Abstract
Neonatal exposure to general anesthetics has been associated with neurotoxicity and morphologic changes in the developing brain. Isoflurane is a volatile anesthetic widely used in pediatric patients to induce general anesthesia, analgesia, and perioperative sedation. In the present study, we investigated the effects of a single neonatal isoflurane (3% in oxygen, 2 h) exposure in rats at postnatal day (PND) 7, in short-term (24 h — PND8) and long-term (adulthood) protocols. In PND8, ex vivo analysis of hippocampal and frontal cortex slices evaluated cell viability and susceptibility to in vitro glutamate challenge. In adult rats, behavioral parameters related to anxiety-like behavior, short-term memory, and locomotor activity (PND60-62) and ex vivo analysis of cell viability, membrane permeability, glutamate uptake, and susceptibility to in vitro glutamate challenge in hippocampal and cortical slices from PND65. A single isoflurane (3%, 2 h) exposure at PND7 did not acutely alter cell viability in cortical and hippocampal slices of infant rats (PND8) per se and did not alter slice susceptibility to in vitro glutamate challenge. In rat’s adulthood, behavioral analysis revealed that the neonatal isoflurane exposure did not alter anxiety-like behavior and locomotor activity (open field and rotarod tests). However, isoflurane exposure impaired short-term memory evaluated in the novel object recognition task. Ex vivo analysis of brain slices showed isoflurane neonatal exposure selectively decreased cell viability and glutamate uptake in cortical slices, but it did not alter hippocampal slice viability or glutamate uptake (PND65). Isoflurane exposure did not alter in vitro glutamate-induced neurotoxicity to slices, and isoflurane exposure caused no significant long-term damage to cell membranes in hippocampal or cortical slices. These findings indicate that a single neonatal isoflurane exposure did not promote acute damage; however, it reduced cortical, but not hippocampal, slice viability and glutamate uptake in the adulthood. Additionally, behavioral analysis showed neonatal isoflurane exposure induces short-term recognition memory impairment, consolidating that neonatal exposure to volatile anesthetics may lead to behavioral impairment in the adulthood, although it may damage brain regions differentially.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Although general anesthesia is frequently required to facilitate medical and surgical pediatric procedures, acting as a powerful modulator of neuronal activity, there are ongoing concerns that anesthetic drugs may harm human neurodevelopment. Besides promoting neuronal apoptosis (Creeley et al. 2013), preclinical studies have shown that general anesthetics can also impair neuronal cytoarchitecture (Patel and Sun 2009). These anesthetics modulate cortical connectivity and may impair appropriate circuit assembly during critical periods of neurodevelopment by inducing spine growth, the formation of functional synapses (Roo et al. 2009), and increasing the dendritic spine density in young mice (Briner et al. 2010). One of the most commonly used inhalational anesthetics in the clinic is isoflurane.
Isoflurane (2-chloro-2-(difluoromethoxy)-1,1,1-trifluoroethane) is a volatile anesthetic that decreases excitatory neurotransmission by reducing the presynaptic Ca2+ influx in glutamatergic synapses (Baumgart et al. 2015). It was recently suggested that isoflurane blocks glutamatergic neurotransmission by a dual presynaptic mechanism (Wang et al. 2020). In fact, isoflurane reduced exocytosis by inhibiting Ca2+ currents evoked by a short presynaptic depolarization, while it inhibited exocytosis evoked by a prolonged depolarization via directly blocking exocytic machinery downstream of Ca2+ influx (Wang et al. 2020). Moreover, isoflurane acts on γ-aminobutyric acid type A receptors (GABAAR), increasing the affinity for GABA, thus extending the duration of GABA-mediated synaptic inhibition (Garcia et al. 2010).
An increasing number of studies demonstrated possible neurotoxicity promoted by isoflurane chronic and acute exposure in animal models of developing brain. In infant non-human primates, a repetitive exposure to isoflurane promoted long-term behavioral consequences, such as motor reflex deficits and increased anxiety (Coleman et al. 2017). It was recently shown that 5-h exposures of non-human primates to isoflurane during infancy are associated with decreased close social behavior after multiple exposures (three times), and anxiety-related behaviors after one exposure, but they do not affect the cognitive domains tested (Neudecker et al. 2021). Moreover, isoflurane exposure increased cortical neuroapoptosis in neonatal macaque brain (Brambrink et al. 2010; Noguchi et al. 2017).
A single exposure to isoflurane (3.5%) has been shown to influence neurogenesis, decreasing progenitor cell proliferation in the hippocampal dentate gyrus until 5 days after anesthesia, and also caused deficits in fear conditioning and spatial reference memory tasks in infant rats (Stratmann et al. 2009a). Additionally, 4 h of maternal anesthesia with isoflurane (1.4%) in rodents caused region-specific cell loss in the hippocampus of adult male offspring (Palanisamy et al. 2017). However, the effects of a single isoflurane exposure on neurodevelopment and long-term neurobehavioral aspects of neonates and infants need to be further investigated.
Therefore, this study aimed to evaluate the effects of a 2-h exposure to isoflurane (3%) on postnatal day 7 (PND7), in short term (24 h — PND8) and long term (in adulthood — PND65), by analyzing slices from two brain regions, the frontal cortex and the hippocampus. In PND8, cell viability and the susceptibility to in vitro challenge with glutamate were evaluated in slices of hippocampus and frontal cortex. In adult rats, neurobehavioral parameters related to anxiety-like behavior, short-term memory, and locomotor activity (PND60-62) were evaluated, and in the hippocampal and cortical slices obtained at PND65, cell viability, susceptibility to in vitro glutamate challenge, membrane permeability, and glutamate uptake were analyzed.
Material and Methods
Animals
All experimental procedures involving the animals were performed following National Institute of Health Guide for the Care and Use of Laboratory Animals (NIH Publications, 8th edition 2011) and were designed to minimize suffering and limit the number of animals used. The experiments were performed after approval of the protocol by the local Institutional Ethics Committee for Animal Research (CEUA/UFSC PP955). Dams were monitored, and the day of birth of pups was noted. Wistar rat pups (from the Federal University of Santa Catarina breeding colony) were randomly assigned to treatment groups and were maintained with their dams in sets of four females and four males at PND2. On PND7, rats were randomly assigned to saline or isoflurane groups. For all experimental interventions, care was taken to minimize the duration of maternal separation and handling of pups, and this was the same for both control and treatment groups. The animals were maintained in an air-conditioned room at 22 ± 2 ºC on a 12-h light/dark cycle (lights on at 7:00 a.m.). Rats were housed to a maximum of 6 in plastic cages with food and water ad libitum, with pups weaned into same-sex cages at PND21 and maintained until PND65.
