Abstract
Oil sands tailings, a slurry of alkaline water, silt, clay, unrecovered bitumen, and residual hydrocarbons generated during bitumen extraction, are contained in ponds. Indigenous microbes metabolize hydrocarbons and emit greenhouse gases from the tailings. Metabolism of hydrocarbons in tailings ponds of two operators, namely, Canadian Natural Upgrading Limited (CNUL) and Canadian Natural Resources Limited (CNRL), has not been comprehensively investigated. Previous reports have revealed sequential and preferential hydrocarbon degradation of alkanes in primary cultures established from CNUL and CNRL tailings amended separately with mixtures of hydrocarbons (n-alkanes, iso-alkanes, paraffinic solvent, or naphtha). In this study, activation pathway of hydrocarbon biodegradation in these primary cultures was investigated. The functional gene analysis revealed that fumarate addition was potentially the primary activation pathway of alkanes in all cultures. However, the metabolite analysis only detected transient succinylated 2-methylpentane and 2-methylbutane metabolites during initial methanogenic biodegradation of iso-alkanes and paraffinic solvent in all CNUL and CNRL cultures amended with iso-alkanes and paraffinic solvent. Under sulfidogenic conditions (prepared only with CNUL tailings amended with iso-alkanes), succinylated 2-methylpentane persisted throughout incubation period of ~ 1100 days, implying dead-end nature of the metabolite. Though no metabolite was detected in n-alkanes- and naphtha-amended cultures during incubation, assA/masD genes related to Peptococcaceae were amplified in all CNUL and CNRL primary cultures. The findings of this present study suggest that microbial communities in different tailings ponds can biodegrade hydrocarbons through fumarate addition as activation pathway under methanogenic and sulfidogenic conditions.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Alkanes constitute major proportion of crude oil and its derived products, which can impact many environments, naturally or anthropogenically. Alkanes are inert in nature and have high C-H bond dissociation energy [1]. Under aerobic condition, aerobic microorganisms utilize highly reactive oxygen species to overcome the high C-H bond dissociation energies of alkanes via mono and/or dioxygenase reactions [2]. However, under anaerobic conditions, anaerobic microorganisms must activate alkanes via alternative mechanisms. For the last two decades, fumarate addition has been implicated as the main hydrocarbon activation pathway in many anaerobic cultures amended with (1) n-alkanes [3,4,5,6,7], (2) iso-alkanes [8, 9], (3) monoaromatics [10,11,12,13], and (4) polyaromatics [13,14,15,16]. Other anaerobic hydrocarbon activation pathways including carboxylation and hydroxylation [17] have also been implicated and highly debated but not widely reported.
Oil sands tailings ponds are engineered environments that are impacted by fugitive hydrocarbons containing a large proportion of alkanes (n-alkanes, iso-alkanes, and cycloalkanes). Extraction solvents/diluents used in the process of bitumen extraction from oil sands ores are recovered but their residual concentrations in generated waste tailings escape to oil sands tailings ponds. Indigenous methanogenic microbial communities in oil sands tailings [18] metabolize a suite of alkanes (n-and iso-alkanes) and some monoaromatics [19,20,21] contributing to methane (CH4) emissions from tailings ponds [22]. Most previous studies investigated microbial degradation of hydrocarbons in the primary cultures established using fluid fine tailings (FFT) collected from Syncrude Canada Ltd. Mildred Lake Settling Basin (MLSB). These primary cultures metabolized a wide range of alkanes to CH4. The metabolite and functional gene analyses performed on these primary cultures revealed possibility of fumarate-addition as the primary activation pathway of n- and iso-alkanes biodegradation in MLSB [8, 9].
The biodegradation pathways of alkanes in tailings ponds operated by Canadian Natural Upgrading Limited (CNUL) and Canadian Natural Resources Ltd. (CNRL) remain relatively unexplored. Previous studies [23,24,25,26,27] have focused on the biodegradation of specific hydrocarbon diluents used in bitumen extraction processes at these sites. While these studies characterized the microbial communities involved in degradation, the exact metabolic pathways for hydrocarbon activation are still unknown.
CNUL uses primarily paraffinic solvents (C5–C6; n- and iso-alkanes), whereas CNRL employs a more complex diluent such as naphtha (n-, iso-, and cycloalkanes, and alkylbenzenes primarily in the C6–C10 range). Despite identifying key microbial players, understanding the specific pathways by which these hydrocarbons are activated remains as a knowledge gap.
To address this gap, the present study is aimed to conduct functional gene and metabolite analyses on the previously studied primary cultures from CNUL and CNRL. By analyzing these cultures’ metabolites and functional genes, the primary activation pathways employed by the indigenous microbial communities in these tailings ponds will be elucidated. This research will significantly contribute to existing understanding of alkane activation mechanisms and anaerobic hydrocarbon biodegradation processes in distinct tailings pond environments.
