Abstract
Purpose of Review
To review the methods of improving surgical, oncological, and functional outcomes in women with bladder cancer treated with radical cystectomy.
Recent Findings
Οrthotopic urinary diversion (ONB) is a safe option for well-selected women as it combines high rates of daytime and nighttime continence with exceptional oncologic outcomes. It is considered safe even for patients with limited lymph node disease and trigone involvement, as long as a preoperative biopsy of the bladder neck or an intraoperative frozen section analysis of distal urethral margin rules out malignant disease. Nerve-sparing techniques have shown promising results. For well-selected patients with early invasive disease, sparing of internal genitalia has proven to be oncologically safe. Yet, generally accepted and evidence-based oncological and functional follow-up schemes for women after radical cystectomy are still lacking.
Summary
Properly designed prospective studies are needed with adequate number of participants in order to safely conclude about a broader use of pelvic organ-sparing cystectomy.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
According to epidemiological studies, bladder cancer (BCa) ranks ninth among all malignancies while it is considered to be the 13th most common cause of cancer-related death in the world [1, 2]. With regard to histologic classification, urothelial carcinoma predominates in the developed countries of Europe and North America, with non-urothelial histologies being more common in other regions, such as the Middle East.
Two studies reported that over a period of two decades (1985–2005), the diagnosis of BCa in the USA increased by more than 50%, while from 1975 to 1996 the 5-year survival rate increased by 6% [3, 4]. Similarly, there is a downward trend of mortality rates among several European nations, with the exception of some eastern European countries [5].
Typically, BCa is diagnosed in older individuals with the mean age of diagnosis being 69 years for men and 71 years for women [6]. In terms of incidence, the diagnosis was found to be three to four times less common in women than in men [7, 8] but at the same time women are more likely to receive a diagnosis of locally advanced disease [6, 8,9,10,11].
So far, cigarette smoking has been shown to be the most common risk factor for BCa development [12]. A number of occupational exposures such as dye and petroleum products have also been linked to BCa [13, 14] but to a lesser extent than smoking [15, 16]. It is worth mentioning that even controlled for exposure to smoking and occupational hazards, the diagnosis of the disease is still far more common in men [17].
Interestingly, while female gender is considered to be protective in terms of cancer-specific survival for most types of cancer, this is not the case for women suffering from BCa, since they have been found to experience higher mortality than men [6, 9, 10, 18,19,20]. One possible explanation could be the aforementioned advanced stage at the time of diagnosis in women, though in a study published by Mungan et al., female patients were shown to have worse outcomes across all stages of the disease when compared to men [11]. During the last decade, several studies trying to shed some light on the possible causes of gender disparities reported that women with hematuria are more likely to receive a false diagnosis of urinary tract infection [21], thus missing the opportunity to undergo a prompt and complete hematuria evaluation [22,23,24]. According to Hollenbeck et al. a subsequent delay in the diagnosis of BCa is clearly associated with a higher cancer-specific mortality [25].
Preoperative Aspects of Orthotopic Diversions in Females
Surgical Considerations
Nowadays, radical cystectomy with bilateral pelvic lymhadenectomy has been established as the gold standard therapy for patients with localized (cT2N0M0) or locally advanced (cT3a-T4a, N0–Nx, M0) BCa, since it provides excellent locoregional control of the disease [26, 27]. It has been more than 150 years since ureterosigmoidostomy was introduced as the first mechanism of urinary diversion following cystectomy [28]. After almost a century, it was replaced by the ileal conduit described by Bricker [29], but it was not until the early 1990s that urologists developed ileal neobladder to allow urine storage and voiding through the native urethra [30, 31]. While it was initially limited to male patients [30], increasing experience and improved understanding of the female pelvic anatomy contributed to orthotopic neobladder becoming an option for many women undergoing radical cystectomy [32,33,34,35]. It was of utmost importance that several studies demonstrated the oncological safety of orthotopic diversion in carefully selected female patients, since the fear of cancer cells spreading to the female urethra made that procedure initially unavailable to women [36, 37].
Before proceeding with the operation, the surgeon should have a thorough discussion with the patient in order to decide which type of urinary diversion would yield the best oncological and functional outcomes taking into consideration several patient-centered factors such as biological age, manual dexterity, and history of radiotherapy [38•, 39]. Regarding the ideal timeline that should be followed, the operation should take place within 12 weeks from the diagnosis (based on data from the SEER-Medicare database) [40], since any delay could negatively affect long-term survival [41].
Age
As expected, age of the patient plays a major role in the selection of the appropriate urinary diversion technique after radical cystectomy. According to a study published by Aghazadeh et al., advanced age is linked to complications occurring both during the procedure and the following period of recovery [42]. Subsequent studies reported that although disease-specific survival was similar among different age groups, advanced age was associated with increased burden of postoperative complications [43,44,45]. The above findings provide an explanation why elderly patients are more often offered ileal conduit as the preferred type of urinary diversion instead of an orthotopic neobladder [27, 46]. In 2010, Stenzl et al. concluded that older patients will finally achieve similar continence rates with their younger counterparts, although this may take longer [47]. Metcalfe et al. reported that younger age is independently associated with better quality of life in patients undergoing radical cystectomy [48]. On the other hand, according to the findings of a recent meta-analysis, advanced age is not an independent parameter affecting quality of life when ileal conduit is compared to orthotopic neobladder [49]. Taken together, it is suggested that the patient selection should be based on surgical fitness and functional status, since biological rather than chronological age seems to be closely related to the final outcome [50, 51].
Manual Dexterity
Every time a surgeon considers one of her or his patients for orthotopic diversion, a thorough discussion about the possible need for clean intermittent self-catheterization (CISC) during the postoperative period is mandatory [52]. Patients should be aware about the efforts required during the rehabilitation period and that they should have sufficient manual dexterity in order to master CIC if needed. In a series of 121 female patients with neobladder substitution, more than half of them required periodic CISC after surgery [53]. Similar results were obtained by one of the largest urethra-sparing cystectomy series to date [37]. All of the above underscore the need for evaluation of manual dexterity before proceeding with the operation, having in mind that most of the patients with muscle-invasive bladder cancer (MIBC) are expected to experience significant mental and physical decline before and after the diagnosis [54].
Anterior Exenteration and Pelvic Lymphadenectomy
Anterior pelvic exenteration is considered to be the standard of care when treating MIBC and recurrent high-grade urothelial tumors [27]. When performed in female patients, the technique involves removal of the bladder, urethra, distal ureters, uterus, fallopian tubes, ovaries, and anterior vaginal wall [27, 37, 55,56,57].
By definition a radical cystectomy should also include a meticulous bilateral pelvic lymph node dissection (PLND) [27, 58,59,60,61]. The extent of PLND is directly related to the perioperative morbidity and survival rates after radical cystectomy [62]. The treating surgeon should also have in mind that an extended PLND may improve 5-year progression-free survival [58, 63]. Nevertheless, there is no commonly accepted threshold regarding the number of lymph nodes that need to be removed, since there are considerable differences among individuals [59, 62, 64]. Summarizing, an extended PLND is defined as the removal of all lymphatic tissue from the common iliac vessels cranially to the genitofemoral nerve laterally, the circumflex iliac vein and node of Cloquet distally, the hypogastric vessels along with the obturator fossa medially, and presacral nodes posteriorly [65].
