Abstract
Understanding the role different habitats play in the life histories of fishes is important for the development of holistic aquatic ecosystem management plans. We used otolith trace element analysis to reconstruct the life history of estuarine triplefin Forsterygion nigripenne and infer its habitat use. Analysis of otoliths using laser ablation inductively coupled plasma mass spectrometry showed an elemental profile without prominent changes in elemental concentration. The otolith elemental profiles were not indicative of movement between waters with substantial Sr:Ca and Ba:Ca differences. The Sr:Ca and Ba:Ca profiles were indicative of within—and near—estuary movements encompassing short time periods. Overall, the otolith microchemical profile was likely indicative of a life history closely associated with estuaries. However, it is difficult to rule out brief excursions to the marine environment given that may not have been of sufficient duration to be recorded in the elemental composition of the otolith. This suggests that F. nigripenne is the only species in southern New Zealand known to be closely associated with estuarine habitat for its entire life cycle.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Understanding the ways in which fish use different habitats is vital for effective aquatic ecosystem management, and the mitigation of threats facing these habitats through well-informed conservation practices (Peterson et al. 2000; Whitfield 2002; Courrat et al. 2009; Delpech et al. 2010). Estuaries in particular play an essential role in supporting many fish species, contributing in different ways to a variety of life history strategies (Whitfield 1999; Potter et al. 2001; Elliott et al. 2007; Hoeksema et al. 2009). Estuaries can serve as nursery habitats for fish from both freshwater and saltwater environments because of their high productivity and capacity to act as refuges from predators and adverse environmental conditions (Paterson and Whitfield 2000; Beck et al. 2001). Whilst many fish species only make transitory use of estuaries as migration routes, some physiologically tolerant species complete their entire life cycle within estuaries (Elliott et al. 2007).
A range of techniques can be used to study how fish utilize estuaries, including tissue stable isotope analysis, otolith microchemistry, and acoustic/radio telemetry (Gillanders and Kingsford 2000; Childs et al. 2008; Fry and Chumchal 2011). As each of these techniques collect different types of information, the most appropriate approach is dependent on the objectives of the study. However, the information that tissue stable isotope analysis provides is limited to relatively short time periods due to tissue turnover rates (Buchheister and Latour 2010). The use of electronic tagging is restricted to post-juvenile and adult life stages of larger-bodied species, as the relatively large tags make it difficult or impossible to use the technique on early life-stages and small individuals (Miles et al. 2013; Dawson et al. 2015), which is problematic given that many larval and juvenile fish either rear in estuaries or briefly migrate through them. Otoliths, however, record ambient environmental conditions continuously over the lifetime of an individual fish, potentially enabling an understanding of migration patterns and habitat use of individuals of all sizes and across all life stages (Campana 1999; Elsdon et al. 2008).
The use of otolith trace element analysis to elucidate the life history of a fish depends on the predictability of the relationship that exists between the otolith chemical composition of the species in question and the ambient water chemistry (Elsdon et al. 2008). It is generally assumed that ratios of trace elements to calcium (Ca), particularly strontium (Sr) and barium (Ba), are deposited chronologically into the otolith material consistently relative to ambient concentrations in the surrounding water (Izzo et al. 2018). Therefore, otolith Sr:Ca and Ba:Ca have been used as tracers of the past habitat use by estuarine fish (Gillanders and Kingsford 2000; Gillanders 2002a; Dorval et al. 2005b; Schilling et al. 2018). The predictive relationships between trace elements (i.e., Sr and Ba) and salinity where high Sr/low Ba associated with seawater, and low Sr/high Ba associated with freshwater habitats are often used to reconstruct fish life histories (Elsdon et al. 2008).
Estuarine triplefin Forsterygion nigripenne is a species endemic to New Zealand that occurs in estuaries and coastal areas, including the Chatham Islands (Francis et al. 2005; Wellenreuther et al. 2007; Feary et al. 2009; Kohn and Clements 2011). F. nigripenne spawns in nests built on shallow and sheltered habitats wherein mud and sand are the dominant substrates (Wellenreuther and Clements 2007). This species is reported to exhibit 69 days of mean pelagic larval duration (PLD) before settlement to the adult habitat (Kohn and Clements 2011), a relatively long PLD which provides the potential to promote the dispersal of larvae between estuaries through the marine environment. However, gene flow studies indicate that F. nigripenne has limited dispersal potential and low migration rates (Hickey et al. 2009; Kohn and Clements 2011), suggesting the possibility that they remain within estuaries throughout their life cycle. Nevertheless, studies on estuaries located on the South Island of New Zealand have suggested that the life history strategy of F. nigripenne comprises a marine larval/juvenile phase (Jellyman et al. 1997; Sutherland and Closs 2001).
