Abstract
The present study was considered to assess the protective effects of boron (B) on gentamicin-induced oxidative stress, proinflammatory cytokines, and histopathological changes in rat kidneys. Rats were split into eight equal groups which were as follows: control (fed with low-boron diet); gentamicin group (100 mg/kg, i.p.); B5, B10, and B20 (5, 10, and 20 mg/kg B, i.p.) groups; gentamicin (100 mg/kg, i.p.) plus B5, B10, and B20 (5, 10, and 20 mg/kg B, i.p.) groups. B was given to rats 4 days before the gentamicin treatment and B administration was completed on the 14th day. Gentamicin administration was started on the 4th day and finished on the 12th day. Gentamicin increased malondialdehyde levels, while reduced glutathione levels in the blood and kidney. Furthermore, superoxide dismutase and catalase activities of erythrocyte were decreased. Besides, serum and kidney nitric oxide and 8-dihydroxyguanidine levels were increased by gentamicin. Additionally, serum levels and kidney mRNA expressions of TNF-α, NFκB, IL-1β, and IFN-γ were found to be the highest in the gentamicin group. Histopathologically, interstitial hemorrhage and tubular necrosis were detected in the kidneys of the gentamicin group. Nonetheless, B administration reversed gentamicin-induced lipid peroxidation, antioxidant status, and inflammation. In conclusion, B has a preventive effect against gentamicin-induced nephrotoxicity and ameliorates kidney tissues of the rat.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Among aminoglycosides, gentamicin has the widest spectrum and the highest bactericidal potency after amoxicillin. It is particularly effective on strains of Enterobacteriaceae (Escherichia coli, Klebsiella, Aerobacter, etc.) bacteria and Gram-negative bacilli such as Pseudomonas aeruginosa and methicillin-resistant Staphylococcus aureus [1]. Clinical usage of gentamicin is important due to its effect and low price [2, 3]. The most important side effects of aminoglycoside antibiotics are kidney dysfunction, loss of balance, and hearing. Especially in chronic diseases, renal failure, and metabolic diseases, adverse effects on the kidneys can be seen obviously. The formation of free oxygen radicals such as superoxide and hydroxyl radicals caused by gentamicin has been proposed to explain the gentamicin-mediated nephrotoxicity. Numerous studies investigated the side effect mechanisms of gentamicin and concluded that there may be a relationship between the increased lipid peroxidation (LPO) and nitric oxide (NO) with side effects in target tissues [4,5,6]. Several in vivo and in vitro studies suggest that gentamicin administration could result in inflammation due to one of the mechanisms of nephrotoxicity. In this toxicity, gentamicin activates tumor necrosis factor (TNF-α), interleukin (IL-1β), and interferon (IFN-γ) which are responsible for inflammation [7, 8].
Boron is mainly provided in trace amount by plants and also, recent studies indicated that B showed beneficial effects on inflammatory and oxidative stress, DNA damage, and S-adenosylhomocysteine involvement in methylation and homocysteine levels. By means of these effects, nutritional intakes of B moderate or alleviate several pathological conditions related to aging, cancer, sarcopenia, cognitive decline, and bone health [9]. B also improves fetal development and enhances early embryonic gene expressions of rats [10]. In order to investigate the effects of B compounds on biochemical parameters such as thyroid hormones, leptin, insulin, and carnitine levels were analyzed and it was determined that the effect of borax on hormonal status was more dominant [11]. In other studies, boric acid and borax were used together; it was expressed that they decreased LPO levels and DNA damage by enhancing the antioxidant activity [12,13,14]. Additionally, B compounds have a protective effect against malathion- [15], bisphenol- [16], arsenic- [17], cadmium- [18], and acrylamide-induced oxidative stress [19] in rats. So far, there have been no reports on the effect of B compounds against gentamicin-induced nephrotoxicity. Therefore, we hypothesized that B compounds could suppress inflammation, oxidative, and DNA damage caused by gentamicin.
In this study, it was aimed to determine the effect of B on gentamicin-induced DNA damage {8-dihydroxyguanidine (8-OHDG)}, LPO, and antioxidant markers {malondialdehyde (MDA), reduced glutathione (GSH), NO, superoxide dismutase (SOD), and catalase (CAT)} in the blood and kidney of rats. In addition, serum and kidney TNF-α, IFN-γ, IL-1β, and NFκB levels were analyzed by ELISA and mRNA expressions of these genes in the kidney were determined by real-time PCR. Also, histopathological changes in the kidney were evaluated by light microscope.
