Abstract
Purpose
Analysis and comparison of the helminth assemblages in Antarctic rockcod Notothenia coriiceps collected near the UAS “Akademik Vernadsky” (Argentine Islands, West Antarctica) in 2002 and 2014–2015 were performed to characterise the parasite community and investigate the temporal changes in helminth assemblages and infection parameters.
Methods
All specimens of N. coriiceps (n = 194) were caught at depths of 10–30 m. Parasites (22,856 helminth specimens and 15,057 cysts) were collected manually and identified based on their morphology. Statistical analysis of the quantitative data was performed using the Quantitative Parasitology 3.0 (QP 3.0), Paleontological Statistics (PAST v. 3.1), and PRIMER 6 software.
Results
Twenty-seven species of four taxonomic groups were recorded: trematodes (8 species), cestodes (4), nematodes (5), and acanthocephalans (10). Helminth samples collected in 2002 and 2014–2015 showed a rather high similarity in species composition. The species richness was higher in the sample collected in 2014–2015, while the evenness and diversity in the two samples were similar. The dissimilarity between helminth infracommunities in the two samples appeared to be statistically significant. Larval cestodes Diphyllobotrium sp., the acanthocephalan Metacanthocephalus rennicki, and the trematode Neoleoburia antarctica were found to make the most significant impact on the dissimilarity.
Conclusions
The analysis of the composition and structure of helminth community in N. coriiceps revealed the changes that have happened during the last decade. At least some of the changes are attributed to the changes in marine ecosystems in Western Antarctica.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Parasites of Antarctic fish from various parts of the Southern Ocean have been studied for over 150 years [1]. During this time, more than 100 papers on metazoan parasites of Antarctic fishes have been published; presently, the list of parasites includes nearly 250 species found in 142 species of host fishes [2]. Most of the publications were devoted to the study of the taxonomy of specific parasite groups [3,4,5,6,7,8,9,10,11] or to the studies of parasites from individual host species [12,13,14,15,16,17]. The most studied are the parasites of economically important fish species; attempts have been made to use parasites as biological tags to identify fish stocks [18,19,20,21,22,23,24]. However, despite a large number of parasitological publications [see 2], there are still very few works on the temporal changes in the parasite communities of separate species of Antarctic fishes [17, 24, 25].
A number of metazoan parasites of fish, particularly helminths, have complex life cycles involving various species of invertebrate and vertebrate hosts. Due to this, these parasites can be used as sensitive indicators of ecological and environmental changes in marine ecosystems [26,27,28,29,30]. The analysis of changes in the species diversity and structure of parasite communities of various animal hosts in Antarctic ecosystems has drawn more attention in recent decades [1, 31]. Since most species of Antarctic birds and mammals are under protection [32], the investigations of their parasites are restricted. At the same time, the current state of Antarctic fish populations allows their commercial fishing and scientific research. Many species of Antarctic birds and mammals are migratory and acquire parasites from elsewhere; while teleost fishes are restricted to the Antarctic waters, and, thus, best suited to assess the local helminth fauna. Numerous non-commercial fish species, such as Antarctic black rockcod, Notothenia coriiceps Richardson, 1844, are promising objects for long-term monitoring studies of parasite communities. Notothenia coriiceps is abundant in different parts of the Southern Ocean, including West Antarctica [33,34,35]. Studies in recent decades showed that the stock of N. coriiceps did not demonstrate any tendency of decreasing [33, 36]. The black rockcod inhabits shallow waters and may be easily caught with simple equipment (fishing rods, nets). Therefore, there are abundant data on its parasite fauna in different parts of the Southern Ocean [11, 13, 17, 24, 37,38,39]. This allows us to consider N. coriiceps as one of the most suitable fish species for long-term studies of the parasite community state and to use its parasites as indicators for monitoring of Antarctic marine ecosystems [31].
Detailed studies of the helminth species diversity in N. coriiceps from the waters of the Ukrainian Antarctic Station (UAS) “Akademik Vernadsky” area (Galindez Island, Argentine Islands, West Antarctica) were carried out twice: in 2002 [13] and 2014–2015 [17]. In total, 28 helminth species were found to parasitize N. coriiceps. Preliminary comparison of the published data collected in 2002 and the data collected in 2014–2015 showed some changes in the prevalence and intensity of fish infection by several helminth species over 12 years [17]. However, the detailed analysis of all these data on the infracommunity or component community levels has not been performed yet. Recent complementary study of these two samples made it possible to carry out their comprehensive comparative analysis.