A year-round monthly sampling programme on one of the permanently open estuaries in Otago, southern New Zealand (Waikouaiti Estuary) indicated that F. nigripenne occurs in large numbers in the estuary throughout the year, from settlement-age larvae with a translucent body and well-developed fins, to post-spawning adults (Taddese and Closs 2020). Conversely, in a recent study, F. nigripenne was found to be absent in intermittently closed and open southeastern New Zealand lakes or lagoons (ICOLLs) (Taddese et al. 2018). Thus, whilst it is known that F. nigripenne utilize estuaries for much of their life cycle, the precise details of its use across all life history stages are not understood. Hence, this study aimed to use otolith microchemistry to reconstruct the life history of F. nigripenne to better understand its habitat use over its full life history. Parameters estimated from otolith microstructural analyses were used to compliment understanding of the habitat use patterns of F. nigripenne.
A previous laboratory experiment indicated that otolith trace element analysis is a reliable technique to understand movements of F. nigripenne between waters of different Sr:Ca and Ba:Ca compositions. The results of the experiment showed incorporation of Sr:Ca and Ba:Ca into F. nigripenne otolith is linearly and positively related to their respective values in the ambient water (Taddese et al. 2019). As otolith elemental signatures collected near the core and the edge reflect early life history and habitat use at the time of catch, respectively, (Jessop et al. 2012), we hypothesize that relatively higher Sr:Ca and lower Ba:Ca, indicative of pelagic larval life in the marine environment, will be observed in areas of F. nigripenne otoliths closer to the core. Lower Sr:Ca and higher Ba:Ca relative to the core region will be recorded on the outer edge of F. nigripenne otolith indicating increased exposure to freshwater inputs, suggesting either the use of estuarine, or near-estuarine coastal habitat subject to the influence of freshwater inputs.
Materials and methods
Fish sampling
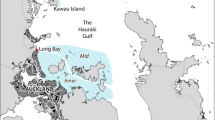
Juveniles of estuarine triplefin were collected using a seine net (L × H = 14 × 1 m; 10-mm mesh size) from the middle and lower sections of two permanently open estuaries located on the coast of Otago (South Island, New Zealand; Fig. 1): Waikouaiti estuary and Tokomairiro estuary. The areas of the Waikouaiti and Tokomairiro estuaries are estimated to be 1.41 km2 and 17.5 km2, respectively. Average salinity at the lower section of the Waikouaiti estuary ranges from 19 (winter) to 28 (summer) (Taddese and Closs 2020). On the other hand, salinity in the lower section of the Tokomairiro estuary fluctuates between 19 and 6, during summer and winter months respectively (Taddese 2020 Unpubl.). Fish [TL mm, 41.8 ± 12 (mean ± SD)] from the Waikouaiti (N = 25) and Tokomairiro (N = 27) were collected in August and November 2016, respectively. Fish were sampled from both the middle (Waikouaiti) and lower (Tokomairiro) reaches of estuaries. Sampling locations were chosen based on prior assessment of the two estuaries (see Taddese et al. 2018). Individuals were euthanized with an overdose of AQUI-S 20E fish anesthetic (AQUI-S New Zealand Ltd, Lower Hutt, New Zealand) and preserved in 70% ethanol for subsequent otolith extraction and analyses.
Otolith extraction, preparation, and analysis
Sagittal otoliths were extracted, cleaned with a nylon brush, air-dried, and stored dry in individual microcentrifuge tubes. Tweezers with plastic tips were used to handle otoliths during extraction and mounting. The tweezers were rinsed with ultrapure water and dried with lint-free wipes before and after handling otoliths from different sites. After extraction, both otoliths of each fish were mounted with the proximal surface upward on a microscope slide using Crystalbond 509 thermoplastic adhesive (SPI Supplies, West Chester, PA), and then polished with 9 µm and 3 µm 3M™ diamond lapping films that are made of polyester until the core was exposed. Once polished, one sagittal otolith from each fish was transferred and mounted on another microscope slide using double-sided adhesive tape in preparation for laser ablation inductively coupled plasma mass spectrometry (LA–ICP–MS) analysis.