Materials and Methods
Chemicals
Gentamicin and boric acid as a boron source were provided from Sigma-Aldrich Co. (MO, USA) and other analytical pure chemicals were supplied from the companies in Turkey.
Experimental Design
After approval of the Local Ethical Committee on Animal Research (49533702-62), 2 months of age male Albino Wistar rats (250–350 g) was obtained from the Afyon Kocatepe University Experimental Animal Implementation and Research Center, Turkey. Animals were kept at suitable conditions (12-h light/dark period, 50–55% humidity, and 25 °C), deionized drinking water was given to rats, and they were fed with low-B diet which was prepared according to Hunt [20] as shown in Table 1.
In the experiment, a total of 56 rats were split into 8 equal groups. Experimental design was carried out as follows: control (fed with low-boron diet); gentamicin group (100 mg/kg, i.p.), B5, B10, and B20 (5, 10, and 20 mg/kg B, i.p.) groups; gentamicin (100 mg/kg, i.p.) plus B5, B10, and B20 (5, 10, and 20 mg/kg B, i.p.) groups. B was administered to rats 4 days before the gentamicin treatment and B administration was completed on the 14th day. Administration of gentamicin was started on the 4th day and finished on the 12th day. Gentamicin and B doses were chosen according to Ateşşahin et al. [22] and Ince et al. [21], respectively. End of the administrations, blood and kidney tissues were collected under anaesthesia (xylazine/ketamine). The method of Winterbourn et al. [23] was carried out to determine the antioxidant enzyme activities of rat erythrocytes. Additionally, biochemical, molecular, and histopathological analyses were performed to detect the effect of B and gentamicin on kidney tissues.
Biochemical Analyses
Blood urea nitrogen (BUN) and creatinine levels of serum, as kidney function parameters, were assayed with commercial kits (Biolabo, Maizy, France). MDA levels [24, 25] in the blood and kidney, GSH levels [26] in the blood and kidney, activities of SOD [27] and CAT [28, 29] in the erythrocyte and kidney, NO levels [30] in the serum and kidney, and protein [31] and hemoglobin levels [32] were measured by Shimadzu 1601-UV spectrophotometer (Tokyo, Japan). Also, TNF-α, NFκB, IL-1β, IFN-γ, and 8-OHdG levels in serum and tissue were determined by commercial rat ELISA kits (MyBioSource, San Diego, CA, USA).
Determination of mRNA Expression Levels of Proinflammatory Cytokines
Total RNA of the kidney was extracted and reversed transcribed using Tripure Reagent (Roche, Germany) and RT2 HT first-strand kit (Qiagen, Germany), respectively following the manufacturer’s protocols. Primers were ordered from Ella Biotech (Martinsried, Germany) and shown in Table 2. Each sample was analyzed in triplicate. Also, normalization was performed according to the housekeeping gene GAPDH expression level. The results are stated as relative gene expression using the delta–delta CT method [33].
Histopathological Analyses
For histopathological analysis, kidney tissues of rats were fixed in 10% formalin solution for 48 h. Fixed tissues were dehydrated by graded alcohol solutions (70–100%). Afterwards, they were objected to xylene and embedded in paraffin blocks which were sliced sections (5–6 μm), stained with hematoxylin–eosin (H&E) and each section was examined under a light microscope (Nikon Eclipse CI, Tokyo, Japan).
Statistical Analyses
Data obtained from experimental animals were expressed as means and standard deviation of means (± SD). Analysis of the data was performed using one-way analysis of variance, followed by the Duncan test on the SPSS 20.0 and p < 0.05 was considered to be significant.
Results
Effect on Kidney Function Parameters
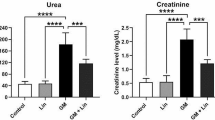
Gentamicin treatment increased BUN (Fig. 1a) and creatinine (Fig. 1b) levels of serum compared with control (p < 0.05). However, only B treatment did not affect these values compared with the control group (p > 0.05). Furthermore, B treatment in a dose-dependent manner decreased these alterations in gentamicin plus B groups compared with the gentamicin group (p < 0.05).
