Abstract
In this study, for the first time, double-layer films based on furcellaran and gelatin were obtained, in which the 1st layer of furcellaran was enriched with various plant extracts. The aim of this work was to improve the composition of the developed innovative packaging material by using two layers of biopolymer complexes and analysing the composites to determine the effect of different model plant extracts on the physicochemical properties of the double-layered materials. Furthermore, the hypothesis that positron annihilation lifetime spectroscopy (PALS) could be correlated with gas barrier properties was verified. The addition of plant extracts significantly influenced the physicochemical parameters of the obtained double-layered materials. The lowest solubility was noted in films with the addition of garlic extract (58.40%) and black pepper extract (59.26%) which also had the lowest water content (8.88%). The film with garlic extract also had the lowest oxygen transfer rate (OTR) value after 6 months of storage which is consistent with PALS. The presented data suggest the possibility of correlating PALS results with the gas barrier properties. Tests carried out using this method will facilitate the selection of new potential ingredients when designing packaging materials based on biopolymers, thus, making it possible to obtain new-generation matrices.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
The increase in food consumption as a consequence of civilization progress and processes of ubiquitous market globalisation, as well as changes in the needs and expectations of consumers, are among the most important factors significantly influencing the speed of development in the packaging industry (Kawecka & Cholewa-Wójcik, 2017). The direction of transformation on the food packaging market is shifting towards the development of innovations meeting the requirements contained in legal regulations regarding the reduction of consuming so-called classic polymer materials, while ensuring the ability to meet the increasingly stringent requirements of the food industry and consumers themselves (de Vargas et al., 2022; Garavand et al., 2017; Neethirajan & Jayas, 2011). Due to the above, research has been carried out for many years on the development and modification of biopolymer film composition as an alternative to synthetic packaging (Grzebieniarz et al., 2023; Mouhoub et al., 2023).
Biopolymers, as alternative substitutes for plastics, seem to be a promising direction of work on innovations in packaging, however, despite intensive research, areas that limit their use are still identified, and which, at the same time, provide space for improving their quality. One of the weaknesses of biopolymers, limiting their widespread applicability, is the not fully satisfactory values of the parameters characterising functional properties of the films produced from them, e.g. relatively low mechanical strength of the film, degradation rate in various environments and, consequently, changes in mechanical, physical as well as thermal properties during storage (Voon et al., 2012). In addition, their resistance to the growth of microorganisms on the surface should be considered, and consequently, the impact on the microbiological purity of the stored products as well as the potential release of harmful compounds into the packaged product (Pérez et al., 2021). In order to overcome these limitations, scientists are making various attempts to modify biopolymer films. The most common of these is a combination of 2 or more biodegradable polymers (Izzi et al., 2023; Khan et al., 2023) plasticiser additives (Hernández et al., 2022), chemical modifications, the addition of nanoparticles or nanofillers (Neethirajan et al., 2011; Wang et al., 2022), as well as active ingredients (Gulati et al., 2023; Verónica et al., 2023). Nanoparticle additives are innovative but controversial approach. Chemical modifications seem to be promising; nonetheless, they have many restrictions resulting from economic- and safety-related aspects, as well as potential application on an industrial scale, especially in the food industry. Hence, the approach taking the concepts of combining polymers with additions of natural active ingredients into account seems to be a justified, economical form of modifying biopolymer films (Dutta & Sit, 2023; Shakouri et al., 2023).
One of the polymers with very great applicative potential is furcellaran. It is a negatively charged copolymer of β-carrageenan and κ-carrageenan obtained from red algae. The feature distinguishing it from other biopolymers is its ability to form gels that are transformed from a ball to a helix, i.e. from a disordered to ordered state. This allows the modification of biopolymer films and shaping the desired functional properties of packaging materials (Marangoni et al., 2020; Schaefer et al., 2020). Recent literature reports demonstrate the validity of combining two polysaccharides in the synthesis of biopolymer films. In their research da Silva Pereira et al. (2021) indicate that the mechanical and barrier properties of protein- and polysaccharide-based films can be improved through interactions between the components. They have shown that these interactions can affect film properties such as texture, thermal stability or chemical structure. Izzi et al. (2023) in their study also demonstrate that combining more than one biopolymer for film formation leads to the precipitation of materials with better mechanical and barrier properties compared to those having only one component. In the present study, a protein-polysaccharide complex was developed using furcellaran and gelatin, both of which are capable of forming gels.
The use of biopolymers capable of forming gels makes it possible to innovatively modify the material by using two biopolymer layers. As demonstrated by Grzebieniarz et al. (2023) and Saini et al. (2021) the synthesis of multilayer biopolymer films can significantly improve the performance of composites by improving, among other things, its barrier properties or increasing the strength of the material. The formation of multilayer biopolymer films is an innovative pathway for the synthesis of biopolymer materials with very high development potential and significant opportunities for their improvement and modification.
In their research Ribeiro et al. (2021) note a recent trend in packaging research focusing on the using natural sources of antimicrobial and antioxidant compounds, such as natural plant extracts. They indicate that this area of research is motivated by environmental and health concerns. As future challenges in this area of research, they identify the need for a comprehensive analysis of the physicochemical and performance properties of biopolymer-based films, especially in the context of assessing the effects of natural extracts on film parameters.
The addition of plant extracts of films has been extensively reported in the literature, however, focusing only on the modification of monolayer films (Kumar et al., 2022; Silva-Weiss et al., 2013). As an active ingredient, it has been shown to improve antimicrobial, barrier, mechanical or thermal properties. Due to the origin of the extract and the solvent used, biopolymeric materials can be successfully modified (da Rosa et al., 2020; Xu et al., 2021). The evaluation of the effects of plant extracts on multilayer films appears to be of great importance in view of the possibility of modifying biopolymer-based materials through the use of natural, non-toxic compounds and the properties of the biopolymers themselves. However, since research in this field has only recently been carried out, there are still relatively few areas that need further investigation with a particular focus on the effect of plant extracts on the performance properties of multilayer biopolymer films.
Due to the fact that the actions undertaken to improve the composition of double-layer films based on furcellaran and gelatin, additionally enriched with additives, are highly innovative, and it is extremely important to subject the produced films to quality assessment considering the features and properties characterising their applicative value. Assessing the value in use, understood as the degree to which the set of properties of the packaging material meets the requirements related to its application, is crucial to determine the potential for using the material in the case of a specific food product group with concrete quality requirements (Cholewa-Wójcik, 2016; Vaziri et al., 2022).