The second sagittal otolith was used to determine age using light microscopy (Olympus® Corporation, Tokyo, Japan) to count daily increments. Images of otoliths were taken by an Olympus SC100 camera mounted on a light microscope and examined using ImageJ (version 1.50, National Institutes of Health, USA) image analysis software. Increments were also counted directly using a microscope when they were difficult to count using image analysis. F. nigripenne otolith microstructure profile is known to show three distinct regions comprising (1) narrow light-colored increments close to the nucleus that become wider and darker in the middle, and (2) a conspicuous dark increment, followed by (3) narrower and lighter increments near the outer edge (Kohn and Clements 2011). The conspicuous dark increment that lies next to the intermediate increments is considered to be a settlement mark (Fig. 2; Kohn and Clements 2011). Hatch, settlement date, and PLD were back-calculated using the settlement mark as a reference from 47 otoliths (N = 23, Waikouaiti; N = 24, Tokomairiro). As five otoliths (two from Waikouaiti and three from Tokomairiro) were damaged during preparation, a total of 47 otoliths were used in microstructural analyses from both estuaries. Each otolith was aged twice with two non-sequential randomized, independent reads by the same reader, and the average percent error was calculated. Since no sample ages exceeded 5% error, all 47 samples were included in the results.
Image of a polished Forsterygion nigripenne otolith. Gray arrow with black border, wider growth increments before the settlement mark; Black arrow, settlement mark; White arrow, narrower growth increments after the settlement mark. The white broken arrow drawn from the primordium to the posterior end indicates the laser ablation path
Data for eight elements (7Li, 23Na, 24Mg, 25Mg, 55Mn, 31P, 39 K, 88Sr, 137Ba and 138Ba), as well as 43Ca were quantified using an Agilent 7900 ICP-MS coupled to an Australia Scientific Instruments (ASI) RESOlution M-50 laser ablation system powered by a Coherent 193 nm ArF excimer laser. The laser ablated along a predetermined linear transect from the primordium to the otolith margin with a spot size of 50 µm and scan speed of 5 µm/s. Laser repetition rate was set at 7 Hz and on-sample fluence was 2.5 J/cm2. A 20 s gas blank was collected prior to each sample, and certified reference materials (the NIST 610 and NIST 612 glass standards, and the calcium carbonate pellet USGS MACS-3) were ablated every 7–8 samples to allow for correction for any background element counts as well as instrument drift and bias. Each otolith was pre-ablated three times to avoid potential surface contamination from sample preparation.
Data processing
Data were processed using the Trace_Elements_IS data reduction scheme in Iolite software (version 3.6) run on Igor Pro 6.37 to convert the raw count rate data to element concentrations after normalization to Ca and calibration with NIST 610. Standard reference materials (NIST 612 and MACS-3) were used to assess the linearity, accuracy, and precision of the measurements (see Online resource, Supplementary Material 1). The results for most elements were within < 3% and < 10% of published values of NIST 612 and MACS-3, respectively (Pearce et al. 1997; Jochum et al. 2012, USGS 2012). All the measurements were above the instrument limit of detection (see Online resource, Supplementary Material 2).
Comparison of mean element to Ca (Me:Ca) values before and after the settlement mark enabled comparison of larval and adult habitat signatures. However, the use of the settlement mark as a borderline between otolith chemical signatures from early and adult life stages can be obscured by the lag effect of elemental uptake in otoliths (see Elsdon and Gillanders 2005). Hence, mean ± SD of Me:Ca from the first and last 50 µm of otolith transects were taken to approximate the values representing the pre and post-settlement periods consistently. Data were smoothed using a three-point moving median.
Data analysis
After conversion to element concentrations and normalization to Ca, the multi-element data were subjected to multivariate statistical analyses using the PRIMER-e multivariate statistical package (version 7; Quest Research Limited, Auckland, New Zealand). Principal coordinates analysis (PCO) was performed using the Euclidean resemblance matrix constructed from a square-root transformed data to explore sample groupings. A cluster analysis was performed to cross-check sample groupings on the PCO plot. A similarity profile test (SIMPROF) was used to identify statistically significant sample groupings by overlaying statistically different SIMPROF clusters on the PCO plot. Although they showed similar patterns with thier respective isotopes, 25Mg and 137Ba were excluded from the PCO to avoid redundancy. The pre- and post-settlement otolith Sr:Ca and Ba:Ca signatures were compared using the Mann–Whitney U test. Violations of parametric test assumptions necessitated the use of the Mann–Whitney U test.
