Abstract
Rheumatoid arthritis (RA) is a complex and challenging autoimmune disease characterized by chronic inflammation of the joints, discomfort, stiffness, functional impairment, and systemic complications that affect millions of people around the world. Despite advances in medication, controlling RA remains difficult due to its complicated pathophysiology and numerous clinical symptoms. Synthetic medications, while effective, frequently cause considerable adverse effects, necessitating the investigation of alternate therapeutic options. This study attempts to provide a complete overview of synthetic medications and natural items used to treat RA. It specifically investigates the pathophysiological mechanisms that underpin RA and the efficacy and safety profiles of synthetic pharmaceuticals. Synthetic medications, such as disease-modifying antirheumatic drugs (DMARDs), and biologics remain important treatments for RA, albeit with hazards. So to explore safer and more effective therapy for the treatment of RA, a need to exploit the possible therapeutic benefits of natural items such as antioxidants, plant secondary metabolites, and traditional herbal remedies arises. By encompassing a spectrum of insights, from the molecular level to holistic traditional practices, this review aims to provide a holistic understanding of the role of natural products and traditional herbal medicines in the managerial landscape of RA. Furthermore, it investigates the effect of nutrition in regulating inflammation and disease development in RA. Integrative techniques that use natural products present intriguing adjuvant therapy, delivering anti-inflammatory, analgesic, and immunomodulatory effects with potentially fewer side effects. Understanding the interaction of synthetic medications and natural products, as well as the role of nutrition, can help enhance RA treatment regimens, improve patient outcomes, and reduce treatment-related problems. This study is a valuable resource for doctors, researchers, and patients looking for evidence-based methods for RA care. The synthesis of this knowledge contributes to the ongoing pursuit of enhanced therapeutic strategies, fostering improved outcomes and quality of life for individuals grappling with rheumatoid arthritis.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Rheumatoid arthritis (RA) is a systemic autoimmune disorder that develops in humans when the immune system works against the body. It mainly develops in the synovium lining of the bone joints, which can result in swelling and inflammation of the synovium. Inflammation of synovial joints can cause permanent damage to cartilage and bone. Furthermore, due to inflammation, the joint gradually loses its normal shape and reduces bone strength and the ability to function properly [1]. In addition, the tendons and ligaments responsible for joint stability can weaken over time. Typically, RA affects the smaller joints of the fingers, wrists, and ankles, but it can also affect various other parts of the body, including the eyes, lungs, blood vessels, heart, and skin. According to the World Health Organisation (WHO), 18 million people suffered from RA in 2019. RA is more prevalent among adults over the age of 50, with the majority among female populations. According to studies, people suffering from RA are around 0.1% of the world’s population. RA affects more than 70% of women and 55% of people over the age of 55 years. Additionally, 13 million people suffer from RA at severity levels (moderate or severe). In India, approximately 0.92% of the adult population have RA [2, 3].
The precise cause of RA remains unclear, but several factors contribute to its development. Previous research revealed that the cause and progression of RA depend on environmental, genetic, and infections. The other minor factors that stimulate the progression of RA include synovial damage, hyperplasia, and autoantigen modification. Environmental factors such as obesity, smoking, and specific pathogens such as Porphyromonas gingivalis can trigger RA factors. Genetic factors such as anomalies in cytokine synthesis, signal transduction, and T- and B-cell activity led to immune cell activation. Epigenetic modifications and genetic variations that affect the antigen response also play an essential role in the development of RA [4, 5]. The synovial damage and hyperplasia processes result in inflammatory conditions that aid in damaging synovial fibroblasts and causing them to grow abnormally. At the same time, autoantigen modification is a process known as citrullination, which eventually alters autoantigens. This alteration leads to neoepitopes because of the modified surface charge and the higher susceptibility to proteolytic degradation. This process leads to the development of RA [6]. A schematic representation of the potential cause of RA is given in Fig. 1. These factors stimulate and result in inflammation of the joints. At the same time, the body begins to oppose an invader that is not there.
Unlike RA, osteoarthritis is a common condition typically associated with joint wear and tear that occurs as people age. Unlike osteoarthritis, RA is an autoimmune disorder that can lead to disability if not adequately treated [7].
The scrutiny of RA causation and the underlying mechanisms, coupled with an in-depth analysis of synthetic drugs, biologics, and compounds that undergo clinical evaluation, underscores the need for a diversified therapeutic approach. The pivotal focus on natural products unveils a promising avenue to elucidate the molecular intricacies by which antioxidants and plant secondary metabolites contribute to RA mitigation. Traditional herbal medicine and extracts, subjected to rigorous examination, offer valuable insight into their pharmacological efficacy and established roles in the management of RA. Furthermore, giving recommended diets offers a pragmatic avenue for individuals to actively participate in their well-being. The review will improve knowledge, well-being, and outcomes for individuals living with RA.
Pathophysiology of RA
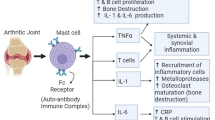
Immune dysregulation leads to autoimmunity in RA, which is thought to develop when genetically susceptible individuals are exposed to environmental triggers. RA can affect several organ systems, including blood vessels. The primary target site of autoimmunity is the cellular lining of the joints, known as the synovium. Both adaptive and innate immune responses contribute to the development of RA, with T-cell lymphocytes, particularly Th1 and Th17 cells, prevalent in the synovium. The stimulated antigen-presenting in the lymph node interacts with T and Bursa-derived cells (B cells), which increases the production of chemokines and cytokines, lymphocyte differentiation, and the formation of autoantibodies. T and B cells then migrate to the joint and interact with synoviocytes, macrophages, dendritic cells, and osteoclasts. As a result, more inflammatory cells enter the joint space, as well as the production of degradative enzymes, inflammatory cytokines (like TNF-alpha, IL-1, and IL-6), synoviocyte hyperplasia, and neoangiogenesis. To combat this perceived invader, Janus kinases and cytokines communicate during the inflammatory cycle, often causing further inflammation [8, 9]. Within the inflamed joint, specific cells such as macrophages, fibroblasts, and osteoclasts begin to damage cartilage and bone, contributing to the progression of the disease [10]. Synovitis is the result of these interactions and presents clinically as swollen, inflamed, and painful joints. Inflammatory destructive pannus is the result of untreated synovitis; damages bone, cartilage, and other articular tissues; and results in chronic pain and bone and joint deformities [8, 9]. A pictorial representation illustrating the pathophysiology of RA is given in Fig. 2.