As part of work on improving the composition of the biopolymer double-layer packaging material under development, the addition of natural plant extracts was introduced in order to determine the effect on the physicochemical properties of multi-layer biopolymer films based on furcellaran and gelatin. In addition to standard methods used to evaluate the quality and performance of biopolymer packaging films, a method of positron annihilation lifetime spectroscopy (PALS) was also used, which may become an interesting tool in the selection and optimisation regarding the composition of biopolymer packaging matrices. Water extracts from commonly available and economical plants: lingonberries, black pepper, garlic, dried tomatoes and tansy herb were prepared. The extracts were added to the 1st furcellaran layer in the double-layer films, and the planned tests were carried out on the composites obtained in this manner. The structure was characterised by atomic force microscopy (AFM) and scanning electron microscopy (SEM). Colour and barrier properties against UV light, water properties such as water content, solubility and contact angle as well as thermal properties (DSC) were determined. Another area of research was gas barrier properties as a function of time. Water vapour transmission rate (WVTR) and oxygen transmission rates (OTR) were analysed over a 6-month period. Due to the fact that the parameters characterising the functional properties of the packaging material are one of the priority components determining the final quality of the packaging, the analysis of the obtained results (so-called technical parameters) was the basis for assessing the quality of composites as innovative packaging materials for food products. The hypothesis of this study was to prove whether the innovative solution to corelate PALS with the gas barrier properties and using active furcellaran/gelatin-based double-layer films are ways to eliminate the functional drawbacks of single-layer films.
Materials and Methods
Materials
Gelatin (GEL) was purchased from Chemland (Stargard, Poland) and furcellaran (FUR) was supplied by Est-Agar AS (Karla Village, Estonia). The declared molecular weight of the FUR was 2.95 × 105 Da, while the chemical composition included carbohydrate content, 79.61/100 g; protein content, 1.18/100 g; and fat content, 0.24/100 g. Glycerol was also purchased from Chemland (Stargard, Poland).
The implemented spices grainy black pepper (Kamis, Poland), granulated garlic (Kamis, Poland) and dried tomatoes (Kamis, Poland) were purchased in a nearby Lidl store (Kraków, Poland), lingonberries came from Natura Wita (Pińczów, Poland), and tansy herb (Tanacetum vulgare) from Plantago (Złotów, Poland).
Preparation of Aqueous Plant Extracts
All the aqueous extracts were prepared in the same way, and a detailed description can be found in the paper by Jamróz et al. (2022a, b, c). Furthermore, 10 g of black pepper fruit (BPE), dried tomatoes (DTE), lingonberry fruit (LBE), tansy herb (TVE) or granulated garlic (GE) were weighed successively and poured into 90 ml of deionised water. The mixture was then stirred at 80 °C for 2 h. The resulting extracts were filtered using quantitative filter paper.
Preparation of Double-Layer Films
The 1st layer of the film was a 1% furcellaran solution with the addition of aqueous plant extracts. Glycerol was used as a plasticiser in a ratio of 1:1, according to polymer weight. In 5 beakers, 370 ml of 1% FUR solutions were prepared, to which 100 ml of water extracts (LBE, BPE, DTE, TVE, GE) were successively added. The solutions were left in conditions of constant stirring at 60 °C for 1 h. Then, the solutions prepared as such were poured onto a mould and allowed to transform into a gel. At this time, the 2nd layer was prepared. Five beakers contained 470 ml of 1% FUR, to which 2.35 g of gelatine were added, while constantly stirring (temperature 60 °C, duration 20 min). The solutions prepared in this way were poured onto the 1st film layer. The films were allowed to dry at room temperature under a lab hood, and after complete drying, they were removed from the moulds for further analysis.
Positron Lifetime Spectroscopy
In the present work, a digital positron lifetime spectrometer purchased from TechnoAP was used (Mawatari, Japan). As the detector, 2 photomultipliers were applied: H3378-50 coupled with BaF2 scintillators in the form of a disc. The time resolution of the spectrometer was about 180 ps. The isotope 22Na was used as the positron source. The isotope was enveloped into a 7-µm thick Kapton film. The activity of the source was approximately 1.9 MBq. Samples comprised 2 stacks of 7 films with a total thickness of about 1.5 mm which sandwiched the positron source. All spectra containing more than 1.5 × 106 counts were deconvoluted using LT code taking the source contribution and subtracting the background into account (Kansy, 1996).
Atomic Force Microscope – AFM
Surface topography images were obtained with the Dimension Icon XR Atomic Force Microscope (AFM) (Bruker, Santa Barbara, CA, USA). The microscope was operated in the air via the Peak Force Tapping (PFT) mode. Standard silicon cantilevers with a nominal spring constant of 0.4 N/m, nominal tip radius < 2 nm and triangular geometry were used in the analysis. Before evaluation, each sample was cut into appropriately sized pieces and glued to a smooth silicon wafer.
Scanning Electron Microscope – SEM
Sample morphologies were analysed using the SEM/FIB Quanta 3D 200i (FEI, Oregon, USA) microscope. The typical parameters were: 5-kV acceleration voltage (current of 0.2 nA) and 10-mm working distance. Magnifications of × 200, × 800 and × 1500 were registered for the top views of the samples. The samples were imaged with no prior preparation. From each sample, 2 different spots were chosen as those most representative. The cross sections of the samples were imaged with the same parameters, magnifications of × 200, × 500 and × 1000.
Optical and Colour Parameters
UV spectra were recorded for the film within the wavelength range of 300–700 nm using the Shimadzu 2101 spectrophotometer (Shimadzu, Kyoto, Japan). The opacity was measured according to the method proposed by Du et al. (2021) and was calculated using the following formula:
where A600 means the absorbance value at the wavelength of 600 nm, while x is the thickness of the film (mm). The film thickness was measured with a handheld device, Mitotuyo, No. 7327 (Kawasaki, Japan).
The surface colour of the composites was evaluated using the reflection method via the Color i5 spectrometer (X-Rite, Grand Rapids MI, USA, illuminant D65) with the following parameters: measuring geometry d/8, observer 10°, spectral range 400–700 nm and measuring gap 25 mm. A white standard plate with the following parameters: L* = 98.82, a* = − 0.36 and b* = 3.28 was used as the background.
