Abstract
Sophora is deemed as one of the most remarkable genera of Fabaceae, and the third largest family of flowering plants. The genus Sophora comprises approximately 52 species, 19 varieties, and 7 forms that are widely distributed in Asia and mildly in Africa. Sophora species are recognized to be substantial sources of broad spectrum biopertinent secondary metabolites namely flavonoids, isoflavonoids, chalcones, chromones, pterocarpans, coumarins, benzofuran derivatives, sterols, saponins (mainly triterpene glycosides), oligostilbenes, and mainly alkaloids. Meanwhile, extracts and isolated compounds from Sophora have been identified to possess several health-promising effects including anti-inflammatory, anti-arthritic, antiplatelets, antipyretic, anticancer, antiviral, antimicrobial, antioxidant, anti-osteoporosis, anti-ulcerative colitis, antidiabetic, anti-obesity, antidiarrheal, and insecticidal activities. Herein, the present review aims to provide comprehensive details about the phytochemicals and biological effects of Sophora species. The review spotlighted on the promising phytonutrients extracted from Sophora and their plethora of bioactivities. The review also clarifies the remaining gaps and thus qualifies and supplies a platform for further investigations of these compounds.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Medicinal plants have been identified and utilized throughout human history. Plants could synthesize a wide variety of bioactive compounds that have several biological functions (Sweelam et al. 2018; Boozari et al. 2019b; Elberry et al. 2020). Fabaceae (Leguminosae, the legume family) is the third largest family of flowering plants (Borges et al. 2013). Fabaceae includes about 20,000 species within 750 genera and it includes herbal trees, herbs, and shrubs distributed worldwide (Simpson 2010). One of the most important genera of this family is Sophora which contains ~ 52 species, 19 varieties, and 7 forms that are widely distributed in Asia and mildly spread in Africa (Simpson 2010; Borges et al. 2013).
More than 15 species of this genus have been historically used in the traditional medicines, shifting the scientific community attention on their bioactive components with multi pharmacological and/or therapeutic benefits (Aly et al. 2019; Boozari et al. 2019b; Cai et al. 2020; Wang et al. 2020a, b, c). Despite the importance of genus Sophora, only few species have been chemically evaluated as previously covered in several reviews (Aly et al. 2019; Boozari et al. 2019b; Elberry et al. 2020). Herein, we illustrated the structure of > 360 isolated compounds from different Sophora species, including alkaloids, chalcones, chromones, pterocarpans, coumarins, benzofuran derivatives, sterols, isoflavonoids, flavonols, flavones, saponins, and stilbene oligomers (Cao and He 2020; Jin et al. 2020; Zhang et al. 2020a). These phytonutrients showed numerous pharmacological activities including anti-inflammatory, anti-arthritic, antiplatelets, antipyretic, anticancer, antiviral, antimicrobial, antioxidant, anti-osteoporosis, anti-ulcerative colitis, antidiabetic, anti-obesity, antidiarrheal, insecticidal, and hair growth promoting, besides their uses in the treatment of skin diseases i.e. eczema, colitis, and psoriasis (Huang et al. 2018a, b; Aly et al. 2019; Boozari et al. 2019b; Wang et al. 2019a, b, 2020a, b, c; Guo et al. 2020; Mao et al. 2020; Li et al. 2021b; Ma et al. 2021).
The review by Krishna et al. (2012) summarized the literature to document the ethnomedical and secondary metabolites that characterize and differentiate between Sophora species. Further, Aly et al. have surveyed the findings during 8 years (2011–2019) on the bioactivities of Sophora species, with particular focus on their anticancer and anti-inflammatory properties. Wang and co-authors have also assessed the reported biological activities of important class of compounds, quinolizidine alkaloids of Sophora species (Wang et al. 2019a), while Boozari et al. has a particular focus on active prenylated flavonoids and their structure–activity relationship (Boozari et al. 2019b). The purpose of the present critical study is to highlight the recent advances in current knowledge on Sophora species. However, the aim is not only to represent the recorded data, but also to address all the defects and gaps required for further future investigation. In the current review, figures are used as the simplest way to simplify the hugely recorded data of Sophora species into informative points. We employed those figures to elaborate and suggest the future scientists’ role for a better prospective investigation regarding Sophora genus. We do believe that this kind of ordered knowledge will guide the researchers to extend their research and explore the Sophora for drug development.
Phytochemistry
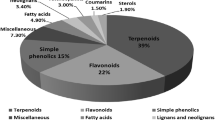
Sophora is a crucial genus, because of the broad variation in its chemical composition. This genus was reported to be rich in polyphenols such as flavanols, flavones, and isoflavonoids along with pterocarpans, chalcones, chromones, and benzofuran derivatives (Tables 1, 2, 3, 4, 5, 6). As tabulated in Tables 7, 8, 9, 10, 11 and 12, stilbenes, oligomers, sterols, triterpenes, coumarins, alkaloids, phenolic acids, and other phenolated compounds were identified from different Sophora species. Other constituents include hydrocarbons and fatty acids methyl esters identified by gas chromatography/mass spectrometry (GC/MS) from Sophora species was also demonstrated in Table 13.
Traditional uses of main Sophora species
Traditionally Sophora species were widely utilized in the treatment of many diseases and ailments (Ahn 1998). Sophora plants, such as the roots of S. flavescens, the roots of S. tonkinensis, and the seeds of S. alopecuroides were commonly used in traditional Chinese medicines for the treatment of fever, bacterial infections, heart disease, rheumatism, eczema, colitis, acute pharyngolaryngeal infection, sore throat, acute dysentery and gastrointestinal hemorrhage, and other gastrointestinal diseases (Fang et al. 2018; Fan et al. 2019; Gu et al. 2020; Jin et al. 2020; Li et al. 2020d; Wang et al. 2020b; Zhang et al. 2020a; Ti et al. 2021). The main species of Sophora that are commonly used in traditional medicine were the following:
S. flavescens
The dried roots of S. flavescens Aiton are traditionally used as an antipyretic medicine to reduce the inflammation (Aly et al. 2019; Wang et al. 2019c). The herb is also used for the treatment of skin and mucosal ulcers, sores, diarrhea, gastrointestinal hemorrhage, arrhythmia, and eczema (Aly et al. 2019; Boozari et al. 2019b). The root of S. flavescens has been extensively used in the clinic to treat cancer, hematochezia, dysentery, jaundice, pruritus vulvae, hepatitis, eczema, and skin diseases in traditional Chinese medicine (Yang et al. 2021). Traditionally, S. flavescens was used for asthma, sores, gastrointestinal hemorrhage and allergy, inflammation, anti-ulcerative, and treating of diarrhea and eczema (Ahn 1998; Chen et al. 2020b).
S. japonica and S. viciifolia
Recently the flower buds of S. japonica have been used as cosmetic whitening agent (Kim et al. 2021). The flower buds of S. japonica, which are a major traditional medicine in many Asian countries, are used to stop bleeding (Kim et al. 2021), and therefore, they are used to treat bleeding haemorrhoids, hypertension, and pyoderma. Meanwhile, S. japonica L. was used as a haemostatic agent to treat hemorrhoids and hematemesis (Aly et al. 2019; Elberry et al. 2020), where S. viciifolia roots were utilized as a Chinese drug to treat fever, cystitis, haematuria, and edema (Ao et al. 2019). The S. davidii plant is synonymous with, and formerly known as, S. viciifolia. The roots of S. davidii have been traditionally used to clear heat, sooth a sore throat, blood cooling, swelling reducing, and treat each of hematochezia, cough, and dysentery, etc. (Li et al. 2021b).
S. alopecuroides
It is benefited for its anti-dysentery effect, a property that is commonly observed in other Sophora genus phytomedicines (Zhou et al. 2020).
S. tonkinensis
In traditional Chinese medicine theory, the root and rhizome are toxic and mainly used in the treatment of pharyngeal and laryngeal diseases (Zhang et al. 2021b).
S. subprostrata and S. mollis
Another example about the roots of S. subprostrata and S. mollis, which were used as an antipyretic, analgesic with a potential anti-tumor activity (Ao et al. 2019; Wang et al. 2019a; Zhang et al. 2020a; Quradha et al. 2021). These roots are also used as a Korean traditional medicine for the treatment of fever, inflammation, peptic ulcer, and cancer (Aly et al. 2019, 2020a; Boozari et al. 2019b; Chen et al. 2020b; Wang et al. 2020b; Yang et al. 2020b). Several Sophora species including S. subprostrata displayed various activities in dispelling dampness, clearing heat, sedation, detoxification, and relieving swelling and pain (Zhou et al. 2020).
S. exigua
Sophora exigua Craib is commonly used in Thailand to increase postpartum breast milk production in women who have hypogalactia and reduce fever (Kaewdana et al. 2021).
Biological effects
Active molecules from the genus Sophora
Worldwide, about 4 billion people today rely on plants as sources of drugs even in the developed countries (Sweelam et al. 2018; Aly et al. 2019; Boozari et al. 2019b). Freshly, medicinal plants gained the attention in term of identifying and exploiting safe and effective remedies for treatment of several chronic and infective diseases (Boozari et al. 2019a, b; Wang et al. 2020a ,b; Zhou et al. 2020). The phytoextracts of Sophora species are rich sources of bioactive phytochemicals, such as prenylated flavonoids, quinolizidine-type alkaloid, terpenes, etc. with health-promoting properties (Tables 14, 15). Biological activities of many Sophora species bioactive compounds are depending on the structure of those constituents.
Cytotoxic and anti-tumor activity
Prenylated flavonoids from Sophora species exert dependently inhibited the Alzheimer’s disease, primarily through their skeleton’s structure (chalcones, flavonols, flavanones), and their lipophilic chain length (lavandulyl and prenyl groups) (Aly et al. 2019; Boozari et al. 2019b). Due to the promising and diverse bioactivities on multitarget tissues of prenylated flavanones, research on such components were recently spotlighted (Awad et al. 2014, 2018; Boozari et al. 2019b; Wang et al. 2020b). For example, sophoraflavanone G (19; from S. flavescens) has revealed cytotoxicity for several tumor cells with IC50-values of 20 mM, which was like cisplatin, commonly used as a recent chemotherapy drug to treat different cancers (Long et al. 2020). It also has reported to induce apoptosis in triple-negative breast cancer cells (Huang et al. 2019, 2020). Kurarinone (23) induced apoptosis in small cell lung carcinoma (SCLC) cells via multiple mechanisms and delayed SCLC-cell’s migration and invasion (Chung et al. 2019). By its underlying mechanism, Kurarinone promoted Fas and TRAIL receptor-1 and -2 expression via the caspase-8/Bid pathway.
A prenylated flavanone from the roots of S. flavescens has recorded anti-proliferative activity against human hepatoma cells (HepG2) (Yang et al. 2021). This lavandulyl flavonoid 2-methoxy-2′,4′,4,6-tetrahydroxy-5-lavanduly dihydrochalcone (100) significantly activated autophagic flux and trigger reactive oxygen species (ROS) release in HepG2. Studies of its mechanism suggested that this compound mediates its anti-proliferative effects through autophagic cell death, which is an apoptosis independent. Western blot experiments demonstrated that the prenylated flavanone 100 could also activate the key signaling protein of autophagy and ROS, while it does not affect the main protein of the apoptosis signaling pathway (Yang et al. 2021).
The lavandulylated flavanones, 2′-methoxykurarinone (24) and kurarinone (23), were isolated from the root of S. flavescens, together with two known lavandulyl flavanones, sophoraflavanone G (19) and leachianone A (20), were exhibited cytotoxic activity against human myeloid leukemia HL-60 cells (Chang et al. 2019a, Huang and Xu 2020). Matrine (246) was recoded to promote human myeloid leukemia cells apoptosis through Warburg effect mediated by hexokinase 2 (Lin et al. 2019b). It (246) also inhibited the growth of natural killer/T-cell lymphoma cells by modulating CaMKIIγ-c-Myc signaling pathway (Gu et al. 2020).
The structure of exiguaflavanones A (61) and B (62), purified from S. exigua has approximately the same carbon skeleton orientation as frullanolide. This compound exhibited potent anti-breast cancer activity in breast cancer cell lines, including MDA-MB- (-231and -468) and MCF-7 (Boozari et al. 2019b; Farhadi et al. 2019; Li et al. 2020a; Yang et al. 2020c).
The isoflavonoid, formononetin (125) isolated from the phytoextract of S. flavescens (PSf), has been well-documented for its anticancer and the status of 125 in cancer therapeutics varied from arresting cell cycle, fights progression of cancer via inducing apoptosis, and halting metastasis via targeting various pathways (Jiang et al. 2019a). It also (125) targeted the fibroblast growth factor 2 and protein kinase-B signaling pathways, leading to the suppression of tumor growth and angiogenesis. The in vivo antiangiogenetic synergetic ability of low dose of oxaliplatin, the most used chemotherapy drug for colorectal cancer, combined with an injection of S. flavescens (Sf-I) against the growth of human umbilical vein endothelial cells was reported (Elberry et al. 2020).
Compounds with 2 isoprenyl groups (one in A-ring and the other in B-ring) tetrapterol G (151) and compound with α, α-dimethylallyl group at C-5′ of B-ring such as secundiflorol D (140), secundiflorol E (141), secundiflorol A (154), and secundifloran (155), showed relatively high cytotoxic activity against tumor and normal human cell lines (Guo et al. 2020; Ledoux et al. 2018). The cytotoxicity effect of different isoflavone glucosides and maackiain (172), isolated from S. alopecuroides seeds, against MCF-7, Hep3B, HeLa, and H1299 cells was reported (Rong et al. 2020; Tsai et al. 2020). The arylbenzofuran, 2-(2′,4′-dihydroxy-phenyl)-5,6-methylene dioxybenzofuran (192), isolated from the roots of PSf and the roots and PSt rhizome, potently inhibited IL-6 production and cytotoxic activity against the KB epidermoid carcinoma cell line (Boozari et al. 2019b; Li et al. 2019c; Jin et al. 2020).
Compound Kushen Injection (CKI), as a formula mainly consisted of matrine 246, has been employed into clinical applications in the adjuvant treatment of breast, lung, colon, gastric, esophageal, liver, and pancreatic cancer (Cho et al. 2018; Ao et al. 2019; Chen et al. 2019c; Hong et al. 2019; Liang et al. 2020; Yao et al. 2021). The reported mechanism of this component (246), to suppress KRAS‐driven pancreatic cancer growth, was by inhibiting autophagy-mediated energy metabolism (Cho et al. 2018). Kushen is the root of S. flavescens based-formula inhibited angiogenesis in a collagen-induced arthritis rat model (Ao et al. 2019; Elberry et al. 2020). Kurarinone (23), a lavandulyl flavanone, isolated from the roots of PSf, was reported to have a cytotoxic activity (IC50 = 22.2 µM) against human MCF-7/6 breast cancer cells, but with a weak estrogenic activity (Boozari et al. 2019b). The inhibitory effects of flavonoid-rich extract of S. flavescens on indoleamine 2, 3-dioxygenase 1 (IDO1), a tryptophan catabolising enzyme, is known as a tumor cell survival factor that causes immune escape in several types of cancer (Kwon et al. 2019). Kushenol (28), a flavonoid isolated from PSf, was reported as IDO1-inhibitor and might be useful in the development of immunotherapeutic agents against cancer.
The matrine (246) has been proved in many studies to inhibit cancer cell proliferation and metastasis, arrest cell cycle, promote apoptosis, and reduce the toxicity of anticancer drugs with reversing effects on anticancer drug resistance (Chen et al. 2019d; Cao and He 2020; Fu et al. 2020; Liu et al. 2020a, b; Zhang et al. 2020a). A nontoxic concentration of Matrine (246) also facilitated intrinsic apoptosis pathway and suppressed the expression of ATP-binding cassette subfamily B member 1 drug transport (Chen et al. 2019d). The same compound induced papillary thyroid cancer cell apoptosis in vitro and suppressed the in vivo tumor growth by downregulating miR-182-5p (Li et al. 2019b; Fu et al. 2020). The inhibitory effect of 246 on human nasopharyngeal carcinoma CNE-2 cells was reported (Wang et al. 2018c). Matrine (246) reversed multidrug resistance of breast cancer MCF-7/ADR cells through PI3K/AKT signaling pathway (Zhou et al. 2018b). Another study suggested that the dependence of 246 suppression of self-renewal, on regulation of LIN28A/Let-7 pathway in breast cancer stem cells (Li et al. 2020c). The quinolizidine-type alkaloid (246) can inhibit the proliferation of breast cancer MCF7, BT-474, and MDA-MB-231 cells, which may be related to the inhibition of inhibitory κB kinase β (IKKβ) regulation of nuclear factor κB (NF-κB) signaling pathway (Cao and He 2020; Zhang et al. 2020a).
The anticancer property of a derivative of matrine 246 was explored (Zhou et al. 2019). The activity of N-methyl cytisine (268) in inhibiting breast cancer cell metastasis was previously demonstrated (Chen et al. 2020a).
Leachianols (208–211), are naturally occurring oligostilbenes; the resveratrol trimers were isolated from S. leachiana roots (Yang et al. 2019; Boozari et al. 2019a). These stilbenoids exhibited a wide range of biological activity including a positive result in a cell-based assay for anti-mitotic activity using HeLa cells with a cytotoxic effect. A prepared injection from S. flavescens and Heterosmilax chinensis was reported to disrupt cell cycle and induce apoptosis in breast cancer (Nourmohammadi et al. 2019). This injection strongly reduced migration of HT-29 and MDA-MB-231 cells, moderately slowed brain cancer cells on migration and invasion of colon, brain, and breast cancer cell lines. The prepared injection decreased metastasis by impairing migration in hepatocellular carcinoma cells and repressing RNA markers associated with tumor metastasis in MCF-7 cells.
Oxysophoridine 242 is an alkaloid extracted from S. alopecuroides Linn. Many in vitro cellular and in vivo animal studies showed that 242 has various pharmacological activities. It suppressed the growth of hepatocellular carcinoma (Yao et al. 2012) and colorectal cancer cells (Jin et al. 2017) by regulating apoptosis associated with the Bcl-2/Bax/caspase-3 signalling pathway, and alleviation of spinal cord injury via anti-inflammatory, anti-oxidative stress, and anti-apoptosis effects (Cao et al. 2018).
The protective effects of oxymatrine (249) on homocysteine-induced endothelial injury were reported (Wu et al. 2019a). The involvement of mitochondria-dependent apoptosis and Akt-eNOS-NO signaling pathways was suggested. The ability of sophoridine (240) to suppress rat liver BRL-3A cells viability in a concentration- and time-dependent manner was also recorded (Qiu et al. 2018). This compound (240) increases cytotoxicity, ROS accumulation, and cell apoptosis in a concentration-dependent manner (Qiu et al. 2018). Sophoridine (240) and oxymatrine (249) exerted a good anti-tumor effect in clinical applications (Cao and He 2020; Liu et al. 2020a, b).
Oxymatrine (249) exerted cytotoxicity against cancer cells through regulation various oncogenic signaling pathways such as the Akt, nuclear factor kappa B (NF-κB) cascades, and epidermal growth factor receptor (EGFR) (Halim et al. 2019). This QA 249 induced A549 human non-small lung cancer cell apoptosis via extrinsic and intrinsic pathways (Zhou et al. 2018d; Izdebska et al. 2019). Another study presented the influence of oxymatrine on non-small cell lung cancer cells (Halim et al. 2019; Izdebska et al. 2019). It suggested that this compound has induced dose-dependent cell death, mainly through ER stress-induced apoptosis pathway. Oxymatrine 249 also reduced the metastatic potential by inhibiting the cancer epithelial-mesenchymal transition process (Chen et al. 2019c). A recent study showed that through in vitro inhibiting colon cancer cell epithelial-mesenchymal transition and NF-κB signaling, the compound 249 reversed the resistance of 5-fluorourfacil (Liang et al. 2020).
The role of matrine (246), oxymatrine (249), and PSf alkaloid gel in cervical cancer has been also investigated (Zhou et al. 2018e). The gel has recorded to restrain cervical cancer cell proliferation, induce cellular apoptosis through stimulation of Bax and E-cadherin, induce cell cycle arrest in G2/M phase, inhibit metastasis, and suppression of MMP2, Bcl-2 and cyclin A. The regulation of the cervical cancer cells was suggested via the suppression of AKT/mTOR signaling pathway (Zhou et al. 2018e). Through activating p53 and Hippo signaling pathways, sophoridine (240) inhibited lung cancer cell growth and enhanced the cisplatin sensitivity (ur Rashid et al. 2020; Zhou et al. 2018d, 2020). The same compound 240 was reported to exert tumor-suppressive activities via promoting ESRRG-mediated β-catenin degradation in gastric cancer (Peng et al. 2020b). Sophoridine (240) also reported to suppress macrophage-mediated immunosuppression through TLR4/IRF3 pathway, and subsequently upregulate CD8 + T cytotoxic function against gastric cancer (Zhuang et al. 2020). Sipi soup, a traditional Chinese medicine, which is consists of the aqueous extract derived from 4 plants including PSj, was reported as an anti-inflammatory component. This medicine has suggested that it may prevent the progression of cervical cancer by inhibiting the activation of CAF, and the inflammatory process by reducing HIPK1-AS expression (Zhou et al. 2018a).
Cytotoxic activities of phenolic acids (324–331) from the seeds of S. alopecuroides and the roots of S. tonkinensis against hepato-cellular carcinoma, HepG2, and Hep3B cell lines were investigated (Song et al. 2018; Hou et al. 2020; Zhang et al. 2021c). Butein (331) significantly inhibited the cancer cell growth in a concentration-dependent manner (Song et al. 2018). Another study confirmed that the weak anticancer activity of compounds (325–328) and (3S,2R)-methyl 2-(4-hydroxybenzyl)tartrate (332) against the same two cell lines (Song et al. 2019).
HPLC fingerprint of chloroform extract from S. tonkinensis Gagnep revealed the presence of ononin (109), genistin (119), genistein (120), isosophoranone (127), trifolirhizin (154), isotrifolirhizin (155), and maackiain (172) (Song et al. 2021; Chen et al. 2020c). The authors explored the mechanisms of this extract on suppression tumors of nasopharyngeal carcinoma cells. They found that the extract inhibited cell viability, clonal growth, and induced cell apoptosis in a dose-dependent manner. This effect was exerted by silencing the PI3K/AKT/mTOR signaling pathway, which is associated with upregulation of cleaved PARP, cleaved caspase 3/7/8/9, caspase 3/7/8/9, Bax, and downregulation of PI3K, P-PI3K, PARP, AKT,P-AKT, mTOR, and P-mTOR and Bcl-2 (Ao et al. 2019; Cao and He 2020; Chen et al. 2020d). Compounds 246 and 249 had direct killing effects on various tumor cells, such as HepG2, BEL-7402, and SGC-7901, and could inhibit the invasion and metastasis of gastric cancer last cells (Huang et al. 2018c; Elberry et al. 2020; Zhang et al. 2020a). Matrine (246) has been reported to inhibit the progression of prostate cancer by promoting expression of GADD45B (Huang et al. 2018a). Oxymatrine (249) exhibits anti-tumor activity in a gastric cancer through inhibiting IL-21R-mediated JAK2/STAT3 pathway and the proliferation of vascular endothelial cells (Huang et al. 2018c; Su et al. 2018; Fan et al. 2019).