Vaginal Sparing Approach
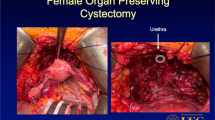
Over the years, efforts to achieve the best possible combination between oncological outcomes and postoperative quality of life have gained increasing attention. The introduction and wide acceptance of orthotopic neobladder has motivated many urologists to experiment on less radical surgical techniques, which are commonly known as pelvic organ-sparing techniques. Historically, pelvic organ-sparing radical cystectomy (POSRC) was initially applied to younger and sexually active men [66]. It was not until the early 2000s that these particular techniques were first used in female patients, after several studies showed that sparing the urethra during RC is oncologically safe [67, 68]. In women undergoing RC, organ-sparing includes preservation of the uterus, anterior vaginal wall, fallopian tubes, and ovaries. It has already been reported that the sparing of the female internal genitalia has a positive impact on postoperative voiding pattern and surgical outcomes following an orthotopic bladder reconstruction [69,70,71,72,73]. Additionally, the preservation of the anterior vaginal wall or interposition of an omental flap between the vagina and the neobladder decreases the risk of a future fistula [52].
Nevertheless, the treating surgeon should never exaggerate with his efforts to achieve a better functional outcome at the expense of oncological results. In several studies including female patients undergoing anterior exenteration, it was found that the local invasion of internal genitalia ranges between 2.7 and 7.5% [74,75,76,77,78,79,80]. According to Djaladat et al. [80], the vagina is the most common location of invasion and it is commonly associated with tumors located at the base of the bladder. This means that the surgeon should perform a meticulous preoperative evaluation of the candidate for POSRC and be ready to abandon organ-sparing with the slightest suspicion of pelvic organ involvement, since a positive surgical margin at RC has been associated with a decreased 5-year survival of only 32% [81].
Urethra-Sparing Approach
Since the preservation of the urethra is a prerequisite for an orthotopic bladder reconstruction, a lot of research has been done in order to establish inclusion criteria for candidates for a urethra-sparing approach. Until the 1990s, en bloc urethrectomy was a common practice for women because of the fear for urethral tumor involvement [82] and the presumption that the remaining urethra in case of partial urethrectomy lacks function [83]. Nowadays, an absolute indication for urethrectomy is a positive urethral surgical margin during RC. Several studies on female population showed that urethral involvement ranges from 2 to 16%, with the major risk factors being location of tumor in the bladder neck and invasion of the anterior wall of the vagina [35, 84, 85]. So far, the use of frozen section analysis intraoperatively has gained wide acceptance as a factor determining whether the surgeon should proceed with the initial plan of orthotopic reconstruction or instead perform a urethrectomy and cutaneous diversion [86,87,88]. In a study published by Osman et al. in 2012, frozen section analysis had a specificity of 99% and sensitivity of 90% in detecting urethral invasion when compared to the gold standard of the final histopathologic diagnosis [89], which confirms previous findings defining frozen section analysis as a valid method of identifying best candidates for orthotopic urinary diversion [36].
Functional Aspects of Orthotopic Neobladder Reconstruction in Females
Continence-Urinary Retention
The ultimate goal of an orthotopic bladder reconstruction is to provide a low-pressure space for urine storage, which allows the patient to maintain a satisfactory level of daytime and nighttime continence without the need for intermittent catheterization.
Based on data from early studies in female patients with orthotopic neobladder, more than 60% of them were able to void spontaneously after the surgery, while the rates of daytime and nighttime continence were 80% and 65–70% respectively [37, 90,91,92,93,94]. In 2012, Anderson et al. reported that daytime incontinence was associated with preceding hysterectomy, while the severity was closely related to the presence of stress incontinence prior to the surgery [95]. In fact, an analysis of urethral pressure profiles confirmed the hypothesis that patients without hysterectomy, thus with less distortion of pelvic anatomy, experience higher rates of continence when compared to their hysterectomized counterparts [96]. According to Rouanne et al., the only factor predicting daytime incontinence in a population of 41 female patients was found to be the age > 65 years [97]. While more than half of the patients experience nighttime incontinence in the immediate postoperative period [95], many of them are expected to have a significant improvement as the neobladder can store more urine over time [98]. Moreover, nerve-sparing techniques besides yielding better daytime continence rates are directly associated to an improvement of nighttime continence by 5 to 25% [34, 99, 100].
When it comes to compare POSRC with the classical radical cystectomy, most authors agree that organ-sparing is linked to higher rates of continence [56, 72, 73, 94]. In two large series of patients who were offered an organ-sparing procedure, the rates of both day- and nighttime continence ranged between 80 and 100% [72, 73]. Nevertheless, we should have in mind that in the case of POSRC, most of the patients have an excellent baseline function and are highly motivated, which could be a bias when attempting a direct comparison with the classical RC.
Several studies conducted in animals stressed the importance of preserving the branches of the pudendal nerve and pelvic plexus in order to preserve the mechanism of continence [101, 102]. In 2007, Bhatta Dhar et al. emphasized the need for fine handling of autonomic nerves passing medial to the ureters and crossing the uterine and vaginal arteries as well the paravaginal region [99]. Of equal importance when performing a nerve-sparing cystectomy is the preservation of somatic nerve bundles originating from S2–4 levels of the spinal cord and running in close proximity with the inferior bladder pedicle [103].
One problem that is significantly more common among women with orthotopic neobladder is that of hypercontinence. In 2002, after performing video-urodynamic investigation in a female population with orthotopic neobladder, Ali-el-Dein et al. concluded that hypercontinence should be attributed to anatomical rather than functional reasons [104]. So far, a possible kinking of the vesicourethral anastomosis, strictures in the anastomosis, pouchocele, and dyssynergia of pelvic floor muscles have been proposed as possible mechanisms responsible for hypercontinence. Interestingly, Gross et al. characterized continence as a dynamic process after finding that many of the patients with urinary retention had a previous normal voiding period of 3–6 months. The above findings were attributed to neuropraxia causing denervation of the proximal urethra during the early post-operative period [96]. In a large retrospective study from Egypt, 16% of the study population reported postoperative hypercontinence [105]. On the contrary, there are other studies with much higher rates of hypercontinence among their patients, like that of Bartsch et al. who reported urinary retention affecting 45% of their study population [106]. Such heterogeneity in findings is most likely due to variable assessment of continence via a patient interview instead of using validated questionnaires. Finally, we should mention that several studies focusing on POPRC have reported rates of hypercontinence that range between 0 and 3% [72, 73, 107, 108].
Sexual Function
Sexual dysfunction is a major, although usually overlooked, factor causing significant distress in the lives of cystectomized patients [109,110,111]. Therefore, it is of utmost importance that the treating surgeon has a thorough discussion on the patient’s expectations and on the realistic outcomes after a radical cystectomy [112], since most of sexually active women would like to preserve a good quality of sexual life after the operation [97]. It is self-understood that the surgeon is asked to achieve a balance between excellent oncologic outcomes and preservation of those anatomic structures that would guarantee a satisfactory sexual function.
