Abstract
Background
To observe the effects of Chinese medicine (CM) Polygonum cuspidatum (PC) on adenosine 5′-monophosphate-activated protein kinase (AMPK), forkhead box O3α (FOXO3α), Toll-like receptor-4 (TLR4), NACHT, LRR and PYD domains-containing protein 3 (NLRP3), and monocyte chemoattractant protein-1 (MCP-1) expression in a rat model of uric acid-induced renal damage and to determine the molecular mechanism.
Methods
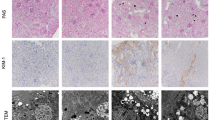
A rat model of uric acid-induced renal damage was established, and rats were randomly divided into a model group, a positive drug group, and high-, medium-, and low-dose PC groups (n=12 per group). A normal group (n=6) was used as the control. Rats in the normal and model groups were administered distilled water (10 mL•kg–1) by intragastric infusion. Rats in the positive drug group and the high-, medium-, and low-dose PC groups were administered allopurinol (23.33 mg•kg–1), and 7.46, 3.73, or 1.87 g•kg–1•d–1 PC by intragastric infusion, respectively for 6 to 8 weeks. After the intervention, reverse transcription polymerase chain reaction, Western blot, enzyme linked immunosorbent assay, and immunohistochemistry were used to detect AMPK, FOXO3α, TLR4, NLRP3, and MCP-1 mRNA and protein levels in renal tissue or serum.
Results
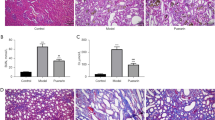
Compared with the normal group, the mRNA transcription levels of AMPK and FOXO3α in the model group were significantly down-regulated, and protein levels of AMPKα1, pAMPKα1 and FOXO3α were significantly down-regulated at the 6th and 8th weeks (P<0.01 or P<0.05). The mRNA transcription and protein levels of TLR4, NLRP3 and MCP-1 were significantly up-regulated (P<0.01 or P<0.05). Compared with the model group, at the 6th week, the mRNA transcription levels of AMPK in the high- and medium-dose groups, and protein expression levels of AMPKα1, pAMPKα1 and FOXO3α in the high-dose PC group, AMPKα1 and pAMPKα1 in the mediumdose PC group, and pAMPKα1 in the low-dose PC group were significantly up-regulated (P<0.01 or P<0.05); the mRNA transcription and protein levels of TLR4 and NLRP3 in the 3 CM groups, and protein expression levels of MCP-1 in the medium- and low-dose PC groups were down-regulated (P<0.01 or P<0.05). At the 8th week, the mRNA transcription levels of AMPK in the high-dose PC group and FOXO3α in the medium-dose PC group, and protein levels of AMPKα1, pAMPKα1 and FOXO3α in the 3 CM groups were significantly up-regulated (P<0.01 or P<0.05); the mRNA transcription levels of TLR4 in the medium- and low-dose PC groups, NLRP3 in the high- and low-dose PC groups and MCP-1 in the medium- and low-dose PC groups, and protein expression levels of TLR4, NLRP3 and MCP-1 in the 3 CM groups were down-regulated (P<0.01 or P<0.05).
Conclusion
PC up-regulated the expression of AMPK and its downstream molecule FOXO3α and inhibited the biological activity of TLR4, NLRP3, and MCP-1, key signal molecules in the immunoinflammatory network pathway, which may be the molecular mechanism of PC to improve hyperuricemia-mediated immunoinflflammatory metabolic renal damage.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Sánchezlozada LG, Tapia E, Santamaría J, Avilacasado C, Soto V, Nepomuceno T, et al. Mild hyperuricemia induces vasoconstriction and maintains glomerular hypertension in normal and remnant kidney rats. Kidney Int 2005;67:237–247.
Mazzali M, Kanellis J, Han L, Feng L, Xia YY, Chen Q, et al Hyperuricemia induces a primary renal arteriolopathy in rats by a blood pressure–independent mechanism. Am J Physiol Renal Physiol 2002;282: F991–F997.