Experimental Design
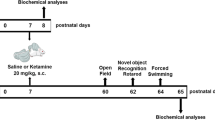
The experimental protocol conducted in the present study is summarized in Fig. 1. Rats were divided into two groups: control (room air) and isoflurane. Animals at PND7 were placed in induction acrylic chamber (Bonther, SP, Brazil) vaporized with 3% isoflurane in 30% oxygen/air or maintained at room air for 2 h, according to protocol previously reported by Stratmann et al. 2009a, b; Tsukamoto et al. 2017) with modification of the exposure time to 2 h. Animals were kept normothermic throughout experiments, closely monitored until they regained consciousness after anesthesia, and were returned to their dams in their home cages after the procedures. Two protocols were carried out independently: short- and long-term protocols. In the short-term protocol, the rats from one brooding were euthanized by decapitation 24 h after room air or isoflurane exposure (PND8; n = 4 animals per group). In the long-term protocol, rats from three broodings were also subjected to air or isoflurane exposure, maintained with their dams until weaning, and then separated at same-sex cages. Rats were subjected to behavioral tests from PND60 (n = 7–9 animals per group). At PND65, the animals were euthanized by decapitation. Hippocampi and frontal cortices were removed in both protocols, and the biochemical analyses were performed.
Schematic representation of experimental procedure protocols. Rats at PND7 were placed in induction chamber gassed with 3% isoflurane in 30% oxygen/air or to room air for 2 h. A Short-term protocol: Biochemical analyses in hippocampal and frontal cortical slices were performed 24 h after the isoflurane exposure (PND8). B Long-term protocol: Behavioral tests began at PND60. At PND65, biochemical assays were carried out in the hippocampal and frontal cortical slices
Biochemical Analyses
Preparation and Incubation of Hippocampal and Frontal Cortical Slices
Hippocampi and frontal cortices were placed in an ice-cold Krebs–Ringer bicarbonate buffer (KRB) containing, in mM: 122 NaCl, 3 KCl, 1.2 MgSO4, 1.3 CaCl2, 0.4 KH2PO4, 25 NaHCO3, and 10 d-glucose. The buffer was bubbled with 95% O2–5% CO2 up to pH 7.4 (Dal-Cim et al. 2013). Slices (0.4 mm) were prepared using a McIlwain Tissue Chopper, separated in KRB at 4 ºC, using a soft brush. Immediately after sectioning, slices were transferred to a 24-well plate and were maintained in KRB at 35 °C for 30 min to recover from slicing trauma (pre-incubation period).
Glutamate Toxicity
A previously established protocol of evaluation of glutamate toxicity in vitro was used (Ludka et al. 2017; Molz et al. 2011, 2009). After a pre-incubation period, slices were exposed to 10 mM glutamate (in KRB) for 1 h. The medium was then removed, and slices were maintained during 4 h in the nutritive incubation medium (NIM) composed of 50% KRB, 50% Dulbecco’s modified Eagle’s medium (DMEM, Gibco), and 20 mM HEPES, at 37 °C in a CO2 atmosphere. The slices corresponding to the control groups were incubated only in KRB solution for 1 h and then maintained in the NIM, to preserve slice viability in a medium similar to the physiological conditions.
Cellular Viability
The ability of cells to reduce 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) was used to determine cell viability as previously described (Mosmann 1983). Slices from the hippocampus and frontal cortex were incubated with MTT (0.5 mg/mL) in KRB for 30 min at 37 °C. The tetrazolium ring of MTT can be cleaved by active dehydrogenases to produce a precipitated formazan in which reduction can be used as an index of cellular viability. The medium was withdrawn, and precipitated formazan was solubilized with dimethyl sulfoxide (DMSO) and quantified spectrophotometrically at a wavelength of 540 nm.
PI Incorporation
Propidium iodide (PI) incorporation is a parameter to evaluate cellular membrane damage as indicative of cell death. Cell damage was assessed by evaluating the uptake of the fluorescent exclusion dye, PI, which is a polar compound that enters only cells with damaged membranes. Once inside the cells, PI complexes with DNA and emits an intense red fluorescence (630 nm) when excited by green light (495 nm). Hippocampal and frontal cortical slices were prepared and incubated with PI (7 μg/mL) for 30 min at 37 °C, and then washed with KRB for analysis on a fluorescence microplate reader (TECAN) (Piermartiri et al. 2009).
L-[3H]glutamate Uptake
L-[3H]glutamate uptake into hippocampal and frontal cortical slices was evaluated as previously described (Molz et al. 2009). Slices were prepared and incubated in KRB for 30 min at 37 ºC to normalize physiological conditions. Hippocampal and frontal cortical slices were then washed for 15 min at 37 ºC in Hank’s balanced salt solution (HBSS), composition in mM: 1.29 CaCl2, 136.9 NaCl, 5.36 KCl, 0.65 MgSO4, 0.27 Na2HPO4, 1.1 KH2PO4, and 5 HEPES. Uptake was assessed by adding 0.33 μCi/mL L-[3H]glutamate in the presence of 100 μM unlabeled glutamate in a final volume of 300 μL. Incubation was stopped immediately after 7 min by discarding the incubation medium, and slices were subjected to two ice-cold washes with 1 mL HBSS. Slices were solubilized by adding a solution with 0.1% NaOH/0.01% SDS and incubated overnight. Aliquots of slice lysates were taken for determination of the intracellular content of L-[3H]glutamate by scintillation counting. Sodium-independent uptake was determined by using choline chloride, instead of sodium chloride in the HBSS buffer. Unspecific sodium-independent uptake was subtracted from total uptake to obtain the specific sodium-dependent glutamate uptake. Results were obtained and presented as nmol of L-[3H]glutamate taken up per milligram of protein per minute.
Protein Measurement
Protein content was evaluated by the method of Lowry and collaborators (Lowry et al. 1951), using bovine serum albumin (Sigma-Aldrich Chemical Co, MO, USA) as standard.