Materials and Methods
Description of Primary Cultures Established
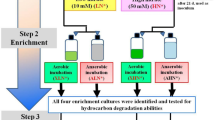
The primary cultures (in triplicate) were established by incubating 50 mL of FFT collected from CNUL’s Muskeg River Mine tailings pond or CNRL’s Horizon tailings pond FFT with 50 mL of methanogenic medium or sulfate-reducing medium in a 158-mL serum bottle. Heat-killed (autoclaved at 121 °C and 20 psi for 4 consecutive days) and unamended microcosms were also prepared in duplicates to consider abiotic degradation and the production of CH4 resulting from residual indigenous substrates in the FFTs, respectively. All primary cultures analyzed here has been described in detail in previous studies (Table 1). All cultures were incubated at room temperature (~ 20 °C) statically in the dark until CH4 production or sulfate-reduction plateaued. The CH4 concentration was measured by analyzing the headspace of all microcosms using gas chromatography equipped with flame ionization detector (GCFID) whereas sulfate concentration in all sulfate-reducing microcosms was analyzed using ion chromatography following method as described in a recent report [27]. The spiked hydrocarbon concentrations in all cultures were examined using purge-trap gas chromatography with flame ionization detection (GCFID), following the methodology outlined in Siddique et al. [20]. Additional PONAU (paraffins, olefins, naphthenes, aromatics, and unknown components) analysis was performed on microcosms amended with paraffinic solvent and naphtha as detailed in the previous reports [23, 24]. These primary cultures are discussed briefly in Supplementary Information.
Determination of Metabolites During Hydrocarbon Biodegradation
The cultures from all studies were sampled for metabolite analysis at two time points: first sampling was performed during the exponential production of CH4 or reduction of sulfate, and second sampling was done after CH4 production or sulfate reduction plateaued [28]. Two to four milliliters of cultures were drawn from all replicates of each treatment and pooled in 20-mL EPA vials (Fisher Scientific). One microgram of 4-fluoro-1-naphthoic acid (4F1N; CAS#573–03-5; Alfa Aesar) was then added to all samples as a surrogate standard [29]. Using concentrated hydrochloric acid (CAS#7647–01-0; Sigma-Aldrich), all samples were acidified to pH < 2 [30]. The acidified samples were extracted three times with 15 mL of ethyl acetate (CAS#141–78-6; Fisher Scientific) and dried overnight in a fume hood. The dried samples were dissolved in 1.5 mL ethyl acetate and transferred into 2-mL GC vials. The solution was concentrated to ~ 100 µL under N2 flow and derivatized with 100 µL of N,O-bis(trimethylsilyl)trifluoroacetamide (BSTFA; CAS#25561–30-2; Thermo Scientific) at 70 °C for ~ 90 min. The derivatized samples were diluted to 500 µL with ethyl acetate before analysis by gas chromatography-mass spectrometry (GC–MS). One microliter of derivatized sample was analyzed by TRACE 1300 gas chromatograph equipped with a TraceGold TG-5MS GC column (30 m by 0.25 mm internal diameter; Thermo Scientific) in splitless mode. The initial temperature of the oven was held at 65 °C for 5 min, increased at 5 °C min−1 to 280 °C and then held at 280 °C for 15 min. Mass spectra of metabolites were obtained using ISQ LT Single Quadrupole mass spectrometer (Thermo Scientific), and the data was acquired in the scan mode from 50 to 600 mass units.
Nucleic Acid Extraction and Functional Gene Amplification
Triplicate 300 μL volumes of primary culture were collected from each of three replicate primary cultures described above at the end of incubation for functional genes (assA/masD) analysis. The total DNA was extracted from the cultures using the protocol described in detail in previous report [25]. The recovered DNA from each replicate of a single treatment was pooled before PCR amplification. The assA/masD gene was amplified in triplicate from pooled DNA using primers 1432F (5′-CCNACCACNAAGCAYGG-3′) and 1936R (5′-TCRTCATTNCCCCAYTTNGG-3′) [28, 31]. The temperature program for amplification in a S1000™ Thermal Cycler (BIO RAD) was as follows: 95 °C for 3 min followed by 40 cycles of 96 °C for 45 s, 52 °C for 30 s, 72 °C for 45 s, and a final extension step at 72 °C for 10 min. The PCR reaction (25 μL) contained 12.5 μL AccuStart II PCR ToughMix (Quanta Biosciences, Gaithersburg, MD), 1 μL extracted DNA (~ 20 ng/μL), 2.5 μL (10 μM) of each primer, and 6.5 μL of sterile nuclease-free water. Negative controls containing only PCR reagents and nuclease-free water were also included with every set of samples to maintain quality control. The triplicate amplifications from single treatment were pooled, and the amplicons’ size was confirmed via 1% agarose gel electrophoresis. All amplicons with the right size were excised and purified using Qiagen’s QIAquick Gel Extraction kit according to the manufacturer’s procedure.