Effects of gentamicin (100 mg/kg) and gentamicin (100 mg/kg) plus boron (B) at doses of 5 (B5), 10 (B10), and 20 (B20) mg/kg on blood urea nitrogen (BUN; A), and creatinine (B) in serum of male rats. Values are mean ± standard deviations (n: 7). (a, b, c, d) In the same column, values with different letters show statistically significant differences
Effect on Lipid Peroxidation and Antioxidant Enzyme Activities
Gentamicin treatment increased MDA levels in whole blood (Table 3) and kidney tissue (Table 4) compared with control (p < 0.05), and B administration (especially at 10 and 20 mg/kg) decreased MDA levels compared with the control group (p < 0.05). Moreover, B plus gentamicin treatment decreased MDA levels compared with the gentamicin group (p < 0.05). On the other hand, gentamicin treatment decreased GSH levels in whole blood (Table 3) and kidney tissues (Table 4) compared with control (p < 0.05) whereas B plus gentamicin treatment enhanced GSH levels compared with the gentamicin group (p < 0.05). In addition, gentamicin treatment decreased SOD and CAT activities in whole blood (Table 3) and kidney tissue (Table 4) compared with control (p < 0.05); nonetheless, B plus gentamicin treatment ameliorated these enzyme activities compared with the gentamicin group (p < 0.05). Also, only B treatment did not affect SOD and CAT activities compared with control (p > 0.05). Gentamicin treatment increased NO levels of serum (Table 3) and kidney tissue (Table 4) compared with control (p < 0.05) yet, B plus gentamicin treatment decreased NO levels compared with gentamicin group (p < 0.05). Additionally, only B treatment in a dose-dependent manner increased NO levels of kidney compared with the control group (p < 0.05).
Effect on Proinflammatory Cytokine Levels
It was found that gentamicin treatment increased TNF-α, NFκB, IL-1β, IFN-γ, and 8-OHdG levels of serum compared with control (p < 0.05) which were presented in Table 5. However, B plus gentamicin treatment decreased their serum levels in a dose-dependent manner compared with the gentamicin group (p < 0.05). Additionally, gentamicin treatment increased TNF-α, NFκB, IL-1β, IFN-γ, and 8-OHdG levels of kidney tissue (Table 6) compared with control, and B plus gentamicin treatment in a dose-dependent manner reversed these alterations compared with the gentamicin group (p < 0.05). Also, fold changes in mRNA expression levels of TNF-α (Fig. 2a), NFκB (Fig. 2b), IL-1β (Fig. 2c), and IFN-γ (Fig. 2d) in the kidney of rats were found to be increased in the gentamicin group compared with control (p < 0.05). Moreover, B plus gentamicin treatment in a dose-dependent manner suppressed mRNA expression of these genes compared with the gentamicin group.
Effects of gentamicin (100 mg/kg) and gentamicin (100 mg/kg) plus boron (B) at doses of 5 (B5), 10 (B10), and 20 (B20) mg/kg on TNF-α (A), NFĸB (B), IL1-β (C), and IFN-γ (D) gene expressions by RT-PCR in kidney of male rats. Values are mean ± standard deviations (n: 7). (a, b, c, d, e, f) In the same column, values with different letters show statistically significant differences
Effect on Histopathological Changes
Gentamicin treatment evoked hemorrhage in interstitial areas, necrosis in tubular epithelial cells, eruption of epithelial cells, and hyaline cylinders in tubules lumen of kidneys. Also, there were not found to be any histopathological changes in the control and alone B treatment groups. The histopathological alterations of rat kidney were detailed in Table 7 and were also demonstrated in Fig. 3.
Histopathological appearance of male rat kidney (stained with H&E; dimensions 20 × 100 μm; n: 7). Red arrow, interstitial hemorrhage; arrow, spilled epithelial cells in tubulus lumen; arrowhead, hyaline cylinders in tubulus lumen. A1, control; A2, boron (B) at 5 mg/kg (B5); A3, B at 10 mg/kg (B10); A4, B at 20 mg/kg (B20); A5, gentamicin (100 mg/kg); A6, B5 + gentamicin (100 mg/kg); A7, B10+gentamicin (100 mg/kg); A8, B20+gentamicin (100 mg/kg) groups
Discussion
The important side effect of gentamicin is nephrotoxicity which is mainly determined by serum BUN and creatinine levels [3, 34, 35], whereas several substances such as lycopene [3], green tea extract [34], and cilostazol [35] reduced the nephrotoxicity. In this study, gentamicin treatment increased BUN and creatinine levels, whereas B administration decreased these values in a dose-dependent manner. This suggests that B has a protective role in the kidney tissue. A number of researchers have reported that gentamicin in a dose- and/or time-dependent manner increases MDA level and decreases GSH level and activities of SOD and CAT by inducing oxidative stress [3, 4, 36, 37]. Karahan et al. [3] treated rats with 100 mg/kg gentamicin i.p. for 6 days and increased MDA levels, whereas decreased glutathione peroxidase and CAT activities were determined in the kidney tissue. Similarly, Kuhad et al. [36] administered gentamicin (100 mg/kg, i.p.) for 8 days and observed increased MDA levels, reduced GSH levels, and SOD and CAT activities in rat kidney. Khan