Abstract
Overexposure to food colorants above the allowed daily intake (ADI) level can provoke hyperactivity and other disturbed behaviors especially in children. Two new methods were developed to separate five synthetic colorants, which were Tartrazine (E102), Sunset Yellow (E110), Allura Red (E129), Carmoisine (E122), and Brilliant Blue (E133). They are labeled on a large variety of commercial food products in the Egyptian market without mentioning their definite concentrations. Therefore, there was a real need to determine these colorants with simple, accurate, and fast methods. This is the first study to determine these colorants in a wide variety of food products present in the Egyptian market. The HPLC approach with photodiode array detection was developed to quantify these colorants, on a C18 column, with a mobile phase composed of acetonitrile and water containing 1% ammonium acetate (pH 6.8), separation was carried out using a gradient program. The colorants were eluted and efficiently separated within 9 min. Then, as a complementary technique to HPLC, the UPLC-ESI–MS/MS approach was developed for identification and accurate mass measurement of the colorants found in high concentrations, the colorants were obtained simultaneously in negative mode, the run time was only 3 min. These developed methods were validated according to ICH recommendations and they were applied to analyze 65 food products including jelly powder, puddings, ice cream powders, concentrated soft drink powders, carbonated drinks, chewing gums, and sugar confectionery.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Recently, food safety has become a vital issue to the general public, medical, nutritional experts, and food science researchers. Food additives are widely used to confer a desirable appearance and enhance the nutritional properties of commercial products (Ntrallou, 2020; Bordagaray, 2018; Bordagaray, 2019). Colorants are chemicals which are applied during development or processing of foods and soft drinks. However, some of these chemicals represent potential risks to human health and result in adverse effects like metabolic acidosis, tremors, and hyperpnoea (Sorouraddin, 2015), especially if they are used in exceeded amounts (Sierra-Rosales, 2017). Therefore, regulated use of these colorants in different food items must be ensured (Islam, 2019).
Children are the most vulnerable as they consume a lot of candies and drinks, especially those rich in synthetic colorants. Owing to their lower weight relative to adults, acceptable daily intake values (ADIs) are easily reached so that the adverse effects are more obvious in this population. Centered on agencies’ international guidelines, such as the U.S. Department of Agriculture (USDA) and the U.S. Food and Drug Administration (FDA), all food colorants must be listed (Mazdeh, 2016). The analysis of food needs quick methods for routine control and high sample throughput. Chromatographic separation allows multiple compounds in complex matrices to be quantified simultaneously, many methods have been involved to improve separation efficiency and decrease the time of analysis (Lhotská, 2018).
The investigated synthetic colorants were (Tartrazine, Sunset Yellow, Allura Red, Carmoisine, and Brilliant Blue). Chemical structures, numbers of the European community (E numbers), and denominations of the color index (CI numbers) were represented in (Fig. S1). Upon observation, they were among the dyes most labeled in Egyptian food products such as ice cream, puddings, sweets, drinks, and chewing gums; however, their concentrations were not mentioned. Mentioning their concentrations is valuable to be sure that ADIs are not exceeded particularly by young children. This work aimed to monitor various Egyptian food products which are consumed daily especially by children, using fast simple methods. Chromatographic analysis was successfully performed on 65 food products.
Various analytical approaches have been reported to determine a wide variety of synthetic food colorants, like thin-layer chromatography (TLC) (Oka, 1994; de Andrade, 2014), adsorptive voltammetry (Ni, 1997), differential pulse polarography (Combeau, 2002), and electrophoresis (Cifuentes, 2006; Dossi, 2007; Prado, 2006). However, most of them require time-consuming pretreatment and cannot be applied to complicated color mixtures (Mazdeh, 2016). Different chromatographic methods have been reported including HPLC with UV detection (Demiralay, 2006; Garcı́a, 2003; Lino, 2010; Saad, 2005; Techakriengkrai, 2007; Tfouni, 2002; Brazeau, 2018), ion chromatography with UV detection (Dossi, 2006), molecular absorption spectrophotometry (Cantarelli, 2009), GC using flame ionization detection (Dong, 2006, Wang,2006), flow injection analysis (FI) with the aid of UV detection ( García-Jiménez, 2007; García‐Jiménez, 2006). High-performance ion chromatography (Lucena, 