Construction of assA/masD Gene Clone Libraries and Phylogenetic Analysis
The gel purified amplicons were quantified on NanoDrop-1000 spectrophotometer v3.3 before used for cloning with Invitrogen’s TA Cloning Kit (pCR™2.1-TOPO® vector and DH5α-T1R E. coli competent cells) according to manufacturer’s procedure. All clones were first checked for inserts via PCR amplification with insert-specific primers and resolved by gel electrophoresis. The amplicons were sequenced using T7 primers and BigDye Terminator v3.1 cycle sequencing kit (Applied Biosystem) on an ABI 3730 sequencer (Applied Biosystem, Foster City, CA) in Molecular Biology Services Unit (MBSU) at the University of Alberta. Initial analysis showed that the assA/masD genes in the samples had minimal diversity; therefore, we performed restriction analysis on all amplicons by MspI digestion for restriction fragment pattern similarities to reduce the number of clones for sequencing [32]. The sequences from assA/masD clone libraries were trimmed to remove vector sequences. The trimmed sequences were then compared against GenBank Database using BLASTX algorithm to determine closest assA/masD-derived amino acid sequences. The assA/masD gene sequences were aligned with reference sequences from GenBank using MUSCLE [33] and manually edited. The sequences were then clustered into OTUs and translated using Geneious R8 (Biomatters Ltd, New Zealand). Maximum likelihood tree with WAG model and 100 bootstrap replicates was constructed using PhyML [34]. All cloned sequences were deposited in GenBank under the accession numbers KU840850–KU840905.
Results
Detection of Succinylated Metabolites in Primary Cultures
During incubation, different primary cultures started biodegrading the amended hydrocarbons at different times (Supplementary Figs. A–F). However, two sampling time points were selected for all cultures to determine metabolites: first, during the active biodegradation of the amended hydrocarbons and, second, at the end of incubation to determine if the metabolites detected (if any) persisted after prolonged incubation (sampling times from all cultures are indicated in Table 2). In all methanogenic CNUL and CNRL primary cultures amended with Naph, 2-Alkanes, and 4-Alkanes, no metabolite was detected at both sampling points. However, in methanogenic samples collected from CNUL Par, CNRL Par, CNRL M-3I, and CNUL M-3I, succinylated metabolites were detected at the first sampling point but the same metabolites were not detected at second sampling point. The succinylated metabolites, however, were detected in both sampling points in samples collected from sulfidogenic CNUL S-3I (Table 2). In all samples where succinylated metabolites were detected, a cluster of two GC–MS peaks at 25.9 and 26.1 min were detected with mass spectra profiles identical to mass spectra expected from (2-methylpentyl)succinic acid (Fig. 1A, B). The mass spectra of putative double-derivatized trimethylsilyl (di-TMS) ester of (2-methylpentyl)succinic acid comprised an (M-15)+ ion with m/z 331 and other key ion fragments (m/z 73, 147, 174, 217, and 262) (Fig. 1B), which were similar to previously reported spectra of derivatized fumarate-added 2-methylpentane (or 1,3-dimethylbutylsuccinic acid) [9].
Postulated TMS-derivatized metabolites extracted from CNUL M-3I detected using GC–MS. A Retention time of the postulated TMS-derivatized C5 and C6 iso-alkylsuccinate. Surrogate standard: 4-fluoro-1-naphthoic acid added in all samples prior to metabolite extraction procedure. B Mass spectrum of putative TMS-derivatized 2-methylpentylsuccinic acid. C Mass spectrum of putative TMS-derivatized 2-methylbutylsuccinic acid. Inferred putative structures and diagnostic (M-15)+ ion fragments of m/z 317 and 331 are shown in the insets. Chromatograms (A, B) were used as representative chromatograms for metabolites detected in CNUL S-3I, CNRL M-3I, CNUL Par, and CNRL Par
Additionally, we also detected another cluster of two peaks at 23.9 and 24.1 min exclusively at the first sampling point from CNUL M-3I (Fig. 1A, C). The mass spectral profiles of both peaks were similar, and the profiles were consistent with derivatized fumarate-added 2-methylbutane with diagnostic (M-15)+ ion fragments of m/z 317 and other key fumarate-added ion fragments (m/z 73, 147, 172, 217, and 262) (Fig. 1A, C). The key ion fragments were the same as the ion fragments associated with fumarate addition metabolites reported in previous studies [8, 35,36,37,38]. These peaks were not detected in the second sampling point from CNUL M-3I.
assA/masD Gene Detection During Anaerobic Biodegradation of Alkanes
Because alkylsuccinate metabolite was detected in some of the primary cultures and the fact that the amendments used in all the treatments were completely or mostly consisted of alkanes, functional genes encoding catalytic subunits of alkylsuccinate synthase (assA), also known asmethylalkylsuccinate synthase (masD), were chosen to be investigated. The PCR was performed to amplify partial assA/masD genes in all the primary cultures to support the detection of postulated succinylated metabolites and to ascertain if fumarate addition was the potential activation pathway in cultures where activated metabolites were not detected.
assA/masD-like genes that are considered as key functional genes involved in activation of alkanes via fumarate addition pathway were found in the total DNA extracted from all the amended primary cultures. However, no amplification of assA/masD-like genes was observed from unamended cultures, which might indicate low abundance of key bacteria carrying the functional genes in alkane-deficient cultures. The restriction analysis revealed only one gene fragmentation pattern in all amended primary cultures except CNUL and CNRL Naph, which exhibited two and three unique fragmentation patterns, respectively (reflected in the number of OTUs present in each treatment; Fig. 2), indicating low diversity of assA/masD-like genes in the cultures. This observation coincided with observations from previous studies [8, 9] which also reported low diversity of assA/masD-like genes in FFT-derived enrichment cultures incubated with various n- and iso-alkanes amendments.
