Abstract
Purpose of Review
Tyrosine kinase inhibitors (TKI) and monoclonal antibodies (mAbs) that target the epidermal growth factor receptor (EGFR) have changed the therapeutic landscape across a range of solid malignancies. However, there is little data regarding the cardiovascular (CV) impact of these agents. The purpose of this review is to discuss reported CV effects, pathophysiology, pre-treatment screening, diagnostic workup, and treatment recommendations in this patient population.
Recent Findings
It is apparent that CV events are not class dependent, and while infrequently reported in clinical trials, unique CV toxicity may occur with EGFR inhibitors, including structural, electrical, and vascular events.
Summary
There remains an unmet need to fully elucidate the spectrum of CV events associated with EGFR inhibitors. Early CV screening, close clinical monitoring, coupled with a multidisciplinary approach between medical and cardio-oncology is needed to minimize the potentially detrimental impact of cardiotoxicity in this patient population.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The advent and evolution of targeted anti-neoplastic therapy against EGFR represents a paradigm shift in cancer treatment, allowing a tailored approach focusing on the unique genomic aberrancies of various malignancies. The United States Food and Drug Administration (FDA) has approved multiple EGFR-TKIs targeting receptor and non-receptor tyrosine kinases and mAbs against growth factor receptor kinases for the treatment of multiple cancer types, including non-small cell lung cancer (NSCLC), colorectal cancer (CRC), squamous cell carcinomas of the head and neck, medullary thyroid carcinoma, and HER2-amplified breast cancer [1]. While patient outcomes are improving, CV adverse effects (AE) are becoming increasingly recognized, encompassing structural (left ventricular (LV) dysfunction, heart failure), electrical (arrhythmias, QTc prolongation, sudden cardiac arrest (SCA)), and vascular (hypertension, coronary artery disease (CAD), myocardial infarction (MI)) AE. This review examines the CV pathophysiological and clinical consequences of EGFR inhibition, providing a guide to the screening and diagnosis of cardiotoxicity associated with EGFR-TKIs and mAbs.
Pathophysiologic Role in Cardiovascular Development and Function
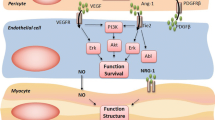
EGFR is a receptor tyrosine kinase in the erythroblastic leukemia viral oncogene homolog (ErbB)/HER (human epidermal growth factor receptor) family, which consists of four different receptor/tyrosine kinase domains: EGFR/HER1/ErbB1, ErbB2/Neu/HER2, ErbB3/HER3, and ErbB4/HER4 [2]. The ErbB receptor pathway is necessary in the homeostatic regulation of cellular proliferation, differentiation, and apoptosis [2, 3]. Signal transduction pathways are propagated following ligand-induced receptor homo- or heterodimerization and activation of downstream tyrosine kinase activity [4] (Fig. 1).
The ErbB receptor family is necessary for the development and maintenance of the CV system [3]. In utero, the ErbB2/ErbB4/NRG-1 signaling cascade stimulates CV development and remains expressed in low concentrations localized to the T-tubules in the adult heart [5]. Preclinical and translational models assessing mutations or blockade of the ErbB2, ErbB3, and ErbB4 pathways have impacts on myocardial development and function. For example, human-induced pluripotent stem cell–derived cardiomyocytes of trastuzumab-treated patients have impaired contractility and calcium handling properties from mitochondrial dysfunction and impaired cardiac energy metabolism related to alteration in the AMP-activated protein kinase pathway [6]. Deletions of ErbB2, ErbB4, or NRG-1 in mutant embryonic mice cells resulted in abnormal ventricular trabeculation and embryonic lethality [7,8,9]. Erickson and colleagues also noted that mice embryos deficient in ErbB2 died from a lack of ventricular cardiomyocyte differentiation [10]. ErbB2 also appears to be necessary in adults; young adult mice with an acquired mutation in ErbB2 led to severe cardiomyopathy and sudden death [5]. EGFR/ErbB1 has been shown to play a role in vascular remodeling. Schreier et al. found that targeted EGFR deletion in vascular smooth muscle isolated from mice aortas led to spontaneous cell death, resulting from reduced pentose phosphate pathway activity, disturbed cellular matrix homeostasis, and diminished induction of ERK1/2 phosphorylation in response to oxidative stress [11•]. Crosstalk between EGFR and ErbB2 can also occur; a murine model showed targeted blockade of ErbB1 signaling also resulted in the inhibition of ErbB2 signaling, culminating in cardiomyocyte hypertrophy, chamber dilation, and dilated cardiomyopathy [12•].
EGFR-TKIs and mAbs have a unique array of “on-target” and “off-target” CV events, suggesting drug-specific mechanisms [3]. For instance, the EGFR and HER2 inhibitor, lapatinib, downregulates genes involved in small molecule metabolism and reduces glucose uptake in induced pluripotent stem cell–derived cardiomyocytes [13]. Vandetanib, a TKI targeting the VEGF receptor, EGFR, and RET proto-oncogene, inhibits the rapidly activating delayed rectifier K + channel, via the blockade of the human Ether-à-go-go-Related Gene (hERG) channel, prolonging action potential duration. In addition, its action in human-induced pluripotent stem cell–derived cardiomyocytes markedly reduces the maximum rate of depolarization during the action potential upstroke [14]. Since CV risk related to EGFR-TKIs is drug-specific rather than class-specific, it is prudent that medical oncologists and cardiologists are familiar with the following agents.
Drug-Specific Cardiovascular Effects
NSCLC (1st Generation)
Erlotinib
Erlotinib is a small molecule EGFR TKI approved as maintenance and second-line therapy for NSCLC patients with locally advanced or metastatic disease and as first-line therapy along with gemcitabine in locally advanced, unresectable, or metastatic pancreatic cancer. It is also approved by the European Medicine Agency (EMA) as a first-line therapy for EGFR-mutated NSCLC [15,16,17].
A highly selective EGFR inhibitor, erlotinib has a low CV risk profile [18, 19]. Mice models have shown upregulation of the JAK/STAT pathway, which may provide cardioprotective effects [19]. While earlier trials in NSCLC did not report cardiac events, several subsequent trials have reported acute coronary syndrome (ACS) and heart failure in patients receiving erlotinib as monotherapy or in combination with other agents. In the National Cancer Institute of Canada trial assessing gemcitabine ± erlotinib in advanced pancreatic cancer, there was a higher incidence of reported coronary artery events (2.3% vs 1.2%) [20•, 21•]. In a study assessing maintenance, erlotinib in stage IIIB and IV NSCLC cardiac AE included one patient with cardiopulmonary failure, one with cardiopulmonary arrest, and one patient with heart failure leading to death [22]. In a phase II trial investigating the addition of erlotinib to bevacizumab in renal cell carcinoma, one patient out of 51 treated with erlotinib had an arterial thromboembolic event [18, 23]. In a phase III trial comparing bevacizumab with or without erlotinib, there were similar rates of hypertension, “other cardiovascular events,” and thromboembolic events [24]. Subsequent case reports and series have reported rare cardiac events, including dilated cardiomyopathy and ST-elevation myocardial infarction (STEMI) (Table 1, Fig. 2) [25, 26].
Gefitinib
Gefitinib is an oral selective EGFR TKI approved for EGFR exon 19 deletion or exon 21 substituted NSCLC [60, 61]. CV events related to gefitinib are uncommon, although several trials excluded patients with clinically relevant CV disease [62,63,64,65,66]. However, there are rare reports of fatal cardiotoxicity reported with gefitinib, including myocarditis, cardiomyopathy, MI, atrial fibrillation, and left bundle branch block (Table 1, Fig. 2) [21•,27,28,29,30,30]. Additional studies are needed to ascertain CV risk in this patient population.
Necitumumab
Necitumumab is a 2nd-generation monoclonal IgG1 antibody directed against EGFR approved for advanced squamous NSCLC [51, 67, 68]. Clinical trials have rarely reported cases of arterial thromboembolism, but the incidence was similar in the necitumumab plus chemotherapy arm and the chemotherapy alone arms [49••, 51]. In a phase III trial comparing necitumumab, cisplatin, and gemcitabine to cisplatin and gemcitabine alone for stage IV squamous NSCLC, cardiopulmonary or SCA occurred in approximately 3% of the treatment arm versus less than 1% of patients who were on chemotherapy alone, leading to an FDA black box warning [49••, 68, 69]. In a phase 1b trial investigating the addition of necitumumab to pembrolizumab in patients with stage IV NSCLC of both squamous and non-squamous histology, 1 patient had SCA (Table 1, Fig. 2) [50].
NSCLC (2nd Generation)
Afatinib
Afatinib is an irreversible second-generation EGFR/HER2-TKI approved for first-line treatment of metastatic NSCLC [70].
CV AE are rarely reported with afatinib. In a pooled analysis of 49 trials, there were no reports of heart failure or decreased LVEF [70]. One reported case of symptomatic systolic and diastolic heart failure and pericardial effusion occurred but resolved with temporary cessation of therapy (Table 1, Fig. 2) [31].
Dacomitinib
Dacomitinib is an irreversible, pan-HER TKI approved for first-line treatment of patients with metastatic NSCLC EGFR exon 19 deletion or exon 21 L858R substitution mutations [63]. In a phase III study comparing dacomitinib and gefitinib as front line therapy in newly diagnosed NSCLC, no cardiovascular AE were reported, but patients were excluded if they had untreated or substantial CV disease [63, 71]. There do not appear to be any significant reports of CV toxicity. Furthermore, there do not appear to be any clinically relevant impacts to the QTc interval with dacomitinib, based on a phase II study (Table 1, Fig. 2) [72•].
