Abstract
This study focused on freshwater hyphomycetes associated with submerged decaying leaves of Calophyllum brasiliense in three streams in the semiarid region of Brazil. Leaves were enclosed in litter bags and deployed into streams on two occasions (dry and wet seasons). Samples of leaf litter were collected every two months (November 2013 to January 2015). Unlike other studies, we specifically targeted ecologically distinct groups of freshwater hyphomycetes by using two methods to induce fungal sporulation: submerged incubation (SI) of leaf litter on an orbital shaker and incubation in moist chambers (MC). We aimed to analyze and compare the structure of freshwater hyphomycete communities in the streams using both methods, detect possible successional patterns, and evaluate if environmental variables have influenced fungal diversity and sporulation rates. Sixty-nine taxa of freshwater hyphomycetes were observed. Of these, 26 were found under SI and 56 in MC. We observed large differences in fungal communities recovered by SI vs. MC that demonstrates the importance of using several methodological approaches to maximize the number of taxa recovered in ecological studies of litter-associated fungi. The highest sporulation rates under SI were observed during the wet season. Results of community ordination suggested that environmental variables affected the structure of fungal communities; for example, water velocity showed a positive effect on fungal diversity, while higher oxygen availability was associated with lower diversity. This study advances our understanding of the freshwater hyphomycete communities in tropical streams where ecological studies of aquatic fungi are still uncommon.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Freshwater fungi are important decomposers of allochthonous plant litter entering rivers and streams from the riparian zone (Sridhar et al. 2010; Ferreira et al. 2014). Fungi mediate carbon, energy, and nutrient flow in freshwater ecosystems including the increase in the quality of decomposing leaf detritus, which makes it more palatable and nutritious food source to aquatic invertebrate consumers (Gulis et al. 2006). In aquatic ecosystems, successional studies of fungal communities have been conducted on both submerged wood (Shearer and Webster 1991; Ho et al. 2002) and leaves (Shearer and Webster 1985; Gessner et al. 1993; Schoenlein-Crusius and Milanez 1998; Newman et al. 2015). On submerged leaves, both chytridiomycetes and zygomycetes can be detected, but the fungal communities are dominated by ascomycetes and basidiomycetes (Schoenlein-Crusius and Milanez 1989; Gessner et al. 1993).
Freshwater hyphomycetes include diverse aquatic fungi found in continental aquatic ecosystems that, as a group, have a worldwide distribution (Shearer et al. 2007). Freshwater hyphomycetes encompass three ecological groups that have been studied for decades (Hyde et al. 2015). The most recognized group is called Ingoldian fungi in honor of Dr. Ingold for his pioneering contribution to their studies (Descals et al. 1977). They represent the true aquatic hyphomycetes due to their unique adaptations to stream and river environments, such as tetraradial or sigmoid shape of conidia and fast germination that facilitate underwater dispersal and colonization of submerged plant litter. The aero-aquatic hyphomycetes display mycelial growth on submerged substrates; however, even though sporulation typically does not occur underwater, the conidia demonstrate adaptations to water dispersal (Fisher 1977). Species of facultative aquatic hyphomycetes can sporulate below or above water but typically lack specific adaptation for water dispersal; they represent a continuous group with terrestrial hyphomycetes (with conidia often showing thick walls, i.e., being dematiaceous (Descals and Moralejo 2001)).
The structure of freshwater hyphomycete communities can be assessed by a variety of techniques that are aimed to induce sporulation: incubation of submerged plant material on a shaker or in bubble chambers simulating stream conditions or, if fungi produce spores in air, moist chamber incubation (Graça et al. 2016). Submerged incubation of plant material, mainly leaves, yields a large number of Ingoldian fungi or aquatic hyphomycetes (Chan et al. 2000; Ghate and Sridhar 2016), while incubation of leaf litter or wood in moist chambers reveals mainly facultative aquatic hyphomycetes, aero-aquatic hyphomycetes, and freshwater ascomycetes (Sridhar et al. 2010; Barbosa et al. 2013) that are quite diverse in the Neotropics (Castañeda-Ruiz et al. 2016).
Several studies have indicated that environmental variables, such as water temperature, oxygen concentration, turbulence, and pH, may influence the diversity, activity, and sporulation of freshwater hyphomycetes (Webster 1975; Hissy et al. 1992; Chauvet and Suberkroop 1998; Rajashekhar and Kaveriappa 2000; Medeiros et al. 2009). The effects of dissolved inorganic nutrients (nitrogen and phosphorus), which are often elevated in rivers affected by human activity, have recently received considerable attention (Gulis and Suberkropp 2003; Gulis et al. 2008; Ferreira et al. 2015; Kominoski et al. 2015). Another important factor affecting aquatic fungal communities is the diversity of plant litter and riparian vegetation (Gulis 2001; Chauvet et al. 2016). For this study, Calophyllum brasiliense Cambess was chosen due to its high abundance and ecological importance in the riparian corridors in Chapada Diamantina including Rio de Contas.