Specific blood tests, including erythrocyte sedimentation rate (ESR), C-reactive protein (CRP), rheumatoid factor (RF), and anti-cyclic citrullinated peptide (anti-CCP) antibodies, are used in the diagnosis of RA [11]. Laboratory markers are essential, including elevated levels of CRP and ESR in blood serum, as well as detection of specific autoantibodies for RA, all of which are carefully examined by a medical professional as part of the diagnostic process. Furthermore, to diagnose and monitor the progression of RA in patients, medical professionals often recommend performing ultrasound and magnetic resonance imaging. In addition, the diagnosis of RA includes a complete assessment of the patient’s condition. This assessment considers various factors, including the patient’s symptoms, such as recent joint pain and swelling, morning stiffness, family history, and joint ultrasound sonography [12, 13].
Treatment for RA
The exact reason for RA is not known, but once damage to bone joints occurs, it cannot be reversed by anti-RA therapy. However, there are various drugs, supplements, and therapies available to minimize the effects of RA symptoms. The primary objective of RA treatment is to stop this inflammatory process and prevent further joint damage [2, 3]. Fortunately, there are several classes of drugs available that can reduce RA symptoms. Possible forms of treatment include disease-modifying antirheumatic drugs (DMARDs), methotrexate being a standard first-line therapy for RA. Furthermore, targeted synthetic DMARDs inhibit various kinases, including JAK or MAPK. Other treatment approaches include non-steroidal anti-inflammatory drugs (NSAIDs), glucocorticoids, joint stress reduction, physical and occupational therapy, and, when necessary, surgical intervention. These conventional medications are effective against RA symptoms and suppress inflammatory factors in vivo but show minimal effects on the improvement of the disease [14]. These drugs show some adverse side effects, such as substantial hepatotoxicity, cardiovascular disease, and others, which limit their therapeutic and clinical use [15]. Although these treatments can initially alleviate symptoms, people can eventually lose their ability to them; in that case, biologics are prescribed. Biologics are a class of medications that target specific parts of the immune system involved in inflammation and joint damage in RA. Biologics can significantly improve symptoms and slow disease progression. However, these biologics are associated with increased risks of severe infections such as tuberculosis and reactivation of hepatitis B and some of the rare side effects such as drug-induced lupus, demyelinating diseases, and lymphoma. Compared to DMARDs, biologics substantially increase treatment costs [16]. In severe cases, anti-TNF/Il6 biologics are prescribed, but their enhanced therapeutic potential is offset by reduced patient compliance due to invasive administration and a variety of adverse effects. These adverse effects include the gradual onset of immune deficiencies or autoimmune flares and the development of antidrug antibodies, resulting in decreased treatment effectiveness [17]. Treatment for RA, including synthetic drugs and biologics, is given in Table 1, and a list of compounds under clinical evaluation for the treatment of RA is shown in Table 2.
Treating RA is associated with challenges, such as a complex autoimmune nature, variability in the disease course, difficulty in early diagnosis, complex medication regimens, side effects of medications, cost of biologics, patient compliance, joint damage prevention, management of comorbidities, impact on mental health, and access to specialized care. RA management is a complex and personalized process that requires careful monitoring and coordination among healthcare providers. Given all the limitations, there is a pressing need for safer, more effective, and more affordable therapy options to alleviate and cure RA. A promising approach can be natural products such as traditional herbal products; it has been the result of a thoughtful and meticulous investigation of medications that have been used for many years and are known to be safer, with easy access to the general public [15].
Natural Products as a Promising Therapy to Tackle RA
Natural products, such as traditional herbal drugs, have potential benefits for the treatment of RA. They have fewer side effects than synthetic drugs and biologics and offer anti-inflammatory properties and pain relief. Herbal products are often used alongside conventional drugs as complementary therapies. Numerous extracts, active components, and isolated compounds from traditional herbal medicines (THMs), including alkaloids, flavonoids, triterpenes, and phenolic acids, have shown potential anti-RA properties based on emerging data and research. These bioactive substances alleviate RA symptoms by interacting with various targets involved in immune regulation, oxidative stress, miRNA, angiogenesis, and inflammatory responses [38].
Managing RA with Antioxidants
Several studies suggested the substantial involvement of oxidative stress involved in the development of RA. Oxidative stress, defined as an imbalance between the creation of harmful reactive oxygen species (ROS) and the body’s ability to mitigate their impact, has been linked to inflammation in tissue and is responsible for the harm in RA. In addition to inflammatory cytokines and matrix metalloproteinases, RA causes an excess release of free radicals. This excessive release contributes to inflammation and facilitates interactions among host cells, including chondrocytes, fibroblasts, and osteoclasts [39, 40]. Due to inflammatory responses, inflammatory cells (neutrophils) produce oxidant substances. In the plasma membrane, neutrophils have NADPH oxidase enzyme. Superoxide radicals of O2 are a form of ROS produced when bacteria and immunological complexes stimulate cells. Other ROS are produced as a result of radical reactions [41]. Monocytes, macrophages, and granulocytes produce free radicals. Monocytes, for example, generate 2.7 times more oxygen radicals in individuals with RA than in healthy individuals. During the electron transport process, oxidative phosphorylation is a source of mitochondrial reactive oxygen species (mtROS) within mitochondria. In addition to ROS, including peroxide, superoxide, and hydroxyl, free radicals also encompass RNS. The production of reactive nitrogen and oxygen species is a defense mechanism of the body against various bacterial infections, but their continuous excess results in the degeneration of cartilage and bone. Free radicals can damage biological molecules such as proteins, low-density lipoprotein (LDL), lipids, and DNA; they can also damage unsaturated fatty acids in cell membranes, leading to loss of cellular integrity and functional changes in cells’ receptors and enzymes [42, 43]. Through the MAPK pathway, mtROS promotes inflammatory cytokines (IL-6 s and TNF-α). To some extent, increased ROS production and loss in the body’s capacity to remove them are responsible for the progression of RA. The claim mentioned above is supported by the metabolites induced by free radicals observed in synovial fluid [44].