Maximum likelihood tree showing the affiliation of translated sequences coding for the alpha subunit of alkylsuccinate synthase (assA) to selected reference sequences. AssA-like sequences from current study (bold) was aligned with closely related AssA, BssA, and NmsA reference sequences recovered from NCBI nr-database through BLASTX searches. Maximum likelihood tree was constructed using PhyML with WAG model and 100 bootstrap replicates. Pyruvate formate lyase sequence from Desulfobacula sp. TS was used as the outgroup. The GenBank accession numbers are indicated in parentheses
Clone libraries generated from all CNUL and CNRL amended primary cultures revealed that all the cloned sequences were related to assA/masD genes (Fig. 2). Subsequent analysis performed at the protein level of the assA/masD genes to construct maximum-likelihood tree revealed that the translated assA/masD fragments were closely related to a putative methylalkylsuccinate subunit (MasD) from an uncultured bacterium recovered from gas seepage-impacted and pristine cold marine sediments and putative AssA from an uncultured Peptococcaceae recovered from a methanogenic short-chain alkane-degrading enrichment culture (SCADC) [9, 39] [9]. All AssA/MasD sequences had high similarity at protein level (≥ 98% identity) to each other and to AssA related to Peptococcaceae SCADC except CNUL-Naph OTU 1, CNUL M-3I, and CNRL-Naph OTU 3 which had ~ 94–95% identity to AssA from Peptococcaceae SCADC (Fig. 2).
Discussion
In recent years, there has been increasing evidence indicating that hydrocarbons are primarily activated via fumarate activation pathway under anaerobic conditions. Fumarate addition has been demonstrated under various reducing conditions, and the corresponding succinylated metabolites have been detected in various hydrocarbon-impacted environments and hydrocarbon-degrading enrichment and pure cultures [17, 40,41,42,43,44]. Fumarate addition has also been demonstrated in MLSB FFT-derived enrichment cultures grown on various alkanes [8, 9]. However, no similar study on hydrocarbon activation pathway has been reported in FFTs from relatively “younger” oil sands tailings ponds managed by CNUL and CNRL, which employed different oil sands extraction process and treated their tailings differently for solid consolidation enhancement before being deposited into the tailings ponds. Initial studies on biodegradation of different hydrocarbon mixtures in CNUL and CNRL FFT [23,24,25] revealed the enrichment of different microbial taxa during the biodegradation process and distinct biodegradation pattern in CNUL and CNRL FFT-associated microbial communities. These differences beg the question whether hydrocarbons in CNUL and CNRL tailings ponds were also activated differently in the biodegradation process.
To identify the metabolites resulting from activation of the amended alkanes and to discover if the detected metabolites persist in the cultures during extended incubation, culture samples retrieved from two sampling points (sampling time indicated in Table 2) from all primary cultures were analyzed. Succinylated metabolites were only detected in CNUL M-3I, CNUL S-3I, CNRL M-3I, CNUL Par, and CNRL Par (Table 2) but not in the other primary cultures. The putative metabolites were only detected during active biodegradation of iso-alkanes; however, the metabolites appeared to be transient because the metabolites were not detected at second sampling point excepting CNUL S-3I. Similarly, in previous reports which examined methanogenic biodegradation of alkanes, succinylated metabolites were only transiently detected in cultures grown on iso-alkanes [8, 9] but not in cultures amended with n-alkanes [9, 45]. This phenomenon could be attributed to the recalcitrance nature of iso-alkanes over n-alkanes, which have been reported previously [23, 24], where iso-alkanes were shown to have lower conversion rate to CH4 than n-alkanes. Thus, although under methanogenic condition the succinylated iso-alkanes were only transient products, the succinylated iso-alkanes were not further oxidized as rapidly as succinylated n-alkanes, allowing the metabolic intermediates to transiently accumulate and, thus, be detected during the exponential iso-alkanes degradation phase. The reason for the unsuccessful detection of any metabolites from methanogenic cultures amended with n-alkanes, therefore, may be explained by (1) non-accumulating metabolites resulting from the close link of alkane activation with metabolites turnover and (2) instrumentation limitation in detecting low yield metabolites.
Nonetheless, iso-alkanes also constitute a major portion of naphtha (~ 39 wt% [24]. However, naphtha comprised a wide range of iso-alkanes (C6-C10) as opposed to paraffinic solvent, which only has three major iso-alkane components constituting ~ 58 wt% of paraffinic solvent’s total mass [23]. Therefore, the concentration of individual iso-alkanes in naphtha is low compared to the concentration of individual iso-alkanes in paraffinic solvent; hence, the concentration of any metabolites that might arise from the oxidation of the iso-alkanes in naphtha amended cultures may be too low to be detected by GC–MS. Interestingly under sulfate-reducing conditions, the succinylated iso-alkane, 1,3-dimethylbutylsuccinic acid persisted throughout ~ 1100 days incubation even though sulfate concentration has plateaued since day ~ 900 (Supplementary Fig. F). Accumulation of 1,3-dimethylbutylsuccinic acid in CNUL S-3I might be attributed to the absence or low growth of microbes carrying the enzymes appropriate for oxidation of 1,3-dimethylbutylsuccinic acid. The accumulation of 1,3-dimethylbutylsuccinic acid might have also resulted in inhibition of further degradation of 2-methylpentane in CNUL S-3I despite the fact that high concentration of sulfate was still present in the culture (Supplementary Fig. F). The derivatized metabolites from 2-methylbutane and 2-methylpentane exhibited a cluster of two GC–MS peaks. These peaks might have represented diastereomers formed by enzymatic mechanisms as described by [46], which have also been reported in previous studies examining anaerobic biodegradation of iso-alkanes [8, 9, 47].