In this study, we investigated communities of freshwater hyphomycetes associated with submerged decomposing leaves of C. brasiliense in three streams of the Rio de Contas basin. Unlike other studies, we specifically targeted ecologically distinct groups of freshwater hyphomycetes by using two methods to induce fungal sporulation: submerged incubation (SI) of leaf litter on an orbital shaker and incubation in moist chambers (MC). We aimed to (i) analyze and compare the structure of fungal communities in the experimental streams using both methods; (ii) detect possible successional patterns in the structure of freshwater hyphomycete communities; and (iii) evaluate if environmental variables (water temperature, pH, dissolved oxygen, water velocity, and total nitrogen and phosphorus) have influenced the diversity and sporulation rates of fungal communities.
Materials and methods
Study area
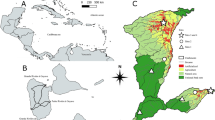
The present study was conducted in three streams of the Rio de Contas basin: Rio de Contas (RC), Patricinho (P1), and Patricio (P2), located in Piatã, Serra da Tromba, Chapada Diamantina, a semiarid region in the northeastern Brazil (Table 1; Fig. S1). The RC has predominantly grassland vegetation, some trees, shrubs, and lianas in the riparian corridor, but it does not have a closed canopy; P1 has less grassland vegetation in the riparian corridor, while P2 has even more trees and nearly closed canopy (Table S2).
Sampling and laboratory analyses
In September 2013 and May 2014, we visited the study area to collect leaves of Calophyllum brasiliense, an evergreen tree. Green leaves were collected directly from trees in the riparian forest for two of our experiments. Experiment 1 (E1) was carried out from November 2013 to May 2014 and experiment 2 (E2) from July 2014 to January 2015, approximately 8-month duration each to allow sufficient leaf litter decomposition and fungal succession. The rainfall in the study area was approximately two times higher than historical means during December to April period (2013–2014), and the collections made from January 2014 to May 2014 and in January 2015 were categorized as “wet season” while the rest of the samples were considered as “dry season” (Fig. 1). The rainfall and temperature data were obtained from the Instituto Nacional de Metereologia (INMET).
For both experiments, five leaves were enclosed in each fine-mesh litter bag (30 × 30-cm, 0.5-mm mesh size) which were then deployed in streams by tying with nylon line to trees or other riparian vegetation. Every two months, we collected six litter bags from each stream (6 litter bags × 3 streams × 4 collection dates × 2 experiments for the total of 144 bags and 720 leaves). Litter bags were placed in plastic bags and transported to the Laboratory of Mycology (LAMIC), Universidade Estadual de Feira de Santana in a cooler. At each sampling date, we also collected water samples (1 L) that were later analyzed for total nitrogen and phosphorus according to Koroleff (1976) in the Laboratory of Environmental Microbiology, Universidade Federal da Bahia (UFBA). The environmental variables: water temperature, pH, and dissolved oxygen, were measured in the field on each sampling date with portable equipment (Horiba U-50) and the water velocity with the flow meter (Global Water FP111 flow probe).
In the laboratory, leaf litter samples were processed using two methodological approaches: to induce sporulation of freshwater fungi: (1) submerged incubation and (2) incubation in moist chambers.
Submerged incubation following Bärlocher (2005): on each sampling date, leaves from three litter bags per stream were rinsed and twenty 12-mm-diameter discs were cut with a cork borer from each litter bag. These discs were incubated for 48 h at 18–20 °C on a shaker (100 rpm) in 100-mL Erlenmeyer flasks containing 30 mL of sterile distilled water to induce fungal sporulation. Spore suspensions were filtered through membrane filters (5-μm pore size, Millipore), and the filters were then mounted on slides with cotton blue in lactic acid. In addition, the leaf discs were also mounted on slides and examined under compound microscope. All spores on filters and slides with leaf discs were identified and counted.
Incubation in moist chambers following Castañeda-Ruiz et al. (2016): leaf litter samples (from three litter bags per stream) were rinsed, placed in Petri dishes (moist chambers) and incubated in a polystyrene box with sterile water plus glycerol for 30 days. Fungal structures were mounted on slides with lactic acid and sealed with nail polish. Fungi from each slide were identified and counted. Some fungi were also isolated into pure culture to confirm identification.
Data analysis
Litter-associated fungal communities from experimental streams were evaluated using Simpson diversity index and evenness (Magurran 1988). Chao 1 was used to estimate the number of taxa expected in the communities (Chao 1984). While analyzing the relative importance of individual taxa in the communities, the relative abundance was expressed as a percentage of total conidia produced by all taxa (SI), and a percentage of total fungi slides by taxa (MC). Analysis of variance (ANOVA) was performed to test for the effects of stream, experiment, and collection day on fungal sporulation rate (SI). Similarity percentage analysis (SIMPER) was performed to identify the most influential taxa that contributed to the dissimilarity of fungal communities among streams. ANOSIM (Analysis of Similarity) permutation test was also performed to assess the dissimilarities of communities among streams (Clarke 1993). Non-metric Multidimensional Scaling (NMDS) was used to analyze the effects of environmental variables (nutrient availability, oxygen, pH, temperature, and water velocity) on communities of fungi, using Bray-Curtis dissimilarity matrix (Clarke 1993). Successional changes in fungal communities were evaluated through the constancy of species, which was calculated as: C = p × 100/P, (where p = number of collections in which the taxon was present, and P = total number of collections.). We used the following constancy classes: accidental taxon (C ≤ 25%), accessory taxon (25% < C ≤ 50%), and constant taxon (C > 50%) (Dajoz 1983). To compare the methodologies (SI vs. MC), we performed Unweighted Pair Group Method with Arithmetic Mean (UPGMA) cluster analysis based on the Dice similarity matrix.