The measurement of total antioxidant capacity (TAC) can be used to assess serum antioxidant status. A lower TAC indicates an increased free radical activity. In patients with RA, the antioxidant defense system experiences decreased levels of TAC, antioxidant enzymes (superoxide dismutase, glutathione peroxidase, and catalase), and high malondialdehyde secretion [45]. Epidemiologic studies have also shown an unfavorable correlation between dietary antioxidant intake and the appearance of RA. Dietary intake of antioxidants is lower in RA patients, and their blood levels showed decreased antioxidant concentrations. Compared to a healthy person, in RA patients, synovial fluid and serum contain lower antioxidant levels. Yet the ongoing generation of free radicals in inflamed joints results in the downside of the antioxidant system (both enzymatic and non-enzymatic), leading to tissue destruction [46]. A possible explanation for this condition could be the inadequate intake of natural antioxidants from food, as we discovered from the literature that the dietary intake of micronutrients with vitamin E (alpha-tocopherol), vitamin C, provitamin A, selenium, zinc, and beta-carotene was less than the recommended amounts in RA patients. The patient’s food intake is affected by loss of appetite and impaired absorption and metabolism of dietary micronutrients. Other causes may include the inability to prepare food and the avoidance of certain foods due to fictitious diets [47]. These are some of the antioxidant supplements that have been researched concerning RA.
Vitamins as Antioxidants and Their Managerial Role in RA Therapy
Vitamin C is a potent antioxidant. It is present in oranges, grapefruits, lemons, strawberries, kiwis, bell peppers, and broccoli. Some studies have suggested that it might help reduce inflammation and oxidative stress in patients with RA. Vitamin C is capable of neutralizing harmful free radicals. Additionally, vitamin C is vital for collagen production, a structural protein in cartilage, connective tissues, and bones. Furthermore, adequate vitamin C intake can support tissue repair and potentially decelerate the progression of joint deterioration [48]. Vitamin C has the potential to improve iron absorption without heme and may also impact the absorption of other minerals and medications, including NSAIDs and DMARDs, frequently used in the treatment of RA [49].
Vitamin E is a fat-soluble protein that has been investigated for its potential anti-inflammatory properties and protects cells and tissues from oxidative damage. It is commonly found in nuts, including almonds and hazelnuts, seeds such as sunflower seeds and pumpkin seeds, spinach, avocados, and vegetable oils [50]. Vitamin E, particularly alpha-tocopherol, is a powerful antioxidant that can be beneficial in neutralizing free radicals. The antioxidant properties of vitamin E can help decrease oxidative stress and protect joint tissues. Vitamin E may have a mild analgesic (pain reliever) effect. Although it is not a substitute for RA medications, it can contribute to overall pain management when used as part of a comprehensive treatment plan [51].
Beta-carotene is an antioxidant and belongs to a class of carotenoids. Foods rich in beta-carotene, a precursor to vitamin A, include carrots, sweet potatoes, butternut squash, and dark leafy greens such as spinach and kale. Beta-carotene acts as a potential antioxidant, countering free radicals, and highly reactive molecules that can foster oxidative stress and tissue harm. In RA, where oxidative stress can induce joint damage and inflammation, reducing it could prove advantageous for those affected. Moreover, beta-carotene is essential for a healthy immune system. Some research suggests that a diet rich in carotenoids, including beta-carotene, may have mild anti-inflammatory effects [52].
Elements as Antioxidants and Their Managerial Role in RA Therapy
Selenium is a trace mineral that acts as an antioxidant. Some studies have explored its potential benefits in people with RA conditions. Selenium is found in foods such as nuts, fish, and whole grains [53]. Selenium is a component of selenoproteins, including glutathione peroxidases (GPx), which function as antioxidants in the body. These selenoproteins aid in counteracting detrimental free radicals and diminishing oxidative stress. In RA, oxidative stress can contribute to joint inflammation and damage, and selenium’s antioxidant properties can help mitigate these effects. Selenium is crucial for supporting joint health by diminishing inflammation and curbing oxidative damage within the joints. It has the potential to decelerate the progression of joint damage in RA [54].
Zinc is a mineral found in lean meats, nuts, and legumes. It supports the immune system and helps to repair tissues. It is a cofactor for multiple antioxidant enzymes, including superoxide dismutase (SOD), which helps neutralize harmful superoxide radicals in the body. These enzymes work to counteract oxidative stress and reduce the detrimental impacts of free radicals, which are implicated in RA inflammation and joint damage [55]. Moreover, zinc is essential to maintain a healthy immune system. In RA, the immune system becomes disrupted, leading to chronic inflammation and joint tissue attacks. Adequate zinc levels may support immune function and help modulate the autoimmune response observed in RA. Zinc synthesizes collagen. Furthermore, sufficient zinc levels in the blood can support tissue repair and potentially slow the progression of joint damage [56].
Fatty Acids as Antioxidants and Their Managerial Role in RA Therapy
Omega-3 fatty acids are found in fatty fish such as salmon, mackerel, sardines, flaxseeds, and walnuts and have anti-inflammatory properties. Some research suggests that omega-3 fatty acids may help reduce inflammation and joint pain in RA. Omega-3 fatty acids (specifically EPA and DHA) have been shown to reduce inflammation in the body. It can regulate the immune response and reduce the generation of pro-inflammatory substances, which can alleviate RA symptoms [57]. Additionally, omega-3 fatty acids have well-documented cardiovascular benefits, such as reducing triglycerides, improving blood vessel function, and potentially lowering the risk of heart disease, which can be particularly relevant for individuals with RA [58].
Plant Secondary Metabolites as a Managerial Role in RA Therapy
Certain secondary plant metabolites with managerial roles in RA therapy are given in Table 3, and peptides as potential RA therapy are shown in Table 4.
The plausible mechanism of action of secondary metabolites as anti-RA is depicted in Fig. 3.
Traditional Herbal Medicines and Herbal Extracts as a Managerial Therapy of RA
Green Tea
Green tea extract comprises antioxidants called catechins, with the most abundant and well-studied catechin being epigallocatechin gallate (EGCG). Green tea extract has gained attention for its possible health benefits, including its antioxidant and anti-inflammatory properties. The anti-inflammatory properties of green tea extract could potentially reduce inflammation and relieve RA symptoms [91]. Green tea extract may also support the immune system by modulating immune cell activity. Polyphenols from green tea, including EGCG, can inhibit enzyme activity involved in oxidative stress, such as xanthine oxidase and myeloperoxidase. By inhibiting these enzymes, green tea extract may help reduce oxidative damage [92].
Willow Bark
Willow bark, which has been used for generations to reduce pain and inflammation, is likely to be used as a complementary therapy for the treatment of RA. Bark extracts have been used in areas including South America, Egypt, Classical Greece, and China, according to historical documents [93]. Willow bark contains salicin, a precursor to salicylic acid, which is also found as an active component in aspirin. This substance inhibits the enzymes involved in prostaglandin formation, which reduces inflammation and pain. Salicylic acid and other components of willow bark contribute to its anti-inflammatory actions, which may help patients with RA with joint swelling, stiffness, and discomfort. Like aspirin, willow bark also helps to lower body temperature, offering relief from RA-related fever [94].
