Abstract
The Gulf killifish, Fundulus grandis, is a vital component of saltmarsh ecosystems and an indicator species for environmental impacts, because of strong site fidelity. Also, their otoliths can provide a record of environmental conditions because they are metabolically inert, grow continuously with the fish, and incorporate trace elements from the environment. We used laser ablation inductively coupled plasma mass spectrometry (LA-ICP-MS) to determine chemical composition differences in Gulf killifish otoliths across the northern Gulf of Mexico. Fish collections started in fall 2012 and continued through summer 2013. Concentrations of Mn, Sr, and Ba varied among sites and allowed for discrimination of fish between estuaries in Louisiana (elevated Ba concentrations) and the west side of Mobile Bay, Alabama (elevated Mn concentrations). However, elemental signatures of otoliths from Mississippi, Florida, and the east side of Alabama could not be discriminated from one another. Regional differences in otolith elemental signatures in Louisiana and west Alabama appear to provide unique chemical tags for these waters and, thus, may have utility for nursery habitat determination for species with estuarine-dependent juveniles. Otoliths of F. grandis that had been exposed to oil (either from the 2010 Deepwater Horizon oil spill or because of close proximity to an oil refinery) did not differ in elemental signature between paired oiled and non-oiled sites. Therefore, the otoliths did not contain trace metals associated with oil. Also, the relative condition of F. grandis did not differ between paired sites. The presence of F. grandis at all sites, the lack of effect of oiling on relative condition, and no signal of oil-related elements in the otoliths suggest minimal long-term impact of the Deepwater Horizon oil spill on F. grandis.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
The Gulf killifish, Fundulus grandis, is a vital component of saltmarsh ecosystems along the US Gulf of Mexico coast. This fish is one of the most abundant nekton species found in saltmarshes and provides an important prey resource for many piscivores, including commercially valuable fishes (Rozas and Reed 1993; Rozas and Zimmerman 2000). F. grandis is thought to be very similar to its Atlantic coast congener Fundulus heteroclitus (Mummuichog) sharing a recent ancestor in the family Fundulidae (Whitehead 2010) with some evidence of hybridization when ranges overlap (Gonzalez et al. 2009). These two species also exhibit similar spawning (Kneib and Stiven 1978; Greeley and MacGregor 1983) and feeding behaviors (Kneib and Stiven 1978; Rozas and LaSalle 1990) and both are saltmarsh residents that exhibit strong site fidelity (Lotrich 1975; Teo and Able 2003; Able et al. 2006; Nelson et al. 2014). This marsh residency and strong site fidelity is the reason why F. heteroclitus (Courtenay et al. 2002; Finley et al. 2009; Dibble and Meyerson 2012, 2014) and recently F. grandis (Whitehead et al. 2012) have been used as indicator species to assess environmental impacts and changes in saltmarshes.
Otolith chemistry has been used for the assessment of past environmental conditions experienced by a fish (Lowe et al. 2009, 2011; Farmer et al. 2013), stock discrimination (Campana et al. 2000; Sohn et al. 2005; Niklitschek et al. 2010), retrospective tracking of fish movements (Walther et al. 2011; Albuquerque et al. 2012; Farmer et al. 2013), and identification of nursery habitat (Thorrold et al. 1998; Gillanders and Kingsford 2003; Vasconcelos et al. 2007; Tanner et al. 2011). This is because otoliths are metabolically inert and therefore not reworked throughout life as bone can be, and they provide records of fish growth and incorporate trace chemical compositions from the environment (Campana and Neilson 1985; Campana 1999; Campana and Thorrold 2001; Sturrock et al. 2012). These trace chemicals are incorporated into the otolith through substitution with Ca in the CaCO3 aragonite matrix (Doubleday et al. 2014) and come from surrounding water (Farrell and Campana 1996; Walther and Thorrold 2006) and/or diet items (Kennedy et al. 2000; Buckel et al. 2004).
The utility of otolith microchemistry as a tool depends strongly on environmental conditions and, specifically, chemical gradients, in which a fish lives. Significant differences in otolith microchemical signatures have been found in fishes collected from different areas within estuarine (Vasconcelos et al. 2007; Bradbury et al. 2008; Tanner et al. 2011), marine (Ashford and Jones 2007; Longmore et al. 2010; Ferguson et al. 2011), and freshwater systems (Brazner et al. 2004; Zeigler and Whitledge 2010; Wolff et al. 2012). Otolith microchemistry is particularly effective in estuarine systems, due to varying amounts of chemical inputs among watersheds (Oros et al. 2007; Reay 2009; Statham 2012; Wang et al. 2012), variable salinity, and variation in freshwater input (Rooker et al. 2004; Lowe et al. 2011). Because otolith microchemical signatures can provide a record of environmental conditions experienced throughout the life of a fish, otolith microchemistry can be a useful tool in environmental impact studies. Elements (Na, Mg, Cr, and Sr) found in crude oil from the 2002 Prestige oil spill in northwestern Spain, were incorporated in otoliths of benthic juvenile Turbot (Scophthalmus maximus) in the laboratory (Morales-Nin et al. 2007).
The Deepwater Horizon oil spill (referred to hereafter as DHOS) in the Gulf of Mexico was the largest oil spill in U.S. history (Abbriano et al. 2011) and possibly the largest offshore spill to have ever occurred (Camilli et al. 2010). It released an estimated 4 million barrels of Louisiana crude oil into the northern Gulf of Mexico (NGOM) during summer 2010 (Camilli et al. 2010). Initial studies on environmental impacts of the DHOS spill suggested that nearshore (Fodrie and Heck 2011; Moody et al. 2013) and offshore fishes (Rooker et al. 2013; Szedlmayer and Mudrak 2014) have been relatively resilient to adverse effects of oiling in the NGOM. However, detection of small genetic differences in F. grandis from Barataria Bay, LA were presumed to be an effect of the DHOS oil (Whitehead et al. 2012). An independent marker such as elements associated with oil being identified in fish otoliths would allow potential impacts, such as the timing or perhaps the magnitude of exposure, to be more clearly assessed. Analysis of DHOS oil using inductively coupled plasma mass spectrometry (ICP-MS) found Al, V, Cr, Co, Ni, Cu, Zn, Se, and Sn in the oil and a lack of heavier metals suggesting that it was a lighter oil (Grosser et al. 2012). Additionally, Se, Ni, and V have been found in other crude oils and refinery effluents (Miekeley et al. 2005; De Almeida et al. 2009; Ellis et al. 2011).
The objectives of this study were to determine whether (1) the elemental composition of F. grandis otoliths across the broader northern Gulf of Mexico could be used to discriminate the source of fish on both small and large spatial scales and (2) oil exposure could be detected using the chemical compositions of F. grandis otoliths, which was accomplished by comparing the otoliths of fish collected from paired oiled and non-oiled marshes.
Methods
Field Collection
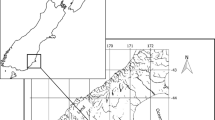
Collection of F. grandis occurred at a total of 10 saltmarshes across the northern Gulf of Mexico (Fig. 1). The amount of DHOS oiling at sites was assessed using the NOAA Environmental Response Management Application (ERMA) (gomex.erma.noaa.gov) and oiled sites were paired with non-oiled ones. Louisiana sites were located in Barataria Bay near Grand Isle, LA, and included one that was heavily oiled from the DHOS oil spill (Barataria A, BA) and one that did not receive DHOS oil (Barataria B, BB). Sites in Mississippi were both near Pascagoula. The refinery site (RF) was along a dredge ditch adjacent to an oil refinery (assumed to have been exposed regularly to non-DHOS oil and refinery effluents) and the Crooked Creek site (CK) was a control site in the Grand Bay National Wildlife Refuge (and not regularly exposed to oil). Three sites were located in Alabama, west of Mobile Bay: Fowl River (FR), Fowl B (FB), and Fowl A (FA), and FA received light oiling during the DHOS spill. Two more Alabama sites, Wolf Bay (WF) and Perdido (PD), were located east of Mobile Bay, near Orange Beach, AL. The Perdido site also received light oiling during the DHOS spill. The site in Florida was in Hogtown Bayou (HT), which is part of the larger Choctawhatchee Bay and was not oiled.