The aim of the present study was to characterise the helminth populations and communities in N. coriiceps from the area near the UAS “Akademik Vernadsky” (Argentine Islands, West Antarctica) in 2002 and 2014–2015 and investigate the possible changes in helminth assemblages and infection parameters.
Materials and Methods
Helminthological data analysed in the present study were collected in coastal waters of Galindez Island, Argentine Islands Archipelago, Western Antarctica near the Ukrainian Antarctic station “Akademik Vernadsky” (65°15′ S, 64°16′ W) during the Ukrainian Antarctic expeditions in February–March 2002 and from April 2014 till January 2015. All specimens of N. coriiceps used in the analysis (93 specimens collected in 2002 and 103 specimens collected in 2014–2015) were caught using a fishing rod at the shore of the Galindez Island at depths from 10 to 30 m. All fishes collected were immediately transported to the laboratory where they were measured and examined using the standard parasitological techniques [13, 17]. In the combined sample (n = 196), the fish total body length (TBL) was 29.4 ± 7.4SD (11.8–48.0) cm; it was 26.4 ± 7.9SD (11.8–48.0) cm in the sample collected in 2002, and 32.2 ± 5.8 (21.5–44.5) cm in the sample collected in 2014–2015.
Parasites were collected manually from the skin, body cavity, stomach, intestine, liver, and mesentery. All collected parasites were washed in saline and fixed in 70% ethanol. Acanthocephalans were kept in tap water for 30 min to 3 h for proboscis evagination prior to their fixation in ethanol. Identification of the parasites was performed in the laboratories of the W. Stefansky Institute of Parasitology in Warsaw, Poland, and in the I. I. Schmalhausen Institute of Zoology in Kyiv, Ukraine, under light microscopes using the morphological criteria. Totally, 14,458 helminth specimens and 7460 cysts were collected in 2002 [13] and 8398 helminth specimens and 7597 cysts were collected in 2014–2015 [17]. All helminth specimens were deposited in the parasitological collections of the Institute of Parasitology in Warsaw, Poland and in the Institute of Zoology in Kyiv, Ukraine.
The prevalence, the mean abundance, and the intensity of infection were calculated for each helminth species in each of the two samples, except Corynosoma spp. collected in 2014–2015. Confidence intervals (95%) were identified for the prevalence of infection and mean abundance using the Quantitative Parasitology 3.0 (QP 3.0) software [40]. Prevalence of fish infection with separate helminth species in the two samples was compared using the unconditional exact test [41], while the mean abundance and mean intensity of infection were compared using the bootstrap-t test in the QP 3.0 following the recommendations of Rózsa et al. [40].
In the analysis of helminth communities’ parameters, we excluded the fish specimens with TBL < 20 cm from the sample. All excluded specimens were from the sample collected in 2002. Therefore, we used the data on helminths’ abundance and occurrence in 73 host specimens collected in 2002 and 103 hosts collected in 2014–2015 for comparative analysis of helminth communities. The comparison of species richness and abundance in the helminth infracommunities was performed using the Mann–Whitney test for equal medians in the Paleontological Statistics Software (PAST v. 3.1) [42]. The estimated species richness in the helminth component community was identified separately in the two samples using Chao1, jackknife, and bootstrap methods in the PRIMER 6 software [43]. The same program was used to calculate the diversity indices in the helminth component communities and to identify the similarities/differences between the helminth infracommunities using the Bray–Curtis index of similarity, nMDS (non-metric multidimensional scaling), ANOSIM (analysis of similarity), and SIMPER (similarity percentage) routines. The similarity matrix based on the Bray–Curtis index was composed in PRIMER 6 after the ‘log(x + 1)’ transformation of initial data on helminth species abundance in separate infracommunities, and ‘sample by total’ standardisation.
Interspecific associations and covariations of helminth species in the infracommunities were analysed using the procedure described in Ludwig and Reynolds [44] in the combined sample of hosts (n = 196). The associations were identified based on the Chi-square test, and the covariations were estimated based on Spearman’s coefficient of correlation (rS). All calculations were performed in PAST v. 3.1 [42].