Stinging Nettle
Stinging nettle (Urtica dioica) belongs to a rare herbaceous perennial flower plant with stinging hairs native to Asia, Africa, and Europe. The nettle leaf extract complimented each other in the experimental, clinical, and trial phases. It is a well-known plant with a long history of using stems, leaves, and roots. It is used as a source of energy, such as soup or curry, as well as fiber, and as a medicinal herb. Nettle leaf extracts are used as anti-inflammatory treatments for RA [95]. Nettle includes several anti-inflammatory substances, including histamine, serotonin, and chlorogenic acid. These can help RA sufferers minimize joint swelling, pain, and stiffness. The stinging sensation caused by the hairs of the nettles functions as a counterirritant, diverting the brain’s attention away from the discomfort caused by inflamed joints. Nettle may also have an indirect effect on pain pathways. Nettle acts as a diuretic, encouraging the elimination of excess fluids and toxins that could contribute to inflammation in patients with RA [96].
Thunder God Vine
Thunder god vine (Tripterygium wilfordii Hook F), also known as Lei Gong Teng, has been adopted for generations in traditional Chinese medicine to treat inflammation. Extraction of Tripterygium wilfordii Hook F has been shown in animal studies to suppress both inflammatory and immunological reactions. In vitro studies have revealed that Tripterygium wilfordii Hook F components reduce immunological responses by decreasing the transcription of cytokine genes such as IL-2 and gamma interferon [97]. Thunder god vine includes various active substances with anti-inflammatory effects, including triptolide and celastrol. These substances reduce joint inflammation and pain by suppressing the activity of inflammatory agents such as cytokines and enzymes. It has the potential to modify the immune system, inhibiting hyperactive immunological responses that contribute to the development of RA. This can help to regulate an autoimmune attack on joint tissues. Thunder god vine has antioxidant activity, which means it can eliminate harmful free radicals that cause tissue damage and inflammation in RA patients [98].
Olive Oil
Olive oil contains polyphenols, including hydroxytyrosol and oleuropein, which have antioxidant properties. Antioxidants help to eliminate potentially harmful free radicals and reduce oxidative stress, two factors in RA that can lead to joint damage and inflammation. A higher risk of cardiovascular disease is associated with RA. Monounsaturated fats in olive oil and their antioxidant and anti-inflammatory compounds can positively impact cardiovascular health, which can be particularly relevant for people with RA [99].
Recommended Diets to Manage RA
A well-defined balanced diet that incorporates various antioxidant-rich foods and other healthy lifestyle habits can improve overall well-being. However, it is crucial to collaborate with your healthcare provider to establish a comprehensive strategy for effectively managing your RA [100]. The food pyramid for RA patients is given in Fig. 4.
Conclusion and Future Perspective
RA is an autoimmune disorder that affects joints, cartilage, and synovium lining of bones, finally leading to permanent damage to the bones and joints. Several treatments for RA include DMARDs, NSAIDs, glucocorticoids, and biologics. However, these therapies have significant drawbacks, including adverse effects, and biologics increase the risk for several pathogenic infections. Thus, there is a need for alternative managerial therapies which can support the RA treatment.
Herbal products can be used alongside conventional drugs as complementary therapies. Several plant-based secondary metabolites and herbal extracts have shown potential anti-RA properties. We would like to highlight some of the significant contributions of anti-RA, including vitamins, elements, secondary metabolites, and extracts that could be used as next-generation anti-RA managerial therapies. For example, vitamin C is essential for cartilage and bones and supports tissue repairs. At the same time, vitamin E possesses mild analgesic effects, and beta-carotene helps scavenge the ROS. In the element’s category, selenium and zinc are part of the body’s antioxidant machinery, which helps diminish free radicals and oxidative stress. Among secondary metabolites, flavonoids such as hesperidin, liquiritin, tangeretin, genistein, apigenin, quercetin, and diarylheptanoids such as curcumin reduce inflammation by acting against several anti-inflammatory pathways. In addition, peptides such as apitoxin, agkistrodon, GLPP, psalmotoxin 1, and scolopendrasin IX showed promising anti-inflammatory activity by working against several inflammatory mediators.
The pivotal focus on natural products unveils a promising avenue to elucidate the molecular intricacies by which antioxidants and plant secondary metabolites contribute to RA mitigation. THM and extracts, subjected to rigorous examination, offer valuable insight into their pharmacological efficacy and established roles in the management of RA. The above-discussed secondary metabolites, herbal extracts, elements, and peptides are based on the in vitro studies. Thus, these secondary metabolites, peptides, elements, and vitamins should be evaluated with the anti-RA drugs to confirm their therapeutic potential in the in vivo model of RA. Also, these should be assessed for their synergistic activity with the known anti-inflammatory and anti-RA drugs to develop the possible combination therapy.
Abbreviations
- Anti-CCP:
-
Anti-cyclic citrullinated peptide
- AP-1:
-
Activated protein-1
- B cells:
-
Bursa-derived cells
- CRP:
-
C-reactive protein
- DMARD:
-
Disease-modifying antirheumatic drugs
- ESR:
-
Erythrocyte sedimentation rate
- IκB:
-
Inhibitor of kappa B
- IL-1:
-
Interleukin-1
- IL-6:
-
Interleukin-6
- JNK:
-
Jun N-terminal kinase
- LDL:
-
Low-density lipoprotein
- LPS:
-
Lipopolysaccharide
- MAPKs:
-
Mitogen-activated protein kinases
- MMPs:
-
Matrix metalloproteinases
- mtROS:
-
Mitochondrial reactive oxygen species
- NLRP3:
-
NOD-like receptor protein 3
- NF-κB:
-
Nuclear factor kappa B
- Nrf-2:
-
Nuclear factor erythroid 2-related factor 2
- NSAIDs:
-
Non-steroidal anti-inflammatory drugs
- RF:
-
Rheumatoid factor
- ROS:
-
Reactive oxygen species
- OCP:
-
Osteoclast precursors
- RA:
-
Rheumatoid arthritis
- STAT3:
-
Signal transducer and activator of transcription 3
- T cell:
-
Thymus cells
- TAC:
-
Total antioxidant capacity
- TAK1:
-
Transforming growth factor-β-activated kinase 1
- THMs:
-
Traditional herbal medicines
- TNF:
-
Tumor necrosis factor
- VGEF:
-
Vascular endothelial growth factor
References
Debreova M, Culenova M, Smolinska V, Nicodemou A, Csobonyeiova M, Danisovic L. Rheumatoid arthritis: from synovium biology to cell-based therapy. Cytotherapy. 2022;24(4):365–75.