Behavioral Tests
Behavioral tests were carried out from PND60 to PND62. All tests were performed between 9:00 a.m. and 02:00 p.m., and they were scored by the same rater in an observation sound-attenuated room under low-intensity light (12 lx), where the rats had been habituated for at least 1 h before the beginning of the tests. Behavior was monitored through a video camera positioned above the apparatuses, and the videos were later analyzed with the ANY Maze® video tracking system (Stoelting Co., Wood Dale, IL, USA). The apparatuses were cleaned with 10% ethanol between animals to avoid odor cues.
Open Field
Spontaneous locomotor activity was assessed in the open-field apparatus. Rats were placed in the center of a wooden arena (100 × 100 cm, grey walls, and grey floor) and allowed to freely explore it for 10 min. The total distance traveled, the number of crossings, and the average speed as indicators of spontaneous locomotor activity. The number of center visits and time spent in the center of the open field was used as a measure of anxiety-like behavior (Walsh and Cummins 1976).
Rotarod
The balance and motor coordination of rats were addressed in the accelerating rotarod apparatus (Insight Scientific Equipments, Ribeirão Preto, SP, Brazil). Rotarod apparatus consists of a grooved metal roller (6 cm in diameter) separated by 9-cm-wide compartments, elevated 16 cm. First, a habituation session was performed, in which each rat remained on the apparatus (absent rotation) for 30 s. Rats were allowed as many trials as necessary to reach this criterion. Next, a training session was performed, in which the animals had three trials to stay for 90 s on the rotating apparatus (5 rpm). Those animals that were able to perform for 90 s during the training session were chosen for the experiment. During the test session (performed 30 min after training), the starting speed was 5 rpm, and it was increased by 0.1 revolutions per second over a maximal period of 300 s, and the latency to fall (in seconds) from the accelerating rotarod was determined (Jiang et al. 2004).
Novel Object Recognition Task
The short-term recognition memory was addressed in a novel object recognition (NOR) task, performed as previously described (Abe et al. 2004). The task consists of three phases: habituation, training, and test phases. The habituation phase consisted of 2 days before testing, in which all rats were allowed to explore the open-field arena for 15 min once a day. The habituation phase aims to reduce stress, anxiety, and environmental exploration of animals on test day. The training and testing phases occurred 24 h after the last habituation day, for 3 min each, separated by an interval of 30 min to evaluate the short-term memory. The time spent by animals investigating each object in both phases was recorded. In the training phase, rats were exposed to two identical objects (A1 and A2) for 3 min. These objects were fixed in opposite corners 20 cm away from walls and 60 cm apart from each other. In the test phase, rats were exposed for 3 min to one of the familiar objects, and the other was replaced by a new object (B), which had a similar shape and size with a different color.
Statistical Analysis
The results are presented as the mean ± S.E.M. Comparisons between control and isoflurane groups were performed by the unpaired Student’s t test. Data from cell viability evaluation were analyzed by two-way ANOVA followed by post hoc of Newman-Keuls. Additionally, we used a t test to compare the percentage of recognition against a hypothetical value of 50% in the NOR task. Probability values less than 0.05 (P < 0.05) were considered statistically significant. Statistical analysis was performed using the GraphPad Prism 6.0 (GraphPad, San Diego, CA, USA) and Statistica 13.0 software (StatSoft Inc., La Jolla, CA, USA).
Results
Effects of a Single Neonatal Isoflurane Exposure on Cellular Viability Following In Vitro Glutamate Challenge in Hippocampal and Frontal Cortical Slices of Neonatal and Adult Rats
Neonatal rats (PND7) were subjected to isoflurane exposure (3%), and at PND8, their hippocampi and frontal cortices were isolated, sliced, and assessed for cellular viability. Moreover, the response of the brain slices to glutamate toxicity was also evaluated in this short-term protocol (Fig. 2A, B).
Cellular viability evaluation in hippocampal and frontal cortical slices from neonatal and adult rats subjected to a single isoflurane neonatal exposure. Cellular viability at PND8 of A hippocampal and B frontal cortical slices obtained from control (air) or isoflurane-exposed rats. C Hippocampal and D frontal cortical slices viability of rats at PND65. Data are presented as means ± S.E.M. of 4 animals per group. *P < 0.05 or **P < 0.01 represent means significantly different from air-control group (two-way ANOVA followed by Newman-Keuls post hoc test)
Twenty-four hours after air or isoflurane exposure, the hippocampal (Fig. 2A) and frontal cortical (Fig. 2B) slices of air-exposed neonatal rats (PND8) did not show a reduction in cellular viability caused by glutamate toxicity, as we previously showed (Sampaio et al. 2018). Similarly, the isoflurane exposure did not decrease the cellular viability in hippocampal and frontal cortical from both control and glutamate-challenged slices as well.
When performing the long-term protocol, hippocampal and frontal cortical slices obtained from adult rats (PND65) displayed a susceptibility to glutamate challenge (Fig. 2C, D). In hippocampal slices, in vitro glutamate challenge decreased cellular viability in both air- and isoflurane-exposed groups [P = 0.0134] (Fig. 2C). On the other hand, the neonatal isoflurane exposure did not alter the cellular viability in comparison to hippocampal slices from the air-exposed group in PND65. Furthermore, in frontal cortical slices, in vitro glutamate challenge also decreased cellular viability in both air-exposed and isoflurane-exposed [P = 0.0273] groups (Fig. 2D). Differently from hippocampal slices, neonatal isoflurane exposure per se decreased the cellular viability in frontal cortical slices from adult rats when compared to the air-exposed group [P = 0.0058], but it did not alter the excitotoxicity imposed by glutamate challenge (Fig. 2D).
Long-Term Effects of a Single Neonatal Isoflurane Exposure on PI Incorporation and Glutamate Uptake in Hippocampal and Frontal Cortical Slices of Adult Rats
The PI incorporation, which is a marker of damaged cellular membranes, was evaluated in hippocampal and frontal cortical slices obtained from adult rats (PND65), following isoflurane exposure. A single neonatal isoflurane exposure did not significantly change PI incorporation in both hippocampal (Fig. 3A) and frontal cortical (Fig. 3B) slices obtained from adult rats (PND65).