NSCLC (3rd Generation)
Osimertinib
Osimertinib is a third-generation irreversible EGFR-TKI approved for advanced NSCLC harboring EGFR activating mutation, advanced NSCLC following progression after first-line EGFR TKI therapy harboring T790M mutation, and as adjuvant therapy for stage IB-IIIA NSCLC who had EGFR exon 19 deletions or exon 21 mutations. In a phase III trial comparing osimertinib to pemetrexed amongst T790M EGFR mutation-positive NSCLC who had progressed after first-line EGFR-TKI therapy, decreases in LVEF ≥ 10% and an absolute LVEF < 50% occurred in 5% of the osimertinib arm with a median time to onset of 5.5 months, with cardiac failure reported in 3% in the osimertinib group and none in the pemetrexed treatment arm. Also, 4% of the osimertinib arm had QTc prolongation compared to 1% in the control arm [32•]. In a phase III trial comparing frontline osimertinib to gefitinib or erlotinib for advanced NSCLC, 10% of osimertinib treated patients had QTc prolongation compared to 4% in the control arm. Grade ≥ 3 QTc prolongation occurred in 1% of each arm. Furthermore, decreased LVEF occurred in 5% of osimertinib-treated patients compared to 2% in the control arm [33•]. In a retrospective study of 123 cases of advanced NSCLC receiving osimertinib, 5% of patients had cardiac AE including MI, heart failure, and valvular heart disease [35]. In a pharmacovigilance study based on FDA adverse events database, reporting odds ratio of heart failure, QTc prolongation and atrial fibrillation were higher with osimertinib compared to first- and second-generation EGFR TKIs [36•]. Another study based on the World Health Organization (WHO) pharmacovigilance database, VigiBase, found that osimertinib was associated with higher odds of QTc prolongation, supraventricular tachycardia, and heart failure [37]. In a phase III study assessing adjuvant osimertinib compared to placebo in stage IB to IIIA NSCLC, the osimertinib arm reported cardiac AE in 5% (reduced EF, cardiomyopathy, pulmonary edema, and cardiomyopathy compared to 3% in the placebo group (Table 1, Fig. 2) [34•].
Mobocertinib
Mobocertinib is an irreversible EGFR/HER2-TKI recently approved for the treatment of NSCLC harboring the EGFR exon 20 insertion mutation [38, 73]. A pooled safety analysis of 250 patients reported QTc prolongation and heart failure as the most common CV events [38]. In the analysis, 1.2% of patients developed a QTc interval > 500 ms, and around 11% had a relative 60 ms increase in QTc interval from baseline with one instance of torsades de pointes (TdP). Heart failure occurred in 2.7% of patients. Prior phase I/II trials excluded patients with prolonged QTc [74]. Ongoing phase III trials are evaluating the CV safety profile of mobocertinib [75] (Table 1, Fig. 2).
Amivantamab-vmjw
Amivantamab-vmjw is a bispecific antibody directed against epidermal growth factor (EGF) and MET receptors, for adult patients with locally advanced or metastatic non-small cell lung cancer (NSCLC) with EGFR exon 20 insertion mutations who have progressed on platinum-based chemotherapy [59••, 76]. In the phase I trial, acute coronary symptom, atrial flutter, and cardio-respiratory distress were reported in one patient each out of 114 patients [59••]. There is currently little data on the cardiac impacts on this bispecific antibody, and additional assessments in upcoming phase III studies are needed (Table 1).
Breast Cancer
Neratinib
Neratinib is a small molecule, irreversible, dual pan-HER TKI approved for extended adjuvant treatment of early-stage, HER2-positive breast cancer and in combination with capecitabine for patients with advanced/metastatic HER2-positive disease after ≥ 2 prior lines of HER2-directed therapy [39•, 77, 78].
CV AE are rare. In a phase III trial comparing neratinib plus capecitabine to lapatinib plus capecitabine for HER2-positive, pretreated metastatic breast cancer, cardiac monitoring was performed at the start of cycles 3 and 6 and every 6 cycles afterward using a single standard 12-lead digital electrocardiogram (ECG) and multiple-gated acquisition scans or transthoracic echocardiograms. The incidence of arrhythmia was 3.3%, ischemic heart disease was 0.7%, QTc prolongation was 2.3%, and decreased LVEF in 4.3%. Many patients in this study received two or more HER2-directed regimens in the neoadjuvant (16.9%) and adjuvant (47.6%) setting [39•]. In a phase III trial assessing extended adjuvant neratinib therapy for patients with hormone receptor positive, HER2-positive, early-stage breast cancer, QTc prolongation occurred in 3% compared to 7% in the placebo group, and a decreased LVEF occurred 1% of patients in both the treatment and placebo arms [42•]. In the extended analysis, no additional cardiac toxicities were reported [77]. In a phase III trial comparing neratinib plus paclitaxel to trastuzumab plus paclitaxel, grade 3 or higher cardiac events were reported in 1.3% of patients in the neratinib group compared to 3% in the trastuzumab group, with toxicities including decreased LVEF and heart failure [43] (Table 1, Fig. 2).
Lapatinib Ditosylate
Lapatinib ditosylate is a reversible, small molecule EGFR/HER2-TKI that is approved for the treatment of advanced or metastatic HER2-positive breast cancer in combination with capecitabine in women who have previously received trastuzumab, and in combination with letrozole in women with hormone receptor positive, HER2-positive metastatic breast cancer [79–81].
In preclinical canine models, therapeutic doses of lapatinib have been demonstrated to increase total peripheral vascular resistance, QTc interval, and the plasma concentration of cardiac troponin I [82]. However, reports of CV toxicity vary widely, with clinical trials and retrospective reports reporting AE ranging from 0.0 to 19.2% [39•, 41, 83, 84, 85, 86, 87••]. In the phase III study mentioned above, arrhythmias occurred in 3.5%, ischemic heart disease in 0.6%, QTc prolongation in 3.9%, and decreased LVEF in 2.3% of patients treated in the lapatinib plus capecitabine arm [39•]. In a phase III trial comparing trastuzumab emtansine and lapatinib-capecitabine in previously treated, metastatic breast cancer, there were no cardiac events attributed to the lapatinib group, compared to < 1% of cardiac dysfunction in the trastuzumab arm [41]. In a phase III study of 8,381 women with HER2-positive breast cancer treated with either trastuzumab alone or adjuvant lapatinib plus trastuzumab, 2–3% developed heart failure, which did not substantially differ from trastuzumab plus lapatinib or trastuzumab alone (2–3%) [40]. In a recent subanalysis of a trial comparing adjuvant lapatinib plus trastuzumab vs trastuzumab alone, 8.6% of 4190 patients developed CV events. Of these, 7.9% occurred in the lapatinib + trastuzumab arm and 9.3% in trastuzumab alone arm. Predisposing CV risk factors included LVEF < 55%, diabetes mellitus, BMI > 30 kg/m2, cumulative dose of doxorubicin ≥ 240 mg/m2, and of epirubicin ≥ 480 mg/m2 (Table 1, Fig. 2) [87••].
Head and Neck, Colon Cancer
Cetuximab
Cetuximab is a recombinant chimeric human/mouse IgG1 monoclonal antibody which binds to EGFR and competitively inhibits the binding of other ligands such as TGF-beta. It is approved for metastatic, KRAS wild type, EGFR expressing colorectal cancer in combination with folinic acid, 5-fluorouracil, and irinotecan (FOLFIRI), in combination with irinotecan in patients who are refractory to fluoropyrimidine and oxaliplatin therapy, and as a single agent in patients who have failed oxaliplatin and irinotecan-based chemotherapy [ 88,89,90]. In addition to colon cancer, cetuximab is approved for locally/regionally advanced squamous cell carcinoma of the oropharynx, hypopharynx, or larynx in combination with high-dose radiotherapy and in combination with a platinum-fluorouracil chemotherapy backbone for metastatic disease [52, 91].
Clinical trials of cetuximab for colorectal cancer seldomly reported CV AE, and in several clinical trials, no CV events were reported [88, 89, 92]. However, in an extended analysis of a phase III trial of cetuximab + irinotecan following first-line treatment failure in metastatic colorectal cancer, one case of treatment-related, new-onset heart failure resulting in death occurred. In addition, there was another death in the cetuximab + irinotecan arm due to cardiac failure, which was attributed to irinotecan [53]. In an analysis of 2,126 cetuximab-treated patients alone or in combination with chemotherapy for metastatic colorectal cancer, cardiotoxicity was reported in 0.9%, with 5 cases of grades 3 or 4 events, including coronary spastic angina, myocardial infarction, and heart failure, resulting in 4 deaths. All deaths occurred with concomitant FOLFIRI or irinotecan [54•]. In an analysis of cardiotoxicity comparing single agent cetuximab and panitumumab in metastatic, chemotherapy refractory colorectal adenocarcinoma, troponin elevation occurred in 34.4%, nonspecific ST changes in 32.7%, and QTc prolongation in 36.1%, with resolution occurring within 10 months. Chest pain occurred in 8.1% (one grade 3 event) and 4.8% developed arrhythmias requiring medical management [56••]. In a case control study of 27,992 patients receiving TKIs for various malignancies, cetuximab was associated with higher odds of new onset heart failure. Pre-existing comorbidities that posed a risk included diabetes mellitus, hypertension, chronic renal failure, ischemic heart disease, valvular heart disease, arrhythmia, and smoking [55•].
Cetuximab-associated CV toxicity is also reported in patients receiving treatment for head and neck cancers, although radiotherapy and platinum-based therapy are confounding factors. In a phase III trial assessing platinum-fluorouracil-cetuximab vs platinum-fluorouracil as front-line therapy in metastatic head and neck cancer, CV events were reported in 7% and 4% of patients in the platinum-fluorouracil-cetuximab treatment arm and control arm, respectively, including heart failure (n = 4 vs 1), MI or ischemia (n = 7 vs 2), and SCA (n = 3 vs 1), which did not significantly differ (p = 0.22) [52]. A phase II study of cetuximab plus cisplatin, with or without paclitaxel, reported CV AE in 6% of the treatment arm and 5% of the placebo arm with one patient in each arm developing grade 3 or higher AEs (Table 1, Fig. 2) [93].