Rheumatoid arthritis. 2023. https://www.who.int/news-room/fact-sheets/detail/rheumatoid-arthritis.
Gupta BM, Gupta R, Kumar A, Bansal M. Rheumatoid arthritis research in India: a scientometric assessment of publications during 2007–2016. SF J Orthopedic Rheumatol 2018.
Liao KP, Alfredsson L, Karlson EW. Environmental influences on risk for rheumatoid arthritis. Curr Opin Rheumatol. 2009;21(3):279.
McGraw WT, Potempa J, Farley D, Travis J. Purification, characterization, and sequence analysis of a potential virulence factor from Porphyromonas gingivalis, peptidylarginine deiminase. Infect Immun. 1999;67(7):3248–56.
Lin Y-J, Anzaghe M, Schülke S. Update on the pathomechanism, diagnosis, and treatment options for rheumatoid arthritis. Cells. 2020;9(4):880.
Anderson AS, Loeser RF. Why is osteoarthritis an age-related disease?, Best practice & research. Clin Rheumatol. 2010;24(1):15–26.
Littlejohn EA, Monrad SU. Early diagnosis and treatment of rheumatoid arthritis. Primary care: Clinics in office practice. 2018;45(2):237–55.
Guo Q, Wang Y, Xu D, Nossent J, Pavlos NJ, Xu J. Rheumatoid arthritis: pathological mechanisms and modern pharmacologic therapies. Bone res. 2018;6(1):15.
Cojocaru M, Cohocaru IM, Chico B. New insight into the rheumatoid vasculitis. Rom J Intern Med. 2015;53(2):128–32.
Charleson K. Types of rheumatoid arthritis. 2023. https://www.verywellhealth.com/types-of-rheumatoid-arthritis-5093089#citation-7. Accessed 11/11/2023 2023.
Aletaha D, Smolen JS. Diagnosis and management of rheumatoid arthritis: a review. JAMA. 2018;320(13):1360–72.
Burmester GR, Pope JE. Novel treatment strategies in rheumatoid arthritis. The Lancet. 2017;389(10086):2338–48.
Watanabe R, Okano T, Gon T, Yoshida N, Fukumoto K, Yamada S, Hashimoto M. Difficult-to-treat rheumatoid arthritis: current concept and unsolved problems. Front Med (Lausanne). 2022;9:1049875.
Wang Y, Chen S, Du K, Liang C, Wang S, Boadi EO, Li J, Pang X, He J. Chang Y-x, Traditional herbal medicine: therapeutic potential in rheumatoid arthritis. J Ethnopharmacol. 2021;279: 114368.
Joensuu JT, Aaltonen KJ, Aronen P, Sokka T, Puolakka K, Tuompo R, Korpela M, Vasala M, Ilva K, Nordström D. Cost-effectiveness of biologic compared with conventional synthetic disease-modifying anti-rheumatic drugs in patients with rheumatoid arthritis: a register study. Rheumatology. 2016;55(10):1803–11.
Papadopoulou D, Roumelioti F, Tzaferis C, Chouvardas P, Pedersen A-K, Charalampous F, Christodoulou-Vafeiadou E, Ntari L, Karagianni N, Denis MC. Repurposing the antipsychotic drug amisulpride for targeting synovial fibroblast activation in arthritis. JCI insight. 2023;8:9.
Benjamin O, Goyal A, Lappin SL. Disease-modifying antirheumatic drugs (DMARD). In: StatPearls. Treasure Island (FL): StatPearls Publishing; 2023.
Choi J, Fenando A. Sulfasalazine. StatPearls, StatPearls Publishing; 2022.
Ghlichloo I, Gerriets V. Nonsteroidal anti-inflammatory drugs (NSAIDs). StatPearls, StatPearls Publishing; 2023.
Waller DG, Sampson AP. 30 - Rheumatoid arthritis, other inflammatory arthritides and osteoarthritis, in: D.G. Waller, A.P. Sampson (Eds.). Medical Pharmacology and Therapeutics (Fifth Edition). Elsevier; 2018. pp. 373–383.
Cohen S, Tuckwell K, Katsumoto TR, Zhao R, Galanter J, Lee C, Rae J, Toth B, Ramamoorthi N, Hackney JA. Fenebrutinib versus placebo or adalimumab in rheumatoid arthritis: a randomized, double-blind, phase II trial. Arthritis Rheumatol. 2020;72(9):1435–46.
Wu H, Yan S, Chen J, Luo X, Li P, Jia X, Dai X, Wang C, Huang Q, Liu L. JAK1-STAT3 blockade by JAK inhibitor SHR0302 attenuates inflammatory responses of adjuvant-induced arthritis rats and decreases Th17 and total B cells. Joint Bone Spine. 2016;83(5):525–32.
Conaghan PG, Nowak M, Du S, Luo Y, Landis J, Pachai C, Fura A, Catlett IM, Grasela DM, Østergaard M. Evaluation of BMS-986142, a reversible Bruton’s tyrosine kinase inhibitor, for the treatment of rheumatoid arthritis: a phase 2, randomised, double-blind, dose-ranging, placebo-controlled, adaptive design study. Lancet Rheumatol. 2023;5(5):e263–73.
Tanaka Y, Genovese MC, Matsushima H, Long-term safety, efficacy, and patient-centered outcomes of filgotinib in the treatment of rheumatoid arthritis: current perspectives, patient preference and adherence 2023;2499–2516.
Peterfy CG, Strand V, Friedman A, Hall S, Mysler E, Durez P, Baraliakos X, Enejosa JV, Shaw T, Li Y. Inhibition of structural joint damage progression with upadacitinib in rheumatoid arthritis: 1-year outcomes from the SELECT phase 3 program. Rheumatology. 2022;61(8):3246–56.
Daien C, Krogulec M, Gineste P, Steens J-M, Du Roure LD, Biguenet S, Scherrer D, Santo J, Ehrlich H, Durez P. Safety and efficacy of the miR-124 upregulator ABX464 (obefazimod, 50 and 100 mg per day) in patients with active rheumatoid arthritis and inadequate response to methotrexate and/or anti-TNFα therapy: a placebo-controlled phase II study. Ann Rheum Dis. 2022;81(8):1076–84.
Yoon S-B, Hong H, Lim H-J, Choi JH, Choi YP, Seo SW, Lee HW, Chae CH, Park W-K, Kim HY. A novel IRAK4/PIM1 inhibitor ameliorates rheumatoid arthritis and lymphoid malignancy by blocking the TLR/MYD88-mediated NF-κB pathway. Acta Pharmaceutica Sinica B. 2023;13(3):1093–109.
