J Cell Biochem 120(3):3747–3756. https://doi.org/10.1002/jcb.27655
Marquez-Jurado S, Diaz-Colunga J, das Neves RP et al (2018) Mitochondrial levels determine variability in cell death by modulating apoptotic gene expression. Nat Commun 9(1):389. https://doi.org/10.1038/s41467-017-02787-4
Martens JA, Laprade L, Winston F (2004) Intergenic transcription is required to repress the Saccharomyces cerevisiae SER3 gene. Nature 429(6991):571–574. https://doi.org/10.1038/nature02538
Meng S, Jian Z, Yan X et al (2019) LncRNA SNHG6 inhibits cell proliferation and metastasis by targeting ETS1 via the PI3K/AKT/mTOR pathway in colorectal cancer. Mol Med Rep. https://doi.org/10.3892/mmr.2019.10510
Mitra A, Mishra L, Li S (2015) EMT, CTCs and CSCs in tumor relapse and drug-resistance. Oncotarget 6(13):10697–10711
Ping G, Xiong W, Zhang L et al (2018) Silencing long noncoding RNA PVT1 inhibits tumorigenesis and cisplatin resistance of colorectal cancer. Am J Transl Res 10(1):138–149
Ponting CP, Oliver PL, Reik W (2009) Evolution and functions of long noncoding RNAs. Cell 136(4):629–641. https://doi.org/10.1016/j.cell.2009.02.006
Qu L, Ding J, Chen C et al (2016) Exosome-transmitted lncarsr promotes sunitinib resistance in renal cancer by acting as a competing endogenous RNA. Cancer Cell 29(5):653–668. https://doi.org/10.1016/j.ccell.2016.03.004
Quinn JJ, Chang HY (2016) Unique features of long non-coding RNA biogenesis and function. Nat Rev Genet 17(1):47–62. https://doi.org/10.1038/nrg.2015.10
Rebelo A, Molpeceres J, Rijo P et al (2017) Pancreatic cancer therapy review: from classic therapeutic agents to modern nanotechnologies. Curr Drug Metab 18(4):346–359. https://doi.org/10.2174/1389200218666170201151135
Ren H, Yang X, Yang Y et al (2018a) Upregulation of LncRNA BCYRN1 promotes tumor progression and enhances EpCAM expression in gastric carcinoma. Oncotarget 9(4):4851–4861. https://doi.org/10.18632/oncotarget.23585
Ren J, Ding L, Zhang D et al (2018b) Carcinoma-associated fibroblasts promote the stemness and chemoresistance of colorectal cancer by transferring exosomal lncRNA H19. Theranostics 8(14):3932–3948. https://doi.org/10.7150/thno.25541
Rivandi M, Pasdar A, Hamzezadeh L et al (2019) The prognostic and therapeutic values of long noncoding RNA PANDAR in colorectal cancer. J Cell Physiol 234(2):1230–1236. https://doi.org/10.1002/jcp.27136
Sha S, Yuan D, Liu Y et al (2017) Targeting long non-coding RNA DANCR inhibits triple negative breast cancer progression. Biol Open 6(9):1310–1316. https://doi.org/10.1242/bio.023135
Shan Y, Ying R, Jia Z et al (2017) LINC00052 promotes gastric cancer cell proliferation and metastasis via activating the Wnt/beta-catenin signaling pathway. Oncol Res 25(9):1589–1599. https://doi.org/10.3727/096504017x14897896412027
Shang C, Sun L, Zhang J et al (2017) Silence of cancer susceptibility candidate 9 inhibits gastric cancer and reverses chemoresistance. Oncotarget 8(9):15393–15398. https://doi.org/10.18632/oncotarget.14871
Shang AQ, Wang WW, Yang YB et al (2019) Knockdown of long non-coding RNA PVT1 suppresses cell proliferation and invasion of colorectal cancer via upregulation of microRNA-214-3p. Am J Physiol Gastrointest Liver Physiol. https://doi.org/10.1152/ajpgi.00357.2018
Shen J, Hong L, Yu D et al (2019) LncRNA XIST promotes pancreatic cancer migration, invasion and EMT by sponging miR-429 to modulate ZEB1 expression. Int J Biochem Cell Biol 113:17–26. https://doi.org/10.1016/j.biocel.2019.05.021