The results are demonstrated as L* (lightness), a* (red–green) and b* (yellow–blue). The whiteness (WI) and yellowness indices (YI) were also estimated based on the formulas below:
Water Content and Solubility in Water
The water properties were determined according to the method proposed by Kavoosi et al. (2014) and Souza et al. (2017). From each film, 20 × 20-mm squares were cut out and then weighed to the nearest 0.0001 to determine the initial weight (W1). The samples were dried for 24 h at 70 °C, and re-weighed (initial dry weight—W2). Each sample was placed in 30 ml of deionised water and stored at room temperature. After 24 h, the samples were dried again at 20 °C for 24 h and weighed (final insoluble mass—W3). Before the analysis, the samples were conditioned by storing them in a desiccator with a saturated solution of Mg(NO3)2 (RH = ~ 53%) at room temperature for 48 h. Water content and solubility were determined using the following formulas:
Water Vapour Transmission Rate (WVTR) and Oxygen Transmission Rate (OTR)
Water vapour transmission rate (WVTR) and oxygen transmission rates (OTR) were determined as a function of time using the gravimetric method according to ISO 2528:2017. A special vessel was filled with a drying substance (silica gel), which was then covered with the tested material and placed in a chamber with a regulated microclimate. Conditions of 23 °C and 85% humidity were used to establish the WVTR value, and 23 °C and 50% humidity were implemented to determine the OTR value. The analysis included 4 measurements immediately after film preparation and after 1, 4 and 6 months.
Contact Angle
The contact angle of the tested films with respect to water was determined via the sessile drop method at room temperature (~ 23 °C) using a video contact angle measurement system (OCA, Dataphysics, Germany). A drop of deionised water (8 µl in volume) was dotted onto the surface of the film with a micro-injector, and then an image was obtained and analysed.
Thermal Properties–Differential Scanning Calorimetry (DSC)
The thermodynamic characteristics of the films were determined by using the DSC 4000 differential scanning calorimeter (PerkinElmer, USA). Samples (approximately 5 mg) were hermetically closed in aluminium pans and heated from 30 to 300 °C at a rate of 10 °C/min. An empty aluminium pan was used as a reference. Temperatures and enthalpy of thermal transitions were determined with the use of Pyris software (PerkinElmer, USA).
Statistical Analysis
All analyses were performed in at least 3 replicates. Differences between the means of the data were tested for statistical significance at p < 0.05 using Statistica software (Stat Soft, Inc., USA). The Saphiro-Wilk test was used to check normality of distribution, while one-way ANOVA was applied to assess the significance of differences between the mean values. Turkey’s post-hoc tests were also implemented.
Results
Positron Annihilation Lifetime Spectroscopy (PALS)
A nucleus of 22Na undergoes the β+-decay emitting a positron which enters the material studied. Emission of the positron is followed by emission of a 1.28-MeV photon. The detection of the photon indicates ‘birth’ of the positron. The detection of one of the annihilation photons of energy totalling 511 keV marks ‘death’ of the positron allows to determine its lifetime. This is the basis of PALS. In molecular materials, the positron and electron pair can form a bound state before annihilation, which is an analogue of the hydrogen atom, called positronium (Ps). Ps has can be formed in regions of low electron density such as pores or inter- and intra-molecular spaces. In the case of polymers, the latter are called local free volumes or holes. Due to the arrangement of the spins of both particles, Ps can exist in 2 states, so-called para-Ps (p-Ps), when the spins are set anti-parallel and orto-Ps (o-Ps), when the spins are parallel. When Ps is created in a vacuum, the ratio of the p-Ps to o-Ps is 1:3 and p-Ps annihilates into 2 photons with the lifetime of 125 ps for, while o-Ps annihilates into 3 photons with the lifetime of 142 ns. In matter, due to the presence of other electrons, the positron forming o-Ps can annihilate with a ‘foreign’ electron with anti-parallel spin into 2 photons in a process called pick-off annihilation. This causes significant shortening of the o-Ps lifetime to a few nanoseconds. Moreover, this lifetime depends on the size of the local free volume holes. The positron lifetime spectrum can be described as the sum of exponential decay functions. Each exponential decay function, called a positron lifetime component, is defined by its intensity I and lifetime τ. For materials where Ps is formed, there are usually 3 positron lifetime components. The shortest one, τ1, comes from annihilation of p-Ps, the medium one, τ2, within the range of 0.3 to 0.45 ns, originates from annihilation of positrons which do not form Ps, i.e. free positrons and the longest one, τ3, come from annihilation of o-Ps in the pick-off process.
To obtain information on the size of the local free volume hole, the following relationship is commonly used (Eldrup et al., 1981; Tao et al., 2020). It links the radius of free volume R with the o-Ps lifetime τ3:
where Δ = 0.1656 nm and this is an empirical parameter describing the depth of penetrating the positronium wave function into the area surrounding the free volume hole where the electrons are located. The increase in the value of radius R causes a non-linear increase in the value of the lifetime component τ3.
In Fig. 1, the results of the positron lifetime measurements are presented for the as received biopolymers and after storage for 6 months. Shown are the o-Ps lifetime, τ3 (Fig. 1A) and the average volume of the free volume holes Vh = 4/3πR3/3 (Fig. 1B) calculated using the free volume radius given by Eq. (1).
Another important parameter characterising the properties of the material is the fraction of free volumes fv in the material under study. There have been attempts to connect it with the intensity of the o-Ps component, I3, in the spectrum via the following formula (Wang et al., 1990).
where fv is given in %, Vh = 4/3πR3/3 is expressed in Å3, and the coefficient a = 0.0018 (Wang et al., 1990). However, this equation should be used with caution because various factors can influence I3 and not only changes in the fraction of the free volume. Results of the I3 intensity component were higher for the reference than for furcellarans with extracts. In the case of the obtained biopolymers, the value for the reference was 24.4 ± 0.1%, and after storage, 25.4 ± 0.1%. For all furcellarans with extracts, the values were similar both for the achieved fims and after 6 months of storage, the average value being 22.2 ± 0.1%. Results for value of the radius R and fraction of free volumes fv are given in Table 1.
Uncertainty related to the quantities in Table 1 are as follows: R – 0.001 nm, fv – 0.1%.
The introduction of additives to the base polymer causes a decrease in τ3 and hence a reduction in the average size of the free volume holes in comparison to the reference sample. However, these changes are small. The changes are more visible for TVE, DTE and GE than for the BPE and LBE films. The situation is similar for the intensity of the o-Ps component, I3, and therefore, the free volume fraction fv.
Storing the films for 6 month causes a decrease in the average size of the free volume holes for all samples. This decrease may correlate with the decrease in water vapour transmission rate. The smallest size was observed for the TVE and GE films.
The application of PALS in the study of coatings reported in literature, films and food packaging allowed to indicate that the method is able to demonstrate differences in porosity at the molecular level in the tested materials (Reineccius & Yan, 2016). Small changes in size of the free volume holes significantly affect diffusion due to the exponential dependence of diffusion on these sizes. This relationship has some support in the free volume theory by Cohen and Turnbull (1959). Within the framework of this theory, the relationship between the diffusion coefficient of a penetrant, D, and the average free volume of a polymer, Vh, is the following:
where R is the gas constant, while A and B are empirical constants. The reciprocal correlation between the size of the free volume holes and logarithm of gas permeability is as was shown for polyhydroxyimides (Nocoń-Szmajda et al., 2021).