Results
Helminth Community Composition and Parameters of Infection
All fish specimens examined in 2002 and 2014–2015 were found infected with helminths. Collected helminths were assigned to 27 species of four taxonomic groups: trematodes, cestodes, nematodes, and acanthocephalans (Table 1). Eight species of trematodes were all represented by adults (maritae) in the digestive tract. Four cestode species were found as the larval stage (metacestodes) in the digestive tract; one was identified as Diphyllobothrium sp.; three were assigned to the family Tetrabothriidae and identified as monolocular, bilocular, and trilocular metacestodes based on their distinctive morphology. Cysts with metacestodes were not examined separately. Two nematode species, Ascarophis nototheniae Johnston et Mawson, 1945 and Dichelyne fraseri (Baylis, 1929), were found on adult stage in the digestive tract; three species of anisakid nematodes were represented by the third-stage larvae located mostly on the liver and in the body cavity (Table 1). Five out of ten species of acanthocephalans were represented by adult worms in the host intestine; five species of Corynosoma Lühe, 1904 were the cystacanths found free in the intestine and encysted in the body cavity.
Twenty helminth species were found in the sample collected in 2002; the trematode Derogenes johnstoni Prudhoe et Bray, 1973, monolocular metacestodes, the acanthocephalans Metacanthocephalus campbelli (Leiper and Atkinson, 1914), M. rennicki (Leiper and Atkinson, 1914), and Corynosoma evae Zdzitowiecki, 1984, the nematodes Dichelyne fraseri (Baylis, 1929) and Anisakis sp. were not recorded. However, the presence of the latter nematode species appeared to be statistically insignificant in the sample collected in 2014–2015, for the minimum limit of the confidence interval of its prevalence (0.36%) was lower than the minimum possible prevalence in the sample (0.99%) (see Table 1). Therefore, the absence of Anisakis sp. in 2002 and its presence in 2014–2015 is not considered as a significant difference between the samples. The absence of Corynosoma bullosum (Linstow, 1892) and C. shackletoni Zdzitowiecki, 1978 in the sample collected in 2014–2015 is uncertain, for not all the larval acanthocephalans were excysted and identified in the sample. If the two latter species are excluded from the calculations, the similarity between the samples based on the presence/absence of species is estimated as rather high: Jaccard index 0.72 and Sørensen index 0.84.
Statistical comparison of the infection prevalence of helminths in two samples showed that it differed significantly in seven species (Table 1). The prevalence of infection was significantly higher in the nematodes A. nototheniae and Pseudoterranova sp., the acanthocephalans Aspersentis megarhynchus (Linstow, 1892) and Metacanthocephalus johnstoni Zdzitowiecki, 1983, and the cestode Diphyllobothrium sp. in 2014–2015 compared to that in 2002. On the contrary, the prevalence of infection of the trematode Neolebouria antarctica (Szidat et Graefe, 1967) and of bilocular metacestodes was significantly higher in 2002. Three species had no differences in the infection prevalence in two samples: the nematode Contracaecum sp., and the trematodes Lecithaster macrocotyle Szidat et Graefe, 1967 and Macvicaria microtestis Zdzitowiecki and Cielecka, 1997. On the other hand, the latter two species were statistically insignificant in both samples based on the confidence intervals of the infection prevalence (Table 1); therefore, the similarity in their occurrence is uncertain. In other helminth species, the differences in infection prevalence were not statistically significant in the two samples.
Statistically significant differences in the abundance and infection intensity in the samples collected in 2002 and 2014–2015 were recorded in the same species which had the differences in the infection prevalence (Table 1). The only exception is the acanthocephalan Aspersentis austrinus Van Cleave, 1929 (= A. megarhynchus in [13]), which had similar abundance and infection intensity in two samples, though its infection prevalence differed significantly.
Characterisation of Helminth Communities
In the statistical comparison of helminth communities, we combined five acanthocephalan species of the genus Corynosoma in each of the two samples, since not all the cystacanths were identified in the sample collected in 2014–2015. Therefore, all the cystacanths in both samples were treated as a separate unit, Corynosoma spp. in further calculations. The species richness in the helminth component community in 2002 was 17 species. The estimated species richness did not differ dramatically from the observed one, it equalled 17 (Chao1), 19 (jackknife), or 18 (bootstrap). In 2014–2015, the observed species richness was 24, while the estimated species richness was 24 (Chao1), 27 (jackknife), or 25 (bootstrap). Margalef’s index of species richness was expectedly lower in 2002 (1.68) than in 2014–2015 (2.38). On the other hand, the evenness in the community estimated by Pielou’s index was almost identical in the two samples: 0.57 in 2002 vs. 0.56 in 2014–2015. Consequently, the diversity indices were rather similar: Shannon index was 1.60 in 2002 vs. 1.77 in 2014–2015; Simpson index was 0.68 in 2002 vs. 0.72 in 2014–2015.