Sample sites in the northern Gulf of Mexico. Boxes on the map at the top of the figure correspond to sample areas that are indicated by numbers below the boxes. Enlarged panels of each sample area are shown and denoted with the corresponding number and name of the area in the bottom right of each panel. Within each panel, sites are labeled and denoted with an oiled or non-oiled symbol
Fish were collected in fall 2012, winter 2013, spring 2013, and summer 2013 from all sites using minnow traps baited with cut menhaden (Brevoortia sp.). At each site, eight traps were placed in saltmarsh habitat and left for a soak time of 1 to 1.5 h. All F. grandis in traps were euthanized, stored on ice, and returned to the lab. During each fish collection, water temperature and salinity were measured using a salinity-conductivity meter (Yellow Springs Instruments model 30). Water samples for chemical analysis were collected during the winter, spring, and summer sampling events. Each water sample was filtered through a 0.45-μm filter, preserved with 125 μL nitric acid, stored in 125 ml acid washed polypropylene bottles, and refrigerated until processing.
Lab Processing
Fish were either processed immediately after returning from the field or frozen for later processing. Up to 20 F. grandis were selected from each site for otolith analysis. These 20 fish consisted of four fish from each of five different size ranges (50 to 59, 60 to 69, 70 to 79, 80 to 89, 90 to 100+ mm total length [TL]), if present. If all size ranges were not represented, then the numbers selected were chosen equally from represented size classes to reach a total of 20 individuals per location. Lengths and weights of the selected fish were recorded and their sagittal otoliths were removed. Otoliths were then rinsed in 30 % H2O2 to clean off any residual tissue, followed by triple distilled ultra-filtered water (DIUF) and stored dry in vials until preparation for microchemistry.
For each fish, one of the sagittal otoliths was prepared for microchemistry analysis. First, the otolith was re-washed in 30 % H2O2 and rinsed with DIUF. The otolith was then inspected under a dissecting scope to look for remaining tissue, and if present, it was removed with forceps and the otolith was rinsed again with DIUF. After cleaning, otoliths were polished until a smooth surface was achieved using progressively finer grit 3 M® lapping film (grit sizes 30 to 0.3 μm). After polishing, otoliths were re-rinsed with DIUF and mounted to petrographic slides using Weldbond® glue and stored until microchemical analysis.
Otolith Microchemistry
Otolith microchemical analyses were conducted at the University of Windsor, Great Lakes Institute for Environmental Research (GLIER), Windsor, Ontario, Canada. Trace metal compositions of otoliths from F. grandis were quantified using a laser ablation inductively coupled plasma mass spectrometry (LA-ICP-MS) system comprising a high energy, pulsed, ultrafast femtosecond laser (Integra-C® by Quantronix®, East Setauket, New York) and a Thermo-Electron® X Series-II® ICP-MS. Straight line laser ablations were conducted using a 20-μm diameter laser beam and extended from one edge to the other, across the otolith core. The average element concentration for this entire ablation track was defined as whole otolith analysis, given that this track encompasses the entire life of the fish twice and this term (whole otolith analysis) will be used throughout the rest of the paper. Ablation speed for all otoliths was set at 5 μm/s using a computer-controlled stage. Ablated material was carried to the ICP-MS using Ar gas. The laser energy was 27 μJ, measured after a 2.5-mm beam-constricting aperture, pulse frequency was 100 Hz, pulse width was 130 fs, and wavelength was 785 nm. The ICP-MS was operated with 1200 W RF power, a nebulizer gas flow rate of 1.01 L/min, coolant gas rate of 14 L/min, and a ICP sampling depth of 120 mm. The signal intensities in integrated counts/second (icps) for 33 isotopes were measured and were used to quantify the concentrations of 24 elements (Supplemental Table 1). The signal intensities of multiple isotopes for some elements (e.g., Ca, Ni, Zn, Se, Sr, Sn, Ba, and Pb) were measured for internal standardization and interference correction purposes. Given the speed of the ablation and the number of isotopes measured, concentrations were output roughly every 0.4 second of the ablation.
Two runs ablating synthetic borosilicate glass standard reference material (National Institute of Standards and Technology [NIST] 610) with known concentrations of the elements of interest were analyzed both before and after approximately every 16 otolith samples to provide external calibration standards, calculate the limits of detection (LOD) for each element, and to correct for instrumental and Ar carrier gas background levels (Longerich et al. 1996; Ludsin et al. 2006). For an element to be included in the data comparative analysis, it had to be detected above the LOD in at least 50 % of the samples. Estimated instrument precision and relative standard deviation (RSD %) of each element was obtained from a validation study (Shaheen et al. 2008), which measured performance of the LA-ICP-MS system used in our study. For otolith analyses, Ca was used as the internal calibration standard and was assigned a constant concentration of 400,432 ppm (the stoichiometric abundance of Ca in aragonite (CaCO3)), to correct for differences in mass of ablated material (Campana et al. 1994) and to convert raw isotope signal intensity measurements (icps) to concentration units (ppm) (Ludden et al. 1995; Longerich et al. 1996; Ludsin et al. 2006).
Water samples were analyzed using solution-mode ICP-MS, after a 100-fold dilution with an internal standard solution of 1 % HNO3 containing internal standards spikes of Be, In, and Tl at concentrations of 10, 1, and 2 ppb, respectively, to correct for instrumental mass bias and drift, as well as matrix effects on an individual sample basis (Lowe et al. 2009, 2011).
Statistical Analysis
Fish Length and Condition
A two-way ANOVA with site, collection season, and the site × season interaction was used to determine whether fish lengths differed across sites and seasons. This ANOVA was run using type-III sums of squares and was checked for violation of assumptions using Shapiro-Wilks and Levene’s tests.
Since standard weight equations have not been developed for F. grandis, relative condition was compared across sites, seasons, and lengths using a two-way ANOVA with type-III sums of squares. Relative condition is the ratio of an individual weight to the expected weight of the fish as determined by the length-weight regression for all fish in the study (Neumann et al. 2012). The main effects of site and season and the covariate length, as well as all two-way interactions, were included in the ANOVA. Differences between sites and seasons were compared using Tukey pairwise comparisons, obtained from the most parsimonious model, which was chosen by removing nonsignificant main effects (main effects were only removed if a significant interaction did not include the main effect) and interactions from the model until only significant ones remained. This ANOVA was also checked for violation of assumptions using Shapiro-Wilks and Levene’s tests.
Whole Otolith Analysis
To determine if elemental concentrations differed among sites and between paired oiled and non-oiled sites for whole otolith averages (average element concentration for the entire ablation), a two-way ANOVA with site, collection season, a covariate fish length (TL), and all possible two-way interactions were included in the first model. The most parsimonious model was chosen by removing nonsignificant (p ≥ 0.05) main effects and interactions and re-running the ANOVA until only significant variables remained. If an interaction was significant, but a main effect included in that interaction was not, then the main effect was retained in the final model. All tests were run using type-III sums of squares (Speed et al. 1978; Shaw and Mitchellolds 1993) and Tukey pairwise comparisons were performed when significant site or season effects were shown. Each final ANOVA was tested for violation of assumptions using the Shapiro-Wilks and Levene’s tests. Although the majority of ANOVAs failed these tests and log data transformations did not help, the ANOVA test is robust to violations in normality and variance and has greater power than the similar nonparametric Kruskal-Wallis or rank-sum approaches (Brownie and Boos 1994; Kahn and Rayner 2003); therefore, ANOVA was used.
Elements that showed significant differences between sites in the Tukey tests were used in a linear discriminant function analysis (LDFA) with leave one out cross validation, to determine if fish could be correctly classified to site of collection (prior probabilities set at 0.1 for each site given the ten sites) and then region of collection (prior probabilities set at 0.25 for each of the four regions) based on otolith chemical signatures. Before LDFA was performed, all chemical data were scaled using z-scores. Using the same scaled data from the LDFA, a hierarchical cluster analysis was performed based on a Euclidean distance matrix and Ward’s method of cluster classification (Ward 1963; Wishart 1969). This cluster analysis was performed for all sites to see how sites were grouped and to see if classification of fish to states or some other regional scheme was most appropriate.
Otolith Edge and Core Analysis
To compare elemental concentrations in the cores versus the edges of otoliths, the laser ablation time-resolved analyses were subdivided into core and edge components for a subsample of 200 otoliths. This subsample consisted of one fish from each size class (if present) for each site and season (n = 50/season, total n = 200). If all size classes were not represented in a collection, then the five fish were chosen from size classes that were present. The time-resolved ICP-MS signal for a standard distance of 113 μm was chosen to represent the edge and core signatures. This represents a distance equal to 10 % of the ablation when averaged across all of the smallest (50 to 59 mm TL) fish in the edge/core subsample. Therefore, each edge chemical signature was measured by averaging data obtained for all ICP-MS time slices (~57) over the 113 μm at the end of the ablation, and each core chemical signature was measured by averaging data obtained for all ICP-MS time slices (~57) over the 113 μm straddling the otolith core, where the center of the core was at 56.5 μm, such that an equal number of data points was used on either side of the core. Otolith cores were defined as the small, opaque section in the approximate center of the otoliths.