Results
Hatch and settlement months
The mean ± SD PLD of F. nigripenne was 48.6 ± 11 and 40 ± 6.8 days for fish collected from Waikouaiti and Tokomairiro, respectively (Fig. 3a, b). Most fish from the Waikouaiti settled in their habitats in June and July (Fig. 3c, d), whereas the settlement months for most samples from Tokomairiro were September and October. Hatch periods of fish from the Waikouaiti were evenly distributed across April to June (Fig. 3e, f). In Tokomairiro, most fish hatched in July and August. The estimated age was 97.4 ± 22.9 (mean ± SD) and 98.7 ± 31.8 (mean ± SD) days for samples collected from the Waikouaiti and Tokomairiro estuaries, respectively.
Otolith chemistry
Overall, the otolith chemistry data of F. nigripenne showed Sr:Ca and Ba:Ca profiles without prominent changes in elemental concentrations across the core to edge transect (Figs. 4, 5). However, in some otoliths from the Waikouaiti estuary, smaller Ba:Ca peaks covering short time periods were evident nearer the otolith edge (Fig. 4a, d). In contrast, in some otoliths from the Tokomariro estuary the Sr:Ca and Ba:Ca ratios co-fluctuated along the transect (Fig. 5a–e).
The PCO analysis revealed that the first two principal coordinates (PCO1 and PCO2) contributed 65.9% the explained variance in the data. Specifically, PCO1 and PCO2 contributed 46.9% and 19% of the explained variance in the data. Otolith Me:Ca values were grouped between estuaries and pre- and post-settlement mark otolith regions (Fig. 6).The pre-settlement otolith Me:Ca values of fish from the Waikouaiti estuary were significantly different from the Tokomairiro values. The post-settlement otolith Me:Ca values from both estuaries overlapped to a large extent on the PCO plot. However, some of the post-settlement otolith Me:Ca values from the Waikouaiti estuary were significantly different from the Tokomairiro values.
Principal coordinates (PCO) analysis plot of Me:Ca values quantified from pre- and post-settlement mark parts of otoliths collected from two estuaries [i.e., Waikouaiti (Wk) and Tokomairiro (Toko)]. The plot is based on a resemblance matrix constructed from a square-root transformed data. Each circle represents a significantly different group as identified by the similarity profile (SIMPROF) test (see the supplementary material)
Sr:Ca and Ba:Ca values from the outer edge region were higher than the values from the core region of otoliths. The pre-settlement period Sr:Ca ratios ranged from 2.1 mmol/mol to 3.2 mmol/mol for fish collected from the Waikouaiti (Fig. 7a), whereas the post-settlement Sr:Ca values were between 2.9 mmol/mol to 4.7 mmol/mol (Fig. 7a). Ranges of pre-and post-settlement Ba:Ca were between 0.3 µmol/mol to 1.5 µmol/mol and between 0.5 µmol/mol to 4.7 µmol/mol, respectively for samples collected from the Waikouaiti (Fig. 7b). Similarly, the pre-and post-settlement Sr:Ca values of fish sampled from the Tokomairiro varied between 2.4 mmol/mol to 3.6 mmol/mol and 2.9 mmol/mol to 4.6 mmol/mol, respectively (Fig. 7a). Ba:Ca ranged from 0.3 µmol/mol to 2.1 µmol/mol and from 0.8 µmol/mol to 2.1 µmol/mol for samples collected from the Tokomairiro (Fig. 7b). The results of the Mann–Whitney U test supported the differences between the pre- and post-settlement mark otolith Sr:Ca and Ba:Ca values (Table 1).
