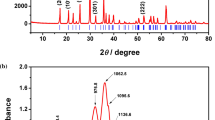
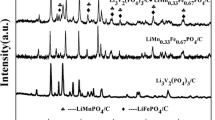
Abstract
Phspho-olivine LiFePO4 was synthesized from the relatively insoluble lithium source Li2CO3, proper iron and phosphorus sources (n(Li):n(Fe):n(P)=1:1:1) by a novel hydrothermal method. Afterwards, the optimal sample was mixed with glucose and two-step calcinated (500 °C and 750 °C) under high-purity N2 to obtain the LiFePO4/C composite. The resultant samples were characterized by X-ray diffraction (XRD), atomic absorption spectrometry (AAS), scanning electron microscops (SEM), transmission electron microscopy (TEM), energy dispersive spectrometry (EDS), elementary analysis (EA) and electrochemical tests. The results show that the optimal reaction condition is to set the reactant concentration at 0.5 mol·L−1, the reaction temperature at 180 °C for 16 h duration. During the reaction course, an intermediate product NH4FePO4·H2O was first synthesized, and then it reacted with Li+ to form LiFePO4. The optimized LiFePO4 sample with an average particle size (300 to 500 nm) and regular morphology exhibits a relatively high discharge capacity of 84.95 mAh· g−1 at the first charge-discharge cycle (0.1C, 1C=170 mA·g−1). Moreover, the prepared LiFePO4/C composite shows a high discharge capacity of 154.3 mAh·g−1 at 0.1C and 128.2 mAh·g−1 even at 5C. Besides it has good reversibility and stability in CV test.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Padhi AK, Nanjundaswamy KS, Goodenough JB. Phospho-Olivines as Positive-Electrode Materials for Rechargeable Lithium Batteries[J]. J. Electrochem. Soc., 1997, 144: 1 188–1 194
Meligrana G, Gerbald C, Tue A, et al. Hydrothermal Synthesis of High Surface LiFePO4 Powders as Cathodes for Li-ion Cells[J]. J. Power Sources, 2006, 160: 516–522
Shu HB, Wang XY, Wu Q, et al. The Effect of Ammonia Concentration on the Morphology and Electrochemical Properties of LiFePO4 Synthesized by Ammonia Assisted Hydrothermal Route[J]. Eletrochim. Acta, 2012, 76, 120–129
Shenouda AY, Liu HK. Studies on Electrochemical Behaviour of Zinc-Doped LiFePO4 for Lithium Battery Positive Electrode[J]. J. Alloys Compd., 2009, 477: 498–503
Lee J, Teja AS. Synthesis of LiFePO4 Micro and Nanoparticles in Supercritical Water[J]. Mater. Lett., 2006, 60: 2 105–2 109
Vujković M, Stojković I, Cvjetićanin N, et al. Gel-Combustion Synthesis of LiFePO4/C Composite with Improved Capacity Retention in Aerated Aqueous Electrolyte Solution[J]. Eletrochim. Acta, 2013, 92: 248–256
Jones JL, Huang JT, Meng YS. Intermittent X-ray Diffraction Study of Kinetics of Delithiation in Nano-Scale LiFePO4[J]. J. Power Sources, 2009, 189: 702–705
Yu F, Zhang JJ, Yang YF, et al. Preparation and Characterization of Porous Spherical LiFePO4/C by Spray Drying and Carbothermal Method[J]. J. Wuhan University of Technology-Mater. Sci. Ed., 2007, 22: 853–855
Murugan AV, Muraliganth T, Manthiram A. Comparison of Microwave Assisted Solvothermal and Hydrothermal Syntheses of LiFePO4/C Nanocomposite Cathodes for Lithium Ion Batteries[J]. J. Phys. Chem. C, 2008, 112: 14 665–14 671
Yang JM, Bai Y, Qing CB, et al. Electrochemical Performances of Co-Doped LiFePO4/C Obtained by Hydrothermal Method[J]. J. Alloys Compd., 2011, 509: 9 010–9 104
Liang GC, Wang L, Ou XQ, et al. Lithium Iron Phosphate with High-Rate Capability Synthesized through Hydrothermal Reaction in Glucose Solution[J]. J. Power Sources, 2008, 184: 538–542