Evaluation of cellular membrane damage due to propidium iodide (PI) incorporation and glutamate uptake into hippocampal and frontal cortical slices from adult rats (PND65) subjected to a single isoflurane neonatal exposure. PI incorporation in A hippocampal and B frontal cortical slices of rats at PND65. Na+-dependent L-[.3H]glutamate uptake in the C hippocampus and D frontal cortex slices of rats at PND65. Data are presented as means ± S.E.M. of 5 animals per group. *P < 0.05 represents mean significantly different from air-control group (unpaired Student’s t test)
The L-[3H]glutamate uptake analysis was performed on hippocampal and frontal cortical slices obtained from adult rats (PND65) following a single neonatal isoflurane exposure. In the hippocampal slices from adult rats, no changes were observed in Na+-dependent L-[3H]glutamate uptake (Fig. 3C). However, in the frontal cortical slices obtained from adult rats following neonatal isoflurane exposure, L-[3H]glutamate uptake was shown to significantly decrease [P = 0.0420] (Fig. 3D).
Long-Term Neurobehavioral Effects of a Single Neonatal Isoflurane Exposure
To verify putative alterations on spontaneously locomotor, exploration activity, and anxiety-like behaviors of the adult rats (PND60) subjected to a neonatal isoflurane exposure, we carried out the open-field test (Fig. 4). No significant changes were observed in the following analyzed parameters: total distance traveled (Fig. 4A), mean speed (Fig. 4B), number of crossings (Fig. 4C), number of visits to the center (Fig. 4D), and time in the center (Fig. 4E).
Evaluation of long-term effects of a single isoflurane neonatal exposure on spontaneous locomotor activity and anxiety-like behaviors of adult rats (PND60) evaluated in the open-field test. The following parameters were analyzed: total distance traveled (A), mean speed (B), number of crossings (C), number of visits to the center (D), and time in the center (E). Data are presented as means ± S.E.M. of 8–9 animals per group (unpaired Student’s t test)
The rotarod test was performed to evaluate the impact of a neonatal exposure to isoflurane onto balance and motor coordination of adult rats (PND62). The latency to fall in air- or isoflurane-exposed group was not statistically different (Fig. 5).
Motor performance evaluation in the rotarod test in adult rats (PND62) subjected to a single-isoflurane neonatal exposure. Latency to fall of rod in the rotarod test of adult rats (PND62) subjected to isoflurane neonatal exposure. Data are presented as means ± S.E.M. of 8–9 animals per group (unpaired Student’s t test)
Regarding cognitive function, short-term recognition memory was evaluated by the NOR task in adult rats (PND62). As shown in Fig. 6A, the two identical objects (A1 and A2) were equally explored in the training phase. According to Student’s t test, the obtained data shows a significant difference between the percentage of novel object recognition control group and the hypothetical value of 50% in the test phase [t(8) = 2.499; P = 0.0370] (Fig. 6B). This result indicates that the animals from the control group spent more time exploring the novel object, which was not observed in the isoflurane-exposed group, demonstrating a short-term memory impairment induced by a single isoflurane neonatal exposure.
Short-term memory evaluation in the novel object recognition (NOR) task in adult rats (PND62) subjected to a single isoflurane neonatal exposure. The time of objects (A1 and A2) exploration in the training phase (A) and the percentage of the novel object recognition (discrimination index) in the test phase (B) are presented. Data are presented as means ± S.E.M. of 7 animals per group. *P < 0.05 as compared to a hypothetical value of 50% (unpaired Student’s t test)
Discussion
The first study to indicate a possible relationship between general anesthesia and neurodevelopment impairment of young children was published by Eckenhoff (Eckenhoff 1953). Since then, molecular methods have been applied to examine the underlying mechanisms involved in the potential neurotoxicity of volatile anesthetics in the mammalian brain (Hogan 2004; Futterer et al. 2004), and the number of experimental and clinical research concerning developmental anesthesia neurotoxicity has been growing (Davidson 2011; McCann and Soriano 2019). Isoflurane, a volatile anesthetic commonly used in pediatric and obstetric surgery, has been investigated under distinct protocols in preclinical studies, with different doses and duration of exposure pointing to neurodevelopmental toxicity (Stratmann et al. 2010).
By using a novel approach, the present study demonstrated that a single isoflurane (3%) exposure at PND7 did not acutely alter the cellular viability in cortical and in hippocampal slices of infant rats (PND8), nor did it alter resistance to glutamatergic toxicity in vitro at that age. However, a decreased cellular viability and glutamate uptake was observed in cortical slices only, but not in hippocampal slices from adult rats (PND65) subjected to neonatal isoflurane exposure. Additionally, these rats also demonstrated short-term memory impairment in the adulthood, as assessed by the novel object recognition (NOR) task.
Stratmann et al. (Stratmann et al. 2009a) showed different outcomes in PND7 rats that were exposed to 2 or 4 h of isoflurane (1 minimum alveolar concentration — 1 MAC). Although both interventions caused significant cell death 12 h after the exposure, only 4 h of isoflurane promoted a long-term neurocognitive deficit in adult rats (PND60), which was evident in spatial reference memory and spatial working memory tasks. In contrast, in our study, we were able to identify short-term memory impairment on adult rats exposed to only 2 h of isoflurane (3%), as assessed by the NOR task. Those different findings possibly resulted from the fact that distinct isoflurane concentrations were used, since 1 MAC of isoflurane in rats at postnatal maturation is around 1.15% (Frink et al. 1992; Ganzberg 2017), but it varies between PND7-9 (Orliaguet et al. 2001), and in the above study, it was assumed to be 3.5% for PND7 (Stratmann et al. 2009a). Additionally, one should stress that different types of memory were assessed between studies: object recognition memory vs. spatial memory. Interestingly, exposure of PND7 mice to 1.5% isoflurane for 4 h induced deficits in object recognition at 6 weeks (Schaefer et al. 2020). One may argue that the neurocognitive development impairments are dependent on isoflurane dose, exposure onset and time duration, analyzed species and type of assessed memory. In the present study, we used the NOR test because it holds some advantages over other rodent memory tests (Lueptow 2017). The main advantage is that it relies on rodents’ natural tendency to explore novelty and the conditions of the NOR task more closely resemble those used in studying human cognition, increasing the ecological validity of the test when compared to other rodent memory tests (Lueptow 2017).