et al. [4] treated rats with gentamicin (80 mg/kg, i.p.) for 10 days and they found increased LPO and reduced CAT and SOD activities in rat kidney. In accordance with other studies, in this study, it was determined that gentamicin treatment increased MDA levels of the blood and kidney tissue. However, B treatment with gentamicin significantly decreased LPO compared with only gentamicin-treated rats. Also, only B treatment decreased MDA levels compared with control. It has been observed that B has hydroxyl radical, superoxide, and singlet oxygen molecule scavenger roles and reduces LPO [12, 13, 21]. Previous studies have presented that GSH plays a fundamental role in the detoxification of gentamicin and when GSH stores are exhausted cell death begins [22]. Ince et al. [13, 21] showed that B administration induced GSH activity in rats. In this study, an increase in LPO and depletion in GSH were observed in the gentamicin group. This may be a consequence of increased LPO and decreased GSH may arise from the depletion of GSH deposits. It was observed that B in a dose-dependent manner ameliorated the GSH levels in gentamicin-induced rats. This proposes that B reduced peroxidase activity between cells. It was reported that in animals, SOD and CAT activities are reduced by nephrotoxic agent such as gentamicin [3, 4, 36]. Also, the results of our study showed that gentamicin treatment decreased SOD and CAT activities of erythrocytes and tissues. Increased oxidative stress may deplete these enzymes. SOD and CAT activities of B plus gentamicin treatment groups were significantly higher compared with the gentamicin group. These findings suggested that B dose dependently increased the ability of these enzymes.
Nitric oxide has a notable role in the maintenance of local blood flow in the renal cortex of rats, which can also be affected by intraglomerular dynamics [38]. Moreira et al. [39] emphasized that gentamicin administration (100 mg/kg, i.p.) in rats increased serum NO level while vitamin C, as a protective agent, decreased this level. Parlakpinar et al. [40] investigated the protective effect of caffeic acid phenyl esterase (10 μmol/kg i.p) against gentamicin 100 mg/kg i.p-induced renal toxicity in rats and they reported increased NO production as well as increased LPO. Yaman and Balikci [41] evaluated the protective effect of Nigella sativa against gentamicin-induced renal damage and reported that gentamicin increased the NO level. In this study, it was determined that gentamicin increased NO levels, whereas B administration inhibited these increases. This suggested that B inhibits NO formation against oxidative damage. It was also found that only B administration (particularly at higher doses) increased kidney NO levels compared with control. This suggested that high dose of B could enhance synthesis of NO in endothelial cells of the kidney.
In this study, 100 mg/kg (i.p.) gentamicin administration caused to increased parameters of inflammation in the serum and kidney. Samarghandian et al. [42] reported that administration of 100 mg/kg gentamicin for 8 days increased serum levels of IL-6, IL-18, IL-1β, and TNF-α. However, administration of 10–30 mg/kg thymoquinone for 28 days decreased these values. Salem et al. [34] reported that subcutaneously injected gentamicin (50 mg/kg twice daily during 15 consecutive days) administration increased serum TNF-α and oxidative stress parameters (MDA, GSH, SOD, and CAT) in rats, but gentamicin plus green tea extract (oral, 300 mg/kg) significantly decreased these levels. Likewise, Mahmoud et al. [43] reported that serum TNF-α and oxidative stress parameters (MDA, GSH, SOD, and GPx) were increased in gentamicin-treated rats, while thymoquinone and curcumin (oral, 20 mg/kg) treatment decreased these levels. Fouad et al. [44] evaluated the protective effect of oral 50 mg/kg naringenin against gentamicin (80 mg/kg, i.p.) treatment. They found that renal oxidative stress parameters (MDA, NO, and GPx) and IL-8 levels are increased and naringenin reduced these parameters. In this study, it was observed that serum and renal inflammatory parameters (TNF-α, NFκB, IL-1β, and IFN-γ) levels were increased with gentamicin in rats but B application decreased the level of these parameters. This demonstrated that B has protective effects on cells against gentamicin-induced damage [24].
Chemical substances and drugs cause DNA damage due to alteration of DNA-binding protein and guanosine C-8 position [45, 46]. Arsenic, cadmium, and mercury have been reported to increase 8-OHDG levels in the kidney [47,48,49]. In this study, it was observed that gentamicin treatment caused increased levels of 8-OHdG in rat kidney and serum. This suggests that the main target of ROS formed by gentamicin is DNA. On the contrary, B dose dependently inhibited the formation of ROS levels. Furthermore, the administration of B in a dose-dependent manner increased the levels of cytokines and 8-OHDG in the kidney compared with the control group, which is due to its effect on kidney functions.