Box plots show the pre-settlement and post-settlement otolith Sr:Ca (a) and Ba:Ca (b) of Forsterygion nigripenne collected from the Waikouaiti (N = 25) and Tokomairiro (N = 27) estuaries. The boxes show the interquartile range, with the median value indicated by the horizontal line; whiskers show the range. LH life history, WK Waikouaiti estuary, TK Tokomairiro estuary, Pre pre-settlement mark, post post-settlement mark, SrCa Sr:Ca (mmol/mol), BaCa, Ba:Ca (µmol/mol)
Discussion
Other life history parameters inferred from otolith microstructure (i.e., time of hatch and settlement month) were indicative of continuous recruitment of juveniles within estuaries over an extended period. This pattern aligns well with the consistent presence of transparent juveniles with fully developed fins at the middle section of the Waikouaiti (Taddese 2019) and other estuaries (Jellyman et al. 1997; Sutherland and Closs 2001) between January and June. Fish sampling time difference between the Waikouaiti and Tokomairiro caused slight variation in settlement and hatch months among samples from both sites. Fish were collected in August and November from the Waikouaiti and Tokomairiro estuaries, respectively. This was reflected in settlement months, whereby most fish from the Waikouaiti settled in June and July, and those from the Tokomairiro settled in September and October. The average PLD observed in this study (44.1 ± 9.9 days, mean ± SD) was also slightly lower than the previously reported PLD in the North Island, New Zealand (69 days, Kohn and Clements 2011). Given the association of cold temperature with longer larval life (Green and Fisher 2004), PLD was expected to be longer in the South Island where temperature is relatively lower. Therefore, the lower PLD of F. nigripenne observed in the present study could be a result of other biotic and abiotic factors (e.g., food availability) that affect larval traits. It is known that higher food abundance can increase in larval growth rate (Claramunt and Wahl 2000). It is likely that larvae that grow faster will be able to settle in the adult habitat relatively faster than those which grow slowly (e.g., Kaemingk et al. 2019).
Our findings showed that the changes in magnitudes of otolith element to Ca (Me:Ca) across transects of F. nigripenne otoliths were not substantial enough to suggest movement across waters of markedly different Sr:Ca and Ba:Ca values. The most plausible explanation for this steady otolith elemental profile between different life stages is either continuous estuarine residency, with perhaps some movement into coastal habitat closely associated with, and strongly influenced by, estuarine inputs. In a previous experiment, otolith elemental profiles of juveniles of this species reared in water spiked with different Sr and Ba concentrations showed significant differences in otolith Sr:Ca and Ba:Ca values (Taddese et al. 2019). Therefore, given that otolith elemental uptake in F. nigripenne is directly related to the concentrations in the ambient water, the general otolith Me:Ca profile observed in this study was indicative of a life history closely associated with estuaries. The absence of marked variations in Sr:Ca and Ba:Ca along the otolith transects suggests that F. nigripenne is likely associated with estuaries over its entire life history, and is perhaps the only species with such a life history strategy in southern New Zealand.
If the larval life stage of F. nigripenne involved movement to the wider marine environment for long periods, the pre-settlement Me:Ca profile would have shown higher Sr:Ca and lower Ba:Ca values than the post-settlement values, such as those seen in species undertaking an amphidromous migration, passing from the marine environment through the estuary and into freshwater (see example profiles in Hicks et al. 2017). Alternatively, Me:Ca signatures close to the core would have shown higher Ba:Ca and lower Sr:Ca ratios had the early life history of F. nigripenne consisted of a significant stay in the upper freshwater dominated sections of estuaries where Sr concentration is markedly lower.
The pre-settlement otolith Me:Ca variation between the two estuaries seen in the PCO plot could indicate subtle differences in larval rearing habitat. Whereas the overlap among most of the post-settlement mark otolith Me:Ca values could mean that fish stay in the estuaries after completing their early life. The estuary-specific difference in post-settlement mark otolith Me:Ca values may suggest that fish from the Waikouaiti estuary undertake short-term excursions to the near shore areas.
A steady Sr:Ca ratio and minor Ba:Ca peaks were observed in samples from the Waikouaiti estuary. The minor Ba:Ca peaks observed in Waikouaiti samples are presumably the result of Ba concentration variability in the ambient estuarine water, most likely associated with seasonal and periodic increased freshwater inflow. Low salinity measurements were recorded in the Waikouaiti estuary during spring and winter months due to increased freshwater inputs (Taddese and Closs 2020). Temporal Ba:Ca variations correlated to freshwater flow were reported for juvenile spotted seatrout Cynoscion nebulosus otoliths in Chesapeake Bay (Dorval et al. 2005b). Furthermore, the steady Sr:Ca profile with small Ba:Ca peaks could indicate continuance of saltwater inputs with relatively constant Sr composition in this very open and well-mixed estuary.