Split-plot ANOVA (SP-ANOVA) with individual fish as the whole plot and otolith region as the split plot was performed to see if otolith region (core and edge) chemical signatures differed within otoliths. The interactions of season, site, and length with otolith region were used in the SP-ANOVA to determine if differences between the core and edge were consistent across these effects. The most parsimonious model was chosen by removing nonsignificant (p ≥ 0.05) interactions or main effects, with the exception of otolith region and re-running the model until only significant variables remained. These split-plot ANOVAs were run using type-III sums of squares.
Differences in detectable element (>50 % LOD) concentrations among sites and seasons at both cores and edges of otoliths were analyzed with two-way ANOVAs, using type-III sums of squares. The main effects of site, season, and the site × season interaction were used in the first model, and if the interaction was significant (p ≤ 0.05), then no main effects could be removed and this was considered to be the best model. If the interaction was not significant, the remaining significant main effects were used in the final model. If either site or season was shown to be significant, then Tukey tests were performed to identify differences among sites and seasons. ANOVAs for this section were checked for violations of assumptions using Shapiro-Wilks and Levene’s tests, and once again, the majority of tests failed; however, we continued to use ANOVAs because of their robustness.
Salinity and Temperature
Differences in salinity and water temperature across sites and seasons were analyzed using a one-way ANOVA. If the site or season ANOVA was significant, a Tukey honest significant difference (HSD) test was used to find differences among sites and/or seasons. All ANOVAs were tested for assumptions using Shapiro-Wilks and Levene’s tests.
Water Elemental Concentrations
One-way ANOVAs were used to compare water sample elemental concentrations among sites and seasons. If either the site or season ANOVA was significant, a Tukey HSD test was used to identify differences among sites and seasons, and ANOVAs were checked for violation of assumptions. For an element to be included in the evaluation of water chemistry, it had to be above LOD in >50 % of otolith samples and >50 % of water samples. All elemental abundances in water samples were converted into molar ratios with calcium (Ca) prior to analysis and are reported in element (mmol) per Ca (mol) water.
Results
Field Collections
Gulf killifish were successfully captured from every sample site during each collection event. It was not always possible to collect four fish in each of the five size classes (≤59, 60 to 69, 70 to 79, 80 to 89, ≥90 mm TL) as per design. In total, 18 to 20 otoliths were obtained from each site on each sample date for otolith microchemistry (OMC) analysis, except for winter samples from BB and FR, which had 11 and 13 otoliths, respectively. This resulted in a final sample size of 773 otoliths (180 to 200 per season) for OMC analysis, which were used for whole otolith comparisons. Average fish length did not differ significantly among sites (ANOVA: F 9, 733 = 1.330, p = 0.217), but captured fish were significantly smaller in winter than any other season (ANOVA: F 3, 733 = 5.491, p < 0.001). Also, the site × season interaction was significant (ANOVA: F 27, 733 = 4.069, p < 0.001), due to differences among sites that were not consistent between seasons.
Relative Condition
Gulf killifish relative condition differed across sites (ANOVA: F 9, 729 = 9.108, p < 0.001) and seasons (ANOVA: F 3, 729 = 10.304, p < 0.001). Both the site × season (ANOVA: F 27, 729 = 6.377, p < 0.001) and season × length (ANOVA: F 3, 729 = 2.796, p = 0.040) interaction terms were significant, but length alone did not have a significant effect on relative condition (ANOVA: F 1, 729 = 0.331, p = 0.565, Fig. 2a–c). Paired oiled and non-oiled sites did not differ in relative condition, except that the oiled PD site had higher relative conditions than WF. Relative condition was highest at BB and BA, and RF did not differ from these sites (Fig. 2a). Also, relative condition was highest in summer (Fig. 2b).
F. grandis relative condition (mean [±1 SE]) across sites, with letters above bars corresponding to significant differences among sites based on the results of Tukey pairwise comparisons (a). Mean relative condition across sites by seasons, with letters beside seasons in the legend corresponding to significant differences among seasons based on the results of Tukey pairwise comparisons (b) and mean relative condition across fish length classes by seasons (c). Lines connecting points in b–c help to visualize patterns and separate seasons and length classes; they do not depict relationships between sites or length classes
Otolith Differences Among Sites
Detectable Elements
Out of the 33 isotopes that were measured using the ICP-MS, 20 (Supplemental Table 1) met the threshold criterion (>50 % LOD) for inclusion in OMC comparisons. For elements where more than one isotope met the inclusion criterion (Zn, Sr, Sn, Ba, Pb), the isotopes were regressed against one another, and if regressions varied too far from the 1:1 slope line (R 2 < 0.90), then both isotopes were considered in OMC comparisons. Only isotopes of Sr (R 2 = 0.361) and Zn (R 2 = 0.878) differed from the 1:1 relationship. The number of samples above LOD for 67Zn was less than 66Zn, which resulted in observations of zero for 67Zn but not 66Zn when concentrations were low, causing deviation from the 1:1 line. Therefore, 66Zn was the only Zn isotope used in OMC comparisons. Both isotopes of Sr were used given the relatively low correlation between 88Sr and 86Sr. The possible cause(s) of this lack of correlation between Sr isotopes (e.g., LOD, systematic interference effects), however, is not readily apparent. This resulted in use of 13 isotopes of 12 elements in the OMC statistical analysis (Table 1).
Concentration Comparisons Among Sites
Entire otolith average concentrations of Mn, Fe, Rb, Sr, Ba, and U differed across sites. Whole otolith average concentrations of Mn differed across sites (ANOVA: F 9, 720 = 8.841, p < 0.001) and collection seasons (ANOVA: F 3, 720 = 7.995, p < 0.001), but length alone did not have a significant effect (ANOVA: F 1, 720, p = 0.060). All three interaction terms were significant for Mn, site × length (ANOVA: F 9, 720 = 5.223, p < 0.001), site × season (ANOVA: F 27, 720 = 3.11, p < 0.001), and length × season (ANOVA: F 3, 720 = 8.357, p < 0.001) (Fig. 3a–d). Paired oiled and non-oiled sites did not differ in Mn concentrations, except FA (light oiling) was higher than FB (non-oiled), but concentrations were similar to the non-oiled FR site located farther up the watershed. FR and FA also had the highest Mn concentrations of any site, while FB and BB composed the next highest group (Fig. 3a). Despite these statistical interactions, the patterns of Mn concentrations were consistent across sites (Fig. 3b–d). Core Mn concentrations differed across sites (ANOVA: F 9, 190 = 6.856, p < 0.001) and the concentration patterns were similar to those seen in whole otolith analysis, while no site differences were observed in otolith edges. Manganese core concentrations were also higher than otolith edge concentrations (SP-ANOVA: F 1, 175 = 156.53, p < 0.001) across all sites and seasons.
Mean (±1 SE) whole otolith Mn concentrations (ppm) across sites, where bars with different letters indicate significant differences among sites based on Tukey pairwise comparisons (a). Mean whole otolith Mn concentrations across sites by season, where letters beside season in the legend correspond to significant differences among seasons, based on Tukey pairwise comparisons (b). Mean whole otolith Mn concentrations across sites by fish length class (c) and across fish length classes by season (d). Lines connecting points in b–d help to visualize patterns and separate seasons and lengths; they do not depict relationships between sites or length classes
Whole otolith concentrations of iron differed across sites (ANOVA: F 9, 729 = 21.812, p < 0.001) and seasons (ANOVA: F 3, 729 = 9.421, p < 0.001), but length (ANOVA: F 1, 729 = 3.708, p = 0.055) alone was not related. The interactions of site × season (ANOVA: F 27, 729 = 18.979, p < 0.001) and season × length (ANOVA: F 3, 729 = 4.140, p = 0.006) were both significant as well (Fig. 4a–c). The FB site (non-oiled) had significantly higher mean Fe concentrations than other sites, with FA having the second highest concentrations (Fig. 4a). Besides this, no other differences were observed between paired oiled and non-oiled sites. Differences among sites in both otolith cores (ANOVA: F 9, 160 = 5.532, p < 0.001) and edges (ANOVA: F 9, 160 = 4.750, p < 0.001), as well as seasons (core-ANOVA: F 3, 160 = 5.069, p = 0.002; edge-ANOVA: F 3, 160 = 3.438, p = 0.02), were similar to those seen in whole otolith comparisons and the interactions between site × season (core-ANOVA: F 27, 160 = 6.823, p < 0.001; edge-ANOVA: F 27, 160 = 6.070, p < 0.001) were similar as well.