Li XF, Zhang Z, Liu F, et al. Synthesis of Spherical LiFePO4/C Composites as Cathode Material of Lithium-ion Batteries by a Novel Glucose Assisted Hydrothermal Method[J]. Adv. Mater. Res., 2013, 787: 58–64
Li XF, Zhang X, Zhang Z. Synthesis of High-Performance LiFePO 4/C Composite with a Grape-Bunch Structure through the Hydrothermal Method [J]. Ionics, 2014, 20: 1 275–1 283
Gao G, Liu AF, Hu ZH, et al. Synthesis of LiFePO4/C as Cathode Material by a Novel Optimized Hydrothermal Method[J]. Rare Metals, 2011, 30: 433–438
Brochu F, Guerfi A, Trottier J, et al. Structure and Electrochemistry of Scaling Nano C-LiFePO4 Synthesized by Hydrothermal Route: Complexing agent effect[J]. J. Power Source, 2012, 214: 1–6
Wang XF, Li T, Wu J, et al. Electrochemical Properties of Nano-LiFePO4 Prepared by Hydrothermal Reaction[J]. J. Wuhan University of Technology-Mater. Sci. Ed., 2011, 26 (4): 624–627
Kuwahara A, Suzuki S, Miyayama M, et al. Hydrothermal Synthesis of LiFePO4 with Small Particle Size and Its Electrochemical Properties[J]. J. Electroceram., 2010, 24, 69–75
Chen J, Whittingham MS. Hydrothermal Synthesis of Lithium Iron Phosphate[J]. Electrochem. Commun., 2006, 8: 855–858
Recham N, Armand M, Laffont L, et al. Eco-Efficient Synthesis of LiFePO4 with Different Morphologies for Li-ion Batteries[J]. Electrochem. Solid-State Lett., 2009, 12: A39–A44
Doeff MM, Hu YQ, Mclarnn F, et al. Effect of Surface Carbon Structure on the Electrochemical Performance of LiFePO4[J]. Electrochem. Solid-State Lett., 2003, 6: A207–A209
Yang ST, Zhao NH, Dong HY, et al. Synthesis and Characterization of LiFePO4 Cathode Material Dispersed with Nano-Structured Carbon[J]. Electrochim. Acta., 2005, 51: 166–171
Zhang Z, Peng SF, Liu DC. Principle and Technology for Inorganic Fine Chemicals[M]. Beijing: Chemical Industry Press, 2005:136–147
Zhang Z, Li XF, Hu YL, et al. Chinese Patent, 2012, CN102790214A
Kanamura K, Koizumi S, Dokko K. Hydrothermal Synthesis of LiFePO4 as Cathode Material for Lithium Batteries[J]. J. Mater. Sci., 2008, 43: 2 138–2 142
Ou XQ, Xu SZ, Liang GC, et al. Effect of Fe(III) Impurity on the Electrochemical Performance of LiFePO4 Prepared by Hydrothermal Process[J]. Sci. China Ser. E, 2009, 52: 264–268
Lee J, Teja AS. Characteristics of Lithium Iron Phosphate (LiFePO4) Particles Synthesized in Subcritical and Supercritical Water[J]. J. Supercrit. Fluids, 2005, 35: 83–90
Yang SF, Zavalij PY, Whittingham MS. Hydrothermal Synthesis of Lithium Iron Phosphate Cathodes[J]. Electrochem. Commun., 2001, 3: 505–508
Padhi AK, Nanjundaswamy KS, Masquelier C, et al, Effect of Structure on the Fe3+ /Fe2+ Redox Couple in Iron Phosphates[J]. J. Electrochem. Soc., 1997, 144: 1 609–1 613
Li G, Yang ZX, Yang WS. Effect of FePO4 Coating on Electrochemical and Safety Performance of LiCoO2 as Cathode Material for Li-ion Batteries[J]. J. Power Sources, 2008, 183: 741–748
Le ZQ, Bo SM, Wang GJ. Inorganic Fine Chemicals Manual[M]. Beijing: Chemical Industry Press, 2004:140–141
Author information
Authors and Affiliations
Corresponding author
Additional information
Funded by the National Natural Science Foundation of China (No. 51004074)
Rights and permissions
About this article
Cite this article
Li, X., Hu, Y., Liu, F. et al. Synthesis of low-cost LiFePO4 from Li2CO3 by a novel hydrothermal method and investigation on the reaction mechanism. J. Wuhan Univ. Technol.-Mat. Sci. Edit. 30, 223–230 (2015). https://doi.org/10.1007/s11595-015-1129-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11595-015-1129-2