Li J, Li M, Wu D, Zhou J. Leung S-o, Zhang F, SM03, an anti-human CD22 monoclonal antibody, for active rheumatoid arthritis: a phase II, randomized, double-blind, placebo-controlled study. Rheumatology. 2022;61(5):1841–8.
Gordon D, Kivitz A, Singhal A, Burt D, Bangs MC, Huff EE, Hope HR, Monahan JB. Selective inhibition of the MK2 pathway: data from a phase IIa randomized clinical trial in rheumatoid arthritis. ACR Open Rheumatology. 2023;5(2):63–70.
Robak E, Robak T. Bruton’s kinase inhibitors for the treatment of immunological diseases: current status and perspectives. J Clin Med. 2022;11(10):2807.
Montalban X, Wallace D, Genovese MC, et al. Characterisation of the safety profile of evobrutinib in over 1000 patients from phase II clinical trials in multiple sclerosis, rheumatoid arthritis and systemic lupus erythematosus: an integrated safety analysis. J Neurol Neurosurg Psychiatry. 2023;94(1):1–9.
Abuelazm M, Ghanem A, Mahmoud A, et al. The efficacy and safety of olokizumab for rheumatoid arthritis: a systematic review, pairwise, and network meta-analysis. Clin Rheumatol. 2023;42(6):1503–20.
Takeuchi T, Chino Y, Kawanishi M, Nakanishi M, Watase H, Mano Y, Sato Y, Uchida S, Tanaka Y. Efficacy and pharmacokinetics of ozoralizumab, an anti-TNFα NANOBODY® compound, in patients with rheumatoid arthritis: 52-week results from the OHZORA and NATSUZORA trials. Arthritis Res Ther. 2023;25(1):60.
Taylor PC, Weinblatt ME, McInnes IB, Atsumi T, Strand V, Takeuchi T, Bracher M, Brooks D, Davies J, Goode C. Anti-GM-CSF otilimab versus sarilumab or placebo in patients with rheumatoid arthritis and inadequate response to targeted therapies: a phase III randomised trial (contRAst 3). Ann Rheum Dis. 2023;82(12):1527–37.
Muralidharan S, Njenga M, Garron T, Bondensgaard K, Paolini JF. Preclinical immunopharmacologic assessment of KPL-404, a novel, humanized, non-depleting antagonistic anti-CD40 monoclonal antibody. J Pharmacol Exp Ther. 2022;381(1):12–21.
Buttgereit F, Aelion J, Rojkovich B, Zubrzycka-Sienkiewicz A, Chen S, Yang Y, Arikan D, D’Cunha R, Pang Y, Kupper H. Efficacy and safety of ABBV-3373, a novel anti–tumor necrosis factor glucocorticoid receptor modulator antibody–drug conjugate, in adults with moderate-to-severe rheumatoid arthritis despite methotrexate therapy: a randomized, double-blind, active-controlled proof-of-concept phase IIa trial. Arthritis & Rheumatology. 2023;75(6):879–89.
Deligiannidou G-E, Gougoula V, Bezirtzoglou E, Kontogiorgis C, Constantinides TK. The role of natural products in rheumatoid arthritis: current knowledge of basic in vitro and in vivo research. Antioxidants. 2021;10(4):599.
Edilova MI, Akram A, Abdul-Sater AA. Innate immunity drives pathogenesis of rheumatoid arthritis. biomed j. 2021;44(2):172–82.
Laurindo LF, de Maio MC, Barbalho SM, Guiguer EL, Araújo AC, de Alvares GR, Flato UAP, Júnior EB, Detregiachi CRP, dos Santos Haber JF. Organokines in rheumatoid arthritis: a critical review. Int J Mol Sci. 2022;23(11):6193.
Veselinovic M, Barudzic N, Vuletic M, Zivkovic V, Tomic-Lucic A, Djuric D, Jakovljevic V. Oxidative stress in rheumatoid arthritis patients: relationship to diseases activity. Mol Cell Biochem. 2014;391:225–32.
Wang X, Fan D, Cao X, Ye Q, Wang Q, Zhang M, Xiao C. The role of reactive oxygen species in the rheumatoid arthritis-associated synovial microenvironment. Antioxidants. 2022;11(6):1153.
Finaud J, Lac G, Filaire E. Oxidative stress: relationship with exercise and training. Sports Med. 2006;36:327–58.
Naik E, Dixit VM. Mitochondrial reactive oxygen species drive proinflammatory cytokine production. J Exp Med. 2011;208(3):417–20.
Mititelu RR, Pădureanu R, Băcănoiu M, Pădureanu V, Docea AO, Calina D, Barbulescu AL, Buga AM. Inflammatory and oxidative stress markers—mirror tools in rheumatoid arthritis. Biomedicines. 2020;8(5):125.
Hitchon CA, El-Gabalawy HS. Oxidation in rheumatoid arthritis. Arthritis Res Ther. 2004;6(6):1–14.
Cerhan JR, Saag KG, Merlino LA, Mikuls TR, Criswell LA. Antioxidant micronutrients and risk of rheumatoid arthritis in a cohort of older women. Am J Epidemiol. 2003;157(4):345–54.
Carr AC, Maggini S. Vitamin C and immune function. Nutrients. 2017;9(11):1211.
Hemilä H. Vitamin C and infections. Nutrients. 2017;9(4):339.
Traber MG. Atkinson J. Vitamin E, antioxidant and nothing more, free radical biology and medicine. 2007;43(1):4–15.
Kamanlı A, Nazıroğlu M, Aydılek N, Hacıevlıyagil C. Plasma lipid peroxidation and antioxidant levels in patients with rheumatoid arthritis. Cell Biochem Funct. 2004;22(1):53–7.
Elmadfa R. Majchrzak. Wagner, Genser, Lettner, Pinter, Effects of beta-carotene supplementation on free radical mechanism in healthy adult subjects, International journal for vitamin and nutrition research. 2004;74(2):147–52.
Hu W, Zhao C, Hu H, Yin S. Food sources of selenium and its relationship with chronic diseases. Nutrients. 2021;13(5):1739.
Turrubiates-Hernández FJ, Márquez-Sandoval YF, González-Estevez G, Reyes-Castillo Z, Muñoz-Valle JF. The relevance of selenium status in rheumatoid arthritis. Nutrients. 2020;12(10):3007.
Prasad AS. Discovery of human zinc deficiency: its impact on human health and disease. Adv Nutr. 2013;4(2):176–90.