Combined chemotherapy can effectively reduce or stabilize the tumors, improve quality of life, and significantly alleviate the cancer-induced pain (Fan et al. 2019). Radix S. flavescens contained serum can inhibit cell proliferation and induce apoptosis of human colon cell line SW480 (Liang et al. 2020). This induction of apoptosis was reported as one of the suggested anti-neoplasm mechanisms of the medicine (Elberry et al. 2020). With the effective components of oxysophocarpine (236), matrine (246), and oxymatrine 249 (Cao and He 2020), it has been widely applied to treating various kinds of malignant tumors in China, including colon carcinoma (Liang et al. 2020; Liu et al. 2020a, b). Matrine (246) effectively inhibited the proliferation and anchorage-independent growth of human colon cancer HT29 cells (Fang et al. 2018).
Exposure of gastric cancer cells (AGS and HGC 27) to oxymatrine (249), contributed to the suppression of cell proliferation and invasion (Huang et al. 2018c). The anti-tumor effect of 249 was exhibited via regulation of JAK/STAT signaling pathway. Anti-proliferation effects of phytoextract of S. moorcroftiana (PSm) seeds on HepG2 were also reported (Su et al. 2018). Sophocaprine (235), matrine (246), and aloperine (284) were identified in the extract of PSm with low polarity. Matrine (246) has reported to reverse the Warburg effect and it suppresses colon cancer cell growth via negatively regulating HIF-1α (Hong et al. 2019). This compound (246) has reported to trigger colon cancer cell apoptosis and G0/G1 cell cycle arrest, where 284 has induced apoptosis and G2/M cell cycle arrest in HepG2 (Liu et al. 2019a, 2020b). Matrine (246) induced apoptosis in human esophageal squamous cancer KYSE-150 cells through increasing ROS and inhibiting mitochondrial function (Jiang et al. 2018). It also inhibited the growth of oral squamous cell carcinoma cells in vitro and in vivo (Li et al. 2019d). Matrine (246) has exhibited its effect via mediation of microRNA-22 (ur Rashid et al. 2019; Liu et al. 2020b), where aloperine (284) was through PI3K/Akt signaling pathway (Liu et al. 2019a).
Matrine (246) has also involved in the progression of gastric cancer through inhibiting miR‐93‐5p and upregulating the expression of target gene AHNAK (Liu et al. 2020b). The effect of matrine (246) from S. flavecens on hepatocellular carcinoma, the one of the most prevalent and lethal cancer with high metastasis and recurrence rates, has been investigated (Dai et al. 2021). This significant anti-metastatic effect was attributed to enhanced miR-199a-5p expression and subsequently impaired hypoxia-inducible factor-1α signaling and epithelial-mesenchymal transition. Thus, matrine (246) was suggested as a promising component with anti-metastatic medication for hepatocellular carcinoma therapy.
The mechanism of allomatrine (248), isolated from the bark of S. japonica, in invasion and proliferation inhibition of human lung cancer A549 cell line was also investigated. This alkaloid inhibited the proliferation and invasion in vitro by promoting apoptosis, inducing ROS production, inhibiting ubiquitin proteasome, arresting cell cycle, and regulating tumor related gene expression (Liu et al. 2020a, b).
The quinolizidine-type alkaloid aloperine (284) extracted from PSa, showed some therapeutic effects against multiple myeloma and colon cancer though increasing cell apoptosis (Yin et al. 2018; Li et al. 2020d).
The quinolizidine alkaloid 284 exerted the most potent cytotoxic activity on leukemia cell lines U937, HL-60, and K562, lung cancer cells, glioma cells, ovarian cancer cells, oesophageal cancer EC109 cells, and HepG2 cell lines (Qiu et al. 2020b; Zhou et al. 2020).
The combination of compound 284-Adbic (adenoviral vector expressing p14ARF/p53) can produce a synergistic effect as anti-cancer mixtures at low doses. This may offer less toxic and more effective treatment strategy for non-small cell lung cancer (Muhammad et al. 2020). Research conducted by Song’s group has recorded that 284 significantly inhibited the viability of bladder cancer cells, mostly via suppressing hypoxia induced epithelial-mesenchymal transition activation of mTOR/p70S6K/4E-BP1 pathway (Zhou et al. 2020).
The anti-tumor effect of total alkaloids of S. alopecuroides (TASa) in H22 tumor-bearing mice was investigated. It was found that different dose groups of TASa extract could apparently inhibit the solid H22 tumor in mice, with a clear dose–effect relationship (Huang et al. 2018c; Jia et al. 2020).
The potential cytotoxic, antimicrobial, antioxidant, and anti-enzyme of S. alopecuroides L. seeds was recently reported (Zahra et al. 2021). Twelve quinolizidine alkaloids; sophalodes A–L (307–319), which were matrine-type alkaloids isolated from S. alopecuroides seeds (Li et al. 2021a). Structurally, sophalodes A-D (307–310) were the examples of C-11 oxidized matrine-type alkaloids from Sophora plants.
The sophalodes (307–319) A-L inhibited nitric oxide production induced by lipopolysaccharide in RAW 264.7 macrophages, among them, compounds sophalode K 318 exhibited the most significant activity with IC50-values of 29.19 μM.
Evaluation of anticancer activity of isolated compounds from S. mollis (Royle) Graham Ex Baker, including scopoletin 216 and β-sitosterol glucoside 218 have revealed weak effect against HeLa and 3T3 cell lines (Quradha et al. 2021). The major compounds derived from Sophora species that having cytotoxic and anti-tumor activities were illustrated in Table 16.
Neurological activity
Many studies have reported the neuroprotective effects of QA isolated from Sophora (Wang et al. 2019a, d). The useful use of PSf and the isolated active alkaloid (246), as natural alternatives for hypnotic medicine, was suggested (Wang et al. 2019a). They have alleviated the hyperactivity induced by caffeine and promoted a shift toward non-rapid eye movement sleep. The mechanism was suggested to decrease wake time by the activation of the neurons of ventrolateral preoptic nucleus, and through modulating serotonergic transmission (Wang et al. 2019a). Aloperine (284) has significant neuroprotective effects, in an Alzheimer’s disease cellular model, attributing to its anti-oxidative stress (Wang et al. 2019d). This compound 284 ameliorated oxidative stress patterns in presenilin 1 exon 9 deletion mutant cells by reducing the production of 4-hydroxy-2-nonenal and ROS (Zhao et al. 2018). Its treatment increased mitochondrial membrane potential (MMP) in N2a/Swe.D9 cells as Alzheimer’s disease cellular model.
The phytoextract of S. flavescens (PSf) and its prenylated flavonoids; possessing low molecular weights and lipophilic moieties, may be potent therapeutic and preventive candidates for Alzheimer’s disease (Boozari et al. 2019b; Cho et al. 2020).
β-Site APP cleaving enzyme 1 (BACE1) activities in Alzheimer’s disease were significantly inhibited by kushenol (28), isoxanthohumol (32), leachianone A (20), and kuraridin (184) with IC50-values of 5.45, 7.19, 8.56, and 6.03 μM, respectively. These compounds were isolated from the lead bioactive fractions EtOAc and CH2Cl2 of the crude PSf (Boozari et al. 2019b; Chen et al. 2019a).
Maackiain (172) has emerged as a valuable agent for Parkinson’s disease treatment, with remarkable improvements in food-sensing behavior and lifespan (Tsai et al. 2020) mainly by inhibiting monoamine oxidase B. Maackiain (172) significantly reduced dopaminergic neuron damage in 6-hydroxydopamine-exposed worms of the BZ555 strain. The neurological activity was illustrated in Table 17.
Antimicrobial activity
Sophora exigua has reported with antimicrobial activities against Candida albicans, Staphylococcus epidermidis, and Pseudomonas aeruginosa (Kaewdana et al. 2021). Sophoraflavanone G (19), isolated from S. exigua also exerted an antibacterial effect by reducing the fluidity of cellular membranes (Boozari et al. 2019b; Farhadi et al. 2019).
Flavanones (19) and (23) exhibited potent antibacterial activities (10 µg/disk) against Gram-positive bacteria (Farhadi et al. 2019). The phytoextract of S. alopecuroides roots and its content of pterocarpan derivative, maackiain (172) helped individuals with androgenetic alopecia as the derivative promoted the proliferation of human hair keratinocytes (Chang et al. 2018; Wang et al. 2018f; Huh et al. 2020).
Sophoraflavanone B (48) also caused cell wall weakening and consequently membrane damage had occurred, and intracellular constituents leaked from the cell as the proposed mechanism of its antimicrobial activity against methicillin-resistant-Staph. aureus (anti-MRSA) (Farhadi et al. 2019; Hadadi et al. 2020). An antibacterial synergism of compound (48) with antibiotics, including ampicillin, gentamicin, and oxacillin anti-MRSA, was previously determined (Farhadi et al. 2019). While the other prenylated flavonoid, sophoraflavanone G (19; from S. flavescens and S. exigua) potentiated the effect of ampicillin or oxacillin against the MRSA infection (Farhadi et al. 2019). In addition, the compound (19) showed significant antibiofilm formation against Bacillus subtilis, Staph. aureus, and Staph. epidermidis with MIC-values ranging from 3.1 to 12.5 μg mL−1 (Farhadi et al. 2019). The lavandulyl flavanones isolated from the roots of S. exigua (PSe) (Li et al. 2020a; Yang et al. 2020c) were proposed to be potent phytotherapeutic agents against MRSA infection (Farhadi et al. 2019). A complete growth inhibition of 21 MRSA-strains (3.13–6.25 μg mL−1) was exerted by exiguaflavanones-A (61) and B (62), the isolated flavanones from PSe with a lavandulyl residue (Farhadi et al. 2019). Partial synergistic effects with anti-MRSA antibiotics, such as minocycline, rifampicin, and vancomycin, were also reported. Lavandulyl flavanones were also reported as anti-malarial, anti-inflammatory, and cytotoxic agents (Boozari et al. 2019b; Li et al. 2020a; Kaewdana et al. 2021).
The potentials of total alkaloids of S. alopecuroides (TASa) and matrine (246) in prevention of biofilm formation in Staph. epidermidis infections were indicated (Jia et al. 2019). A previous study evidenced that the isoflavonoid genistein (120), isolated from S. moorcroftiana, could be a potential inhibitor of NorA, a multi-drug transporter of Staph. aureus (Farhadi et al. 2019). The mode of action of the aglycone on different bacterial cells was studied and the changes exerted in the cell morphology of bacteria were suggested as the underlying mechanism. Additionally, mixing this compound with the bacterial culture immediately and significantly inhibited the global synthesis of RNA and DNA (Farhadi et al. 2019).
The chalcone isobavachalcone (185) has reported with the lowest MIC-value (0.3 μg mL−1) of Gram-positive bacteria, which was fourfold lower than the MIC-value (4.9 μg mL−1) of the conventional drug gentamicin (Aly et al. 2019; Farhadi et al. 2019; Rammohan et al. 2020).
Matrine (246) and oxymatrine (249) have anti-Helicobacter pylori (Hp) infection effects, the closely related to the digestive diseases (Li et al. 2020d; Wang et al. 2020b, c). The compounds used for oral ulcers and have the potential of antibacterial inflammation, and alleviating swelling, pain, and sore myogenic (Li et al. 2020d; Wang et al. 2020b). Additionally, ASa combined with omeprazole or bismuth pectin has promising antimicrobial activity against Hp, and conventional triple therapy through inhibition of Hp-induced IL-8, COX-2, and NF-кB expression. Summary of findings on the antimicrobial activity of Sophora compounds was also illustrated in Table 18.
Anti-osteoporosis activity
In a sub-cytotoxic concentration range, a derivative of genistein (120) showed a significant proliferative activity in estrogen-dependent cell line of MCF-7. In a previous report, prominent anti-osteoporosis effect of the CH2Cl2 fraction from PSj fruits was attributed to the high content of isoflavone (120). This aglycone has a peculiar anti-osteoporotic dual mode of action. It was recorded to be the most effective isoflavone in preserving bone health and rebalancing bone turnover towards bone formation through its positive ability to regulate bone cell metabolism (Yang et al. 2020c). This isoflavone inhibited osteoclast and stimulated osteoblast function, mainly through the osteoprotegerin-sRANKL system.
Compounds genistein 120 and sophoricoside 134 from S. japonica was also reported to prevent the osteoporosis (Yang et al. 2020c). Chemically, isoflavonoids are compounds with ring C in position 3 instead of position 2. The isoflavone aglycone genistein (120) and flavonol aglycone kaempferol (2) were found to be the main phytoestrogens in the naringinase-treated phytoextract of S. japonica (PSj) seeds (Yang et al. 2020c). S. japonica seeds extract PSj prevented bone loss, partly by inhibiting the osteoclastic activity. The alcoholic PSj showed a significant estrogenic activity only after naringinase treating. Kaempferol (2), was nearly equipotent to genistein (120) as an estrogen agonist (Yang et al. 2020c), which was reported to prevent the osteoporosis (Yang et al. 2020c). Fructus Sophorae (FS) or Huaijiao, the dry mature fruit of S. japonica, were proved to prevent bone loss in ovariectomized rat’s model (Shim et al. 2005). The effect of flavonoids, including rutin (3), genistein (120), pratensein (129), biochanin (130), prunetin (131) sophoricoside (134), and rutin (3), on rat osteoblasts was also reported (Yang et al. 2020c). They were act as estrogen-like reagents and significantly increased the cellular activity of MC3T3-E1 cells. Another study disclosed the effects of alkaloids on osteoblasts infected with Staph. aureus and osteoclasts (Wang et al. 2018e).
Matrine (246) inhibited TGF-β induced Smad2/3 phosphorylation and transcription of runt-related transcription factor-2, alkaline phosphatase, and osteocalcin after osteoinduction (Mao et al. 2020). Matrine 246 inhibited heterotopic ossification by suppressing the migration and osteogenic differentiation of TGF-β (Mao et al. 2020).
Sophocarpine (235) also exerted a protective effect against ox-LDL-induced endothelial damage (Fang et al. 2020), via regulating NF-κB signaling pathway (Wu et al. 2019b). The same compound (235) suppressed NF-κB signalling pathway and attenuated wear particle-induced implant loosening by inhibiting osteoclastogenesis and bone resorption (Zhou et al. 2018c).
Hepatoprotective activity
Sophora fruit tea is a traditional Chinese herb tea rich in sophoricoside (134). This compound is an isoflavone glycoside (genistein-4′-O-β-d-glucopyranoside) isolated from dried S. japonica fruit (Li and Lu 2018). The hepatoprotective effect of 134 was exerted against fructose‐induced liver injury via regulating oxidation, lipid metabolism, and inflammation in mice. Other alkaloids, such as sophoridine (240) and oxymatrine (249), were the key components of S. flavescens (Sf -I). The Sf-I can improve immunity, protect the liver against oxidation, and provide a chemotherapeutic sensitivity (Cao and He 2020; Liu et al. 2020a, b). The possible mechanism of hepatotoxicity induced by phytoextract of S. flavescens (PSf) has been explored (Jiang et al. 2020c). It has been suggested that PSf exhibited a metabolism disturbance of the bile acids, fatty acids, glycerophospholipids, and amino acids of orally exposed rats. The hepatoprotective activity of the benzoic acid derivatives sophophenoside A (322) and sophophenoside B (323) on the cytotoxic effect of D-galactosamine in HL-7702 cells was assayed (Jiang et al. 2020b). Sophophenoside A (322) exhibited a moderate hepatoprotective activity at a concentration of 10 μM. A combinational treatment of S. moorcroftiana alkaloids with albendazole displayed better therapeutic effects than albendazole alone against liver echinococcosis, and alleviated liver injury (Zhang et al. 2018a). A recent study also suggested that aloperine (284) could prevent the early development of hepatocellular carcinoma like lesions in rat liver (Shi et al. 2019).
Muscle and vasorelaxation activity
Kushenol (28) and sophoraflavanoneB (48) strongly induced the relaxation of porcine coronary arteries with respective ED50-values of 8.6 and 12.4 µM, separately (Aly et al. 2019). Relaxation of bladder smooth muscle by flavonoids of S. flavescens was also recorded (Han et al. 2018), via direct activation of the large‐conductance calcium‐activated potassium channel. Matrine (246) also improved skeletal muscle atrophy by mostly inhibiting E3 ubiquitin ligases and activating the Akt/mTOR/FoxO3α signaling pathway (Chen et al. 2019b). Authors also demonstrated that comound (246) could increase muscle fiber size and muscle mass in an in vivo CT26 colon adenocarcinoma cachexia mouse model (Chen et al. 2019b). Furthermore, Yang et al. 2018 suggested that aloperine (284) induced vasodilatory effects, in rat aorta, mainly due to the operations of voltage-operated potassium channels and ATP-sensitive potassium channels. The vascular relaxant effect of ethanolic extract of S. flavescens roots was found, via endothelium-dependent NO-soluble guanylyl cyclase–cGMP signaling pathway (Aly et al. 2019; Boozari et al. 2019a, b).
Antiviral activity
In addition to the ability of various plants secondary metabolites to interfere directly with viral replication, these metabolites may also modulate the host immune response against infection with limited side-effects (Abd-Alla et al. 2009; Yang et al. 2019; Ma et al. 2020a). S. subprostrata Chun and T. chen radix (EC90 = 82.2 ± 8.2 and 27.5 ± 1.1 μg mL−1, respectively with SI = 11.1) was one of the most active 22 plants against mouse hepatitis virus (MHV)-A59-infected mouse dihydrolipoamide-branched chain transacylase E2 cells. The plant extract significantly reduced the viral replication after 6 h of exposure following infection. In another screening study on 19 plants, S. flavescens Aiton radix was confirmed to be an effective on the viral replication in MHV-A59-infected cells (Kim and Yun-Choi 2008; Orhan and Deniz 2020). Interestingly, S. subprostrata has the capacity to inhibit coronaviruses replication in vitro which holds a promise for a prospect drug against the emerging COVID-19 (Fuzimoto and Isidoro 2020; Ghoran et al. 2021).
The lavandulylated flavanone, kurarinol (29), chalcone, and kuraridin (184), isolated from S. flavescens roots have been reported to have antiviral activities (Boozari et al. 2019b; Yang et al. 2019). Compound (29) also inhibited rotavirus-induced cytopathic effects, probably due to its ability to modulate the rotavirus replication and toll-like receptor 3-mediated inflammatory pathways. This chalcone inhibited the adsorption and replication of reoviruses (human type 1–3 reoviruses and Korean porcine reovirus), by inhibiting viral RNA, protein synthesis, hemagglutination, and shedding (Boozari et al. 2019b; Chen et al. 2020d). Many flavonoids, including 6 isoflavones and three pterocarpans, isolated from S. davidii flowers have reported to exhibit protective effects against Tobacco Mosaic virus replication with a higher inhibition rate than ningnanmycin as a positive control (Wang et al. 2018d; Yang et al. 2019).
Exiguaflavanone A (61) has been reported as one of the anti-Zika virus phytochemicals, by strongly interacting with the ATP site of helicase (Farhadi et al. 2019). The anti-influenza activity of the decoction of San Wu Huangqin D-SWH, a traditional Chinese medicine formula, was suggested to be related with its regulation ability on the immune system or/and inhibitory effects on major virus proteins (Ma et al. 2018, 2020a, b; Li et al. 2020d). Interestingly, Chinese patients (~ 80%) with the chronic hepatitis B and ~ 4 billion of the world population rely on traditional herbal medicines (Liu et al. 2018b; Mohamed et al. 2019). The main herbal component of this formula is S. flavescens, the well-known phytoextract, having a wide spectrum of biological properties. S. flavescens exerted broad antiviral effects versus influenza, hepatitis B virus (HBV) (both wild-type and entecavir-resistant), enterovirus71, coxsackie B3, and respiratory syncytia, due to its key bioactive substances, including flavonoids, triterpenoids, and quinolizidine-type alkaloid (QA) (Liu et al. 2018b; Ma et al. 2018, 2020a, b; Ren et al. 2019; Wang et al. 2019a).
QA were extracted from many Sophora species (Table 11) and have been exerted various excellent activities such as sophocarpine (235), sophoridine (240), matrine (246), oxymatrine (249), and aloperine (284) (Huang et al. 2018c; Ao et al. 2019; Zhang et al. 2020a).
Matrine-family alkaloids, as tetracycloquinolizindine analogues, have aroused great interests over the past decades (Chen et al. 2018d; Wang et al. 2018b; Fu et al. 2020; Cai et al. 2020), which possessed various pharmacological activities (Wang et al. 2019a; Cai et al. 2020). These alkaloids showed a potent antiviral activity against HBV-infection (Zhang et al. 2018b, c; Parvez et al. 2019). Matrine (246) and oxymatrine (249), as common alkaloids, have been used for the clinical treatment of hepatitis in China (Zhang et al. 2020a). Many matrine-type alkaloids isolated from the PSf roots were investigated for their antiviral activities against HBV (Zhang et al. 2018b, 2020a). The combined matrine (246) and oxymatrine (249) capsules were used for the treatment of chronic HBV infection (Parvez et al. 2019). Many matrine-type alkaloids, isolated from the rhizomes of S. tonkinensis, have also exhibited antiviral activity (Pan et al. 2015).
9α-Hydroxysophocarpine (237), 12β-hydroxyoxysophocarpine (239), 5α-hydroxymatrine, and sophoranol (251) have been exhibited antiviral activity against the coxsackie virus B3, with IC50-values of 26.62–252.18 μM (Pan et al. 2015; Ti et al. 2021). The alkaloids 12α-hydroxysophocarpine (238), 239, and sophoramine (243) inhibited influenza virus A/Hanfang/359/95 replication with IC50-values of 63.07–242.46 µM. Oxymatrine (249), sophoridine (240), alopecurine B (286), flavesine G (287), and flavesine J (290) also effectively inhibited HBsAg secretion at noncytotoxic concentrations than lamivudine, as a positive control (Ti et al. 2021).
Matrine (246) in vitro inhibited the replication of porcine reproductive and respiratory syndrome virus co-infection in PAM cells. The underlying antiviral mechanism of 246 may be mediated through its ability to partly regulate TLR3,4/NF-κB/TNF-α pathway (Sun et al. 2018; Ti et al. 2021).
The polysaccharide isolated from S. subprosrata regulated the inflammatory response of mouse mononuclear macrophage cell line RAW264.7 cells infected with porcine circovirus type 2 (Li et al. 2019a; Yang et al. 2020b). The bush Sophora root polysaccharide and its sulfate as well as baicalin-phospholipid complex (BPC) inhibited duck hepatitis A virus type 1 (DHAV-1) adsorption, replication, and release (Chen et al. 2018a, b). This effect may be due to drop the RNA synthesis and the protein translation via reducing the 3D protein stability, inhibiting cellular Hsp70 expression, and suppressing DHAV-1 IRES activity (Chen et al. 2018c).