Shi H, Li K, Feng J et al (2020) LncRNA-DANCR interferes with miR-125b-5p/HK2 axis to desensitize colon cancer cells to cisplatin vis activating anaerobic glycolysis. Front Oncol 10:1034. https://doi.org/10.3389/fonc.2020.01034
Song C, Sun P, He Q et al (2019) Long non-coding RNA LINC01287 promotes breast cancer cells proliferation and metastasis by activating Wnt/ss-catenin signaling. Eur Rev Med Pharmacol Sci 23(10):4234–4242. https://doi.org/10.26355/eurrev_201905_17928
Sun J, Zhang Y (2019) LncRNA XIST enhanced TGF-beta2 expression by targeting miR-141-3p to promote pancreatic cancer cells invasion. Biosci Rep. https://doi.org/10.1042/bsr20190332
Sun K, Hu P, Xu F (2018a) LINC00152/miR-139-5p regulates gastric cancer cell aerobic glycolysis by targeting PRKAA1. Biomed Pharmacother 97:1296–1302. https://doi.org/10.1016/j.biopha.2017.11.015
Sun Z, Zhang B, Cui T (2018b) Long non-coding RNA XIST exerts oncogenic functions in pancreatic cancer via miR-34a-5p. Oncol Rep 39(4):1591–1600. https://doi.org/10.3892/or.2018.6245
Sun L, Liu L, Yang J et al (2019) SATB1 3’-UTR and lncRNA-UCA1 competitively bind to miR-495-3p and together regulate the proliferation and invasion of gastric cancer. J Cell Biochem 120(4):6671–6682. https://doi.org/10.1002/jcb.27963
Tan HY, Wang C, Liu G et al (2019) Long noncoding RNA NEAT1-modulated miR-506 regulates gastric cancer development through targeting STAT3. J Cell Biochem 120(4):4827–4836. https://doi.org/10.1002/jcb.26691
Tao Y, Han T, Zhang T et al (2017) LncRNA CHRF-induced miR-489 loss promotes metastasis of colorectal cancer via TWIST1/EMT signaling pathway. Oncotarget 8(22):36410–36422. https://doi.org/10.18632/oncotarget.16850
Verdecchia A, Francisci S, Brenner H et al (2007) Recent cancer survival in Europe: a 2000–02 period analysis of EUROCARE-4 data. Lancet Oncol 8(9):784–796. https://doi.org/10.1016/s1470-2045(07)70246-2
Wang W, Li Y, Zhang X et al (2014) Evaluating the significance of expression of CEA mRNA and levels of CEA and its related proteins in colorectal cancer patients. J Surg Oncol 109(5):440–444. https://doi.org/10.1002/jso.23503
Wang G, Pan J, Zhang L et al (2017a) Long non-coding RNA CRNDE sponges miR-384 to promote proliferation and metastasis of pancreatic cancer cells through upregulating IRS1. Cell Prolif. https://doi.org/10.1111/cpr.12389
Wang L, Dong P, Wang W et al (2017b) Gemcitabine treatment causes resistance and malignancy of pancreatic cancer stem-like cells via induction of lncRNA HOTAIR. Exp Ther Med 14(5):4773–4780. https://doi.org/10.3892/etm.2017.5151
Wang CL, Wang D, Yan BZ et al (2018a) Long non-coding RNA NEAT1 promotes viability and migration of gastric cancer cell lines through up-regulation of microRNA-17. Eur Rev Med Pharmacol Sci 22(13):4128–4137. https://doi.org/10.26355/eurrev_201807_15405
Wang H, Li H, Zhang L et al (2018b) Overexpression of MEG3 sensitizes colorectal cancer cells to oxaliplatin through regulation of miR-141/PDCD4 axis. Biomed Pharmacother 106:1607–1615. https://doi.org/10.1016/j.biopha.2018.07.131
Wang H, Zhang M, Sun G (2018c) Long non-coding RNA NEAT1 regulates the proliferation, migration and invasion of gastric cancer cells via targeting miR-335-5p/ROCK1 axis. Pharmazie 73(3):150–155. https://doi.org/10.1691/ph.2018.7877
Wang M, Han D, Yuan Z et al (2018d) Long non-coding RNA H19 confers 5-Fu resistance in colorectal cancer by promoting SIRT1-mediated autophagy. Cell Death Dis 9(12):1149. https://doi.org/10.1038/s41419-018-1187-4