Mean (±1 SE) whole otolith Fe concentrations (ppm) across sites, where bars with different letters indicate significant differences among sites based on Tukey pairwise comparisons (a). Mean whole otolith Fe concentrations across sites by seasons, where letters beside season in the legend correspond to significant differences among seasons based on Tukey pairwise comparisons (b). Mean whole otolith Fe concentrations across fish length classes for each season (c). Lines connecting points in b–c help to visualize patterns and separate seasons; they do not depict relationships between sites or length classes
The Florida site, Hogtown Bayou (HT), had significantly lower mean Rb concentrations for entire otoliths than FB and WF, while Rb concentrations were similar to one another at other sites (ANOVA: F 9, 733 = 21.812, p < 0.001) (Fig. 5). Also, no difference in Rb was observed between any paired oiled and non-oiled sites. Mean Rb whole otolith concentrations were less in the fall than the three other seasons (ANOVA: F 3, 733 = 9.421, p < 0.001), and the interaction between these two main effects (ANOVA: F 27, 733, p < 0.001) was significant (Fig. 5b). Rubidium did not exhibit significant differences among sites in either otolith core or edge comparisons.
Mean (±1 SE) whole otolith Rb concentrations (ppm) across sites, where bars with different letters indicate significant differences among sites based on Tukey pairwise comparisons (a). Mean whole otolith Rb concentrations across sites by seasons, where letters beside season in the legend correspond to significant differences among seasons based on Tukey pairwise comparisons (b). Lines connecting points in b help to visualize patterns and separate seasons; they do not depict relationships between sites
Whole otolith 86Sr concentrations differed among sites (ANOVA: F 9, 720 = 3.425, p < 0.001) and seasons (ANOVA: F 3, 720 = 41.1627, p < 0.001) being highest in the fall (Fig. 6a, b). All interaction terms, site × season (ANOVA: F 27, 720 = 5.586, p < 0.001), site × length (ANOVA: F 9, 720 = 2.276, p = 0.016), and season × length (ANOVA: F 3, 720 = 20.524, p < 0.001), were significant, but the covariate length was not (ANOVA: F 1, 720 = 0.192, p = 0.661). BB had the lowest mean 86Sr out of all sites, followed by BA and FR, which did not differ. CK and PD had the highest mean 86Sr concentrations and also did not differ (Fig. 6a). Although present, significant interactions did not affect overall patterns across sites (Fig. 6b–d). Core and edge concentration differences among sites (core-ANOVA: F 9, 187 = 10.738, p < 0.001, edge-ANOVA: F 9, 160 = 7.623, p < 0.001) and seasons (core-ANOVA: F 3, 187 = 34.438, p < 0.001, edge-ANOVA: F 3, 160 = 8.162, p < 0.001) were similar to whole otolith comparisons, and the interaction of site × season (ANOVA: F 27, 160 = 2.635, p < 0.001) was only significant for otolith edge comparisons.
Mean (±1 SE) whole otolith 86Sr concentrations (ppm) across sites, where bars with different letters indicate significant differences among sites based on Tukey pairwise comparisons (a). Mean whole otolith 86Sr concentrations across sites by seasons, where letters beside season in the legend correspond to significant differences among seasons based on Tukey pairwise comparisons (b). Mean whole otolith 86Sr concentrations across sites for each fish length class (c) and across fish length classes for each season (d). Lines connecting points in b–d help to visualize patterns and separate seasons and lengths; they do not depict relationships between sites or length classes
Concentrations of 88Sr across whole otoliths also differed among sites (ANOVA: F 9, 720 = 2.774, p = 0.003) and seasons (ANOVA: F 3, 720 = 49.674, p < 0.001) (Table 4, Fig. 7a, b). Length (ANOVA: F 1, 720 = 0.400, p = 0.527) alone was not significant, but the interactions of site × length (ANOVA: F 9, 720 = 2.045, p = 0.032) and season × length (ANOVA: F 3, 720 = 20.515, p < 0.001) were. The interaction of site × season (ANOVA: F 27, 720 = 8.031, p < 0.001) was also significant. Similar to 86Sr, BB had the lowest mean 88Sr concentration followed by BA, which did not differ from FR. CK and PD had the highest 88Sr concentrations for all sites and they did not differ (Fig. 7a). Winter samples had a lower mean 88Sr concentration than other seasons, fall and spring did not differ, and summer had the highest concentration (Fig. 7b). General patterns of concentrations across sites were consistent (Fig. 7b, c). Differences in 88Sr concentrations across sites in both cores (ANOVA: F 9, 160 = 5.239, p < 0.001) and edges (ANOVA: F 9, 160 = 7.779, p < 0.001) of otoliths were significant and similar to whole otolith patterns. Seasonal differences were only significant in edge comparisons (ANOVA: F 3, 160 = 15.364, p < 0.001), but the season × site interaction was significant (core-ANOVA: F 27, 160 = 2.041, p = 0.004; edge-ANOVA: F 27, 160 = 3.477, p < 0.001) in both core and edge analysis. Although both 86Sr and 88Sr differed between paired oiled and non-oiled sites, differences were not consistently in the same direction; rather, Sr concentration patterns followed salinity trends (Figs. 6 and 7).
Mean (±1 SE) whole otolith 88Sr concentrations (ppm) across sites, where bars with different letters indicate significant differences among sites based on Tukey pairwise comparisons (a). Mean whole otolith 88Sr concentrations across sites by seasons, where letters beside season in the legend correspond to significant differences among seasons based on Tukey pairwise comparisons (b). Mean whole otolith 88Sr concentrations across sites for each fish length class (c) and across fish length classes for each season (d). Lines connecting points in b–d help to visualize patterns and separate seasons and lengths; they do not depict relationships between sites or length classes
Concentrations of barium across the whole otolith differed across sites (ANOVA: F 9, 729 = 227.113, p < 0.001) and seasons (ANOVA: F 3, 729 = 39.786, p < 0.001) and were negatively related to fish length (ANOVA: F 1, 729 = 7.595, p = 0.006) (Fig. 8a–c). The interactions of site × season (ANOVA: F 27, 729 = 12.417, p < 0.001) and season × length (ANOVA: F 3, 729 = 7.935, p < 0.001) were also both significant. Barium concentrations were highest at the Louisiana sites (BB, BA) and FR had the closest mean concentration to these sites (Fig. 8a). Also, barium concentrations generally decreased with fish length for each collection (Fig. 8c). Although paired oiled and non-oiled sites differed in Ba concentrations, oiled sites were not always higher than non-oiled ones. The patterns of Ba concentrations in otolith cores and edges were similar to those seen in whole otoliths and were also different across sites (core-ANOVA: F 9, 160 = 24.760, p < 0.001; edge-ANOVA: F 9, 160 = 66.413, p < 0.001) and seasons (core-ANOVA: F 3, 160 = 6.644, p < 0.001; edge-ANOVA: F 3, 160 = 31.187, p < 0.001), with a significant site × season interaction (core-ANOVA: F 27, 160 = 2.146, p = 0.002; edge-ANOVA: F 27, 160 = 4.808, p < 0.001). The PD site exhibited higher Ba in cores than edges, and the within otolith comparison predicted that otolith cores had higher concentrations than edges (SP-ANOVA: F 1, 175 = 17.65, p < 0.001). However, this was not always the case at all sites and seasons, leading to significant otolith region × site (SP-ANOVA: F 18, 175 = 49.660, p < 0.001) and region × season interactions (SP-ANOVA: F 6, 175 = 9.61, p < 0.001).
Mean (±1 SE) whole otolith Ba concentrations (ppm) across sites, where bars with different letters indicate significant differences among sites based on Tukey pairwise comparisons (a). Mean whole otolith Ba concentrations across sites by seasons, where letters beside season in the legend correspond to significant differences among seasons (b). Mean whole otolith Ba concentrations across fish length classes for each season (c). Lines connecting points in b–c help to visualize patterns and separate seasons and lengths; they do not depict relationships between sites or length classes
Uranium concentrations in otoliths were higher at BA than HT (ANOVA: F 9, 733 = 2.776, p = 0.003), but paired oiled and non-oiled sites did not differ (Fig. 9a). Fall and winter samples had higher concentrations than spring and summer samples (ANOVA: F 3, 733 = 7.206, p < 0.001) and the significant site × season interaction (ANOVA: F 27, 733 = 1.920, p = 0.004) was driven by different patterns across sites during the fall and winter seasons (Fig. 9b). Also, U was below detection limits in the cores and edges of otoliths, so it was not considered in those comparisons.