2005), reversed-phase liquid chromatography (Garcıa-Falcón, 2005; Kirschbaum, 2003; Prado, 2002; Mathiyalagan, 2019), and ion-pair liquid chromatography (Fuh, 2002; Gianotti, 2005; González, 2003; Gallego, 2003; Ishikawa, 2003; Kiseleva, 2003). There were some methods to determine the studied colorants in presence of others in different matrices using HPLC with UV/Vis or photodiode array detection (Al-Degs, 2009; Enríquez-Gabeiras, 2012; Kirschbaum, 2006; Long, 2009; Olgun,2012; Serdar, 2009; Iammarino, 2019). HPLC coupled with UV/Vis or DAD detection is the most common approach as colorants are highly absorbed at the UV and/or visible wavelength. UPLC-MS/MS methods for analysis of colorants in foods have also been reported (Chen, 2014; Gao, 2015; Guerra, 2018; Tsai, 2013; Zou, 2013). Ultraviolet (UV) detectors were replaced by MS detectors as they provide more structural details and can adhere to the confirmatory process requirements set out in European Commission Decision 2002/657/EC. Recently, E102,E110, E122, E129, and E 133 were separated in the presence of some other colorants in food products consumed by children in Saudi Arabia using only an HPLC–DAD method and the method developed was not completely validated (Ahmed, 2021), while E102, E110, E129, and E 133 were separated in the presence of some other colorants in only chewing gums and soft drinks present in Korea using HPLC–DAD method and LC–MS/MS method, where the limit of detection of the HPLC–DAD method was almost similar to our proposed HPLC–DAD method while the limit of detection of our LC–MS/MS method was lower than the reported LC–MS/MS method (Jang,2021). Also, E102,E110, E129 were separated using HPLC–UV method in soft drinks, powder juice and candies (Al-Khateeb, 2021). Nguyen has separated E102,E110, E122, E129, and E 133 in the presence of other colorants in food products in Korea (Nguyen, 2021), using only HPLC–DAD method with longer run time and higher limit of detection than our method.
In our study, a reversed-phase high-performance liquid chromatography with diode array detector (RP-HPLC–DAD) method and a UPLC-ESI–MS/MS method were developed for the determination of five water-soluble synthetic food colorants (E102, E110, E129, E122, and E133) in a set of 65 food products present in the Egyptian market. These colorants were chosen owing to their abundance among food products in the local market in Egypt. The proposed methods included a simple pretreatment procedure for the samples and allowed the detection of colorants at very low concentrations. It is worth mentioning that this study is considered to be the first one to determine the concentrations of the synthetic food colorants (E102, E110, E129, E122, and E133) in a wide variety of food products such as sweets, drinks, ice cream powders, puddings, and chewing gums in the Egyptian market, which are consumed mainly by children, and to compare the results with international limits.
Experimental
Materials and Reagents
High purity standard of Tartrazine (E102) (99.1% purity), Sunset Yellow (E110) (98.1% purity), Allura Red (E129) (98.1% Purity), Carmoisine (E122) (98.0% purity), and Brilliant Blue (133) (98.5% purity) were obtained from Sigma-Aldrich. HPLC-grade methanol was obtained from Fisher Scientific. Ammonium acetate (98%) and acetonitrile (HPLC grade) were purchased from Sigma-Aldrich. The ultra-pure water was prepared by distillation in glass and passage through a Milli-Q water system, passed through a 0.45-μm membrane filter, and degassed for 30-min in an ultrasonic bath. Food samples were purchased from Egyptian supermarkets.
RP-HPLC–DAD Analysis
The HPLC separation and quantitation were achieved on Waters LC 2695, (Milford, MA, USA) with a gradient pump using low-pressure mixing system, vacuum membrane degasser, an autosampler with a 100 μl sample loop and a capacity of 120 vials, and a Waters module 2996 photodiode array detector. The column C18 (100 mm × 4.6 mm (i.d.)) packed with Inertsil ODS-3 V (5-μm particle diameter, GL Sciences, Tokyo, Japan) and an HPLC column oven (DALIAN REPLETE®, Hong Kong) was used. Waters Empower software was used for Data acquisition. The mobile phase consisted of eluent A, water containing 1% ammonium acetate (with pH 6.8, adjusted with ammonium hydroxide); and eluent B, acetonitrile. RP-HPLC–DAD assay was carried out by constructing a gradient elution program of 5% B at 0–3 min, 10% B at 3–9 min, 40% B at 9–9.5 min, and 70% B at 9.5–12 min. In the end, a 3 min equilibrium phase to the column was made to recover first condition of 95% A: 5% B for the next run. The acquisition wavelength of DAD was set to scan between 200 and 800 nm. At column temperature 25 °C, all determinations were performed. The injected volume was 20 μL and the run time was 12 min. The flow rate set at 1.0 mL min−1. The mobile phase was filtered through a 0.45-μm membrane filter and degassed under vacuum prior to use.