Wang H, Chen W, Yang P et al (2019a) Knockdown of linc00152 inhibits the progression of gastric cancer by regulating microRNA-193b-3p/ETS1 axis. Cancer Biol Ther 20(4):461–473. https://doi.org/10.1080/15384047.2018.1529124
Wang J, Lv B, Su Y et al (2019b) Exosome-mediated transfer of lncRNA HOTTIP promotes cisplatin resistance in gastric cancer cells by regulating HMGA1/miR-218 axis. Onco Targets Ther 12:11325–11338. https://doi.org/10.2147/ott.S231846
Wang M, Hu H, Wang Y et al (2019c) Long non-coding RNA TUG1 mediates 5-fluorouracil resistance by acting as a ceRNA of miR-197-3p in colorectal cancer. J Cancer 10(19):4603–4613. https://doi.org/10.7150/jca.32065
Wang X, Lai Q, He J et al (2019d) LncRNA SNHG6 promotes proliferation, invasion and migration in colorectal cancer cells by activating TGF-beta/Smad signaling pathway via targeting UPF1 and inducing EMT via regulation of ZEB1. Int J Med Sci 16(1):51–59. https://doi.org/10.7150/ijms.27359
Wang X, Lan Z, He J et al (2019e) LncRNA SNHG6 promotes chemoresistance through ULK1-induced autophagy by sponging miR-26a-5p in colorectal cancer cells. Cancer Cell Int 19:234. https://doi.org/10.1186/s12935-019-0951-6
Wei W, Liu Y, Lu Y et al (2017) LncRNA XIST promotes pancreatic cancer proliferation through miR-133a/EGFR. J Cell Biochem 118(10):3349–3358. https://doi.org/10.1002/jcb.25988
Weidle UH, Birzele F, Kollmorgen G et al (2017) Long Non-coding RNAs and their role in metastasis. Cancer Genomics Proteom 14(3):143–160. https://doi.org/10.21873/cgp.20027
Wu X, Zheng Y, Han B et al (2018) Long noncoding RNA BLACAT1 modulates ABCB1 to promote oxaliplatin resistance of gastric cancer via sponging miR-361. Biomed Pharmacother Biomed Pharmacother 99:832–838. https://doi.org/10.1016/j.biopha.2018.01.130
Wu H, Zou Q, He H et al (2019) Long non-coding RNA PCAT6 targets miR-204 to modulate the chemoresistance of colorectal cancer cells to 5-fluorouracil-based treatment through HMGA2 signaling. Cancer Med 8(5):2484–2495. https://doi.org/10.1002/cam4.1809
Wu C, Hu Y, Ning Y et al (2020a) Long noncoding RNA plasmacytoma variant translocation 1 regulates cisplatin resistance via miR-3619-5p/TBL1XR1 axis in gastric cancer. Cancer Biother Radiopharm. https://doi.org/10.1089/cbr.2019.3342
Wu Q, Ma J, Wei J et al (2020b) FOXD1-AS1 regulates FOXD1 translation and promotes gastric cancer progression and chemoresistance by activating the PI3K/AKT/mTOR pathway. Mol Oncol. https://doi.org/10.1002/1878-0261.12728
Xi Z, Si J, Nan J (2019) LncRNA MALAT1 potentiates autophagyassociated cisplatin resistance by regulating the microRNA30b/autophagyrelated gene 5 axis in gastric cancer. Int J Oncol 54(1):239–248. https://doi.org/10.3892/ijo.2018.4609
Xiao J, Lv Y, Jin F et al (2017a) LncRNA HANR promotes tumorigenesis and increase of chemoresistance in hepatocellular carcinoma. Cell Physiol Biochem 43(5):1926–1938. https://doi.org/10.1159/000484116
Xiao Y, Yurievich UA, Yosypovych SV (2017b) Long noncoding RNA XIST is a prognostic factor in colorectal cancer and inhibits 5-fluorouracil-induced cell cytotoxicity through promoting thymidylate synthase expression. Oncotarget 8(47):83171–83182. https://doi.org/10.18632/oncotarget.20487
Xiao Z, Qu Z, Chen Z et al (2018) LncRNA HOTAIR is a prognostic biomarker for the proliferation and chemoresistance of colorectal cancer via MiR-203a-3p-mediated Wnt/ss-catenin signaling pathway. Cell Physiol Biochem 46(3):1275–1285. https://doi.org/10.1159/000489110