In the sample of N. coriiceps collected in 2002 (n = 73), the species richness in helminth infracommunities, i.e., the number of helminth species in a host individual, ranged from 2 to 13 species; the mean number of species in an infracommunity was 7.7 with a median of 8.0 species. In the sample collected in 2014–2015 (n = 101), the species richness in helminth infracommunities ranged from 3 to 14 species; the mean number of species was 8.7 with a median of 9.0 species. A significant difference between two samples in median values of species richness in helminth infracommunities was confirmed by the Mann–Whitney test: U = 2734.5; z = − 3.1067; p = 0.002. The abundance in helminth infracomunities, i.e., the number of specimens in a host individual, ranged from 26 to 813 in the sample collected in 2002, and from 9 to 467 in the sample collected in 2014–2015. The mean value of the abundance was 190.2 in 2002 and 152.3 in 2014–2015; the median value was 140 in 2002 and 136 in 2014–2015. According to the Mann–Whitney test, the difference in abundance between the two samples was not significant (p = 0.72).
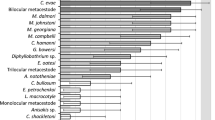
We estimated the similarities between helminth infracommunities using the Bray–Curtis index. The results were visualised by nMDS plot (Fig. 1). For the majority of infracommunities (all except 4), the distance based on the Bray–Curtis index was within 0.5. On the other hand, samples collected in 2002 and 2014–2015 formed two distinct groups on the plot. ANOSIM routine confirmed a moderate and significant dissimilarity between the groups: ANOSIM R = 0.47, p = 0.001. Average similarity within each of the two groups identified by SIMPER analysis was almost identical: 63.8% in the sample collected in 2002 and 63.9% in the sample collected in 2014–2015, while the average dissimilarity between the groups was 45.7%. According to SIMPER, the following ten helminth species had the largest contribution (81.5% in total) to the dissimilarity between the samples: Corynosoma spp. (contribution 11.33%), M. rennicki (10.67%), M. georgiana (10.19%), bilocular metacestodes (8.69%), M. johnstoni (8.03%), Diphyllobothrium sp. (7.98%), Pseudoterranova sp. (7.84%), G. bowersi (6.92%), E. oatesi (5.19%), and N. antarctica (4.67%).
Since the samples were collected in different seasons: in February and March of 2002 and from April 2014 till January 2015, we analysed seasonal changes in the prevalence and abundance of ten helminth species contributing to the differences between the samples. The sample collected in 2014–2015 was divided into five subsamples, each containing hosts collected during 2 months, and the prevalence and abundance of each species were calculated in each subsample and compared to those in the sample collected in 2002 (Table 2).
Significant differences in prevalence and abundance between the sample collected in February–March 2002 and all the subsamples collected in 2014–2015 were observed in M. rennicki (the species was absent in 2002 and had high prevalence and abundance in 2014–2015), Diphyllobothrium sp. (both prevalence and abundance were higher in 2014–2015), and N. antarctica (absence or low infection rates in 2014–2015 vs. significantly higher infection prevalence and abundance in 2002). Bilocular metacestodes had significantly lower infection prevalence from April till November in 2014 than those in February–March of 2002, while the prevalence was similar in December–January 2014–2015 and in 2002. On the other hand, the abundance of bilocular metacestodes was significantly lower in all subsamples collected in 2014–2015 compared to that in the sample collected in 2002. In April–September of 2014, the infection prevalence of M. georgiana, G. bowersi, and E. oatesi was different from that in 2002, while in the samples collected in October 2014–January 2015, the prevalence was similar to that in 2002. The abundance of these species changed similarly in 2014–2015, except for the abundance of G. bowersi which in all subsamples of 2014–2015 was similar to that in 2002. Corynosoma spp., M. johnstoni, and Pseudoterranova sp. had similar infection rates in the subsamples collected in 2014–2015 and in the sample collected in 2002, except for the abundance of Pseudoterranova sp. was higher in all subsamples collected in 2014–2015 (Table 2).
We analysed the helminth species associations and covariations in a whole sample of 196 infracommunities collected in 2002 and 2014–2015. For the analyses, we selected ten helminth species that had a prevalence of infection higher than 30%. Interactions of M. rennicki with other species were studied only in the sample collected in 2014–2015 (n = 101), for this species was absent in 2002. We found a total of 50 statistically significant interactions (36 associations and 14 covariations) out of 90 possible ones; none of 10 species had completely independent occurrence or abundance (Table 3). Metacanthocephalus rennicki demonstrated the least number of interactions; it had a positive association and covariation only with M. johnstoni. Other species had positive associations with two or more species. The largest number of interactions was observed in the nematode Pseudoterranova sp. (5 associations and 4 covariations), the trematode G. bowersi (5 associations and 2 covariations), and the acanthocephalan M. johnstoni (4 associations and 3 covariations). Negative covariation between M. johnstoni and bilocular metacestode (rS = − 0.24) was the only negative interaction revealed in the sample analysed; all other covariations and all associations were positive.