Gentamicin administration causes inflammation due to the formation of tubular necrosis in the kidney, which results in increased monocyte and macrophage migration in the tissues due to the kidney damage [46, 47]. Activation and translocation of NFκB which regulates gene expression of adhesion molecules, cytokines, and chemokines against oxidative stress take place during the inflammatory process [6]. Inhibiting the inflammatory mediators (TNF-α and IL) involved in its activation is considered to be important in renal nephrotoxicity [50, 51]. Subramanian et al. [52] treated rats with gentamicin (100 mg/kg, i.p.) and observed immunomodulation due to nephrotoxicity, abnormal cell growth, with increased gene expression of KIM-1, COX-2, osteopontin, Nrf2, NFκB, TNF-α, IL-6, and iNOS. Park et al. [7] and Quirós et al. [53] reported that gentamicin-induced renal damage is upregulated by inflammation cytokines (TNF-α, IL-1β, and IFN-γ). Additionally, it was reported that various antioxidant substances such as hesperidin [51], paricalcitol [7], and cardiotrophin-1 [53] inhibited gentamicin-induced inflammatory cytokines formation. In this study, expression of inflammatory cytokines (TNF-α, NFκB, IL-1β, and IFN-γ) increased in the kidney tissue which was similar to other studies. On the other hand, B treatment decreased the expression of inflammatory parameters in a dose-dependent manner due to inhibitory effect of B on ROS products [24]. Armstrong and Spears [54] reported that B supplementation to pig diet resulted in increased TNF-α and IFN-γ levels. In the study, it was observed that 20 mg/kg B administration caused an increase in the inflammatory cytokines (TNF-α, NFκB, IL-1β, and IFN-γ) expression levels. This indicates that kidney functions are affected by a high dose of boron.
Gentamicin administration has caused leukocyte infiltration, lumen spillage, tubular necrosis, and formation of hyaline cylinders in the kidney tubules of rat kidney [53]. Salem et al. [34] reported that gentamicin application resulted in degeneration, dense cellular infiltration, tubular dilation, and varying degrees of necrosis in the kidney of rats. Hegazy et al. [55] reported that gentamicin treatment caused vacuolar degeneration, tubular dilatation, interstitial hemorrhage, and cellular infiltration in the rat kidney. Stojiljkovic et al. [56] performed a study with rats and they assessed the effects of vitamin C (200 mg/kg) against gentamicin (100 mg/kg, 8 days). They reported that gentamicin treatment caused coagulation-type necrosis with black inclusions in kidney proximal tubular epithelial cells. Veljković et al. [52] evaluated the effect of green tea (300 mg/kg/day) against gentamicin (100 mg/kg, 8 days) and necrosis in proximal tubules of the kidney, dense mononuclear cell infiltration in interstitial area, and cytoplasmic gaps were observed. Sun et al. [57] evaluated the effects of sodium butyrate (50, 100, and 200 mg/kg) against gentamicin toxicity (100 mg/kg, 8 days). They observed that tubulointerstitial lesions, hyperplasia, and fibrosis. Yaman and Balikci [41] analyzed the protective effect of Nigella sativa (0.2 and 0.4 mL/kg) against 100 mg/kg gentamicin toxicity in rats and they reported that gentamicin caused necrosis in proximal tubules of the kidney, vacuolization, desquamation, and necrosis in epithelial cells, and mononuclear cell infiltrations also damaged integrity of glomerulus and membrane. These studies stated that all substances given against gentamicin toxicity reduced the kidney damage. In the present study, gentamicin treatment caused similar symptoms as hemorrhage in interstitial areas, necrosis in tubular epithelial cells, eruption of epithelial cells, and hyaline cylinders in tubules lumen of kidneys. However, B in a dose-dependent manner decreased these alterations due to its protective effects [21] against gentamicin treatment.
Consequently, it was detected that gentamicin caused loss of kidney functions and antioxidant enzyme activities whereas increases LPO level and expression of inflammation cytokines in this study. On the other hand, B reduced the gentamicin-induced formation of free radicals and inflammatory cytokines and strengthened the antioxidant system. Therefore, this study introduces a novel approach to B supplementation and suggests that B may carry potent use against gentamicin-induced nephrotoxicity. To achieve the abovementioned suggestions, other necessary studies and clinical trials are required.