Sophoridine (240) has exhibited obvious antiviral effects both in vitro and in vivo against CoxsackievirusB3 (CV-B3), by regulating cytokine expression. It is likely that the alkaloid (240) itself, not its metabolites, is mainly responsible for the antiviral activities (Ma et al. 2018; Ren et al. 2019). The serum samples obtained from rats with oral administration of compound 240 reduced this virus, a major pathogen for acute and chronic viral myocarditis, titers in infected myocardial cells. Matrine (246) has been reported to suppress the viral RNA copy number on rhabdomyosarcoma cells (Ma et al. 2018; Ren et al. 2019). Moreover, 246- treatment of mice challenged with a lethal dose of human enterovirus 71, one of the major causative agents of hand, foot, and mouth disease in children less than 6 years old, reduced the mortality and relieved clinical symptoms (Ren et al. 2019). The matrine-type alkaloid sophoridine (240) inhibited EV71 infection in Vero cells, and it was highly effective against EV71 when Vero cells were pretreated with 240 for 2 h. This compound was highly effective at inhibiting EV71 attachment at a concentration over 250 μg mL−1 (Ren et al. 2019; Ti et al. 2021). The role of 249 and anagyrine (273), as the potent anti-respiratory syncytial virus components (Ma et al. 2018; Parvez et al. 2019) that have been isolated from the active PSf was recorded. The suggested mode of 284 to inhibit human immunodeficiency virus (HIV)-1 infection, was by blocking virus entry and inhibited the virus envelope-mediated cell–cell fusion (at low concentrations) (Aly et al. 2019; Wang et al. 2019a). This compound has approximately sevenfold more potent activity against HIV-1 than influenza virus (Wang et al. 2019a).
Matrine-based alkaloids (279–281), isolated from S. alopecuroides seeds, were reported with a similar or even better efficacy compared to the treatment with lamivudine and/or interferons (Wang et al. 2019a). Compounds 279 and 280 possess unprecedented 6/6/6/4 and 6/5/6/6 ring systems, respectively, while 281 has a pair of stereoisomeric matrine-acetophenone alkaloids with an unusual skeleton. A rare 1,4-diazaindan-type alkaloid; flavascensine (279) was also isolated from S. flavescens roots (Wang et al. 2019a; Ti et al. 2021). Sophaline E (282), the first example of sparteine-indolizine alkaloid, sophaline G (283a) and sophaline H (283b), the epimeric normatrine-julolidine alkaloids with unusual skeletons were isolated from S. alopecuroides and have showed antiviral activities against HBV (Wang et al. 2019a; Li et al. 2020d; Majnooni et al. 2021). A quinolizidine alkaloid with an unusual endocyclic ring system, aloperine (284) was extracted from the seeds and leaves of S. alopecuroides L. (Majnooni et al. 2021). Compound 284 has been identified as a potent hepatitis virus C inhibitor with EC50-value of 4.23 ± 0.99 μM (Dang et al. 2016; Ti et al. 2021).
HIV-1-NL4-3 NanoLuc-sec virus infection of MT4 cells was performed, in the presence of various concentrations of 284 to evaluate its anti-HIV activity. The quinolizidine alkaloid 284 showed anti-HIV activity with EC50-value of 1.75 ± 0.59 μM. BMS-806-resistant, Env-mediated cell–cell fusion, such as that mediated by YU2-T198P and 8x, may be inhibited by 284 (Ti et al. 2021). Through endocytosis inhibition, the quinolizidine alkaloid 284 effectively prevents the propagation of hepatitis virus C (HCV) in Huh7.5 cell line and primary human hepatocytes without cytotoxicity (Lv et al. 2020; Zhou et al. 2020).
Recently, Zhang et al. 2021c isolated unusual matrine–adenine hybrids from S. davidii with inhibitory effects on human cytomegalovirus (HCMV). Sophovicines B (233) and C (234) can inhibit HCMV replication effectively with IC50-values of 7.12 and 7.32 μM, respectively.
Pulmonary hypertension
The alterations of phosphodiesterase (PDE) isozyme related to various pathologies and the design of specific PDE inhibitors might lead to the development of new specific therapeutic strategies in numerous pathologies (Farhadi et al. 2019). The prenylated flavonol, kushenol (28), mediated mTOR pathway by inhibiting PDE and Akt activity to induce apoptosis in non-small-cell lung cancer cells (Chen et al. 2019a). PDE5‐inhibiting drugs are also used in the treatment of erectile dysfunction and pulmonary hypertension. A strong and selective inhibitory activity against rat diaphragm phosphodiesterase 5 (IC50 = 4.77 mg/mL) was exerted by the methanolic extract of S. flavescens roots (Boozari et al. 2019b). The isoflavonoid aglycones, biochanin (130) and prunetin (131), have selectively inhibited phosphodiesterase (PDE) isozyme PDE4. Orobol (5,7,3′,4′-tetrahydroxyisoflavone) was obtained from the ethyl acetate fraction of S. davidii (Franch.) Skeels (Li et al. 2021b). One of orobol synonyms is isoluteolin (126). A derivative of the flavone luteolin (4) exhibited a dual inhibition on PDE2/PDE4 with an IC 50 ~ 40 μM, where quercetin (1) selectively inhibited PDE3/PDE4 (Boozari et al. 2019b; Farhadi et al. 2019).
A study from Chang and his coauthors provided information about the role of 284 to suppress human pulmonary vascular smooth muscle cell proliferation via inhibiting inflammatory response (Chang et al. 2019b). Aloperine (284) could protect the mice against bleomycin -induced pulmonary fibrosis by attenuated fibroblast proliferation and differentiation (Yin et al. 2018; Chang et al. 2019b). Mechanistic studies revealed that 284 could regulate the phosphatidylinositol-3-kinase/protein kinase B/mammalian target of rapamycin (PI3K/AKT/mTOR) and TGF-β/Smad signaling pathways by reducing fibroblast proliferation and differentiation, respectively (Yang et al. 2018; Yin et al. 2018; Wang et al. 2019d). While the quinolizidine alkaloids, isolated from S. viciifolia flowers including three matrine-type alkaloids (291–293) and the sparteine-type alkaloid (294), showed no activity as anti-inflammatory and anti-influenza virus (Gao et al. 2020).
Anti-inflammatory activity
Kushenol (28) was reported to inhibit autophagy and impairs lysosomal positioning via VCP/p97 inhibition (Kwon et al. 2020). A recent study has demonstrated that kushenol dose-dependently suppressed the production of inflammatory mediators in lipopolysaccharide-stimulated RAW264.7 macrophages (Cho et al. 2020). In HaCaT cells, the compound exerted antioxidant stress activity and prevented DNA damage as well as cell death by upregulating the endogenous antioxidant defense system.
The potent inflammasome-activating effect of maackiain (172) was also reported (Huh et al. 2020). The compound (172) has been suggested to exert an immunostimulatory effect by promoting IL-1β production via activation of the inflammasome/caspase-1 pathway and this may be clinically useful as an acute immune-stimulating agent.
The plant phytoextracts are also used for conditions involving the liver, heart, intestinal tract, and skin (Boozari et al. 2019b; Quradha et al. 2021). Experimental investigations indicated that its phytoextracts stimulated anticancer, antibacterial, antiviral, anti-inflammatory, and antipruritic responses with wound healing benefits (Huang et al. 2018a; Su et al. 2018; Boozari et al. 2019b).
Recently, S. gibbosa extract-loaded microemulsion-impregnated gelatin/chitosan was suggested to be a potential candidate for the wound healing (Shalaby et al. 2021).
The prenylated flavonoids, namely kushenol (28) and sophoraflavanone B (48), are used in wound healing (Chen et al. 2019a) and also exerted anti-inflammatory and anti-oxidative stress activities (Cho et al. 2020). Compound (28) is a noncompetitive potent inhibitor of tyrosinase, the enzyme responsible for synthesizing melanin, thereby has potential cosmetic applications as a skin whitener (Boozari et al. 2019a, b; Cho et al. 2020). Decoction of an herbal Chinese formula comprises the S. flavescens rootstock, has been reported as a botanical remedy for treatment of skin inflammation (Chang et al. 2018). ASa was clinically used for the treatment of chronic prostatitis and psoriasis (Aly et al. 2019; Boozari et al. 2019b; Hadadi et al. 2020). The isolated prenylated flavonoids from S. flavescens roots also can inhibit the cell growth and production of pro-inflammatory mediators which were related to skin diseases such as psoriasis (Chang et al 2018; Boozari et al. 2019b; Yang et al. 2020a, b, c). The anti-proliferation effect of oxymatrine (249) on human skin keratinocytes has been reported to be related with the suppression of p63 expression. This compound does not affect the formation of basement membrane, which is very important to maintain the normal function of human skin keratinocytes (Aly et al. 2019; Wang et al. 2020a, b).
Many dosage forms were prepared from S. alopecuroides and applied to the clinical therapy e.g., the tablet (colitis enteritis and dysentery), suppository (cervical erosion and gynecological inflammation), and oil liniment (neurodermatitis and eczema) (Chen et al. 2020b; Jia et al. 2020; Li et al. 2020d).
The effect of many prenylated flavonoids, isolated from roots and rhizomes of S. tonkinensis, on the expression of inflammatory mediators and proprotein convertase subtilisin/kexin type 9 (PCSK9) was also studied (Ahn et al. 2019). Lonchocarpol A (56) at concentrations of 20 μM downregulated PCSK9 mRNA expression in HepG2 cells (Ahn et al. 2019).
Matrine (246) can induce endoplasmic reticulum stress in MCF-7 cells, downregulate the expression of hexokinase II, and inhibit the energy metabolism. In addition, studies have reported that matrine (246) has a therapeutic effect on Alzheimer’s disease, encephalomyelitis, asthma, myocardial ischemia, rheumatoid arthritis, osteoporosis, and its mechanism is mainly related to the inhibition of inflammatory response and apoptosis (Liu et al. 2020a, b; Yang et al.2020a, b, c; Zhang et al. 2020a).
Matrine (246) has recorded to alleviate Staph. aureus lipoteichoic acid-induced endometritis via suppression of TLR2-mediated NF-κB activation (Jiang et al. 2019b).
The combination of S. japonica L. and Scutellaria baicalensis Georgi improved the intestinal barrier function, reduced inflammation, increased short-chain fatty acids, decreased indoxyl sulfate, and inhibited oxidative stress reactions (Guan et al. 2021). Both plants have anti-hypertensive effects with clinical benefits against kidney injury.
The pretreatment with aloperine (284) protected mice against ischemia reperfusion-induced acute renal injury as manifested by reducing tubular apoptosis, attenuating inflammatory infiltration, and/or improving renal function (Wang et al. 2019a; Boozari et al. 2019a). By inhibiting inflammation and infiltration of macrophages, Psf exhibited a protection against mycobacterial trehalose dimycolate-induced lung granuloma (Liu et al. 2018a). Mechanistic studies revealed that QA (284) selectively repressed IFN-γ and IL-1β expression by NFκB transcriptional activity and regulating PI3K/Akt/mTOR signaling (Yin et al. 2018; Ye et al. 2020). Matrine (246) prevented adriamycin-induced nephropathy (AIN) through the modification of disordered plasma lipids and recovery of renal function, and this bioactivity is partly attributed to the suppression of renal inflammation and Treg/Th17 imbalance regulation (Aly et al. 2017; Wang et al. 2019a). This compound (246) also protected PC12 cells from lipopolysaccharide-evoked inflammatory injury via upregulation of miR-9 (Jiang et al. 2020a), and normalized regulatory T cells (Treg)/T-helper17 cells (Th17) ratio in peripheral blood mononuclear cells of AIN rats (Wang et al. 2019d; Zhang et al. 2020a, 2021a, b, c).
Besides, alkaloids (Table 11) are one of the main effective substances of Sophora species with anti-inflammatory activity. The total alkaloids of phytoextract of S. alopecuroides (TASa) protected against colitis, probably due to the downstream pro-inflammatory mediators and the inhibition of nuclear transcription factor κB (NF-κB) activation (Halim et al. 2019; Chen et al. 2020b). The TASa increase the expression of CD4+ CD25+Tregs and IL-10 in experimental colitis (Fan et al. 2019; Li et al. 2020d).
Compound scopoletin 216, isolated from S. mollis (Royle) Graham Ex Baker, showed a significant anti-inflammatory effect as it reduced edema of the paw (83.98%), which was more potent than the standard drug (ibuprofen, showed an inhibition percentage of 73.22%). A docking study on a phenolic acid derivative into the binding site of cyclooxygenase isoform showed a better protective effect against COX-1 than the COX-2 isoform (Quradha et al. 2021).
The ability of S. flavescens containing Chinese formula to elicit anti-inflammatory and anti-oxidative stress response in vitro has been reported. The formula blocked cellular transformation and protected against dextran sulfate sodium-induced colitis in a mouse model (Fang et al. 2018). Sophocarpine (235) was reported to ameliorate dextran sodium sulfate-induced colitis by regulating cytokine balance (Chen et al. 2020b). This compound has prevented cigarette smoke-induced restenosis in rat carotid arteries after angioplasty (Yang et al. 2020a). The compound can play the anti-inflammatory role in cells. The alkaloid (249) has an anti-inflammatory effect by interfering with the IκB-α protein expression and inhibiting NF-κB activity in colitis cells (Chen et al. 2020b).
Among QA from S. tonkinensis, sophocarpine (235), (−)-anagyrine (273), 14β-hydroxymatrine (301), and 7β-sophoramine (302) had high anti-inflammatory activities (in-vitro). While sophocarpine (235), 5α-hydroxymatrine (284) (−)-anagyrine (273), and 5α,14β-dihydroxymatrine (300) exhibited higher anti-inflammatory effects (in vivo) (He et al. 2019). Oxymatrine (249) prevented synovial inflammation and migration via blocking activation in rheumatoid fibroblast-like synoviocytes (Zhou et al. 2016; Liang et al. 2018). Suppression of the activation of NF-κB and MAPK in human chondrocytes and inhibition of IL-1β-induced expression of matrix metalloproteinases were exhibited by matrine (246) (Fan et al. 2019; Li et al. 2020d). Aloperine (284) showed a suppressive effect on the swelling of hind paw inrats which induced by carrageenin, macostatin, PGE2, histamine, and 5-HT. It also increased the permeability of capillaries caused by histamine and the leukocytic migratory response (Wang et al. 2020b). Different concentrations of matrine (246) solution all had inhibitory effects on the growth of C6 cell lines, which showed an apparent dose- anti-glioma effect relationship (Zhang et al. 2020a, 2021a, b, c).
Recently (Li et al. 2021a), a quinolizidine alkaloid of matrine-type; sophalode K (318); isolated from the seeds of S. alopecuroides suppressed the protein levels of iNOS and COX-2, revealing its anti-inflammatory potential.
Summary of findings on the anti-inflammatory effects of compounds derived from Sophora species was illustrated in Table 19.
Anti-allergy activity
The anti-allergy and anti-inflammatory effects of the root of S. flavescens were investigated using 1-fluoro-2,4-dinitrofluorobenzene-induced contact dermatitis mouse model and in vitro using RBL-2H3 cells (Aly et al. 2019; Yoo et al. 2019). The release of histamine and β-hexosaminidase and migration was inhibited by treatment with this extract. Anti-asthma herbal medicine intervention is an aqueous extract of three herbs, including S. flavescens (Yoo et al. 2019). It prevented allergic asthma airway hyper-reactivity in mice and inhibited acetylcholine induced airway smooth muscle contraction in tracheal rings from allergic asthmatic mice. The methanol extract of S. flavescens was reported as antipruritic agent that could affect acute and chronic pruritus (Aly et al. 2019). This extract inhibited serotonin (5-HT)-induced scratching (an itch-related response) in a dose-dependent manner, without affecting the locomotor activity. Anti-allergic prenylated flavonoids from the roots of S. flavescens were also recorded (Boozari et al. 2019b). Sophoraflavanone G (19), leachianone A (20) and kushenol (28) inhibited the release of β-hexosaminidase from cultured RBL-2H3 cells (IC50 values ranging from 15 -30 μM). The regulatory effect of ethanolic extract of Flos S. japonica on allergic mediators produced by intracellular calcium ionophore activation in mast cells was reported (Peng et al. 2018).
At nontoxic concentrations, a prenylated flavonoid sophoraflavanone M 101 isolated from S. flavescens (as an anti-inflammatory herb), reduced LPS-induced production of inflammatory mediators IL-6, NO, TNF-α, and MCP-1 in mouse primary peritoneal macrophages. The compound inhibited two important inflammatory signaling pathways, NF-κB and JNK/AP-1 (Han et al. 2021).
Recently, the effect of sophoricoside 134 from the mature seeds of S. japonica on the allergic chronic inflammatory lung disorder asthma was investigated (Kim and Lee 2021). The compound 134 reduced the allergic and asthmatic symptoms by suppressing airway inflammation and antibody-antigen reaction in mouse models. It improved allergic asthma by suppressing mast cell activation and CD4+ T cell differentiation. This isoflavone glycoside 134 suppressed immune cell recruitments in the airway lumens of the lungs and production of pro-inflammatory cytokines in the bronchoalveolar lavage fluid of ovalbumin (OVA)-induced mice. It also decreased the amounts of histamine and arachidonic acid metabolites released in OVA-induced mice and antibody-antigen stimulated mast cells. In addition, compound 134 decreased differentiation of naïve CD4+ T cells into T helper type 1 (Th1), Th2, and Th17 cells.
The anti-allergic and anti-inflammatory actions of herbal combination four herbs including S. flavescens was recorded (Yoo et al. 2019). Strong anti-degranulation actions were recorded and the formation of prostaglandin D2, interleukin-4, tumor necrosis factor-α, and leukotriene C4 was inhibited.
The isoflavone glycoside sophoricoside 134 inhibited the bioactivities of interleukins (IL-3, 5 and 6), and granulocyte–macrophage colony-stimulating factor in BAF/BO3, Y16, MH60/BSF-2, and TF-1 cells. IL-1β and α-tumor necrosis factor in A375.S2 and WEHI-164 cells were not affected (Kim et al. 2003).
Prenylated flavonoids and 2-arylbenzofuran (194) could be responsible for the anti-allergic effect of S. tonkinensis by inhibiting IL-6 production (Boozari et al. 2019b; Zhang et al. 2021b). S. davidii stems and leaves affected the ear and toe swelling of the mice. They could reduce the capillary permeability of the mice and decrease the number and the duration of itching (Yoo et al. 2019). Aloperine (284) has been used widely in clinical settings to treat inflammatory diseases, including allergic dermatitis (Ye et al. 2020). The anti-inflammatory and anti-allergy effects of compounds derived from Sophora sp are summarized in Table 19.
Anti-parasite
Most of the species of genus Sophora (approximately 70 species) are distributed in tropical and temperate zones and serve as pesticides and/or nectariferous plants (Zhou et al. 2020). S. flavescens exhibited an anti-protozoal activity (in vitro) as it inhibits Toxoplasma gondii proliferation at lower concentrations (Youn et al. 2003). S. flavescens exhibited an anti-neosporal activity to inhibit parasite proliferation, colonies containing Neospora caninum tachyzoites. The isoflavone was used to investigate the antileishmanial and trypanocidal compounds. Genistein (120) displayed a promising antileishmanial activity (IC50 4.2 μM) and selectivity (IC50 32.9 μM, versus Vero cells).
Many prenylated isoflavanones sophoronol (158), tetrapterol C-E (148–150), tetrapterol G-I (151–153) and tetrapterol F (159) were isolated from the roots of S. mollis and exhibited anitplasmodial activity against the CQS D10 strain of Plasmodium falciparum (de Andrade et al. 2020; Li et al. 2020a; Ma et al. 2020b). Sophoronol C 104 and sophoronol E 106 exhibited moderate anitplasmodial activity, with IC50 values of 12.9 and 12.8 μM, respectively. A fraction of PSm seeds has oxysophocarpine (236) and oxymatrine (249) as the main ingredients which showed no anti-parasite effect. While the activity of another fraction has the low polarity compounds; sophocaprine (235) and matrine (246) as the main phytoconstituents, were evaluated. The fraction presented stronger protoscolicidal activity in vivo and obvious inhibiting effects against growth of Echinococcus granulosus cyst (Luo et al. 2018). It was suggested that the oxygen group in the alkaloid molecule might affect the activity of PSm against echinococcosis in experimentally protoscolex-infected mice.
The major phytoconstituents of phytoextracts of S. alopecuroides PSa and S. flavescens PSf are sophocarpine (235), matrine (246), and anagyrine (273). These alkaloids presented high mortality against the third-instar larvae of Aedes albopictus (Shoukat et al. 2020). Additionally, PSa extract was more effective than a single active alkaloid due to the synergism of its active ingredients, which may be effective in managing the resistant population of Ae. albopictus. A concentration-dependent manner was recorded, as larval mortality increased with the increase in the PSa concentration and its constituents (235 and 246). Maximum mortality was achieved at higher concentrations (Shoukat et al. 2020).
Sophocarpine (235) and matrine (246); isolated from S. alopecuroides, are the main constituents for the broad bioactivities on insect pests, especially on aphids (Ma et al. 2020b). The acaricidal bioactivity of an oxymatrine 249-based commercial formulation was assessed against a pest in coffee cropping systems (Oligonychus ilicis) (de Andrade et al. 2020). QAs are widely used insecticides against citrus psyllids. Among these alkaloids, the dominant alkaloids sophocarpine (235) and sophoridine (240) were reported as the alkaloids repelled Asian citrus psyllid (Rizvi et al. 2019). The aphicidal action of sophocarpine (235) and matrine (246); isolated from S. alopecuroides, was reported and they have certain similarities to that of avermectin, a positive control (Ismail et al. 2020; Ma et al. 2020b). The lupin alkaloid (246) and its unsaturated derivatives 235 had intense nematicidal activity against nematodes. Sophoramine (243) had such activity, but it was less than that of 235 (Zhang et al. 2020a, 2021a, b, c; Ma et al. 2020b). Aso, cytisine-type alkaloids; N-methyl cytisine (268) and anagyrine (273) alkaloids were more active against the nematodes than the lupin-type alkaloids; sophocarpine 235, matrine 246 (Wang et al. 2019a; de Andrade et al. 2020; Huang and Xu 2020a; Ismail et al. 2020; Ma et al. 2020b). The nematocidal effect of aqueous extract of roots and stems of S.mollis against root-knot nematodes (Meloidogyne incognita) was reported (Ismail et al. 2020).A study confirmed the inhibition effect of S. flavescens ethanol extract on Phytophthora nicotianae (a pathogen of tobacco black shank disease) which cause huge economic losses each year (Li et al. 2020b). This effect could be achieved by inhibiting the production of sporangia and the release of zoospore release.
The quinolizidine-type alkaloid 284 had significant nematocidal and insecticidal activities via binding to the nicotinic acetylcholine receptor (Zhou et al. 2020).
The cytotoxic activity of alkaloid fraction of leaves of S. secundiflora and S. tomentosa and isolated alkaloids was also studied using crystal violet assay against MCF-7 and HEPG-2 cell lines (Aly et al. 2021).
Using different concentrations and mortality rate, the insecticidal activity of 70% methanol extract of leaves of S. tomentosa, S. secundiflora, and the isolated alkaloids were assessed against 3rd instar larvae of Culex pipiens (Diptera: Culicidae) (Aly et al. 2021). Anagyrine (273) exhibited high insecticidal activity with LC50 value of 3.42 ppm after 24 h of exposure (Aly et al. 2021).