Analyses were conducted in Past 3.14 (Hammer et al. 2013) and Primer 5.2.6 software (Clarke and Gorley 2006).
Results
Environmental variables
RC, P1, and P2 are oligotrophic streams, with moderate-to-high concentrations of dissolved oxygen and slightly acidic pH. P1 had the highest temperatures while P2 had slightly cooler waters (Table 1).
Sporulation rates in SI
Sporulation rates of freshwater hyphomycetes under submerged incubation conditions (Fig. 2) were significantly higher in E1 than in E2 (ANOVA, F1,60 = 14.1, P = 0.0004). In E1, high sporulation rates were found in RC and P2 on day 120, and P1 on day 180, while in E2, the highest peaks in all streams were achieved on days 180 or 240. These patterns generally corresponded to increased precipitation during the wet seasons (Fig. 1). No differences in sporulation rates among streams or collection days were found (ANOVA, P > 0.05).
Fungal community structure
SI
Taxa richness and diversity
Twenty-six taxa of freshwater hyphomycetes were recorded from submerged leaves of C. brasiliense: one was an aero-aquatic hyphomycete, 13 were Ingoldian fungi, and 12 were facultative aquatic hyphomycetes (Table 2, S3–S5). Taxa richness was comparable among streams with 16, 19, and 22 species, while 11 species were shared by all three streams (Table 2). Dendrosporium lobatum was recorded only in RC, while Scutisporus brunneus was found only in P1, and four species were exclusive to P2: Dactylellina ellipsospora, Dictyotrichocladium aquaticum, Spirosphaera carici-graminis, and Subulispora procurvata. The highest taxa richness was observed during E1 that mostly coincided with the wet season, while fewer species were recorded during E2 (mostly dry season) (Fig. 3, Table 2). Estimates of Chao 1 were close to the actual number of taxa recorded in our samples suggesting an adequate sampling effort. Diversity estimates (Simpson index) and evenness of spore distribution among taxa were generally higher during the E2 than E1 (Table 2).
Succession
The temporal changes in relative abundances of dominant taxa for all streams and both experiments (E1 and E2) are illustrated in Fig. 4. Triscelophorus acuminatus was a dominant or co-dominant species in most cases, with its relative abundance increasing at later stages of decomposition by the end of E2. Morphotype 1 displayed high relative abundance during the middle stages of decomposition (day 60 and day 120), especially during the E2 and in P2. Monotosporella sp. was the dominant species in P1 during E1, while during the E2, it was mostly recorded during the middle stages of decomposition. Overall, Ingoldian fungi were mostly associated with submerged leaves of Calophyllum brasiliense during early-to-middle stages of succession in all streams during E1 and in RC in E2 (Tables S3–S5), while they were more abundant during the later successional stages in P1 and P2 during E2. The constancy of fungal taxa in each stream and experiments (Table S6) shows that during E1, all streams had more constant taxa (2, 5, and 5 in RC, P1, and P2, respectively) than during E2 (3, 4, and 3 taxa), while the number of accessory and accidental species did not show a clear pattern.
Dissimilarity of communities and effects of environmental variables
Similarity percentage analysis (SIMPER) showed that Triscelophorus acuminatus, Monotosporella sp., and Morphotype 1 contributed the most to the differentiation of fungal communities among streams (Table S7). No significant differences in freshwater hyphomycete communities among streams were found by ANOSIM.
UPGMA cluster and NMDS analyses generally showed separation of experiments based on their fungal communities (Figs. 5 and 6). NMDS analysis also allowed to relate fungal communities to environmental variables. Oxygen availability was important in separating fungal communities along coordinate 1 axis, and it was mainly related to the communities found in P1 and P2 during the dry season. Fungal communities were separated along coordinate 2 axis, mainly due to pH being slightly higher in P2 vs. RC on most days. Higher water velocity was associated with more diverse fungal communities (mainly RC_E1_2 and P1_E1_3).
Results of NMDS analysis showing ordination of leaf-associated freshwater hyphomycete communities from submerged incubations. The effects of environmental factors are also shown: total nitrogen (TN), total phosphorus (TP), water temperature (Temp), pH, dissolved oxygen (O2), and water velocity (WV). For sample labels, RC, P1, and P2 refer to streams (Rio de Contas, Patricinho, and Patricio, respectively); E1 and E2 to experiment 1 and 2; and numbers 1 through 4 to sampling days 60, 120, 180, and 240, respectively
Results of UPGMA cluster analysis of leaf-associated freshwater hyphomycete communities from submerged incubations and moist chambers. Treatments are defined by stream (RC, Rio de Contas; P1, Patricinho; P2, Patricio), experiments (E1 or E2), and methodology (SI for submerged incubations and MC for moist chambers)
The UPGMA cluster analysis with samples incubated under submerged conditions and in moist chambers (see below) showed clear separation by the type of incubation (Fig. 6). Moreover, the similarity among samples was higher in case of submerged incubation (> 65%).
