Summary
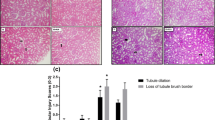
The aim of the present study was to observe the protective effects of α-lipoic acid (ALA) on vascular injury in rats with hyperuricemia (HUA). The ALA treatment groups (10, 30 and 90 mg/kg, respectively) were administered with ALA via gavage for 2 weeks. Subsequently, the levels of blood urea nitrogen (BUN), creatinine (CREA), uric acid (UA), total cholesterol (TC), high density lipoprotein-C (HDL-C) and low density lipoprotein-C (LDL-C) were measured; the activities of glutathione peroxidase (GSH-Px), catalase (CAT), malonaldehyde (MDA), superoxide dismutase (SOD) and xanthine oxidase (XOD) were also determined. The thoracic aorta of rats in each experimental group was observed under a light microscope; ultrastructural analysis was performed. SOD and CAT protein contents were investigated by Western blotting. The results revealed that: i) Compared with the model group, the levels of UA were decreased in the ALA groups and the levels of BUN, CREA, TC, and LDL-C decreased in the 30 and 90 mg/kg ALA groups (P<0.05); ii) compared with the model group, the activities of GSH-Px, SOD and XOD were increased and the levels of MDA were reduced in the 90 mg/kg ALA group (P<0.05); and iii) in the model and 10 mg/kg ALA groups, edema and shedding were observed in endothelial cells. Compared with the model and 10 mg/kg ALA groups, the 30 and 90 mg/kg ALA groups exhibited fewer swollen endothelial cells. In summary, the results of the present study indicated that HUA resulted in vascular oxidative stress injury and decreased the activity of antioxidative enzymes, which leads to endothelial cell damage and vascular lesions. ALA may serve as a therapeutic agent for the treatment of HUA-induced endothelial dysfunction.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Bardin T, Richette P. Definition of hyperuricemia and gouty conditions. Curr Opin in Rheumatol, 2014,26(2):186–191
Peng TC, Wang CC, Kao TW, et al. Relationship between hyperuricemia and lipid profiles in US adults. Biomed Res Int, 2015,2015:127596
Van DC, Van Echteld IA, Falzon L, et al. Cardiovascular risk factors and comorbidities in patients with hyperuricemia and/or gout: a systematic review of the literature. J of Rheumatol Suppl, 2014,92:9–14
Jalal DI. Hyperuricemia, the kidneys, and the spectrum of associated diseases: a narrative review. Curr Med Res Opin, 2016,32(11):1863–1869
Cheng S, Yang Y, Zhou Y, et al. Influence of different concentrations of uric acid on oxidative stress in steatosis hepatocytes. Exp Ther Med, 2018,15(4):3659–3665
Wu SH, Shu XO, Milne G, et al. Uric acid correlates to oxidation and inflammation in opposite directions in women. Biomarkers, 2015,20(4):225–231
Johnson RJ, Nakagawa T, Sanchezlozada LG, et al. Sugar, Uric Acid, and the Etiology of Diabetes and Obesity. Diabetes, 2013,62(10):3307–3315
Zhang JX, Zhang YP, Wu QN, et al. Uric acid induces oxidative stress via an activation of the renin-angiotensin system in 3T3-L1 adipocytes. Endocrine, 2015,48(1):135–142
Choi YJ, Shin HS, Choi HS, et al. Uric acid induces fat accumulation via generation of endoplasmic reticulum stress and SREBP-1c activation in hepatocytes. Lab Invest, 2014,94(10):1114–1125
Zhu Y, Hu Y, Huang T, et al. High uric acid directly inhibits insulin signalling and induces insulin resistance. Biochem Biophys Res Commun, 2014,447(4):707–714
Zhuang Y, Feng Q, Ding G, et al. Activation of ERK1/2 by NADPH oxidase-originated reactive oxygen species mediates uric acid-induced mesangial cell proliferation. Am J Physiol Renal Physiol, 2014,307(4):F396–406
Luo C, Lian X, Hong L, et al. High Uric Acid Activates the ROS-AMPK Pathway, Impairs CD68 Expression and Inhibits OxLDL-Induced Foam-Cell Formation in a Human Monocytic Cell Line, THP-1. Cell Physiol Biochem, 2016,40(3–4):538–548
Shay KP, Moreau RF, Smith EJ, et al. Alpha-lipoic acid as a dietary supplement: molecular mechanisms and therapeutic potential. Biochim Biophys Acta, 2009,1790(10):1149–1160