Antidiabetic activity
A flavonoid-rich extract of S. davidii (Franch.) Skeels exhibited antidiabetic activity via activation of AMP-activated protein kinase in KK-Ay mice (Huang et al. 2018b; Wang et al. 2019b; Yang et al. 2019). This extract promoted the glucose transporter 4 (GLUT-4) translocation and improved glucose uptake in L6 cells (Huang et al. 2018b). Several isoflavone glycosides isolated from the stem bark of S. japonica demonstrated inhibitory effects on aldose reductase in vitro (Aly et al. 2019). Among these compounds, daidzein (108), paratensein-7-O-glucoside (113), and puerol A 115 displayed inhibitory effects, with IC50 values of 3.2, 6.4, and 1.9 µM, respectively.
Flavonoid-rich extract from S. alopecuroides ameliorated dyslipidemia, hyperglycemia, and insulin resistance in diabetic mice (Lv et al. 2021). This was due to the activation of PKC/GLUT4 pathway and the regulation of PPARα and PPARγ expression in white adipose tissue and liver, thereby ameliorating dyslipidemia.
Flavanones were the main active principles responsible for GLUT-4 translocation activities of petroleum ether and the ethyl acetate fractions of the roots of Sophora davidii (Franch.) Skeels (Ma et al. 2021). The flavanones; davidones A-E 94–98 and acacetin 99 promoted GLUT-4 translocation by the range of 1.35–3.00 folds (Ma et al. 2021).
2-(2′,4′-Dihydroxyphenyl)-5,6-methylenedioxybenzofuran 192 and three 2-arylbenzofuran dimers; Shandougenine A-C (193–195) were obtained from the ethyl acetate fraction of S. davidii (Franch.) Skeels (Li et al. 2021b). These compounds were tested for their potential GLUT-4 translocation activities. L6 cell line, which stably expressed Myc-GLUT-4-mOrange, was used to evaluate these effects. The positive control was insulin (100 nM) and 2-arylbenzofurans showed weak activity. Isoluteolin (126), isolated from the same plant, exhibited the most potent GLUT-4 translocations with 1.60-fold enhancement (Li et al. 2021b). It is considered as an active antidiabetic constituent in the plants of genus Sophora.
A lavandulyl flavanone; leachianone A (20) from the roots of S. davidii had GLUT-4 translocation activities (Ma et al. 2021).
(+)-Lirioresinol-A 196 was obtained from the ethyl acetate fraction of S. davidii (Franch.) Skeels (Li et al. 2021b) showed moderate translocation activity, increasing GLUT-4 translocation by 1.39-fold. It could be considered as a potentially active anti-diabetic constituent in the plants of genus Sophora.
Isopimpinellin (215) isolated from S. interrupta has antidiabetic activity and inhibitory activity against the enzyme adenine phosphoribosyltransferase from Leishmania (Boozari et al. 2019b; Rammohan et al. 2020). Oxymatrine 249 reported to ameliorate diabetes-induced aortic endothelial dysfunction via the regulation of eNOS and NOX4 (Wang et al. 2019b).
Antioxidant activity
Owing to tremendous antioxidant potential (Ledoux et al. 2018; Guo et al. 2020), phytoextracts of Sophora are extensively used in numerous pharmaceutical formulations. Antioxidant films were prepared by mixing phytoextract of S. japonica (PSj) with Artemisia sphaerocephala gum (Guo et al. 2020). DPPH scavenging activity of the film increased significantly with PSj addition. Antioxidant activity of the edible flowers of S. viciifolia was reported (Lin et al. 2019a; Guo et al. 2020). Recently, Kaewdana et al. (2021) found that the ethanolic extract of S. exigua root could inhibit the superoxide anions, DPPH radicals, and hydroxyl radicals, with half-maximal inhibitory concentration (IC50) values of 129.78 ± 0.65, 24.63 ± 1.78, and 30.58 ± 1.19 μg mL−1, respectively.
Shandougenine B (194) is the first naturally occurring dimeric 2-arylbenzofuran with a novel C-3 C-3″ bond linkage and shandougenine A (193) is a unique dimeric 2-arylbenzofuran with a C-3-C5‴ bond linkage. The 2-arylbenzofuran (194) had a higher ABTS cation radical scavenging capacity and DPPH free radical than compound 174 (Boozari et al. 2019b).
C-3″ bond linkage and shandougenine A (193) is a unique dimeric 2-arylbenzofuran with a C-3-C5‴ bond linkage. The 2-arylbenzofuran (194) had a higher ABTS cation radical scavenging capacity and DPPH free radical than compound 174 (Boozari et al. 2019b).
Data suggested that the inhibition of macrophage NO production by these isoflavanones may, at least in part, be explained by their radical scavenging or reduction activity (Boozari et al. 2019b; Lin et al. 2019a). The prenylated chalcone, kuraridin (184) and prenylated flavonol kushenol (28), have more potent scavenging/inhibitory activities than the prenylated flavanones, sophoraflavanone G (19) and kurarinone (23) (Chen et al. 2019a). These prenylated flavonoids, isolated from S. flavescens, demonstrated significant inhibitory activities against intracellular ROS levels as well as nuclear factor-kappaB activation by tert-butylhydroperoxide (Farhadi et al. 2019). A study pointed out that flavonols have the high scavenging activity compared to flavanones and this can be related to the presence the C2=C3 double bond and the 3-OH group in the flavonol skeleton. The flavonol kushenol (28) presented the highest scavenging activity like that of the positive control, L-ascorbic acid followed by kuraridin (184) (Chen et al. 2019a). The flavanones sophoraflavanone G (19), leachianone A (20), kurarinone (23), and kurarinol (29) were moderately effective scavengers, with inhibition percentages less than 50 (at 200 µg/mL). Also, sophoraflavanone G (19), kurarinone (23) exhibited IC50 values of 5.26 and 7.73 µg/mL, respectively, in DPPH· scavenging assay (Boozari et al. 2019b; Cho et al. 2020).
2-Propenoic acid, 3-(3,4-dihydroxyphenyl)-octacosyl ester 320, isolated from S. mollis (Royle) Graham Ex Baker, demonstrated significant free-radical scavenging activity using the 2,2-diphenyl-1-picryl-hydrazyl-hydrate (DPPH) free-radical assay. The recorded inhibition percentage of DPPH was 95.646 ± 0.003, 94.766 ± 0.014, and 94.516 ± 0.011% at concentrations of 400, 200, and 100 μg mL−1, respectively (Quradha et al. 2021).
Miscellaneous activities
Malaria is a disease caused by Plasmodium parasites, which infect humans and female Anopheles mosquitoes. The lavandulyl flavanones; sophoraflavanone G (19), leachianone A (20), and methoxykurarinone (24), isolated from the roots of S. flavescens, showed moderate antimalarial activities (in vitro) against P. falciparum (Li et al. 2020a; Ma et al. 2020b; Kaewdana et al. 2021). Their -half-maximal inhibitory concentration (IC50) values were 2.6, 2.1, and 2.4 μM, respectively. Recently, the aqueous extract of S. exigua root at doses of 200, 400, and 600 mg/kg had a stronger antimalarial activity than the ethanolic extract (Kaewdana et al. 2021). The aqueous extract at 600 mg/kg exhibited 60.46% suppression of parasitemia (Uzor 2020).
The phytoextract of S. tonkinensis rhizoma (PSt) exhibited antidiarrheal activity. The extract (0.01–10 mg mL−1) has spasmolytic effect and inhibited the spontaneous contraction of rabbit jejunum in a concentration-dependent manner like verapamil (Li et al. 2019c). The relationship between PSt and L-type calcium channel, concentration–response relationship curves of cumulative calcium chloride were determined. The phytoconstituents of Sophora species contained terpenes, sterols, flavonoids, and alkaloids, and these substances are linked to the antidiarrheal activity (Boozari et al. 2019b; Li et al. 2019c). A combination of PSf extract and a compound from Stevia rebaudiana, stevioside improved the small intestinal lesion score in piglets, alleviate diarrhea and decrease rotaviral shedding in feces (Boozari et al. 2019b; Wang et al. 2019a).
The lipoidal matters were reported with in vitro cytotoxic activity towards HCT-116 carcinoma cell line (IC50-value of 97.00 and 38.76 μg mL−1 for S. secundiflora and S. tomentosa, respectively) using the MTT assay (Aly et al. 2020a). Palmitic acid 346 is the major saturated fatty acid in both lipoidal matters; it showed anti-inflammatory and selective cytotoxic activity towards the human leukemia cell line MOLT-4. Linolenic 349 bioactive lipophilic compounds in S. secundiflora acid in the lipoidal matter decreased the growth of transplanted prostate, colon, and breast cancer cells in vivo. Regarding the in vitro studies, compound 349 was able to inhibit growth and promote apoptosis of transplanted prostate, colon, and breast cancer cells. Linolenic acid 349-induced apoptosis of colon and breast cancer cells via a mitochondrial-mediated pathway.
The dominant compounds in the unsaponifiable matter of S. secundiflora were α-amyrin (230, 9.73%) and β-amyrin acetate (231, 55.20%). While n-nonacosane (362, 43.80%) and 2-methyltriacontane (364, 11.94% were the main components in S. tomentosa. In the saponifiable fraction, the content of saturated fatty acids identified in S. tomentosa (58.37%) was higher than S. secundiflora (29.0%). The percentage of unsaturated fatty acids identified in S. secundiflora (62.67%) was higher than S. tomentosa (34.51%). Methyl linolenate 349, 36.62%) and methyl palmitate (346, 40.02%) are the major compounds in S. secundiflora and S. tomentosa. The dominant compounds in the unsaponifiable matter of S. tomentosa were n-nonacosane (362, 43.80%) and 2-methyltriacontane (364, 11.94%). α-Amyrin (230, 9.73%) and β-amyrin acetate (231, 55.20%) were the main components in S. secundiflora (Aly et al. 2020a).
Phytol (347) is a cyclic diterpene and a member of branched-chain unsaturated alcohols with antioxidant activity related to antinociceptive activities (Aly et al. 2020a). α- and β-Amyrins are pentacyclic triterpenes with anti-inflammatory, antimicrobial, antioxidant, and anticancer properties. Linolenic acid (349, C18:3) is a polyunsaturated fatty acid called omega-3 fatty acid relative to its three double bonds (Boozari et al. 2019b; Aly et al. 2020a).
Many isoprenoid flavonoids, with particularly an isoprenyl and a lavandulyl group in backbone structures were isolated from S. flavescens and S. pachycarpa and S. japonica (Boozari et al. 2019a, b; Li et al. 2020a). The anti-ulcer activities of many phytoextracts of Sophora species (e.g. S. subprostrata, S. japonica and S. interrupta) were reported due to the presence of phytochemical constituents such as saponins and isoflavonoids (Boozari et al. 2019b; Farhadi et al. 2019; Chen et al. 2020b; Yang et al. 2020b, c). Genistein (120) and biochanin A (130) showed about 2.5–6.5 folds greater inhibitory effects on arachidonic acid and U46619 induced platelet aggregation. Irisolidone (132) was ∼22–40 folds stronger inhibitor than acetylsalicylic acid on arachidonic acid and U46619 induced aggregation (Kim and Yun-Choi, 2008; Chen et al. 2020d).
Three isoflavones: genistein (120), biochanin A (130) and irisolidone (132); and four were identified as isoflavone-glycosides: sissotrin (117), tectoridin 118, genistin (119), and sophorabioside (134) from the stems, fruits and leaves of S. japonica (Kim and Yun-Choi 2008; Chen et al. 2020d). The isoflavone-glycoside tectoridin 118 showed inhibitory activity on U46619 and arachidonic acid-induced aggregation (IC50 of 123.4 and 25.9 µM, respectively) which had an activity similar to that of genistein (120) and biochanin A (130), but less than that of irisolidone (132) (Kim and Yun-Choi 2008; Chen et al. 2020d).
The extract of S. alopecuroides reduced the acute opioid withdrawal symptoms (Kianbakht et al. 2020). It decreased the clinical opiate withdrawal scale score (at days 3 and 8 significantly) compared with the placebo and had good safety and tolerability. The activity may be attributed to its major alkaloids; sophoramine (243), sophocarpine (235) and matrine (246). Through regulating TGF-β/Smad signaling in rats, matrine (246) suppressed the pancreatic fibrosis (Liu et al. 2019b). The mechanisms of protective effects of oxymatrine (249), on damaged organs and tissues are mainly related to its anti-inflammatory, anti-oxidative stress, anti- or pro-apoptotic, anti-fibrotic, metabolism-regulation, and anti-nociceptive functions (Lan et al. 2020).
Accumulating evidence indicates that sophocarpine (235) and oxymatrine (249) may protect against myocardial ischemic damage. The protective effect of 235 on the cardiovascular system was reported. This compound ameliorates cardiac hypertrophy through the activation of autophagic responses (Lin et al. 2020).
The total alkaloids extract derived from S.alopecuroides L improved depression-like behaviors and depression-related indicators in mice (Zhang et al. 2021a). A recent study indicated that S. alopecuroides L.-derived alkaloids improved depression in mice through modulating gut microbiota (Zhang et al. 2021a). The results of HPLC–MS (positive ionization) for total alkaloids from the seeds of S. alopecuroides confirmed the presence of six main alkaloids. They were identified as, sophocarpine 235, sophoridine (240), oxysophoridine 242, matrine (246), oxymatrine (249), and lupanine 253. The role of the hippocampal PI3K/Akt/mTOR signaling of matrine 246 was reported (Wu et al. 2019a, b). Matrine 246 exerts antidepressant-like effects on mice. It ameliorates anxiety and depression-like behaviour by targeting hyperammonemia-induced neuroinflammation and oxidative stress in CCl4 model of liver injury (Li et al. 2019a; Zhang et al. 2020a).
Many therapeutics isolated from PSf are considered as alternative anti-caries agents such as quercetin (1), kaempferol (2) and sophoraflavanone G (19) due to their inhibitory activities against Streptococcus mutans biofilms and antibacterial properties against several strains of mutans streptococci (Farhadi et al. 2019; Wang et al. 2019a). The application of deep eutectic solvent on the extraction of rutin from S. japonica bud was reported (Peng et al. 2018). Another styudy ivestigatedusing a combination of near-infrared spectroscopy and chemometrics as a rapid, quantitative analysis of the rutin (3) proportion in differently processed products of S. japonica L (Chen et al. 2018d).
Phytoextracts: a structure–activity relationship
The structure elucidation and characterization of bioactive compounds supposed that the functionality is based on structural chemistry (Boozari et al. 2019b). Previous structural modification and structure–activity relationship (SAR) studies revealed that presence of lavandulyl or isoprenyl substitution in C-8 position of flavanone structure increased the cytotoxic and antibacterial effect (Farhadi et al. 2019). Prenylated flavonoids with hydroxyl (OH) groups at the C3 and C6 and prenyl moieties at C8 site can inhibit human carboxylesterase 2 that promote the metabolic activation of ester drugs and lipid metabolism (Mukai, 2018). Also, the relationship between their structure and antibacterial activity indicates the importance of OH group on the B ring. This group can control hydrophilicity (Farhadi et al. 2019). While the presence of prenyl or lavandulyl, as an aliphatic group, could contribute to the hydrophobicity at the A ring (Boozari et al. 2019b).
Most prenylated flavonoids with lavandulyl or isoprenyl moieties at C8 displayed potent antibacterial activity. However, the position of methoxyl groups in the skeletons of compounds 19, 20 and 24 contribute to the antimalarial activity as they did not show selective toxicity (Li et al. 2020a; Ma et al. 2020b). The isoflavone skeleton has been deserved to investigate antileishmanial and trypanocidal compounds (Boozari et al. 2019b).
Sophoraflavanone G (19), kurarinone (23), and kurarinol (29), are other flavanones from the phytoextract of S. flavescens roots have a strong tyrosinase inhibitory activity. The results of molecular modeling study have suggested that the terminal hydroxy function within the lavandulyl group was important for optimal binding. This group within kurarinol (29) is instrumental in the interaction with the enzyme (Farhadi et al. 2019). The presence of lavandulyl groups also attributed to the trypanocidal activity of prenylated flavonoids. Alopecurone G (18), sophoraflavanone G (19), leachianone A (20), kurarinone (23), and methoxykurarinone (24) showed a stronger activity compared to other prenylated flavonoids (Boozari et al. 2019b; Chang et al. 2019a). The presence of lavandulyl groups also attributed to the antibacterial activity (Farhadi et al. 2019). The additional active group of 8-lavandulyl facilitated the interaction between compound 19 (sophoraflavanone G) and bacterial cellular membranes, resulting in an enhancement of the membrane effect. Compound 19 equally reduced the fluidity of outer and inner layers of membranes compared with naringenin (worked on outer layers more than inner ones) (Farhadi et al. 2019). The isogeranyl at C‐8 and OH at 3, 2′, and 4′ on the A and B rings of prenylated flavonoid improved the effects of ampicillin or oxacillin against MRSA infection (Farhadi et al. 2019).
Sophoraflavanone G (19) and sophoraflavanone I (67) (stilbene residue on B ring of flavonoid structure) showed a cytotoxic activity against tumor cells (HSC‐2 and HSG). Also, alopecurones (stilbene residue on A ring of flavonoid structure) exhibited an inhibitory activity against multi-drug resistance associated protein 1 (Boozari et al. 2019b). The lavandulyl flavanones (19) and (24) suppressed glutamate-induced neurotoxicity and ROS generation in HT22 cells via the induction of the expression of heme oxygenase (HO)-1 and increased HO activity dose- and time-dependently. Despite structural similarities, other lavandulyl flavanones (20) and (23) did not stimulate HO-1 expression, ROS scavenging, or cytoprotection and thus they did not show protection (Chang et al. 2019a).
The flavonostilbenes alopecurone A (57), B (65), and D (59) are considered as therapeutic reversal agents against multidrug resistance-associated protein 1 (MDRP1). They had an inhibitory activity and dramatically increased the accumulation of 6-carboxyfluorescein diacetate and doxorubicin in MDRP1-transfected U-2 OS cells (Boozari et al. 2019a,b; Yang et al. 2019; Wang et al. 2020b). The beneficial protective effects of HO-1 in inflammation were mediated not only via enzymatic degradation of pro-inflammatory free heme but also via production of the anti-inflammatory compound’s bilirubin and carbon monoxide (Wang et al. 2018a).
A decoction of a Chinese herbal formula has been reported as a botanical remedy for treatment of skin inflammation (Chang et al. 2018). A resveratrol glucoside is considered as one of the core anti-C–C motif chemokine ligand 17 (CCL17) bioactive ingredients in a Chinese formula, comprises the rootstock of S. flavescens (Chang et al. 2018).
The prenyl/or lavandulyl group at the C-8 position, along with a 3,4′-dihydroxyl group, could play a predominant role in antidiabetic complications and anti‐Alzheimer activities (Boozari et al. 2019b; Chang et al. 2019a). A previous review analyzed the SAR of prenylated flavonoid and clarified that the substitution of the flavonoid ring system with prenylated groups increased the lipophilicity and confers upon the molecule a strong affinity to biological membranes (Boozari et al. 2019b).
A study suggested that the β-D-glucose substitute at C-3, in the structure of flavonoids isolated from S. davidii, was necessary for the anti- Tobacco Mosaic Virus (TMV) activity (Yang et al. 2019). The presence of 1,3-benzodioxole ring could lead to increasing the protective effects against TMV replication.
Recently, the effect of substitutions at isoflavone derivatives on the binding affinity against the human ACE2 (hACE2), targeting COVID-19, was investigated (Alesawy et al. 2021). It was found that the substitutions at 5-position with hydroxyl (genistein 120) and methoxy group (glycitein) increased the binding of isoflavones against hACE2, with an increased affinity of hydroxyl derivative (Alesawy et al. 2021). Isoflavonoids can strongly bind the hACE-2 and viral main protease Mpro with great binding modes.
Studies show that prenylation decreases the flavonoids transportation from intestinal epithelia and reduces their absorption (Boozari et al. 2019b; Chang et al. 2019a). Despite the lower absorption of prenylated flavonoids compared with flavonoids, higher accumulation was found in the liver and kidney and exerted strong biological activity.
Previous structural modification and structure–activity relationship (SAR) studies revealed that N12 modifications on 284 scaffolds could enhance the anti-HIV activity (Lv et al. 2020). N12 substitutions improved their EC50’s 1.5- to 2.5-fold compared to that of unmodified 284. The favorable modifications for the increased anti-HIV-1 activity include a spacer of 4 carbons in length attached to N12 with the other end linked to a para-substituted phenyl group through an amide bond.
A previous study compared the SAR of flavesine J (290) with flavesine G (287) revealed that the introduction of a pyridine ring to a carboxylic acid group at C-15 might enhance the antiviral activity against HBV. Also, the structure of alopecurine A (285, 14.1%) and alopecurine B (286, 46.0%) and the HBsAg inhibitory activity relationship implied that the cleavage of the C-5–C-6 bond could produce a positive effect on antiviral activity against HBV. Whereas the cleavage of the C6–C-7 bond seems to be unfavorable for this activity (Zhang et al. 2018b, c). SAR with respect to the delta-lactam moiety of the matrine-type alkaloids and nematicidal activity was examined (Zhang et al. 2020a).
The key bioactive constituents of many Sophora species that exhibited sedative, depressant, analgesic, hypothermic, anti-tumor, and antipyretic activities, were the quinolizidine-type alkaloids (Aly et al. 2019, 2020a, b, c, 2021; Wang et al. 2019a; Cao and He 2020; Gao et al. 2020). Quinolizidine alkaloids (QA) were chemically divided into matrine-, sparteine-, cytisine-, and aloperine- types alkaloids (Li et al. 2021a).
QA possess extensive pharmacological effects on immune system as well as the cardiovascular and the nervous systems (Ren et al. 2019). A chloro-containing matrine-type alkaloid alopecine D (298) was isolated from the seeds of S. alopecuroides. This compound, of unusual matrine-type alkaloids featuring with additional di -chloromethyl attached on the D ring displayed immunosuppressive activity (Wang et al. 2020b, c). The compound exhibitedinhibitory effects on LPS-induced B cells or the proliferation of ConA-induced T lymphocytes with IC50-value of 3.74 or 3.98 μM, respectively.
The toxicological action of sophocarpine (235) and matrine (246) is related to the regulation of glutamate and γ‐aminobutyric acid systems. It has certain similarities to that of avermectin, a positive control (Ma et al. 2020b). The lupin alkaloid (246) and its unsaturated derivative (235) had a strong nematicidal activity. Another unsaturated derivative, sophoramine (243), had such activity, but it was less than the activity of 235. The degree of unsaturation in the delta-lactam ring of these lupin alkaloids was necessary to enhibit pinewood nematodes (Ismail et al. 2020; Ma et al. 2020b; Zhang et al. 2020a). The crude fraction containing cytisine-type alkaloids; N-methyl cytosine (268) and anagyrine (273) was more active against the nematodes than the lupin-type alkaloids; sophocarpine (235), matrine (246) (Wang et al. 2019a; Chen et al. 2020a; Huang and Xu 2020a; Ma et al. 2020b).