MC
Taxa richness and diversity
Fifty-six taxa of freshwater hyphomycetes were recorded in moist chambers; of these, six were aero-aquatic hyphomycetes, six were Ingoldian fungi, and 44 were facultative aquatic hyphomycetes (Table 3, S8–S10). RC had the highest taxa richness with 41 species, while samples from P1 and P2 yielded 32 and 19 species, respectively. All streams shared 10 taxa, while RC, P1, and P2 had 18, 9, and 3 exclusive taxa, respectively. RC and P2 showed the highest taxa richness during E2 (Table 3), while in P1, it was the same in both experiments. In contrast to submerged incubations, estimates of Chao 1 for all streams and both experiments were considerably higher than the number of taxa actually recorded. Simpson diversity indices were comparable across streams and experiments, while the evenness among taxa was considerably lower in E2 than E1 in RC and P2 (Table 3).
Succession
Overall, the highest number of taxa was generally found at later stages of decomposition (day 240) in all streams and in both experiments (Tables S8–S10). In RC, species of Thozetella were co-dominant in E1, being especially important during earlier stages of decomposition (Fig. 7). In P2, Kionochaeta spissa and Chalara alabamensis were highly important on day 180 of E1, while the latter was the dominant species on the very last sampling date in E2. Beltrania rhombica was an important early colonizer in P2 in both experiments, while Dictyochaeta simplex had high relative abundances in P1 throughout both experiments. Overall, the number of constant taxa varied from 2 to 5 among streams and experiments, while the number of accidental taxa was generally higher during E2 than E1 (e.g., up to 24 accidental taxa in RC) (Table S11).
Dissimilarity of communities and effects of environmental variables
SIMPER analysis showed that Dictyochaeta simplex, Triscelophorus acuminatus, and Chalara alabamensis contributed the most to the differentiation of fungal communities among streams (Table S12). ANOSIM showed that streams significantly affected the structure of fungal communities (R = 0.47, P = 0.0001). In contrast to data from submerged incubations, NMDS analysis based on fungal data from moist chambers was not significant and did not provide meaningful ordination (not shown).
Discussion
A total of 69 species of freshwater hyphomycetes have been recorded in this study using two methodologies. However, since SI and MC targeted (and yielded) largely different ecological communities of fungi, the observed patterns will be discussed separately.
SI
Taxa richness and diversity
Most species of freshwater hyphomycetes recovered from C. brasiliense under submerged conditions are Ingoldian fungi (13 species). This was expected since the methodology of Bärlocher (2005) promotes sporulation of this group. The taxa richness recorded is slightly higher than what was previously reported from submerged leaves in Brazil (Schoenlein-Crusius and Milanez 1989; Schoenlein-Crusius and Milanez 1998; Sales et al. 2014). However, some studies in the Neotropics have found a higher number of Ingoldian fungi, e.g., Bärlocher et al. (2010) reported 32 taxa from streams in Panama.
Jabiol et al. (2013) studied aquatic hyphomycete communities along a broad latitudinal gradient and found relatively low diversity in the tropics, which corroborates the earlier observation of Shearer et al. (2007) that aquatic hyphomycetes display the highest diversity in temperate regions. Duarte et al. (2016) reviewing the distribution of aquatic hyphomycetes worldwide came to the same conclusion. Graça et al. (2016) suggested some explanations of the low diversity of aquatic hyphomycetes in tropical streams. One of those is the relatively low dissolved nutrient availability, which was also the case in our study. Another factor that may explain the low diversity in tropical streams is the low quality of substrate, such as leaf litter. Many species of plants in tropical regions produce compounds (tannins and phenols) that inhibit the colonization by fungi and decreasing litter decomposition rates (Canhoto and Graça 1996; Hoorens et al. 2003; Moretti et al. 2007). Calophyllum brasiliense used in this study produces phenolic compounds and calcium oxalate crystals (Junior et al. 2005).
Succession
Most Ingoldian fungi together with some facultative hyphomycetes were found in pioneer stage of succession on decaying leaves of C. brasiliense. Earlier studies of succession on submerged leaves of Ficus microcarpa, Quercus robur, Alchornea triplinervia, Caesalpinia echinata, and Campomanesia phaea conducted in the Atlantic forest of Brazil indicated that Ingoldian fungi typically occur in later stages of succession while facultative aquatic or terrestrial fungi are found during the early phases (Schoenlein-Crusius and Milanez 1989; Schoenlein-Crusius et al. 1990; Schoenlein-Crusius and Milanez 1998; Moreira 2011). Gessner et al. (1993) working with alder leaves in temperate region also found higher richness of Ingoldian fungi at later successional stages. In our study, we did not find clear replacement of species through stages of succession; some taxa were constant from pioneer stages until the end of the study, while a few taxa occurred just in early stages. Gessner et al. (1993) also did not observe clear species replacement as expected from classical succession studies.
All streams in E1 showed pioneer communities with high species richness. The impoverished communities at the later stages of decomposition were in agreement with a pattern suggested by Yanna et al. (2002). In E2, we did not generally observe this pattern probably because of delay in colonization due to the presence of green spots in collected leaves. According to Bärlocher (1991), when the leaching does not occur, the colonization by aquatic hyphomycetes could be delayed.
Beltrania rhombica and Thozetella havanensis were observed only in early stages (until 120 days) of succession. According to Hyde et al. (2015), some species found in terrestrial situations cannot survive for a long time under submerged conditions.
MC
Taxa richness and diversity
Moist chamber incubations favor sporulation by taxa of facultative aquatic hyphomycetes and aero-aquatic hyphomycetes due to simulation of a terrestrial humid environment. Some Ingoldian fungi may also sporulate as previously observed from Brazilian Caatinga and Amazon (Fiuza et al. 2017).