UPLC-MS/MS Analysis
Separation was made on a Waters 3100 instrument (Milford, MA, USA) with a binary solvent manager pump, autosampler, and thermostatic column compartment. An API 5000 triple quadrupole (TQ detector, Acquity UPLC, USA) was the tandem mass spectrometer. A UPLC acquity® BEH shield RP18 column (150 mm × 2.1 mm, 1.7 μm) was used. Mass lynx software (Version 4.1) was used for data analysis and system operation. UPLC/MS/MS assay was carried out using an isocratic system with a flow rate of 0.3 mL min−1. Acetonitrile: 4 mM ammonium acetate (80:20, v/v) was the mobile phase. By electrospray ionization mass spectrometry, mass spectra were obtained simultaneously in negative mode (ESI/MS). The injected volume was 2 μl through a 3-min run time.
The tuning parameters were set for detection of the investigated colorants optimally: desolvation temperature, 400 °C; ion source temperature, 120 °C; capillary voltage, 3.5 kV; desolvation gas flow, 600L/h. The proper collision energy, cone voltage and representative product ions values for these five colorants were listed in Table 1.
Preparation of Standards of Colorants and Calibration
Dissolving 10.0 mg of pure coloring agents into 10.0 mL of methanol was used to prepare individual standard stock solutions containing each colorant. The solutions were freshly prepared, tightly closed to amber colored volumetric flasks, and kept at 4 °C. For calibration of HPLC method, these stock solutions were diluted with methanol to obtain working standard solutions of suitable concentrations within the concentration range (0.5–50 μg mL−1). For calibration of UPLC-MS/MS method, accurate volumes of each of E102, E110, E129, E122, and E133 stock solutions were transferred into volumetric flasks (10 mL) and diluted to volume with methanol to make synthetic mixtures in the concentration range (0.01–5 µg mL−1) of each compound.
Preparation of Commercial Samples
All of the sixty five tested samples were brought from the Egyptian markets and categorized to four categories: jelly powders, puddings, ice cream powders (A), concentrated fruit-flavored soft drinks powders (B), carbonated fruit-flavored drinks (C), chewing gums, candy smartiz, and sugar confectionery(D). Category A samples were numbered from A 1 to A28, category B samples were numbered from B 29 to B43, category C samples were numbered from B 44 to B51, and category D samples were numbered from B 52 to B65.
Samples Analyzed by RP-HPLC–DAD Technique
Treatment of Carbonated Fruit-Flavored Drinks
Twenty-five milliliters of the sample were adequately transferred to a 50-mL volumetric flask and 20 mL of HPLC water was added. The samples were subjected to sonication for 15 min and completed with HPLC water to the mark. After filtering through 0.45-μm disposable syringe filters, 20 μl of the solutions were injected.
Treatment of Concentrated Fruit-Flavored Soft Drink Powders
Two grams of each sample was weighed and diluted with 30 mL HPLC water in a 50-mL volumetric flask. The samples were subjected to sonication for 15 min and completed to the mark with HPLC water. After filtering through 0.45-μm disposable syringe filters, all solutions were injected (20 μl).
Treatment of Jelly Powder, Ice Cream Powder, Puddings, and Mehalabia
Two grams of each sample was weighed and diluted with HPLC water in a 25-mL volumetric flask. The sample solutions were subjected to sonication for 15 min and completed with HPLC water to the mark. After filtering through 0.45-μm disposable syringe filters, all solutions were injected (20 μl).
Treatment of Chewing Gums, Candy Smartiz, and Sugar Confectionary
Five grams of each sample was weighed then sliced and transferred to a beaker containing 5 ml of 1% ammonia solution (Khanavi, 2012). Twenty-five milliliters of HPLC water was added, the sample solutions were subjected to sonication for 30 min, filtered through a folded filter paper, and the filtrate was collected in a 50-ml volumetric flask and completed with water. After filtering through 0.45-μm disposable syringe filters, the solutions were injected (20 μl).
Samples Analyzed by UPLC-MS/MS Technique
The selected samples (A5, A11, A13, A19, B31, B40, B41, B42, B43, and D 53) were treated as follows: accurately, 0.5 gm of each sample was weighed and dissolved in 10 mL of water (50 mg/mL), except savory red sampleD53) was diluted to 5 mg/mL, as it is highly concentrated and vortexed for 1 min. All solutions were subjected to sonication for 30 min and further diluted by HPLC water. All sample solutions were filtered by a 0.22-μm syringe filter then injected.