Sophora species and pandemic coronavirus disease
Coronavirus disease 2019 (COVID-19) is an illness caused by a novel coronavirus called severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) (Zhang et al. 2020a, b, c; Majnooni et al. 2021). The ongoing COVID-19 pandemic has created an alarming situation due to its high morbidity and mortality rates, economic losses, and the threat to global health security. When the paper was submitted, about 220 countries and territories around the world have reported more than 188,616,093 million confirmed cases of SARS-CoV-2, including more than 4,065,804 deaths (https://www.worldometers.info/coronavirus/#countries). In Egypt, the total cases were 283,320 cases and 16,412 deaths. Infections caused by viruses pose a significant challenge due to their wide spectrum of clinical presentations (Zhou et al. 2020). The critical cases of coronaviruses are linked to aberrant immune response, severe acute respiratory infection, and thrombosis. In vitro antiviral assays of extracts of some medicinal plants revealed the potential of using S. subprostrata as an anti-SARS-CoV2 candidate for further clinical studies (Fuzimoto and Isidoro 2020; Ghoran et al. 2021).
Most of the active natural compounds with inhibitory activity towards various types of coronaviruses could inhibit SARS-CoV-2, are belongs to polyphenols and flavonoids (e.g. quercetin 1 and luteolin 4) (Orhan et al. 2020). Quercetin (1) was tested for its SARS-CoV-3CLpro inhibitory effect. The compound has reportedly inhibited cleavage activity of the 3CLpro in a cell-based assay in a dose-dependent manner (Majnooni et al. 2021).
Other medicinal plants that produce various phenolic compounds and sterols are distinguished for their beneficial effects against human coronavirus (HCoV)-229E (Alesawy et al. 2021; Ghoran et al. 2021).
The isoflavonoids are an important polyphenolic subclass of flavonoids with a skeleton based on a 3-phenylchroman structure and their antiviral activity was proved in several scientific reports (Boozari et al. 2019b; Farhadi et al. 2019). Some isoflavonoids (e.g. isoflavonoid genistein 120) could inhibit SARS-CoV-2 which target the human ACE2 (hACE2) (Alesawy et al. 2021). This compound (120) is the major isoflavonoid of soybean seeds, and it could inhibit herpes simplex virus HSV-2 (333 strain), HSV-1 (29R strain), and KOS strains) replications with IC50-values of 14.12, 7.76 and 14.02, respectively (Alesawy et al. 2021).
Phenols and polyphenols (flavonoids) attack viral proteins present in the viral membrane or inside the virus particle. Phenolics are active against free viral particles but not—or to a lesser degree—after a virus have entered a host cell. Another group of PSMs is directed against DNA or RNA. These are DNA intercalators such as emetine and other isoquinoline alkaloids, and quinoline alkaloids. These alkaloids can inhibit viral development and viral replication in cells, as shown for SARS-CoV and other viruses.
A coumarin isopimpinellin 215; from the leaf ethanol extract of Angelica keiskei (Apiaceae), targeting the SARS-CoV 3CLpro and PLpro by cell-based and cell-free cleavage assays possessed a deficient inhibition against SARS-CoV 3CLpro with 10% at 200 μM. While the isolated isobavachalcone (185) (IC50 = 39.4 ± 5.2 μM for cell-free, IC50 = 11.9 ± 2.8 μM for cell-based assay, selectivity index SI = 1.3) displayed competitive inhibition towards SARS-CoV 3CLpro with a dose-dependent manner. The same alkylated chalcone (185) isolated from the extract of the seed ethanol extract of Psoralea corylifolia L. (Fabaceae) exhibited a high inhibitory effect (IC50 = 15 μg mL−1) against SARS-CoV (papain-like) PLpro (Orhan et al. 2020).
The pterocarpan derivative, maackiain (172) was isolated from the seeds of S. alopecuroides (Tsai et al. 2020; Rong et al. 2020). Concerning the activity of different pterocarpan derivatives; it was noted that the compound which contained an additional tetrahydrofuran ring attached to the chromene ring showed a better binding affinity inside the human ACE2 (hACE2) than the compounds which contained free OH groups at the chromene ring (Alesawy et al. 2021).
Generally, different tested isoflavones and pterocarpans as COVID-19 agents in a screening study decreased the affinity against hACE-2 in descending order of isoflavone derivatives > pterocarpan derivatives (Alesawy et al. 2021; Ghoran et al. 2021).
Thus, to find natural compounds with inhibitory activity against SARS-CoV, many researchers evaluated several natural compounds, including flavonoids, sterols, and fatty acids, against the activity of SARS helicase. They used colorimetry-based ATP hydrolysis assay or a fluorescence resonance energy transfer-based double-strand DNA unwinding assay to evaluate the inhibitory activity (Orhan and Deniz 2020; Ghoran et al. 2021; Remali and Aizat, 2021).
Alkaloids are a group of naturally occurring chemical compounds that contain mostly basic nitrogen atoms. Various studies showed that quinolizidine alkaloid matrine 246 exhibited antiviral activities against coxsackievirus B3 in Vero cells and influenza H3N2 virus in MDCK cells (Liu et al. 2018b; Ma et al. 2018, 2020a, b; Ren et al. 2019; Wang et al. 2019a; Sun et al. 2020).
The mechanism of anti-coronavirus effect of S. flavescens Aiton could be illustrated from multiple pathways, such as matrine 246 alkaloids and type I interferon, ERK signal pathway, NF-κB signal pathway and PI3K/Akt signal pathway (Zou et al. 2019; Li and Yu 2020). It provided a reference for clinical treatment of coronavirus infection pneumonia and research and development of related drugs of S. flavescens. The extracts of this species inhibited coronavirus replication (in vitro) and reduced the intracellular viral RNA concentration (Remali and Aizat 2021). The profiles of S. flavescens were analyzed to obtain biomarkers detected in different root tissues using combining metabolomes and transcriptomes (Wei et al. 2021). The expression levels of different transcripts (AO-2 & 6, LYS-A1 & A2, PMT-1, 17, 34, and 35) were highly and positively correlated with alkaloids (e.g., sophoridine (240), oxymatrine (249), and matrine (246) content (Wei et al. 2021).
Another extract of S. subprostrata Chun could inhibit RdRp activity through decreasing the protein expression in murine coronavirus (Remali and Aizat 2021). The methanol extract of S. subprostrata radix decreased production, the intracellular viral RNA and protein expression of some coronaviruses-single-stranded, positive-strand RNA virus with a helical nucleocapsids such as mouse hepatitis virus and porcine epidemic diarrhea virus (Sun et al. 2018; Chen et al. 2020d; Remali and Aizat 2021). Of note, S. subprostrata radix constituents could contribute to the race for anti-COVID-19 chemotherapeutic discovery (Fuzimoto and Isidoro 2020; Ghoran et al. 2021). This herb Sophora radix showed inhibition effects against mouse hepatitis virus (MHV)-59/CoV (EC50 = 0.8 μg mL−1) than ribavirin (EC50 = 17.5 μg mL−1).
Recent animal and clinical studies revealed a therapeutic potential of quinolizidine alkaloid matrine 246 (from S. flavescens Radix) on COVID-19 patients (Ren et al. 2019; Wang et al. 2019a; Sun et al. 2020). The alkaloid 246 reduced IL-16, IL-10, and TNF-α levels and the viral load in lung tissues. Consequently, 246 can significantly shorten the time to viral clearance in COVID-19 patients, leading to earlier negative SARS-CoV-2 test. Concomitantly, S. flavescens Radix extract can moderately inhibit anti-SARS-CoV1 3CL protease (Luo et al. 2009).
The therapeutic effect of sodium chloride salt of 246 in a mouse pneumonia model infected with HCoV-229E (Choudhry et al. 2020; Jin et al. 2020). The intraperitoneal injection of alkaloid salt significantly decreased the pathological damage to the lung tissue and reduced the lung index. The percentage of T cells of CD4+ and CD8+, the number of B cells in peripheral blood, the production of TNF-α, IFN-γ, IL-6, IL-10, and the viral load in the lung was significantly low compared to those in the controls. Therefore, alkaloid salts have therapeutic effect on HCoV-229E-infected mice via a mechanism related to the regulation of immune function (Choudhry et al. 2020; Sun et al. 2020).
Network pharmacology and molecular docking study simulation demonstrated that 246 could interact with the three key target proteins Mpro, ACE2, and RdRp during the SARS-CoV-2 infection (Peng et al. 2020a).
Another quinolizidine alkaloid oxysophoridine (242) inhibited the viral replication in Vero-E6 cells at noncytotoxic concentrations (Batalha et al. 2021). In the last decades, the applications of gemcitabine as a potent antiviral and chemotherapy agent attracted growing attention. Gemcitabine exhibited in vitro activity against SARS-CoV-2, with EC50 = values of 1.24 µM (Zhang et al. 2020a, b, c; Batalha et al. 2021). A combination of gemcitabine with 242 had an antiviral effect against SARS-CoV-2 (Zhang et al. 2020a, b, c).
Oxysophoridine 242 exhibited a dose-dependent inhibition of 2019-CoV replication in infected cells similar to chloroquine, the reported reference drug (Zhang et al. 2020a, b, c). The compound 242 may have a broad-spectrum antiviral activity against RNA viruses. The EC50 value of this compound was 0.18 μM compared with 1.36 μM for chloroquine (Zhang et al. 2020a, b, c). The 50% cytotoxic concentration (CC50) value was > 40 μM. The selectivity index: SI (CC50/EC50), in Vero cells was higher than 222 compared with just above 30 for chloroquine (the positive control). Further studies are needed to investigate the antiviral mechanism against SARS-CoV-2, toxicological evaluation and the safety profile of compound 242. A dose-dependent inhibition of 242 on SARS-CoV-2 infectivity in Huh-7 cells was reported (Fielding et al. 2020; Zhang et al. 2020a, b, c).
Discussion and future perspectives
The present review summarizes the research progress regarding Sophora species, with a particular focus on the traditional uses, the phytochemical and biological characteristics. The trend in drug discovery aimed to promote the utilization of natural and traditional resources for contemporary health care, including food/diet therapy (Tsai et al. 2020). Sophora has been used for a long time in different standard medicine formulas as an alternative traditional medicine in many countries, particularly in China and South Korea. More than 85% of Chinese medical material originates from plants (Yao et al. 2021).
Pharmacological studies on crude extracts and pure metabolites provided details on its traditional uses, as Sophora has been effectively used traditionally for treating hematochezia, asthma, allergy, sores, inflammation, gastrointestinal hemorrhage, dysentery, clear heat, cool the blood and reduce swelling, soothing the sore throat, and cough (Boozari et al. 2019b; Li et al. 2021b).
The presented data stated that all the reported phytochemical and pharmacological studies focused mainly on some species such as S. flavescens and S. japonica (Fig. 1); however, the majority of Sophora species still need further investigations. The relative percentage of all published chemical and biological reports regarding Sophora species is shown in Fig. 1.
Additionally, the state of the art on Sophora chemistry provides considerable opportunities for future discoveries. Approximately 370 chemical constituents have been isolated from different Sophora species (Fig. 2). The distribution of the secondary metabolites among Sophora species is illustrated in Fig. 2. These metabolites belong to different classes, including alkaloids, flavonols and flavones, isoflavonoids, pterocarpans, stilbene oligomers, phenolic acids, and other phenolic compounds.
This review sheds light on the promising chemical compounds extracted from Sophora and their bioactivities. This could make these chemical compounds suitable candidates for further development through clinical trials. There are still scientific gaps needs to be filled regarding studying chemical compositions of different species and their therapeutic effects.
First, Alkaloids are unique to Sophora and showed valuable pharmacological activities. The total alkaloids of S. alopecuroides TASa and matrine 246 inhibited auto-inducer 2 in the biofilms of Staphylococcus epidermidis. Also, this TASa ameliorated murine colitis by regulating bile acid metabolism and gut microbiota and had a protective effect on colitis, associated with downstream pro-inflammatory mediators and inhibition of nuclear transcription factor κB (NF-κB) activation (Halim et al. 2019; Izdebska et al. 2019; Chen et al. 2020b). Therefore, future studies are required for precise isolation and identification of each alkaloid structure, other than the major compound 246, using advanced characterization techniques. Also, the antimicrobial, cytotoxic, antioxidant and enzyme inhibition potentials of the methanol extract of S.alopecuroides seeds need to be investigated for the identification and isolation of bioactive compounds. In future studies, we recommend further in vivo toxicity assays and clinical efficacies to further evaluate different biomedical properties of the bioactive compounds (Zahra et al. 2021).
Second, prenylated isoflavonoids and matrine 246-based alkaloids in addition to lavandulyl flavanones and phenolic acids were mainly isolated from the root and rhizome parts of Sophora species. However, screening other parts may provide more chances for finding new bioactive substances, such as matrine-acetophenone alkaloid 281. This alkaloid with an unusual skeleton is mainly isolated from the seeds and roots of Sophora organs. While the first example of sparteine-indolizine (282) and the epimeric normatrine-julolidine alkaloids with uncommon skeletons (283a and 283b) were isolated from the roots of S. flavescens and S. alopecuroides, respectively.
The class of isoflavonoids could prevent and cure some diseases. The various therapeutic effects of prenylated isoflavonoids were described in other reviews primarily through several biochemical, cellular, and molecular mechanisms (Ahn et al. 2019). However, more standardization and documentation are needed for the isolation and identification of the prenylated isoflavonoids to validate their health benefit claims. Moreover, Prenylated isoflavonoids exhibited various pharmacological properties, including anticancer, anti-inflammatory, and antioxidant activities (Boozari et al. 2019b; Cho et al. 2020). Further studies could contribute to a new era of prenylated isoflavonoids (isolated from genus Sophora)-based medicinal and nutraceutical agents for the treatment of oxidative stress mediated diseases.
Third, in the last two decades, prenylated coumarins displayed promising and effective pharmacological properties, including antimicrobial, anti-inflammatory, and antioxidant activities (Lin et al. 2014; Hassanein et al. 2020). However, more research is needed to isolate and identify the prenylated coumarins from other Sophora species other than S. interrupta (the species characterized by the presence of di-C-prenylated coumarin 214) (Rammohan et al. 2015).
Fourth, the studies on oligostilbenes, pterocarpans and triterpenes are scarce when compared with the studies conducted on the isoflavonoids, flavonols, flavones and alkaloids. While the investigation of benzofuran derivatives, steroids, phenolic acids and chalcones compounds is still in the initial stage. Thus, more bioactive components could be identified using bioactivity guided isolation strategies.
Fifth, Fig. 3 illustrates the relative percentage of the secondary metabolites isolated from each Sophora species under investigation.
Figure 4 illustrates the type and the relative percentage of each chemical class isolated from Sophora species as a guide for further in-depth phytochemical scanning. In the current review, we discussed the structure–activity relationships SAR (Huang and Xu 2020a; Li et al. 2020d).
These results indicated that S. flavescens and S. alopecuroides are the main biologically explored species ((Boozari et al. 2019b; Wang et al. 2020b), while other species such as; S. mollis, S. tomentosa, S. interrupta, and S. moorcroftiana are insufficiently chemically studied. Therefore, more studies are needed for better understanding their chemical structures and properties to explain their biological activities.
Furthermore, there are still several gaps in our understanding of the applications of different Sophora species, despite many performed pharmacological studies on their medicinal importance. Although a large dose compound S. flavescens Ait injection is safe and effective to treat the advanced malignant tumors (recommended dose of 20 mL/day), some in vitro and in vivo studies were conducted at high doses for a clinical study (Ao et al. 2019; Kianbakht et al. 2020). For example, doses of Sophora extracts that were applied to evaluate antipruritic, anti-inflammatory effects (administrated personally at 200 mg kg −1 of extract in mice) were too high for the application in clinical studies (Aly et al. 2019; Yoo et al. 2019). The methanol extracts of S. flavescens significantly inhibited a serotonin-induced itch-related response (scratching), a mouse model of atopic dermatitis. The plant and its constituents affected acute and chronic pruritus and could be considered as new antipruritic agents.
The different pharmacological activities performed on Sophora species are illustrated in Fig. 5. The collected data indicated extensive pharmacological studies of some species e.g. S.alopecuroides, S. flavescens and S. tomentosa, while other species such as S. chrysophylla, S. exigua and S. viciifolia remain insufficiently studied. Other species e.g. S. franchetiana S. griffithii, S. korensis and S. macrocarpa, were pharmacologically not reported till now.
Sixth, many species of Sophora were tested as anti- herpesviruses (Zhou et al. 2010; Hassan, 2020). Herpesviruses are one of the most important viruses that infect humans and animals. The correlation between herpesviruses and coronaviruses is limited to the induced complications following the infections (Hassan 2020). An attention to some natural anti-herpesvirus alkaloid compounds, which have recently been proven to have excellent inhibitory efficacy against SARS-CoV-2 replication, was drawn by Hassan (2020). This was an attempt to explore various treatment options to combat COVID-19 based on available drugs with antiviral properties. Total alkaloids extracted from S. alopecuroides (TASa) were useful in the treatment of oral herpes catarrhalis, dental ulcer and herpes zoster (Zhou et al. 2010). To obtain the best performance, several factors affecting the encapsulation efficiency of TASa binary ethosome were investigated. A transmembrane pH gradient active loading method to prepare TASa binary ethosomes was developed (Zhou et al. 2010). The external preparation is limited by its low percutaneous penetration and bitter taste.
A combination of gemcitabine with oxysophoridine 242 had an additive antiviral effect against SARS-CoV-2 (Zhang et al. 2020a, b, c). Gemcitabine has been reported to block infection of diverse DNA and RNA viruses. Its activity against SARS-CoV-2 has been recently reported in Vero E6 and Huh7 cells. Gemcitabine as an antiviral drug, is not yet extensively investigated, particularly in terms of SAR or combination with other alkaloids (Zhang et al. 2020a, b, c). Overall, little is known about the antiviral mode of action of oxysophoridine 242; thus, future studies are needed to determine how this alkaloid interferes with coronaviral replication (Fielding et al. 2020).
A plant gel containing S. alopecuroides extracts (as an antiviral agent) was prepared. A patent has described the use of this gel as the external preparation for treating herpes zoster (Google patents CN102100734A). The gel can relieve the pain and has less side effects.
The gut microbes are closely related to depression as recently suggested (Zhang et al. 2021a). The depression in both patients and model animals significantly altered the gut microbiota. A recent study indicated that S. alopecuroides L.-derived alkaloids improved depression in mice through modulating gut microbiota (Zhang et al. 2021a). Therefore, further studies are needed to illustrate the activity of other Sophora species.
Seventh, a study was conducted to develop and evaluate chitosan/gelatin-based S. gibbosa extract-loaded microemulsion as a wound dressing (Shalaby et al. 2021). The dressing displayed superior wound repair compared to the control in terms of histological examination, proliferating cell nuclear antigen determination and alpha-smooth muscle actin (Shalaby et al. 2021). Thus, this natural product-loaded microemulsion-impregnated gelatin/chitosan could be a potential candidate for wound healing preparations.
Eighth, the potential economic use of Sophora species for the enhance crop yield and quality was suggested. In an effort to find growth-promoting strains that could be applied to enhance crop yield and quality, the fungal endophytes isolated from S. flavescens is gaining attention (Turbat et al. 2020). The symbiotic effects of endophytic fungi in association with their host plant can improve plant growth and reduce the adverse effects of both biotic and abiotic stresses. The plant growth-promoting activities of endophytic fungi isolated from various parts of S. flavescens have been revealed.
Nineth, a study demonstrated the use of S. flavescens in antimicrobial nanoparticles coated onto carbon fiber (CF) filters (Sim et al. 2014). Recent studies are now focusing on nanosized materials and their antimicrobial activities; this could facilitate the preparation of CF with reduced bio-contamination (Haroun et al. 2017; Rizk et al. 2018). This may be a scientific basis for controlling both gaseous and bioaerosol pollutants using antimicrobial CF filters coated with S. flavescens nanoparticles. The efficiency of Staphylococcus epidermidis inactivation increased with the concentration of S. flavescens nanoparticles in the activated CF filter coating. Reported information focused on the use of other species nanoparticles and their pharmacological aspects are scarce.
Tenth, the promising results confirmed by animal models should be further investigated by clinical studies. For example, SKI3301 (a purified herbal extract from S. tonkinensis), inhibited airway inflammation and bronchospasm in allergic asthma animal models (in vivo) (Yoo et al. 2017). The novel herbal cocktail (F-PASA) with anti-allergic properties which was also proved by investigating the IgE/antigen-mediated allergic responses in RBL-2H3 cells and passive cutaneous anaphylaxis in mice (Yoo et al. 2019). Other preparation like compound Kushen injection has combined with chemotherapy on postoperative patients with breast cancer as plant-derived anticancer agents (Ao et al. 2019). The extract of S. alopecuroides also reduced acute opioid withdrawal symptoms (Kianbakht et al. 2020). It decreased the clinical opiate withdrawal scale score and seems to be safe and tolerable.
Conclusions and further needs
Sophora species (family Fabaceae) are intriguing medicinal plants and rich sources of diverse and chemical structures especially in terms of the alkaloids and prenylated flavonoids. The claimed health benefits of these classes of compounds include anti-inflammatory, antiviral, anti-cancer, and other promising effects that were thoroughly summarized. However, although the current evidence is promising, further clinical studies are needed to evaluate the role of these compounds to support human health. Besides, the mechanisms by which they confer the health benefits, the bioavailability studies on Sophora’s alkaloids and prenylated flavonoids are also scarce. Furthermore, Sophora species are characterized by rare and unique skeletons of matrine-acetophenone and normatrine-julolidine alkaloids type and the 2-arylbenzofuran dimers. Biologically, Sophora species naturally synthesize secondary metabolites with outstanding biological potential, especially anti-inflammatory and anti-cancer activities. Sophora species are widely used in folk medicine, but they still deserve to be investigated for their clinical and medicinal properties. The current review provides details on some Sophora species that need further phytochemical and/ or pharmacological investigations. Further studies on this genus are recommended to study other Sophora species and to better correlate the bioactivity and chemical properties of these species. Moreover, S. subprostrata constituents could be considered as a candidate for future clinical trials to develop effective chemical drugs against SARS-CoV-2.