Most studies of freshwater hyphomycetes in moist chambers that dealt with submerged wood showed higher diversity of facultative aquatic hyphomycetes (up to 115 taxa) than what was found in the present study, while observations from leaf litter yielded comparable results (Tsui et al. 2001; Castañeda-Ruiz et al. 2010, 2016; Fiuza et al. 2015; Hyde et al. 2015; Kodsueb et al. 2016).
Succession
In most cases, we found the highest richness of freshwater hyphomycetes at later stages of the experiments, which may correspond to a mature stage of succession as predicted by Dix and Webster (1995). Communities of saprotrophic fungi often demonstrate species replacement as succession progresses (Gessner et al. 1993; Rayner and Todd 1979), and we have found a similar pattern in moist chambers as suggested by high number of accessory or rare species in all streams.
Cylindrocladiella infestans that occurred during the first sampling date (day 60) in all streams in E2 is often isolated as endophyte or pathogen from leaves (Brown and Ferreira 2000; Evans et al. 2003). Since our leaves had large green spots when collected, it is likely that it continued growing and then sporulated in moist chambers, a saprotrophic pattern described for some endophytes earlier (Promputtha et al. 2007, 2010).
Comparison between methodologies
We did not see a great overlap between submerged and moist chamber incubations with respect to the recovered fungal taxa (only 13 of 69 taxa were recorded by both techniques). This suggests that using several methodological approaches may help maximize the number of taxa recovered in ecological studies of litter-associated fungi in streams. Sridhar et al. (2010) studying aquatic fungi on wood using damp incubation and bubble chamber methodologies have found even smaller overlap of just one species.
In most cases, moist chamber incubations yielded higher fungal diversity (Simpson index) than submerged incubations because of the greater degree of dominance displayed by samples in the latter method. Moreover, evenness was also higher in moist chambers. UPGMA results clearly showed separation of samples based on methodology.
Sporulation rates, multivariate analyses, and effects of environmental variables
We observed the highest sporulation rates and species richness during E1 that was started at the end of the dry season and spanned all of the wet season. In Brazil, the litterfall occurs at the end of the dry season due to hydrological stress, and with the beginning of the wet season, increases in leaf litter standing stock in streams coincide with inputs of nutrients from the riparian zone stimulating microbial activity (Gonçalves et al. 2014; Rezende et al. 2016). Sales et al. (2014), who also worked in Bahia (Chapada Diamantina), found peaks of fungal sporulation during the wet season. Ghate and Sridhar (2016) studying aquatic hyphomycetes on leaves of palm trees also detected the highest richness during the wet season. Thus, it appears that both in tropical and temperate regions, the seasonal patterns of activity of leaf-associated fungi in streams are driven by the availability of resources (litterfall). However, in this and some other studies in the tropics, the timing of the litterfall was determined by seasonal precipitation patterns, while in the temperate regions, it is driven by temperature declines in the fall (Shearer et al. 2007; Sridhar and Sudheep 2010; Graça et al. 2016). Graça et al. (2016) also suggested that increased water turbulence during the wet season in tropical regions stimulates activity; sporulation; and, hence, concentration of conidia of aquatic hyphomycetes in water, which facilitates colonization of new substrates.
In moist chamber incubations, the highest richness of freshwater hyphomycetes was observed in streams RC and P2 during the second experiment (E2), which was conducted mostly during the dry season, when the stream flow is low and some plant litter may become partially emergent. According to SIMPER results, fungal communities in these incubations were dominated by fungi common in terrestrial ecosystems, with the exception of Triscelophorus acuminatus. For example, Dictyochaeta simplex is usually found on submerged or terrestrial leaves, twigs, and grasses (Wong and Hyde 2001; Paulus et al. 2006), while Chalara alabamensis has been recorded several times on leaves or wood in terrestrial situations (Morgan-Jones and Ingram 1976; Rambelli et al. 2004).
According to SIMPER analysis, Triscelophorus acuminatus was the dominant or co-dominant species in both submerged and moist chamber incubations in all streams. It is a species with worldwide distribution (Fiuza and Gusmão 2013); however, it is often reported from tropical areas or from temperate streams during the warmer seasons (Jabiol et al. 2013). In tropical areas of India, Sridhar and Sudheep (2010) observed the high production of spores by T. acuminatus, as well as Amniculicola longissima, Flagellospora curvula, Lunulospora curvula, and T. monosporus, which are also reported in the current study.