Several studies have shown that isoflurane exposure affects cell viability in a dose- and time-dependent manner. For example, PC12 cells exposed to 2.4% isoflurane exhibited lower cell viability than that of cells exposed to 1.2% isoflurane. Prolonged exposure (6 h vs. 24 h) to 2.4% isoflurane resulted in a profound reduction in PC12 cell viability (Kim et al. 2016). In C57BL/6 mice exposed to 2 or 4 h of isoflurane (1.4%), caspase-3 cleavage was elevated 24 h post-anesthesia in the hippocampus. Moreover, 4 h of isoflurane exposure triggered long-term caspase-3 activation 2 weeks-post anesthesia (Liu et al. 2014). These findings showed that isoflurane induces apoptosis, which may contribute to long-term neurotoxicity.
The present findings showed a reduction in cellular viability in frontal cortical slices, but not in hippocampus slices, of adult rats subjected to isoflurane neonatal exposure. However, the neonatal isoflurane exposure did not acutely (PND8) alter frontal cortex or hippocampal viability. The lack of frontal cortical neurotoxicity in the early time point is consistent with results presented by Zou et al. (2008). These authors demonstrated that PND7 rat pups, which were exposed to a low dose of isoflurane (0.55%) for up to 8 h, did not show any significant neurotoxic effect in frontal cortex 6 h after completion of anesthetic administration. Moreover, these authors further stated that no significant cell damage was observed in other brain regions, including the hippocampus.
Our data in the adult rats (PND65) also indicate that hippocampus and frontal cortex were differentially affected by isoflurane exposure, which may be linked to a heterogeneous central nervous system development in different brain regions that changes their vulnerability to neurotoxic agents (Zhao et al. 2016). Although we did not see any changes in cellular viability and evidence of membrane damage in hippocampal slices, we cannot rule out that hippocampal cellular dysfunction is in place since this brain region is important for object recognition memory (Broadbent et al. 2010).
Besides evaluating isoflurane effects in adult rats, we also assessed a short-term effect 24 h after isoflurane exposure and a putative susceptibility to glutamate toxicity. As previously shown, slices obtained from both control and isoflurane exposed neonatal rats (PND8) are less susceptible to in vitro glutamate challenge than slices from adult brains, as indicated by lack of cellular metabolic activity reduction. This is due to the fact that glutamate receptors and transporter expression are developmentally regulated, and their immature expression and distribution in neonatal brain may avoid excitotoxic events, as previously shown by others and by us (Sampaio et al. 2018; Hestrin 1992; Dumas 2005; Yuan and Bellone 2013). Therefore, isoflurane did not alter cell viability per se, neither alter slice susceptibility to glutamate challenge at PND8. However, isoflurane exposure triggered a significant cellular viability decrease in cortical, but not hippocampal, slices obtained from adult rats (PND65), although cortical slice viability was not further changed by in vitro glutamate challenge. These observations suggest that metabolic perturbations in frontal cortex imposed by isoflurane exposure reached a stable nadir. Considering glutamate (in air-exposed rats) and isoflurane exposure per se induced cellular viability impairments which were not significantly different, it is feasible to suggest they may involve similar toxicity pathways, as caspase-3 activation induced by glutamate in slices, which we previously showed (Molz et al. 2008).
Albeit its main target is the GABA receptor, isoflurane can also exert effects on glutamatergic system including glutamate receptors (Zhang et al. 2008) and transporters (Qu et al. 2013). Importantly, the clearance of released glutamate occurs mainly by astrocytic glutamate transporters GLAST (or EAAT1) and GLT-1 (or EAAT2) (Robinson and Jackson 2016). These transporters can efficiently control the concentration of glutamate in the synaptic cleft, contributing to glutamate turnover to neurons and protecting from excitotoxicity. Isoflurane exposure (1.2% for 2 h) to old rats promoted spatial learning and memory impairment and displayed higher extracellular levels of glutamate and upregulation of EAAT1 on hippocampus (Qu et al. 2013). In another study, protein levels of NMDA receptor subunit GluN2A and EAAT1 were significantly increased in the hippocampus of rats following neonatal exposure to isoflurane (2% for 4 h; PND7) from 2 h to 3 days post-exposure, but they were not altered 7 and 28 days after exposure (Wang et al. 2019). Zuo (2001) demonstrated that isoflurane at clinically relevant concentrations (1–3%) caused a time-, sodium-, and concentration-dependent increase of glutamate uptake in primary cultures of rat cerebral mixed glial cells.
These data prompted us to evaluate the impact of isoflurane exposure on glutamate uptake into slices from frontal cortex and from hippocampus in adulthood. Neonatal isoflurane exposure did not alter hippocampal glutamate uptake in the adulthood. On the contrary, glutamate uptake was significantly decreased in cortical slices, at the same period where a decreased cellular viability was observed (PND65). Considering glutamate transporter activity may be sensitive to isoflurane, our data suggests that a reduction on cortical glutamate uptake may be contributing to increasing extracellular glutamate levels, and consequently glutamate excitotoxicity, thus promoting a reduced frontal cortical slice viability following a neonatal isoflurane exposure. However, to prove this hypothesis, studies evaluating whether neonatal isoflurane exposure may also alter glutamate transporter expression, or whether isoflurane interferes with the activity of glutamatergic neurons will be necessary to clarify the exact mechanism of action of isoflurane. Although, regarding glutamate transporters, it seems the neonatal alterations in protein levels induced by isoflurane are not observed for longer periods (Zhang et al. 2008). Then, functional data, as the evaluation of glutamate uptake, seems to be of more relevance for neurotoxicity assessment (Dal-Cim et al. 2019).
To further evaluate the long-term impact of neonatal isoflurane exposure on brain functional alterations, we also analyzed general locomotor activity (through of the total distance traveled, number of crossings, and average speed in the open field), anxiety-like behavior (number of center visits and time spent in the center of the open field), and motor balance in the rotarod test. Isoflurane neonatal exposure did not change any of the parameters evaluated. This is suggestive that isoflurane did not trigger obvious behavioral alterations beyond short-term memory impairment. This is consistent with data presented by Rosenholm et al. (Rosenholm et al. 2017), which showed negligible behavioral changes in mice subjected to repeated brief isoflurane anesthesia during early postnatal development. Furthermore, single neonatal exposure to isoflurane did not induce long-term motor behavioral alterations in rhesus macaques, but repeated isoflurane exposure had motor consequences (Coleman et al. 2017). Overall, these studies may suggest that longer and repeated exposures to isoflurane may be detrimental to cognitive as well as to locomotor and emotional behavior.