Results and Discussion
Optimization of the Separation
Optimization of the HPLC–DAD Method
Many mobile phase compositions have been tried, methanol and phosphate buffer (50:50, v/v); methanol and phosphate buffer (70:30, v/v), methanol and ammonium acetate buffer (80:20, v/v); methanol and ammonium acetate solution 1% (w/v) (70:30,v/v); and acetonitrile and ammonium acetate buffer (20:80, v/v). The separation was poor for these trials. E122 and E133 colorants were not well separated and the peaks E102 and E110 were forked. Isocratic elution was not appropriate and gradient elution was then tried to achieve better separation. The use of gradient elution to separate colorants was also present in previous methods (Kirschbaum, 2003; Serdar, 2009; Iammarino, 2019; Filiz, 2019; Ahmed, 2021, Jang, 2021). Different amounts of ammonium acetate in the mobile phase were tested. Experiments and a reported method (Minioti, 2007) revealed that the most effective amount for ammonium acetate is 1% (w/v). Different pH values for the ammonium acetate buffer were also examined (6, 6.8, 7.5, and 7.9), and increasing pH to more than 7 leads to poor resolution. Several time programs with different initial percentage of acetonitrile were tested, increased acetonitrile concentration to more than 10%, resulted in insufficient separation and characteristic overlap of E122 and E133 colorants, while lower acetonitrile concentration (about 5%) resulted in excessive tailing and longer retention time of E133. Different column temperatures were examined (25 ºC, 30 ºC, and 35 ºC) and no significant effect was observed upon increasing temperature more than 25 ºC. The best results were achieved by using mobile phase consisting of water containing 1% ammonium acetate at pH 6.8 and acetonitrile using gradient elution program and column temperature kept at 25 °C. Optimum resolution, consistent baseline separation of the studied compounds, was achieved by the previous condition (Fig. 1). The chromatographic conditions used allowed us to achieve the separation of colorants within 9 min.
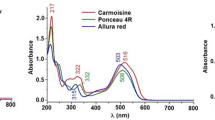
A chromatogram was established with scanning in the wavelength range from 200 to 800 nm, continuously. The spectral data were very useful to confirm peak purity. The absorption maxima of the studied colorants were 426 nm, 482 nm, 508 nm, 517 nm, and 629 nm for E102, E110, E129, E122, and E133, respectively. Quantitation was achieved at 388 nm, where reasonable linearity ranges, LOD, and LOQ were achieved for all of the colorants studied. The average retention time ± standard deviation for E102, E110, E129, E122, and E133 were found to be 3.561 ± 0.02, 6.533 ± 0.04, 7.109 ± 0.05, 8.394 ± 0.07, and 8.864 ± 0.06 min, respectively, for seven replicates. The first to be eluted from the chromatographic column were the azo compounds, followed by non-azo compounds. The last synthetic colorant eluted was E133 which is derived from triphenyl methane and has an apolar character. For that, this compound interacted longer with the column and was the last to be eluted. The selectivity of the HPLC method was demonstrated in Table 2.
Optimization of the UPLC-MS/MS Method
In the LC–ESI–MS/MS system, selection of the mobile phase composition is of high importance because it must offer an acceptable compromise between good chromatographic elution, separation, analytes ionization, and efficient desolvation of charged species in the MS detector. Satisfactory identification was performed with a mobile phase consisting of acetonitrile: 4 mM ammonium acetate (80:20 v/v) using isocratic elution (Fig. 2). The optimum flow rate was 0.3 mL min−1. These followed conditions, allowed us to quantify the studied colorants in 3 min.
Under these operating conditions, E102, E110, E129, E122, and E133 were negatively charged due to their chemicophysical properties. They undergo deprotonation due to the presence of acidic sites. For this reason, MS detection was performed in the negative ESI mode.
Structural Analyses of E102, E110, E129, E122, and E133 by UPLC-ESI–MS/MS
Upon observing the structural analyses of the studied colorants, E102 exhibited an (M-H)− ion at m/z 233.02 and eluted at 0.75 min, its MS/MS spectrum produced fingerprint fragment ions at m/z 210.91 and 197.58 (Fig. S2), E110 exhibited an (M-H)− ion at m/z 203.05 eluted at 0.79 min, its MS/MS spectrum produced fingerprint fragment ions at m/z 170.74, 155.78, and 106.68 (Fig. S3), E129 exhibited an (M-H)− ion at m/z 225.17 and eluted at 0.79 min, its MS/MS spectrum produced fingerprint fragment ions at m/z 235.92, 206.76, 200.03, 181.05, 171.75, and 135.98 (Fig. S4); E122 exhibited an (M-H)− ion at m/z 228.06 and eluted at 0.79 min, its MS/MS spectrum produced fingerprint fragment ions at m/z 220.96, 206.08, 169.98, and 79.69 (Fig. S5); and E133 exhibited an (M-H)− ion at m/z 373.32 and eluted at 0.78 min, its MS/MS spectrum produced fingerprint fragment ions at m/z 481.40, 333.33, and 169.99 (Fig. S6).
Validation of the Methods
Linearity
The linearity of the methods proposed was tested by evaluating the various concentrations of each compound. Nine concentrations were chosen in the 0.5–50 μg mL−1 range for the HPLC–DAD process, while concentrations in the range 0.01–5 μg mL−1 were used for the tested colorants in the UPLC-MS/MS range. Three injections were made for each concentration. The high value of the correlation coefficient was used to assess the calibration graphs’ linearities. The regression parameters for the methods obtained by the least square treatment of the results were shown in Table 3.