Research on glassy carbohydrate encapsulation systems performed by Drusch et al. (2012) and further, by Serfert et al. (2013), allowed to note that an increase in the free volume holes negatively influences oxygen diffusion. However, according to those authors, oxygen diffusion rates may depend not only on free volume size but also on oxygen solubility in a matrix. This may shed light on oxygen permeability results regarding the films under study, for which it was shown that despite the same size of the free volume holes for TVE and GE films, only the latter exhibited significant decrease in oxygen permeability after 6 months of storage (Fig. 2).
Recently, PALS has shown great potential in differentiating the size of free volume elements that present similar bulk properties but significant differences in the stability of the microencapsulated oil, showing that the oxygen diffusivity through the matrix must be a key determinant in auto-oxidation of the encapsulated oil.
Water Vapour Transmission Rate (WVTR) and Oxygen Transmission Rate (OTR)
WVTR (A) and OTR (B) parameters of the analysed composites are presented in Fig. 2. It has been demonstrated that in the case of water vapour permeability, the results significantly differed (p < 0.05) depending on the type of plant extract.
A statistically significant (p < 0.05) decrease was also observed in the WVTR value after the first month of film analysis. Jamróz et al. (2022a, b, c) and Bonilla and Sobral (2016) showed that the addition of plant extracts did not significantly improve the WVTR value. However, in the study, it was indicated that there are significant differences between the various extracts, and the lowest value was observed for LBE, which exhibited the most hydrophobic nature. Jamróz et al. (2022a, b, c) observed a completely different phenomenon. These authors enriched double-layer films based on furcellaran and CMC with lingonberry extract, and the obtained values were within the range of 852.00–957.47 g/m2 x d. Not only is the selection of the active ingredient important, but also the fine-tuning of the film-forming matrix is of great significance as well.
The analysis of oxygen barrier properties showed that the type of film is not significant (p > 0.05), however, as in the case of the WVTR value as a function of time, a statistically significant (p < 0.05) decrease was observed. The lowest OTR values were noted in films enriched with garlic extract. The obtained data confirm the validity of PALS analysis, which showed that after 6 months of storing the tested films (Table 1), the GE-enriched biopolymer matrix reduces the size of free volumetric holes during storage, making it difficult for oxygen molecules to penetrate. The barrier properties of biopolymer films depend on many factors, including material thickness and composition, permeability as well as microclimate conditions such as temperature and humidity (Jamróz et al., 2019). Lower WVTR and OTR values are a desirable feature in the case of biopolymer films, especially those with potential use for food storage. Barriers to water vapour can reduce the migration of moisture, and thus, contribute to extending the shelf-life of products (Jamróz et al., 2020).
Atomic Force Microscopy (AFM) and Scanning Electron Microscopy (SEM) Analysis
Atomic force microscopy (AFM) and scanning electron microscopy (SEM) were used to visualise the topography and surface structure of the film. These results are shown in Fig. 3.
All films, both the 1st (A) and 2nd layers (B) of the composites, were analysed. For all the obtained composites, the structure of the 1st layer was much rougher, which may have been due to the addition of water extracts. Medina-Jaramillo et al. (2017) demonstrated that there is a significant relationship between the roughness of the material and its hydrophobicity/hydrophilicity. Therefore, the surface roughness of the composites results from the hydrophilicity of the added composites. These authors achieved a similar result to Ju et al. (2019a, b) as well as Jamróz et al. (2022a). The 2nd layer of the composites was characterised by lower roughness compared to the 1st one. Pure FUR and GEL films have a dense and non-porous structure (Jamróz et al., 2022b; Makarova et al., 2022; Yao et al., 2019). The observed roughness may have been caused by the presence of insoluble compounds in the extracts and the movement of hydrophilic molecules to the surface of the film during the drying process (Ju et al., 2019a, b).
Both the structure of furcellaran and gelatin have a specific smooth, compact surface (Jamróz et al., 2018). The addition of plant extracts significantly affected its appearance (Fig. 3C). Here, a spongy structure can be observed that is very ordered, which can also directly affect the gas barrier properties. The ordered, compact structure of the film can be a strong barrier for gases, hindering the migration path. Moreover, the separation of the layers was not as pronounced as was the case in other double-layer films (Jamróz et al., 2022a, b, c), however, this may indicate high compatibility between the 2 biopolymers present in the films.
Optic and Colour Parameters
All films showed barrier properties against UV light, which is shown in Fig. 4. The highest UV barrier was shown in the case of films with the addition of lingonberry extract, pepper and tansy, and the lowest with garlic and tomato extract. The barrier effect of LBE against UV light has been observed and previously described by Jamróz et al. (2022a, b, c) and indicates the action of phenolic components. For the black pepper extract, the maximum absorbance value is observed at a wavelength of about 350 nm, which is related to the presence of piperine in its composition, an alkaloid that gives the spice a characteristic taste (Aziz et al., 2015). The high barrier properties against UV light in the case of tansy is associated with the presence of flavonoids. Several main flavonoids found in this plant can be distinguished, namely: apigenin, acacetin, luteolin, cynaroside, eupatilin and jaceidin (Kurkina et al., 2011). The main active substance of garlic is allicin, which is formed as a result of the action of the enzyme allinase on allin. It is a phytoncide with strong bactericidal effects and a characteristic smell. Bocchini et al. (2001) conducted a UV analysis of allicin, obtaining the highest absorbance value within the range of 300 and 200 nm, indicating that allicin is mainly responsible for the barrier properties of the composite containing garlic extract. In the case of the tomato extract, the highest absorbance value was observed between 400 and 550 nm. The decrease in light transmittance can be explained by the presence of lutein, lycopene and β-carotene in the sample. The increase in absorbance due to the presence of lutein and β-carotene was observed at the level of 420–450 nm, and for lycopene, 470–500 nm (Siti Imama et al., 2020).
As expected, all films showed high opacity (Table 2). There are many studies in which the effects of plant extracts have been noted with regard to the degree of film opacity, due to the colour resulting from the presence of dyes, which, at the same time, contributes to the reduction of UV light transmission (Gulati et al., 2023; Munir et al., 2019). In all the films, the values of parameters L*, a* and b* differed significantly and were consistent with the visual observation (Table 2). The highest value of the L* (lightness) parameter with the highest transparency was obtained for the film with the addition of GE, and the lowest for the film with the addition of LBE. A similar effect was obtained for the film with lingonberry extract Jamróz et al. (2022a, b, c) noted that the degree of opacity increased with the concentration of the extract, and Gulati et al. (2023) noted that the total colour difference ∆E also increased with increasing extract concentration. This may be due to the selective absorption of low-wavelength light by phenolic compounds (Fabra et al., 2018), and the same UV light barrier mechanism for garlic extract was indicated by Salim et al. (2022), who have also shown high barrier properties for films containing garlic extracts.