Discussion
The species composition of N. coriiceps helminth community in the area of Galindez Island has changed over 12 years: 20 helminth species were found in 2002, while 25 helminth species were found in 2014–2015 (Table 1). In studies of 2014–2015, seven additional species were recorded, namely the trematode Derogenes johnstoni, monolocular metacestode, the nematodes Dichelyne fraseri and Anisakis sp., and the acanthocephalans Metacanthocephalus campbelli and M. rennicki. On the other hand, two species of acanthocephalans, namely Corynosoma bullosum and C. shackletoni, were not found. Analysis of the infection prevalence of helminth species including the calculation of 95% confidence intervals revealed that De. johnstoni, Di. fraseri, monolocular metacestode, Anisakis sp., and C. shackletoni were “statistically insignificant species”, because the minimum limit of the confidence interval of their prevalence was lower than the minimum possible prevalence in the sample. Therefore, their contribution to the difference in the species composition was not significant. Also, two species of acanthocephalans, namely Corynosoma bullosum and C. shackletoni, found exclusively in cysts collected from the fish body cavity and internal organs [13] could be easily missed in samples of 2014–2015 when all 7597 cysts collected were not excysted and identified.
Comparative analysis of two samples of helminths in N. coriiceps revealed significantly larger species richness in the component community in 2014–2015. We presume that the increase in the infection of fish could be associated with both an increase in organic pollution in the water area of the UAS “Akademik Vernadsky” over a 12-year period [45] and with global ecological changes in the coastal ecosystems of the Antarctic and the Argentine Islands. Similar tendencies of increase of N. coriiceps infection with acanthocephalans associated with organic pollution were previously observed near the Polish Antarctic research station on King George Island [24]. In addition, over the past decades, the increasing number of birds, in particular, gentoo penguins (Pygoscelis papua) and Antarctic shags (Phalacrocorax bransfieldensis) and seals has been observed in the area of UAS [46,47,48]. Fish-eating birds and seals are definitive hosts of diphyllobothriid cestodes, anisakid nematodes, and acanthocephalans of the genus Corynosoma. Therefore, the growth of their populations undoubtedly increases the contamination of the coastal environment with the eggs of these parasites and, ultimately, increases the infection of teleost fish with their larvae. However, more data collected over a longer period of time are needed to clarify the trends in the species richness and structure of the N. coriiceps helminth community.
Despite the species richness in the helminth component community was different in the two studied samples, the diversity indices appeared to be rather similar. This may suggest that no dramatic changes have occurred in the helminth community of N. coriiceps during 12–13 years. Besides, we observed the similarity also in the abundance in helminth infracommunities in the two samples. On the other hand, the species richness in the infracommunities was higher in the sample collected in 2014–2015. The difference was confirmed by two statistical tests: the Mann–Whitney U test and ANOSIM routine and, therefore, is considered as significant. SIMPER analysis named ten helminth species contributing to the differences. An increase of species richness in the infracommunities might have occurred due to the higher prevalence of at least some of those species. The difference might be either due to seasonal changes in helminth prevalence of infection or because of the long-term changes of the prevalence. We analysed seasonal changes in prevalence and abundance of those 10 species in 2014–2015 to found the species which influenced the differences in the species richness in infracommunities. Only three species, namely the cestode larvae Diphyllobotrium sp., the acanthocephalan Metacanthocephalus rennicki, and the trematode Neoleoburia antarctica, had the prevalence constantly different in 2014–2015 compared to that in 2002. We conclude, therefore, that those three species contributed to the long-term changes in the helminth community in N. coriiceps. Furthermore, the two former species had a significantly higher prevalence in 2014–2015, therefore, they apparently contributed to the higher species richness in the infracommunities in the sample. We suppose that an increase in the N. coriiceps infection with the cestode Diphyllobotrium sp. is associated with an increase in populations of the seals, in particular, the Weddell seals Leptonychotes weddellii near the UAS “Akademik Vernadsky” over the last few decades [47]. Seals are the definitive hosts of these cestodes in the Argentine Islands region.