In summary, our findings indicate that a single neonatal isoflurane exposure did not alter short-term brain slice viability, but it induced a reduction in cellular viability and glutamate uptake observed in adulthood in the frontal cortex, but not in the hippocampus. Additionally, neonatal isoflurane exposure resulted in an impairment on the short-term recognition memory.
Altogether, our data consolidate previous observations demonstrating that exposure of developing brains to inhaled general anesthetics including isoflurane as well as sevoflurane may produce neurotoxicity leading to cognitive abnormalities later in life (Gupta and Datta 2020; Murphy and Baxter 2013; Shen et al. 2013). Since many healthy young children are exposed to general anesthetics, a careful assessment of the benefit/risk balance of the long-term consequences of exposure to anesthetics including isoflurane in neonates is still needed.
Disclaimer
The financial support agencies had no further role in study design; in the collection, analysis, and interpretation of data; in the writing of the report; and in the decision to submit the paper for publication.
Data Availability
All data generated or analysed during this study are included in this published article.
References
Abe H, Ishida Y, Iwasaki T (2004) Perirhinal N-methyl-D-aspartate and muscarinic systems participate in object recognition in rats. Neurosci Lett 356(3):191–194. https://doi.org/10.1016/j.neulet.2003.11.049
Baumgart JP, Zhou ZY, Hara M, Cook DC, Hoppa MB, Ryan TA, Hemmings HC Jr (2015) Isoflurane inhibits synaptic vesicle exocytosis through reduced Ca2+ influx, not Ca2+-exocytosis coupling. Proc Natl Acad Sci USA 112(38):11959–11964. https://doi.org/10.1073/pnas.1500525112
Brambrink AM, Evers AS, Avidan MS et al (2010) Isoflurane-induced neuroapoptosis in the neonatal rhesus macaque brain. Anesthesiology 112(4):834–841. https://doi.org/10.1097/ALN.0b013e3181d049cd
Briner A, De Roo M, Dayer A, Muller D, Habre W, Vutskits L (2010) Volatile anesthetics rapidly increase dendritic spine density in the rat medial prefrontal cortex during synaptogenesis. Anesthesiology 112(3):546–556. https://doi.org/10.1097/ALN.0b013e3181cd7942
Broadbent NJ, Gaskin S, Squire LR, Clark RE (2010) Object recognition memory and the rodent hippocampus. Learn Mem 17(1):5–11. https://doi.org/10.1101/lm.1650110
Coleman K, Robertson ND, Dissen GA et al (2017) Isoflurane anesthesia has long-term consequences on motor and behavioral development in infant rhesus macaques. Anesthesiology 126(1):74–84. https://doi.org/10.1097/ALN.0000000000001383
Creeley C, Dikranian K, Dissen G, Martin L, Olney J, Brambrink A (2013) Propofol-induced apoptosis of neurones and oligodendrocytes in fetal and neonatal rhesus macaque brain. Br J Anaesth 110(Suppl 1):i29-38. https://doi.org/10.1093/bja/aet173
Dal-Cim T, Ludka FK, Martins WC et al (2013) Guanosine controls inflammatory pathways to afford neuroprotection of hippocampal slices under oxygen and glucose deprivation conditions. J Neurochem 126(4):437–450. https://doi.org/10.1111/jnc.12324
Dal-Cim T, Poluceno GG, Lanznaster D, de Oliveira KA, Nedel CB, Tasca CI (2019) Guanosine prevents oxidative damage and glutamate uptake impairment induced by oxygen/glucose deprivation in cortical astrocyte cultures: involvement of A1 and A2A adenosine receptors and PI3K, MEK, and PKC pathways. Purinergic Signal 15(4):465–476. https://doi.org/10.1007/s11302-019-09679-w
Davidson AJ (2011) Anesthesia and neurotoxicity to the developing brain: the clinical relevance. Paediatr Anaesth 21(7):716–721. https://doi.org/10.1111/j.1460-9592.2010.03506.x
De Roo M, Klauser P, Briner A et al (2009) Anesthetics rapidly promote synaptogenesis during a critical period of brain development. PLoS ONE 4(9):e7043. https://doi.org/10.1371/journal.pone.0007043
Dumas TC (2005) Late postnatal maturation of excitatory synaptic transmission permits adult-like expression of hippocampal-dependent behaviors. Hippocampus 15(5):562–578. https://doi.org/10.1002/hipo.20077
Eckenhoff JE (1953) Relationship of anesthesia to postoperative personality changes in children. AMA Am J Dis Child 86(5):587–591. https://doi.org/10.1001/archpedi.1953.02050080600004
Frink EJ Jr, Malan TP, Atlas M, Dominguez LM, DiNardo JA, Brown BR Jr (1992) Clinical comparison of sevoflurane and isoflurane in healthy patients. Anesth Analg 74(2):241–245. https://doi.org/10.1213/00000539-199202000-00012
Futterer CD, Maurer MH, Schmitt A, Feldmann RE Jr, Kuschinsky W, Waschke KF (2004) Alterations in rat brain proteins after desflurane anesthesia. Anesthesiology 100(2):302–308. https://doi.org/10.1097/00000542-200402000-00019
Ganzberg S (2017) The FDA warning on anesthesia drugs. Anesth Prog 64(2):57–58. https://doi.org/10.2344/0003-3006-64.2.57
Garcia PS, Kolesky SE, Jenkins A (2010) General anesthetic actions on GABA(A) receptors. Curr Neuropharmacol 8(1):2–9. https://doi.org/10.2174/157015910790909502
Gupta A, Datta PK (2020) Sevoflurane consumption during inhalational induction in children: a randomized comparison of minute ventilation-based techniques with standard fixed fresh gas flow technique. AANA J 88(3):177–182
Hestrin S (1992) Developmental regulation of NMDA receptor-mediated synaptic currents at a central synapse. Nature 357(6380):686–689. https://doi.org/10.1038/357686a0
Hogan K (2004) Long-lasting changes in brain protein expression after exposure to an anesthetic. Anesthesiology 100(2):209–212. https://doi.org/10.1097/00000542-200402000-00003