As Ortiz-Duarte (2021) and Júnior (2021) have shown, the characteristics of optical properties and opacity are of great importance in the context of the potential application of films in the food industry. The use of films with high barrier properties makes it possible to create a protective barrier and support for food to improve its sensory properties. Light passing through the packaging material may promote oxidation reactions of photosensitive compounds, consequently, shortening their shelf-life. The primary function of packaging materials and packaging made of them should therefore be to protect products, e.g. against the negative effects of UV light and, as a result, to extend shelf-life, protect against loss of colour and nutritional value as well as the presence of an unpleasant taste. In addition, the use of natural dyes allows to reduce the share of synthetic dyes in the food industry (Arfat et al., 2017; Salim et al., 2022).
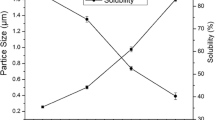
FUR-based films are hydrophilic, and the addition of plant extracts affects both the 1st and 2nd film layers (Jamróz et al., 2021). Various effects of plant extracts on water parameters were observed (Table 2). There are 2 types of water-biopolymer interactions. If the water content is less than 100 g per 1 kg of biopolymer, hydrogen bonds cause the formation of a water-biopolymer system. However, if the water content reaches a level above this value, water-water interactions occur inside the biopolymer matrix (Zając et al., 2021). For the LBE and GE films, an increase in the value towards hydrophobic properties was observed, while for the lingonberry extract, we noted a WCA value above 90°, which proves the hydrophobicity of the composite. Similarly, it was for these films that the lowest water content was found. For the remaining films, the addition of plant extracts increased the hydrophilicity, which may be related to the presence of numerous phenolic groups in the extracts, further improving the interaction of the film with water molecules by reducing the contact angle and increasing the water content in the composite (Sun et al., 2017).
As described by Júnior (2021) biopolymer films tend to be hydrophilic, which results in low barrier properties against water vapour and, consequently, in reduced functional values and potential applications. Therefore, it seems important to analyse the possibility of modifying and altering biopolymer films to increase hydrophobic properties and improve barrier properties. Both furcellaran and gelatin films are hydrophilic and have high solubility (Jamróz et al., 2021; Tagrida et al., 2023), and additionally, plant extracts contribute to increasing the solubility of biopolymer films (Tagrida et al., 2023). Presenting these components in a different configuration and arrangement can eliminate the problem of excessive solubility. The obtained double-layer films based on furcellaran, gelatin and plant extracts are characterised by good water properties, which may have resulted from the formation of a furcellaran-gelatin complex, already presented earlier by our team (Jamróz et al., 2018).
Thermal Properties
In Table 2, the melting point (Tm) and enthalpy (ΔH) of the film with added extracts are shown. The endothermic peak of the furcellaran-based film is approximately 206 °C, while for gelatin films, it occurs between 130–140 °C, which is most likely associated with a partial change regarding the helix-coil of some proteins (Jamróz et al., 2019). The observed value of the melting point is within the range characteristic for the FUR/GEL composite. The highest enthalpy value was obtained for the film containing LBE. This may be due to the presence of water-polymer hydrogen bonds, which can stabilise the structure of the composite, good miscibility of the component polymers or the interaction of the FUR matrix with the phenolic compounds of the lingonberry extract. The reinforced film structure showed higher thermal stability, requiring more energy to break down the internal interactions (Jamróz et al., 2021, 2022c). In the work by Jamróz et al. (2022c), the authors added lingonberry extract to a double-layer film based on furcellaran and CMC and observed that as the concentration of lingonberry extract elevated, the ΔH value also increased. However, even at the highest concentration of the extract, the value of ΔH was lower (164.1 J/g) than in the case of double-layer films based on furcellaran and gelatin enriched with the blueberry extract. These results indicate that the film-forming matrix is of great importance in the preparation of materials based on biopolymers.
Conclusions
Evaluating the quality of double-layer films based on FUR/GEL films with the addition of aqueous plant extracts, carried out on the basis of the analysis of parameters characterising the functional properties, showed that these films have great applicative potential and can be used as an alternative to food packaging based on plastics. A decrease in the WVTR and OTR values was observed, which is a desirable phenomenon due to the possibility of ensuring better conditions, e.g. for stored food. Water, colour and optical properties depend on the type of extract used, in particular, its active substance or the amount of phenolic compounds. All the analysed films showed good barrier properties against UV light. The type of water extract does not significantly affect the water vapour permeability of the film, but it differentiates the degree of oxygen permeability. With the passage of storage time, the WVTR and OTR values of the produced biopolymer films decrease, which may be related to changes in the micro-environment. In addition, the conducted research using the PALS method allowed to confirm that the addition of plant extracts, in particular, those from garlic, affects the parameters of gas permeability after 6 months of storage, which is a highly satisfactory result. In this work, a new direction of research is indicated, concerning the determination of the correlation between PALS analysis and the performance parameters of biopolymer films. The remaining results of research on the quality assessment of the produced biopolymer films were confirmed by relatively stable performance parameters. Nevertheless, due to the developmental nature of this work, further research is necessary to determine the impact of the packaging material on changes in the shelf-life of the product packed in it. The acquired knowledge will allow for the identification of further activities in the field of developing the concept of innovation regarding integrated products, as well as managing their life cycle.
Data Availability
The datasets generated and/or analysed during the current study are available from the corresponding author on a reasonable request.
References
Arfat, Y. A., Ahmed, J., Hiremath, N., Auras, R., & Joseph, A. (2017). Thermo-mechanical, rheological, structural and antimicrobial properties of bionanocomposite films based on fish skin gelatin and silver-copper nanoparticles. Food Hydrocolloids, 62, 191–202. https://doi.org/10.1016/j.foodhyd.2016.08.009
Aziz, D. M., Hama, J. R., & Alam, S. M. (2015). Synthesising a novel derivatives of piperine from black pepper (Piper nigrum L.). Journal of Food Measurement and Characterization, 9(3), 324–331. https://doi.org/10.1007/s11694-015-9239-2
Bocchini, P., Andalò, C., Pozzi, R., Galletti, G. C., & Antonelli, A. (2001). Determination of diallyl thiosulfinate (allicin) in garlic (Allium sativum L.) by high-performance liquid chromatography with a post-column photochemical reactor. Analytica Chimica Acta, 441(1), 37–43. https://doi.org/10.1016/S0003-2670(01)01104-7
Bonilla, J., & Sobral, P. J. A. (2016). Investigation of the physicochemical, antimicrobial and antioxidant properties of gelatin-chitosan edible film mixed with plant ethanolic extracts. Food Bioscience, 16, 17–25. https://doi.org/10.1016/j.fbio.2016.07.003
Cholewa-Wójcik, A. (2016). The analysis of change in selected functional properties of plastic films as a result of aging process. Towaroznawcze Problemy Jakości, (3), 178–193.