A co-fluctuating pattern of Sr:Ca and Ba:Ca was seen in fish from the Tokomairiro estuary. This could reflect short-lived movements between different sections of the estuary. Small-scale within estuary variations in water trace element concentration can occur (Dorval et al. 2005a; Williams et al. 2018) and influence otolith composition (Gillanders and Kingsford 2000; Gillanders 2002a, b; Sanchez-Jerez et al. 2002; Dorval et al. 2007). We presume that, given the facilitation of Ba incorporation by Sr in F. nigripenne otoliths (Taddese et al. 2019), the somewhat co-variability of Sr:Ca with Ba:Ca observed in the present study suggests brief movements within the estuary from/to the section(s) where Sr is possibly higher (i.e., the lower reach). A snap-shot measurement of Sr and Ba from the Waikouaiti estuary indicated that Sr is higher in the deep middle sections (Taddese 2019; Taddese et al. 2019). It is possible that F. nigripenne moves between the estuary and the nearby coastal marine environment strongly influenced by the estuarine inputs. However, the influence of estuarine inputs, in terms of Sr and Ba, is unlikely to extend far into the marine environment given that estuaries studied are all relatively small in size.
A slight increase in Me:Ca ratios from the portion of the otoliths close to the core to the section representing the time of catch was observed in this study, suggesting that F. nigripenne probably spends its larval life in relatively deeper water columns (i.e., the main river channel) of estuaries where the Sr concentration is relatively lower (Taddese 2019; Taddese et al. 2019). An alternative explanation for this pattern could be ontogenetic changes related to dietary shifts between different life stages (Buckel et al. 2004; Engstedt et al. 2012). However, diet is known to have minimal influence on otolith chemistry relative to ambient water conditions (Milton and Chenery 2001; Walther and Thorrold 2006).
To our knowledge, the present study is the first to examine the life history of an estuarine dependent fish species in southern New Zealand estuaries using otolith chemical analysis. Our results highlight the valuable functions estuaries play in supporting the life history of an endemic fish species. The interpretations of otolith Me:Ca patterns in this study were based on a previous laboratory experiment conducted to validate the relationship between ambient water and otolith chemistry of the study species (Taddese et al. 2019). Taddese et al. (2019) confirmed the extent to which ambient water chemistry influences elemental accretion in F. nigripenne otoliths. The findings of the experiment established that Sr and Ba incorporated into F. nigripenne otoliths at a rate that reflects the ambient concentrations. Despite the complexity of estuarine water chemistry, the continuous presence of both Sr and Ba in F. nigripenne otoliths indicates the species is estuary-dependent over its entire life history. Relating a time-series estuarine water Me:Ca data to otolith values in future studies will further help in understanding the life history of F. nigripenne.
References
Beck MW, Heck KL, Able KW et al (2001) The identification, conservation, and management of estuarine and marine nurseries for fish and invertebrates. Bioscience 51:633–641
Buchheister A, Latour RJ (2010) Turnover and fractionation of carbon and nitrogen stable isotopes in tissues of a migratory coastal predator, summer flounder (Paralichthys dentatus). Can J Fish Aquat Sci 67:445–461
Buckel JA, Sharack BL, Zdanowicz VS (2004) Effect of diet on otolith composition in Pomatomus saltatrix, an estuarine piscivore. J Fish Biol 64:1469–1484
Campana SE (1999) Chemistry and composition of fish otoliths: pathways, mechanisms and applications. Mar Ecol Prog Ser 188:263–297
Childs AR, Cowley PD, Næsje TF et al (2008) Do environmental factors influence the movement of estuarine fish? A case study using acoustic telemetry. Estuar Coast Shelf Sci 78:227–236
Claramunt RM, Wahl DH (2000) The effects of abiotic and biotic factors in determining larval fish growth rates: a comparison across species and reservoirs. Trans Am Fish Soc 129:835–851
Courrat A, Lobry J, Nicolas D et al (2009) Anthropogenic disturbance on nursery function of estuarine areas for marine species. Estuar Coast Shelf Sci 81:179–190