Mean (±1 SE) whole otolith U concentrations (ppm) across sites, where bars with different letters indicate significant differences among sites based on Tukey pairwise comparisons (a). Mean whole otolith U concentrations across sites by seasons, where letters beside season in the legend correspond to significant differences among seasons based on Tukey pairwise comparisons (b). Lines connecting points in b help to visualize patterns and separate seasons; they do not depict relationships between sites
Discrimination Among Sites and Regions
Concentrations of Mn, Fe, Rb, Sr, Ba, and U in otoliths were used in the LDFA because the concentrations of these elements differed significantly among sites. This LDFA analysis, where each site was used as a group in the function, assigned fish to the correct collection sites with a relatively low degree of accuracy. Among sites, FR had the highest correct classification percentage (86 %), followed by BB (68 %) and BA (64 %). Also, when misclassifications for BB and BA occurred, these two sites were classified as the other most of the time. Classification was poorest at CK (13 %) and HT (14 %), followed by PD, WF, FA, and FB (all around 40 %, Table 2). The elements that contributed most strongly to the discrimination functions were Ba, Sr, Fe, and Mn (decreasing importance, respectively, Table 3) and sites that were misclassified to one another had similar concentrations of these elements. The hierarchical cluster analysis also had similar results to the LDFA, with BB and BA forming a discrete cluster and FR, FA, and FB being grouped together (Fig. 10). Sites in Mississippi, on the east side of Alabama, and in Florida were clustered without geographical distinction.
To relate broader regional patterns that might be related to major watershed differences, sites were grouped into four collection regions and another LDFA was performed. These four regions consisted of Louisiana sites (BB, BA), MS sites (RF, CK), Alabama sites on the west side of Mobile Bay (FB, FA, and FR), and Alabama sites on the east side of Mobile Bay combined with the Florida site (WF, PD, HT). Classification rates for these collection regions were better than for individual sites, especially in the LA sampling region (97 % classification success, Table 4). The MS and AL east + FL regions were misclassified to each other, resulting in the low classification percentages for these groups (46 and 58 %, respectively), and the AL west grouping (67 % successful) was misclassified to both MS and AL east + FL 15 % of the time. The main elements contributing to discrimination were similar as in the LDFA above with Ba having the most influence followed by Sr and Mn, respectively (Table 5).
Salinity and Temperature
Each site experienced varying salinities and temperatures throughout the study. Salinity differed significantly across sites (ANOVA: F 9, 30 = 7.530, p < 0.001), but not seasons (ANOVA: F 3, 36 = 1.796, p = 0.165, Fig. 11), and temperature differed significantly across seasons (ANOVA: F 3, 36 = 42.810, p < 0.001), but not sites (ANOVA: F 9, 30 = 0.227, p < 0.001).
Water Chemistry
Only four of the elements that differed across sites in otolith analysis (Mn, Fe, Sr, and Ba) were also present above the analytical LOD at least 50 % of the time in water samples, and of these, all but Fe were detectable in 100 % of samples. Concentrations of manganese (Mn/Ca) in water samples differed across sites (ANOVA: F 9, 20 = 4.086, p = 0.004), but not seasons (ANOVA: F 2, 27 = 0.212, p = 0.810). The concentration of Mn was highest in FR, which is similar to otolith Mn patterns. However, unlike otolith concentrations, water samples from FA did not have higher Mn concentrations (Fig. 12a). Iron (Fe/Ca) concentrations in water samples did not differ across sites (ANOVA: F 9, 20 = 1.519, p = 0.208) or seasons (ANOVA: F 2, 27 = 1.071, p = 0.357). Strontium (Sr/Ca) concentrations in water samples did not vary across sites (ANOVA: F 9, 20 = 1.497, p = 0.216), but concentrations were higher in summer than in spring, and concentrations in winter did not differ from either spring or summer (ANOVA: F 2, 27 = 4.692, p = 0.018) (Fig. 12b). Barium (Ba/Ca) differed across sites (ANOVA: F 9, 20 = 10.32, p < 0.001), but not seasons (ANOVA: F 2, 27 = 0.3663, p = 0.697), and concentrations were highest in water samples from BB, which did not differ from FA. FA was also similar to BA, FB, and FR (Fig. 12c).
Discussion
Oil Indicators
Other than carbon, hydrogen, and oxygen (the elements that make up the majority of hydrocarbons), several elements have been identified in crude and weathered DHOS oil (Mg, Al, V, Cr, Co, Ni, Cu, Zn, As, Se, Sn, and Pb) (Grosser et al. 2012; Liu et al. 2012), refinery effluents (Se) (Miekeley et al. 2005; De Almeida et al. 2009), and other crude oils (Ni and V) (Ellis et al. 2011). In addition, several elements (Na, Mg, and Cr) have been shown to be incorporated into fish otoliths when exposed to oil (Morales-Nin et al. 2007). In our study, many of these elements were either below detection limits (V, Cr, Ni, and Se) or did not vary among sites (Mg, Al, Cu, Zn, Sn, Pb). Given this and the absence of significant differences between oiled versus non-oiled sites in concentration patterns for elements that did vary across sites, we found no detectable indication of impact by metals as a result of oil exposure from the DHOS oil or refinery processes in F. grandis otoliths.
We expected that otoliths from F. grandis collected at the oiled site in Barataria Bay (BA) Louisiana would likely have a trace metal signature that reflected oil exposure, given that this site received heavy amounts of oiling (gomex.erma.noaa.gov/erma.html). Also, weathered DHOS oil in marshes has a higher concentration of metals than crude oil (Liu et al. 2012), and visible oil was still present in this area of marsh as late as October 2013 (gomex.erma.noaa.gov/erma.html, 25-Mar-14 SCAT Oiling Ground Observations). Our fish collections did not start until 2012, but given that F. grandis do not exhibit large-scale movements (Nelson et al. 2014) and assuming they exhibit similar age and growth patterns (50 to 90 mm TL = 1- to 4-year-old fish) to F. heteroclitus (Fritz and Garside 1975; Dibble and Meyerson 2012), then all fish from this site should have been exposed to some oil throughout their lives.
Otoliths from F. grandis collected at the refinery site (RF) were also expected to exhibit chemical signatures indicative of oil exposure, because this site is located in a borrow ditch beside an oil refinery, where oil exposure is likely to be occurring continuously. Not finding elements in otoliths that were associated with oil exposure in AL sites (FA, PD) was not surprising, given that the amount of oiling in these marshes was light and patchy. Also, a similar study conducted in close proximity to our killifish collection sites in Alabama did not find an effect of DHOS oil on trace element or stable isotope uptake into oyster shells (Carmichael et al. 2012).
In contrast to our results, Morales-Nin et al. (2007) were able to detect an oil signature in Turbot (S. maximus) otoliths, perhaps because in their experiment oil was added directly to food sources; therefore, the level of contamination in the lab experiment was likely much higher than the levels experienced by fish in a natural environment. The differences in incorporation could also be based on physiological differences between Gulf killifish and turbot. Even species within a family have been shown to incorporate elements into the otolith differently (Rooker et al. 2004).
Another possible explanation as to why fish that were exposed to oil did not exhibit elevated levels of oil-related metals in their otoliths is the physiological regulation of trace elements and the pathway of otolith incorporation. It has been well documented that elements similar to Ca readily assimilate into the otolith aragonite matrix (e.g., Sr and Ba) through a substitution with Ca. This is because they are not bound elsewhere in the body and can easily pass from the water into the blood and then the endolymph (Sturrock et al. 2012). Therefore, these elements and resulting element/Ca ratios in the otolith exhibit similar concentration patterns to the surrounding water element/Ca ratios (Walther and Limburg 2012). Other trace metals, including many associated with oil (e.g., Zn, Cu, Pb, Se), can be bound to blood proteins and readily excreted through the liver or other pathways (Campana 1999; Sturrock et al. 2012), resulting in incorporation concentrations which could be much lower than ambient water compositions.
Differences Among Sites
Although differences among sites in whole otolith elemental and isotopic composition that could be attributed to oil exposure were not seen, Mn, Fe, Rb, Sr, Ba, and U concentrations differed significantly among sites. Manganese, Sr, and Ba contributed most to the distinction among both sites and regions in the linear discriminant function analyses, and paired sites within estuaries expressed similar concentrations of these elements. Differences in Sr, Ba, and Mn have also been used in many other studies (Thorrold et al. 1998; Campana et al. 2000; Gillanders and Kingsford 2003; Vasconcelos et al. 2007; Tanner et al. 2011) to classify fish to collection areas.