Cohen, M. H., & Turnbull, D. (1959). Molecular transport in liquids and glasses. The Journal of Chemical Physics, 31(5), 1164–1169.
da Rosa, G. S., Vanga, S. K., Gariepy, Y., & Raghavan, V. (2020). Development of biodegradable films with improved antioxidant properties based on the addition of carrageenan containing olive leaf extract for food packaging applications. Journal of Polymers and the Environment, 28(1), 123–130. https://doi.org/10.1007/s10924-019-01589-7
da Silva Pereira, G. V., da Silva Pereira, G. V., Xavier Neves, E. M. P., Albuquerque, G. A., de Arimatéia Rodrigues do Rêgo, J., Cardoso, D. N. P., & Joele, M. R. S. P. (2021). Effect of the mixture of polymers on the rheological and technological properties of composite films of acoupa weakfish (Cynoscion acoupa) and cassava starch (Manihot esculenta C.). Food and Bioprocess Technology, 14(7), 1199–1215. https://doi.org/10.1007/s11947-021-02622-1
de Vargas, V. H., Marczak, L. D. F., Flôres, S. H., & Mercali, G. D. (2022). Advanced technologies applied to enhance properties and structure of films and coatings: a review. Food and Bioprocess Technology, 1–24.
Drusch, S., Serfert, Y., Berger, A., Shaikh, M., Rätzke, K., Zaporojtchenko, V., & Schwarz, K. (2012). New insights into the microencapsulation properties of sodium caseinate and hydrolyzed casein. Food Hydrocolloids, 27(2), 332–338.
Du, H., Liu, C., Unsalan, O., Altunayar-Unsalan, C., Xiong, S., Manyande, A., & Chen, H. (2021). Development and characterization of fish myofibrillar protein/chitosan/rosemary extract composite edible films and the improvement of lipid oxidation stability during the grass carp fillets storage. International Journal of Biological Macromolecules, 184, 463–475. https://doi.org/10.1016/j.ijbiomac.2021.06.121
Dutta, D., & Sit, N. (2023). Application of natural extracts as active ingredient in biopolymer based packaging systems. Journal of Food Science and Technology, 60(7), 1888–1902. https://doi.org/10.1007/s13197-022-05474-5
Eldrup, M., Lightbody, D., & Sherwood, J. N. (1981). The temperature dependence of positron lifetimes in solid pivalic acid. Chemical Physics, 63(1–2), 51–58.
Fabra, M. J., Falcó, I., Randazzo, W., Sánchez, G., & López-Rubio, A. (2018). Antiviral and antioxidant properties of active alginate edible films containing phenolic extracts. Food Hydrocolloids, 81, 96–103.
Garavand, F., Rouhi, M., Razavi, S. H., Cacciotti, I., & Mohammadi, R. (2017). Improving the integrity of natural biopolymer films used in food packaging by crosslinking approach: A review. International Journal of Biological Macromolecules, 104, 687–707. https://doi.org/10.1016/j.ijbiomac.2017.06.093
Grzebieniarz, W., Biswas, D., Roy, S., & Jamróz, E. (2023). Advances in biopolymer-based multi-layer film preparations and food packaging applications. Food Packaging and Shelf Life, 35, 101033. https://doi.org/10.1016/j.fpsl.2023.101033
Gulati, M., & P.S., K. M., & Reddy, J. P. (2023). Effect of onion peel extract on structural, mechanical, thermal, and antioxidant properties of methylcellulose films. Food and Bioprocess Technology. https://doi.org/10.1007/s11947-023-03054-9
Hernández, V., Ibarra, D., Triana, J. F., Martínez-Soto, B., Faúndez, M., Vasco, D. A., & Garmulewicz, A. (2022). Agar biopolymer films for biodegradable packaging: a reference dataset for exploring the limits of mechanical performance. Materials, 15(11), 3954.
Izzi, Y. S., Gerschenson, L. N., Jagus, R. J., & Ollé Resa, C. P. (2023). Edible films based on tapioca starch and WPC or gelatine plasticized with glycerol: Potential food applications based on their mechanical and heat-sealing properties. Food and Bioprocess Technology. https://doi.org/10.1007/s11947-023-03089-y
Jamróz, E., Cabaj, A., Tkaczewska, J., Kawecka, A., Krzyściak, P., Szuwarzyński, M., & Juszczak, L. (2022a). Incorporation of curcumin extract with lemongrass essential oil into the middle layer of triple-layered films based on furcellaran/chitosan/gelatin hydrolysates - In vitro and in vivo studies on active and intelligent properties. Food Chemistry, 476. https://doi.org/10.1016/j.foodchem.2022.134476
Jamróz, E., Janik, M., Juszczak, L., Kruk, T., Kulawik, P., Szuwarzyński, M., & Khachatryan, K. (2021). Composite biopolymer films based on a polyelectrolyte complex of furcellaran and chitosan. Carbohydrate Polymers, 118627. https://doi.org/10.1016/j.carbpol.2021.118627
Jamróz, E., Janik, M., Marangoni, L., Vieira, R. P., Tkaczewska, J., Kawecka, A., & Juszczak, L. (2022b). Double-layered films based on furcellaran, chitosan, and gelatin hydrolysates enriched with AgNPs in yerba mate extract, montmorillonite, and curcumin with rosemary essential oil. Polymers, 14.