Temperature is known to affect sporulation rate of aquatic hyphomycetes (Chauvet and Suberkropp 1998), plant litter decomposition (Boyero et al. 2016), and fungal communities in streams due to differences in temperature optima displayed by fungal species (i.e., cold-water vs. warm-water species) (Duarte et al. 2013). In our study, most species were typical of warm waters, water temperature did not vary greatly throughout the experiments, and thus, NMDS analysis did not show a great effect of temperature on fungal communities. On the other hand, NMDS revealed an apparent effect of oxygen concentration in structuring fungal communities driven mostly by samples from the second experiment with higher O2 concentrations. These fungal communities (P1 and P2 in dry season) had lower diversity and abundance of freshwater hyphomycetes conidia. Experimental studies that examined the wide range of oxygen concentrations found positive effects on diversity and sporulation of aquatic hyphomycetes (Medeiros et al. 2009). However, in our experiments, stream oxygen availability did not vary to the same extent, and it is also possible that our observed pattern could be due to the confounding effects of other variables. The positive effect of water velocity on NMDS ordination was driven mostly by fungal communities during the first experiment (mainly RC_E1_2 and P1_E1_3). Sanders and Webster (1980) found that higher flow can stimulate fungal sporulation due to enhanced water turbulence. Higher pH and total nitrogen in water were related to fungal communities in P2 during the first experiment. The effect of total phosphorus was rather minor. Several studies on the effects of nutrient enrichment (Gulis and Suberkropp 2003, 2004; Ferreira et al. 2015) found that dissolved inorganic nutrients can greatly stimulate the activity of aquatic litter-associated fungi, including their sporulation rate with concomitant changes in community structure, and ultimately affect fungi-driven decomposition of leaf litter in streams and rivers.
Conclusions
Our data demonstrate the importance of using several methodological approaches to maximize the number of taxa recovered in ecological studies of litter-associated fungal communities. Submerged and aerial incubations provided complementary insights into fungal community structure since they favored fungal groups adapted to different sporulation environments. Successional patterns also differed between the two methodologies: the submerged incubations showed the highest richness of freshwater hyphomycetes during pioneer and mature stages of succession, while in moist chambers, many taxa were observed at the final stages of the experiment. Overall, the richness (69 taxa) of freshwater hyphomycetes associated with submerged leaves of C. brasiliense was lower than often reported from temperate streams. At the same time, we found that samples in submerged incubations were more sensitive to differences in environmental variables driven by seasonal precipitation. Fungal communities differed between the experiments, and the highest fungal sporulation rates were observed during the wet season. The input of leaves, changes in oxygen concentration, and water velocity likely affected the communities of freshwater hyphomycetes associated with submerged leaf litter.
This study improves our understanding of the freshwater hyphomycete communities in streams of the semiarid regions of Brazil and the environmental variables that control the structure of these communities, which is especially important since ecological studies of aquatic fungi in Brazil and the tropical regions in general are still uncommon.
References
Barbosa FR, Raja HA, Shearer CA, Gusmão LFP (2013) Some freshwater fungi from the Brazilian semi-arid region, including two new species of hyphomycetes. Cryptogam Mycol 34:243–258. https://doi.org/10.7872/crym.v34.iss2.2013.243
Bärlocher F (1991) Fungal colonization of fresh and dried leaves in the River Teign (Devon, England). Nova Hedwigia 52(3–4):349–357
Bärlocher F (2005) Sporulation of aquatic hyphomycetes. In: Graça MAS, Bärlocher F, Gessner MO (eds) Methods to study litter decomposition: a practical guide. Springer, Netherlands, pp 185–187
Bärlocher F, Helson JE, Williams DD (2010) Aquatic hyphomycete communities across a land-use gradient of Panamanian streams. Fundam Appl Limnol 177(3):209–221. https://doi.org/10.1127/1863-9135/2010/0177-0209
Boyero L, Pearson RG, Hui C, Gessner M, Pérez J et al (2016) Biotic and abiotic variables influencing plant litter breakdown in streams: a global study. Proc R Soc B 283(1829):1–10. https://doi.org/10.1098/rspb.2015.2664
Brown BN, Ferreira FA (2000) Disease during propagation of eucalypts. In: Keane PJ, Kile GA, Podger FD, Brown BN (eds) Diseases and Pathogens of Eucalypts. Csiro Publishing, Melbourne, pp 119–151
Canhoto C, Graça MAS (1996) Decomposition of Eucalyptus globulus leaves and three native leaf species Alnus glutinosa, Castanea sativa and Quercus faginea in a Portuguese low order stream. Hydrobiologia 333:79–85. https://doi.org/10.1007/bf00017570
Castañeda-Ruiz RF, Minter DW, Stadler M, Gené J, Guarro J, Cano J (2010) Two new anamorphic fungi from Cuba: Endophragmiella profusa sp. nov. and Repetoblastiella olivacea gen. & sp. nov. Mycotaxon 113:415–422. https://doi.org/10.5248/113.415
Castañeda-Ruiz RF, Heredia G, Gusmão LFP, Li D-W (2016) Fungal diversity of Central and South America. In: Li D-W (ed) Biology of microfungi. Springer, Switzerland, pp 197–217
Chan SY, Goh TK, Hyde KD (2000) Ingoldian fungi in Lam Tsuen River and Tai Po Kau Forest Stream, Hong Kong. In: Hyde KD, Ho WH, Pointing SB (eds) Aquatic mycology across the Millenium, 5. Fungal Diversity press, Thailand, pp 109–118.
Chao A (1984) Non-parametric estimation of the number of classes in a population. Scand J Stat 11:265–270
Chauvet E, Suberkropp K (1998) Temperature and sporulation of aquatic hyphomycetes. Appl Environ Microbiol 64:1522–1525
Chauvet E, Cornut J, Sridhar KR, Selosse M, Bärlocher F (2016) Beyond the water column: aquatic hyphomycetes outside their preferred habitat. Fungal Ecol 19:112–127. https://doi.org/10.1016/j.funeco.2015.05.014
Clarke KR (1993) Non-parametric multivariate analysis of changes in community structure. Aust J Ecol 18:117–143. https://doi.org/10.1111/j.1442-9993.1993.tb00438.x
Clarke KR, Gorley RN (2006) PRIMER v6: User Manual/Tutorial. PRIMER-E, Plymouth.