Xiong Y, Wang J, Zhu H et al (2018) Chronic oxymatrine treatment induces resistance and epithelialmesenchymal transition through targeting the long non-coding RNA MALAT1 in colorectal cancer cells. Oncol Rep 39(3):967–976. https://doi.org/10.3892/or.2018.6204
Xiong G, Liu C, Yang G et al (2019) Long noncoding RNA GSTM3TV2 upregulates LAT2 and OLR1 by competitively sponging let-7 to promote gemcitabine resistance in pancreatic cancer. J Hematol Oncol 12(1):97. https://doi.org/10.1186/s13045-019-0777-7
Xu CG, Yang MF, Ren YQ et al (2016) Exosomes mediated transfer of lncRNA UCA1 results in increased tamoxifen resistance in breast cancer cells. Eur Rev Med Pharmacol Sci 20(20):4362–4368
Xu TP, Wang WY, Ma P et al (2018) Upregulation of the long noncoding RNA FOXD2-AS1 promotes carcinogenesis by epigenetically silencing EphB3 through EZH2 and LSD1, and predicts poor prognosis in gastric cancer. Oncogene 37(36):5020–5036. https://doi.org/10.1038/s41388-018-0308-y
Xu M, Chen X, Lin K et al (2019) lncRNA SNHG6 regulates EZH2 expression by sponging miR-26a/b and miR-214 in colorectal cancer. J Hematol Oncol 12(1):3. https://doi.org/10.1186/s13045-018-0690-5
Yan K, Tian J, Shi W et al (2017) LncRNA SNHG6 is associated with poor prognosis of gastric cancer and promotes cell proliferation and EMT through epigenetically silencing p27 and sponging miR-101-3p. Cell Physiol Biochem 42(3):999–1012. https://doi.org/10.1159/000478682
Yang Z, Klionsky DJ (2010) Eaten alive: a history of macroautophagy. Nat Cell Biol 12(9):814–822. https://doi.org/10.1038/ncb0910-814
Yang X, Duan B, Zhou X (2017a) Long non-coding RNA FOXD2-AS1 functions as a tumor promoter in colorectal cancer by regulating EMT and Notch signaling pathway. Eur Rev Med Pharmacol Sci 21(16):3586–3591
Yang ZY, Yang F, Zhang YL et al (2017b) LncRNA-ANCR down-regulation suppresses invasion and migration of colorectal cancer cells by regulating EZH2 expression. Cancer Biomark 18(1):95–104. https://doi.org/10.3233/cbm-161715
Yang F, Li X, Zhang L et al (2018a) LncRNA TUG1 promoted viability and associated with gemcitabine resistant in pancreatic ductal adenocarcinoma. J Pharmacol Sci 137(2):116–121. https://doi.org/10.1016/j.jphs.2018.06.002
Yang Z, Shi X, Li C et al (2018b) Long non-coding RNA UCA1 upregulation promotes the migration of hypoxia-resistant gastric cancer cells through the miR-7-5p/EGFR axis. Exp Cell Res 368(2):194–201. https://doi.org/10.1016/j.yexcr.2018.04.030
Yang J, Du YM, Li B (2019a) LncRNA BX357664 inhibits the proliferation and invasion of non-small cell lung cancer cells. Eur Rev Med Pharmacol Sci 23(2):660–669. https://doi.org/10.26355/eurrev_201901_16880
Yang Z, Sun Y, Liu R et al (2019b) Plasma long noncoding RNAs PANDAR, FOXD2-AS1, and SMARCC2 as potential novel diagnostic biomarkers for gastric cancer. Cancer Manag Res 11:6175–6184. https://doi.org/10.2147/cmar.s201935
Yang Q, Li K, Huang X et al (2020) lncRNA SLC7A11-AS1 promotes chemoresistance by blocking SCF(β-TRCP)-mediated degradation of NRF2 in pancreatic cancer. Mol Ther Nucleic Acids 19:974–985. https://doi.org/10.1016/j.omtn.2019.11.035
Ye Y, Gu B, Wang Y et al (2019a) YY1-induced upregulation of long noncoding RNA ARAP1-AS1 promotes cell migration and invasion in colorectal cancer through the wnt/beta-catenin signaling pathway. Cancer Biother Radiopharm. https://doi.org/10.1089/cbr.2018.2745
Ye Y, Yang S, Han Y et al (2019b) HOXD-AS1 confers cisplatin resistance in gastric cancer through epigenetically silencing PDCD4 via recruiting EZH2. Open Biol 9(9):190068. https://doi.org/10.1098/rsob.190068