For reference purposes, sexual dysfunction following RC in female patients is grouped into sexual pain disorders (vulvodynia, dyspareunia, vaginismus) and sexual arousal disorders secondary to devascularization of the clitoris [113]. In 2004, Volkmer et al. [114] retrospectively reviewed the data of 29 female patients with orthotopic neobladder and found that the ability to achieve and maintain lubrication was the function with the greatest decline in the postoperative period as assessed by the use of the Female Sexual Function Index (FSFI) [115]. During the same period, Zippe et al. [116] published the results of a study following up 34 cystectomized women for a median period of 24 months and concluded that the most common complaint was a difficulty in reaching orgasm, while more than half of the participants (52%) complained of significantly decreased sexual satisfaction. Similarly, two subsequent studies reported rates of sexual dysfunction ranging from 46 to 59% [117, 118].The aforementioned disorders are mainly attributed to a possible injury of the neurovascular bundles which follow a course along the lateral vaginal wall [34, 119] or a severe devascularization of the clitoris during transection of the urethra [119, 120]. According to a study released by Bhatt et al., patients who underwent neurovascular preservation during orthotopic cystectomy reported better results in every aspect of sexual function when compared non-neurovascular preservation counterparts [121]. That means that it is really important to carefully dissect pelvic structures and avoid opening the endopelvic fascia in order to preserve most of the autonomic nervous fibers that are critical for the preservation of both continence and sexual function. Regarding POSRC techniques, Ali-El-Dein et al. showed that genital sparing provides better sexual outcomes without compromising oncological results [73]. Towards the same direction, it has been proposed that when the anterior vaginal wall is removed, a reconstruction should be performed by retubularization of the posterior vaginal wall or by using bowel segments, flaps of rectus abdominis and gracilis, or skin grafts to form a neovagina [114, 122,123,124,125]. Of all the above, it is easily understood that although oncologic outcomes and safety are of paramount importance for these patients, efforts should not be precluded to improve their quality of life.
Risk of Urethral Recurrence
Since the advent of urethra-sparing techniques in women undergoing radical cystectomy, urologists have to confront the challenge of a new population of patients at risk for postoperative urethral recurrence (UR). According to a study published in 2013, male population is at a higher risk of developing UR [65]. This could be partially explained by the fact that the squamous metaplasia, present normally in postmenopausal women even up to the area of the bladder trigone, poses a barrier to the implantation of neoplastic urothelial cells.
A recent meta-analysis on a mixed population (both males and females) of 9498 individuals showed that pathological stage, non-orthotopic diversion, positive urethral margins, male gender, multifocality, and involvement of the bladder neck had a statistically significant positive correlation with the risk for UR [126]. Early series of female patients with orthotopic diversion report UR rates ranging from 0 to 2% [53, 56, 91, 127,128,129]. In 2012, a large single-institution study showed a UR rate equal to 0.8% after analyzing the data of 121 women with orthotopic neobladder and a median follow-up of 56 months [53]. In one of the largest series to date (1506 individuals) including both male and female patients, Boorjan et al. reported 85 cases of UR, which was three times more common in cutaneous when compared to orthotopic diversion [130]. A large multi-institutional study assessed the risk of UR in a female population of 297 individuals after orthotopic bladder reconstruction and showed that only 2.4% of them developed a UR during the follow-up period [131•]. Authors of the same study reported that lymph node metastasis harbors a higher risk for overall but not for UR, confirming the assumption that patients with limited lymph node disease should not be denied the opportunity to receive an orthotopic diversion. Finally, with regard to the time frame and symptomatology, cases of UR most commonly present with discharge and pain [130], while most of them are diagnosed within 3 years after the surgery [132, 133].
Follow-Up
It is common sense that every patient describing suspicious signs and symptoms like urethral bleeding and pain after orthotopic neobladder reconstruction should receive prompt evaluation in order to exclude a possible recurrence of the disease. So far, due to the relative rarity of UR, the urologic community has not reached a consensus as to what a proper follow-up scheme should include [133]. Current literature describes the urine cytology as the most common screening method for recurrence. According to Varol et al., cytology should be used twice a year for the first 2 years and once a year thereafter [134]. Another protocol suggests the use of urethroscopy, cytology, and urethral washings on a quarterly basis for 2 years and semiannually after that period [135]. Nevertheless, there are authors defying the diagnostic value of cytology in the case of urinary diversion [136], since deciduous and necrotic intestinal cells in the urine could be confused with neoplastic urothelial cells, while the time that the urine remains in the neobladder could also lead to anaplasia of normal urothelial cells. Moreover, it is critical to note that although urethroscopy and biopsy remains the gold standard for the diagnosis of recurrence, the use of urethroscopy as a screening tool on a regular basis is discouraged by many authors, since it does not seem to provide any benefit in terms of survival [133, 134, 137,138,139].
On the other hand, it is really important that the follow-up plan also includes periodic screening of the upper urinary tract, since 2–9% of patients undergoing radical cystectomy will develop metachronous recurrences after a median period of 24–41 months [140,141,142,143,144]. Although most of the patients are diagnosed with advanced disease predestinating a generally poor prognosis, an early detection in the context of a typical follow-up is associated with significantly higher survival rate [143]. Towards the direction of early diagnosis, most authors recommend periodic cytology [143, 145, 146], while others support the use of IVP at frequent intervals for all patients undergoing RC [147]. As expected, ureterorenoscopy with biopsy is the definitive method for the diagnosis of metachronous upper tract recurrence [148].
Finally, the urologist should also be aware of the medical complications following a radical cystectomy. This means that the follow-up should also include periodic screening for complications of chronic metabolic acidosis [149] as well as assessment for B12 deficiency after the second postoperative year [150].
Conclusions
Radical cystectomy has long since been established as the procedure of choice for muscle-invasive bladder cancer in both male and female patients. The reconstruction of the urinary tract is obviously a challenging process, fruit of the efforts of pioneer surgeons trying to maximize quality of life in this group of patients. So far, several studies confirmed that the orthotopic diversion is a safe option for carefully selected women as it combines high rates of day- and nighttime continence with exceptional oncologic outcomes. It is considered safe even for patients with limited lymph node disease and trigone involvement, as long as a preoperative biopsy of the bladder neck or an intraoperative frozen section analysis of distal urethral margin rules out malignancy. However, it is really important that every patient besides possessing adequate manual dexterity to perform self-catheterization upon request should also consent to a rather demanding rehabilitation process. Special efforts have been described towards preservation of sexual function, with nerve-sparing techniques yielding promising results. For well-selected patients, sparing of internal genitalia seems to be oncological safe with acceptable results. Nevertheless, we certainly need properly designed prospective studies with adequate number of participants in order to safely conclude about a broader use of POSRC. Finally, a lot of work needs to be done towards establishment of a commonly accepted follow-up scheme in order to allow treating surgeons to be more confident when dealing with their patients during the postoperative period.
References
Papers of particular interest, published recently, have been highlighted as: • Of importance •• Of major importance
Ferlay J, Soerjomataram I, Ervik M, et al. GLOBOCAN 2012 v1.0, cancer incidence and mortality worldwide: IARC CancerBase No. 11. International Agency for Research on Cancer: Lyon; 2013.
Torre LA, Bray F, Siegel RL, Ferlay J, Lortet-Tieulent J, Jemal A. Global cancer statistics, 2012. CA Cancer J Clin. 2015;65(2):87–108. https://doi.org/10.3322/caac.21262.
Jemal A, Murray T, Ward E, Samuels A, Tiwari RC, Ghafoor A, et al. Cancer statistics, 2005. CA Cancer J Clin. 2005;55(1):10–30.
Jemal A, Siegel R, Xu J, Ward E. Cancer statistics, 2010. CA Cancer J Clin. 2010;60(5):277–300. https://doi.org/10.3322/caac.20073.