Panitumumab
Panitumumab is a monoclonal human IgG2 antibody against EGFR that is approved for the treatment of metastatic colorectal cancer that progressed on treatment with oxaliplatin, fluoropyrimidine, and irinotecan-containing regimens [94]. CV toxicities including heart failure and arrhythmias have been reported [55•, 56••, 57]. A nested case–control population-based study including 27,000 patients who received TKIs and/or chemotherapy showed an increased risk of heart failure in patients receiving panitumumab (OR 3.01, 95% CI 1.02–8.85) [55•]. In addition to heart failure, arrhythmias have been documented [56••, 57]. Furthermore, EKG changes including ST segment changes and QTc prolongation have been reported, none of which were considered “serious” [56••]. A post-marketing study involving more than 3,000 colorectal cancer patients in Japan reported 0.2% incidence of CV AE with only one case of grade 3 cardiotoxicity (Table 1, Fig. 2) [58].
Thyroid Cancer
Vandetanib
Vandetanib is a non-specific TKI with activity against VEGF receptor, EGFR, and rearranged during transfection (RET) tyrosine kinases, approved for the management of unresectable, locally advanced, or metastatic medullary thyroid cancer in adult patients [44•, 95].
CV toxicity is reported in clinical studies as well as post-marketing studies, and the risk of QTc interval prolongation, TdP, and SCA are included in FDA prescriber warnings [95]. QTc prolongation is commonly reported, is dose dependent, and has an incidence ranging from 5.4 to 44.1 [44•, 45, 46]. Furthermore, meta-analyses have reported that the risk of all-grade QTc prolongation and grade 3 or higher QTc prolongation is higher with vandetanib compared to placebo or other VEGF inhibitors [47, 48]. Based on this data, patients should be monitored closely for QTc prolongation given the possibility of life-threatening arrhythmias, such as TdP and SCA [95].
Hypertension is a common AE reported with vandetanib in clinical trials, ranging from 16 to 56% [44•, 45, 46]. In a phase 3 trial assessing vandetanib compared to erlotinib in previously treated advanced NSCLC, hypertension was reported in 16% of the treatment arm compared to 2% of the erlotinib group, and 0.4% of patients randomized to vandetanib had to discontinue treatment due to poorly controlled hypertension [45]. In addition, meta-analyses have found that vandetanib increases the risk of hypertension compared to similar class agents (Table 1, Fig. 2) [47, 48].
Screening, Diagnosis, Management, and Prevention of EGFR Tyrosine Kinase Inhibitor Cardiotoxicity
Baseline Cardiovascular Risk Assessment and Prevention
The spectrum of possible EGFR-TKI-associated cardiotoxicity varies depending on the specific agent and is impacted by baseline CV disease or risk factors (Table 1, Fig. 2). Cancer and CV disease share many common risk factors, and a baseline CV risk assessment should be obtained before initiation of potentially cardiotoxic EGFR-TKIs [96, 97, 98, 99, 100••, 101, 102]. Modifiable risk factors including obesity, diabetes mellitus, hypertension, current tobacco use, and red/processed meat consumption all should be addressed with patients before and during EGFR-TKI therapy as each predisposes to adverse CV events and may negatively impact oncological outcomes [96, 102].
For example, low-density lipoprotein molecules are independently associated with CAD, and patients with intermediate-to-high risk of atherosclerotic CV disease should be on statin therapy [103]. Current tobacco consumption is associated with major adverse CV events and worse clinical outcomes during treatment of lung cancer [104]. Hence, medical therapy should be optimized for diabetes, hypertension, dyslipidemia, and lifestyle modification and tobacco cessation encouraged [105].
Concomitant or prior cancer therapies should be considered in CV risk stratification before administration of potentially cardiotoxic EGFR-TKIs. Anthracyclines, platinum-based therapy, alkylating agents, HER2 inhibitors, VEGF inhibitors, immune checkpoint inhibitors, and chest radiotherapy have been associated with LV dysfunction, acute heart failure, systemic hypertension, and ischemic heart disease [106,107,108,109,110,111,112]. Radiotherapy, particularly targeting the mediastinum, left chest wall, and head and neck, is associated with accelerated coronary, cerebrovascular, and peripheral artery atherosclerosis [113, 114]. Targeted therapy with EGFR-TKIs may increase the risk of major adverse CV events in patients who are concurrently or previously received any of the above therapeutics.
Indications and Rationale for Screening
The increasing complexity of targeted oncological therapies and a discrepancy in adverse CV events reported in clinical trials of EGFR-TKIs compared to pharmacovigilance studies highlight the importance of referring patients to a cardio-oncologist for the prevention, detection, and management of CV disease before initiation of potentially cardiotoxic EGFR-TKIs [98, 99, 100••, 101, 114, 115]. EGFR-TKIs generally have a better cardiac safety profile compared to other antineoplastic therapies, and research is ongoing regarding the CV risk posed by these agents [116]. Accordingly, clinical guidelines and guidance statements from professional societies have lagged in offering specific recommendations regarding risk stratification of patients treated with EGFR-TKIs alone with the American Society of Clinical Oncology (ASCO) leaving no formal recommendations for this patient population [98].
Cardiotoxicity related to EGFR-TKIs appears to be a drug-specific rather than class specific, and screening tests should be tailored to the AE associated with the drug (e.g., obtaining a 12-lead ECG to evaluate QTc interval duration prior to administration of vandetanib or a baseline echocardiogram before starting osimertinib in a patient with known CAD). The 2020 European Society of Medical Oncology (ESMO) guidelines for the management of cardiac disease in cancer patients accounts for the heterogeneous effects of specific EGFR-TKIs, recommending a baseline 12-lead ECG and electrolytes in patients before administering a drug associated with QTc prolongation and serial quantitative assessment of LVEF and diastolic function with the same imaging modality before initiation of EGFR-TKIs associated with cardiomyopathy and heart failure [100••] (Fig. 3).
Screening and management of EGFR-TKI cardiovascular toxicities. *Low to intermediate CV risk patients in which decision about statin use is uncertain. #Baseline echocardiography (may consider strain protocol if available) is recommended in high-risk patients or patients pending initiation of agents associated with heart failure listed in Fig. 2. May consider baseline stress test or coronary CT angiography in patients with stable ischemic heart disease or congestive heart failure and no recent ischemic evaluation. ∫Cardiac MRI with stress preferred over myocardial perfusion imaging or coronary CT angiography for evaluation of ejection fraction and ischemia unless contraindications present
Role of Screening Electrocardiography and Arrhythmia Management
Arrhythmias are not uncommon among patients undergoing treatment with certain EGFR-TKIs. Atrial fibrillation has been associated with vandetanib, osimertinib, and lapatinib in pharmacovigilance studies [36•, 116]. Rate-control medications, primarily beta blockers, are preferred over nondihydropyridine calcium channel blockers and antiarrhythmics for medical management of atrial fibrillation related to EGFR-TKIs due to less drug-drug interactions [99].
Caution should be exercised when using direct oral anticoagulants in patients receiving vandetanib or lapatinib as both are CYP3A4-type cytochrome P450 and p-glycoprotein inhibitors and can increase risk of bleeding events [117]. Left atrial appendage occlusion devices are promising alternatives to long-term anticoagulation for patients with atrial fibrillation on these agents, although these technologies have not been specifically evaluated in patients with cancer [118].
A baseline standard 12-lead ECG should be obtained in patients who are starting arrhythmogenic EGFR-TKIs or mAbs, as summarized in Table 1 and Fig. 2. The QT interval should be measured manually and QTc estimated by the Fridericia formula [QT interval/(RR interval)1/3] as it is more accurate at extreme heart rates [67, 101, 118]. Risk of arrhythmia is increased with high-grade QTc prolongation (QTc > 500 ms), and an alternative agent should be considered in these patients [98]. ECGs should be obtained frequently every 3–4 weeks during initial treatment. Nonessential QTc prolonging medications should be discontinued, and electrolytes obtained before treatment. Nausea and vomiting which may induce hypokalemia and QTc prolongation should be addressed before and during treatment.
If ventricular arrhythmia or cardiogenic syncope occurs, the drug should be discontinued. Intravenous magnesium sulfate should be administered for TdP, and refractory TdP should be addressed with isoproterenol or temporary transvenous pacing to maintain the heart rate > 100 beats/minute [118].
Role of Serum Biomarkers
Currently, there is insufficient evidence regarding the utility of serum cardiac troponin, N-terminal pro-B-type natriuretic peptide (NT pro-BNP), or BNP levels for the surveillance of cardiotoxicity related to EGFR-TKIs [99]. Both the European Society of Cardiology and ESMO recommend obtaining baseline serum troponin I and NT-proBNP or BNP levels in high-risk patients or all patients starting potentially cardiotoxic therapies, although evidence in support of the practice is currently limited to observational studies [100••, 119]. Just as in all other patients, cardiac biomarkers should be obtained when clinical signs or symptoms of heart failure or myocardial infarction is suspected during treatment with EGFR-TKIs and should be considered in baseline assessment of patients with history of heart failure.