Dajoz R (1983) Ecologia Geral. Vozes, Petrópolis.
Descals E, Sanders PF, Ugalde UO (1977) Hifomicetos ingoldianos del pais vasco. Munibe 29:237–260
Descals E, Moralejo E (2001) Water and asexual reproduction in the Ingoldian fungi. Botanica Complutensis 25:13–71
Dix NJ, Webster J (1995) Fungal ecology. Chapman and Hall, Springer, London.
Duarte S, Fernandes I, Nogueira MJ, Cássio F, Pascoal C (2013) Temperature alters interspecific relationships among aquatic fungi. Fungal Ecol 6:187–191. https://doi.org/10.1016/j.funeco.2013.02.001
Duarte S, Bärlocher F, Pascoal C, Cassio F (2016) Biogeography of aquatic hyphomycetes: current knowledge and future perspectives. Fungal Ecol 19:169–181. https://doi.org/10.1016/j.funeco.2015.06.002
Evans HC, Holmes KA, Thomas SE (2003) Endophytes and mycoparasites associated with an indigenous forest tree, Theobroma gileri, in Ecuador and a preliminary assessment of their potential as biocontrol agents of cocoa diseases. Mycol Prog 2(2):149–160. https://doi.org/10.1007/s11557-006-0053-4
Ferreira V, Gulis V, Pascoal C, Graça MAS (2014) Stream pollution and fungi. In: Jones EBG, Hyde KD, Pang K-L (eds) Freshwater fungi: and fungi-like organisms. Walter de Gruyter GmbH and Co KG, Berlin, pp 389–412
Ferreira V, Castagneyrol B, Koricheva J, Gulis V, Chauvet E, Graça MAS (2015) A meta-analysis of the effects of nutrient enrichment on litter decomposition in streams. Biol Rev 90(3):669–688. https://doi.org/10.1111/brv.12125
Fisher PJ (1977) New methods of detecting and studying saprophytic behavior of aero-aquatic hyphomycetes from stagnant water. Trans Br Mycol Soc 68:407–411. https://doi.org/10.1016/s0007-1536(77)80194-0
Fiuza PO, Gusmão LFP (2013) Ingoldian fungi from the semi-arid Caatinga biome of Brazil. Mycosphere 4:1133–1150. https://doi.org/10.5943/mycosphere/4/6/10
Fiuza PO, Gusmão LFP, Castañeda-Ruiz RF (2015) Conidial fungi from the semiarid Caatinga biome of Brazil: a new species of Selenosporella from submerged leaves. Mycotaxon 1303:601–605. https://doi.org/10.5248/130.601
Fiuza PO, Cantillo-Pérez T, Gulis V, Gusmão LFP (2017) Ingoldian fungi of Brazil: some new records and a review including a checklist and a key. Phytotaxa 306(3):171–200. https://doi.org/10.11646/phytotaxa.306.3.1
Gessner MO, Thomas M, Jean-Louis AM, Chauvet E (1993) Stable successional patterns of aquatic hyphomycetes on leaves decaying in a summer cool stream. Mycol Res 97:163–172. https://doi.org/10.1016/s0953-7562(09)80238-4
Ghate SD, Sridhar KR (2016) Aquatic hyphomycetes associated with leaves, leaf detritus and crown humus in palm canopies. Czech Mycol 68(2):111–126
Gonçalves JF, Rezende RS, Gregório RS, Valentin GC (2014) Relationship between dynamics of litterfall and riparian plant species in a tropical stream. Limnologica 44:40–48. https://doi.org/10.1016/j.limno.2013.05.010
Graça MAS, Hyde K, Chauvet E (2016) Aquatic hyphomycetes and litter decomposition in tropical e subtropical low order streams. Fungal Ecol 19:182–189. https://doi.org/10.1016/j.funeco.2015.08.001
Gulis V (2001) Are there any substrate preferences in aquatic hyphomycetes? Mycol Res 1059:1088–1093. https://doi.org/10.1016/s0953-7562(08)61971-1
Gulis V, Suberkropp K (2003) Leaf litter decomposition and microbial activity in nutrient-enriched and unaltered reaches of a headwater stream. Freshw Biol 48:123–134. https://doi.org/10.1046/j.1365-2427.2003.00985.x
Gulis V, Suberkropp K (2004) Effects of whole-stream nutrient enrichment on the concentration and abundance of aquatic hyphomycete conidia in transport. Mycologia 96:57–65. https://doi.org/10.1080/15572536.2005.11832997
Gulis V, Kuehn KA, Suberkropp K (2006) The role of fungi in carbon and nitrogen cycles in freshwater ecosystems. In: Gadd GM (ed) Fungi in biogeochemical cycles. Cambridge University Press, Cambridge, pp 404–435
Gulis V, Suberkropp K, Rosemond AD (2008) Comparison of fungal activities on wood and leaf litter in unaltered and nutrient-enriched headwater streams. Appl Environ Microbiol 744:1094–1101. https://doi.org/10.1128/aem.01903-07
Hammer Ø, Harper DAT, Ryan PD (2013) PAST: paleontological statistics software package for education and data analysis. Paleontological Statistics, PAST, 3.0 the Past of the Future. http://folk.uio.no/ohammer/past/
Ho WH, Yanna HKD, Hodgkiss IJ (2002) Seasonality and sequential occurrence of fungi on wood submerged in Tai Po Kau Forest Stream, Hong Kong. In: Hyde KD, Jones EBG (eds) Fungal succession. Fungal Diversity Springer, Netherlands, pp 21–43
Hoorens B, Aerts R, Stroetenga M (2003) Does initial litter chemistry explain litter mixture effects on decomposition? Oecologia 137:578–586. https://doi.org/10.1007/s00442-003-1365-6
Hissy FT, Khallil AM, Abdel-R Raheem AA (1992) Occurrence and distribution of zoosporic fungi and aquatic hyphomycetes in Upper Egypt. J Islamic Acadf Sci 53:173–179.