Activation of alkanes via fumarate addition pathway is carried out by glycyl radical alkylsuccinate or methylalkylsuccinate synthase (Ass or Mas, respectively). To determine the presence of genes encoding Ass/Mas and diversity of the genes in all the cultures, amplification of assA/masD genes, which encode for the catalytic subunit of Ass/Mas, was performed using degenerate assA/masD primers. assA/masD genes were amplified from all CNUL and CNRL amended primary cultures, indicating that microbial communities in all the primary cultures have the potential for activating alkanes via fumarate addition pathway despite that the expected alkysuccinate metabolites were not detected in all the cultures, possibly due to quick further transformation. The diversity of the translated assA/masD genes in all the cultures, however, is low and has high similarity to Peptococcaceae SCADC (Fig. 2). Incidentally, all the primary cultures exhibited enrichment of Peptococcaceae (~ 36–77% sequence reads) during biodegradation of the amended alkanes [23,24,25], and similar enrichment of Peptococcaceae has also been reported in n- and iso-alkane-degrading cultures from FFT [8, 9, 21, 25,26,27, 39, 48], providing further evidence for implication of Peptococcaceae as important degraders of alkanes in oil sands tailings ponds.
Conclusion
In summary, this study significantly advances the overall understanding of hydrocarbon activation pathways under methanogenic and sulfidogenic conditions, particularly highlighting the pivotal role played by the fumarate addition pathway. Specifically, this study underscores the complexities involved in studying hydrocarbon metabolism due to the transient nature of metabolites during biodegradation process. Moreover, the findings underscore the significance of Peptococcaceae in the degradation of hydrocarbons within oil sands tailings ponds. The metabolite and functional gene profiling data, combined with results from previous studies [8, 9, 49], strongly indicate the prevalence of the fumarate addition pathway across various oil sands tailings ponds, despite differences in extraction techniques and tailings management practices among operators. Nevertheless, the potential existence of alternative metabolic routes, such as carboxylation and/or hydroxylation for hydrocarbon activation within these environments, is acknowledged.
Future investigations should delve deeper into elucidating the governing mechanisms behind these pathways and explore the factors influencing the persistence of specific metabolites. Such endeavors will undoubtedly propel our understanding of anaerobic hydrocarbon biodegradation in intricate environments to new frontiers, further enriching our knowledge base and potentially informing improved environmental management strategies, not only for remediation oil sands tailings ponds but also other hydrocarbon-impacted environments.
Data Availability
All data generated or analyzed during this study are included in this published article and its supplementary information files.
References
Blanksby, S. J., & Ellison, G. B. (2003). Bond dissociation energies of organic molecules. Accounts of chemical research, 36(4), 255–263. https://doi.org/10.1021/ar020230d
Boll, M., & Heider, J. (2010). Anaerobic degradation of hydrocarbons: Mechanisms of C-H-bond activation in the absence of oxygen. In Handbook of Hydrocarbon and Lipid Microbiology (Vol. 1, pp. 1011–1024). Springer. https://doi.org/10.1007/978-3-540-77587-4
Callaghan, A. V., Wawrik, B., Ní Chadhain, S. M., Young, L. Y., & Zylstra, G. J. (2008). Anaerobic alkane-degrading strain AK-01 contains two alkylsuccinate synthase genes. Biochemical and biophysical research communications, 366(1), 142–148. https://doi.org/10.1016/j.bbrc.2007.11.094
Zedelius, J., Rabus, R., Grundmann, O., Werner, I., Brodkorb, D., Schreiber, F., … Widdel, F. (2011). Alkane degradation under anoxic conditions by a nitrate-reducing bacterium with possible involvement of the electron acceptor in substrate activation. Environmental Microbiology Reports, 3(1), 125–135. https://doi.org/10.1111/j.1758-2229.2010.00198.x
Khelifi, N., Amin Ali, O., Roche, P., Grossi, V., Brochier-Armanet, C., Valette, O., … Hirschler-Réa, A. (2014). Anaerobic oxidation of long-chain n-alkanes by the hyperthermophilic sulfate-reducing archaeon, Archaeoglobus fulgidus. The ISME Journal, 8(11), 2153–2166. https://doi.org/10.1038/ismej.2014.58