Role of Multimodality Imaging
Baseline echocardiography should be obtained before starting potentially cardiotoxic therapies and in high-risk patients (e.g., established CV disease, ≥ 3 CV risk factors, survivors of childhood or young adulthood cancers, prior chest or mediastinal radiotherapy, and prior cardiotoxic systemic therapy per ASCO and the American Society of Echocardiography [98, 120]. The best available method is recommended for evaluation (i.e., three-dimensional preferred over two-dimensional) and the modified Simpson’s biplane method is preferred with two-dimensional echocardiography [120]. Speckle tracking echocardiography with global longitudinal strain (GLS) is recommended for detection of subtle changes in LV contractility, and the same vendor and software should be utilized in each echocardiogram [120,121,122]. A GLS > − 16% is considered abnormal and indicative of subclinical LV dysfunction, and cancer therapeutics-related cardiac dysfunction is defined as a > 10% absolute reduction in EF from baseline or a relative reduction in GLS > 15% [122]. The utility of strain protocol echocardiography and optimal screening interval in asymptomatic patients receiving EGFR-TKIs remains ill-defined, although it should incorporate each patient’s risk as well as the risk of the specific chemotherapy [99, 100••, 122, 123].
Cardiac magnetic resonance imaging (CMR) is the most accurate modality for quantification of LVEF and provides further assessment of myocardial inflammation and tissue characterization compared to echocardiography. CMR should be considered in cases where echocardiogram images are suboptimal, situations where drug discontinuation is considered, and accurate quantitative assessment of LVEF is desired [97, 98, 120]. However, observational studies have yet to demonstrate a clear pattern in myocardial inflammation and fibrosis with EGFR-TKI cardiotoxicity [124].
Diagnosis and Management of Stable Ischemic Heart Disease and Myocardial Infarction
Certain EGFR-TKIs are associated with ischemic heart disease [20•, 21•, 28, 29, 53, 91] (Fig. 2, Table 1). Patients with CV risk factors should be considered for anatomic or functional ischemic evaluation for risk stratification and initiation of cardioprotective medications before commencing treatment with these agents [26]. Coronary artery calcium scanning involves non-contrast computed tomography (CT) imaging to evaluate coronary plaque burden and prognosticates risk of major adverse CV events in asymptomatic patients with low to intermediate pretest probability of CAD [125]. Coronary CT angiography utilizes ECG gating for noninvasive assessment of coronary arteriosclerosis in patients with stable angina and recommended every 5 years by the Society of Cardiovascular Angiography and Interventions for asymptomatic patients requiring cancer therapies associated with CAD [126].
Stress echocardiography and CMR with perfusion mapping is preferred over single-photon emission CT myocardial perfusion imaging for functional assessment of ischemia, as the former have been evaluated in the cardio-oncology population and offer additional information regarding subclinical LV dysfunction, contractile reserve, myocardial perfusion reserve, and myocardial scar burden [120, 127, 128]. Patients with ACS or an abnormal stress test should undergo cardiac catheterization to allow for therapeutic percutaneous coronary intervention of culprit lesions and physiologic assessment of intermediate lesions [126].
Management of Cardiomyopathy and Acute Decompensated Heart Failure
Echocardiography and cardiac biomarkers should be obtained in patients who are to receive lapatinib, neratinib, osimertinib, and vandetanib due to the increased risk of cardiomyopathy reported in clinical trials and post-marketing surveillance of these medications [20•, 21•, 27, 28, 30, 36•,119,120,121,122, 124, 127, 128]. Risk factors and comorbidities should be addressed at baseline and subsequent visits, although there is insufficient evidence to support starting cardioprotective medications such as beta blockers, angiotensin convertase enzyme inhibitors, or aldosterone receptor blockers pre-emptively in patients with normal LV function [100••]. Alternative agents should be considered in asymptomatic patients with a baseline EF < 50% or impaired GLS < − 16%, although lapatinib and neratinib may be continued in patients with an EF 40–49% who are initiated on cardioprotective medications and undergo close clinical and serial echocardiography every 3 months based on the SAFE-HEaRT protocol [129]. Treatment should be held in patients who develop heart failure and a cardiology consultation obtained for initiation of diuretics and guideline-directed medical therapy (GDMT) per current guidelines [101, 102].
Diagnosis and Management of Pericardial Disease
Pericardial effusions are not uncommon in patients with lung cancer and may be a clinical predictor of treatment resistance to EGFR-TKIs [130]. Pericarditis and cardiac tamponade have been reported with use of erlotinib, gefitinib, afatinib, and osimertinib, although these findings are likely confounded by progression of NSCLC, prior or concurrent radiotherapy, or immune checkpoint inhibitors [36•]. Patients with cardiac tamponade should undergo urgent pericardiocentesis and recurrent pericardial effusions warrant cardiothoracic surgery consultation regarding pericardial window [131].
Conclusions
EGFR/ErbB1 targeted therapy has improved outcomes across a range of solid malignancies. The spectrum of CV toxicity is variable and appears to be drug dependent. While infrequently reported in clinical trials, it is plausible that the true gamut of CV events with EGFR TKIs is underreported due to lack of pre-specified endpoints in clinical trials or trial exclusion of patients with pre-existing cardiac disease. Additional studies are needed to ascertain the true breadth of drug-specific cardiovascular toxicity. A balance of care and close collaboration between medical oncology and cardio-oncology is crucial in ensuring the minimization of unique, drug specific, adverse cardiovascular events outcomes that could result in significant mortality or morbidity, delayed oncologic treatment, or premature discontinuation of therapy.
Abbreviations
- ACS:
-
Acute coronary syndrome
- AE:
-
Adverse effects
- CAD:
-
Coronary artery disease
- CMR:
-
Cardiac magnetic resonance imaging
- CRC:
-
Colorectal cancer
- CT:
-
Computed tomography
- CV:
-
Cardiovascular
- ECG:
-
Electrocardiogram
- EGFR:
-
Epidermal growth factor receptor
- FDA:
-
United States Food and Drug Administration
- FOLFIRI:
-
Folinic acid, 5-fluorouracil, irinotecan
- GLS:
-
Global longitudinal strain
- HER:
-
Human epidermal growth factor receptor
- KRAS:
-
Kirsten rat sarcoma virus
- LV:
-
Left ventricle
- LVEF:
-
Left ventricular ejection fraction
- mAb:
-
Monoclonal antibody
- MET:
-
Mesenchymal epithelial transition factor
- MI:
-
Myocardial infarction
- NR:
-
Not reported
- NSCLC:
-
Non-small cell lung cancer
- RET:
-
Rearranged during transfection
- SCA:
-
Sudden cardiac arrest
- SCC:
-
Squamous cell carcinoma
- TdP:
-
Torsades de pointes
- TKI:
-
Tyrosine kinase inhibitor
- VEGF:
-
Vascular endothelial growth factor
References
Papers of particular interest, published recently, have been highlighted as: • Of importance •• Of major importance
Chen MH, Kerkelä R, Force T. Mechanisms of cardiac dysfunction associated with tyrosine kinase inhibitor cancer therapeutics. Circulation. 2008;118(1):84–95. https://doi.org/10.1161/CIRCULATIONAHA.108.776831.
Holbro T, Hynes NE. ErbB receptors: directing key signaling networks throughout life. Annu Rev Pharmacol Toxicol. 2004;44:195–217. https://doi.org/10.1146/annurev.pharmtox.44.101802.121440.
Hervent AS, De Keulenaer GW. Molecular mechanisms of cardiotoxicity induced by ErbB receptor inhibitor cancer therapeutics. Int J Mol Sci. 2012;13(10):12268–86. https://doi.org/10.3390/ijms131012268.
Yarden Y, Sliwkowski MX. Untangling the ErbB signalling network. Nat Rev Mol Cell Biol. 2001;2(2):127–37. https://doi.org/10.1038/35052073.
Ozcelik C, Erdmann B, Pilz B, et al. Conditional mutation of the ErbB2 (HER2) receptor in cardiomyocytes leads to dilated cardiomyopathy. Proc Natl Acad Sci U S A. 2002;99(13):8880–5. https://doi.org/10.1073/pnas.122249299.
Kitani T, Ong SG, Lam CK, et al. Human-induced pluripotent stem cell model of trastuzumab-induced cardiac dysfunction in patients with breast cancer. Circulation. 2019;139(21):2451–65. https://doi.org/10.1161/CIRCULATIONAHA.118.037357.
Lee KF, Simon H, Chen H, et al. Requirement for neuregulin receptor erbB2 in neural and cardiac development. Nature. 1995;378(6555):394–8. https://doi.org/10.1038/378394a0.
Gassmann M, Casagranda F, Orioli D, et al. Aberrant neural and cardiac development in mice lacking the ErbB4 neuregulin receptor. Nature. 1995;378(6555):390–4. https://doi.org/10.1038/378390a0.
Meyer D, Birchmeier C. Multiple essential functions of neuregulin in development. Nature. 1995;378(6555):386–90. https://doi.org/10.1038/378386a0.Erratum.In:Nature1995;378(6558):753.
Erickson SL, O’Shea KS, Ghaboosi N, et al. ErbB3 is required for normal cerebellar and cardiac development: a comparison with ErbB2-and heregulin-deficient mice. Development. 1997;124(24):4999–5011.
Schreier B, Döhler M, Rabe S, et al. Consequences of epidermal growth factor receptor (ErbB1) loss for vascular smooth muscle cells from mice with targeted deletion of ErbB1. Arterioscler Thromb Vasc Biol. 2011;31(7):1643–52. https://doi.org/10.1161/ATVBAHA.111.223537. This was a preclinical model of vascular smooth muscle isolated from mice aortas with EGFR deletion and abolished signaling. EGFR deletion led to diminished calcium signaling, decreased ERK1/2 activation in response to oxidative stress, and enhanced spontaneous cell death
Rajagopalan V, Zucker IH, Jones JA, et al. Cardiac ErbB-1/ErbB-2 mutant expression in young adult mice leads to cardiac dysfunction. Am J Physiol Heart Circ Physiol. 2008;295(2):H543–54. https://doi.org/10.1152/ajpheart.91436.2007. Blockade of ErbB-1 signaling in young adult mice. This led to blockade of ErbB-2 signaling, cardiac hypertrophy, chamber dilation, and depressed cardiac function, indicating the need of ErbB-1 signaling in normal cardiac homeostasis in adult mice.