Albertoni G, Maquigussa E, Pessoa E, Barreto JA, Borges F, Schor N. Soluble uric acid increases intracellular calcium through an angiotensin–dependent mechanism in immortalized human mesangial cells. Exp Biol Med 2010;235:825–832.
Curhan GC, Mitch WE. Diet and kidney disease. In: Brener BM, ed. The Kidney. 8th ed. Philadelphia: Saunders Elsevier; 2008:1827–1847.
Zhang YK, Shen YP. Controversy and progress in the treatment of hyperuricemia in chronic kidney disease. Chin J Nephrol (Chin) 2011;27(2):75–76.
Wang DP, Zeng L, Shang SC, eds. Laboratory animal blood physiological and biochemical reference manual. Beijing: Science Press; 2011:129.
Gordon S. Pattern recognition receptors: doubling up for the innate immune response. Cell 2002;111:927–930.
Wolfs TG, Buurman WA, van Schadewijk A, de Vries B, Daemen MA, Hiemstra PS, et al. In vivo expression of Tolllike receptor 2 and 4 by renal epithelial cells: IFN–gamma and TNF–alpha mediated up–regulation during inflammation. J Immunol 2002;168:1286–1293.
So A. Recent advances in the pathophysiology of hyperuricemia and gout. Rev Med Suisse 2007;3:720,722–724.
Li QI, Wang CN, Song YL. Research progress on NALP3 inflammasome and related diseases. Chin J Conserv Dent (Chin) 2011;21:355–359,328.
Zhou Y, Fang L, Jiang L, Wen P, Cao H, He W, et al. Uric acid induces renal inflammation via activating tubular NF–κB signaling pathway. PLoS One 2012;7:e39738.
Chen DY, Song ZF, Li F, Zhang DQ. Expression of FOXO1 and FOXO3α on peripheral blood mononuclear cells and their clinical roles in patients with systemic lupus erythemtosus. Chin J Rheumatol (Chin) 2008;12:629–631.
Hwang JW, Rajendrasozhan S, Yao H, Chung S, Sundar IK, Huyck HL, et al. FOXO3 deficiency leads to increased susceptibility to cigarette smoke–induced inflammation, airspace enlargement, and chronic obstructive pulmonary disease. J Immunol 2011;187:987–998.
Qiu H, Chen C, Wang RL, Luo H, Zhao BX. Progressive chronic inflammatory reaction in spleens of FoxO3a gene knockout mice. Chin J Biol (Chin) 2012;25:1650–1652,1657.
Lin L, Hron JD, Peng SL. Regulation of NF–kappaB, Th activation, and autoinflammation by the forkhead transcription factor Foxo3α. Immunity 2004;21:203–213.
Im J, Hergert P, Nho RS. Reduced FoxO3a expression causes low autophagy in idiopathic pulmonary fibrosis fibroblasts on collagen matrices. Am J Physiol Lung Cell Mol Physiol 2015;309:L552–L561.
Togher S, Larange A, Schoenberger SP, Feau S. FoxO3 is a negative regulator of primary CD8+ T–cell expansion but not of memory formation. Immunol Cell Biol 2015;93:120–125.
Tikhanovich I, Kuravi S, Campbell RV, Kharbanda KK, Artigues A, Villar MT, et al. Regulation of FOXO3 by phosphorylation and methylation in hepatitis C virus infection and alcohol exposure. Hepatology 2014;59:58–70.
Xuan LL, Hou Q. Recent advances in the study of AMPK and inflammatory pulmonary disease. Acta Pharm Sin (Chin) 2014;49:1089–1096.
Author information
Authors and Affiliations
Corresponding author
Additional information
Supported by the National Natural Science Foundation of China (No. 81473516 and No. 30973918)
Electronic supplementary material
Rights and permissions
About this article
Cite this article
Ma, Wg., Wang, J., Bu, Xw. et al. Effects of Polygonum cuspidatum on AMPK-FOXO3α Signaling Pathway in Rat Model of Uric Acid-Induced Renal Damage. Chin. J. Integr. Med. 25, 182–189 (2019). https://doi.org/10.1007/s11655-017-2979-6
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11655-017-2979-6