Zhou, L., Li, K.-P., Mbadinga, S. M., Yang, S.-Z., Gu, J.-D., & Mu, B.-Z. (2012). Analyses of n-alkanes degrading community dynamics of a high-temperature methanogenic consortium enriched from production water of a petroleum reservoir by a combination of molecular techniques. Ecotoxicology, 21(6), 1680–1691. https://doi.org/10.1007/s10646-012-0949-5
Bian, X.-Y., Maurice Mbadinga, S., Liu, Y.-F., Yang, S.-Z., Liu, J.-F., Ye, R.-Q., … Mu, B.-Z. (2015). Insights into the anaerobic biodegradation pathway of n-alkanes in oil reservoirs by detection of signature metabolites. Scientific Reports, 5, 9801. https://doi.org/10.1038/srep09801
Abu Laban, N., Dao, A., Semple, K., & Foght, J. M. (2015). Biodegradation of C7 and C8 iso-alkanes under methanogenic conditions. Environmental Microbiology, 17(12), 4898–4915. https://doi.org/10.1111/1462-2920.12643
Tan, B., Semple, K., & Foght, J. M. (2015). Anaerobic alkane biodegradation by cultures enriched from oil sands tailings ponds involves multiple species capable of fumarate addition. FEMS Microbiology Ecology, 91(5), iv042. https://doi.org/10.1093/femsec/fiv042
Beller, H. R., & Spormann, A. M. (1998). Analysis of the novel benzylsuccinate synthase reaction for anaerobic toluene activation based on structural studies of the product. Journal of Bacteriology, 180(20), 5454–5457. Retrieved from http://www.pubmedcentral.nih.gov/articlerender.fcgi?artid=107597&tool=pmcentrez&rendertype=abstract
Bozinovski, D., Herrmann, S., Richnow, H.-H., von Bergen, M., Seifert, J., & Vogt, C. (2012). Functional analysis of an anaerobic m-xylene-degrading enrichment culture using protein-based stable isotope probing. FEMS Microbiology Ecology, 81(1), 134–144. https://doi.org/10.1111/j.1574-6941.2012.01334.x
Fowler, S. J., Dong, X., Sensen, C. W., Suflita, J. M., & Gieg, L. M. (2012). Methanogenic toluene metabolism: community structure and intermediates. Environmental microbiology, 14(3), 754–764. https://doi.org/10.1111/j.1462-2920.2011.02631.x
Gieg, L. M., & Suflita, J. M. (2002). Detection of anaerobic metabolites of saturated and aromatic hydrocarbons in petroleum-contaminated aquifers. Environmental Science & Technology, 36(17), 3755–3762. Retrieved from http://www.ncbi.nlm.nih.gov/pubmed/12322748
Selesi, D., Jehmlich, N., Von Bergen, M., Schmidt, F., Rattei, T., Tischler, P., … Meckenstock, R. U. (2010). Combined genomic and proteomic approaches identify gene clusters involved in anaerobic 2-methylnaphthalene degradation in the sulfate-reducing enrichment culture N47. Journal of Bacteriology, 192(1), 295–306. https://doi.org/10.1128/JB.00874-09
Berdugo-Clavijo, C., Dong, X., Soh, J., Sensen, C. W., & Gieg, L. M. (2012). Methanogenic biodegradation of two-ringed polycyclic aromatic hydrocarbons. FEMS Microbiology Ecology, 81(1), 124–133. https://doi.org/10.1111/j.1574-6941.2012.01328.x
Annweiler, E., Materna, A., Safinowski, M., Kappler, A., Richnow, H. H., Michaelis, W., & Meckenstock, R. U. (2000). Anaerobic degradation of 2-methylnaphthalene by a sulfate-reducing enrichment culture. Applied and Environmental Microbiology, 66(12), 5329–5333. https://doi.org/10.1128/AEM.66.12.5329-5333.2000.Updated
Callaghan, A. V. (2013). Enzymes involved in the anaerobic oxidation of n-alkanes: From methane to long-chain paraffins. Frontiers in Microbiology, 4, 89. https://doi.org/10.3389/fmicb.2013.00089
Siddique, T., Penner, T., Klassen, J., Nesbø, C., & Foght, J. M. (2012). Microbial communities involved in methane production from hydrocarbons in oil sands tailings. Environmental Science & Technology, 46(17), 9802–9810. https://doi.org/10.1021/es302202c
Siddique, T., Fedorak, P. M., MacKinnon, M. D., & Foght, J. M. (2007). Metabolism of BTEX and naphtha compounds to methane in oil sands tailings. Environmental Science & Technology, 41(7), 2350–2356. Retrieved from http://www.ncbi.nlm.nih.gov/pubmed/17438786
Siddique, T., Fedorak, P. M., & Foght, J. M. (2006). Biodegradation of short-chain n-alkanes in oil sands tailings under methanogenic conditions. Environmental Science & Technology, 40(17), 5459–5464. Retrieved from http://www.ncbi.nlm.nih.gov/pubmed/16999125
Siddique, T., Semple, K., Li, C., & Foght, J. M. (2020). Methanogenic biodegradation of iso-alkanes and cycloalkanes during long-term incubation with oil sands tailings. Environmental Pollution, 258. https://doi.org/10.1016/j.envpol.2019.113768