Pelucchi C, Bosetti C, Negri E, Malvezzi M, La Vecchia C. Mechanisms of disease: the epidemiology of bladder cancer. Nat Clin Pract Urol. 2006;3(6):327–40.
Scosyrev E, Noyes K, Feng C, Messing E. Sex and racial differences in bladder cancer presentation and mortality in the US. Cancer. 2009;115(1):68–74. https://doi.org/10.1002/cncr.23986.
Siegel RL, Miller KD, Jemal A. Cancer statistics, 2015. CA Cancer J Clin. 2015;65(1):5–29. https://doi.org/10.3322/caac.21254.
Fajkovic H, Halpern JA, Cha EK, Bahadori A, Chromecki TF, Karakiewicz PI, et al. Impact of gender on bladder cancer incidence, staging, and prognosis. World J Urol. 2011;29(4):457–63. https://doi.org/10.1007/s00345-011-0709-9.
Messer JC, Shariat SF, Dinney CP, Novara G, Fradet Y, Kassouf W, et al. Female gender is associated with a worse survival after radical cystectomy for urothelial carcinoma of the bladder: a competing risk analysis. Urology. 2014;83(4):863–7. https://doi.org/10.1016/j.urology.2013.10.060.
Kluth LA, Rieken M, Xylinas E, Kent M, Rink M, Rouprêt M, et al. Gender-specific differences in clinicopathologic outcomes following radical cystectomy: an international multi-institutional study of more than 8000 patients. Eur Urol. 2014;66(5):913–9. https://doi.org/10.1016/j.eururo.2013.11.040.
Mungan NA, Kiemeney LA, van Dijck JA, van der Poel HG, Witjes JA. Gender differences in stage distribution of bladder cancer. Urology. 2000;55(3):368–71.
Strope SA, Montie JE. The causal role of cigarette smoking in bladder cancer initiation and progression, and the role of urologists in smoking cessation. J Urol. 2008;180(1):31–7. https://doi.org/10.1016/j.juro.2008.03.045 discussion 37.
Brown T, Slack R, Rushton L, British Occupational Cancer Burden Study Group. Occupational cancer in Britain. Urinary tract cancers: bladder and kidney. Br J Cancer. 2012;107(Suppl 1):S76–84. https://doi.org/10.1038/bjc.2012.121.
Samanic CM, Kogevinas M, Silverman DT, Tardón A, Serra C, Malats N, et al. Occupation and bladder cancer in a hospital-based case-control study in Spain. Occup Environ Med. 2008;65(5):347–53.
Jung I, Messing E. Molecular mechanisms and pathways in bladder cancer development and progression. Cancer Control. 2000;7:325–34.
Cole P, Hoover R, Friedell GH. Occupation and cancer of the lower urinary tract. Cancer. 1972;29(5):1250–60.
Hartge P, Harvey EB, Linehan WM, Silverman DT, Sullivan JW, Hoover RN, et al. Unexplained excess risk of bladder cancer in men. J Natl Cancer Inst. 1990;82(20):1636–40.
Afshar N, English DR, Thursfield V, Mitchell PL, Te Marvelde L, Farrugia H, et al. Differences in cancer survival by sex: a population-based study using cancer registry data. Cancer Causes Control. 2018;29(11):1059–69. https://doi.org/10.1007/s10552-018-1079-z.
Mungan NA, Aben KK, Schoenberg MP, Visser O, Coebergh JW, Witjes JA, et al. Gender differences in stage-adjusted bladder cancer survival. Urology. 2000;55(6):876–80.
Tracey E, Roder D, Luke C, Bishop J. Bladder cancer survivals in New South Wales, Australia: why do women have poorer survival than men? BJU Int. 2009;104(4):498–504. https://doi.org/10.1111/j.1464-410X.2009.08527.x.
Cohn JA, Vekhter B, Lyttle C, Steinberg GD, Large MC. Sex disparities in diagnosis of bladder cancer after initial presentation with hematuria: a nationwide claims-based investigation. Cancer. 2014;120(4):555–61. https://doi.org/10.1002/cncr.28416.
Garg T, Pinheiro LC, Atoria CL, Donat SM, Weissman JS, Herr HW, et al. Gender disparities in hematuria evaluation and bladder cancer diagnosis: a population based analysis. J Urol. 2014;192(4):1072–7. https://doi.org/10.1016/j.juro.2014.04.101.
Bassett JC, Alvarez J, Koyama T, Resnick M, You C, Ni S, et al. Gender, race, and variation in the evaluation of microscopic hematuria among Medicare beneficiaries. J Gen Intern Med. 2015;30(4):440–7. https://doi.org/10.1007/s11606-014-3116-2.
Lyratzopoulos G, Abel GA, McPhail S, Neal RD, Rubin GP. Gender inequalities in the promptness of diagnosis of bladder and renal cancer after symptomatic presentation: evidence from secondary analysis of an English primary care audit survey. BMJ Open. 2013;3:e002861.
Snyder C, Harlan L, Knopf K, Potosky A, Kaplan R. Patterns of care for the treatment of bladder cancer. J Urol. 2003;169(5):1697–701.
Hautmann RE, Gschwend JE, de Petriconi RC, Kron M, Volkmer BG. Cystectomy for transitional cell carcinoma of the bladder: results of a surgery only series in the neobladder era. J Urol. 2006;176(2):486–92 discussion 491-2.
Stein JP, Lieskovsky G, Cote R, Groshen S, Feng AC, Boyd S, et al. Radical cystectomy in the treatment of invasive bladder cancer: long-term results in 1,054 patients. J Clin Oncol. 2001;19(3):666–75.
Simon J. Operation for directing the ureteral orifices into the rectum, temporary success, subsequent death, autopsy. Lancet. 1852;2:568–70.
BRICKER EM. Bladder substitution after pelvic evisceration. Surg Clin North Am. 1950;30(5):1511–21.
Hautmann RE, Egghart G, Frohneberg D, Miller K. The ileal neobladder. J Urol. 1988;139(1):39–42.
Hautmann RE, Miller K, Steiner U, Wenderoth U. The ileal neobladder: 6 years of experience with more than 200 patients. J Urol. 1993;150(1):40–5.
Gschwend JE. Bladder substitution. Curr Opin Urol. 2003;13(6):477–82.
Stenzl A, Höltl L. Orthotopic bladder reconstruction in women—what we have learned over the last decade. Crit Rev Oncol Hematol. 2003;47(2):147–54.
Stenzl A, Colleselli K, Poisel S, Feichtinger H, Pontasch H, Bartsch G. Rationale and technique of nerve sparing radical cystectomy before an orthotopic neobladder procedure in women. J Urol. 1995;154(6):2044–9.
Stenzl A, Draxl H, Posch B, Colleselli K, Falk M, Bartsch G. The risk of urethral tumors in female bladder cancer: can the urethra be used for orthotopic reconstruction of the lower urinary tract? J Urol. 1995;153(3 Pt 2):950–5.
Stein JP, Penson DF, Wu SD, Skinner DG. Pathological guidelines for orthotopic urinary diversion in women with bladder cancer: a review of the literature. J Urol. 2007;178(3 Pt 1):756–60.
Stein JP, Penson DF, Lee C, Cai J, Miranda G, Skinner DG. Long-term oncological outcomes in women undergoing radical cystectomy and orthotopic diversion for bladder cancer. J Urol. 2009;181(5):2052–8; discussion 2058-9. https://doi.org/10.1016/j.juro.2009.01.020.