Necela BM, Axenfeld BC, Serie DJ, et al. The antineoplastic drug, trastuzumab, dysregulates metabolism in iPSC-derived cardiomyocytes. Clin Transl Med. 2017;6:5. https://doi.org/10.1186/s40169-016-0133-2.
Lee HA, Hyun SA, Byun B, et al. Electrophysiological mechanisms of vandetanib-induced cardiotoxicity: comparison of action potentials in rabbit Purkinje fibers and pluripotent stem cell-derived cardiomyocytes. PLoS ONE. 2018;13(4):e0195577. https://doi.org/10.1371/journal.pone.0195577.
Dowell J, Minna J, Kirkpatrick P. Erlotinib hydrochloride. Nat Rev Drug Discovery. 2005;4(5):S14–5.
[Internet]. Accessdata.fda.gov. 2021 [cited 6 September 2021]. Available from: https://www.accessdata.fda.gov/drugsatfda_docs/label/2010/021743s14s16lbl.pdf.
[Internet]. European Medicine Agency. 2021 [cited 6 September 2021]. Available from: https://www.ema.europa.eu/en/documents/variation-report/tarceva-h-c-618-ii-0043-epar-assessment-report-variation_en.pdf
Petrelli F, Cabiddu M, Borgonovo K, et al. Risk of venous and arterial thromboembolic events associated with anti-EGFR agents: a meta-analysis of randomized clinical trials. Ann Oncol. 2012;23(7):1672–9. https://doi.org/10.1093/annonc/mdr592.
Stuhlmiller TJ, Zawistowski JS, Chen X, et al. Kinome and transcriptome profiling reveal broad and distinct activities of erlotinib, sunitinib, and sorafenib in the mouse heart and suggest cardiotoxicity from combined signal transducer and activator of transcription and epidermal growth factor receptor inhibition. J Am Heart Assoc. 2017;6(10):e006635. https://doi.org/10.1161/JAHA.117.006635.
Moore MJ, Goldstein D, Hamm J, National Cancer Institute of Canada Clinical Trials Group, et al. Erlotinib plus gemcitabine compared with gemcitabine alone in patients with advanced pancreatic cancer: a phase III trial of the National Cancer Institute of Canada Clinical Trials Group. J Clin Oncol. 2007;25(15):1960–6. https://doi.org/10.1200/JCO.2006.07.9525. A phase III trial comparing erlotinib/gemcitabine to placebo/gemcitabine in locally advanced or metastatic pancreatic adenocarcinoma. There was a higher incidence of coronary artery events reported in the treatment arm (2.3% vs 1.2%).
Zaborowska-Szmit M, Krzakowski M, Kowalski DM, et al. Cardiovascular complications of systemic therapy in non-small-cell lung cancer. J Clin Med. 2020;9(5):1268. https://doi.org/10.3390/jcm9051268A comprehensive review of cardiotoxicity with various treatment modalities in patients with NSCLC, including several approved EGFR TKIs, including Erlotinib, Gefitinib, Afatinib, and Osimertinib.
Cicènas S, Geater SL, Petrov P, et al. Maintenance erlotinib versus erlotinib at disease progression in patients with advanced non-small-cell lung cancer who have not progressed following platinum-based chemotherapy (IUNO study). Lung Cancer. 2016;102:30–7. https://doi.org/10.1016/j.lungcan.2016.10.007.
Bukowski RM, Kabbinavar FF, Figlin RA, et al. Randomized phase II study of erlotinib combined with bevacizumab compared with bevacizumab alone in metastatic renal cell cancer. J Clin Oncol. 2007;25(29):4536–41. https://doi.org/10.1200/JCO.2007.11.5154.
Johnson BE, Kabbinavar F, Fehrenbacher L, et al. ATLAS: randomized, double-blind, placebo-controlled, phase IIIB trial comparing bevacizumab therapy with or without erlotinib, after completion of chemotherapy, with bevacizumab for first-line treatment of advanced non-small-cell lung cancer. J Clin Oncol. 2013;31(31):3926–34. https://doi.org/10.1200/JCO.2012.47.3983.
Pinquié F, de Chabot G, Urban T, et al. Maintenance treatment by erlotinib and toxic cardiomyopathy: a case report. Oncology. 2016;90(3):176–7. https://doi.org/10.1159/000444186.
Kus T, Aktas G, Sevinc A, et al. Could erlotinib treatment lead to acute cardiovascular events in patients with lung adenocarcinoma after chemotherapy failure? Onco Targets Ther. 2015;8:1341–3. https://doi.org/10.2147/OTT.S84480.
Truell JS, Fishbein MC, Figlin R. Myocarditis temporally related to the use of gefitinib (Iressa). Arch Pathol Lab Med. 2005;129(8):1044–6. https://doi.org/10.5858/2005-129-1044-MTRTTU.
Lynch DR Jr, Kickler TS, et al. Recurrent myocardial infarction associated with gefitinib therapy. J Thromb Thrombolysis. 2011;32(1):120–4. https://doi.org/10.1007/s11239-010-0539-4.
Yamaguchi K, Kanazawa S, Kinoshita Y, et al. Acute myocardial infarction with lung cancer during treatment with gefitinib: the possibility of gefitinib-induced thrombosis. Pathophysiol Haemost Thromb. 2005;34(1):48–50. https://doi.org/10.1159/000088548.
Omori S, Oyakawa T, Naito T, et al. Gefitinib-induced cardiomyopathy in epidermal growth receptor-mutated NSCLC. J Thorac Oncol. 2018;13(10):e207–8. https://doi.org/10.1016/j.jtho.2018.05.020.
Nuvola G, Dall’Olio FG, Melotti B, et al. Cardiac toxicity from afatinib in EGFR-mutated NSCLC: a rare but possible side effect. J Thorac Oncol. 2019;14(7):e145–6. https://doi.org/10.1016/j.jtho.2019.02.027.
Mok TS, Wu Y-L, Ahn M-J, et al. Osimertinib or platinum-pemetrexed in EGFR T790M-positive lung cancer. N Engl J Med. 2017;376(7):629–40. https://doi.org/10.1056/NEJMoa1612674. A phase III trial assessing Osimertinib in T790M-positive advanced non–small-cell lung cancer, who had disease progression after first-line EGFR-TKI therapy. A decrease in LVEF ≥10% and an absolute LVEF <50% occurred in 5% of the osimertinib arm with a median time onset of 5.5 months. Furthermore, 3% of the osimertinib arm had QTc prolongation compared to 0% in the control arm.
Ramalingam SS, Vansteenkiste J, Planchard D, et al. Overall survival with osimertinib in untreated, EGFR-mutated advanced NSCLC. N Engl J Med. 2020;382(1):41–50. https://doi.org/10.1056/NEJMoa1913662. A phase III trial comparing frontline osimertinib to gefitinib or erlotinib for advanced NSCLC. In this study, 10% of osimertinib treated patients had QTc prolongation, with ≥ grade 3 QTc prolongation in 1%, and decreased LVEF occurred in 5% of osimertinib treated patients.
Wu YL, Tsuboi M, He J, et al. Osimertinib in resected EGFR-mutated non-small-cell lung cancer. N Engl J Med. 2020;383(18):1711–23. https://doi.org/10.1056/NEJMoa2027071. A phase III study assessing adjuvant Osimertinib compared to placebo in stage IB to IIIA NSCLC. The Osimertinib arm reported cardiac AE in 5% (reduced EF, cardiomyopathy, pulmonary edema and cardiomyopathy compared to 3% in the placebo group.
Kunimasa K, Kamada R, Oka T, et al. Cardiac adverse events in EGFR-mutated non-small cell lung cancer treated with osimertinib. JACC CardioOncol. 2020;2(1):1–10. https://doi.org/10.1016/j.jaccao.2020.02.003.
Anand K, Ensor J, Trachtenberg B, et al. Osimertinib-induced cardiotoxicity: a retrospective review of the FDA Adverse Events Reporting System (FAERS). JACC CardioOncol. 2019;1(2):172–8. https://doi.org/10.1016/j.jaccao.2019.10.006. A pharmacovigilance study that showed a statistically significant reporting odds ratio of heart failure, QTc prolongation and atrial fibrillation in Osimertinib treated patients compared to first and second-generation EGFR TKIs and total reported events in the database.
Waliany S, Zhu H, Wakelee H, et al. Pharmacovigilance analysis of cardiac toxicities associated with targeted therapies for metastatic NSCLC. J Thorac Oncol. 2021;S1556–0864(21):02377–87. https://doi.org/10.1016/j.jtho.2021.07.030.
[Internet]. Accessdata.fda.gov. 2021 [cited 20 September 2021]. Available from: https://www.accessdata.fda.gov/drugsatfda_docs/label/2021/215310s000lbl.pdf.
Saura C, Oliveira M, Feng YH, et al. Neratinib plus capecitabine versus lapatinib plus capecitabine in HER2-positive metastatic breast cancer previously treated with ≥ 2 HER2-directed regimens: phase III NALA Trial. J Clin Oncol. 2020;38(27):3138–49. https://doi.org/10.1200/JCO.20.00147. A phase III trial comparing neratinib/capecitabine to lapatinib/capecitabine. Arrhythmia (3.3% vs 3.5%), ischemic heart disease (0.7% vs 0.6%), QTc prolongation (2.3% vs 3.9%), and decreased LVEF (4.3% vs 2.3%) was reported in both treatment arms.