References
Abd-Alla HI, Moharram FA, Gaara AH, El-Safty MM (2009) Phytoconstituents of Jatropha curcas L. leaves and their immunomodulatory activity on humoral and cell-mediated immune response in chicks. Z Naturforsch 64C:495–501. https://doi.org/10.1515/znc-2009-7-805
Ahn DK (1998) Illustrated Book of Korean Medicinal Herbs. Kyo-Hak Publisher, Seoul, p 199
Ahn J, Kim YM, Chae HS, Choi YH, Ahn HC, Yoo H, Kang M, Kim J, Chin YW (2019) Prenylated flavonoids from the roots and rhizomes of Sophora tonkinensis and their effects on the expression of inflammatory mediators and proprotein convertase subtilisin/kexin type 9. J Nat Prod 82(2):309–317. https://doi.org/10.1021/acs.jnatprod.8b00748
Alesawy MS, Abdallah AE, Taghour MS, Elkaeed EB, Eissa H, Metwaly AM (2021) In silico studies of some isoflavonoids as potential candidates against covid-19 targeting human ACE2 (hACE2) and viral main protease (Mpro). Molecules 26(9):2806. https://doi.org/10.3390/molecules26092806
Aly HF, Abd-Alla HI, Ali SA, Alez RA, Abu-Krisha T, Mamdouh MM (2017) Bioinformatics: inflammatory cytokines and attenuation of diabetes hypercholesterolemia-induced renal injury using morning glory and necklace pod extracts. Asian J Pharm Clin Res 10:347–355. https://doi.org/10.22159/ajpcr.2017.v10i11.20119
Aly SH, Elissawy AM, Eldahshan OA, Elshanawany MA, Efferth T, Singab AN (2019) The pharmacology of the genus Sophora (Fabaceae): an updated review. Phytomedicine 64:153070. https://doi.org/10.1016/j.phymed.2019.153070
Aly HS, Elissawy AM, Eldahshan OA, Elshanawany MM, Singab AN (2020a) Phytochemical investigation using GC/MS analysis and evaluation of antimicrobial and cytotoxic activities of the lipoidal matter of leaves of Sophora secundiflora and Sophora tomentosa. Arch Pharm Sci Ain Shams Univ. 4(2):207–214. https://doi.org/10.21608/APS.2020.38371.1039
Aly SH, Elissawy AM, Eldahshan OA, Elshanawany MA, Singab AN (2020b) Variability of the chemical composition of the essential oils of flowers and the alkaloid contents of leaves of Sophora secundiflora and Sophora tomentosa. J Essent Oil-Bear Plants 23(3):442–452. https://doi.org/10.1080/0972060X.2020.1750489
Aly SH, Elissawy AM, Fayez AM, Eldahshan OA, Elshanawany MA, Singab AN (2020c) Neuroprotective effects of Sophora secundiflora, Sophora tomentosa leaves and formononetin on scopolamine-induced dementia. Nat Prod Res. https://doi.org/10.1080/14786419.2020.1795853
Aly SH, Elissawy AM, Allam AE, Farag SM, Eldahshan OA, Elshanawany MA, Singab AN (2021) New quinolizidine alkaloid and insecticidal activity of Sophora secundiflora and Sophora tomentosa against Culex pipiens (Diptera: Culicidae). Nat Prod Res. https://doi.org/10.1080/14786419.2021.1919108
Ao M, Xiao X, Li Q (2019) Efficacy and safety of compound Kushen injection combined with chemotherapy on postoperative patients with breast cancer: a meta-analysis of randomized controlled trials. Medicine 98:e14024. https://doi.org/10.1097/MD.0000000000014024
Awad HM, Abd-Alla HI, Mahmoud KH, El-Toumy SA (2014) In vitro anti-nitrosative, antioxidant, and cytotoxicity activities of plant flavonoids: a comparative study. Med Chem Res 23:3298–3307. https://doi.org/10.1007/s00044-014-0915-2
Awad HM, Abd-Alla HI, Ibrahim MA, El-Sawy ER, Abdalla MM (2018) Flavones from Heavenly Blue as modulators of Alzheimer’s amyloid-beta peptide (Aβ) production. Med Chem Res 27:768–776. https://doi.org/10.1007/s00044-017-2100-x
Batalha PN, Forezi LSM, Lima CGS, Pauli FP, Boechat FCS, de Souza MCBV, Cunha AC, Ferreira VF, da Silva FDC (2021) Drug repurposing for the treatment of COVID-19: Pharmacological aspects and synthetic approaches. Bioorg Chem 106:104488. https://doi.org/10.1016/j.bioorg.2020.104488
Bisby FA, Buckingham J, Harborne JB (eds) (1994) Phytochemical dictionary of the Leguminosae. CRC Press, Boca Raton
Boozari M, Ebrahimi SN, Soltani S, Tayarani-Najaran Z, Emami SA, Asili J, Iranshahi M (2019a) Absolute configuration and anti-cancer effect of prenylated flavonoids and flavonostilbenes from Sophora pachycarpa: possible involvement of Wnt signaling pathway. Bioorg Chem 85:498–504. https://doi.org/10.1016/j.bioorg.2019.01.051
Boozari M, Soltani S, Iranshahi M (2019b) Biologically active prenylated flavonoids from the genus Sophora and their structure–activity relationship—a review. Phytother Res 33:546–560. https://doi.org/10.1002/ptr.6265
Borges L, Bruneau A, Cardoso D, Crisp M, Delgado-Salinas A, Doyle JJ, Egan A, Herendeen PS, Hughes C, Kenicer G, Klitgaard B (2013) Towards a new classification system for legumes: progress report from the 6th International Legume Conference. S Afr J Bot 89:3–9. https://doi.org/10.1016/j.sajb.2013.07.022
Cai XH, Zhang HY, Xie B (2020) Matrine-family alkaloids: versatile precursors for bioactive modifications. Med Chem 16(4):431–453. https://doi.org/10.2174/1573406415666190507121744
Cao X, He Q (2020) Anti-tumor activities of bioactive phytochemicals in Sophora flavescens for breast cancer. Cancer Manag Res 12:1457–1467. https://doi.org/10.2147/CMAR.S243127
Cao Z, Chen L, Liu Y, Peng T (2018) Oxysophoridine rescues spinal cord injury via antiinflammatory, antioxidative stress and antiapoptosis effects. Mol Med Rep 17:2523–2528. https://doi.org/10.3892/mmr.2017.8170
Chang JB, Lane ME, Yang M, Heinrich M (2018) Disentangling the complexity of a hexa-herbal Chinese medicine used for inflammatory skin conditions—predicting the active components by combining lc-ms-based metabolite profiles and in vitro pharmacology. Front Pharmacol 9:1091. https://doi.org/10.3389/fphar.2018.01091
Chang HC, Liu KF, Teng CJ, Lai SC, Yang SE, Ching H, Wu CR (2019a) Sophora tomentosa extract prevents MPTP-induced parkinsonism in C57BL/6 mice via the inhibition of GSK-3β phosphorylation and oxidative stress. Nutrients 11:252. https://doi.org/10.3390/nu11020252
Chang Z, Zhang P, Zhang M, Jun F, Hu Z, Yang J, Wu Y, Zhou R (2019b) Aloperine suppresses human pulmonary vascular smooth muscle cell proliferation via inhibiting inflammatory response. Chin J Physiol 62(4):157–165. https://doi.org/10.4103/CJP.CJP_27_19
Chen Y, Yang Y, Wang F, Yang X, Yao F, Ming K, Yuan W, Zeng L, Liu J (2018a) Antiviral effect of baicalin phospholipid complex against duck hepatitis A virus type 1. Poult Sci 97(8):2722–2732. https://doi.org/10.3382/ps/pey155
Chen Y, Yang Y, Yuan W, Wang Z, Ming K, Zeng L, Liu J (2018b) Effects of Bush Sophora Root polysaccharide and its sulfate on DHAV-1 replication. Carbohydr Polym 197:508–514. https://doi.org/10.1016/j.carbpol.2018.06.039
Chen Y, Yuan W, Yang Y, Yao F, Ming K, Liu J (2018c) Inhibition mechanisms of baicalin and its phospholipid complex against DHAV-1 replication. Poult Sci 97(11):3816–3825. https://doi.org/10.3382/ps/pey255
Chen Y, Zhong Y, Qi Y, Qiu Y, Yang L, Liang S (2018d) Near-infrared spectroscopy for rapid evaluation of different processing products of Sophora japonica L. Spectr Lett 51(1):37–44. https://doi.org/10.1080/00387010.2017.1416478
Chen H, Yang J, Hao J, Lv Y, Chen L, Lin Q, Yuan J, Yang X (2019a) A novel flavonoid kushenol Z from Sophora flavescens mediates mTOR pathway by inhibiting phosphodiesterase and Akt activity to induce apoptosis in non-small-cell lung cancer cells. Molecules 24(24):4425. https://doi.org/10.3390/molecules24244425
Chen L, Chen L, Wan L, Huo Y, Huang J, Li J, Lu J, Xin B, Yang Q, Guo C (2019b) Matrine improves skeletal muscle atrophy by inhibiting E3 ubiquitin ligases and activating the Akt/mTOR/FoxO3α signaling pathway in C2C12 myotubes and mice. Oncol Rep 42(2):479–494. https://doi.org/10.3892/or.2019.7205
Chen Y, Chen L, Zhang JY, Chen ZY, Liu TT, Zhang YY, Fu LY, Fan SQ, Zhang MQ, Gan SQ, Zhang NL (2019c) Oxymatrine reverses epithelial-mesenchymal transition in breast cancer cells by depressing αVβ3 integrin/FAK/PI3K/Akt signaling activation. OncoTargets Ther 12:6253. https://doi.org/10.2147/OTT.S209056
Chen Z, Nishimura N, Okamoto T, Wada K, Naora K (2019d) Molecular mechanism of matrine from Sophora alopecuroides in the reversing effect of multi-anticancer drug resistance in K562/ADR cells. BioMed Res Int. https://doi.org/10.1155/2019/1269532
Chen F, Yin X, Wang Y, Lv Y, Sheng S, Ouyang S, Zhong Y (2020a) Pharmacokinetics, tissue distribution, and druggability prediction of the natural anticancer active compound cytisine n-isoflavones combined with computer simulation. Biol Pharm Bull. https://doi.org/10.1248/bpb.b20-00004
Chen L, Shao J, Luo Y, Zhao L, Zhao K, Gao Y, Wang S, Liu Y (2020b) An integrated metabolism in vivo analysis and network pharmacology in UC rats reveal anti-ulcerative colitis effects from Sophora flavescens EtOAc extract. J Pharm Biomed Anal 186:113306. https://doi.org/10.1016/j.jpba.2020.113306
Chen N, Qiao Z, Xu J, Wei K, Li M (2020c) Physiological mechanism and developmental events in differentiating floral buds of Sophora tonkinensis gagnep. Pharmacogn Mag 16(67):83. https://doi.org/10.4103/pm.pm_174_19
Chen Y, Luo Q, Li S, Li C, Liao S, Yang X, Zhou R, Zhu Y, Teng L, Chen H, Yang Y (2020d) Antiviral activity against porcine epidemic diarrhea virus of Pogostemon cablin polysaccharide. J Ethnopharmacol 259:113009. https://doi.org/10.1016/j.jep.2020.113009
Cho YR, Lee JH, Kim JH, Lee SY, Yoo S, Jung MK, Kim SJ, Yoo HJ, Pack CG, Rho JK, Son J (2018) Matrine suppresses KRAS-driven pancreatic cancer growth by inhibiting autophagy-mediated energy metabolism. Mol Oncol 12(7):1203–1215. https://doi.org/10.1002/1878-0261.12324
Cho BO, Che DN, Kim JS, Kim JH, Shin JY, Kang HJ, Jang SI (2020) In vitro anti-inflammatory and anti-oxidative stress activities of kushenol C isolated from the roots of Sophora flavescens. Molecules 25:1768. https://doi.org/10.3390/molecules25081768
Choudhry N, Zhao X, Xu D, Zanin M, Chen W, Yang Z, Chen J (2020) Chinese therapeutic strategy for fighting COVID-19 and potential small-molecule inhibitors against severe acute respiratory syndrome Coronavirus 2 (SARS-CoV-2). J Med Chem 63(22):13205–13227. https://doi.org/10.1021/acs.jmedchem.0c00626
Chung TW, Lin CC, Lin SC, Chan HL, Yang CC (2019) Antitumor effect of kurarinone and underlying mechanism in small cell lung carcinoma cells. OncoTarg Ther 12:6119. https://doi.org/10.2147/OTT.S214964
Dai M, Chen N, Li J, Tan L, Li X, Wen J, Lei L, Guo D (2021) In vitro and in vivo anti-metastatic effect of the alkaloid matrine from Sophora flavescens on hepatocellular carcinoma and its mechanisms. Phytomedicine 87:153580. https://doi.org/10.1016/j.phymed.2021.153580
Dang Z, Zhu L, Lai W, Bogerd H, Lee KH, Huang L, Chen CH (2016) Aloperine and its derivatives as a new class of HIV-1 entry inhibitors. ACS Med Chem Lett 7(3):240–2444. https://doi.org/10.1021/acsmedchemlett.5b00339
de Andrade DJ, da Rocha CM, de Matos ST, Zanardi OZ (2020) Oxymatrine-based bioacaricide as a management tool against Oligonychus ilicis (McGregor)(Acari: Tetranychidae) in coffee. Crop Protec 134:105182. https://doi.org/10.1016/j.cropro.2020.105182
Elberry A, Mufti S, Al-Maghrabi J, Ghareib S, Mosli H, El-Halawany A, Abdel-Sattar E (2020) The protective effect of Sophora japonica on prostatic hypertrophy and inflammation in rat. Inflammopharmacology. https://doi.org/10.1007/s10787-020-00723-5
Fan CL, Zhang YB, Chen Y, Xie P, Wang GC, Tian HY, Li YL, Huang XJ, Zhang XQ, Li ZY, Liu JS (2019) Alopecuroides A-E, matrine-type alkaloid dimers from the aerial parts of Sophora alopecuroides. J Nat Prod 82:3227–3232. https://doi.org/10.1021/acs.jnatprod.8b01081
Fang R, Wu R, Zuo Q, Yin R, Zhang C, Wang C, Guo Y, Yang AY, Li W, Lin L, Kong AN (2018) Sophora flavescens containing-QYJD formula activates Nrf2 anti-oxidant response, blocks cellular transformation and protects against DSS-induced colitis in mouse model. Am J Chin Med 46(07):1609–1623. https://doi.org/10.1142/S0192415X18500829
Fang H, Bo T, Zi X, Tan D, Liu X, Jiang C, Lei G, Deng Y, Cui L (2020) Sophocarpine exert protective effect against ox-LDL-induced endothelial damage via regulating NF-κB signaling pathway. Biosci Biotechnol Biochem 84(10):2104–2112. https://doi.org/10.1080/09168451.2020.1787813
Farhadi F, Khameneh B, Iranshahi M, Iranshahy M (2019) Antibacterial activity of flavonoids and their structure–activity relationship: an update review. Phytother Res 33:13–40. https://doi.org/10.1002/ptr.6208
Fielding BC, da Silva Maia Bezerra Filho C, Ismail NS, Sousa DP (2020) Alkaloids: therapeutic potential against human coronaviruses. Molecules 25(23):5496. https://doi.org/10.3390/molecules25235496
Fu S, Zhao N, Jing G, Yang X, Liu J, Zhen D, Tang X (2020) Matrine induces papillary thyroid cancer cell apoptosis in vitro and suppresses tumor growth in vivo by downregulating miR-182–5p. Biomed Pharmacother 128:110327. https://doi.org/10.1016/j.biopha.2020.110327
Fuzimoto AD, Isidoro C (2020) The antiviral and coronavirus-host protein pathways inhibiting properties of herbs and natural compounds—additional weapons in the fight against the COVID-19 pandemic? J Trad Compl Med 10(4):405–419. https://doi.org/10.1016/j.jtcme.2020.05.003
Gao DD, Zhang ZJ, Li HM, Chen XQ, Liu D, Li RT (2020) Four new quinolizidine alkaloids from the flowers of Sophora viciifolia. Phytochem Lett 36:91–94. https://doi.org/10.1016/j.phytol.2020.01.007
Ghoran SH, El-Shazly M, Sekeroglu N, Kijjoa A (2021) Natural products from medicinal plants with anti-human coronavirus activities. Molecules 26(6):1754. https://doi.org/10.3390/molecules26061754
Google patents (2011) Sophora alopecuroide total alkali medicine for treating herpes zoster Publication number CN102100734A, priority date 22.6.2011.
Gu J, Zhang Y, Wang X, Xiang J, Deng S, Wu D, Chen J, Yu L, Zhou Y, Wang Y, Shen J (2020) Matrine inhibits the growth of natural killer/T-cell lymphoma cells by modulating CaMKIIγ-c-Myc signaling pathway. BMC Complem Med Ther 20(1):1–1. https://doi.org/10.1186/s12906-020-03006-2
Guan Y, Chen K, Quan D, Kang L, Yang D, Wu H, Yan M, Wu S, Lv L, Zhang G (2021) The combination of Scutellaria baicalensis Georgi and Sophora japonica L. ameliorate renal function by regulating gut microbiota in spontaneously hypertensive rats. Front Pharmacol 11:2322. https://doi.org/10.3389/fphar.2020.575294
Guo Z, Wu X, Zhao X, Fan J, Lu X, Wang L (2020) An edible antioxidant film of Artemisia sphaerocephala Krasch gum with Sophora japonica extract for oil packaging. Food Packag Shelf Life 24:100460. https://doi.org/10.1016/j.fpsl.2019.100460
Hadadi Z, Nematzadeh GA, Ghahari S (2020) A study on the antioxidant and antimicrobial activities in the chloroformic and methanolic extracts of 6 important medicinal plants collected from North of Iran. BMC Chem 14:1–1. https://doi.org/10.1186/s13065-020-00683-5
Halim CE, Xinjing SL, Fan L, Vitarbo JB, Arfuso F, Tan CH, Narula AS, Kumar AP, Sethi G, Ahn KS (2019) Anti-cancer effects of oxymatrine are mediated through multiple molecular mechanism (s) in tumor models. Pharmacol Res 147:104327. https://doi.org/10.1016/j.phrs.2019.104327
Han J, Lee S, Lee N, Jo HJ, Park CS (2018) Relaxation of bladder smooth muscle by flavonoids from Sophora flavescens via direct activation of the large-conductance calcium-activated potassium channel. FASEB J 32:533–577. https://doi.org/10.1096/fasebj.2018.32.1
Han Y, Zhang X, Kang Y, Gao Y, Li X, Qi R, Cai R, Qi Y (2021) Sophoraflavanone M, a prenylated flavonoid from Sophora flavescens Ait., suppresses pro-inflammatory mediators through both NF-κB and JNK/AP-1 signaling pathways in LPS-primed macrophages. Europ J Pharmacol 9:174246. https://doi.org/10.1016/j.ejphar.2021.174246
Haroun AA, Elnahrawy AM, Abd-Alla HI (2017) Sol-gel preparation and in vitro cytotoxic activity of nanohybrid structures based on multi-walled carbon nanotubes and silicate. Inorg Nano-Metal Chem 47(7):1023–1027. https://doi.org/10.1080/24701556.2017.1284087
Hassan ST (2020) Shedding light on the effect of natural anti-herpesvirus alkaloids on SARS-CoV-2: a treatment option for COVID-19. Viruses 12(4):476. https://doi.org/10.3390/v12040476
Hassanein EH, Sayed AM, Hussein OE, Mahmoud AM (2020) Coumarins as modulators of the Keap1/Nrf2/ARE signaling pathway. Oxid Med Cell Longev 2020:1675957. https://doi.org/10.1155/2020/1675957
He LJ, Liu JS, Luo D, Zheng YR, Zhang YB, Wang GC, Li YL (2019) Quinolizidine alkaloids from Sophora tonkinensis and their anti-inflammatory activities. Fitoterapia 139:104391. https://doi.org/10.1016/j.fitote.2019.104391
Hong X, Zhong L, Xie Y, Zheng K, Pang J, Li Y, Yang Y, Xu X, Mi P, Cao H, Zhang W (2019) Matrine reverses the Warburg effect and suppresses colon cancer cell growth via negatively regulating HIF-1α. Front Pharmacol 10:1437. https://doi.org/10.3389/fphar.2019.01437
Hou M, Hu W, Hao K, Xiu Z (2020) Flavonoids and phenolic acids from the roots of Sophora tonkinensis Gagnep. Biochem Syst Ecol 89:104011. https://doi.org/10.1016/j.bse.2020.104011
Huang H, Wang Q, Du T, Lin C, Lai Y, Zhu D, Wu W, Ma X, Bai S, Li Z, Liu L (2018a) Matrine inhibits the progression of prostate cancer by promoting expression of GADD45B. Prostate 78(5):327–335. https://doi.org/10.1002/pros.23469
Huang Y, Hao J, Tian D, Wen Y, Zhao P, Chen H, Lv Y, Yang X (2018b) Antidiabetic activity of a flavonoid-rich extract from Sophora davidii (Franch.) Skeels in KK-Ay mice via activation of AMP-activated protein kinase. Front Pharmacol 9:760. https://doi.org/10.3389/fphar.2018.00760
Huang Y, Zhang J, Wang G, Chen X, Zhang R, Liu H, Zhu J (2018c) Oxymatrine exhibits anti-tumor activity in gastric cancer through inhibition of IL-21R-mediated JAK2/STAT3 pathway. Int J Immunopathol Pharmacol 32:2058738418781634. https://doi.org/10.1177/2058738418781634
Huang WC, Gu PY, Fang LW, Huang YL, Lin CF, Liou CJ (2019) Sophoraflavanone G from Sophora flavescens induces apoptosis in triple-negative breast cancer cells. Phytomedicine 61:152852. https://doi.org/10.1016/j.phymed.2019.152852
Huang X, Xu H (2020) Advances on the bioactivities, total synthesis, structural modification, and structure-activity relationships of cytisine derivatives. Mini Rev Med Chem 20(5):369–395. https://doi.org/10.2174/1389557519666191104121821
Huang XB, Yuan LW, Shao J, Yang Y, Liu Y, Lu JJ, Chen L (2020) Cytotoxic effects of flavonoids from root of Sophora flavescens in cancer cells. Nat Prod Res 11:1–6. https://doi.org/10.1080/14786419.2020.1712382
Huh JW, Lee JH, Jeon E, Ryu HW, Oh SR, Ahn KS, Jun HS, Ha UH (2020) Maackiain, a compound derived from Sophora flavescens, increases IL-1β production by amplifying nigericin-mediated inflammasome activation. FEBS Open Bio 10(8):1482–1491. https://doi.org/10.1002/2211-5463.12899
Ismail M, Fayyaz S, Kowsar A, Javed S, Ali I, Ali S, Hussain F, Ali H (2020) Evaluation of nematocidal effects of some medicinal plant extracts against root-knot nematodes (Meloidogyne incognita). Ital J Agron 15(1):63–69. https://doi.org/10.4081/ija.2020.1475
Izdebska M, Zielińska W, Hałas-Wiśniewska M, Mikołajczyk K, Grzanka A (2019) The cytotoxic effect of oxymatrine on basic cellular processes of A549 non-small lung cancer cells. Acta Histochem 121(6):724–731. https://doi.org/10.1016/j.acthis.2019.06.008