The regional LDFA was more successful and also had classification accuracies similar to other studies (Thorrold et al. 1998; Vasconcelos et al. 2007; Tanner et al. 2011), but the reason for the low percentage of classification accuracy in both the MS and AL east + FL grouping is that these regional groups exhibited similar otolith Sr, Ba, and Mn concentrations. Perhaps the addition of δ13C and δ18O stable isotope analysis, as well as high precision heavy isotope analysis, to our study could have improved the correct classification to these regions, given that the addition of these stable isotopes was able to improve the classification percentages of Thorrold et al. (1998) and Tanner et al. (2011).
Manganese concentrations were highest at the Fowl River site, followed by Fowl A and Fowl B, which is the main reason for the Alabama west grouping being identified in the cluster analysis and the regional LDFA. We expected that patterns of Mn concentrations in water samples would be similar to otolith concentrations, and while this was the case at one site (FR), correlation was low at the other two west Alabama sites (FA and FB). One plausible explanation for these discrepancies between water samples and otoliths as well as the high Mn concentrations in otoliths is the redox chemistry of Mn. When water is hypoxic, more free dissolved Mn is released into the water column and fish exposed to hypoxic waters have exhibited increased otolith Mn concentrations (Limburg et al. 2014). These conditions are probable for FR because low O2 and hypoxic conditions were observed in bottom waters near this site as part of another study, during the fall in both 2012 (1.72–2.75 mg/L) and 2013 (3.08 mg/L). Perhaps short-lived hypoxic events are also occurring at the FA and FB sites, although they have not been observed, or increased dissolved Mn concentrations could periodically be flowing down river to these sites. Also, given that our water sampling only occurred during each fish collection trip, there is a good possibility that it did not capture increased Mn events at the FA and FB sites.
Otolith core concentrations of Mn were also higher than edges at every site. This higher concentration of Mn in the otolith core has been documented previously in other species (Morales-Nin et al. 2005; Ruttenberg et al. 2005; Melancon et al. 2008) and is the reason why whole otolith Mn concentrations differed across sites, even though no site differences were found in otolith edges. Mn may prove valuable as a natural tag for estuarine nursery habitat for waters on the west side of Alabama, given that the otolith core would be the part of the structure analyzed to determine nursery habitat use.
86Sr and 88Sr were the second and third best discriminators among sites, respectively, with concentration patterns across sites being similar for both isotopes, as well as being similar to salinity patterns. This was expected given that a positive correlation between otolith Sr and salinity has been found previously (Bath et al. 2000; Lowe et al. 2009; Albuquerque et al. 2012). In addition, these published relationships show that otolith Sr concentrations approach asymptotic levels at around 15 ppt, which explains why Sr concentrations in the otolith were similar between PD and CK, even though salinities were higher at PD.
Barium was the best discriminator among sites and regions in the LDFAs, because Ba concentrations at the Louisiana sites (BB, BA) were much higher than all other sites. The primary source of Ba in estuaries has been shown to be riverine suspended particulate matter (SPM) (Hanor and Chan 1977; Coffey et al. 1997) and the Mississippi River is a very large drainage containing high SPM loads, resulting in high concentrations of Ba (Hanor and Chan 1977; Coffey et al. 1997). This occurred even when salinities around 15 ppt were experienced in the fall at the BA site, demonstrating that Ba concentration in otoliths is not purely a function of salinity. Given that fish collected in Louisiana had much higher otolith concentrations of Ba than at other sites, this could prove to be a reliable tag for nursery area discrimination or stock discrimination for members of a species that use estuarine waters close to the Mississippi River.
Conclusions
Mn, Sr, and Ba were the best discriminators among sites and the use of these elements led to a reasonably successful regional classification, particularly for the Louisiana sites. Mn may be useful as an otolith chemical tag to identify nursery habitat use in estuarine waters on the west side of Alabama, especially proximal to Fowl River. Also, Ba may prove to be a reliable otolith chemical tag for estuarine waters near the Mississippi River, providing a useful tool for nursery habitat identification and stock discrimination.
Otolith chemical signatures between paired oiled and non-oiled sites did not differ in trace elements likely associated with oil exposure, and the relative condition of F. grandis also did not differ between paired sites. Given previous published evidence that fish otoliths incorporate elements specific to oil exposure in the lab (Morales-Nin et al. 2007), our results support a minimal effect of the DHOS spill on F. grandis, a trend that has also been observed for other nearshore fauna (Fodrie and Heck 2011; Carmichael et al. 2012; Moody et al. 2013) in the northern Gulf of Mexico.
References
Abbriano, R.M., M.M. Carranza, S.L. Hogle, R.A. Levin, A.N. Netburn, K.L. Seto, S.M. Snyder, and P.J.S. Franks. 2011. Deepwater Horizon oil spill: a review of the planktonic response. Oceanography 24: 294–301.
Able, K.W., S.M. Hagan, and S.A. Brown. 2006. Habitat use, movement, and growth of young-of-the-year Fundulus spp. in southern New Jersey salt marshes: comparisons based on tag/recapture. Journal of Experimental Marine Biology and Ecology 335: 177–187.
Albuquerque, C.Q., N. Miekeley, J.H. Muelbert, B.D. Walther, and A.J. Jaureguizar. 2012. Estuarine dependency in a marine fish evaluated with otolith chemistry. Marine Biology 159: 2229–2239.
Ashford, J., and C. Jones. 2007. Oxygen and carbon stable isotopes in otoliths record spatial isolation of Patagonian toothfish (Dissostichus eleginoides). Geochimica Et Cosmochimica Acta 71: 87–94.
Bath, G.E., S.R. Thorrold, C.M. Jones, S.E. Campana, J.W. McLaren, and J.W.H. Lam. 2000. Strontium and barium uptake in aragonitic otoliths of marine fish. Geochimica Et Cosmochimica Acta 64: 1705–1714.
Bradbury, I.R., S.E. Campana, and P. Bentzen. 2008. Estimating contemporary early life-history dispersal in an estuarine fish: integrating molecular and otolith elemental approaches. Molecular Ecology 17: 1438–1450.
Brazner, J.C., S.E. Campana, and D.K. Tanner. 2004. Habitat fingerprints for Lake Superior coastal wetlands derived from elemental analysis of yellow perch otoliths. Transactions of the American Fisheries Society 133: 692–704.
Brownie, C., and D.D. Boos. 1994. Type I error robustness of ANOVA and ANOVA on ranks when the number of treatments is large. Biometrics 50: 542–549.
Buckel, J.A., B.L. Sharack, and V.S. Zdanowicz. 2004. Effect of diet on otolith composition in Pomatomus saltatrix, an estuarine piscivore. Journal of Fish Biology 64: 1469–1484.
Camilli, R., C.M. Reddy, D.R. Yoerger, B.A.S. Van Mooy, M.V. Jakuba, J.C. Kinsey, C.P. McIntyre, S.P. Sylva, and J.V. Maloney. 2010. Tracking hydrocarbon plume transport and biodegradation at Deepwater Horizon. Science 330: 201–204.
Campana, S.E. 1999. Chemistry and composition of fish otoliths: pathways, mechanisms and applications. Marine Ecology Progress Series 188: 263–297.
Campana, S.E., and J.D. Neilson. 1985. Microstructure of fish otoliths. Canadian Journal of Fisheries and Aquatic Sciences 42: 1014–1032.
Campana, S.E., and S.R. Thorrold. 2001. Otoliths, increments, and elements: keys to a comprehensive understanding of fish populations? Canadian Journal of Fisheries and Aquatic Sciences 58: 30–38.
Campana, S.E., A.J. Fowler, and C.M. Jones. 1994. Otolith elemental fingerprinting for stock identification of Atlantic cod (Gadus morhua) using laser-ablation ICPMS. Canadian Journal of Fisheries and Aquatic Sciences 51: 1942–1950.
Campana, S.E., G.A. Chouinard, J.M. Hanson, A. Frechet, and J. Brattey. 2000. Otolith elemental fingerprints as biological tracers of fish stocks. Fisheries Research 46: 343–357.
Carmichael, R.H., A.L. Jones, H.K. Patterson, W.C. Walton, A. Perez-Huerta, E.B. Overton, M. Dailey, and K.L. Willett. 2012. Assimilation of oil-derived elements by oysters due to the Deepwater Horizon oil spill. Environmental Science & Technology 46: 12787–12795.
Coffey, M., F. Dehairs, O. Collette, G. Luther, T. Church, and T. Jickells. 1997. The behaviour of dissolved barium in estuaries. Estuarine, Coastal and Shelf Science 45: 113–121.
Courtenay, S.C., K.R. Munkittrick, H.M.C. Dupuis, R. Parker, and J. Boyd. 2002. Quantifying impacts of pulp mill effluent on fish in Canadian marine and estuarine environments: problems and progress. Water Quality Research Journal of Canada 37: 79–99.