Jiang C, Wan X, Jankovic J et al (2004) Dopaminergic properties and experimental anti-parkinsonian effects of IPX750 in rodent models of Parkinson disease. Clin Neuropharmacol 27(2):63–73. https://doi.org/10.1097/00002826-200403000-00004
Kim GH, Lee JJ, Lee SH, Chung YH, Cho HS, Kim JA, Kim MK (2016) Exposure of isoflurane-treated cells to hyperoxia decreases cell viability and activates the mitochondrial apoptotic pathway. Brain Res 1636:13–20. https://doi.org/10.1016/j.brainres.2016.01.052
Liu J, Wang P, Zhang X, Zhang W, Gu G (2014) Effects of different concentration and duration time of isoflurane on acute and long-term neurocognitive function of young adult C57BL/6 mouse. Int J Clin Exp Pathol 7(9):5828–5836
Lowry OH, Rosebrough NJ, Farr AL, Randall RJ (1951) Protein measurement with the Folin phenol reagent. J Biol Chem 193(1):265–275
Ludka FK, Dal-Cim T, Binder LB, Constantino LC, Massari C, Tasca CI (2017) Atorvastatin and fluoxetine prevent oxidative stress and mitochondrial dysfunction evoked by glutamate toxicity in hippocampal slices. Mol Neurobiol 54(5):3149–3161. https://doi.org/10.1007/s12035-016-9882-6
Lueptow LM (2017) Novel object recognition test for the investigation of learning and memory in mice. J Vis Exp (126). https://doi.org/10.3791/55718
McCann ME, Soriano SG (2019) Does general anesthesia affect neurodevelopment in infants and children? BMJ 367:l6459. https://doi.org/10.1136/bmj.l6459
Molz S, Decker H, Dal-Cim T, Cremonez C, Cordova FM, Leal RB, Tasca CI (2008) Glutamate-induced toxicity in hippocampal slices involves apoptotic features and p38 MAPK signaling. Neurochem Res 33(1):27–36. https://doi.org/10.1007/s11064-007-9402-1
Molz S, Dal-Cim T, Tasca CI (2009) Guanosine-5’-monophosphate induces cell death in rat hippocampal slices via ionotropic glutamate receptors activation and glutamate uptake inhibition. Neurochem Int 55(7):703–709. https://doi.org/10.1016/j.neuint.2009.06.015
Molz S, Dal-Cim T, Budni J et al (2011) Neuroprotective effect of guanosine against glutamate-induced cell death in rat hippocampal slices is mediated by the phosphatidylinositol-3 kinase/Akt/ glycogen synthase kinase 3beta pathway activation and inducible nitric oxide synthase inhibition. J Neurosci Res 89(9):1400–1408. https://doi.org/10.1002/jnr.22681
Mosmann T (1983) Rapid colorimetric assay for cellular growth and survival: application to proliferation and cytotoxicity assays. J Immunol Methods 65(1–2):55–63. https://doi.org/10.1016/0022-1759(83)90303-4
Murphy KL, Baxter MG (2013) Long-term effects of neonatal single or multiple isoflurane exposures on spatial memory in rats. Front Neurol 4:87. https://doi.org/10.3389/fneur.2013.00087
National Research Council (2011) Guide for the Care and Use of Laboratory Animals: Eighth Edition. Washington, DC: The National Academies Press. https://doi.org/10.17226/12910
Neudecker V, Perez-Zoghbi JF, Coleman K et al (2021) Infant isoflurane exposure affects social behaviours, but does not impair specific cognitive domains in juvenile non-human primates. Br J Anaesth 126(2):486–499. https://doi.org/10.1016/j.bja.2020.10.015
Noguchi KK, Johnson SA, Dissen GA et al (2017) Isoflurane exposure for three hours triggers apoptotic cell death in neonatal macaque brain. Br J Anaesth 119(3):524–531. https://doi.org/10.1093/bja/aex123
Orliaguet G, Vivien B, Langeron O, Bouhemad B, Coriat P, Riou B (2001) Minimum alveolar concentration of volatile anesthetics in rats during postnatal maturation. Anesthesiology 95(3):734–739. https://doi.org/10.1097/00000542-200109000-00028
Palanisamy A, Crosby G, Culley DJ (2017) Early gestational exposure to isoflurane causes persistent cell loss in the dentate gyrus of adult male rats. Behav Brain Funct 13(1):14. https://doi.org/10.1186/s12993-017-0132-5
Patel P, Sun L (2009) Update on neonatal anesthetic neurotoxicity: insight into molecular mechanisms and relevance to humans. Anesthesiology 110(4):703–708. https://doi.org/10.1097/ALN.0b013e31819c42a4
Piermartiri TC, Vandresen-Filho S, de Araujo HB et al (2009) Atorvastatin prevents hippocampal cell death due to quinolinic acid-induced seizures in mice by increasing Akt phosphorylation and glutamate uptake. Neurotox Res 16(2):106–115. https://doi.org/10.1007/s12640-009-9057-6
Qu X, Xu C, Wang H et al (2013) Hippocampal glutamate level and glutamate aspartate transporter (GLAST) are up-regulated in senior rat associated with isoflurane-induced spatial learning/memory impairment. Neurochem Res 38(1):59–73. https://doi.org/10.1007/s11064-012-0889-8
Robinson MB, Jackson JG (2016) Astroglial glutamate transporters coordinate excitatory signaling and brain energetics. Neurochem Int 98:56–71. https://doi.org/10.1016/j.neuint.2016.03.014
Rosenholm M, Paro E, Antila H, Voikar V, Rantamaki T (2017) Repeated brief isoflurane anesthesia during early postnatal development produces negligible changes on adult behavior in male mice. PLoS ONE 12(4):e0175258. https://doi.org/10.1371/journal.pone.0175258
Sampaio TB, de Oliveira LF, Constantino LC et al (2018) Long-term neurobehavioral consequences of a single ketamine neonatal exposure in rats: effects on cellular viability and glutamate transport in frontal cortex and hippocampus. Neurotox Res 34(3):649–659. https://doi.org/10.1007/s12640-018-9927-x