Piccart-Gebhart M, Holmes E, Baselga J, et al. Adjuvant lapatinib and trastuzumab for early human epidermal growth factor receptor 2-positive breast cancer: results from the randomized phase III adjuvant lapatinib and/or trastuzumab treatment optimization trial. J Clin Oncol. 2016;34(10):1034–42. https://doi.org/10.1200/JCO.2015.62.1797. Epub 2015 Nov 23. Erratum in: J Clin Oncol. 2019 Feb 1; 37(4):356.
Pivot X, Manikhas A, Żurawski B, et al. CEREBEL (EGF111438): A phase III, randomized, open-label study of lapatinib plus capecitabine versus trastuzumab plus capecitabine in patients with human epidermal growth factor receptor 2-positive metastatic breast cancer. J Clin Oncol. 2015;33(14):1564–73. https://doi.org/10.1200/JCO.2014.57.1794.
Chan A, Delaloge S, Holmes FA, et al. Neratinib after trastuzumab-based adjuvant therapy in patients with HER2-positive breast cancer (ExteNET): a multicentre, randomised, double-blind, placebo-controlled, phase 3 trial. Lancet Oncol. 2016;17(3):367–77. https://doi.org/10.1016/S1470-2045(15)00551-3A phase III trial assessing extended adjuvant neratinib in patients with HER2 positive breast cancer and had aready received adjuvant trastuzumab. In this study, QTc prolongation occurred in 3% and decreased LVEF in 1%.
Awada A, Colomer R, Inoue K, et al. Neratinib plus paclitaxel vs trastuzumab plus paclitaxel in previously untreated metastatic ERBB2-positive breast cancer: the NEfERT-T randomized clinical trial. JAMA Oncol. 2016;2(12):1557–64. https://doi.org/10.1001/jamaoncol.2016.0237.
Wells SA Jr, Robinson BG, Gagel RF, et al MJ. Vandetanib in patients with locally advanced or metastatic medullary thyroid cancer: a randomized, double-blind phase III trial. J Clin Oncol. 2012;30(2):134–41. https://doi.org/10.1200/JCO.2011.35.5040. Epub 2011 Oct 24. Erratum in: J Clin Oncol. 2013 Aug; 31 (24):3049. A phase III study of Vandetanic in patients with locally advanced or metastatic medullary thyroid cancer. Hypertension was reported in 32% and QTc prolongation in 14%, with SCA occurring in one patient.
Natale RB, Thongprasert S, Greco FA, et al. Phase III trial of vandetanib compared with erlotinib in patients with previously treated advanced non-small-cell lung cancer. J Clin Oncol. 2011;29(8):1059–66. https://doi.org/10.1200/JCO.2010.28.5981.
Kiura K, Nakagawa K, Shinkai T, et al. A randomized, double-blind, phase IIa dose-finding study of Vandetanib (ZD6474) in Japanese patients with non-small cell lung cancer. J Thorac Oncol. 2008;3(4):386–93. https://doi.org/10.1097/JTO.0b013e318168d228.
Liu Y, Liu Y, Fan ZW, et al. Meta-analysis of the risks of hypertension and QTc prolongation in patients with advanced non-small cell lung cancer who were receiving vandetanib. Eur J Clin Pharmacol. 2015;71(5):541–7. https://doi.org/10.1007/s00228-015-1831-1.
Zang J, Wu S, Tang L, et al. Incidence and risk of QTc interval prolongation among cancer patients treated with vandetanib: a systematic review and meta-analysis. PLoS ONE. 2012;7(2):e30353. https://doi.org/10.1371/journal.pone.0030353.
•• Paz-Ares L, Mezger J, Ciuleanu TE, et al. Necitumumab plus pemetrexed and cisplatin as first-line therapy in patients with stage IV non-squamous non-small-cell lung cancer (INSPIRE): an open-label, randomised, controlled phase 3 study. Lancet Oncol. 2015;16(3):328–37. https://doi.org/10.1016/S1470-2045(15)70046-X. A phase III study comparing cisplatin/pemetrexed/necitumumab to cisplatin/pemetrexed/placebo as first line in stage IV SCC NSCLC. In the treatment arm, pulmonary thromboembolism was reported in 5% and SCA was reported in 3% of the treatment arm.
Besse B, Garrido P, Cortot AB, et al. Efficacy and safety of necitumumab and pembrolizumab combination therapy in patients with Stage IV non-small cell lung cancer. Lung Cancer. 2020;142:63–9. https://doi.org/10.1016/j.lungcan.2020.02.003.
Thatcher N, Hirsch FR, Luft AV, et al. Necitumumab plus gemcitabine and cisplatin versus gemcitabine and cisplatin alone as first-line therapy in patients with stage IV squamous non-small-cell lung cancer (SQUIRE): an open-label, randomised, controlled phase 3 trial. Lancet Oncol. 2015;16(7):763–74. https://doi.org/10.1016/S1470-2045(15)00021-2.
Vermorken JB, Mesia R, Rivera F, et al. Platinum-based chemotherapy plus cetuximab in head and neck cancer. N Engl J Med. 2008;359(11):1116–27. https://doi.org/10.1056/NEJMoa0802656.
Sobrero A, Lenz HJ, Eng C, et al. Extended RAS Analysis of the phase III EPIC trial: irinotecan + cetuximab versus irinotecan as second-line treatment for patients with metastatic colorectal cancer. Oncologist. 2021;26(2):e261–9. https://doi.org/10.1002/onco.13591.
Ishiguro M, Watanabe T, Yamaguchi K, et al. A Japanese post-marketing surveillance of cetuximab (Erbitux®) in patients with metastatic colorectal cancer. Jpn J Clin Oncol. 2012;42(4):287–94. https://doi.org/10.1093/jjco/hys005. A post-marketing study of Japanese patients receiving cetuximab with our without chemotherapy for metastatic colorectal cancer. Cardiotoxicity occurred in 0.9% of 2,126 patients, with 4 deaths attributed to cardiotoxicity.
Gronich N, Lavi I, Barnett-Griness O, et al. Tyrosine kinase-targeting drugs-associated heart failure. Br J Cancer. 2017;116(10):1366–73. https://doi.org/10.1038/bjc.2017.88A nested case-control analysis that found both cetuximab and panitumumab had a higher odds of reported heartfailure, particulary in patients with history of diabetes mellitus, hypertension, chronic renal failure, ischemic heart disease, valvular heart disease, arrhythmia, and smoking.
Tang XM, Chen H, Li Q, et al. Assessment of the cardiac safety between cetuximab and panitumumab as single therapy in Chinese chemotherapy-refractory mCRC. Onco Targets Ther. 2017;11:123–9. https://doi.org/10.2147/OTT.S149716. A study to assess cardiac safety with either single agent cetuximab or panitumumab used as single agent in refractory metastatic colorectal cancer. No significant difference between the two treatment arms, but EKG changes, and troponin elevations were frequently reported.
Qi WX, Zhao S, Chen J. Risk factors for developing cardiac toxicities in cancer patients treated with panitumumab combination therapy. Future Oncol. 2020;16(19):1359–70. https://doi.org/10.2217/fon-2020-0050.
Boku N, Sugihara K, Kitagawa Y, et al. Panitumumab in Japanese patients with unresectable colorectal cancer: a post-marketing surveillance study of 3085 patients. Jpn J Clin Oncol. 2014;44(3):214–23. https://doi.org/10.1093/jjco/hyt196.
Park K, Haura EB, Leighl NB, et al. Amivantamab in EGFR Exon 20 Insertion–mutated non–small-cell lung cancer progressing on platinum chemotherapy: initial results from the CHRYSALIS phase I study. J Clin Oncol. 2021; JCO.21.00662. https://doi.org/10.1200/jco.21.00662. A phase I, open-label, dose-escalation, and dose-expansion study assessing amivantamab in patients with EGFR Exon 20 insertion mutations in NSCLC. In the safety population (n=114), there was one case of acute coronary syndrome and once case of atrial flutter.
Giaccone G. The role of gefitinib in lung cancer treatment. Clin Cancer Res. 2004;10(12 Pt 2):4233s–7s. https://doi.org/10.1158/1078-0432.CCR-040005 ([Internet]).
Accessdata.fda.gov. 2021 [cited 6 September 2021]. Available from: https://www.accessdata.fda.gov/drugsatfda_docs/label/2018/206995s003lbl.pdf-ADDED.
Maemondo M, Inoue A, Kobayashi K, et al. Gefitinib or chemotherapy for non-small-cell lung cancer with mutated EGFR. N Engl J Med. 2010;362(25):2380–8. https://doi.org/10.1056/NEJMoa0909530.
Wu YL, Cheng Y, Zhou X, et al. Dacomitinib versus gefitinib as first-line treatment for patients with EGFR-mutation-positive non-small-cell lung cancer (ARCHER 1050): a randomised, open-label, phase 3 trial. Lancet Oncol. 2017;18(11):1454–66. https://doi.org/10.1016/S1470-2045(17)30608-3.
Mitsudomi T, Morita S, Yatabe Y, et al. Gefitinib versus cisplatin plus docetaxel in patients with non-small-cell lung cancer harbouring mutations of the epidermal growth factor receptor (WJTOG3405): an open label, randomised phase 3 trial. Lancet Oncol. 2010;11(2):121–8. https://doi.org/10.1016/S1470-2045(09)70364-X.
Park K, Tan E, O’Byrne K, et al. Afatinib versus gefitinib as first-line treatment of patients with EGFR mutation-positive non-small-cell lung cancer (LUX-Lung 7): a phase 2B, open-label, randomised controlled trial. Lancet Oncol. 2016;17(5):577–89.
Yang JJ, Zhou Q, Yan HH, et al. A phase III randomised controlled trial of erlotinib vs gefitinib in advanced non-small cell lung cancer with EGFR mutations. Br J Cancer. 2017;116(5):568–74. https://doi.org/10.1038/bjc.2016.456.