References
Lode H, Kemmerich B, Koeppe P (2012) Comparative clinical pharmacology of gentamicin, sisomicin, and tobramycin. Antimicrob Agents Chemother 8:396–401
Martin J, Barras M, Ah Yui N, Kirkpatrick C, Kubler P, Norris R (2012) Gentamicin monitoring practices in teaching hospitals–time to undertake the necessary randomised controlled trial. J Clin Toxicol 2(8):1–5
Karahan I, Ateşşahin A, Yilmaz S, Ceribaşi AO, Sakin F (2005) Protective effect of lycopene on gentamicin-induced oxidative stress and nephrotoxicity in rats. Toxicology 215:198–204
Khan SA, Priyamvada S, Farooq N, Khan S, Khan MW, Yusufi AN (2009) Protective effect of green tea extract on gentamicin-induced nephrotoxicity and oxidative damage in rat kidney. Pharmacol Res 59:254–262
Lee IC, Kim SH, Lee SM, Baek HS, Moon C, Kim SH, Park SC, Kim HC, Kim JC (2012) Melatonin attenuates gentamicin-induced nephrotoxicity and oxidative stress in rats. Arch Toxicol 86:1527–1536
Randjelovic P, Veljkovic S, Stojiljkovic N, Jankovic-Velickovic L, Sokolovic D, Stoiljkovic M, Ilic I (2012) Salicylic acid attenuates gentamicin-induced nephrotoxicity in rats. Sci World J 2012:1–6
Park JW, Bae EH, Kim IJ, Ma SK, Choi C, Lee J, Kim SW (2010) Renoprotective effects of paricalcitol on gentamicin-induced kidney injury in rats. Am J Physiol Physiol 298:301–313
Bae EH, Kim IJ, Joo SY, Kim EY, Choi JS, Kim CS, Ma SK, Lee J, Kim SW (2014) Renoprotective effects of the direct renin inhibitor aliskiren on gentamicin-induced nephrotoxicity in rats. J Renin-Angiotensin-Aldosterone Syst 15:348–361
Nielsen FH (2018) Boron in aging and longevity. In: Trace elements and minerals in health and longevity. Springer, Cham, pp 163–177
Ince S, Erdogan M, Demirel HH, Agca Y, Dal G, Uguz C (2018) Boron enhances early embryonic gene expressions and improves fetal development of rats. J Trace Elem Med Biol 50:34–46
Kucukkurt I, Akbel E, Karabag F, Ince S (2015) The effects of dietary boron compounds in supplemented diet on hormonal activity and some biochemical parameters in rats. Toxicol Ind Health 31:255–260
Türkez H, Geyikoğlu F, Tatar A, Keleş S, Özkan A (2007) Effects of some boron compounds on peripheral human blood. Zeitschrift fur Naturforsch - Sect C J Biosci 62:889–896
Ince S, Kucukkurt I, Cigerci IH, Fidan AF, Eryavuz A (2010) The effects of dietary boric acid and borax supplementation on lipid peroxidation, antioxidant activity, and DNA damage in rats. J Trace Elem Med Biol 24:161–164
Çelikezen FÇ, Toğar B, Özgeriş FB, Izgi MS, Türkez H (2016) Cytogenetic and oxidative alterations after exposure of cultured human whole blood cells to lithium metaborate dehydrate. Cytotechnology 68(4):821–827
Coban FK, Ince S, Kucukkurt I, Demirel HH, Hazman O (2015) Boron attenuates malathion-induced oxidative stress and acetylcholinesterase inhibition in rats. Drug Chem Toxicol 38:391–399
Acaroz U, Ince S, Arslan-Acaroz D, Gurler Z, Demirel HH, Kucukkurt I, Eryavuz A, Kara R, Varol N, Zhu K (2019) Bisphenol-A induced oxidative stress, inflammatory gene expression, and metabolic and histopathological changes in male Wistar albino rats: Protective role of boron. Toxicol Res 8:262–269
Ince S, Kucukkurt I, Acaroz U, Arslan-Acaroz D, Varol N (2019) Boron ameliorates arsenic-induced DNA damage, proinflammatory cytokine gene expressions, oxidant/antioxidant status, and biochemical parameters in rats. J Biochem Mol Toxicol 33:1–7
Yildirim S, Celikezen FC, Oto G, Sengul E, Bulduk M, Tasdemir M, Cinar DA (2018) An investigation of protective effects of litium borate on blood and histopathological parameters in acute cadmium-induced rats. Biol Trace Elem Res 182(2):287–294