• Mischinger J, Abdelhafez MF, Todenhöfer T, Schwentner C, Aufderklamm S, Stenzl A, et al. Quality of life outcomes after radical cystectomy: long-term standardized assessment of Studer Pouch versus I-Pouch. World J Urol. 2015;33(10):1381–7. https://doi.org/10.1007/s00345-014-1461-8 A prospective study reporting on quality of life issues between refluxing and antirefluxing neobladder techniques.
Schilling D, Gakis G, Sievert KD, Stenzl A. Vordere Exenteration der Frau. Aktuelle Urol. 2015;46(1):72–81. https://doi.org/10.1055/s-0034-1545277.
Gore JL, Lai J, Setodji CM, Litwin MS. Saigal CS; Urologic Diseases in America Project. Mortality increases when radical cystectomy is delayed more than 12 weeks: results from a Surveillance, Epidemiology, and End Results-Medicare analysis. Cancer. 2009;115(5):988–96. https://doi.org/10.1002/cncr.24052.
Dalbagni G, Vora K, Kaag M, Cronin A, Bochner B, Donat SM, et al. Clinical outcome in a contemporary series of restaged patients with clinical T1 bladder cancer. Eur Urol. 2009;56(6):903–10. https://doi.org/10.1016/j.eururo.2009.07.005.
Aghazadeh MA, Barocas DA, Salem S, Clark PE, Cookson MS, Davis R, et al. Determining factors for hospital discharge status after radical cystectomy in a large contemporary cohort. J Urol. 2011;185(1):85–9. https://doi.org/10.1016/j.juro.2010.08.016.
Lughezzani G, Sun M, Shariat SF, Budäus L, Thuret R, Jeldres C, et al. A population-based competing-risks analysis of the survival of patients treated with radical cystectomy for bladder cancer. Cancer. 2011;117(1):103–9. https://doi.org/10.1002/cncr.25345.
Donat SM, Siegrist T, Cronin A, Savage C, Milowsky MI, Herr HW. Radical cystectomy in octogenarians—does morbidity outweigh the potential survival benefits? J Urol. 2010;183(6):2171–7. https://doi.org/10.1016/j.juro.2010.02.015.
Koppie TM, Serio AM, Vickers AJ, Vora K, Dalbagni G, Donat SM, et al. Age-adjusted Charlson comorbidity score is associated with treatment decisions and clinical outcomes for patients undergoing radical cystectomy for bladder cancer. Cancer. 2008;112(11):2384–92. https://doi.org/10.1002/cncr.23462.
Madersbacher S, Hochreiter W, Burkhard F, Thalmann GN, Danuser H, Markwalder R, et al. Radical cystectomy for bladder cancer today—a homogeneous series without neoadjuvant therapy. J Clin Oncol. 2003;21(4):690–6.
Stenzl A, Sherif H, Kuczyk M. Radical cystectomy with orthotopic neobladder for invasive bladder cancer: a critical analysis of long term oncological, functional and quality of life results. Int Braz J Urol. 2010;36(5):537–47.
Metcalfe M, Estey E, Jacobsen NE, Voaklander D, Fairey AS. Association between urinary diversion and quality of life after radical cystectomy. Can J Urol. 2013;20(1):6626–31.
Cerruto MA, D'Elia C, Siracusano S, Gedeshi X, Mariotto A, Iafrate M, et al. Systematic review and meta-analysis of non RCT’s on health related quality of life after radical cystectomy using validated questionnaires: better results with orthotopic neobladder versus ileal conduit. Eur J Surg Oncol. 2016;42(3):343–60. https://doi.org/10.1016/j.ejso.2015.10.001.
Sogni F, Brausi M, Frea B, Martinengo C, Faggiano F, Tizzani A, et al. Morbidity and quality of life in elderly patients receiving ileal conduit or orthotopic neobladder after radical cystectomy for invasive bladder cancer. Urology. 2008;71(5):919–23. https://doi.org/10.1016/j.urology.2007.11.125.
Clark PE, Stein JP, Groshen SG, Cai J, Miranda G, Lieskovsky G, et al. Radical cystectomy in the elderly: comparison of clinical outcomes between younger and older patients. Cancer. 2005;104(1):36–43.
Gakis G, Jentzmik F, Schrader M, Stenzl A, Sievert KD. Benefits and risks of orthotopic neobladder reconstruction in female patients. Aktuelle Urol. 2011;42(2):109–14. https://doi.org/10.1055/s-0031-1271413.
Jentzmik F, Schrader AJ, de Petriconi R, Hefty R, Mueller J, Doetterl J, et al. The ileal neobladder in female patients with bladder cancer: long-term clinical, functional, and oncological outcome. World J Urol. 2012;30(6):733–9. https://doi.org/10.1007/s00345-012-0837-x.
Fung C, Pandya C, Guancial E, Noyes K, Sahasrabudhe DM, Messing EM, et al. Impact of bladder cancer on health related quality of life in 1,476 older Americans: a cross-sectional study. J Urol. 2014;192(3):690–5. https://doi.org/10.1016/j.juro.2014.03.098.
Marshall FF, Treiger BF. Radical cystectomy (anterior exenteration) in the female patient. Urol Clin North Am. 1991;18(4):765–75.
Granberg CF, Boorjian SA, Crispen PL, Tollefson MK, Farmer SA, Frank I, et al. Functional and oncological outcomes after orthotopic neobladder reconstruction in women. BJU Int. 2008;102(11):1551–5. https://doi.org/10.1111/j.1464-410X.2008.07909.x.
Stenzl A, Nagele U, Kuczyk M, Sievert KD, Anastasiadis A, Seibold J, et al. Cystectomy - technical considerations in male and female patients. Eur Urol Suppl. 2005;3(3):138-46.
Leissner J, Ghoneim MA, Abol-Enein H, Thüroff JW, Franzaring L, Fisch M, et al. Extended radical lymphadenectomy in patients with urothelial bladder cancer: results of a prospective multicenter study. J Urol. 2004;171(1):139–44.
Dhar NB, Klein EA, Reuther AM, Thalmann GN, Madersbacher S, Studer UE. Outcome after radical cystectomy with limited or extended pelvic lymph node dissection. J Urol. 2008;179(3):873–8; discussion 878. https://doi.org/10.1016/j.juro.2007.10.076.
Steven K, Poulsen AL. Radical cystectomy and extended pelvic lymphadenectomy: survival of patients with lymph node metastasis above the bifurcation of the common iliac vessels treated with surgery only. J Urol. 2007;178(4 Pt 1):1218–23 discussion 1223-4.
Fleischmann A, Thalmann GN, Markwalder R, Studer UE. Extracapsular extension of pelvic lymph node metastases from urothelial carcinoma of the bladder is an independent prognostic factor. J Clin Oncol. 2005;23(10):2358–65.
Herr HW, Faulkner JR, Grossman HB, Natale RB. deVere White R, Sarosdy MF, et al. Surgical factors influence bladder cancer outcomes: a cooperative group report. J Clin Oncol. 2004;22(14):2781–9.
Bochner BH, Cho D, Herr HW, Donat M, Kattan MW, Dalbagni G. Prospectively packaged lymph node dissections with radical cystectomy: evaluation of node count variability and node mapping. J Urol. 2004;172(4 Pt 1):1286–90.
Koppie TM, Vickers AJ, Vora K, Dalbagni G, Bochner BH. Standardization of pelvic lymphadenectomy performed at radical cystectomy: can we establish a minimum number of lymph nodes that should be removed? Cancer. 2006;107(10):2368–74.