Jamróz, E., Khachatryan, G., Kopel, P., Juszczak, L., Kawecka, A., Krzyściak, P., & Zimowska, M. (2020). Furcellaran nanocomposite films: The effect of nanofillers on the structural, thermal, mechanical and antimicrobial properties of biopolymer films. Carbohydrate Polymers, 116244. https://doi.org/10.1016/j.carbpol.2020.116244
Jamróz, E., Kopel, P., Juszczak, L., Kawecka, A., Bytesnikova, Z., Milosavljević, V., & Adam, V. (2018). Development and characterisation of furcellaran-gelatin films containing SeNPs and AgNPs that have antimicrobial activity. Food Hydrocolloids, 83, 9–16. https://doi.org/10.1016/j.foodhyd.2018.04.028
Jamróz, E., Kopel, P., Juszczak, L., Kawecka, A., Bytesnikova, Z., Milosavljevic, V., & Makarewicz, M. (2019). Development of furcellaran-gelatin films with Se-AgNPs as an active packaging system for extension of mini kiwi shelf life. Food Packaging and Shelf Life, 21, 100339. https://doi.org/10.1016/j.fpsl.2019.100339
Jamróz, E., Tkaczewska, J., Juszczak, L., Zimowska, M., Kawecka, A., Krzyściak, P., & Skóra, M. (2022c). The influence of lingonberry extract on the properties of novel, double-layered biopolymer films based on furcellaran, CMC and a gelatin hydrolysate. Food Hydrocolloids, 124, 107334.
Ju, A., Baek, S.-K., Kim, S., & Song, K. B. (2019a). Development of an antioxidative packaging film based on khorasan wheat starch containing moringa leaf extract. Food Science and Biotechnology, 28(4), 1057–1063. https://doi.org/10.1007/s10068-018-00546-9
Ju, T., Zhang, Z., Li, Y., Miao, X., & Ji, J. (2019b). Continuous production of lignin nanoparticles using a microchannel reactor and its application in UV-shielding films. RSC Advances, 9(43), 24915–24921.
Júnior, M. (2021). Sustainable packaging films composed of sodium alginate and hydrolyzed collagen: Preparation and characterization. Food and Bioprocess Technology, 14(12), 2336–2346. https://doi.org/10.1007/s11947-021-02727-7
Kansy, J. (1996). Nuclear Instruments and Methods in Physics Research Section A, 374, 235–244.
Kavoosi, G., Rahmatollahi, A., Dadfar, M. M., & S., & Mohammadi Purfard, A. (2014). Effects of essential oil on the water binding capacity, physico-mechanical properties, antioxidant and antibacterial activity of gelatin films. LWT - Food Science and Technology, 57(2), 556–561. https://doi.org/10.1016/j.lwt.2014.02.008
Kawecka, A., & Cholewa-Wójcik, A. (2017). Jakość opakowania jako determinanta bezpieczeństwa żywności w kontekście wymagań społecznych konsumentów. Żywność Nauka Technologia Jakość, 24(3).
Khan, A., Priyadarshi, R., Bhattacharya, T., & Rhim, J.-W. (2023). Carrageenan/alginate-based functional films incorporated with Allium sativum carbon dots for UV-barrier food packaging. Food and Bioprocess Technology. https://doi.org/10.1007/s11947-023-03048-7
Kumar, L., Ramakanth, D., Akhila, K., & Gaikwad, K. K. (2022). Edible films and coatings for food packaging applications: A review. Environmental Chemistry Letters, 20(1), 875–900. https://doi.org/10.1007/s10311-021-01339-z
Kurkina, A. V., Khusainova, A. I., Daeva, E. D., & Kadentsev, V. I. (2011). Flavonoids from tanacetum vulgare flowers. Chemistry of Natural Compounds, 47(2), 284–285. https://doi.org/10.1007/s10600-011-9906-4
Makarova, A. O., Derkach, S. R., Kadyirov, A. I., Ziganshina, S. A., Kazantseva, M. A., Zueva, O. S., & Zuev, Y. F. (2022). Supramolecular structure and mechanical performance of κ-carrageenan–gelatin Gel. Polymers, 14.
Marangoni, L., Vieira, R. P., Jamróz, E., & Anjos, C. A. R. (2020). Furcellaran: an innovative biopolymer in the production of films and coatings. Carbohydrate Polymers, 117221. https://doi.org/10.1016/j.carbpol.2020.117221
Medina-Jaramillo, C., Ochoa-Yepes, O., Bernal, C., & Famá, L. (2017). Active and smart biodegradable packaging based on starch and natural extracts. Carbohydrate Polymers, 176, 187–194. https://doi.org/10.1016/j.carbpol.2017.08.079
Mouhoub, A., Guendouz, A., El Alaoui-Talibi, Z., Ibnsouda Koraichi, S., Delattre, C., & El Modafar, C. (2023). Development of food packaging with desirable properties and activities using chitosan and Mentha piperita, Salvia officinalis, Melaleuca quinquenervia, and Eucalyptus globulus EOs. Food and Bioprocess Technology. https://doi.org/10.1007/s11947-023-03106-0
Munir, S., Hu, Y., Liu, Y., & Xiong, S. (2019). Enhanced properties of silver carp surimi-based edible films incorporated with pomegranate peel and grape seed extracts under acidic condition. Food Packaging and Shelf Life, 19, 114–120. https://doi.org/10.1016/j.fpsl.2018.12.001
Neethirajan, S., & Jayas, D. S. (2011). Nanotechnology for the food and bioprocessing industries. Food and Bioprocess Technology, 4(1), 39–47. https://doi.org/10.1007/s11947-010-0328-2
Nocoń-Szmajda, K., Wolińska-Grabczyk, A., Jankowski, A., Dryzek, J., Dryzek, E., Janeczek, H., & Musioł, M. (2021). Effects of ionic liquid doping on gas transport properties of thermally rearranged poly. Separation and Purification Technology, 254, 117664.
Ortiz-Duarte, G. (2021). Evaluation of biopolymer films containing silver–chitosan nanocomposites. Food and Bioprocess Technology, 14(3), 492–504. https://doi.org/10.1007/s11947-021-02585-3
Pérez, M. J., Moreno, M. A., Martínez-Abad, A., Cattaneo, F., Zampini, C., Isla, M. I., & Fabra, M. J. (2021). Interest of black carob extract for the development of active biopolymer films for cheese preservation. Food Hydrocolloids, 113, 106436.
Reineccius, G. A., & Yan, C. (2016). Factors controlling the deterioration of spray dried flavourings and unsaturated lipids. Flavour and Fragrance Journal, 31(1), 5–21.
Ribeiro, A. M., Estevinho, B. N., & Rocha, F. (2021). Preparation and incorporation of functional ingredients in edible films and coatings. Food and Bioprocess Technology, 14(2), 209–231. https://doi.org/10.1007/s11947-020-02528-4
Saini, A., Sharma, D., Xia, Y., Saini, A., You, X., Su, Y., & Li, X. (2021). Layer-by-layer assembly of cationic guar gum, cellulose nanocrystals and hydroxypropyl methylcellulose based multilayered composite films. Cellulose, 28(13), 8445–8457. https://doi.org/10.1007/s10570-021-04064-6
Salim, M. H., Kassab, Z., Abdellaoui, Y., Cruz, G., & A., Soumare, A., Ablouh, E.-H., & El Achaby, M. (2022). Exploration of multifunctional properties of garlic skin derived cellulose nanocrystals and extracts incorporated chitosan biocomposite films for active packaging application. International Journal of Biological Macromolecules, 210, 639–653. https://doi.org/10.1016/j.ijbiomac.2022.04.220
Schaefer, E. W., Pavoni, J. M. F., Luchese, C. L., Faccin, D. J. L., & Tessaro, I. C. (2020). Influence of turmeric incorporation on physicochemical, antimicrobial and mechanical properties of the cornstarch and chitosan films. International Journal of Biological Macromolecules, 148, 342–350.