Precision and Accuracy
Analysis of three concentration levels of working solutions of each compound on the same day determined the intra-day accuracy and also precision (each concentration was repeated three times). Inter-day accuracy and precision were measured on three consecutive days by evaluating the three concentration levels of working solutions. The acceptability of the data included accuracy stated as relative error (RE %) and precision stated as relative standard deviation (RSD %). Tables S1 and S2 summarized the results of intra-day and inter-day precision and accuracy.
The maximum measured relative standard deviation of the measurements was approximately 2.1% for UPLC-MS/MS and 2.6% for HPLC–DAD, suggesting the excellent precision of the analytical methods proposed at both repeatability and intermediate levels.
Limits of Detection and Quantification
According to ICH recommendations (ICH, 2005), the limit of detection (LOD) and the limit of quantification (LOQ) is determined as the ratio of 3.3 and 10 standard deviations of the blank (n = 9), respectively, and the slope of the calibration line. Table 3 presented the detection and quantitation limits calculated for E102, E110, E129, E122, and E133.
Selectivity
The selectivity of the methods was established by preparing five mixtures of the studied colorants within the linearity range at different concentrations. The mixtures were analyzed according to previously discussed procedures. Appropriate recoveries (Table S3) were generated, revealing the good selectivity of the methods proposed to concurrently analyze E102, E110, E129, E122, and E133.
Analytical Solution Stability
The analysis demonstrated that the standard colorants solutions were stable for about 3 h at room temperature, for 1 week if kept at 4 °C and for 1 month when kept in the freezer at − 20 °C.
Application to Real Samples
Analysis of Food Samples Using HPLC–DAD Method
The developed HPLC approach was efficiently applied to determine E102, E110, E129, E122, and E133 in sixty five food products which are consumed daily in Egypt especially by children, including carbonated fruit-flavored drinks, concentrated fruit-flavored soft drinks, chewing gums, candy smartiz, puddings, jellies, ice cream powders, and sugar confectionery. Upon reviewing literature for determination of these colorants in relatively similar matrices, the previous HPLC method (Al-Degs, 2009) for determination of only E129, E110, and E102 in commercial soft drinks showed long elution time of 10 min, while the reported method (Olgun, 2012), E102 and E110 and some other colorants were separated in beverages at very long retention time of 14 min and 19 min with the same limit of detection of our developed method and the previous method (Brazeau, 2018) needed a complicated sample pretreatment. This study is considered the first one to be conducted to determine the concentrations of the synthetic food colorants (E102, E110, E129, E122, and E133) in a huge amount of food products in Egypt and to compare the results with international limits with simple sample treatment, good sensitivity and short elution time.
The concentrations obtained by the developed HPLC method for the investigated colorants were shown in Table 4. The concentrations of colorants varied from 0.0252 mg kg−1 (sample D61) to 213.696 mg kg−1 (sample D53). A strange fact is that some manufacturers report that their product contains a certain colorant while a different colorant was detected and quantified by our method (sample A3). Furthermore, some products contained excess colorants which were not labeled (samples A2, A17, A20, A21, A25, A28, B30, B42, B43, C48, C50, D53, D57, D58, and D61). Some colorants were labeled but not identified as in the samples A6, A14, A17, A24, B30, B34, C45, C51, D52, D64, and D65.
Analysis of Some Selected Samples Using UPLC-MS/MS Method
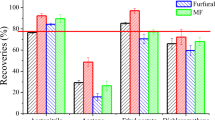
Products which contain very high concentrations of colorants above ADI levels were investigated and characterized by UPLC-ESI–MS/MS technique. Ten food samples (A5, A11, A13, A19, B31, B40, B41, B42, B43, and D53) were selected. Table S4 summarized the results of the ten samples and Fig. 3 showed five food products containing high concentrations of synthetic colorants. According to FDA, ADI of the studied colorants were used to calculate the maximum allowed amount for children weighed 15–25 kg and adults weighed 60–70 kg per day. The data were given in Table S5.
The sample D53 contained the highest concentration among the others, definitely 199.6 mg/Kg of E122 colorant which is not allowed by FDA due to its harmful effects on human health as it has an adverse effect on activity and attention in children. Also, samples B40, B41, B42, and A19 acquired concentrations of colorants above the legal limit. Also in sample B42, the manufacturer reported that the product containing only E102 and E110, and the analysis confirmed the presence of one more synthetic colorant, E129 (Fig. 3). This revealed that our study is suitable for confirmation of the identity and quantity of colorants in foods.
These results have shown that there is a strong need to track the concentrations of synthetic colorants used in food products in order to protect public health from the significant adverse effects of such chemicals and to raise awareness among both consumers and policy makers.