Shankar AH, Prasad AS. Zinc and immune function: the biological basis of altered resistance to infection. Am J Clin Nutr. 1998;68(2):447S-463S.
Calder PC. Marine omega-3 fatty acids and inflammatory processes: effects, mechanisms and clinical relevance. Biochim Biophys Acta Mol Cell Biol Lipids. 2015;1851(4):469–84.
Rizos EC, Ntzani EE, Bika E, Kostapanos MS, Elisaf MS. Association between omega-3 fatty acid supplementation and risk of major cardiovascular disease events: a systematic review and meta-analysis. JAMA. 2012;308(10):1024–33.
Qi W, Lin C, Fan K, Chen Z, Liu L, Feng X, Zhang H, Shao Y, Fang H, Zhao C, Zhang R, Cai D. Hesperidin inhibits synovial cell inflammation and macrophage polarization through suppression of the PI3K/AKT pathway in complete Freund’s adjuvant-induced arthritis in mice. Chem Biol Interact. 2019;306:19–28.
Zhai K-F, Duan H, Cui C-Y, Cao Y-Y, Si J-L, Yang H-J, Wang Y-C, Cao W-G, Gao G-Z, Wei Z-J. Liquiritin from Glycyrrhiza uralensis attenuating rheumatoid arthritis via reducing inflammation, suppressing angiogenesis, and inhibiting MAPK signaling pathway. J Agric Food Chem. 2019;67(10):2856–64.
Li X, Xie P, Hou Y, Chen S, He P, Xiao Z, Zhan J, Luo D, Gu M, Lin D. Tangeretin inhibits oxidative stress and inflammation via upregulating Nrf-2 signaling pathway in collagen-induced arthritic rats. Pharmacology. 2019;104(3–4):187–95.
Cheng W-X, Huang H, Chen J-H, Zhang T-T, Zhu G-Y, Zheng Z-T, Lin J-T, Hu Y-P, Zhang Y, Bai X-L, Wang Y, Xu Z-W, Song B, Mao Y-Y, Yang F, Zhang P. Genistein inhibits angiogenesis developed during rheumatoid arthritis through the IL-6/JAK2/STAT3/VEGF signalling pathway. J Orthop Transl. 2020;22:92–100.
Liu X, Wang Z, Qian H, Tao W, Zhang Y, Hu C, Mao W, Guo Q. Natural medicines of targeted rheumatoid arthritis and its action mechanism. Front Immunol. 2022;13: 945129.
Yuan K, Zhu Q, Lu Q, Jiang H, Zhu M, Li X, Huang G, Xu A. Quercetin alleviates rheumatoid arthritis by inhibiting neutrophil inflammatory activities. J Nutr Biochem. 2020;84: 108454.
Feng Z-T, Yang T, Hou X-Q, Wu H-Y, Feng J-T, Ou B-J, Cai S-J, Li J, Mei Z-G. Sinomenine mitigates collagen-induced arthritis mice by inhibiting angiogenesis. Biomed Pharmacother. 2019;113.
Liang J, Chang B, Huang M, Huang W, Ma W, Liu Y, Tai W, Long Y, Lu Y. Oxymatrine prevents synovial inflammation and migration via blocking NF-κB activation in rheumatoid fibroblast-like synoviocytes. Int Immunopharmacol. 2018;55:105–11.
Xu R, Liu Z, Hou J, Huang T, Yang M. Osthole improves collagen-induced arthritis in a rat model through inhibiting inflammation and cellular stress. Cell Mol Biol Lett. 2018;23(1):1–11.
Lin W, Chen G, Mao Y, Ma X, Zhou J, Yu X, Wang C, Liu M. Imperatorin inhibits proliferation, migration, and inflammation via blocking the NF-κB and MAPK pathways in rheumatoid fibroblast-like synoviocytes. ACS Omega. 2022;7(34):29868–76.
Wang W, Sun W, Jin L. Caffeic acid alleviates inflammatory response in rheumatoid arthritis fibroblast-like synoviocytes by inhibiting phosphorylation of IκB kinase α/β and IκBα. Int Immunopharmacol. 2017;48:61–6.
Fikry EM, Gad AM, Eid AH, Arab HH. Caffeic acid and ellagic acid ameliorate adjuvant-induced arthritis in rats via targeting inflammatory signals, chitinase-3-like protein-1 and angiogenesis. Biomed Pharmacother. 2019;110:878–86.
Liu N, Feng X, Wang W, Zhao X, Li X. Paeonol protects against TNF-α-induced proliferation and cytokine release of rheumatoid arthritis fibroblast-like synoviocytes by upregulating FOXO3 through inhibition of miR-155 expression. Inflamm Res. 2017;66(7):603–10.
Meng D, Li J, Li H, Wang K. Salvianolic acid B remits LPS-induced injury by up-regulating miR-142-3p in MH7A cells. Biomed Pharmacother. 2019;115: 108876.
Mohammadian Haftcheshmeh S, Khosrojerdi A, Aliabadi A, Lotfi S, Mohammadi A, Momtazi-Borojeni AA. Immunomodulatory effects of curcumin in rheumatoid arthritis: evidence from molecular mechanisms to clinical outcomes. Rev Physiol Biochem Pharmacol. 2021;179:1–29.
Chen J, Wu W, Zhang M, Chen C. Taraxasterol suppresses inflammation in IL-1β-induced rheumatoid arthritis fibroblast-like synoviocytes and rheumatoid arthritis progression in mice. Int Immunopharmacol. 2019;70:274–83.
Yu WG, Shen Y, Wu JZ, Gao YB, Zhang LX. Madecassoside impedes invasion of rheumatoid fibroblast-like synoviocyte from adjuvant arthritis rats via inhibition of NF-κB-mediated matrix metalloproteinase-13 expression. Chin J Nat Med. 2018;16(5):330–8.
Gao Q, Qin H, Zhu L, Li D, Hao X. Celastrol attenuates collagen-induced arthritis via inhibiting oxidative stress in rats. Int Immunopharmacol. 2020;84: 106527.
Li N, Gong Z, Li X, Ma Q, Wu M, Liu D, Deng L, Pan D, Liu Q, Wei Z, Wang Q, Han L, Lin C, Chen J. Betulinic acid inhibits the migration and invasion of fibroblast-like synoviocytes from patients with rheumatoid arthritis. Int Immunopharmacol. 2019;67:186–93.
Zhang C, Ma K, Yang Y, Wang F, Li W. Glaucocalyxin A suppresses inflammatory responses and induces apoptosis in TNF-a-induced human rheumatoid arthritis via modulation of the STAT3 pathway. Chem Biol Interact. 2021;341: 109451.