De Almeida, C.M.S., A.S. Ribeiro, T.D. Saint’Pierre, and N. Miekeley. 2009. Studies on the origin and transformation of selenium and its chemical species along the process of petroleum refining. Spectrochimica Acta Part B: Atomic Spectroscopy 64: 491–499.
Dibble, K.L., and L.A. Meyerson. 2012. Tidal flushing restores the physiological condition of fish residing in degraded salt marshes. Plos One 7: e46161. doi:10.1371/journal.pone.0046161.
Dibble, K.L., and L.A. Meyerson. 2014. The effects of plant invasion and ecosystem restoration on energy flow through salt marsh food webs. Estuaries and Coasts 37: 339–353.
Doubleday, Z.A., H.H. Harris, C. Izzo, and B.M. Gillanders. 2014. Strontium randomly substituting for calcium in fish otolith aragonite. Analytical Chemistry 86: 865–869.
Ellis, J., C. Rechsteiner, M. Moir, and S. Wilbur. 2011. Determination of volatile nickel and vanadium species in crude oil and crude oil fractions by gas chromatography coupled to inductively coupled plasma mass spectrometry. Journal of Analytical Atomic Spectrometry 26: 1674–1678.
Farmer, T.M., D.R. DeVries, R.A. Wright, and J.E. Gagnon. 2013. Using seasonal variation in otolith microchemical composition to indicate largemouth bass and southern flounder residency patterns across an estuarine salinity gradient. Transactions of the American Fisheries Society 142: 1415–1429.
Farrell, J., and S.E. Campana. 1996. Regulation of calcium and strontium deposition on the otoliths of juvenile tilapia, Oreochromis niloticus. Comparative Biochemistry and Physiology. Part A, Physiology 115: 103–109.
Ferguson, G.J., T.M. Ward, and B.M. Gillanders. 2011. Otolith shape and elemental composition: Complementary tools for stock discrimination of mulloway (Argyrosomus japonicus) in southern Australia. Fisheries Research 110: 75–83.
Finley, M.A., S.C. Courtenay, K.L. Teather, and M.R. van den Heuvel. 2009. Assessment of northern mummichog (Fundulus heteroclitus macrolepidotus) as an estuarine pollution monitoring species. Water Quality Research Journal of Canada 44: 323–332.
Fodrie, F.J., and K.L. Heck. 2011. Response of coastal fishes to the Gulf of Mexico oil disaster. Plos One 6: e21609. doi:10.1371/journal.pone.0021609.
Fritz, E.S., and E.T. Garside. 1975. Comparison of age composition, growth, and fecundity between 2 populations each of Fundulus heteroclitus and Fundulus diaphanus (Pisces-Cyprinodontidae). Canadian Journal of Zoology-Revue Canadienne De Zoologie 53: 361–369.
Gillanders, B.M., and M.J. Kingsford. 2003. Spatial variation in elemental composition of otoliths of three species of fish (family Sparidae). Estuarine, Coastal and Shelf Science 57: 1049–1064.
Gonzalez, I., M. Levin, S. Jermanus, B. Watson, and M.R. Gilg. 2009. Genetic assessment of species ranges in Fundulus heteroclitus and F-grandis in northeastern Florida salt marshes. Southeastern Naturalist 8: 227–244.
Greeley, M.S., and R. MacGregor. 1983. Annual and semilunar reproductive cycles of the gulf killifish, Fundulus grandis, on the Alabama gulf coast. Copeia 3: 711–718.
Grosser, Z.A., D. Bass, L. Foglio, and L. Davidowski. 2012. Determination of metals as markers oil. Contamination in seafood by ICP-MS. Agro Food Industry Hi-Tech 23: 24–26.
Hanor, J.S., and L.H. Chan. 1977. Non-conservative behavior of barium during mixing of Mississippi River and Gulf of Mexico waters. Earth and Planetary Science Letters 37: 242–250.
Kahn, A., and G.D. Rayner. 2003. Robustness to non-normality of common tests for the many-sample location problem. Journal of Applied Mathematics and Decision Sciences 7: 187–206.
Kennedy, B.P., J.D. Blum, C.L. Folt, and K.H. Nislow. 2000. Using natural strontium isotopic signatures as fish markers: methodology and application. Canadian Journal of Fisheries and Aquatic Sciences 57: 2280–2292.
Kneib, R.T., and A.E. Stiven. 1978. Growth, reproduction, and feeding of Fundulus heteroclitus (L) on a North Carolina salt marsh. Journal of Experimental Marine Biology and Ecology 31: 121–140.
Limburg, K.E., B.D. Walther, Z. Lu, G. Jackman, J. Mohan, Y. Walther, A. Nissling, P.K. Weber, and A.K. Schmitt. 2014. In search of the dead zone: use of otoliths for tracking fish exposure to hypoxia. Journal of Marine Systems. doi:10.1016/j.jmarsys.2014.02.014.
Liu, Z. F., J. Q. Liu, Q. Z. Zhu, and W. Wu. 2012. The weathering of oil after the Deepwater Horizon oil spill: insights from the chemical composition of the oil from the sea surface, salt marshes and sediments. Environmental Research Letters 7. doi:10.1088/1748-9326/1087/1083/035302
Longerich, H.P., S.E. Jackson, and D. Gunther. 1996. Laser ablation inductively coupled plasma mass spectrometric transient signal data acquisition and analyte concentration calculation. Journal of Analytical Atomic Spectrometry 11: 899–904.
Longmore, C., K. Fogarty, F. Neat, D. Brophy, C. Trueman, A. Milton, and S. Mariani. 2010. A comparison of otolith microchemistry and otolith shape analysis for the study of spatial variation in a deep-sea teleost, Coryphaenoides rupestris. Environmental Biology of Fishes 89: 591–605.
Lotrich, V.A. 1975. Summer home range and movements of Fundulus heteroclitus (Pisces Cyprinodontidae) in a tidal creek. Ecology 56: 191–198.
Lowe, M.R., D.R. DeVries, R.A. Wright, S.A. Ludsin, and B.J. Fryer. 2009. Coastal largemouth bass (Micropterus salmoides) movement in response to changing salinity. Canadian Journal of Fisheries and Aquatic Sciences 66: 2174–2188.
Lowe, M.R., D.R. DeVries, R.A. Wright, S.A. Ludsin, and B.J. Fryer. 2011. Otolith microchemistry reveals substantial use of freshwater by southern flounder in the northern Gulf of Mexico. Estuaries and Coasts 34: 630–639.
Ludden, J.N., R. Feng, G. Gauthier, J. Stix, L. Shi, D. Francis, N. Machado, and G.P. Wu. 1995. Applications of LAM-ICP-MS analysis to minerals. Canadian Mineralogist 33: 419–434.
Ludsin, S.A., B.J. Fryer, and J.E. Gagnon. 2006. Comparison of solution-based versus laser ablation inductively coupled plasma mass spectrometry for analysis of larval fish otolith microelemental composition. Transactions of the American Fisheries Society 135: 218–231.
Melancon, S., B.J. Fryer, J.E. Gagnon, and S.A. Ludsin. 2008. Mineralogical approaches to the study of biomineralization in fish otoliths. Mineralogical Magazine 72: 627–637.
Miekeley, N., R.C. Pereira, E.A. Casartelli, A.C. Almeida, and M.D. Carvalho. 2005. Inorganic speciation analysis of selenium by ion chromatography-inductively coupled plasma-mass spectrometry and its application to effluents from a petroleum refinery. Spectrochimica Acta Part B: Atomic Spectroscopy 60: 633–641.
Moody, R.M., J. Cebrian, and K.L. Heck Jr. 2013. Interannual recruitment dynamics for resident and transient marsh species: evidence for a lack of impact by the Macondo oil spill. Plos One 8: e58376. doi:10.1371/journal.pone.0058376.
Morales-Nin, B., S.C. Swan, J.D.M. Gordon, M. Palmer, A.J. Geffen, T. Shimmield, and T. Sawyer. 2005. Age-related trends in otolith chemistry of Merluccius merluccius from the north-eastern Atlantic Ocean and the western Mediterranean Sea. Marine and Freshwater Research 56: 599–607.
Morales-Nin, B., A.J. Geffen, F. Cardona, C. Kruber, and F. Saborido-Rey. 2007. The effect of Prestige oil ingestion on the growth and chemical composition of turbot otoliths. Marine Pollution Bulletin 54: 1732–1741.
Nelson, T.R., D. Sutton, and D. DeVries. 2014. Summer movements of the Gulf Killifish (Fundulus grandis) in a northern Gulf of Mexico salt marsh. Estuaries and Coasts. doi:10.1007/s12237-12013-19762-12235.