Conclusion
At present, there are many food colorants added to food products in order to attract consumers especially children. Some individuals are sensitive to such food colorants; the names of the colors present in the food product must be the same as those on the food product label and the quantity of colorants must be specified in order to ensure food safety. For that, it is necessary to provide for a simplified procedure that allow the determination of these colorants. The current study provided a RP-HPLC–DAD approach for the identification and determination of food colorants definitely Tartrazine E102, Allura Red E129,Sunset Yellow E110, Brilliant Blue E133, and Carmoisine E122 in a set of 65 Egyptian food products from different producers, in which they were well separated in only 9 min with simple sample preparation procedures. In addition a UPLC-ESI–MS/MS approach was developed and utilized as highly precise, sensitive confirmatory method, which could be applied successfully to suspicious marketed products.
Data Availability
All data generated or analyzed during this study are included in this published article and its supplementary information file.
References
Ahmed MA et al (2021) Dietary intake of artificial food color additives containing food products by school-going children. Saudi Journal of Biological Sciences 28:27–34
Al-Degs YS (2009) Determination of three dyes in commercial soft drinks using HLA/GO and liquid chromatography. Food Chem 117:485–490
Al-Khateeb LA (2021) An Eco-Friendly RP-HPLC Method for the separation and trace determination of selected food colorant residues in foodstuffs utilizing superheated Water. J Anal Chem 76:824–833
Bordagaray A et al (2018) Determination of food colorants in a wide variety of food matrices by microemulsion electrokinetic capillary chromatography. Considerations on the found concentrations and regulated consumption limits. Food Chem 262:129–133
Bordagaray A et al (2019) Simultaneous determination of food colorants in liquid samples by UV-Visible spectroscopy and multivariate data analysis using a reduced calibration matrix. J Chemom 33:e3176
Brazeau J (2018) Identification and quantitation of water-soluble synthetic colors in foods by liquid chromatography/ultraviolet–visible method development and validation. ACS Omega 3:6577–6586
Cantarelli MA et al (2009) Simultaneous determination of aspartame and acesulfame-K by molecular absorption spectrophotometry using multivariate calibration and validation by high performance liquid chromatography. Food chemistry.115: 1128–1132
Chen X-H et al (2014) Fast determination of seven synthetic pigments from wine and soft drinks using magnetic dispersive solid-phase extraction followed by liquid chromatography–tandem mass spectrometry. J Chromatogr A 1346:123–128
Cifuentes A (2006) Recent advances in the application of capillary electromigration methods for food analysis. Electrophoresis 27:283–303
Combeau S, Chatelut M, Vittori O (2002) Identification and simultaneous determination of Azorubin, Allura red and Ponceau 4R by differential pulse polarography: application to soft drinks. Talanta 56:115–122
de Andrade FI et al (2014) Determination of synthetic food dyes in commercial soft drinks by TLC and ion-pair HPLC. Food Chem 157:193–198
Demiralay EC, Özkan G, Guzel-Seydim Z (2006) Isocratic separation of some food additives by reversed phase liquid chromatography. Chromatographia 63:91–96
Dong C, Wang W (2006) Headspace solid-phase microextraction applied to the simultaneous determination of sorbic and benzoic acids in beverages. Anal Chim Acta 562:23–29
Dong C, Mei Y, Chen L (2006) Simultaneous determination of sorbic and benzoic acids in food dressing by headspace solid-phase microextraction and gas chromatography. J Chromatogr A 1117:109–114
Dossi N et al (2006) Simultaneous RP-LC determination of additives in soft drinks. Chromatographia 63:557–562
Dossi N et al (2007) (2007) Rapid analysis of azo-dyes in food by microchip electrophoresis with electrochemical detection. Electrophoresis 28:4240–4246
Enríquez-Gabeiras L et al (2012) Interference-free determination of illegal dyes in sauces and condiments by matrix solid phase dispersion (MSPD) and liquid chromatography (HPLC–DAD). Food Chem 135:193–198
Feng F et al (2011) Highly sensitive and accurate screening of 40 dyes in soft drinks by liquid chromatography–electrospray tandem mass spectrometry. J Chromatogr B 879:1813–1818
Filiz Z et al (2019) Determination of synthetic colorants in cosmetic products by reversed-phase high-performance liquid chromatography coupled with diode-array detector. J Res Pharma 23:1048–1059
Fuh M-R, Chia K-J (2002) Determination of sulphonated azo dyes in food by ion-pair liquid chromatography with photodiode array and electrospray mass spectrometry detection. Talanta 56:663–671