Jia F, Zhou Q, Li X, Zhou X (2019) Total alkaloids of Sophora alopecuroides and matrine inhibit auto-inducer 2 in the biofilms of Staphylococcus epidermidis. Microb Pathog 136:103698. https://doi.org/10.1016/j.micpath.2019.103698
Jia YQ, Yuan ZW, Zhang XS, Dong JQ, Liu XN, Peng XT, Yao WL, Ji P, Wei YM, Hua YL (2020) Total alkaloids of Sophora alopecuroides L. ameliorated murine colitis by regulating bile acid metabolism and gut microbiota. J Ethnopharmacol 255:112775. https://doi.org/10.1016/j.jep.2020.112775
Jiang JH, Pi J, Jin H, Yang F, Cai JY (2018) Chinese herb medicine matrine induce apoptosis in human esophageal squamous cancer KYSE-150 cells through increasing reactive oxygen species and inhibiting mitochondrial function. Pathol-Res Pract 214(5):691–699. https://doi.org/10.1016/j.prp.2018.03.015
Jiang D, Rasul A, Batool R, Sarfraz I, Hussain G, Mateen Tahir M, Qin T, Selamoglu Z, Ali M, Li J, Li X (2019a) Potential anticancer properties and mechanisms of action of formononetin. BioMed Res Int. https://doi.org/10.1155/2019/5854315
Jiang K, Guo S, Yang J, Liu J, Shaukat A, Zhao G, Wu H, Deng G (2019b) Matrine alleviates Staphylococcus aureus lipoteichoic acid-induced endometritis via suppression of TLR2-mediated NF-κB activation. Int Immunopharmacol 70:201–207. https://doi.org/10.1016/j.intimp.2019.02.033
Jiang J, Wang G (2020a) Matrine protects PC12 cells from lipopolysaccharide-evoked inflammatory injury via upregulation of miR-9. Pharm Biol 58(1):314–320. https://doi.org/10.1080/13880209.2020.1719165
Jiang JS, Shen Y, Feng ZM, Yang YN, Zhang X, Yuan X, Zhang PC (2020b) New benzoic acid glycosides from Sophora flavescens. J Asian Nat Prod Res. https://doi.org/10.1080/10286020.2020.1801650
Jiang P, Sun Y, Cheng N (2020c) Liver metabolomic characterization of Sophora flavescens alcohol extract-induced hepatotoxicity in rats through UPLC/LTQ-Orbitrap mass spectrometry. Xenobiotica 50(6):670–676. https://doi.org/10.1080/00498254.2019.1687962
Jin SJ, Yang Y, Ma L, Ma BH, Ren LP, Guo LC, Wang WB, Zhang YX, Zhao ZJ, Cui M (2017) In vivo and in vitro induction of the apoptotic effects of oxysophoridine on colorectal cancer cells via the Bcl-2/Bax/caspase-3 signaling pathway. Oncol Lett 14(6):8000–8006. https://doi.org/10.3892/ol.2017.7227
Jin X, Lu Y, Chen S, Chen D (2020) UPLC-MS identification and anticomplement activity of the metabolites of Sophora tonkinensis flavonoids treated with human intestinal bacteFan. J Pharm Biomed Anal 184:113176. https://doi.org/10.1016/j.jpba.2020.113176
Kaewdana K, Chaniad P, Jariyapong P, Phuwajaroanpong A, Punsawad C (2021) Antioxidant and antimalarial properties of Sophora exigua Craib. root extract in Plasmodium berghei-infected mice. Trop Med Health 49(1):24. https://doi.org/10.1186/s41182-021-00314-2
Kianbakht S, Hajiaghaee R, Akhondzadeh S (2020) Efficacy and safety of Sophora alopecuroides var. alopecuroides seed extract for opioid detoxification: a randomized, double-blind, and placebo-controlled clinical trial. Phytother Res 34(5):1108–1113. https://doi.org/10.1002/ptr.6578
Kim JM, Yun-Choi HS (2008) Anti-platelet effects of flavonoids and flavonoid-glycosides from Sophora japonica. Arch Pharm Res 31:886–890. https://doi.org/10.1007/s12272-001-1242-1
Kim BH, Lee S (2021) Sophoricoside from Sophora japonica ameliorates allergic asthma by preventing mast cell activation and CD4+ T cell differentiation in ovalbumin-induced mice. Biomed Pharmacother 133:111029. https://doi.org/10.1016/j.biopha.2020.111029
Kim BH, Chung EY, Ryu JC, Jung SH, Min KR, Kim Y (2003) Anti-inflammatory mode of isoflavone glycoside sophoricoside by inhibition of lnterleukin-6 and cyclooxygenase-2 in inflammatory response. Arch Pharm Res 26(4):306–311. https://doi.org/10.1007/BF02976960
Kim Y, Oh Y, Lee H, Yang B, Choi CH, Jeong H, Kim H, An W (2021) Prediction of the therapeutic mechanism responsible for the effects of Sophora japonica flower buds on contact dermatitis by network-based pharmacological analysis. J Ethnopharmacol 271:113843. https://doi.org/10.1016/j.jep.2021.113843
Krishna PM, KNV R, Sandhya S, BanjiI D (2012) A review on phytochemical, ethnomedical and pharmacological studies on genus Sophora, Fabaceae. Rev Bras Farmacogn 22(5). https://doi.org/10.1590/S0102-695X2012005000043
Kwon M, Ko SK, Jang M, Kim GH, Ryoo IJ, Son S, Ryu HW, Oh SR, Lee WK, Kim BY, Jang JH (2019) Inhibitory effects of flavonoids isolated from Sophora flavescens on indoleamine 2, 3-dioxygenase 1 activity. J Enzyme Inhib Med Chem 34(1):1481–1488. https://doi.org/10.1080/14756366.2019.1640218
Kwon M, Jang M, Kim GH, Oh T, Ryoo IJ, Ryu HW, Oh SR, Kim BY, Jang JH, Ko SK, Ahn JS (2020) Kushenol E inhibits autophagy and impairs lysosomal positioning via VCP/p97 inhibition. Biochem Pharmacol 175:113861. https://doi.org/10.1016/j.bcp.2020.113861
Lan X, Zhao J, Zhang Y, Chen Y, Liu Y, Xu F (2020) Oxymatrine exerts organ-and tissue-protective effects by regulating inflammation, oxidative stress, apoptosis, and fibrosis: from bench to bedside. Pharmacol Res 151:104541. https://doi.org/10.1016/j.phrs.2019.104541
Ledoux A, Cao M, Jansen O, Mamede L, Campos PE, Payet B, Clerc P, Grondin I, Girard-Valenciennes E, Hermann T, Litaudon M (2018) Antiplasmodial, anti-chikungunya virus and antioxidant activities of 64 endemic plants from the Mascarene Islands. Int J Antimicrob Agents 52:622–628. https://doi.org/10.1016/j.ijantimicag.2018.07.017
Li W, Lu Y (2018) Hepatoprotective effects of sophoricoside against fructose-induced liver injury via regulating lipid metabolism, oxidation, and inflammation in mice. J Food Sci 83(2):552–558. https://doi.org/10.1111/1750-3841.14047
Li Z, Yu KX (2020) Exploration on mechanism of anti-coronavirus of Sophora flavescens through multi-pathway. Chin Trad Herbal Drugs 2020:888–894. https://pesquisa.bvsalud.org/global-literature-on-novel-coronavirus-2019-ncov/resource/pt/covidwho-46903
Li L, Huang T, Lan C, Ding H, Yan C, Dou Y (2019a) Protective effect of polysaccharide from Sophora japonica L. flower buds against UVB radiation in a human keratinocyte cell line (HaCaT cells). J Photochem Photobiol B 191:135–142. https://doi.org/10.1016/j.jphotobiol.2018.12.001
Li Q, Zhang S, Wang M, Dong S, Wang Y, Liu S, Lu T, Fu Y, Wang X, Chen G (2019b) Downregulated miR-21 mediates matrine-induced apoptosis via the PTEN/Akt signaling pathway in FTC-133 human follicular thyroid cancer cells. Oncol Lett 18(4):3553–3560. https://doi.org/10.3892/ol.2019.10693
Li Y, Li J, Liu X, Zhang J, Mei X, Zheng R, Chen W, Zheng Q, Zhong S (2019c) Antidiarrheal activity of methanol extract of Sophora tonkinensis in mice and spasmolytic effect on smooth muscle contraction of isolated jejunum in rabbits. Pharm Biol 57:477–484. https://doi.org/10.1080/13880209.2019.1645701
Li Y, Lin H, Deng N, Xie L, Luo R (2019d) Matrine from Vietnamese Sophora root inhibits the growth of oral squamous cell carcinoma cells in vitro and in vivo. Food Sci Technol 39(4):855–858. https://doi.org/10.1590/fst.11518
Li JJ, Zhang X, Shen XC, Xu CY, Tan CJ, Lin Y (2020a) Phytochemistry and biological properties of isoprenoid flavonoids from Sophora flavescens Ait. Fitoterapia 143:104556. https://doi.org/10.1016/j.fitote.2020
Li Q, Dai ML, Xie Y, Yan CB, Liu JH, Zhang YP, Yao BF, Zhang YY (2020b) Inhibition of ethanol extracts of Psoralea corylifolia and Sophora flavescens on Phytophthora nicotianae and their compound effect. Int J Agric Biol 23(1):75–81. https://doi.org/10.17957/IJAB/15.1260
Li X, Liang T, Chen SS, Wang M, Wang R, Li K, Wang JC, Xu CW, Du N, Qin S, Ren H (2020c) Matrine suppression of self-renewal was dependent on regulation of LIN28A/Let-7 pathway in breast cancer stem cells. J Cell Biochem 121(3):2139–2149. https://doi.org/10.1002/jcb.29396
Li Y, Wang G, Liu J, Ouyang L (2020d) Quinolizidine alkaloids derivatives from Sophora alopecuroides Linn: Bioactivities, structure-activity relationships and preliminary molecular mechanisms. Eur J Med Chem 188:111972. https://doi.org/10.1016/j.ejmech.2019.111972
Li JC, Dai WF, Liu D, Zhang ZJ, Jiang MY, Rao KR, Li RT, Li HM (2021a) Quinolizidine alkaloids from Sophora alopecuroides with anti-inflammatory and anti-tumor properties. Bioorg Chem 110:104781. https://doi.org/10.1016/j.bioorg.2021.104781
Li K, Ma Y, Zhou T, Yang X, Choi HY (2021b) Chemical constituents from roots of Sophora davidii (Franch.) Skeels and their glucose transporter 4 translocation activities. Molecules 26(3):756. https://doi.org/10.3390/molecules26030756
Liang J, Chang B, Huang M, Huang W, Ma W, Liu Y, Tai W, Long Y, Lu Y (2018) Oxymatrine prevents synovial inflammation and migration via blocking NF-κB activation in rheumatoid fibroblast-like synoviocytes. Int Immunopharmacol 55:105–111. https://doi.org/10.1016/j.intimp.2017.12.006
Liang L, Wu J, Luo J, Wang L, Chen ZX, Han CL, Gan TQ, Huang JA, Cai ZW (2020) Oxymatrine reverses 5-fluorouracil resistance by inhibition of colon cancer cell epithelial-mesenchymal transition and NF-κB signaling in vitro. Oncol Lett 19(1):519–526. https://doi.org/10.3892/ol.2019.11090
Lin TT, Huang YY, Tang GH, Cheng ZB, Liu X, Luo HB, Yin S (2014) Prenylated coumarins: natural phosphodiesterase-4 inhibitors from Toddalia asiatica. J Nat Prod 77(4):955–962. https://doi.org/10.1021/np401040d
Lin BB, Liu X, Wu SQ, Zheng HX, Huo KK, Qi SS, Chen C (2019a) Phytochemicals content, antioxidant and antibacterial activities of Sophora viciifolia. Chem Biodivers 16(7):e1900080. https://doi.org/10.1002/cbdv.201900080
Lin G, Wu Y, Cai F, Li Z, Su S, Wang J, Cao J, Ma L (2019b) Matrine promotes human myeloid leukemia cells apoptosis through Warburg effect mediated by hexokinase 2. Front Pharmacol 10:1069. https://doi.org/10.3389/fphar.2019.01069
Lin YB, Huang DJ, Huang HL, Chen DX, Huang JH (2020) Sophocarpine ameliorates cardiac hypertrophy through activation of autophagic responses. Biosci Biotechnol Biochem 84(10):2054–2061. https://doi.org/10.1080/09168451.2020.1780111
Liu D, Chan BC, Cheng L, Tsang MS, Zhu J, Wong CW, Jiao D, Chan HY, Leung PC, Lam CW, Wong CK (2018a) Sophora flavescens protects against mycobacterial trehalose dimycolate-induced lung granuloma by inhibiting inflammation and infiltration of macrophages. Sci Rep 8(1):1–4. https://doi.org/10.1038/s41598-018-22286-w
Liu Y, Yao W, Si L, Hou J, Wang J, Xu Z, Li W, Chen J, Li R, Li P, Bo L (2018b) Chinese herbal extract Su-duxing had potent inhibitory effects on both wild-type and entecavir-resistant hepatitis B virus (HBV) in vitro and effectively suppressed HBV replication in mouse model. Antivir Res 155:39–47. https://doi.org/10.1016/j.antiviral.2018.04.017
Liu JS, Huo CY, Cao HH, Fan CL, Hu JY, Deng LJ, Lu ZB, Yang HY, Yu LZ, Mo ZX, Yu ZL (2019a) Aloperine induces apoptosis and G2/M cell cycle arrest in hepatocellular carcinoma cells through the PI3K/Akt signaling pathway. Phytomedicine 61:152843. https://doi.org/10.1016/j.phymed.2019.152843
Liu P, Zhu L, Zou G, Ke H (2019b) Matrine suppresses pancreatic fibrosis by regulating TGF-β/Smad signaling in rats. Yonsei Med J 60(1):79–87. https://doi.org/10.3349/ymj.2019.60.1.79
Liu J, Guo Y, Cao J (2020a) Matrine triggers colon cancer cell apoptosis and G0/G1 cell cycle arrest via mediation of microRNA-22. Phytother Res. https://doi.org/10.1002/ptr.6626
Liu ZM, Yang XL, Jiang F, Pan YC, Zhang L (2020b) Matrine involves in the progression of gastric cancer through inhibiting miR-93-5p and upregulating the expression of target gene AHNAK. J Cell Biochem 121(3):2467–2477. https://doi.org/10.1002/jcb.29469
Long GQ, Wang DD, Wang J, Jia JM, Wang AH (2020) Chemical constituents of Sophora flavescens Ait. and cytotoxic activities of two new compounds. Nat Prod Res. https://doi.org/10.1080/14786419.2020.1765340
Luo W, Su X, Gong S, Qin Y, Liu W, Li J, Yu H, Xu Q (2009) Anti-SARS coronavirus 3C-like protease effects of Rheum palmatum L. extracts. Biosci Trends 3(4):124–126. https://www.biosciencetrends.com/downloadpdf/227
Luo Y, Zhang G, Liu X, Yuan M, Gao Q, Gao H, Ke L, Zhang X, Shi Y, Ma X, Zhang L (2018) Therapeutic and immunoregulatory effects of water-soluble alkaloids E2-a from Sophora moorcroftiana seeds as a novel potential agent against echinococcosis in experimentally protoscolex-infected mice. Vet Res 49:100. https://doi.org/10.1186/s13567-018-0596-9
Lv XQ, Zou LL, Tan JL, Li H, Li JR, Liu NN, Dong B, Song DQ, Peng ZG (2020) Aloperine inhibits hepatitis C virus entry into cells by disturbing internalisation from endocytosis to the membrane fusion process. Eur J Pharmacol 883:173323. https://doi.org/10.1016/j.ejphar.2020.173323
Lv Y, Zhao P, Pang K, Ma Y, Huang H, Zhou T, Yang X (2021) Antidiabetic effect of a flavonoid-rich extract from Sophora alopecuroides L. in HFD-and STZ-induced diabetic mice through PKC/GLUT4 pathway and regulating PPARα and PPARγ expression. J Ethnopharmacol 268:113654. https://doi.org/10.1016/j.jep.2020.113654
Ma Q, Yu Q, Xing X, Liu S, Shi C, Luo J (2018) San Wu Huangqin decoction, a Chinese herbal formula, inhibits influenza a/PR/8/34 (H1N1) virus infection in vitro and in vivo. Viruses 10:11. https://doi.org/10.3390/v10030117
Ma QH, Ren MY, Luo JB (2020a) San Wu Huangqin decoction regulates inflammation and immune dysfunction induced by influenza virus by regulating the NF-κB signaling pathway in H1N1-infected mice. J Ethnopharmacol 264:112800. https://doi.org/10.1016/j.jep.2020.112800
Ma T, Shi X, Ma S, Ma Z, Zhang X (2020b) Evaluation of physiological and biochemical effects of two Sophora alopecuroides alkaloids on pea aphids Acyrthosiphon pisum. Pest Manag Sci. https://doi.org/10.1002/ps.5950
Ma Y, Zhou T, Zhao P, Choi HY, Hao J, Huang H, Wu C, Yang X, Pang K (2021) New flavonoids from the roots of Sophora davidii (Franch.) Skeels and their glucose transporter 4 translocation activities. Bioorgan Chem 106:104500. https://doi.org/10.1016/j.bioorg.2020.104500
Majnooni MB, Fakhri S, Bahrami G, Naseri M, Farzaei MH, Echeverría J (2021) Alkaloids as potential phytochemicals against SARS-CoV-2: approaches to the associated pivotal mechanisms. Evid Based Complement Altern Med 2021:632623. https://doi.org/10.1155/2021/6632623
Mao D, Pan X, Rui Y, Li F (2020) Matrine attenuates heterotopic ossification by suppressing TGF-β induced mesenchymal stromal cell migration and osteogenic differentiation. Biomed Pharmacother 127:110152. https://doi.org/10.1016/j.biopha.2020.110152
Mukai R (2018) Prenylation enhances the biological activity of dietary flavonoids by altering their bioavailability. Biosci Biotechnol Biochem 82(2):207–215. https://doi.org/10.1080/09168451.2017.1415750
Muhammad T, Sakhawat A, Khan AA, Huang H, Khan HR, Huang Y, Wang J (2020) Aloperine in combination with therapeutic adenoviral vector synergistically suppressed the growth of non-small cell lung cancer. J Canc Res Clin Oncol 146(4):861–874. https://doi.org/10.1007/s00432-020-03157-2
Nourmohammadi S, Aung TN, Cui J, Pei JV, De Ieso ML, Harata-Lee Y, Qu Z, Adelson DL, Yool AJ (2019) Effect of compound kushen injection, a natural compound mixture, and its identified chemical components on migration and invasion of colon, brain, and breast cancer cell lines. Front Oncol 9:314. https://doi.org/10.3389/fonc.2019.00314
Orhan IE, Deniz FS (2020) Natural products as potential leads against coronaviruses: could they be encouraging structural models against SARS-CoV-2? Nat Prod Bioprospect 10(4):171–186. https://doi.org/10.1007/s13659-020-00250-4
Pan QM, Li YH, Hua J, Huang FP, Wang HS, Liang D (2015) Antiviral matrine-type alkaloids from the rhizomes of Sophora tonkinensis. J Nat Prod 78(7):1683–1688. https://doi.org/10.1021/acs.jnatprod.5b00325
Parvez MK, Rehman MT, Alam P, Al-Dosari MS, Alqasoumi SI, Alajmi MF (2019) Plant-derived antiviral drugs as novel hepatitis B virus inhibitors: cell culture and molecular docking study. Saudi Pharm J 27:389–400. https://doi.org/10.1016/j.jsps.2018.12.008
Peng F, Xu P, Zhao BY, Zong MH, Lou WY (2018) The application of deep eutectic solvent on the extraction and in vitro antioxidant activity of rutin from Sophora japonica bud. J Food Sci Technol 55(6):2326–2333. https://doi.org/10.1007/s13197-018-3151-9
Peng W, Xu Y, Han D, Feng F, Wang Z, Gu C, Zhou X, Wu Q (2020a) Potential mechanism underlying the effect of matrine on COVID-19 patients revealed through network pharmacological approaches and molecular docking analysis. Arch Physiol Biochem. https://doi.org/10.1080/13813455.2020.1817944
Peng Z, Guan Q, Luo J, Deng W, Liu J, Yan R, Wang W (2020b) Sophoridine exerts tumor-suppressive activities via promoting ESRRG-mediated β-catenin degradation in gastric cancer. BMC Cancer 20(1):582. https://doi.org/10.1186/s12885-020-07067-x
Qiu M, Shi F, Dai F, Song R, Wang S, You Y, Zhao B (2018) A reactive oxygen species activation mechanism contributes to Sophoridine-induced apoptosis in rat liver BRL-3A cells. J Ethnopharmacol 213:376–383. https://doi.org/10.1016/j.jep.2017.10.030
Qiu M, Liu J, Feng P, Su Y, Guo R, Shi F, Wang S, Zhao B (2020a) Cytochrome P450s regulates aloperine-induced pathological changes in mouse liver and kidney. Res Vet Sci 132:97–100. https://doi.org/10.1016/j.rvsc.2020.06.005
Qiu M, Liu J, Su Y, Liu J, Wu C, Zhao B (2020b) Aloperine induces apoptosis by a reactive oxygen species activation mechanism in human ovarian cancer cells. Protein Pept Lett 27(9):860–869. https://doi.org/10.2174/0929866527666200320094313
Quradha MM, Khan R, Adhikari A, Rauf A, Rashid U, Bawazeer S, Al-Awthan YS, Bahattab O, Mubarak MS (2021) Isolation, biological evaluation, and molecular docking studies of compounds from Sophora mollis (Royle) Graham Ex Baker. ACS Omega 6(24):15911–15919. https://doi.org/10.1021/acsomega.1c01532
Rammohan A, Munikishore R, Gunasekar D, Deville A, Bodo B (2015) A new di-C-prenylated coumarin from Sophora interrupta. Nat Prod Res 29:82–85. https://doi.org/10.1080/14786419.2014.959010
Rammohan A, Bhaskar BV, Venkateswarlu N, Gu W, Zyryanov GV (2020) Design, synthesis, docking and biological evaluation of chalcones as promising antidiabetic agents. Bioorg Chem 95:103527. https://doi.org/10.1016/j.bioorg.2019.103527
Remali J, Aizat WM (2021) A review on plant bioactive compounds and their modes of action against coronavirus infection. Front Pharmacol. https://doi.org/10.3389/fphar.2020.589044
Ren G, Ding G, Zhang H, Wang H, Jin Z, Yang G, Han Y, Zhang X, Li G, Li W (2019) Antiviral activity of sophoridine against enterovirus 71 in vitro. J Ethnopharmacol 236:124–128. https://doi.org/10.1016/j.jep.2019.02.045
Rizk MZ, Abo-El-Matty DM, Aly HF, Abd-Alla HI, Saleh SM, Younis EA, Elnahrawy AM, Haroun AA (2018) Therapeutic activity of sour orange albedo extract and abundant flavanones loaded silica nanoparticles against acrylamide-induced hepatotoxicity. Toxicol Rep 5:929–942. https://doi.org/10.1016/j.toxrep.2018.08.021
Rizvi SA, Ling S, Tian F, Liu J, Zeng X (2019) Interference mechanism of Sophora alopecuroides L. alkaloids extract on host finding and selection of the Asian citrus psyllid Diaphorina citri Kuwayama (Hemiptera: Psyllidae). Environ Sci Pollut Res 26(2):1548–1557. https://doi.org/10.1007/s11356-018-3733-0