Acaroz U, Ince S, Arslan-Acaroz D, Gurler Z, Kucukkurt I, Demirel HH, Arslan HO, Varol N, Zhu K (2018) The ameliorative effects of boron against acrylamide-induced oxidative stress, inflammatory response, and metabolic changes in rats. Food Chem Toxicol 118:745–752
Hunt CD (1996) In: Watson RR (ed) Dietary boron deficiency and supplementation. CRC Press, Inc, New York, pp 229–254
Ince S, Kucukkurt I, Demirel HH, Acaroz DA, Akbel E, Cigerci IH (2014) Protective effects of boron on cyclophosphamide induced lipid peroxidation and genotoxicity in rats. Chemosphere 108:197–204
Ateşşahin A, Karahan I, Yilmaz S, Çeribaşi AO, Princci I (2003) The effect of manganese chloride on gentamicin-induced nephrotoxicity in rats. Pharmacol Res 48:637–642
Winterbourn CC, Hawkins RE, Brain M, Carrell RW (1975) The estimation of red cell superoxide activity. J Lab Clin Med 55:337–341
Ohkawa H, Ohishi N, Yagi K (1979) Assay for lipid peroxides in animal tissues by thiobarbituric acid reaction. Anal Biochem 95:351–358
Draper HH, Hardley M (1990) Malondialdehyde determination as index of lipid peroxidation. Methods Enzymol 186:421–431
Beutler E, Duron O, Kelly BM (1963) Improved method for the determination of blood glutathione. J Lab Clin Med 61:882–888
Sun Y, Oberley LW, Li Y (1988) A simple method for clinical assay of superoxidase dismutase. Clin Chem 34:497–500
Luck H (1955) Methods in analysis. In: Methods in analysis. Academy Press, London
Aebi H (1974) Catalase. In: Methods of enzymatic analysis, pp 673–677
Miranda KM, Espey MG, Wink DA (2001) A rapid, simple spectrophotometric method for simultaneous detection of nitrate and nitrite. Nitric Oxide Biol Chem 5:62–71
Lowry O, Rosebrough N, Farr A, Randall R (1951) Protein measurement with the Folin phenol reagent. J Biol Chem 193:265–275
Drabkin DL, Austin JH (1935) Spectrophotometric studies. II. Preparations from washed 112, blood cells; nitric oxide hemoglobin and sulfhemoglobin. J Biol Chem:51–65
Pfaffl MW (2001) A new mathematical model for relative quantification in real-time RT-PCR. Nucleic Acids Res 29:e45
Salem EA, Salem NA, Kamel M, Maarouf AM, Bissada NK, Hellstrom WJ, El-Adl M (2010) Amelioration of gentamicin nephrotoxicity by green tea extract in uninephrectomized rats as a model of progressive renal failure. Ren Fail 32:1210–1215
Abdelsameea AA, Mohamed AM, Amer MG, Attia SM (2016) Cilostazol attenuates gentamicin-induced nephrotoxicity in rats. Exp Toxicol Pathol 68:247–253
Kuhad A, Tirkey N, Pilkhwal S, Chopra K (2006) Effect of Spirulina, a blue green algae, on gentamicin-induced oxidative stres and renal dysfunction in rats. Fundam Clin Pharmacol 20:121–128
Dungca NT (2016) Protective effect of the methanolic leaf extract of Eclipta alba (L.) Hassk. (Asteraceae) against gentamicin-induced nephrotoxicity in Sprague Dawley rats. J Ethnopharmacol 184:18–21
Kasztan M, Jankowski M (2016) Involvement of P2 receptors in regulation of glomerular permeability to albumin by extracellular nucleotides of intra-/extra-glomerular origins. J Physiol Pharmacol 67:177–183
Moreira MA, Nascimento MA, Bozzo TA, Cintra A, da Silva SM, Dalboni MA, Mouro MG, Higa EM (2014) Ascorbic acid reduces gentamicin-induced nephrotoxicity in rats through the control of reactive oxygen species. Clin Nutr 33:296–301
Parlakpinar H, Tasdemir S, Polat A, Bay-Karabulut A, Vardi N, Ucar M, Acet A (2005) Protective role of caffeic acid phenethyl ester (cape) on gentamicin-induced acute renal toxicity in rats. Toxicology 207:169–177
Yaman I, Balikci E (2010) Protective effects of nigella sativa against gentamicin-induced nephrotoxicity in rats. Exp Toxicol Pathol 62:183–190
Samarghandian S, Azimi-Nezhad M, Mehrad-Majd H, Mirhafez SR (2015) Thymoquinone ameliorates acute renal failure in gentamicin-treated adult male rats. Pharmacology 96:112–117