Gao H-G, Gong W-J, Zhao Y-G (2015) Rapid method for quantification of seven synthetic pigments in colored Chinese steamed buns using UFLC-MS/MS without SPE. Anal Sci 31:205–210
García-Jiménez J, Valencia M, Capitán-Vallvey L (2006) Improved Multianalyte determination of the intense sweeteners aspartame and acesulfame-K with a Solid sensing zone implemented in an FIA Scheme. Anal Lett 39:1333–1347
García-Jiménez J, Valencia M, Capitán-Vallvey L (2007) Simultaneous determination of antioxidants, preservatives and sweetener additives in food and cosmetics by flow injection analysis coupled to a monolithic column. Anal Chim Acta 594:226–233
Garcı́a I et al (2003) Advances in methodology for the validation of methods according to the International Organization for Standardization: Application to the determination of benzoic and sorbic acids in soft drinks by high-performance liquid chromatography. J Chromatogr A 992:11–27
Garcıa-Falcón MJ Simal-Gándara, (2005) Determination of food dyes in soft drinks containing natural pigments by liquid chromatography with minimal clean-up. Food Control 16:293–297
Gianotti V et al (2005) Chemometrically assisted development of IP-RP-HPLC and spectrophotometric methods for the identification and determination of synthetic dyes in commercial soft drinks. J Liq Chromatogr Relat Technol 28:923–937
González M, Gallego M, Valcárcel M (2003a) Determination of natural and synthetic colorants in prescreened dairy samples using liquid chromatography-diode array detection. Anal Chem 75:685–693
González M, Gallego M, Valcárcel M (2003b) Liquid chromatographic determination of natural and synthetic colorants in lyophilized foods using an automatic solid-phase extraction system. J Agric Food Chem 51:2121–2129
Guerra E, Llompart M, Garcia-Jares C (2018) Analysis of dyes in cosmetics: challenges and recent developments. Cosmetics 5:47
Holčapek M, Jandera P, Zderadička P (2001) High performance liquid chromatography–mass spectrometric analysis of sulphonated dyes and intermediates. J Chromatogr A 926:175–186
Holčapek M, Volná K, Vaněrková D (2007) Effects of functional groups on the fragmentation of dyes in electrospray and atmospheric pressure chemical ionization mass spectra. Dyes Pigm 75:156–165
Iammarino M et al (2019) Simultaneous determination of twelve dyes in meat products: Development and validation of an analytical method based on HPLC-UV-diode array detection. Food Chem 285:1–9
International Conference on Harmonisation of Technical Requirements for Registration of Pharmaceuticals for Human Use (2005) ICH Harmonised Tripartite Guideline– Validation of Analytical Procedures: Text and Methodology Q2 (R1), Current Step 4 Version, London
Ishikawa F et al (2003) Simultaneous determination of eleven dyes and their aluminum lakes in drugs. J AOAC Int 86:215–221
Islam A et al (2019) Determination of sunset yellow in different brands of orange jellies of Bangladesh by HPLC. Italian Journal of Food Science, 31(1)
Jang GW et al (2021) Method validation of 12 kinds of food dye in chewing gums and soft drinks, and evaluation of measurement uncertainty for soft drinks. Food Chem 356:129705
Khanavi M et al (2012) Development of a green chromatographic method for simultaneous determination of food colorants. Food Anal Methods 5:408–415
Kirschbaum J et al (2003) Development and evaluation of an HPLC-DAD method for determination of synthetic food colorants. Chromatographia 57:S115–S119
Kirschbaum J, Krause C, Brückner H (2006) Liquid chromatographic quantification of synthetic colorants in fish roe and caviar. Eur Food Res Technol 222:572–579
Kiseleva M, Pimenova V, Eller K (2003) Optimization of conditions for the HPLC determination of synthetic dyes in food. J Anal Chem 58:685–690
Lhotská I, Solich P, Šatínský D (2018) A Comparative study of advanced stationary phases for fast liquid chromatography separation of synthetic food colorants. Molecules 23:3335
Lino C, Pena A (2010) Occurrence of caffeine, saccharin, benzoic acid and sorbic acid in soft drinks and nectars in Portugal and subsequent exposure assessment. Food Chem 121:503–508
Long C et al (2009) Synthesis and characterization of a novel molecularly imprinted polymer for simultaneous extraction and determination of water-soluble and fat-soluble synthetic colorants in chilli products by solid phase extraction and high performance liquid chromatography. J Chromatogr A 1216:8379–8385
Lucena R et al (2005) Continuous flow autoanalyzer for the sequential determination of total sugars, colorant and caffeine contents in soft drinks. Anal Chim Acta 530:283–289
Ma M et al (2006) Simultaneous determination of water-soluble and fat-soluble synthetic colorants in foodstuff by high-performance liquid chromatography–diode array detection–electrospray mass spectrometry. J Chromatography A 1103:170–176