Li RJ, Ji WQ, Pang JJ, et al. Alpha-lipoic acid ameliorates oxidative stress by increasing aldehyde dehydrogenase-2 activity in patients with acute coronary syndrome. Tohoku J Exp Med, 2013,229(1):45–51
Ghibu S, Richard C, Vergely C, et al. Antioxidant properties of an endogenous thiol: alpha-lipoic acid, useful in the prevention of cardiovascular diseases. J Cardiovasc Pharmacol, 2009,54(5):391–398
Mohamed WR, Mehany ABM, Hussein RM. Alpha lipoic acid protects against chlorpyrifos-induced toxicity in Wistar rats via modulating the apoptotic pathway. Environ Toxicol Pharmacol, 2018,59:17–23
Gomaa A, Elmottaleb Abd NA, Aamer HA. Antioxidant and anti-inflammatory activities of alpha lipoic acid protect against indomethacin-induced gastric ulcer in rats. Biomed Pharmacother, 2018,101:188–194
Capuano V, Marchese F, Capuano R, et al. Hyperuricemia as an independent risk factor for major cardiovascular events: a 10-year cohort study from Southern Italy. J Cardiovasc Med (Hagerstown), 2017,18(3):159–164
Kuwabara M. Hyperuricemia, Cardiovascular Disease, and Hypertension. Pulse, 2015,3:242–252
Cai W, Duan XM, Liu Y, et al. Uric Acid Induces Endothelial Dysfunction by Activating the HMGB1/RAGE Signaling Pathway. Biomed Res Int, 2017,2017(2):1–11
Chen G, Jia P. Allopurinol decreases serum uric acid level and intestinal glucose transporter-5 expression in rats with fructose-induced hyperuricemia. Pharmacol Rep, 2016,68(4):782–786
Wang CW, Dao RL, Chung WH. Immunopathogenesis and risk factors for allopurinol severe cutaneous adverse reactions. Curr Opin Allergy Clin Immunol, 2016,16(4):339–345
Silva MC, De Sousa CN, Gomes PX, et al. Evidence for protective effect of lipoic acid and desvenlafaxine on oxidative stress in a model depression in mice. Prog Neuro-psychopharmacol Biol Psychiatry, 2016,64:142–148
Petronilho F, Florentino D, Danielski LG, et al. Alpha-Lipoic Acid Attenuates Oxidative Damage in Organs After Sepsis. Inflammation, 2016,39(1):357–365
Renuka M, Vijayakumar N, Ramakrishnan A. Chrysin, a flavonoid attenuates histological changes of hyperammonemic rats: A dose dependent study. Biomed Pharmacother, 2016,82:345–354
Li YP, Wu S, Ran A, et al. Aristolochia Bracteolate Retz. Attenuates Hyperuricemia in a Metabolic Arthritis Rat Model. Afr J Tradit Complement Altern Med, 2017,14(4):180–187
Zhu JX, Wang Y, Kong LD, et al. Effects of Biota orientalis extract and its flavonoid constituents, quercetin and rutin on serum uric acid levels in oxonate-induced mice and xanthine dehydrogenase and xanthine oxidase activities in mouse liver. J Ethnopharmacol, 2004,93(1):133–140
Nguyen MT, Awale S, Tezuka Y, et al. Hypouricemic Effects of Acacetin and 4,5-O-Dicaffeoylquinic Acid Methyl Ester on Serum Uric Acid Levels in Potassium Oxonate-Pretreated Rats. Biol Pharm Bull, 2005,28(12):2231–2234
Zhang S, Zhuang J, Yue G, et al. Lipidomics to Investigate the Pharmacologic Mechanisms of Ginkgo Folium in the Hyperuricemic Rat Model. J Chromatogr B Analyt Technol Biomed Life Sci, 2017,1060:407–415
Ma WG, Wang J, Bu XW, et al. Effects of Polygonum cuspidatum on AMPK-FOXO3alpha signaling pathway in a rat model of uric acid-induced renal damage. Chin J Integr Med, 2019,25(3):182–189
Song C, Zhao X. Uric acid promotes oxidative stress and enhances vascular endothelial cell apoptosis in rats with middle cerebral artery occlusion. Biosci Rep, 2018,38(3). pii: BSR20170939
Aroor AR, Jia G, Habibi J, et al. Uric acid promotes vascular stiffness, maladaptive inflammatory responses and proteinuria in western diet fed mice. Metabolism, 2017,74:32–40
Dai H, Huang Z, Deng Q, et al. The Effects of Lead Exposure on Serum Uric Acid and Hyperuricemia in Chinese Adults: A Cross-Sectional Study. Int J Environ Res Public Health, 2015,12(8):9672–9682
Kuwabara M, Borghi C, Cicero AFG, et al. Elevated serum uric acid increases risks for developing high LDL cholesterol and hypertriglyceridemia: A five-year cohort study in Japan. Int J Cardiol, 2018,261:183–188