Buza V, Rajagopalan B, Curtis AB. Cancer treatment-induced arrhythmias: focus on chemotherapy and targeted therapies. Circ Arrhythm Electrophysiol. 2017;10(8):e005443. https://doi.org/10.1161/CIRCEP.117.005443.
Brinkmeyer JK, Moore DC. Necitumumab for the treatment of squamous cell non-small cell lung cancer. J Oncol Pharm Pract. 2018;24(1):37–41. https://doi.org/10.1177/1078155216682365.
[Internet]. Accessdata.fda.gov. 2021 [cited 7 September 2021]. Available from: https://www.accessdata.fda.gov/drugsatfda_docs/label/2015/125547s000lbl.pdf.
Ewer MS, Patel K, O’Brien D, et al. Cardiac safety of afatinib: a review of data from clinical trials. Cardiooncology. 2015;1(1):3. https://doi.org/10.1186/s40959-015-0006-7.
Mok TS, Cheng Y, Zhou X, et al. Updated overall survival in a randomized study comparing dacomitinib with gefitinib as first-line treatment in patients with advanced non-small-cell lung cancer and EGFR-activating mutations. Drugs. 2021;81(2):257–66. https://doi.org/10.1007/s40265-020-01441-6.
Tan W, Giri N, Quinn S, et al. Evaluation of the potential effect of dacomitinib, an EGFR tyrosine kinase inhibitor, on ECG parameters in patients with advanced non-small cell lung cancer. Invest New Drugs. 2020;38(3):874–84. https://doi.org/10.1007/s10637-019-00887-0. A phase II study assessing the CV safety of Dacomitinib. There were no clinically relevant effect on QTc interval or other ECG parameters.
Pacheco J. Mobocertinib: a potential treatment for NSCLC with EGFR Exon 20 insertions. Cancer Discov. 2021;11(7):1617–9. https://doi.org/10.1158/2159-8290.CD-21-0379.
Riely GJ, Neal JW, Camidge DR, et al. Activity and safety of mobocertinib (TAK-788) in previously treated non-small cell lung cancer with EGFR Exon 20 Insertion Mutations from a Phase I/II Trial. Cancer Discov. 2021;11(7):1688–99. https://doi.org/10.1158/2159-8290.CD-20-1598.
Ramalingam S, Zhou C, Kim T, et al. Mobocertinib (TAK-788) in EGFR exon 20 insertion (ex20ins)+ metastatic NSCLC (mNSCLC): Additional results from platinum-pretreated patients (pts) and EXCLAIM cohort of phase 1/2 study. J Clin Oncol. 2021;39((15_suppl)):9014–9014. https://doi.org/10.1200/JCO.2021.39.15_suppl.9014.
Center for Drug Evaluation and Research. (2021, May 21). FDA grants accelerated approval to amivantamab-vmjw for metastatic non-small cell lung cancer. U.S. Food and Drug Administration. https://www.fda.gov/drugs/resources-information-approved-drugs/fda-grants-accelerated-approval-amivantamab-vmjw-metastatic-non-small-cell-lung-cancer.
Chan A, Moy B, Mansi J, et al. Final efficacy results of neratinib in HER2-positive hormone receptor-positive early-stage breast cancer from the phase III ExteNET Trial. Clin Breast Cancer. 2021;21(1):80-91.e7. https://doi.org/10.1016/j.clbc.2020.09.014.
US Food & Drug Administration: FDA approves neratinib for metastatic HER2-positive breast cancer. https://www.fda.gov/drugs/resources-information-approved-drugs/fda-approves-neratinib-metastatic-her2-positive-breast-cancer#:∼:text=On%20February%2025%2C%202020%2C%20the,regimens%20in%20the%20metastatic%20setting.
Cameron D, Casey M, Oliva C, et al. Lapatinib plus capecitabine in women with HER-2-positive advanced breast cancer: final survival analysis of a phase III randomized trial. Oncologist. 2010;15(9):924–34. https://doi.org/10.1634/theoncologist.2009-0181.
FDA Approves Tykerb for First-Line Combination Treatment of Metastatic Breast Cancer. https://www.drugs.com/newdrugs/gsk-s-tykerb-receives-accelerated-approval-first-line-combination-metastatic-breast-cancer-1979.html.
Johnston S, Pippen J Jr, Pivot X, et al. Lapatinib combined with letrozole versus letrozole and placebo as first-line therapy for postmenopausal hormone receptor-positive metastatic breast cancer. J Clin Oncol. 2009;27(33):5538–46. https://doi.org/10.1200/JCO.2009.23.3734.
Ando K, Wada T, Cao X. Precise safety pharmacology studies of lapatinib for onco-cardiology assessed using in vivo canine models. Sci Rep. 2020; 10 (738). https://doi.org/10.1038/s41598-020-57601-x.
Perez EA, Koehler M, Byrne J, et al. Cardiac safety of lapatinib: pooled analysis of 3689 patients enrolled in clinical trials. Mayo Clin Proc. 2008;83(6):679–86. https://doi.org/10.4065/83.6.679.
Diéras V, Miles D, Verma S, et al. Trastuzumab emtansine versus capecitabine plus lapatinib in patients with previously treated HER2-positive advanced breast cancer (EMILIA): a descriptive analysis of final overall survival results from a randomised, open-label, phase 3 trial. Lancet Oncol. 2017;18(6):732–742. https://doi.org/10.1016/S1470-2045(17)30312-1. Epub 2017 May 16. Erratum in: Lancet Oncol. 2017 Aug;18(8):e433. Erratum in: Lancet Oncol. 2018 Aug; 19(12):e667.
Choi HD, Chang MJ. Cardiac toxicities of lapatinib in patients with breast cancer and other HER2-positive cancers: a meta-analysis. Breast Cancer Res Treat. 2017;166(3):927–36. https://doi.org/10.1007/s10549-017-4460-9.
Dogan E, Yorgun H, Petekkaya I, et al. Evaluation of cardiac safety of lapatinib therapy for ErbB2-positive metastatic breast cancer: a single center experience. Med Oncol. 2012;29(5):3232–9. https://doi.org/10.1007/s12032-012-0253-5.
Eiger D, Pondé NF, Agbor-Tarh D, et al. Long-term cardiac outcomes of patients with HER2-positive breast cancer treated in the adjuvant lapatinib and/or trastuzumab treatment optimization trial. Br J Cancer. 2020;122(10):1453–60. https://doi.org/10.1038/s41416-020-0786-x. A subanalysis assessing cardiac outcomes in patients receiving either adjuvant lapatinib alone or lapatinib/trastuzumab combination. Reported cardiac events were not significant between the two treatment arms and the addition of lapatinib to trastuzumab did not appear to substantially increase the incidence of cardiac events.
Van Cutsem E, Köhne CH, Hitre E, et al. Cetuximab and chemotherapy as initial treatment for metastatic colorectal cancer. N Engl J Med. 2009;360(14):1408–17. https://doi.org/10.1056/NEJMoa0805019.
Sobrero AF, Maurel J, Fehrenbacher L, et al. EPIC: phase III trial of cetuximab plus irinotecan after fluoropyrimidine and oxaliplatin failure in patients with metastatic colorectal cancer. J Clin Oncol. 2008;26(14):2311–9. https://doi.org/10.1200/JCO.2007.13.1193.
Cunningham D, Humblet Y, Siena S, et al. Cetuximab monotherapy and cetuximab plus irinotecan in irinotecan-refractory metastatic colorectal cancer. N Engl J Med. 2004;351(4):337–45. https://doi.org/10.1056/NEJMoa033025.
Bonner JA, Harari PM, Giralt J, et al. Radiotherapy plus cetuximab for squamous-cell carcinoma of the head and neck. N Engl J Med. 2006;354(6):567–78. https://doi.org/10.1056/NEJMoa053422.
Jonker DJ, O’Callaghan CJ, Karapetis CS, et al. Cetuximab for the treatment of colorectal cancer. N Engl J Med. 2007;357(20):2040–8. https://doi.org/10.1056/NEJMoa071834.
Bossi P, Miceli R, Locati LD, et al. A randomized, phase 2 study of cetuximab plus cisplatin with or without paclitaxel for the first-line treatment of patients with recurrent and/or metastatic squamous cell carcinoma of the head and neck. Ann Oncol. 2017;28(11):2820–6. https://doi.org/10.1093/annonc/mdx439.
Rogers JE. Patient considerations in metastatic colorectal cancer - role of panitumumab. Onco Targets Ther. 2017;10:2033–44. https://doi.org/10.2147/OTT.S115430.
U.S. Food and Drug Administration (2011). CAPRELSA® (vandetanib) Tablets, [Online]. https://www.accessdata.fda.gov/drugsatfda_docs/label/2014/022405s007lbl.pdf. Accessed 9/1/2021].
Koene RJ, Prizment AE, Blaes A, et al. Shared risk factors in cardiovascular disease and cancer. Circulation. 2016;133:1104–14. https://doi.org/10.1161/CIRCULATIONAHA.115.020406.
Zamorano JL, Lancellotti P, Rodriguez Munoz D, et al. 2016 ESC Position Paper on cancer treatments and cardiovascular toxicity developed under the auspices of the ESC Committee for Practice Guidelines: the task force for cancer treatments and cardiovascular toxicity of the European Society of Cardiology (ESC). Eur Heart J. 2016;37:2768–801. https://doi.org/10.1093/eurheartj/ehw211.
Armenian SH, Lacchetti C, Barac A, et al. Prevention and monitoring of cardiac dysfunction in survivors of adult cancers: American Society of Clinical Oncology Clinical Practice Guideline. J Clin Oncol. 2017;35:893–911. https://doi.org/10.1200/JCO.2016.70.5400.
Alexandre J, Cautela J, Ederhy S, et al. Cardiovascular toxicity related to cancer treatment: a pragmatic approach to the American and European Cardio-Oncology Guidelines. J Am Heart Assoc. 2020;9:e018403. https://doi.org/10.1161/JAHA.120.018403.