Mathiyalagan S, Mandal BK, Ling Y-C (2019) Determination of synthetic and natural colorants in selected green colored foodstuffs through reverse phase-high performance liquid chromatography. Food Chem 278:381–387
Mazdeh FZ et al (2016) Determination of 8 synthetic food dyes by solid phase extraction and reversed-phase high performance liquid chromatography. Trop J Pharm Res 15:173–181
Minioti KS et al (2007) Determination of 13 synthetic food colorants in water-soluble foods by reversed-phase high-performance liquid chromatography coupled with diode-array detector. Analytica Chimica Acta 583:103–110
Nguyen NVT et al (2021) Simultaneous determination of 11 water-soluble dyes in food products and beverages by high performance liquid chromatography. Int Food Res J 28:120–128
Ni Y, Bai J, Jin L (1997) Multicomponent chemometric determination of colorant mixtures by voltammetry. Anal Lett 30:1761–1777
Ntrallou K, Gika H, Tsochatzis E (2020) Analytical and sample preparation techniques for the determination of food colorants in food matrices. Foods 9:58
Oka H et al (1994) Identification of unlawful food dyes by thin-layer chromatography- fast atom bombardment mass spectrometry. J Chromatogr A 674:301–307
Olgun FAO, Ozturk BD, Apak R (2012) Determination of synthetic food colorants in water-soluble beverages individually by HPLC and totally by Ce (IV)-oxidative spectrophotometry. Food Anal Methods 5:1335–1341
Prado MA et al (2006) Validation of methodology for simultaneous determination of synthetic dyes in alcoholic beverages by capillary electrophoresis. J Chromatography A 1136:231–236
Prado M, Godoy H (2002) Validation of the methodology to determine synthetic dyes in foods and beverages by HPLC. J Liq Chromatogr Relat Technol 25:2455–2472
Saad B et al (2005) Simultaneous determination of preservatives (benzoic acid, sorbic acid, methylparaben and propylparaben) in foodstuffs using high-performance liquid chromatography. J Chromatogr A 1073:393–397
Serdar M, Knežević Z (2009) Simultaneous LC analysis of food dyes in soft drinks. Chromatographia 70:1519
Sierra-Rosales P, Toledo-Neira C, Squella J (2017) Electrochemical determination of food colorants in soft drinks using MWCNT-modified GCEs. Sens Actuators, B Chem 240:1257–1264
Sorouraddin M-H, Saadati M, Mirabi F (2015) Simultaneous determination of some common food dyes in commercial products by digital image analysis. Journal of food and drug analysis 23:447–452
Techakriengkrai I, Surakarnkul R (2007) Analysis of benzoic acid and sorbic acid in Thai rice wines and distillates by solid-phase sorbent extraction and high-performance liquid chromatography. J Food Compos Anal 20:220–225
Tfouni S, Toledo M (2002) Determination of benzoic and sorbic acids in Brazilian food. Food Control 13:117–123
Tsai C-F, Kuo C-H, Shih DY-C (2015) Determination of 20 synthetic dyes in chili powders and syrup-preserved fruits by liquid chromatography/tandem mass spectrometry. J Food Drug Anal 23:453–462
Zou T et al (2013) Determination of seven synthetic dyes in animal feeds and meat by high performance liquid chromatography with diode array and tandem mass detectors. Food Chem 138:1742–1748
Funding
Open access funding provided by The Science, Technology & Innovation Funding Authority (STDF) in cooperation with The Egyptian Knowledge Bank (EKB).
Author information
Authors and Affiliations
Contributions
Ghada H. Abd-ElHamid, Ghada M. Hadad, Randa A. Abdel Salam, and Amany K. Ibrahim, played a major role in preparing methodology, bringing resources, writing the original draft preparation and reviewing. Eman A. Abdel Hameed, Mohamed A. Abdelshakour, and Omayma M. El-Darder played a major role in data interpretation, preparing figures and tables, writing, and reviewing the whole final manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethical Approval
This article does not contain any studies with human participants or animals performed by any of the authors.
Informed Consent
Informed consent was obtained from all individual participants included in this study.
Conflict of interest
Eman A. Abdel Hameed declares that she has no conflict of interest. Ghada H. Abd-ElHamid declares that she has no conflict of interest. Omayma M. El-Darder declares that she has no conflict of interest. Amany K. Ibrahim declares that she has no conflict of interest. Randa A. Abdel Salam declares that she has no conflict of interest. Ghada M. Hadad declares that she has no conflict of interest. Mohamed A. Abdelshakour declares that he has no conflict of interest.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Hameed, E.A.A., Abd-ElHamid, G.H., El-Darder, O.M. et al. Fast Sensitive and Accurate Analysis of the Most Common Synthetic Food Colorants in 65 Egyptian Commercial Products Using New HPLC–DAD and UPLC-ESI–MS/MS Methods. Food Anal. Methods 15, 3444–3457 (2022). https://doi.org/10.1007/s12161-022-02370-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12161-022-02370-8