Wang H, Sun Y, Wang S, et al. Body adiposity index, lipid accumulation product, and cardiometabolic index reveal the contribution of adiposity phenotypes in the risk of hyperuricemia among Chinese rural population. Clin Rheumatol, 2018,37(8):2221–2231
Komori H, Yamada K, Tamai I. Hyperuricemia enhances intracellular urate accumulation via down-regulation of cell-surface BCRP/ABCG2 expression in vascular endothelial cells. Biochim Biophys Acta Biomembr, 2018,1860(5):973–980
Peng TC, Wang CC, Kao TW, et al. Relationship between hyperuricemia and lipid profiles in US adults. Biomed Res Int, 2015,2015:127596
Cibičková L’, Langová K, Vaverková H, et al. Correlation of uric acid levels and parameters of metabolic syndrome. Physiol Res, 2017,66(3):481–487
Stelmach MJ, Wasilewska N, Wicklund-Liland LI, et al. Blood lipid profile and BMI-Z-score in adolescents with hyperuricemia. Ir J Med Sci, 2015,184(2):463–468
Schüller ÁK, Mena Canata DA, Hackenhaar FS, et al. Effects of lipoic acid and n-3 long-chain polyunsaturated fatty acid on the liver ovariectomized rat model of menopause. Pharmacol Rep, 2018,70(2):263–269
Gomes MB, Negrato CA. Alpha-lipoic acid as a pleiotropic compound with potential therapeutic use in diabetes and other chronic diseases. Diabetol Metab Syndr, 2014,6(1):80–97
Mukhopadhyay P, Verzola D, Ratto E, et al. Uric Acid Promotes Apoptosis in Human Proximal Tubule Cells by Oxidative Stress and the Activation of NADPH Oxidase NOX 4. PLoS One, 2014,9(12):e115210
Gomaa AMS, Abd El-Mottaleb NA, Aamer HA. Antioxidant and anti-inflammatory activities of alpha lipoic acid protect against indomethacin-induced gastric ulcer in rats. Biomed Pharmacother, 2018,101:188–194
Khan J, Salhotra S, Ahmad S, et al. The protective effect of α-lipoic acid against bisphenol A-induced neurobehavioral toxicity. Neurochem Int, 2018,118:166–175
Liu Y, Zhang Q, Wang L, et al. The α-lipoic acid improves high-fat diet-induced cerebral damage through inhibition of oxidative stress and inflammatory reaction. Environ Toxicol Pharmacol, 2017,56:219–224
Asakawa S, Shibata S, Morimoto C, et al. Podocyte Injury and Albuminuria in Experimental Hyperuricemic Model Rats. Oxid Med Cell Longev, 2017,2017:3759153
Hong Q, Qi K, Feng Z, et al. Hyperuricemia induces endothelial dysfunction via mitochondrial Na+/Ca2+ exchanger-mediated mitochondrial calcium overload. Cell Calcium, 2012,51(5):402–410
Li P, Zhang L, Zhang M, et al. Uric acid enhances PKC-dependent eNOS phosphorylation and mediates cellular ER stress: A mechanism for uric acid-induced endothelial dysfunction. Int J Mol Med, 2016,37(4):989–997
Yang H, Bai W, Gao L, et al. Mangiferin alleviates hypertension induced by hyperuricemia via increasing nitric oxide releases. J Pharmacol Sci, 2018,137(2):154–161
Liang S, Sun K, Wang Y, et al. Role of Cyt-C/caspases-9,3, Bax/Bcl-2 and the FAS death receptor pathway in apoptosis induced by zinc oxide nanoparticles in human aortic endothelial cells and the protective effect by alpha-lipoic acid. Chem Biol Interact, 2016,258:40–51
Hu H, Wang C, Jin Y, et al. Alpha-lipoic acid defends homocysteine-induced endoplasmic reticulum and oxidative stress in HAECs. Biomed Pharmacother, 2016,80:63–72
Zou H, Wang H, Liu T, et al. Protective role of alpha-lipoic acid in hyperuricemia-induced endothelial dysfunction. Exp Ther Med, 2017,13(6):3047–3054
Acknowledgments
This study was supported by Southwest Medical University and Xinjiang Medical University and we thank them for their funding and technical assistance.
Author information
Authors and Affiliations
Corresponding author
Additional information
Conflict of Interest Statement
The authors declare that they have no competing interests.
Rights and permissions
About this article
Cite this article
Xiang, W., Wang, L., Cheng, S. et al. Protective Effects of α-Lipoic Acid on Vascular Oxidative Stress in Rats with Hyperuricemia. CURR MED SCI 39, 920–928 (2019). https://doi.org/10.1007/s11596-019-2124-1
Received:
Revised:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11596-019-2124-1