Curigliano G, Lenihan D, Fradley M, et al. Management of cardiac disease in cancer patients throughout oncological treatment: ESMO consensus recommendations. Ann Oncol. 2020;31:171–90. https://doi.org/10.1016/j.annonc.2019.10.023. Updated ESMO consensus recommendations for patients who should receive screening EKG, echocardiogram for structural assessment, and patients who are candidates for primary prevention.
Dent SF, Kikuchi R, Kondapalli L, et al. Optimizing cardiovascular health in patients with cancer: a practical review of risk assessment, monitoring, and prevention of cancer treatment-related cardiovascular toxicity. Am Soc Clin Oncol Educ Book. 2020;40:1–15. https://doi.org/10.1200/EDBK_286019.
Cohen JB, Geara AS, Hogan JJ, Townsend RR. Hypertension in cancer patients and survivors: epidemiology, diagnosis, and management. JACC CardioOncol. 2019;1:238–51. https://doi.org/10.1016/j.jaccao.2019.11.009.
Stamler J, Wentworth D, Neaton JD. Is relationship between serum cholesterol and risk of premature death from coronary heart disease continuous and graded? Findings in 356,222 primary screenees of the Multiple Risk Factor Intervention Trial (MRFIT). JAMA. 1986;256:2823–8.
Shaten BJ, Kuller LH, Kjelsberg MO, et al. Lung cancer mortality after 16 years in MRFIT participants in intervention and usual-care groups. Multiple Risk Factor Intervention Trial. Ann Epidemiol. 1997;7:125–36. https://doi.org/10.1016/s1047-2797(96)00123-8.
Arnett DK, Blumenthal RS, Albert MA, et al. 2019 ACC/AHA Guideline on the primary prevention of cardiovascular disease: executive summary. J Am Coll Cardiol. 2019;74:1376–414. https://doi.org/10.1161/CIR.0000000000000677.
Seidman A, Hudis C, Pierri MK, et al. Cardiac dysfunction in the trastuzumab clinical trials experience. J Clin Oncol. 2002;20:1215–21. https://doi.org/10.1200/JCO.2002.20.5.1215.
Trachtenberg BH, Landy DC, Franco VI, et al. Anthracycline-associated cardiotoxicity in survivors of childhood cancer. Pediatr Cardiol. 2011;32:342–53.
Armenian S, Bhatia S. Predicting and preventing anthracycline-related cardiotoxicity. Am Soc Clin Oncol Educ Book. 2018;38:3–12. https://doi.org/10.1007/s00246-010-9878-3.
Salem JE, Manouchehri A, Moey M, et al. Cardiovascular toxicities associated with immune checkpoint inhibitors: an observational, retrospective, pharmacovigilance study. Lancet Oncol. 2018;19:1579–89. https://doi.org/10.1016/S1470-2045(18)30608-9.
Touyz RM, Herrmann J. Cardiotoxicity with vascular endothelial growth factor inhibitor therapy. NPJ Precision Oncology 2018; 2. https://doi.org/10.1038/s41698-018-0056-z.
Drobni ZD, Alvi RM, Taron J, et al. Association between immune checkpoint inhibitors with cardiovascular events and atherosclerotic plaque. Circulation. 2020;142:2299–311. https://doi.org/10.1161/CIRCULATIONAHA.120.049981.
Chitturi KR, Xu J, Araujo-Gutierrez R, et al. Immune checkpoint inhibitor-related adverse cardiovascular events in patients with lung cancer. JACC: CardioOncology. 2019;1:182–92. https://doi.org/10.1016/j.jaccao.2019.11.013.
Darby SC, Ewertz M, McGale P, et al. Risk of ischemic heart disease in women after radiotherapy for breast cancer. N Engl J Med. 2013;368:987–98. https://doi.org/10.1056/NEJMoa1209825.
Gujral DM, Cleator SJ, Bhattacharyya S. Cardiac safety evaluation in cancer clinical trials. Eur J Cancer. 2018;103:143–6. https://doi.org/10.1016/j.ejca.2018.07.141.
Lancellotti P, Suter TM, Lopez-Fernandez T, et al. Cardio-oncology services: rationale, organization, and implementation. Eur Heart J. 2019;40:1756–63. https://doi.org/10.1093/eurheartj/ehy453.
Alexandre J, Salem JE, Moslehi J, et al. Identification of anticancer drugs associated with atrial fibrillation: analysis of the WHO pharmacovigilance database. Eur Heart J Cardiovasc Pharmacother. 2021;7:312–20. https://doi.org/10.1093/ehjcvp/pvaa037.
Steffel J, Verhamme P, Potpara TS, et al. The 2018 European Heart Rhythm Association Practical Guide on the use of non-vitamin K antagonist oral anticoagulants in patients with atrial fibrillation. Eur Heart J. 2018;39:1330–93. https://doi.org/10.1093/eurheartj/ehy136.
Sondergaard L, Wong YH, Reddy VY, et al. Propensity-matched comparison of oral anticoagulation versus antiplatelet therapy after left atrial appendage closure with WATCHMAN. JACC Cardiovasc Interv. 2019;12:1055–63. https://doi.org/10.1016/j.jcin.2019.04.004.
Pudil R, Mueller C, Celutkiene J, et al. Role of serum biomarkers in cancer patients receiving cardiotoxic cancer therapies: a position statement from the Cardio-Oncology Study Group of the Heart Failure Association and the Cardio-Oncology Council of the European Society of Cardiology. Eur J Heart Fail. 2020;22:1966–83. https://doi.org/10.1002/ejhf.2017.
Plana JC, Galderisi M, Barac A, et al. Expert consensus for multimodality imaging evaluation of adult patients during and after cancer therapy: a report from the American Society of Echocardiography and the European Association of Cardiovascular Imaging. J Am Soc Echocardiogr. 2014;27:911–39. https://doi.org/10.1016/j.echo.2014.07.012.
Voigt JU, Pedrizzetti G, Lysyansky P, et al. Definitions for a common standard for 2D speckle tracking echocardiography: consensus document of the EACVI/ASE/Industry Task Force to standardize deformation imaging. J Am Soc Echocardiogr. 2015;28:183–93. https://doi.org/10.1093/ehjci/jeu184.
Liu JE, Barac A, Thavendiranathan P, Scherrer-Crosbie M. Strain imaging in cardio-oncology. JACC: CardioOncology. 2020;2:677–89. https://doi.org/10.1016/j.jaccao.2020.10.011.
Porta-Sanchez A, Gilbert C, Spears D et al. Incidence, diagnosis, and management of QT prolongation induced by cancer therapies: a systematic review. J Am Heart Assoc 2017; 6. https://doi.org/10.1161/JAHA.117.007724.
Harries I, Liang K, Williams M, et al. Magnetic resonance imaging to detect cardiovascular effects of cancer therapy: JACC CardioOncology state-of-the-art review. JACC CardioOncol. 2020;2:270–92. https://doi.org/10.1016/j.jaccao.2020.04.011.
Detrano R, Guerci AD, Carr JJ, et al. Coronary calcium as a predictor of coronary events in four racial or ethnic groups. N Engl J Med. 2008;358:1336–45. https://doi.org/10.1056/NEJMoa072100.
Iliescu CA, Grines CL, Herrmann J, et al. SCAI Expert consensus statement: evaluation, management, and special considerations of cardio-oncology patients in the cardiac catheterization laboratory (endorsed by the Cardiological Society of India, and sociedad Latino Americana de Cardiologia intervencionista). Catheter Cardiovasc Interv. 2016;87:E202–23. https://doi.org/10.1002/ccd.26379.
Celutkiene J, Pudil R, Lopez-Fernandez T, et al. Role of cardiovascular imaging in cancer patients receiving cardiotoxic therapies: a position statement on behalf of the Heart Failure Association (HFA), the European Association of Cardiovascular Imaging (EACVI) and the Cardio-Oncology Council of the European Society of Cardiology (ESC). Eur J Heart Fail. 2020;22:1504–24. https://doi.org/10.1002/ejhf.1957.
Biersmith MA, Tong MS, Guha A, et al. Multimodality cardiac imaging in the era of emerging cancer therapies. J Am Heart Assoc. 2020;9:e013755. https://doi.org/10.1161/JAHA.119.013755.
Lynce F, Barac A, Geng X, et al. Prospective evaluation of the cardiac safety of HER2-targeted therapies in patients with HER2-positive breast cancer and compromised heart function: the SAFE-HEaRt study. Breast Cancer Res Treat. 2019;175:595–603. https://doi.org/10.1007/s10549-019-05191-2.
Hiranuma O, Uchino J, Yamada T, et al. Rationale and design of a phase II trial of osimertinib combined with bevacizumab in patients with untreated epidermal growth factor receptor-mutated non-small-cell lung cancer and malignant pleural and/or pericardial effusion (SPIRAL II Study). Clin Lung Cancer. 2019;20:e402–6. https://doi.org/10.1016/j.cllc.2019.02.016.
Chiabrando JG, Bonaventura A, Vecchie A, et al. Management of acute and recurrent pericarditis: JACC state-of-the-art review. J Am Coll Cardiol. 2020;75:76–92. https://doi.org/10.1016/j.jacc.2019.11.021.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
This article is part of the Topical collection on Cardio-oncology
Rights and permissions
About this article
Cite this article
Chitturi, K.R., Burns, E.A., Muhsen, I.N. et al. Cardiovascular Risks with Epidermal Growth Factor Receptor (EGFR) Tyrosine Kinase Inhibitors and Monoclonal Antibody Therapy. Curr Oncol Rep 24, 475–491 (2022). https://doi.org/10.1007/s11912-022-01215-1
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11912-022-01215-1