Serfert, Y., Schröder, J., Mescher, A., Laackmann, J., Shaikh, M., Rätzke, K., & Moritz, H.-U. (2013). Characterization of the spray drying behaviour of emulsions containing oil droplets with a structured interface. Journal of microencapsulation, 30(4), 325–334.
Shakouri, M., Salami, M., Lim, L.-T., Ekrami, M., Mohammadian, M., Askari, G., & McClements, D. J. (2023). Development of active and intelligent colorimetric biopolymer indicator: anthocyanin-loaded gelatin-basil seed gum films. Journal of Food Measurement and Characterization, 17(1), 472–484. https://doi.org/10.1007/s11694-022-01640-7
Silva-Weiss, A., Ihl, M., Sobral, P. J. A., Gómez-Guillén, M. C., & Bifani, V. (2013). Natural additives in bioactive edible films and coatings: Functionality and applications in foods. Food Engineering Reviews, 5(4), 200–216. https://doi.org/10.1007/s12393-013-9072-5
Siti Imama, K., Mohammad, M., Sutiman Bambang, S., & Sri, W. (2020). The UV-Vis spectrum of antioxidant complex from frozen-dried egg white protein and tomato extract. Berkala Penelitian Hayati, 25(2). https://doi.org/10.23869/110
Souza, V. G. L., Fernando, A. L., Pires, J. R. A., Rodrigues, P. F., Lopes, A. A. S., & Fernandes, F. M. B. (2017). Physical properties of chitosan films incorporated with natural antioxidants. Industrial Crops and Products, 107, 565–572. https://doi.org/10.1016/j.indcrop.2017.04.056
Sun, L., Sun, J., Chen, L., Niu, P., Yang, X., & Guo, Y. (2017). Preparation and characterization of chitosan film incorporated with thinned young apple polyphenols as an active packaging material. Carbohydrate Polymers, 163, 81–91.
Tagrida, M., Nilsuwan, K., Gulzar, S., Prodpran, T., & Benjakul, S. (2023). Fish gelatin/chitosan blend films incorporated with betel (Piper betle L.) leaf ethanolic extracts: characteristics, antioxidant and antimicrobial properties. Food Hydrocolloids, 137, 108316. https://doi.org/10.1016/j.foodhyd.2022.108316
Tao, B., Zhao, W., Lin, C., Yuan, Z., He, Y., Lu, L., & Cai, K. (2020). Surface modification of titanium implants by ZIF-8@Levo/LBL coating for inhibition of bacterial-associated infection and enhancement of in vivo osseointegration. Chemical Engineering Journal, 390, 124621. https://doi.org/10.1016/j.cej.2020.124621
Vaziri, A. S., Sattari, A., & Alemzadeh, I. (2022). Plant-derived biopolymers in food packaging: current status and market potential. In A. DuttTripathi, K. K. Darani, D. C. Rai, & V. Paul (Eds.), Biodegradable Polymer-Based Food Packaging (pp. 13–40). Singapore: Springer Nature Singapore.
Verónica, C. M., Julia, T. M., & M, S. A., Carina, A. M., & Alejandra, B. M. (2023). Antibacterial activity of Bacillus lipopeptides vehiculized and delivered by biopolymeric films. Food and Bioprocess Technology. https://doi.org/10.1007/s11947-023-03139-5
Voon, H. C., Bhat, R., Easa, A. M., Liong, M. T., & Karim, A. A. (2012). Effect of addition of halloysite nanoclay and SiO2 nanoparticles on barrier and mechanical properties of bovine gelatin films. Food and Bioprocess Technology, 5(5), 1766–1774. https://doi.org/10.1007/s11947-010-0461-y
Wang, C., Gong, C., Qin, Y., Hu, Y., Jiao, A., Jin, Z., & Wang, J. (2022). Bioactive and functional biodegradable packaging films reinforced with nanoparticles. Journal of Food Engineering, 312, 110752.
Wang, Y., Nakanishi, H., Jean, Y., & Sandreczki, T. (1990). Positron annihilation in amine-cured epoxy polymers—pressure dependence. Journal of Polymer Science Part B: Polymer Physics, 28(9), 1431–1441.
Xu, D., Chen, T., & Liu, Y. (2021). The physical properties, antioxidant and antimicrobial activity of chitosan–gelatin edible films incorporated with the extract from hop plant. Polymer Bulletin, 78(7), 3607–3624. https://doi.org/10.1007/s00289-020-03294-1
Yao, Y., Wang, H., Wang, R., Chai, Y., & Ji, W. (2019). Fabrication and performance characterization of the membrane from self-dispersed gelatin-coupled cellulose microgels. Cellulose, 26(5), 3255–3269. https://doi.org/10.1007/s10570-019-02263-w
Zając, M., Jamróz, E., Guzik, P., Kulawik, P., & Tkaczewska, J. (2021). Active biopolymer films based on furcellaran, whey protein isolate and Borago officinalis extract: Characterization and application in smoked pork ham production. Journal of the Science of Food and Agriculture, 101(7), 2884–2891. https://doi.org/10.1002/jsfa.10920
Author information
Authors and Affiliations
Contributions
Nikola Nowak, data curation, investigation, methodology, visualisation, writing—original draft, writing—review and editing; Wiktoria Grzebieniarz, investigation, methodology; Agnieszka Cholewa-Wójcik, conceptualization, investigation, methodology; Lesław Juszczak, investigation, methodology; Anna Konieczna-Molenda, investigation, methodology; Ewa Dryzek, investigation, methodology; Maciej Sarnek, investigation, methodology; Michał Szuwarzyński, investigation, methodology; Tomasz Mazur, investigation, methodology; Ewelina Jamróz, supervision, review, editing.
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Nowak, N., Grzebieniarz, W., Cholewa-Wójcik, A. et al. Effects of Selected Plant Extracts on the Quality and Functional Properties of Gelatin and Furcellaran-Based Double-Layer Films. Food Bioprocess Technol 17, 1201–1214 (2024). https://doi.org/10.1007/s11947-023-03190-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11947-023-03190-2