Dawson HA, Potts DD, Maguffee AC, apos, Connor, L.M. (2015) Feasibility of passive integrated transponder technology to study in situ movements of larval sea lamprey. J Fish Wildl Manag 6:71–82 ([In English])
Delpech C, Courrat A, Pasquaud S et al (2010) Development of a fish-based index to assess the ecological quality of transitional waters: the case of French estuaries. Mar Pollut Bull 60:908–918
Dorval E, Jones CM, Hannigan R (2005a) Chemistry of surface waters: distinguishing fine-scale differences in sea grass habitats of Chesapeake Bay. Limnol Oceanogr 50:1073–1083
Dorval E, Jones CM, Hannigan R, van Montfrans J (2005b) Can otolith chemistry be used for identifying essential seagrass habitats for juvenile spotted seatrout, Cynoscion nebulosus, in Chesapeake Bay? Mar Freshw Res 56:645–653
Dorval E, Jones CM, Hannigan R, Montfrans JV (2007) Relating otolith chemistry to surface water chemistry in a coastal plain estuary. Can J Fish Aquat Sci 64:411–424
Elliott M, Whitfield AK, Potter IC et al (2007) The guild approach to categorizing estuarine fish assemblages: a global review. Fish Fish 8:241–268
Elsdon TS, Gillanders BM (2005) Strontium incorporation into calcified structures: separating the effects of ambient water concentration and exposure time. Mar Ecol Prog Ser 285:233–243
Elsdon TS, Wells BK, Campana SE et al (2008) Otolith chemistry to describe movements and life-history parameters of fishes: hypotheses, assumptions, limitations and inferences. In: RN Gibson, RJA Atkinson, JDM Gordon (eds.) Oceanography and marine biology: an annual review, Vol 46. pp. 297–330
Engstedt O, Koch-Schmidt P, Larsson P (2012) Strontium (Sr) uptake from water and food in otoliths of juvenile pike (Esox lucius L.). J Exp Mar Bio Ecol 418–419:69–74
Feary DA, Wellenreuther M, Clements KD (2009) Trophic ecology of New Zealand triplefin fishes (Family Tripterygiidae). Mar Biol 156:1703–1714
Francis MP, Morrison MA, Leathwick J, Walsh C, Middleton C (2005) Predictive models of small fish presence and abundance in northern New Zealand harbours. Estuar Coast Shelf Sci 64:419–435
Fry B, Chumchal MM (2011) Sulfur stable isotope indicators of residency in estuarine fish. Limnol Oceanogr 56:1563–1576
Gillanders BM (2002a) Connectivity between juvenile and adult fish populations: do adults remain near their recruitment estuaries? Mar Ecol Prog Ser 240:215–223
Gillanders BM (2002b) Temporal and spatial variability in elemental composition of otoliths: implications for determining stock identity and connectivity of populations. Can J Fish Aquat Sci 59:669–679
Gillanders BM, Kingsford MJ (2000) Elemental fingerprints of otoliths of fish may distinguish estuarine ‘nursery’ habitats. Mar Ecol Prog Ser 201:273–286
Green BS, Fisher R (2004) Temperature influences swimming speed, growth and larval duration in coral reef fish larvae. J Exp Mar Bio Ecol 299:115–132
Hickey AJR, Lavery SD, Hannan DA, Baker CS, Clements KD (2009) New Zealand triplefin fishes (family Tripterygiidae): contrasting population structure and mtDNA diversity within a marine species flock. Mol Ecol 18:680–696 (In English)
Hicks AS, Jarvis MG, David BO, Waters JM, Norman MD, Closs GP (2017) Lake and species specific patterns of non-diadromous recruitment in amphidromous fish: the importance of local recruitment and habitat requirements. Mar Freshw Res 68:2315–2323
Hoeksema SD, Chuwen BM, Potter IC (2009) Comparisons between the characteristics of ichthyofaunas in nearshore waters of five estuaries with varying degrees of connectivity with the ocean. Estuar Coast Shelf Sci 85:22–35
Izzo C, Reis-Santos P, Gillanders BM (2018) Otolith chemistry does not just reflect environmental conditions: a meta-analytic evaluation. Fish Fish 19:441–454
Jellyman DJ, Glova GJ, Sagar PM, Sykes JRE (1997) Spatio-temporal distribution of fish in the Kakanui River estuary, South Island, New Zealand. NZ J Mar Freshw Res 31:103–118
Jessop BM, Wang C-H, Tzeng W-N, You C-F, Shiao J-C, Lin S-H (2012) Otolith Sr: Ca and Ba: Ca may give inconsistent indications of estuarine habitat use for American eels (Anguilla rostrata). Environ Biol Fishes 93:193–207
Jochum KP, Scholz D, Stoll B et al (2012) Accurate trace element analysis of speleothems and biogenic calcium carbonates by LA-ICP-MS. Chem Geol 318–319:31–44
Kaemingk MA, Swearer SE, Bury SJ, Shima JS (2019) Landscape edges shape dispersal and population structure of a migratory fish. Oecologia 190:579–588