Kong, J. D., Wang, H., Siddique, T., Foght, J., Semple, K., Burkus, Z., & Lewis, M. A. (2019). Second-generation stoichiometric mathematical model to predict methane emissions from oil sands tailings. Science of the Total Environment, 694, 133645. https://doi.org/10.1016/j.scitotenv.2019.133645
Mohamad Shahimin, M. F., & Siddique, T. (2017). Methanogenic biodegradation of paraffinic solvent hydrocarbons in two different oil sands tailings. Science of the Total Environment, 583, 115–122. https://doi.org/10.1016/j.envpol.2016.12.002
Mohamad Shahimin, M. F., & Siddique, T. (2017). Sequential biodegradation of complex naphtha hydrocarbons under methanogenic conditions in two different oil sands tailings. Environmental Pollution, 221, 398–406. https://doi.org/10.1016/j.envpol.2016.12.002
Mohamad Shahimin, M. F., Foght, J. M., & Siddique, T. (2016). Preferential methanogenic biodegradation of short-chain n-alkanes by microbial communities from two different oil sands tailings ponds. Science of Total Environment, 553, 250–257. https://doi.org/10.1016/j.scitotenv.2016.02.061
Mohamad Shahimin, M. F., Foght, J. M., & Siddique, T. (2021). Methanogenic biodegradation of iso-alkanes by indigenous microbes from two different oil sands tailings ponds. Microorganisms, 9(8), 1569. https://doi.org/10.3390/microorganisms9081569
Mohamad Shahimin, M. F., & Siddique, T. (2023). Biodegradation of 2-methylpentane in fluid fine tailings amended with a mixture of iso -alkanes under sulfate-reducing conditions. Canadian Journal of Microbiology. https://doi.org/10.1139/cjm-2023-0022
Mohamad Shahimin, M. F. (2016). Anaerobic biodegradation of hydrocarbons in different oil sands tailings ponds: Key microbial players and main activation pathway of hydrocarbon biodegradation. University of Alberta.
So, C. M., Phelps, C. D., & Young, L. Y. (2003). Anaerobic transformation of alkanes to fatty acids by a sulfate-reducing anaerobic transformation of alkanes to fatty acids by a sulfate-reducing bacterium, strain Hxd3. Applied and Environmental Microbiology, 69(7), 3892–3900. https://doi.org/10.1128/AEM.69.7.3892
Kropp, K. G., Davidova, I. A., & Suflita, J. M. (2000). Anaerobic oxidation of n-dodecane by an addition reaction in a sulfate-reducing bacterial enrichment culture. Applied and Environmental Microbiology, 66(12), 5393–5398. https://doi.org/10.1128/AEM.66.12.5393-5398.2000
Callaghan, A. A. V, Davidova, I. A., Savage-Ashlock, K., Parisi, V. A., Gieg, L. M., Suflita, J. M., … Wawrik, B. (2010). Diversity of benzyl- and alkylsuccinate synthase genes in hydrocarbon-impacted environments and enrichment cultures. Environmental Science & Technology, 44(19), 7287–7294. https://doi.org/10.1021/es1002023
Winderl, C., Schaefer, S., & Lueders, T. (2007). Detection of anaerobic toluene and hydrocarbon degraders in contaminated aquifers using benzylsuccinate synthase (bssA) genes as a functional marker. Environmental Microbiology, 9(4), 1035–1046. https://doi.org/10.1111/j.1462-2920.2006.01230.x
Edgar, R. C. (2004). MUSCLE: a multiple sequence alignment method with reduced time and space complexity. BMC Bioinformatics, 5, 113. https://doi.org/10.1186/1471-2105-5-113
Guindon, S., Gascuel, O., Dufayard, J.-F., Lefort, V., Anisimova, M., & Hordijk, W. (2010). New algorithms and methods to estimate maximim-likelihood phylogenies: assessing the performance of PhyML 3.0. Systematic Biology, 59(3), 307–321. https://doi.org/10.1093/sysbio/syq010
Abu Laban, N., Tan, B., Dao, A., & Foght, J. M. (2015). Draft genome sequence of uncultivated toluene-degrading Desulfobulbaceae bacterium Tol-SR, obtained by stable isotope probing using [13 C 6 ] toluene. Genome Announcements, 3(1), e01423-e1514. https://doi.org/10.1128/genomeA.01423-14
Toth, C. R. A., & Gieg, L. M. (2018). Time course-dependent methanogenic crude oil biodegradation: dynamics of fumarate addition metabolites, biodegradative genes, and microbial community composition. Frontiers in Microbiology, 8(JAN), 1–16. https://doi.org/10.3389/fmicb.2017.02610
Liu, J. F., Lu, Y. W., Zhou, L., Li, W., Hou, Z. W., Yang, S. Z., … Mu, B. Z. (2020). Simultaneous detection of transcribed functional assA gene and the corresponding metabolites of linear alkanes (C4, C5, and C7) in production water of a low-temperature oil reservoir. Science of the Total Environment, 746, 141290. https://doi.org/10.1016/j.scitotenv.2020.141290
Chen, J., Zhou, L., Liu, Y. F., Hou, Z. W., Li, W., Mbadinga, S. M., & Mu, B. Z. (2020). Synthesis and mass spectra of rearrangement bio-signature metabolites of anaerobic alkane degradation via fumarate addition. Analytical Biochemistry, 600(March), 113746. https://doi.org/10.1016/j.ab.2020.113746