In the present study, the co-occurrence of ten helminth species in N. coriiceps was investigated based on the combined sample collected in 2002 and 2014–2015. Helminth species with a high prevalence of infection, e.g., Pseudoterranova sp., M. johnstoni, G. bowersi, and M. georgiana, expectedly had the largest number of interspecific associations due to more probable co-occurrence with other species (Table 3). However, larval nematodes Contracaecum sp. were found to have positive associations with five species: the nematode Pseudoterranova sp., bilocular metacestodes, the trematodes G. bowersi and E. oatesi, and the acanthocephalan M. campbelli, though the prevalence of infection in Contracaecum sp. was just 31.6%. Presumably, those positive associations are based on using the same intermediate hosts by Contracaecum sp. and associated species.
The least number of interspecific associations was observed in the acanthocephalan M. rennicki; it showed positive association and covariation only with M. johnstoni. In our opinion, this positive interaction between these two species suggests that they have the same species of intermediate hosts (Amphipoda), and no competition within the host intestine exists between them. Interestingly, the third species of the genus Metacanthocephalus, M. campbelli showed no associations with its congeners (see Table 3); instead, it presumably shared the transmission routes and intermediate hosts with the nematodes Contracaecum sp. and Pseudoterranova sp., and with bilocular metacestodes.
Thereby, the detailed analysis of the species composition of helminths parasitizing the Antarctic rockcod N. coriiceps and the parameters of fish infection, as well as the analysis of helminth infracommunities, component communities, and species co-occurrence yielded new information on the helminths communities of this fish species in the region of Argentine Islands. We believe that these data will be useful as the baseline for future long-term monitoring studies of teleost fish helminth communities in the region, and for future analysis of the role of helminths as indicators of ecological changes in the marine ecosystems of West Antarctica.
References
MacKenzie K (2017) The history of Antarctic parasitological research. In: Klimpel S, Kuhn T, Mehlhorn H (eds) Biodiversity and evolution of parasitic life in the Southern Ocean, Parasitology Research Monographs, vol 9. Springer Nature, Cham, pp 13–31. https://doi.org/10.1007/978-3-319-46343-8_3
Oğuz MC, Tepe Y, Belk MC, Heckmann RA, Aslan B, Gürgen M, Bray RA, Akgül Ü (2015) Metazoan parasites of Antarctic fishes. Turk J Parasitol 39:174–178. https://doi.org/10.5152/tpd.2015.3661
Zdzitowiecki K (1991) Antarctic Acanthocephala. In: Wägele JW, Sieg J (eds) Synopses of the Antarctic benthos, 3rd edn. Koeltz Scientific Books, Koenigstein, pp 1–116
Zdzitowiecki K (1997) Antarctic Digenea, parasites of fishes. In: Wägele JW, Sieg J (eds) Synopses of the Antarctic benthos, 8th edn. Koeltz Scientific Books, Koenigstein, pp 1–156
Rocka A (2003) Cestodes of the Antarctic fishes. Pol Polar Res 24:261–276
Rocka A (2004) Nematodes of the Antarctic fishes. Pol Polar Res 25:135–152
Rocka A (2006) Helminths of Antarctic fishes: life cycle biology, specificity and geographical distribution. Acta Parasitol 51:26–35. https://doi.org/10.2478/s11686-006-0003-y
Rocka A (2017) Cestodes and nematodes of Antarctic fishes and birds. In: Klimpel S, Kuhn T, Mehlhorn H (eds) Biodiversity and evolution of parasitic life in the Southern Ocean. Parasitology Research Monographs, vol 9. Springer, Cham. https://doi.org/10.1007/978-3-319-46343-8_6
Faltýnková A, Georgieva S, Kostadinova A, Bray RA (2017) Biodiversity and evolution of Digeneans of fishes in the Southern Ocean. In: Klimpel S, Kuhn T, Mehlhorn H (eds) Biodiversity and evolution of parasitic life in the Southern Ocean. Parasitology Research Monographs, vol 9. Springer, Cham. https://doi.org/10.1007/978-3-319-46343-8_5