Schaefer ML, Perez PJ, Wang M et al (2020) Neonatal isoflurane anesthesia or disruption of postsynaptic density-95 protein interactions change dendritic spine densities and cognitive function in juvenile mice. Anesthesiology 133(4):812–823. https://doi.org/10.1097/ALN.0000000000003482
Shen X, Liu Y, Xu S, Zhao Q, Guo X, Shen R, Wang F (2013) Early life exposure to sevoflurane impairs adulthood spatial memory in the rat. Neurotoxicology 39:45–56. https://doi.org/10.1016/j.neuro.2013.08.007
Stratmann G, Sall JW, May LD et al (2009a) Isoflurane differentially affects neurogenesis and long-term neurocognitive function in 60-day-old and 7-day-old rats. Anesthesiology 110(4):834–848. https://doi.org/10.1097/ALN.0b013e31819c463d
Stratmann G, May LD, Sall JW et al (2009b) Effect of hypercarbia and isoflurane on brain cell death and neurocognitive dysfunction in 7-day-old rats. Anesthesiology 110(4):849–861. https://doi.org/10.1097/ALN.0b013e31819c7140
Stratmann G, Sall JW, May LD, Loepke AW, Lee MT (2010) Beyond anesthetic properties: the effects of isoflurane on brain cell death, neurogenesis, and long-term neurocognitive function. Anesth Analg 110(2):431–437. https://doi.org/10.1213/ANE.0b013e3181af8015
Tsukamoto A, Konishi Y, Kawakami T, Koibuchi C, Sato R, Kanai E, Inomata T (2017) Pharmacological properties of various anesthetic protocols in 10-day-old neonatal rats. Exp Anim 66(4):397–404. https://doi.org/10.1538/expanim.17-0037
Walsh RN, Cummins RA (1976) The open-field test: a critical review. Psychol Bull 83(3):482–504
Wang X, Shan Y, Tang Z, Gao L, Liu H (2019) Neuroprotective effects of dexmedetomidine against isoflurane-induced neuronal injury via glutamate regulation in neonatal rats. Drug Des Dev Ther 13:153–160
Wang HY, Eguchi K, Yamashita T, Takahashi T (2020) Frequency-dependent block of excitatory neurotransmission by isoflurane via dual presynaptic mechanisms. J Neurosci 40(21):4103–4115. https://doi.org/10.1523/JNEUROSCI.2946-19.2020
Yuan T, Bellone C (2013) Glutamatergic receptors at developing synapses: the role of GluN3A-containing NMDA receptors and GluA2-lacking AMPA receptors. Eur J Pharmacol 719(1–3):107–111. https://doi.org/10.1016/j.ejphar.2013.04.056
Zhang G, Dong Y, Zhang B et al (2008) Isoflurane-induced caspase-3 activation is dependent on cytosolic calcium and can be attenuated by memantine. J Neurosci 28(17):4551–4560. https://doi.org/10.1523/JNEUROSCI.5694-07.2008
Zhao T, Li C, Wei W, Zhang H, Ma D, Song X, Zhou L (2016) Prenatal ketamine exposure causes abnormal development of prefrontal cortex in rat. Sci Rep 6:26865. https://doi.org/10.1038/srep26865
Zou X, Sadovova N, Patterson TA et al (2008) The effects of L-carnitine on the combination of, inhalation anesthetic-induced developmental, neuronal apoptosis in the rat frontal cortex. Neuroscience 151(4):1053–1065. https://doi.org/10.1016/j.neuroscience.2007.12.013
Zuo Z (2001) Isoflurane enhances glutamate uptake via glutamate transporters in rat glial cells. NeuroReport 12(5):1077–1080. https://doi.org/10.1097/00001756-200104170-00042
Funding
This research was supported by grants from Brazilian funding agencies: Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq) — Instituto Nacional de Ciência e Tecnologia (INCT for Excitotoxicity and Neuroprotection); Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (CAPES/PVE 052/2012; CAPES-FCT 2014), Fundação de Apoio à Pesquisa do Estado de Santa Catarina (FAPESC)—Programa de Apoio aos Núcleos de Excelência (PRONEX — Project NENASC); and by Portuguese funding agencies: Fundação para a Ciência e Tecnologia (FCT, Portugal) — (Strategic Project 2015-UID/NEU/04539/2013); COMPETE-FEDER (POCI-01–0145-FEDER-007400); Centro 2020 Regional Operational Programmes (CENTRO-01–0145-FEDER-000012: HealthyAging2020 and CENTRO-01–0145-FEDER-000008: BrainHealth 2020). C.I.T. and R.D.P. are recipient of research fellowship from CNPq.
Author information
Authors and Affiliations
Contributions
Conceptualization and design of the study: C.I. Tasca, L.F. de Oliveira, R.D. Prediger and F.C. Pereira; material preparation and methodology: L.F. de Oliveira, G.G. Poluceno, T.B. Sampaio, L.C. Constantino, A.P. Costa, W.C. Martins, T. Dal-Cim, K.A. Oliveira, F.K. Ludka; formal analysis and investigation: L.F. de Oliveira, G.G. Poluceno, T.B. Sampaio, L.C. Constantino, A.P. Costa, W.C. Martins, T. Dal-Cim, K.A. Oliveira, F.K. Ludka; writing — original draft preparation: G.G. Poluceno and T.B. Sampaio; writing — review and editing: C.I. Tasca, L.C. Constantino, T. B. Sampaio and F.C. Pereira; funding acquisition: C.I. Tasca, R.D. Prediger and F.C. Pereira; Supervision: C.I. Tasca and F.C. Pereira.
Corresponding authors
Ethics declarations
Ethics Approval
Experiments followed the “Principles of Laboratory Animal Care” (NIH 2011) and were approved by the Committee on the Ethics of Animal Experiments of the Federal University of Santa Catarina (CEUA/UFSC PP955).
Conflict of Interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Ana Paula Costa´s current affiliation: Brain Health Imaging Institute, Department of Radiology, Weill Cornell Medicine, New York, NY, USA.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
de Oliveira, L.F., Poluceno, G.G., Sampaio, T.B. et al. Neonatal Isoflurane Exposure in Rats Impairs Short-Term Memory, Cell Viability, and Glutamate Uptake in Slices of the Frontal Cerebral Cortex, But Not the Hippocampus, in Adulthood. Neurotox Res 40, 1924–1936 (2022). https://doi.org/10.1007/s12640-022-00607-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12640-022-00607-2