Li X, Yuan K, Zhu Q, Lu Q, Jiang H, Zhu M, Huang G, Xu A. Andrographolide ameliorates rheumatoid arthritis by regulating the apoptosis-NETosis balance of neutrophils. Int J Mol Sci. 2019;20:20.
Yuan K, Li X, Lu Q, Zhu Q, Jiang H, Wang T, Huang G, Xu A. Application and Mechanisms of triptolide in the treatment of inflammatory diseases-a review. Front Pharmacol. 2019;10:1469.
Aufschnaiter A, Kohler V, Khalifa S, Abd El-Wahed A, Du M, El-Seedi H, Büttner S. Apitoxin and its components against cancer, neurodegeneration and rheumatoid arthritis: Limitations and possibilities. Toxins. 2020;12(2):66.
Mei L, Lin C, Lei S, Xu L, Fan YS. Small peptides compound isolated from agkistrodon with antiarthritic effect in collagen-induced arthritis rats. Evid Based Complement Alternat Med. 2018;2018:8265150.
Meng M, Wang L, Yao Y, Lin D, Wang C, Yao J, Sun H, Liu M. Ganoderma lucidum polysaccharide peptide (GLPP) attenuates rheumatic arthritis in rats through inactivating NF-κB and MAPK signaling pathways. Phytomedicine. 2023;119: 155010.
Zhou Q, Li T, Fang G, Pang Y, Wang X. Bioactive molecules against rheumatoid arthritis by suppressing pyroptosis. Pharmaceuticals. 2023;16(7):952.
Park YJ, Park B, Lee M, Jeong YS, Lee HY, Sohn DH, Song JJ, Lee JH, Hwang JS, Bae Y-S. A novel antimicrobial peptide acting via formyl peptide receptor 2 shows therapeutic effects against rheumatoid arthritis. Sci Rep. 2018;8(1):14664.
Wang X, Li L, Wang J, Dong L, Shu Y, Liang Y, Shi L, Xu C, Zhou Y, Wang Y. Inhibition of cytokine response to TLR stimulation and alleviation of collagen-induced arthritis in mice by Schistosoma japonicum peptide SJMHE 1. J Cell Mol Med. 2017;21(3):475–86.
Chen Y, Shao S, Huang J, Gu Y, Cheng Y, Zhu X. Therapeutic efficacy of a Trichinella spiralis paramyosin-derived peptide modified with a membrane-targeting signal in mice with antigen-induced arthritis. Front Microbiol. 2020;11: 608380.
Logashina YA, Palikova YA, Palikov VA, Kazakov VA, Smolskaya SV, Dyachenko IA, Tarasova NV, Andreev YA. Anti-inflammatory and analgesic effects of TRPV1 polypeptide modulator APHC3 in models of osteo-and rheumatoid arthritis. Mar Drugs. 2021;19(1):39.
Ni LL, Che YH, Sun HM, et al. The therapeutic effect of wasp venom (Vespa magnifica, Smith) and its effective part on rheumatoid arthritis fibroblast-like synoviocytes through modulating inflammation, redox homeostasis and ferroptosis. J Ethnopharmacol. 2023;317:116700.
Zhang Z, Wan H, Han J, Sun X, Yu R, Liu B, Lu C, Zhou J, Su X. Ameliorative effect of tuna elastin peptides on AIA mice by regulating the composition of intestinal microorganisms and SCFAs. J Funct Foods. 2022;92.
Hussain T, Gupta S, Adhami VM, Mukhtar H. Green tea constituent epigallocatechin-3-gallate selectively inhibits COX-2 without affecting COX-1 expression in human prostate carcinoma cells. Int J Cancer. 2005;113(4):660–9.
Fujiki H, Suganuma M. Green tea: an effective synergist with anticancer drugs for tertiary cancer prevention. Cancer Lett. 2012;324(2):119–25.
Wood JN. From plant extract to molecular panacea: a commentary on Stone (1763) ‘An account of the success of the bark of the willow in the cure of the agues.’ Philos Trans R Soc Lond B Biol Sci. 2015;370:1666.
Vane JR, Botting RM. The mechanism of action of aspirin. Thromb Res. 2003;110(5–6):255–8.
Dhouibi R, Affes H, Ben Salem M, Hammami S, Sahnoun Z, Zeghal KM, Ksouda K. Screening of pharmacological uses of Urtica dioica and others benefits. Prog Biophys Mol Biol. 2020;150:67–77.
Bhusal KK, Magar SK, Thapa R, Lamsal A, Bhandari S, Maharjan R, Shrestha S, Shrestha J. Nutritional and pharmacological importance of stinging nettle (Urtica dioica L.): A review, Heliyon 2022;8(6):e09717.
Lipsky PE, Tao XL. A potential new treatment for rheumatoid arthritis: thunder god vine. Semin Arthritis Rheum. 1997;26(5):713–23.
Tang Y, Liu Q, Feng Y, Zhang Y, Xu Z, Wen C, Zhang Y. Tripterygium ingredients for pathogenicity cells in rheumatoid arthritis. Front Pharmacol. 2020;11: 583171.
Covas M-I, Nyyssönen K, Poulsen HE, Kaikkonen J. Zunft H-JF, Kiesewetter H, Gaddi A, de la Torre R, Mursu J, Bäumler H, The effect of polyphenols in olive oil on heart disease risk factors: a randomized trial. Ann Intern Med. 2006;145(5):333–41.
Radu AF, Bungau SG. Management of rheumatoid arthritis: an overview. Cells. 2021;10:11.
Acknowledgements
The authors thank Dr. Ashok K. Chauhan, Founder President of the Amity group of Institutions, for offering support and providing us with the opportunity to work on this project. Additionally, I would like to express my appreciation to the esteemed teachers for their valuable insights, feedback, guidance, and support throughout the writing process of the article.
Author information
Authors and Affiliations
Contributions
N.G, S.P., and M. wrote the main manuscript G.K. helped in data compilation as well as manuscript structuring, and A.G. was involved in conceptualization, reviewing, and editing the manuscript. All authors reviewed the manuscript.
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Ghosh, N., Pathak, S., Malsawmdawngkimi et al. Natural Products and Traditional Herbal Medicines as Managerial Therapies to Combat Rheumatoid Arthritis. Clin & Translational Metab. 22, 2 (2024). https://doi.org/10.1007/s12018-024-09290-7
Accepted:
Published:
DOI: https://doi.org/10.1007/s12018-024-09290-7