YiRen H, YingCong Y, Sunwu Y et al (2017) Long noncoding RNA MALAT1 regulates autophagy associated chemoresistance via miR-23b-3p sequestration in gastric cancer. Mol Cancer 16(1):174. https://doi.org/10.1186/s12943-017-0743-3
You L, Wang H, Yang G et al (2018) Gemcitabine exhibits a suppressive effect on pancreatic cancer cell growth by regulating processing of PVT1 to miR1207. Mol Oncol 12(12):2147–2164. https://doi.org/10.1002/1878-0261.12393
Yu B, Du Q, Li H et al (2017a) Diagnostic potential of serum exosomal colorectal neoplasia differentially expressed long non-coding RNA (CRNDE-p) and microRNA-217 expression in colorectal carcinoma. Oncotarget 8(48):83745–83753. https://doi.org/10.18632/oncotarget.19407
Yu T, Zhao Y, Hu Z et al (2017b) MetaLnc9 facilitates lung cancer metastasis via a PGK1-activated AKT/mTOR pathway. Cancer Res 77(21):5782–5794. https://doi.org/10.1158/0008-5472.can-17-0671
Yu C, Sun J, Leng X et al (2019a) Long noncoding RNA SNHG6 functions as a competing endogenous RNA by sponging miR-181a-5p to regulate E2F5 expression in colorectal cancer. Cancer Manag Res 11:611–624. https://doi.org/10.2147/cmar.s182719
Yu XH, Wang HF, Wu JB et al (2019b) Non-coding RNAs derailed: the many influences on the fatty acid reprogramming of cancer. Life Sci. https://doi.org/10.1016/j.lfs.2019.05.065
Yue B, Cai D, Liu C et al (2016) Linc00152 functions as a competing endogenous RNA to confer oxaliplatin resistance and holds prognostic values in colon cancer. Mol Ther 24(12):2064–2077. https://doi.org/10.1038/mt.2016.180
Yue B, Liu C, Sun H et al (2018) A positive feed-forward loop between LncRNA-CYTOR and Wnt/beta-catenin signaling promotes metastasis of colon cancer. Mol Ther 26(5):1287–1298. https://doi.org/10.1016/j.ymthe.2018.02.024
Zhang MJ, Huang J (2004) Recent research progress of anti-tumor mechnism matrine. Zhongguo Zhong Yao Za Zhi 29(2):115–118
Zhang Y, Song X, Wang X et al (2016a) Silencing of LncRNA HULC enhances chemotherapy induced apoptosis in human gastric cancer. J Med Biochem 35(2):137–143. https://doi.org/10.1515/jomb-2015-0016
Zhang YH, Fu J, Zhang ZJ et al (2016b) LncRNA-LINC00152 down-regulated by miR-376c-3p restricts viability and promotes apoptosis of colorectal cancer cells. Am J Transl Res 8(12):5286–5297
Zhang W, Shi S, Jiang J et al (2017a) LncRNA MEG3 inhibits cell epithelial-mesenchymal transition by sponging miR-421 targeting E-cadherin in breast cancer. Biomed Pharmacother 91:312–319. https://doi.org/10.1016/j.biopha.2017.04.085
Zhang W, Yuan W, Song J et al (2017b) LncRna CPS1-IT1 suppresses cell proliferation, invasion and metastasis in colorectal cancer. Cell Physiol Biochem 44(2):567–580. https://doi.org/10.1159/000485091
Zhang J, Zhao B, Chen X et al (2018a) Silence of long noncoding RNA NEAT1 inhibits malignant biological behaviors and chemotherapy resistance in gastric cancer. Pathol Oncol Res 24(1):109–113. https://doi.org/10.1007/s12253-017-0233-3
Zhang M, Zhao Y, Zhang Y et al (2018b) LncRNA UCA1 promotes migration and invasion in pancreatic cancer cells via the Hippo pathway. Biochim Biophys Acta Mol Basis Dis 1864(5 Pt A):1770–1782. https://doi.org/10.1016/j.bbadis.2018.03.005
Zhang Q, Chen B, Liu P et al (2018c) XIST promotes gastric cancer (GC) progression through TGF-beta1 via targeting miR-185. J Cell Biochem 119(3):2787–2796. https://doi.org/10.1002/jcb.26447