Kohn YY, Clements KD (2011) Pelagic larval duration and population connectivity in New Zealand triplefin fishes (Tripterygiidae). Environ Biol Fishes 91:275–286
Miles NG, Walsh CT, Butler G, Ueda H, West RJ (2013) Australian diadromous fishes—challenges and solutions for understanding migrations in the 21st century. Mar Freshw Res 65:12–24
Milton DA, Chenery SR (2001) Sources and uptake of trace metals in otoliths of juvenile barramundi (Lates calcarifer). J Exp Mar Bio Ecol 264:47–65
Paterson AW, Whitfield AK (2000) Do shallow-water habitats function as refugia for juvenile fishes? Estuar Coast Shelf Sci 51:359–364
Pearce NJG, Perkins WT, Westgate JA et al (1997) A compilation of new and published major and trace element data for NIST SRM 610 and NIST SRM 612 glass reference materials. Geostandards Newsletter 21:115–144
Peterson CH, Summerson HC, Thomson E et al (2000) Synthesis of linkages between benthic and fish communities as a key to protecting essential fish habitat. Bull Mar Sci 66:759–774
Potter IC, Bird DJ, Claridge PN, Clarke KR, Hyndes GA, Newton LC (2001) Fish fauna of the Severn Estuary. Are there long-term changes in abundance and species composition and are the recruitment patterns of the main marine species correlated? J Exp Mar Bio Ecol 258:15–37
Sanchez-Jerez P, Gillanders BM, Kingsford MJ (2002) Spatial variability of trace elements in fish otoliths: comparison with dietary items and habitat constituents in seagrass meadows. J Fish Biol 61:801–821
Schilling HT, Reis-Santos P, Hughes JM et al (2018) Evaluating estuarine nursery use and life history patterns of Pomatomus saltatrix in eastern Australia. Mar Ecol Prog Ser 598:187–199
Sutherland DL, Closs GP (2001) Spatial and temporal variation in the abundance and composition of ichthyoplankton in a large South Island, New Zealand river estuary. NZ J Mar Freshw Res 35:1061–1069 (In English)
Taddese F (2019) Fish assemblages and life history patterns in estuaries along the Otago coastline, New Zealand. PhD, University of Otago, p 152
Taddese F, Closs GP (2020) Spatiotemporal ichthyofaunal dynamics in a permanently open estuary, Otago, New Zealand. Mar Freshw Res 71:107–116
Taddese F, Schallenberg M, Mikheev P, Jarvis MG, Closs GP (2018) Ichthyofaunal assemblages in shallow littoral habitats of permanently open estuaries and intermittently closed and open lakes or lagoons in Otago, New Zealand. Mar Freshw Res 69:1222–1230
Taddese F, Reid MR, Closs GP (2019) Direct relationship between water and otolith chemistry in juvenile estuarine triplefin Forsterygion nigripenne. Fish Res 211:32–39
USGS (2012) United States Geological Survey certificate of analysis microanalytical standard, MACS-3. US.
Walther BD, Thorrold SR (2006) Water, not food, contributes the majority of strontium and barium deposited in the otoliths of a marine fish. Mar Ecol Prog Ser 311:125–130
Wellenreuther M, Clements KD (2007) Reproductive isolation in temperate reef fishes. Mar Biol 152:619–630
Wellenreuther M, Paul TB, Kendall DC (2007) Ecological diversification in habitat use by subtidal triplefin fishes (Tripterygiidae). Mar Ecol Prog Ser 330:235–246
Whitfield AK (1999) Ichthyofaunal assemblages in estuaries: a South African case study. Rev Fish Biol Fish 9:151–186
Whitfield A (2002) Fishes as indicators of environmental and ecological changes within estuaries: a review of progress and some suggestions for the future. J Fish Biol 61:229–250
Williams J, Jenkins GP, Hindell JS, Swearer SE (2018) Fine-scale variability in elemental composition of estuarine water and otoliths: developing environmental markers for determining larval fish dispersal histories within estuaries. Limnol Oceanogr 63:262–277
Acknowledgements
The authors thank Nicky McHugh and Matthew Downes for providing support in microscopy. Financial assistance for this study was provided by a University of Otago Doctoral Scholarship awarded to F. Taddese. This study was conducted under University of Otago Animal Ethics Committee (AEC) permit number 23/16.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Taddese, F., Reid, M., Heim-Ballew, H. et al. Otolith chemistry of triplefin Forsterygion nigripenne indicates estuarine residency. Fish Sci 87, 271–281 (2021). https://doi.org/10.1007/s12562-021-01501-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12562-021-01501-0