Neumann, R.M., C.S. Guy, and D.W. Willis. 2012. Length, weight, and associated indices. In Fisheries techniques, 3rd ed, ed. A.V. Zale, D.L. Parrish, and T.M. Sutton, 637–676. Bethesda, Maryland: American Fisheries Society.
Niklitschek, E.J., D.H. Secor, P. Toledo, A. Lafon, and M. George-Nascimento. 2010. Segregation of SE Pacific and SW Atlantic southern blue whiting stocks: integrating evidence from complementary otolith microchemistry and parasite assemblage approaches. Environmental Biology of Fishes 89: 399–413.
Oros, D.R., J.R.M. Ross, R.B. Spies, and T. Mumley. 2007. Polycyclic aromatic hydrocarbon (PAH) contamination in San Francisco Bay: a 10-year retrospective of monitoring in an urbanized estuary. Environmental Research 105: 101–118.
Reay, W.G. 2009. Water quality within the York River estuary. Journal of Coastal Research 57: 23–39.
Rooker, J.R., R.T. Kraus, and D.H. Secor. 2004. Dispersive behaviors of black drum and red drum: is otolith Sr : Ca a reliable indicator of salinity history? Estuaries 27: 334–341.
Rooker, J.R., L.L. Kitchens, M.A. Dance, R.J.D. Wells, B. Falterman, and M. Cornic. 2013. Spatial, temporal, and habitat-related variation in abundance of pelagic fishes in the Gulf of Mexico: potential implications of the Deepwater Horizon oil spill. Plos One 8: e76080. doi:10.1371/journal.pone.0076080.
Rozas, L.P., and M.W. LaSalle. 1990. A comparison of the diets of gulf killifish, Fundulus grandis Baird and Girard, entering and leaving a Mississippi brackish marsh. Estuaries 13: 332–336.
Rozas, L.P., and D.J. Reed. 1993. Nekton use of marsh-surface habitats in Louisiana (USA) deltaic salt marshes undergoing submergence. Marine Ecology-Progress Series 96: 147–157.
Rozas, L.P., and R.J. Zimmerman. 2000. Small-scale patterns of nekton use among marsh and adjacent shallow nonvegetated areas of the Galveston Bay Estuary, Texas (USA). Marine Ecology-Progress Series 193: 217–239.
Ruttenberg, B.I., S.L. Hamilton, M.J.H. Hickford, G.L. Paradis, M.S. Sheehy, J.D. Standish, O. Ben-Tzvi, and R.R. Warner. 2005. Elevated levels of trace elements in cores of otoliths and their potential for use as natural tags. Marine Ecology Progress Series 297: 273–281.
Shaheen, M., J.E. Gagnon, Z. Yang, and B.J. Fryer. 2008. Evaluation of the analytical performance of femtosecond laser ablation inductively coupled plasma mass spectrometry at 785 nm with glass reference materials. Journal of Analytical Atomic Spectrometry 23: 1610–1621.
Shaw, R.G., and T. Mitchellolds. 1993. ANOVA for unbalanced data—an overview. Ecology 74: 1638–1645.
Sohn, D., S. Kang, and S. Kim. 2005. Stock identification of chum salmon (Oncorhynchus keta) using trace elements in otoliths. Journal of Oceanography 61: 305–312.
Speed, F.M., R.R. Hocking, and O.P. Hackney. 1978. Methods of analysis of linear-models with unbalanced data. Journal of the American Statistical Association 73: 105–112.
Statham, P.J. 2012. Nutrients in estuaries—an overview and the potential impacts of climate change. Science of the Total Environment 434: 213–227.
Sturrock, A.M., C.N. Trueman, A.M. Darnaude, and E. Hunter. 2012. Can otolith elemental chemistry retrospectively track migrations in fully marine fishes? Journal of Fish Biology 81: 766–795.
Szedlmayer, S.T., and P.A. Mudrak. 2014. Influence of age-1 conspecifics, sediment type, dissolved oxygen, and the Deepwater Horizon oil spill on the recruitment of age-0 red snapper in the Northeast Gulf of Mexico during 2010 and 2011. North American Journal of Fisheries Management 34: 443–452.
Tanner, S.E., R.P. Vasconcelos, P. Reis-Santos, H.N. Cabral, and S.R. Thorrold. 2011. Spatial and ontogenetic variability in the chemical composition of juvenile common sole (Solea solea) otoliths. Estuarine, Coastal and Shelf Science 91: 150–157.
Teo, S.L.H., and K.W. Able. 2003. Habitat use and movement of the mummichog (Fundulus heteroclitus) in a restored salt marsh. Estuaries 26: 720–730.
Thorrold, S.R., C.M. Jones, P.K. Swart, and T.E. Targett. 1998. Accurate classification of juvenile Weakfish Cynoscion regalis to estuarine nursery areas based on chemical signatures in otoliths. Marine Ecology-Progress Series 173: 253–265.
Vasconcelos, R.P., P. Reis-Santos, S. Tanner, V. Fonseca, C. Latkoczy, D. Gunther, M.J. Costa, and H. Cabral. 2007. Discriminating estuarine nurseries for five fish species through otolith elemental fingerprints. Marine Ecology-Progress Series 350: 117–126.
Walther, B.D., and K.E. Limburg. 2012. The use of otolith chemistry to characterize diadromous migrations. Journal of Fish Biology 81: 796–825.
Walther, B.D., and S.R. Thorrold. 2006. Water, not food, contributes the majority of strontium and barium deposited in the otoliths of a marine fish. Marine Ecology-Progress Series 311: 125–130.
Walther, B.D., T. Dempster, M. Letnic, and M.T. McCulloch. 2011. Movements of diadromous fish in large unregulated tropical rivers inferred from geochemical tracers. Plos One 6(4): e18351. doi:10.1371/journal.pone.0018351.
Wang, Y., X. Li, B.H.H. Li, Z.Y.Y. Shen, C.H.H. Feng, and Y.X.X. Chen. 2012. Characterization, sources, and potential risk assessment of PAHs in surface sediments from nearshore and farther shore zones of the Yangtze estuary, China. Environmental Science and Pollution Research 19: 4148–4158.
Ward, J.H. 1963. Hierarchical grouping to optimize an objective function. Journal of the American Statistical Association 58: 236–244.
Whitehead, A. 2010. The evolutionary radiation of diverse osmotolerant physiologies in killifish (Fundulus sp.). Evolution 64: 2070–2085.
Whitehead, A., B. Dubansky, C. Bodinier, T.I. Garcia, S. Miles, C. Pilley, V. Raghunathan, J.L. Roach, N. Walker, R.B. Walter, C.D. Rice, and F. Galvez. 2012. Genomic and physiological footprint of the Deepwater Horizon oil spill on resident marsh fishes. Proceedings of the National Academy of Sciences 109: 20298–20302.
Wishart, D. 1969. An algorithm for hierarchical classifications. Biometrics 25: 165–170.
Wolff, B.A., B.M. Johnson, A.R. Breton, P.J. Martinez, and D.L. Winkelman. 2012. Origins of invasive piscivores determined from the strontium isotope ratio (Sr-87/Sr-86) of otoliths. Canadian Journal of Fisheries and Aquatic Sciences 69: 724–739.
Zeigler, J.M., and G.W. Whitledge. 2010. Otolith trace element and stable isotopic compositions differentiate fishes from the Middle Mississippi River, its tributaries, and floodplain lakes. Hydrobiologia 661: 289–302.
Acknowledgments
We would like to thank Dr. Ash Bullard for the help with site selection and sampling protocol. We would also like to thank all of the people who helped with fish collections, Adrian Stanfill, Emily DeVries, Brooke Merrill, Carl Klimah, Jenny Herbig, Danielle Horn, Jay Foster, and Kyle Gallagher. Special thanks go to Tammy DeVries for spending countless hours pulling miniscule otoliths out of killifish and having to deal with strong H2O2 for cleaning purposes. Finally, we would like to thank Dr. Mohamed Shaheen of the University of Windsor GLIER for the help with the LA-ICP-MS. Funding for this research was provided by British Petroleum.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by Lawrence P. Rozas
Electronic supplementary material
Below is the link to the electronic supplementary material.
Supplementary Table 1
(DOCX 18 kb)
Rights and permissions
About this article
Cite this article
Nelson, T.R., DeVries, D.R., Wright, R.A. et al. Fundulus grandis Otolith Microchemistry as a Metric of Estuarine Discrimination and Oil Exposure. Estuaries and Coasts 38, 2044–2058 (2015). https://doi.org/10.1007/s12237-014-9934-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12237-014-9934-y