Rong ZJ, Hu GS, Lin SY, Yan T, Li N, Zhao Y, Jia JM, Wang AH (2020) Constituents from the seeds of Sophora alopecuroides L. Molecules 25(2):411. https://doi.org/10.3390/molecules25020411
Shalaby K, Mostafa EM, Musa A, Moustafa AE, Ibrahim MF, Alruwaili NK, Zafar A, Elmowafy M (2021) Enhanced full-thickness wound healing via Sophora gibbosa extract delivery based on a chitosan/gelatin dressing incorporating microemulsion. Drug Dev Ind Pharm 47(2):215–224. https://doi.org/10.1080/03639045.2020.1863420
Shen Y, Feng ZM, Jiang JS, Yang YN, Zhang PC (2013) Dibenzoyl and isoflavonoid glycosides from Sophora flavescens: inhibition of the cytotoxic effect of D-galactosamine on human hepatocyte HL-7702. J Nat Prod 76(12):2337–2345. https://doi.org/10.1021/np400784v
Shi J, Han X, Wang J, Han G, Zhao M, Duan X, Mi L, Li N, Yin X, Shi H, Li C (2019) Matrine prevents the early development of hepatocellular carcinoma like lesions in rat liver. Exp Ther Med 18(4):2583–2591. https://doi.org/10.3892/etm.2019.7875
Shim JG, Yeom SH, Kim HJ, Choi YW, Lee DI, Song KY, Kwon SH, Lee MW (2005) Bone loss preventing effect of Sophorae Fructus on ovariectomized rats. Arch Pharm Res 28(1):106–110. https://doi.org/10.1007/s12272-010-2240-8
Shoukat RF, Shakeel M, Rizvi SA, Zafar J, Zhang Y, Freed S, Xu X, Jin F (2020) Larvicidal, ovicidal, synergistic, and repellent activities of Sophora alopecuroides and its dominant constituents against Aedes albopictus. Insects 11:246. https://doi.org/10.3390/insects11040246
Sim KM, Kim KH, Hwang GB, Seo S, Bae GN, Jung JH (2014) Development and evaluation of antimicrobial activated carbon fiber filters using Sophora flavescens nanoparticles. Sci Total Environ 493:291–297. https://doi.org/10.1016/j.scitotenv.2014.06.002
Simpson MG (2010) 8—diversity and classification of flowering plants: eudicots. Plant Syst. https://doi.org/10.1016/B978-0-12-374380-0.50008-7
Song P, Chen H, Wen Y, Lv Y, Deng S, Yang X (2018) Cytotoxic compounds from the seeds of Sophora alopecuroides. Nat Prod Commun 13(8):1934578X1801300818. https://doi.org/10.1177/1934578X1801300818
Song P, Chen H, Huang Y, Wen Y, Hao J, Lv Y, Deng S, Yang X (2019) Phenolic acids from the seeds of Sophora alopecuroides. Chem Nat Compd 55(5):835–838. https://doi.org/10.1007/s10600-019-02826-0
Song Z, Gong X, Ou C, Zhang W, Wang J, Yao C, Qin S, Yan B, Li Q, Wei K, Hou X (2021) Chloroform extract from Sophora tonkinensis Gagnep. inhibits proliferation, migration, invasion and promote apoptosis of nasopharyngeal carcinoma cells by silencing the PI3K/AKT/mTOR signaling pathway. J Ethnopharmacol 271:113879. https://doi.org/10.1016/j.jep.2021.113879
Su G, Yang W, Meng W, Wu Q, Luo Y, Ma X, Zhao S, Xie X, Guo GQ (2018) Anti-proliferation effects of ethanolic extracts from Sophora moorcroftiana seeds on human hepatocarcinoma HepG2 cell line. Nat Prod Res 32:1472–1475. https://doi.org/10.1080/14786419.2017.1353503
Sun P, Sun N, Wang Z, Guo J, He Y, Li H (2018) Matrine inhibits replication of porcine reproductive and respiratory syndrome virus (PRRSV) by influencing the activation of Nsp9. Pak Vet J 38(4):359–364. https://doi.org/10.29261/pakvetj/2018.079
Sun J, Zhao RH, Guo SS, Shi YJ, Bao L, Geng ZH, Gao YJ, Liu J, Li Q, Cui XI (2020) Effect of matrine sodium chloride injection on a mouse model combining disease with syndrome of human coronavirus pneumonia with cold-dampness pestilence attacking the lung. Acta Pharm Sin 55(3):366–373. https://doi.org/10.16438/J.0513-4870.2020-0222
Sweelam HM, Abd-Alla HI, Abdelwahab AB, Gabr MM, Kirsch G (2018) Secondary metabolites and biological activity of Pentas species: a minireview. J Adv Res 10:21–30. https://doi.org/10.1016/j.jare.2017.12.003
Ti H, Zhuang Z, Yu Q, Wang S (2021) Progress of plant medicine derived extracts and alkaloids on modulating viral infections and inflammation. Drug Des Devel Ther 15:1385–1408. https://doi.org/10.2147/DDDT.S299120
Tsai RT, Tsai CW, Liu SP, Gao JX, Kuo YH, Chao PM, Hung HS, Shyu WC, Lin SZ, Fu RH (2020) Maackiain ameliorates 6-hydroxydopamine and SNCA pathologies by modulating the PINK1/Parkin pathway in models of Parkinson’s disease in Caenorhabditis elegans and the SH-SY5Y cell line. Int J Mol Sci 21(12):4455. https://doi.org/10.3390/ijms21124455
Turbat A, Rakk D, Vigneshwari A, Kocsubé S, Thu H, Szepesi Á, Bakacsy L, Škrbić D, Jigjiddorj EA, Vágvölgyi C, Szekeres A (2020) Characterization of the plant growth-promoting activities of endophytic fungi isolated from Sophora flavescens. Microorganisms 8(5):683. https://doi.org/10.3390/microorganisms8050683
ur Rashid H, Xu Y, Muhammad Y, Wang L, Jiang J (2019) Research advances on anticancer activities of matrine and its derivatives: an updated overview. Eur J Med Chem 161:205–238. https://doi.org/10.1016/j.ejmech.2018.10.037
ur Rashid H, Rasool S, Ali Y, Khan K, Martines MA (2020) Anti-cancer potential of sophoridine and its derivatives: recent progress and future perspectives. Bioorg Chem. https://doi.org/10.1016/j.bioorg.2020.103863
Uzor PF (2020) Alkaloids from plants with antimalarial activity: a review of recent studies. Evid-Based Compl Altern Med 2020:8749083. https://doi.org/10.1155/2020/8749083
Wang H, Tong Y, Li W, Zhang X, Gao X, Yong J, Zhao J, Koike K (2018a) Enhanced ultrasound-assisted enzymatic hydrolysis extraction of quinolizidine alkaloids from Sophora alopecuroides L. seeds. J Nat Med 72(2):424–432. https://doi.org/10.1007/s11418-017-1165-7
Wang H, Xiao D, Li W, Zhang X, Gao X, Yong J, Zhao J, Koike K (2018b) A simple and fast quantitative analysis of quinolizidine alkaloids and their biosynthetic precursor, lysine, in Sophora alopecuroides by hydrophilic interaction chromatography coupled with triple-quadrupole tandem mass spectroscopy. Phytochem Anal 29(5):500–506. https://doi.org/10.1002/pca.2757
Wang M, Liu G, Li H (2018c) Extraction of matrine from Sophora flavescens Ait. and evaluation of its inhibitory effects on human nasopharyngeal carcinoma CNE-2 cells. Food Sci Technol 38:333–337. https://doi.org/10.1590/fst.24117
Wang R, Wang S, Pan W, Li Q, Xia Z, Guan E, Zheng M, Pang G, Yang Y, Yi Z (2018d) Strategy of tobacco plant against black shank and tobacco mosaic virus infection via induction of PR-1, PR-4 and PR-5 proteins assisted by medicinal plant extracts. Physiol Molecul Plant Pathol 101:127–145. https://doi.org/10.1016/j.pmpp.2017.07.002
Wang X, Zheng R, Huang X, Mao Z, Wang N, Li H, Wen C, Shou D (2018e) Effects of alkaloids from Sophora flavescens on osteoblasts infected with Staphylococcus aureus and osteoclasts. Phytother Res 32(7):1354–1363. https://doi.org/10.1002/ptr.6069
Wang Y, Xia Z, Liu Z, Wan C, Luo X, Zhang L (2018f) Streptomyces carminius sp. nov., a novel actinomycete isolated from Sophora alopecuroides in Xinjiang, China. Antonie Van Leeuwenhoek 111(10):1807–1814. https://doi.org/10.1007/s10482-018-1069-x
Wang H, Xia C, Chen L, Zhao J, Tao W, Zhang X, Wang J, Gao X, Yong J, Duan JA (2019a) Phytochemical information and biological activities of quinolizidine alkaloids in Sophora: a comprehensive review. Curr Drug Targets 20(15):1572–1586. https://doi.org/10.2174/1389450120666190618125816
Wang L, Li X, Zhang Y, Huang Y, Zhang Y, Ma Q (2019b) Oxymatrine ameliorates diabetes-induced aortic endothelial dysfunction via the regulation of eNOS and NOX4. J Cell Biochem 120(5):7323–7332. https://doi.org/10.1002/jcb.28006
Wang W, Nakashima KI, Hirai T, Inoue M (2019c) Anti-inflammatory effects of naturally occurring retinoid X receptor agonists isolated from Sophora tonkinensis Gagnep. via retinoid X receptor/liver X receptor heterodimers. J Nat Med 73(2):419–430. https://doi.org/10.1007/s11418-018-01277-1
Wang W, Nakashima KI, Hirai T, Inoue M (2019d) Neuroprotective effect of naturally occurring RXR agonists isolated from Sophora tonkinensis Gagnep. on amyloid-β-induced cytotoxicity in PC12 cells. J Nat Med 73(1):154–162. https://doi.org/10.1007/s11418-018-1257-z
Wang H, Tong Y, Xiao D, Xia B (2020a) Involvement of mTOR-related signaling in antidepressant effects of Sophoraflavanone G on chronically stressed mice. Phytother Res. https://doi.org/10.1002/ptr.6675
Wang R, Deng X, Gao Q, Wu X, Han L, Gao X, Zhao S, Chen W, Zhou R, Li Z, Bai C (2020b) Sophora alopecuroides L.: an ethnopharmacological, phytochemical, and pharmacological review. J Ethnopharmacol 248:112172. https://doi.org/10.1016/j.jep.2019.112172
Wang XF, Zhu Z, Hao TT, Fang QQ, Jiang K, Qu SJ, Zuo JP, Zhu W, He SJ, Tan CH (2020c) Alopecines A-E, five chloro-containing matrine-type alkaloids with immunosuppressive activities from the seeds of Sophora alopecuroides. Bioorg Chem 99:103812. https://doi.org/10.1016/j.bioorg.2020.103812
Wei G, Chen Y, Guo X, Wei J, Dong L, Chen S (2021) Biosyntheses characterization of alkaloids and flavonoids in Sophora flavescens by combining metabolome and transcriptome. Sci Rep 11(1):1–2. https://doi.org/10.1038/s41598-021-86970-0
Wu B, Yue H, Zhou GH, Zhu YY, Wu TH, Wen JF, Cho KW, Jin SN (2019a) Protective effects of oxymatrine on homocysteine-induced endothelial injury: involvement of mitochondria-dependent apoptosis and Akt-eNOS-NO signaling pathways. Eur J Pharmacol 864:172717. https://doi.org/10.1016/j.ejphar.2019.172717
Wu D, Zhu X, Kang X, Huang H, Yu J, Pan J, Zhang X (2019b) The protective effect of sophocarpine in osteoarthritis: an in vitro and in vivo study. Int Immunopharmacol 67:145–151. https://doi.org/10.1016/j.intimp.2018.11.046
Yang C, Yu Y, Wu F, Wu Y, Feng J, Yan L, Han L, Ren J, Nie L, Zhou R (2018) Vasodilatory effects of aloperine in rat aorta and its possible mechanisms. Chin J Physiol 61:293–301. https://doi.org/10.4077/CJP.2018.BAH609
Yang J, Fei J, Su H, Tian H, Huang S, Yang P, Mao D, Hu S (2019) Flavonoids from the flowers of Sophora davidii and their anti-tobacco mosaic virus activities. Nat Prod Commun 14(6):1934578X19856786. https://doi.org/10.1177/1934578X19856786
Yang G, Zeng R, Song X, Li T, Liu C, Ni L (2020a) Sophocarpine prevents cigarette smoke-induced restenosis in rat carotid arteries after angioplasty. Ann Palliat Med 9(4):1622–1630. https://doi.org/10.21037/apm-19-568
Yang J, Cao MX, Hu WY, Wei YY, Hu TJ (2020b) Sophora subprotsrata polysaccharides suppress the inflammatory reaction of RAW264. 7 cells infected with PCV2 via regulation NF-κB/MAPKs/c-Jun signal pathway and histone acetylation modification. Int J Biol Macromol 159:957–965. https://doi.org/10.1016/j.ijbiomac.2020.05.128
Yang Y, Tian Y, Zhang Q, Li X, Fu Y, Pei H, Lui D (2020c) Comparative effects of flavonoids from Fructus Sophorae on rat osteoblasts in vitro. Rec Nat Prod 14(1):65–76. https://doi.org/10.25135/rnp.138.19.04.1262
Yang YN, Zhu H, Yuan X, Zhang X, Feng ZM, Jiang JS, Zhang PC (2021) Seven new prenylated flavanones from the roots of Sophora flavescens and their anti-proliferative activities. Bioorg Chem 109:104716. https://doi.org/10.1016/j.bioorg.2021.104716
Yao XQ, Zhang YH, Long W, Liu PX (2012) Oxysophoridine suppresses the growth of hepatocellular carcinoma in mice: in vivo and cDNA microarray studies. Chin J Integr Med 18:209–213. https://doi.org/10.1007/s11655-012-1001-6
Yao CL, Zhang JQ, Li JY, Wei WL, Wu SF, Guo DA (2021) Traditional Chinese medicine (TCM) as a source of new anticancer drugs. Nat Prod Rep. https://doi.org/10.1039/D0NP00057D
Ye Y, Wang Y, Yang Y, Tao L (2020) Aloperine suppresses LPS-induced macrophage activation through inhibiting the TLR4/NF-κB pathway. Inflamm Res 7:1–9. https://doi.org/10.1007/s00011-019-01313-0
Yin W, Han J, Zhang Z, Han Z, Wang S (2018) Aloperine protects mice against bleomycin-induced pulmonary fibrosis by attenuating fibroblast proliferation and differentiation. Sci Rep 8:6265. https://doi.org/10.1038/s41598-018-24565-y
Yoo H, Kang M, Pyo S, Chae HS, Ryu KH, Kim J, Chin YW (2017) SKI3301, a purified herbal extract from Sophora tonkinensis, inhibited airway inflammation and bronchospasm in allergic asthma animal models in vivo. J Ethnopharmacol 206:298–305. https://doi.org/10.1016/j.jep.2017.05.012
Yoo JM, Park KI, Yang JH, Cho WK, Lee B, Ma JY (2019) Anti-allergic actions of F-PASA, a novel herbal cocktail, in IgE/antigen-mediated allergic responses in RBL-2H3 cells and passive cutaneous anaphylaxis in mice. Phytomedicine 55:229–237. https://doi.org/10.1016/j.phymed.2018.06.034
Youn HJ, Lakritz J, Kim DY, Rottinghaus GE, Marsh AE (2003) Anti-protozoal efficacy of medicinal herb extracts against Toxoplasma gondii and Neospora caninum. Vet Parasitol 116:7–14. https://doi.org/10.1016/s0304-4017(03)00154-7
Zahra SA, Iqbal J, Abbasi BA, Yaseen T, Hameed A, Shahbaz A, Kanwal S, Mahmood T, Ahmad P (2021) Scanning electron microscopy of Sophora alopecuroides L. seeds and their cytotoxic, antimicrobial, antioxidant, and enzyme inhibition potentials. Microsc Res Tech. https://doi.org/10.1002/jemt.23740
Zhang F, Hu C, Cheng S, Wang S, Li B, Cao D, Fan H, Pan R, Yang M, Xu Y (2018a) The investigation of the effect and mechanism of Sophora moorcroftiana alkaloids in combination with albendazole on echinococcosis in an experimental rats model. Evid Based Compl Altern Med. https://doi.org/10.1155/2018/3523126
Zhang YB, Luo D, Yang L, Cheng W, He LJ, Kuang GK, Li MM, Li YL, Wang GC (2018b) Matrine-type alkaloids from the roots of Sophora flavescens and their antiviral activities against the hepatitis B virus. J Nat Prod 81:2259–2265. https://doi.org/10.1021/acs.jnatprod.8b00576
Zhang YB, Yang L, Luo D, Chen NH, Wu ZN, Ye WC, Li YL, Wang GC (2018c) Sophalines E-I, Five quinolizidine-based alkaloids with antiviral activities against the hepatitis B virus from the seeds of Sophora alopecuroides. Organ Lett 20(18):5942–5946. https://doi.org/10.1021/acs.orglett.8b02637
Zhang H, Chen L, Sun X, Yang Q, Wan L, Guo C (2020a) Matrine: a promising natural product with various pharmacological activities. Front Pharmacol 11:588. https://doi.org/10.3389/fphar.2020.00588
Zhang Y, Gu D, He S, Meng J, Wang J, Wang Y, Wang Y, Tian J, Yang Y (2020b) Enzyme reaction-guided identification of active components from the flowers of Sophora japonica var. violacea. Food Funct. https://doi.org/10.1039/d0fo00625d
Zhang YN, Zhang QY, Li XD, Xiong J, Xiao SQ, Wang Z, Zhang ZR, Deng CL, Yang XL, Wei HP, Yuan ZM, Ye HQ, Zhang B (2020c) Gemcitabine, lycorine and oxysophoridine inhibit novel coronavirus (SARS-CoV-2) in cell culture. Emerg Microb Infect 9(1):1170–1173. https://doi.org/10.1080/22221751.2020.1772676
Zhang M, Li A, Yang Q, Li J, Wang L, Liu X, Huang Y, Liu L (2021a) Beneficial effect of alkaloids from Sophora alopecuroides L. on CUMS-induced depression model mice via modulating gut microbiota. Front Cell Infect Microbiol 11:665159. https://doi.org/10.3389/fcimb.2021.665159
Zhang SN, Li XZ, Tan LY, Zhu KY (2021b) A review of pharmacological and toxicological effects of Sophora tonkinensis with bioinformatics prediction. Am J Chin Med 49(2):359–389. https://doi.org/10.1142/S0192415X21500178
Zhang ZJ, Dong SW, Gao DD, Du XY, Xie YQ, Xia XS, Li RT (2021c) Unusual matrine–adenine hybrids isolated from Sophora davidii and their inhibitory effects on human cytomegalovirus. Phytochemistry 190:112842. https://doi.org/10.1016/j.phytochem.2021.112842
Zhao J, Zhang G, Li M, Luo Q, Leng Y, Liu X (2018) Neuro-protective effects of aloperine in an Alzheimer’s disease cellular model. Biomed Pharmacother 108:137–143. https://doi.org/10.1016/j.biopha.2018.09.008
Zhao WY, Xiao W, Hao HQ, Zheng K, Zhang DD, Jin Q, Zhu QS (2021) Comparison of activity and components of Sophora flavescens root and seed. IOP Conf Ser 705(1):012021. https://doi.org/10.1088/1755-1315/705/1/012021
Zhou Y, Wei Y, Liu H, Zhang G, Wu XA (2010) Preparation and in vitro evaluation of ethosomal total alkaloids of Sophora alopecuroides loaded by a transmembrane pH-gradient method. AAPS PharmSciTech 11(3):1350–1358. https://doi.org/10.1208/s12249-010-9509-6
Zhou R, Xu Q, Xu Y, Xiong A, Wang Y, Ma P (2016) Oxymatrine attenuated isoproterenol-induced heart failure in rats via relation of COX-2/PGI2 pathway. Biomed Pharmacother 84:1359–1366. https://doi.org/10.1016/j.biopha.2016.10.070
Zhou B, Yu Y, Yu L, Que B, Qiu R (2018a) Sipi soup inhibits cancer-associated fibroblast activation and the inflammatory process by downregulating long non-coding RNA HIPK1-AS. Mol Med Rep 18(2):1361–1368. https://doi.org/10.3892/mmr.2018.9144
Zhou BG, Wei CS, Zhang S, Zhang Z, Gao HM (2018b) Matrine reversed multidrug resistance of breast cancer MCF-7/ADR cells through PI3K/AKT signaling pathway. J Cell Biochem 119(5):3885–3891. https://doi.org/10.1002/jcb.26502
Zhou CH, Shi ZL, Meng JH, Hu B, Zhao CC, Yang YT, Yu W, Chen ZX, Heng BC, Parkman VJ, Jiang S (2018c) Sophocarpine attenuates wear particle-induced implant loosening by inhibiting osteoclastogenesis and bone resorption via suppression of the NF-κB signalling pathway in a rat model. Br J Pharmacol 175(6):859–876. https://doi.org/10.1111/bph.14092
Zhou GZ, Shi YY, Cui LS, Li A, Wang QQ, Liu M (2018d) Oxymatrine induces A549 human non-small lung cancer cell apoptosis via extrinsic and intrinsic pathways. Mol Med Rep 17(1):1071–1076. https://doi.org/10.3892/mmr.2017.7982
Zhou YJ, Guo YJ, Yang XL, Ou ZL (2018e) Anti-cervical cancer role of matrine, oxymatrine and Sophora flavescens alkaloid gels and its mechanism. J Cancer 9(8):1357. https://doi.org/10.7150/jca.22427
Zhou H, Li J, Sun F, Wang F, Li M, Dong Y, Fan H, Hu D (2020) A review on recent advances in aloperine research: pharmacological activities and underlying biological mechanisms. Front Pharmacol 11:538137. https://doi.org/10.3389/fphar.2020.538137
Zhu L, Huang S, Li J, Chen J, Yao Y, Li L, Guo H, Xiang X, Deng J, Xiong J (2020) Sophoridine inhibits lung cancer cell growth and enhances cisplatin sensitivity through activation of the p53 and Hippo signaling pathways. Gene. https://doi.org/10.1016/j.gene.2020.144556
Zhuang H, Dai X, Zhang X, Mao Z, Huang H (2020) Sophoridine suppresses macrophage-mediated immunosuppression through TLR4/IRF3 pathway and subsequently upregulates CD8+ T cytotoxic function against gastric cancer. Biomed Pharmacother 121:109636. https://doi.org/10.1016/j.biopha.2019.109636
Zou Y, Sarem M, Xiang S, Hu H, Xu W, Shastri VP (2019) Autophagy inhibition enhances Matrine derivative MASM induced apoptosis in cancer cells via a mechanism involving reactive oxygen species-mediated PI3K/Akt/mTOR and Erk/p38 signaling. BMC Cancer 19(1):949. https://doi.org/10.1186/s12885-019-6199-7
Acknowledgements
The authors wish to thank the facilities support of the National Research Centre, Egypt. The authors acknowledge the help of Marwa Mostafa, Bab El Shaariya University Hospital, Cairo (Egypt) for drawing the chemical structures of some compounds. Mohamed O. Radwan would like to express heartfelt gratitude to Omnia Fouad for her ongoing and unwavering support. Thank you of being a part of my story.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Abd-Alla, H.I., Souguir, D. & Radwan, M.O. Genus Sophora: a comprehensive review on secondary chemical metabolites and their biological aspects from past achievements to future perspectives. Arch. Pharm. Res. 44, 903–986 (2021). https://doi.org/10.1007/s12272-021-01354-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12272-021-01354-2