Zhang R, Li J, Yan X et al (2018d) Long noncoding RNA plasmacytoma variant translocation 1 (PVT1) promotes colon cancer progression via endogenous sponging miR-26b. Med Sci Monit 24:8685–8692. https://doi.org/10.12659/msm.910955
Zhang S, Zhang X, Sun Q et al (2018e) LncRNA NR2F2-AS1 promotes tumourigenesis through modulating BMI1 expression by targeting miR-320b in non-small cell lung cancer. J Cell Mol Med. https://doi.org/10.1111/jcmm.14102
Zhang X, Feng W, Zhang J et al (2018f) Long noncoding RNA PVT1 promotes epithelialmesenchymal transition via the TGFbeta/Smad pathway in pancreatic cancer cells. Oncol Rep 40(2):1093–1102. https://doi.org/10.3892/or.2018.6462
Zhang Y, Wu J, Jing H et al (2018g) Long noncoding RNA MEG3 inhibits breast cancer growth via upregulating endoplasmic reticulum stress and activating NF-κB and p53. J Cell Biochem. https://doi.org/10.1002/jcb.27982
Zhang Z, Feng L, Liu P et al (2018h) ANRIL promotes chemoresistance via disturbing expression of ABCC1 by regulating the expression of Let-7a in colorectal cancer. Biosci Rep. https://doi.org/10.1042/bsr20180620
Zhang M, Duan W, Sun W (2019a) LncRNA SNHG6 promotes the migration, invasion, and epithelial-mesenchymal transition of colorectal cancer cells by miR-26a/EZH2 axis. Onco Targets Ther 12:3349–3360. https://doi.org/10.2147/ott.s197433
Zhang PF, Wu J, Wu Y et al (2019b) The lncRNA SCARNA2 mediates colorectal cancer chemoresistance through a conserved microRNA-342-3p target sequence. J Cell Physiol 234(7):10157–10165. https://doi.org/10.1002/jcp.27684
Zhang R, Wang Z, Yu Q et al (2019c) Atractylenolide II reverses the influence of lncRNA XIST/miR-30a-5p/ROR1 axis on chemo-resistance of colorectal cancer cells. J Cell Mol Med 23(5):3151–3165. https://doi.org/10.1111/jcmm.14148
Zhang X, Zhao P, Wang C et al (2019d) SNHG14 enhances gemcitabine resistance by sponging miR-101 to stimulate cell autophagy in pancreatic cancer. Biochem Biophys Res Commun 510(4):508–514. https://doi.org/10.1016/j.bbrc.2019.01.109
Zhang Y, Yang H, Du Y et al (2019e) lncRNA TP53TG1 promotes pancreatic ductal adenocarcinoma development by acting as a molecular sponge of miR-96. Cancer Sci. https://doi.org/10.1111/cas.14136
Zhang YF, Li CS, Zhou Y et al (2020a) Propofol facilitates cisplatin sensitivity via lncRNA MALAT1/miR-30e/ATG5 axis through suppressing autophagy in gastric cancer. Life Sci 244:117280. https://doi.org/10.1016/j.lfs.2020.117280
Zhang Z, Zhang Y, Qin X et al (2020b) FGF9 promotes cisplatin resistance in colorectal cancer via regulation of Wnt/beta-catenin signaling pathway. Exp Ther Med 19(3):1711–1718. https://doi.org/10.3892/etm.2019.8399
Zhao J, Du P, Cui P et al (2018) LncRNA PVT1 promotes angiogenesis via activating the STAT3/VEGFA axis in gastric cancer. Oncogene 37(30):4094–4109. https://doi.org/10.1038/s41388-018-0250-z
Zhao Y, Li Y, Sheng J et al (2019) P53–R273H mutation enhances colorectal cancer stemness through regulating specific lncRNAs. J Exp Clin Cancer Res 38(1):379. https://doi.org/10.1186/s13046-019-1375-9
Zheng HC (2017) The molecular mechanisms of chemoresistance in cancers. Oncotarget 8(35):59950–59964. https://doi.org/10.18632/oncotarget.19048
Zhong F, Zhang W, Cao Y et al (2018) LncRNA NEAT1 promotes colorectal cancer cell proliferation and migration via regulating glial cell-derived neurotrophic factor by sponging miR-196a-5p. Acta Biochim Biophys Sin (shanghai) 50(12):1190–1199. https://doi.org/10.1093/abbs/gmy130