Gakis G, Efstathiou J, Lerner SP, Cookson MS, Keegan KA, Guru KA, et al. ICUD-EAU International Consultation on Bladder Cancer 2012: radical cystectomy and bladder preservation for muscle-invasive urothelial carcinoma of the bladder. Eur Urol. 2013;63(1):45–57. https://doi.org/10.1016/j.eururo.2012.08.009.
Spitz A, Stein JP, Lieskovsky G, Skinner DG. Orthotopic urinary diversion with preservation of erectile and ejaculatory function in men requiring radical cystectomy for nonurothelial malignancy: a new technique. J Urol. 1999;161(6):1761–4.
Ali-el-Dein B, el-Sobky E, Hohenfellner M, Ghoneim MA. Orthotopic bladder substitution in women: functional evaluation. J Urol. 1999;161(6):1875–80.
Coloby PJ, Kakizoe T, Tobisu K, Sakamoto M. Urethral involvement in female bladder cancer patients: mapping of 47 consecutive cysto-urethrectomy specimens. J Urol. 1994;152(5 Pt 1):1438–42.
Hautmann RE, Paiss T, de Petriconi R. The ileal neobladder in women: 9 years of experience with 18 patients. J Urol. 1996;155(1):76–81.
Blute ML, Gburek BM. Continent orthotopic urinary diversion in female patients: early Mayo Clinic experience. Mayo Clin Proc. 1998;73(6):501–7.
Nandipati KC, Bhat A, Zippe CD. Neurovascular preservation in female orthotopic radical cystectomy significantly improves sexual function. Urology. 2006;67(1):185–6.
Koie T, Hatakeyama S, Yoneyama T, Hashimoto Y, Kamimura N, Ohyama C. Uterus-, fallopian tube-, ovary-, and vagina-sparing cystectomy followed by U-shaped ileal neobladder construction for female bladder cancer patients: oncological and functional outcomes. Urology. 2010;75(6):1499–503. https://doi.org/10.1016/j.urology.2009.08.083.
Ali-El-Dein B, Mosbah A, Osman Y, El-Tabey N, Abdel-Latif M, Eraky I, et al. Preservation of the internal genital organs during radical cystectomy in selected women with bladder cancer: a report on 15 cases with long term follow-up. Eur J Surg Oncol. 2013;39(4):358–64. https://doi.org/10.1016/j.ejso.2013.02.004.
Chen ME, Pisters LL, Malpica A, Pettaway CA, Dinney CP. Risk of urethral, vaginal and cervical involvement in patients undergoing radical cystectomy for bladder cancer: results of a contemporary cystectomy series from M. D Anderson Cancer Center. J Urol. 1997;157(6):2120–3.
Groutz A, Gillon G, Konichezky M, Shimonov M, Winkler H, Livne PM, et al. Involvement of internal genitalia in female patients undergoing radical cystectomy for bladder cancer: a clinicopathologic study of 37 cases. Int J Gynecol Cancer. 1999;9(4):302–6.
Chang SS, Cole E, Smith JA Jr, Cookson MS. Pathological findings of gynecologic organs obtained at female radical cystectomy. J Urol. 2002;168(1):147–9.
Ali-El-Dein B, Abdel-Latif M, Mosbah A, Eraky I, Shaaban AA, Taha NM, et al. Secondary malignant involvement of gynecologic organs in radical cystectomy specimens in women: is it mandatory to remove these organs routinely? J Urol. 2004;172(3):885–7.
Varkarakis IM, Pinggera G, Antoniou N, Constantinides K, Chrisofos M, Deliveliotis C. Pathological review of internal genitalia after anterior exenteration for bladder cancer in women. Evaluating risk factors for female organ involvement. Int Urol Nephrol. 2007;39(4):1015–21.
Salem H, El-Mazny A. A clinicopathologic study of gynecologic organ involvement at radical cystectomy for bladder cancer. Int J Gynaecol Obstet. 2011;115(2):188–90. https://doi.org/10.1016/j.ijgo.2011.05.026.
Djaladat H, Bruins HM, Miranda G, Cai J, Skinner EC, Daneshmand S. Reproductive organ involvement in female patients undergoing radical cystectomy for urothelial bladder cancer. J Urol. 2012;188(6):2134–8. https://doi.org/10.1016/j.juro.2012.08.024.
Dotan ZA, Kavanagh K, Yossepowitch O, Kaag M, Olgac S, Donat M, et al. Positive surgical margins in soft tissue following radical cystectomy for bladder cancer and cancer specific survival. J Urol. 2007;178(6):2308–12 discussion 2313.
Schilling D, Horstmann M, Nagele U, Sievert KD, Stenzl A. Cystectomy in women. BJU Int. 2008;102(9 Pt B):1289–95. https://doi.org/10.1111/j.1464-410X.2008.07972.x.
Bell CR, Gujral S, Collins CM, Sibley GN, Persad RA. Review. The fate of the urethra after definitive treatment of invasive transitional cell carcinoma of the urinary bladder. BJU Int. 1999;83(6):607–12.
Maralani S, Wood DP Jr, Grignon D, Banerjee M, Sakr W, Pontes JE. Incidence of urethral involvement in female bladder cancer: an anatomic pathologic study. Urology. 1997;50(4):537–41.
Stein JP, Cote RJ, Freeman JA, Esrig D, Elmajian DA, Groshen S, et al. Indications for lower urinary tract reconstruction in women after cystectomy for bladder cancer: a pathological review of female cystectomy specimens. J Urol. 1995;154(4):1329–33.
Kakizoe T, Tobisu K. Transitional cell carcinoma of the urethra in men and women associated with bladder cancer. Jpn J Clin Oncol. 1998;28(6):357–9.
Van Poppel H, Sorgeloose T. Radical cystectomy with or without urethrectomy? Crit Rev Oncol Hematol. 2003;47(2):141–5.
Stenzl A, Cowan NC, De Santis M, Kuczyk MA, Merseburger AS, Ribal MJ, et al. Treatment of muscle-invasive and metastatic bladder cancer: update of the EAU guidelines. Eur Urol. 2011;59(6):1009–18. https://doi.org/10.1016/j.eururo.2011.03.023.
Osman Y, Mansour A, El-Tabey N, Abdel-Latif M, Mosbah A, Hekal I, et al. Value of routine frozen section analysis of urethral margin in male patients undergoing radical cystectomy in predicting prostatic involvement. Int Urol Nephrol. 2012;44(6):1721–5. https://doi.org/10.1007/s11255-012-0276-z.
Chang SS, Cole E, Cookson MS, Peterson M, Smith JA Jr. Preservation of the anterior vaginal wall during female radical cystectomy with orthotopic urinary diversion: technique and results. J Urol. 2002;168(4 Pt 1):1442–5.
Stenzl A, Jarolim L, Coloby P, Golia S, Bartsch G, Babjuk M, et al. Urethra-sparing cystectomy and orthotopic urinary diversion in women with malignant pelvic tumors. Cancer. 2001;92(7):1864–71.
Large MC, Katz MH, Shikanov S, Eggener SE, Steinberg GD. Orthotopic neobladder versus Indiana pouch in women: a comparison of health related quality of life outcomes. J Urol. 2010;183(1):201–6. https://doi.org/10.1016/j.juro.2009.08.148.