Mahmoud AM, Ahmed OM, Galaly SR (2014) Thymoquinone and curcumin attenuate gentamicininduced renal oxidative stress, inflammation and apoptosis in rats. EXCLI J 13:98–110
Fouad AA, Albualib WH, Zahranc A, Gomaa W (2014) Protective effect of naringenin against gentamicin-induced nephrotoxicity in rats. Environ Toxicol Pharmacol 38:420–429
Yoshioka N, Nakashima H, Hosoda K, Eitaki Y, Shimada N, Omae K (2008) Urinary excretion of an oxidative stress marker, 8-hydroxyguanine (8-OH-GUA), among nickel-cadmium battery workers. J Occup Health 50:229–235
Gałazyn-Sidorczuk M, Brzóska MM, Jurczuk M, Moniuszko-Jakoniuk J (2009) Oxidative damage to proteins and DNA in rats exposed to cadmium and/or ethanol. Chem Biol Interact 180:31–38
Iqbal M, Okazaki Y, Okada S (2009) Curcumin attenuates oxidative damage in animals treated with a renal carcinogen, ferric nitrilotriacetate (Fe-NTA): Implications for cancer prevention. Mol Cell Biochem 324:157–164
Li Z, Piao F, Liu S, Wang Y, Qu S (2010) Subchronic exposure to arsenic trioxide-induced oxidative DNA damage in kidney tissue of mice. Exp Toxicol Pathol 62:543–547
Balakumar P, Rohilla A, Thangathirupathi A (2010) Gentamicin-induced nephrotoxicity: do we have a promising therapeutic approach to blunt it? Pharmacol Res 62:179–186
Sahu BD, Tatireddy S, Koneru M, Borkar RM, Kumar JM, Kuncha M, Srinivas R, Shyam-Sunder R, Sistla R (2014) Naringin ameliorates gentamicin-induced nephrotoxicity and associated mitochondrial dysfunction, apoptosis and inflammation in rats: possible mechanism of nephroprotection. Toxicol Appl Pharmacol 277:8–20
Subramanian P, Anandan R, Jayapalan JJ, Hashim OH (2015) Hesperidin protects gentamicin-induced nephrotoxicity via Nrf2/HO-1 signaling and inhibits inflammation mediated by NF-κB in rats. J Funct Foods 13:89–99
Veljković M, Pavlović DR, Stojiljković N, Ilić S, Petrović A, Jovanović I, Radenković M (2016) Morphological and morphometric study of protective effect of green tea in gentamicin-induced nephrotoxicity in rats. Life Sci 147:85–91
Quirós Y, Blanco-Gozaloa V, Sanchez-Gallegoa JI, López-Hernandez FJ, Ruize J, Perez de Obanose MP, López-Novoab JM (2016) Cardiotrophin-1 therapy prevents gentamicin-induced nephrotoxicityin rats. Pharmacol Res 107:137–146
Armstrong TA, Spears JW (2003) Effect of boron supplementation of pig diets on the production of tumor necrosis factor-α and interferon-γ. J Anim Sci 81:2552–2561
Hegazy AMS, Mosaed MM, Elshafey SH, Bayomy NA (2016) 6-Gingerol ameliorates gentamicin induced renal cortex oxidative stress and apoptosis in adult male albino rats. Tissue Cell 48:208–216
Stojiljkovic N, Stoiljkovic M, Randjelovic P, Veljkovic S, Mihailovic D (2012) Cytoprotective effect of vitamin C against gentamicin-induced acute kidney injury in rats. Exp Toxicol Pathol 64:69–74
Sun X, Zhang B, Hong X, Zhang X, Kong X (2013) Histone deacetylase inhibitor, sodium butyrate, attenuates gentamicin-induced nephrotoxicity by increasing prohibitin protein expression in rats. Eur J Pharmacol 707:147–154
Acknowledgment
Also, this study orally presented at 3rd International Turkic World Conference on Chemical Sciences and Technologies (ITWCCST 2017).
Funding
This study was financially supported by a grant (Project Number: 214O661) from the Scientific and Technological Research Council of Turkey.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Ince, S., Kucukkurt, I., Demirel, H.H. et al. Boron, a Trace Mineral, Alleviates Gentamicin-Induced Nephrotoxicity in Rats. Biol Trace Elem Res 195, 515–524 (2020). https://doi.org/10.1007/s12011-019-01875-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12011-019-01875-4