Zhou Y, Chen Y, Ding W et al (2018a) LncRNA UCA1 impacts cell proliferation, invasion, and migration of pancreatic cancer through regulating miR-96/FOXO3. IUBMB Life 70(4):276–290. https://doi.org/10.1002/iub.1699
Zhou Z, Lin Z, He Y et al (2018b) The long noncoding RNA D63785 regulates chemotherapy sensitivity in human gastric cancer by targeting miR-422a. Mol Ther Nucleic Acids 12:405–419. https://doi.org/10.1016/j.omtn.2018.05.024
Zhou P, Liu P, Zhang J (2019a) Long noncoding RNA RUSC1-AS-N promotes cell proliferation and metastasis through Wnt/β-catenin signaling in human breast cancer. Mol Med Rep 19(2):861–868. https://doi.org/10.3892/mmr.2018.9763
Zhou R, Wu Z, Deng X et al (2019b) The long non-coding RNA OLC8 enhances gastric cancer by interaction with IL-11. J Clin Lab Anal 33:e22962. https://doi.org/10.1002/jcla.22962
Zhou C, Yi C, Yi Y et al (2020) LncRNA PVT1 promotes gemcitabine resistance of pancreatic cancer via activating Wnt/β-catenin and autophagy pathway through modulating the miR-619-5p/Pygo2 and miR-619-5p/ATG14 axes. Mol Cancer 19(1):118. https://doi.org/10.1186/s12943-020-01237-y
Zhu J, Zhang R, Yang D et al (2018a) Knockdown of long non-coding RNA XIST inhibited doxorubicin resistance in colorectal cancer by upregulation of miR-124 and downregulation of SGK1. Cell Physiol Biochem 51(1):113–128. https://doi.org/10.1159/000495168
Zhu Y, Qiao L, Zhou Y et al (2018b) Long non-coding RNA FOXD2-AS1 contributes to colorectal cancer proliferation through its interaction with microRNA-185-5p. Cancer Sci 109(7):2235–2242. https://doi.org/10.1111/cas.13632
Zhu Y, Xing Y, Chi F et al (2018c) Long noncoding RNA SNHG6 promotes the progression of colorectal cancer through sponging miR-760 and activation of FOXC1. Onco Targets Ther 11:5743–5752. https://doi.org/10.2147/ott.s170246
Zhu L, Zhu Y, Han S et al (2019a) Impaired autophagic degradation of lncRNA ARHGAP5-AS1 promotes chemoresistance in gastric cancer. Cell Death Dis 10(6):383. https://doi.org/10.1038/s41419-019-1585-2
Zhu M, Wang X, Gu Y et al (2019b) MEG3 overexpression inhibits the tumorigenesis of breast cancer by downregulating miR-21 through the PI3K/Akt pathway. Arch Biochem Biophys 661:22–30. https://doi.org/10.1016/j.abb.2018.10.021
Zou L, Chen FR, Xia RP et al (2019) Long non-coding RNA XIST regulates the EGF receptor to promote TGF-beta1-induced epithelial-mesenchymal transition in pancreatic cancer. Biochem Cell Biol. https://doi.org/10.1139/bcb-2018-0274
Funding
This work was supported by Universiti Sains Malaysia Research University (RUI) Grant [1001.CIPPT.8012304].
Author information
Authors and Affiliations
Contributions
All authors have made significant and direct contribution to the work. WT and ISI provided the idea for the article and critically revised the work; MX mainly drafted this article; WF, SRAR, MAY made contribution on literature search.
Corresponding authors
Ethics declarations
Conflict of interest
The authors have no conflicts of interest to declare that are relevant to the content of this article.
Ethical approval
Not applicable.
Consent to participate
Not applicable.
Consent for publication
All authors have approved this manuscript for publication.
Rights and permissions
About this article
Cite this article
Miao, X., Wang, F., Wang, T. et al. Research updates on the clinical implication of long noncoding RNA in digestive system cancers and chemoresistance. 3 Biotech 11, 423 (2021). https://doi.org/10.1007/s13205-021-02971-w
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s13205-021-02971-w