Klapper R, Münster J, Kochmann J, Klimpel S, Kuhn T (2017) Biodiversity and host specificity of Monogenea in Antarctic fish species. In: Klimpel S, Kuhn T, Mehlhorn H (eds) Biodiversity and evolution of parasitic life in the Southern Ocean. Parasitology Research Monographs, vol 9. Springer, Cham. https://doi.org/10.1007/978-3-319-46343-8_4
Laskowski Z, Zdzitowiecki K (2017) Acanthocephalans in Sub-Antarctic and Antarctic. In: Klimpel S, Kuhn T, Mehlhorn H (eds) Biodiversity and evolution of parasitic life in the Southern Ocean, Parasitology Research Monographs, vol 9. Cham, Springer Nature, pp 141–182. https://doi.org/10.1007/978-3-319-46343-8_8
Zdzitowiecki K (2001) Occurrence of endoparasitic worms in fish, Parachaenichthys charcoti (Bathydraconidae), off the South Shetland Islands (Antarctica). Acta Parasitol 46(1):18–23
Zdzitowiecki K, Laskowski Z (2004) Helminths of an Antarctic fish, Notothenia coriiceps, from the Vernadsky Station (Western Antarctica) in comparison with Admiralty Bay (South Shetland Islands). Helminthologia 41:201–207
Laskowski Z, Zdzitowiecki K (2005) The helminth fauna of some notothenioid fishes collected from the shelf of Argentine Islands, West Antarctica. Pol Polar Res 26(4):315–324
Münster J, Kochmann J, Grigat J, Klimpel S, Kuhn T (2017) Parasite fauna of the Antarctic dragonfish Parachaenichthys charcoti (Perciformes: Bathydraconidae) and closely related Bathydraconidae from the Antarctic Peninsula, Southern Ocean. Parasites Vectors 10:235. https://doi.org/10.1186/s13071-017-2176-7
Kuhn T, Zizka VMA, Münster J, Klapper R, Mattiucci S, Kochmann J, Klimpel S (2018) Lighten up the dark: metazoan parasites as indicators for the ecology of Antarctic crocodile icefish (Channichthyidae) from the north-west Antarctic Peninsula. PeerJ 6:e4638. https://doi.org/10.7717/peerj.4638
Kuzmina TA, Salganskij OO, Lisitsyna OI, Korol EM (2020) Helminths of Antarctic rockcod Notothenia coriiceps (Perciformes, Nototheniidae) from the Akademik Vernadsky Station area (Argentine Islands, West Antarctica): new data on the parasite community. Zoodiversity 54(2):99–110. https://doi.org/10.15407/zoo2020.02.099
Siegel V (1980) Parasite tags for some Antarctic channichthyid fish. Arch FischWiss 31:97–103
Parukhin AM, Lyadov VN (1982) Helminth fauna of food Nototheniidae fishes from Kerguelen Subregion region. Ekologiya Morya 10:49–56 (in Russian)
Parukhin AM (1986) Helminthofauna peculiarities of commercial Nototheniodei from the Subantarktic region of the Indian Ocean. Vest Zool 3:6–10 (in Russian)
Moser M, Cowen RK (1991) The effects of periodic europhication on parasitism and stock identification of Trematomus bernacchii (Pisces: Nototheniidae) in McMurdo Sound, Antaractic. J Parasitol 77:551–556. https://doi.org/10.2307/3283158
George-Nascimento M (1996) Populations and assemblages of parasites in hake, Merluccius gayi, from the southern Pacific Ocean: stock implications. J Fish Biol 48:557–568. https://doi.org/10.1111/j.1095-8649.1996.tb01452.x
Brickle P, MacKenzie K, Pike A (2005) Parasites of the Patagonian toothfish, Dissostichus eleginoides Smitt 1898, in different parts of the Subantarctic. Polar Biol 28:663–671. https://doi.org/10.1007/s00300-005-0737-2
Laskowski Z, Korczak-Abshire M, Zdzitowiecki K (2012) Changes in acanthocephalan infection of the Antarctic fish Notothenia coriiceps in Admiralty Bay, King George Island, over 29 years. Pol Polar Res 33:99–108. https://doi.org/10.2478/v10183-012-0005-4
Laskowski Z, Jezewski W, Zdzitowiecki K (2014) Changes in digenean infection of the Antarctic fish Notothenia coriiceps in Admiralty Bay, King George Island, over three decades. Pol Polar Res 35:513–520. https://doi.org/10.2478/popore−2014−0009
Mouritsen KN, Poulin R (2002) Parasitism, climate oscillations and the structure of natural communities. Oikos 97:462–468. https://doi.org/10.1034/j.1600-0706.2002.970318.x
Mouritsen KN, Tompkins DM, Poulin R (2005) Climate warming may cause a parasite-induced collapse in coastal amphipod populations. Oecologia 146:476–483. https://doi.org/10.1007/s00442-005-0223-0
Hudson PJ, Dobson AP, Lafferty KD (2006) Is a healthy ecosystem one that is rich in parasites? Trends Ecol Evol 21:381–385. https://doi.org/10.1016/j.tree.2006.04.007