Tan, B., Charchuk, R., Li, C., Abu Laban, N., & Foght, J. M. (2014). Draft genome sequence of uncultivated firmicutes (Peptococcaceae SCADC) single cells sorted from methanogenic alkane-degrading cultures. Genome Announcements, 2(5), e00909-e914. https://doi.org/10.1128/genomeA.00909-14
Foght, J. M. (2008). Anaerobic biodegradation of aromatic hydrocarbons: pathways and prospects. Journal of molecular microbiology and biotechnology, 15(2–3), 93–120. https://doi.org/10.1159/000121324
Musat, F. (2015). The anaerobic degradation of gaseous, nonmethane alkanes - from in situ processes to microorganisms. Computational and Structural Biotechnology Journal, 13, 222–228. https://doi.org/10.1016/j.csbj.2015.03.002
Ji, J. H., Zhou, L., Mbadinga, S. M., Irfan, M., Liu, Y. F., Pan, P., … Mu, B. Z. (2020). Methanogenic biodegradation of C9 to C12 n-alkanes initiated by Smithella via fumarate addition mechanism. AMB Express, 10(1). https://doi.org/10.1186/s13568-020-0956-5
Ji, J. H., Liu, Y. F., Zhou, L., Mbadinga, S. M., Pan, P., Chen, J., … Mu, B. Z. (2019). Methanogenic degradation of long n-alkanes requires fumarate-dependent activation. Applied and Environmental Microbiology, 85(16), e00985–19. https://doi.org/10.1128/AEM.00985-19
Oberding, L. K., & Gieg, L. M. (2018). Methanogenic paraffin biodegradation: alkylsuccinate synthase gene quantification and dicarboxylic acid production. Applied and Environmental Microbiology, 84(1), e01773-e1817. https://doi.org/10.1128/AEM.01773-17
Aitken, C. M., Jones, D. M., Maguire, M. J., Gray, N. D., Sherry, A., Bowler, B. F. J., … Head, I. M. (2013). Evidence that crude oil alkane activation proceeds by different mechanisms under sulfate-reducing and methanogenic conditions. Geochimica et Cosmochimica Acta, 109, 162–174. https://doi.org/10.1016/j.gca.2013.01.031
Jarling, R., Sadeghi, M., Drozdowska, M., Lahme, S., Buckel, W., Rabus, R., … Wilkes, H. (2012). Stereochemical investigations reveal the mechanism of the bacterial activation of n-alkanes without oxygen. Angewandte Chemie - International Edition, 51(6), 1334–1338. https://doi.org/10.1002/anie.201106055
Chen, J., Liu, Y., Zhou, L., Mbadinga, S. M., Yang, T., Zhou, J., & Liu, J. (2019). Methanogenic degradation of branched alkanes in enrichment cultures of production water from a high-temperature petroleum reservoir. Applied Microbiology and Biotechnology, 1–4. https://doi.org/10.1007/s00253-018-09574-1
Siddique, T., Mohamad Shahimin, M. F. M. F., Zamir, S., Semple, K., Li, C., & Foght, J. M. J. M. (2015). Long-term incubation reveals methanogenic biodegradation of C5 and C6 iso-alkanes in oil sands tailings. Environmental Science & Technology, 49(24), 14732–14739. https://doi.org/10.1021/acs.est.5b04370
An, D., Brown, D., Chatterjee, I., Dong, X., Ramos-Padron, E., Wilson, S., … Voordouw, G. (2013). Microbial community and potential functional gene diversity involved in anaerobic hydrocarbon degradation and methanogenesis in an oil sands tailings pond. Genome, 56(10), 612–618. https://doi.org/10.1139/gen-2013-0083
Acknowledgements
We thank Canadian Natural Resources Ltd. and Shell CNUL Sands Inc. for providing oil sands tailings samples.
Funding
We gratefully acknowledge funding from the Helmholtz-Alberta Initiative (TS), NSERC Discovery Grant (TS), and Canada Foundation for Innovation (TS). We also acknowledge a Ph.D. scholarship award (King’s Scholarship) and fellowship to MFMS from the Public Service Department of Malaysia and Universiti Malaysia Perlis, respectively.
Author information
Authors and Affiliations
Contributions
Conceptualization: TS and MFMS, data curation: MFMS, formal analysis: MFMS, funding acquisition: TS, investigation: MFMS, methodology: MFMS, project administration: MFMS, resources: TS, supervision: TS, validation: TS, writing original draft: MFMS, and review and editing: MFMS and TS.
Corresponding author
Ethics declarations
Ethics Approval
Not applicable.
Consent to Participate
Not applicable.
Consent for Publication
Not applicable.
Competing Interests
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Mohamad Shahimin, M.F., Siddique, T. Uncovering Anaerobic Hydrocarbon Biodegradation Pathways in Oil Sands Tailings from Two Different Tailings Ponds via Metabolite and Functional Gene Analyses. Appl Biochem Biotechnol (2024). https://doi.org/10.1007/s12010-024-04855-0
Accepted:
Published:
DOI: https://doi.org/10.1007/s12010-024-04855-0