Lee CT, Hafez KS, Sheffield JH, Joshi DP, Montie JE. Orthotopic bladder substitution in women: nontraditional applications. J Urol. 2004;171(4):1585–8.
Abou-Elela A. Outcome of anterior vaginal wall sparing during female radical cystectomy with orthotopic urinary diversion. Eur J Surg Oncol. 2008;34(1):115–21.
Anderson CB, Cookson MS, Chang SS, Clark PE, Smith JA Jr, Kaufman MR. Voiding function in women with orthotopic neobladder urinary diversion. J Urol. 2012;188(1):200–4. https://doi.org/10.1016/j.juro.2012.03.004.
Gross T, Meierhans Ruf SD, Meissner C, Ochsner K, Studer UE. Orthotopic ileal bladder substitution in women: factors influencing urinary incontinence and hypercontinence. Eur Urol. 2015;68(4):664–71. https://doi.org/10.1016/j.eururo.2015.05.015.
Rouanne M, Legrand G, Neuzillet Y, Ghoneim T, Cour F, Letang N, et al. Long-term women-reported quality of life after radical cystectomy and orthotopic ileal neobladder reconstruction. Ann Surg Oncol. 2014;21(4):1398–404. https://doi.org/10.1245/s10434-013-3458-8.
Zahran MH, El-Hefnawy AS, Zidan EM, El-Bilsha MA, Taha DE, Ali-El-Dein B. Health-related quality of life after radical cystectomy and neobladder reconstruction in women: impact of voiding and continence status. Int J Urol. 2014;21(9):887–92. https://doi.org/10.1111/iju.12452.
Bhatta Dhar N, Kessler TM, Mills RD, Burkhard F, Studer UE. Nerve-sparing radical cystectomy and orthotopic bladder replacement in female patients. Eur Urol. 2007;52(4):1006–14.
Turner WH, Danuser H, Moehrle K, Studer UE. The effect of nerve sparing cystectomy technique on postoperative continence after orthotopic bladder substitution. J Urol. 1997;158(6):2118–22.
Hübner WA, Trigo-Rocha F, Plas EG, Tanagho EA. Urethral function after cystectomy: a canine in vivo experiment. Urol Res. 1993;21(1):45–8.
Strasser H, Ninkovic M, Hess M, Bartsch G, Stenzl A. Anatomic and functional studies of the male and female urethral sphincter. World J Urol. 2000;18(5):324–9.
Aboseif SR, Borirakchanyavat S, Lue TF, Carroll PR. Continence mechanism of the ileal neobladder in women: a urodynamics study. World J Urol. 1998;16(6):400–4.
Ali-El-Dein B, Gomha M, Ghoneim MA. Critical evaluation of the problem of chronic urinary retention after orthotopic bladder substitution in women. J Urol. 2002;168(2):587–92.
Ali-el-Dein B, Shaaban AA, Abu-Eideh RH, el-Azab M, Ashamallah A, Ghoneim MA. Surgical complications following radical cystectomy and orthotopic neobladders in women. J Urol. 2008;180(1):206–10. https://doi.org/10.1016/j.juro.2008.03.080 discussion 210.
Bartsch G, Daneshmand S, Skinner EC, Syan S, Skinner DG, Penson DF. Urinary functional outcomes in female neobladder patients. World J Urol. 2014;32(1):221–8. https://doi.org/10.1007/s00345-013-1219-8.
Stenzl A, Colleselli K, Bartsch G. Update of urethra-sparing approaches in cystectomy in women. World J Urol. 1997;15(2):134–8.
Ghoneim MA. Orthotopic bladder substitution in women following cystectomy for bladder cancer. Urol Clin North Am. 1997;24(1):225–39.
Henningsohn L, Wijkström H, Steven K, Pedersen J, Ahlstrand C, Aus G, et al. Relative importance of sources of symptom-induced distress in urinary bladder cancer survivors. Eur Urol. 2003;43(6):651–62.
Kulaksizoglu H, Toktas G, Kulaksizoglu IB, Aglamis E, Unlüer E. When should quality of life be measured after radical cystectomy? Eur Urol. 2002;42(4):350–5.
Henningsohn L, Steven K, Kallestrup EB, Steineck G. Distressful symptoms and well-being after radical cystectomy and orthotopic bladder substitution compared with a matched control population. J Urol. 2002;168:168–74 discussion 174-5.
Salonia A, Zanni G, Gallina A, Briganti A, Saccà A, Suardi N, et al. Unsuccessful investigation of preoperative sexual health issues in the prostate cancer “couple”: results of a real-life psychometric survey at a major tertiary academic center. J Sex Med. 2009;6(12):3347–55. https://doi.org/10.1111/j.1743-6109.2009.01483.x.
Pederzoli F, Campbell JD, Matsui H, Sopko NA, Bivalacqua TJ. Surgical factors associated with male and female sexual dysfunction after radical cystectomy: what do we know and how can we improve outcomes? Sex Med Rev. 2018;6(3):469–81. https://doi.org/10.1016/j.sxmr.2017.11.003.
Volkmer BG, Gschwend JE, Herkommer K, Simon J, Küfer R, Hautmann RE. Cystectomy and orthotopic ileal neobladder: the impact on female sexuality. J Urol. 2004;172(6 Pt 1):2353–7.
Rosen R, Brown C, Heiman J, Leiblum S, Meston C, Shabsigh R. The Female Sexual Function Index (FSFI): a multidimensional self-report instrument for the assessment of female sexual function. J Sex Marital Ther. 2000;26(2):191–208.
Zippe CD, Raina R, Shah AD, Massanyi EZ, Agarwal A, Ulchaker J, et al. Female sexual dysfunction after radical cystectomy: a new outcome measure. Urology. 2004;63(6):1153–7.
Booth BB, Rasmussen A, Jensen JB. Evaluating sexual function in women after radical cystectomy as treatment for bladder cancer. Scand J Urol. 2015;49(6):463–7. https://doi.org/10.3109/21681805.2015.1055589.
Zahran MH, Fahmy O, El-Hefnawy AS, Ali-El-Dein B. Female sexual dysfunction post radical cystectomy and urinary diversion. Climacteric. 2016;19(6):546–50.
Berman L, Berman J, Felder S, Pollets D, Chhabra S, Miles M, et al. Seeking help for sexual function complaints: what gynecologists need to know about the female patient’s experience. Fertil Steril. 2003;79(3):572–6.
Zippe CD, Nandipati KC, Agarwal A, Raina R. Female sexual dysfunction after pelvic surgery: the impact of surgical modifications. BJU Int. 2005;96(7):959–63.
Bhatt A, Nandipati K, Dhar N, Ulchaker J, Jones S, Rackley R, et al. Neurovascular preservation in orthotopic cystectomy: impact on female sexual function. Urology. 2006;67(4):742–5.
Bouman MB, van Zeijl MC, Buncamper ME, Meijerink WJ, van Bodegraven AA, Mullender MG. Intestinal vaginoplasty revisited: a review of surgical techniques, complications, and sexual function. J Sex Med. 2014;11(7):1835–47. https://doi.org/10.1111/jsm.12538.
Schover LR, von Eschenbach AC. Sexual function and female radical cystectomy: a case series. J Urol. 1985;134(3):465–8.
Chen YB, Cheng TJ, Lin HH, Yang YS. Spatial W-plasty full-thickness skin graft for neovaginal reconstruction. Plast Reconstr Surg. 1994;94(5):727–31.