Poulin R (2006) Global warming and temperature-mediated increases in cercarial emergence in trematode parasites. Parasitology 132:143–151. https://doi.org/10.1017/S0031182005008693
Poulin R, Mouritsen KN (2006) Climate change, parasitism and the structure of intertidal ecosystems. J Helminthol 80:183–191. https://doi.org/10.1079/JOH2006341
Kvach Y, Kuzmina T (2020) Parasitological research in Antarctica: review of the issues and future prospects. Ukr Antarct J 1:102–110. https://doi.org/10.33275/1727-7485.1.2020.383 (in Ukrainian)
Shirihai H (2008) A complete guide to Antarctic wildlife, 2nd edn. Princeton University Press, Princeton
Barrera-Oro ER, Marschoff ER, Casaux RJ (2000) Trends in relative abundance of fjord Notothenia rossii, Gobionotothen gibberifrons and Notothenia coriiceps at Potter Cove, South Shetland Islands, after commercial fishing in the area. CCAMLR Sci 7(2000):43–52
Manilo LG (2006) Ichthyofauna and morphobiological characteristics of mass fish species of coastal waters of Argentine Islands (Antarctica). Zbirnyk prats Zoologichnogo Muzeju 38:5–22 (in Ukrainian)
Near TJ (2009) Notothenioid fishes (Notothenioidei). In: Hedges SB, Kumar S (eds) The timetree of life. Oxford University Press, Oxford, pp 339–343
Barrera-Oro ER, Marschoff E (2007) Information on the status of fjord Notothenia rossii, Gobionotothen gibberifrons and Notothenia coriiceps in the lower South Shetland Islands, derived from the 2000–2006 monitoring program at Potter Cove. CCAMLR Sci 14:83–87
Szidat L (1965) Estudios sobre la fauna de parasitos de peces antarticos. I. Los parasitos de Notothenia neglecta Nybelin. Secr Mar Serv Hidrogr Nav H 910:1–84
Palm HW, Reimann N, Splinder M, Plötz J (1998) The role of the rock cod Notothenia coriiceps in the life-cycle of Antarctic parasites. Polar Biol 19:399–406. https://doi.org/10.1007/s003000050265
Laskowski Z, Zdzitowiecki K (2009) Occurrence of Acanthocephalans in Notothenioid fishes in the Beagle channel (Magellanic sub-region, sub-Antarctic). Pol Polar Res 30:179–186. https://doi.org/10.13140/2.1.4863.0402
Rózsa L, Reiczigel J, Majoros G (2000) Quantifying parasites in samples of hosts. J Parasitol 86(2):228–232. https://doi.org/10.1645/0022-3395(2000)086[0228:QPISOH]2.0.CO;2
Reiczigel J, Abonyi-Tóth Z, Singer J (2008) An exact confidence set for two binomial proportions and exact unconditional confidence intervals for the difference and ratio of proportions. Comput Stat Data Anal 52:5046–5053. https://doi.org/10.1016/j.csda.2008.04.032
Hammer Ø (1999–2015) PAST, Paleontological statistics, version 3.10, reference manual. Natural History Museum, University of Oslo
Clarke KR, Gorley RN (2006) PRIMER v6: user manual/tutorial (plymouth routines in multivariate ecological research). PRIMER-E, Plymouth
Ludwig JA, Reynolds JF (1988) Statistical ecology. A primer on methods and computing. John Wiley & Sons, Hoboken
Samyshev EZ (2009) The features of structure and functioning of the ecosystem in the region of the Ukrainian Antarctic station (UAS) Academician Vernadsky. Ukr Antarct J 8:237–266 (in Russian)
Peklo AM (2007) Birds of the Argentine Islands and the Peterman Island. Krivoy Rog: Mineral (in Russian)
Dykyy IV, Peklo AM (2012) Seals of the Argentine Islands (Antarctica). Zbirnyk Prats Zoologichnogo Muzeju 43:104–116 (in Russian)
Smagol VM, Pilipenko DV, Dzhulai AO (2019) Dynamics of numbers and occurrence of colonial species of birds in the area of Ukrainian Antarctic station Akademik Vernadsky. Vest Zool 53(6):491–500. https://doi.org/10.2478/vzoo-2019-0044
Acknowledgements
This study was partially supported by the National Research Foundation of Ukraine (project number 2020.02/0074).
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Kuzmina, T.A., Laskowski, Z., Salganskij, O.O. et al. Helminth Assemblages of the Antarctic Black Rockcod, Notothenia coriiceps (Actinopterygii: Nototheniidae) in Coastal Waters near Galindez Island (Argentine Islands, West Antarctic): Temporal Changes in the Endoparasite Community. Acta Parasit. 67, 207–217 (2022). https://doi.org/10.1007/s11686-021-00448-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11686-021-00448-7