McCraw JB, Massey FM, Shanklin KD, Horton CE. Vaginal reconstruction with gracilis myocutaneous flaps. Plast Reconstr Surg. 1976;58(2):176–83.
Li X, Wang W, Zhu G, He W, Gou X. Risk factors, follow-up, and treatment of urethral recurrence following radical cystectomy and urinary diversion for bladder cancer: a meta-analysis of 9498 patients. Oncotarget. 2017;9(2):2782–96. https://doi.org/10.18632/oncotarget.23451.
Stein JP, Ginsberg DA, Skinner DG. Indications and technique of the orthotopic neobladder in women. Urol Clin North Am. 2002;29(3):725–34.
Ali-el-Dein B, Abdel-Latif M, Ashamallah A, Abdel-Rahim M, Ghoneim M. Local urethral recurrence after radical cystectomy and orthotopic bladder substitution in women: a prospective study. J Urol. 2004;171(1):275–8.
Hassan JM, Cookson MS, Smith JA Jr, Chang SS. Urethral recurrence in patients following orthotopic urinary diversion. J Urol. 2004;172(4 Pt 1):1338–41.
Boorjian SA, Kim SP, Weight CJ, Cheville JC, Thapa P, Frank I. Risk factors and outcomes of urethral recurrence following radical cystectomy. Eur Urol. 2011;60(6):1266–72. https://doi.org/10.1016/j.eururo.2011.08.030.
• Gakis G, Ali-El-Dein B, Babjuk M, Hrbacek J, Macek P, Burkhard FC, et al. Urethral recurrence in women with orthotopic bladder substitutes: a multi-institutional study. Urol Oncol. 2015;33(5):204.e17–23. https://doi.org/10.1016/j.urolonc.2015.01.020 The largest series to date on oncological outcomes after radical cystectomy with ileal neobladder in women.
Huguet J. Diagnosis and treatment of urethral recurrence after radical cystectomy in the male. Actas Urol Esp. 2012;36(1):42–7. https://doi.org/10.1016/j.acuro.2011.06.009.
Stein JP, Clark P, Miranda G, Cai J, Groshen S, Skinner DG. Urethral tumor recurrence following cystectomy and urinary diversion: clinical and pathological characteristics in 768 male patients. J Urol. 2005;173(4):1163–8.
Varol C, Thalmann GN, Burkhard FC, Studer UE. Treatment of urethral recurrence following radical cystectomy and ileal bladder substitution. J Urol. 2004;172(3):937–42.
Nieder AM, Sved PD, Gomez P, Kim SS, Manoharan M, Soloway MS. Urethral recurrence after cystoprostatectomy: implications for urinary diversion and monitoring. Urology. 2004;64(5):950–4.
Yoshimine S, Kikuchi E, Matsumoto K, Ide H, Miyajima A, Nakagawa K, et al. The clinical significance of urine cytology after a radical cystectomy for urothelial cancer. Int J Urol. 2010;17(6):527–32. https://doi.org/10.1111/j.1442-2042.2010.02516.x.
Cho KS, Seo JW, Park SJ, Lee YH, Choi YD, Cho NH, et al. The risk factor for urethral recurrence after radical cystectomy in patients with transitional cell carcinoma of the bladder. Urol Int. 2009;82(3):306–11. https://doi.org/10.1159/000209363.
Nelles JL, Konety BR, Saigal C, Pace J, Lai J. Urethrectomy following cystectomy for bladder cancer in men: practice patterns and impact on survival. J Urol. 2008;180(5):1933–6. https://doi.org/10.1016/j.juro.2008.07.039 discussion 1936-7.
Huguet-Pérez J, Palou J, Millán-Rodríguez F, Salvador-Bayarri J, Villavicencio-Mavrich H, Vicente-Rodríguez J. Upper tract transitional cell carcinoma following cystectomy for bladder cancer. Eur Urol. 2001;40(3):318–23.
Gakis G, Schilling D, Perner S, Schwentner C, Sievert KD, Stenzl A. Sequential resection of malignant ureteral margins at radical cystectomy: a critical assessment of the value of frozen section analysis. World J Urol. 2011;29(4):451–6. https://doi.org/10.1007/s00345-010-0581-z.
Meissner C, Giannarini G, Schumacher MC, Thoeny H, Studer UE, Burkhard FC. The efficiency of excretory urography to detect upper urinary tract tumors after cystectomy for urothelial cancer. J Urol. 2007;178(6):2287–90.
Tollefson MK, Blute ML, Farmer SA, Frank I. Significance of distal ureteral margin at radical cystectomy for urothelial carcinoma. J Urol. 2010;183(1):81–6. https://doi.org/10.1016/j.juro.2009.08.158.
Sanderson KM, Cai J, Miranda G, Skinner DG, Stein JP. Upper tract urothelial recurrence following radical cystectomy for transitional cell carcinoma of the bladder: an analysis of 1,069 patients with 10-year followup. J Urol. 2007;177(6):2088–94.
Schoenberg MP, Carter HB, Epstein JI. Ureteral frozen section analysis during cystectomy: a reassessment. J Urol. 1996;155(4):1218–20.
Soloway MS, Myers GH Jr, Burdick JF, Malmgren RA. Ileal conduit exfoliative cytology in the diagnosis of recurrent cancer. J Urol. 1972;107(5):835–9.
Raj GV, Bochner BH, Serio AM, Vickers A, Donat SM, Herr H, et al. Natural history of positive urinary cytology after radical cystectomy. J Urol. 2006;176(5):2000–5 discussion 2005.
Slaton JW, Swanson DA, Grossman HB, Dinney CP. A stage specific approach to tumor surveillance after radical cystectomy for transitional cell carcinoma of the bladder. J Urol. 1999;162(3 Pt 1):710–4.
Brien JC, Shariat SF, Herman MP, Ng CK, Scherr DS, Scoll B, et al. Preoperative hydronephrosis, ureteroscopic biopsy grade and urinary cytology can improve prediction of advanced upper tract urothelial carcinoma. J Urol. 2010;184(1):69–73. https://doi.org/10.1016/j.juro.2010.03.030.
Fujisawa M, Nakamura I, Yamanaka N, Gotoh A, Hara I, Okada H, et al. Changes in calcium metabolism and bone demineralization after orthotopic intestinal neobladder creation. J Urol. 2000;163(4):1108–11 quiz 1295.
Hautmann RE, Abol-Enein H, Davidsson T, Gudjonsson S, Hautmann SH, Holm HV, et al. International Consultation on Urologic Disease-European Association of Urology Consultation on Bladder Cancer 2012. ICUD-EAU International Consultation on Bladder Cancer 2012: urinary diversion. Eur Urol. 2013;63(1):67–80. https://doi.org/10.1016/j.eururo.2012.08.050.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
Nikolaos Kalampokis, Nikolaos Grivas, Markus Ölschläger, Fahmy Nabil Hassan, and Georgios Gakis each declare no potential conflicts of interest.
Human and Animal Rights and Informed Consent
This article does not contain any studies with human or animal subjects performed by any of the authors.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
This article is part of the Topical Collection on Urothelial Cancer
Rights and permissions
About this article
Cite this article
Kalampokis, N., Grivas, N., Ölschläger, M. et al. Radical Cystectomy in Female Patients - Improving Outcomes. Curr Urol Rep 20, 83 (2019). https://doi.org/10.1007/s11934-019-0951-z
Published:
DOI: https://doi.org/10.1007/s11934-019-0951-z