Hyde KD, Fryar S, Tian Q, Bahkali AH, Xu JC (2015) Lignicolous freshwater fungi along a north-south latitudinal gradient in the Asian/Australian region: can we predict the effects of global warming on biodiversity and function? Fungal Ecology https://doi.org/10.1016/j.funeco.2015.07.002.
Jabiol J, Bruder A, Gessner MO, Makkonen M, McKie BG, Peeters ETHM, Vos VCA, Chauvet E (2013) Diversity patterns of leaf-associated aquatic hyphomycetes along a broad latitudinal gradient. Fungal Ecol 6:439–448. https://doi.org/10.1016/j.funeco.2013.04.002
Junior AG, Ferreira ICP, Nakamura CV, Filho BPD, Jacomassi E, Young MCM, Cortez DAG (2005) Estudo morfo-anatômico das folhas e caule da Calophyllum brasiliense Clusiaceae, uma contribuição ao estudo farmacognóstico da droga vegetal. Acta Farm Bonaer 243:371–376
Kominoski JS, Rosemond AD, Benstead JP, Gulis V, Maerz JC, Manning DWP (2015) Low-to-moderate nitrogen and phosphorus concentrations accelerate microbially driven litter breakdown rates. Ecol Appl 25(3):856–865. https://doi.org/10.1890/14-1113.1
Koroleff F (1976) Determination of nutrients. In: Grasshoffk (ed) Methods of seawater analysis. Verlag Chemie, Weinhein, pp117–181.
Kodsueb R, Lumyong S, McKenzie EHC, Bahkali AH, Hyde KD (2016) Relationships between freshwater and terrestrial lignicolous fungi. Fungal Ecol 19:155–168. https://doi.org/10.1016/j.funeco.2015.09.005
Magurran AE (1988) Ecological diversity and its measurement. Princeton University Press, Princeton
Medeiros AO, Pascoal C, Graça MAS (2009) Diversity and activity of aquatic fungi under low oxygen conditions. Freshw Biol 54:142–149. https://doi.org/10.1111/j.1365-2427.2008.02101.x
Moreira CG (2011) Sucessão de hifomicetos e avaliação da biomassa fúngica durante a decomposição de folhedo de Caesalpinia echinata Lam. e Campomanesia phaea O. Berg. Landrum submersos em lagos artificiais na cidade de São Paulo, SP. In: PhD Thesis. Instituto de Botânica, São Paulo
Moretti M, Gonçalves JF, Callisto M (2007) Leaf breakdown in two tropical streams: differences between single and mixed species packs. Limnologica 373:250–258. https://doi.org/10.1016/j.limno.2007.01.003
Morgan-Jones G, Ingram EG (1976) Notes on hyphomycetes. XII. A new species of Chalara. Mycotaxon. 4(2):489–492
Newman MM, Liles MR, Feminella JK (2015) Litter breakdown and microbial succession on two submerged leaf species in a small forested stream. PLoS One 10(6):1–22. https://doi.org/10.1371/journal.pone.0130801
Paulus BC, Kanowski J, Gadek PA, Hyde KD (2006) Diversity and distribution of saprobic microfungi in leaf litter of an Australian tropical rainforest. Mycol Res 110:1441–1454. https://doi.org/10.1016/j.mycres.2006.09.002
Promputtha I, Lumyong S, Dhanasekaran V, McKenzie EHC, Hyde KD, Jeewon R (2007) A phylogenetic evaluation of whether endophytes become saprotrophs at host senescence. Microb Ecol 53:579–590. https://doi.org/10.1007/s00248-006-9117-x
Promputtha I, Hyde KD, McKenzie EHC, Peberdy JFP, Lumyong S (2010) Can leaf degrading enzymes provide evidence that endophytic fungi becoming saprobes? Fungal Divers 41:89–99. https://doi.org/10.1007/s13225-010-0024-6
Rajashekhar M, Kaveriappa KM (2000) Effects of temperature and light on growth and sporulation of aquatic hyphomycetes. Hydrobiologia 441:149–153
Rambelli A, Mulas B, Pasqualetti M (2004) Comparative studies on microfungi in tropical ecosystems in Ivory Coast forest litter: behaviour on different substrata. Mycol Res 108(3):325–336
Rayner ADM, Todd NK (1979) Population and community structure and dynamics of fungi in decaying wood. Adv Bot Res 7:333–420. https://doi.org/10.1016/s0065-2296(08)60090-7
Rezende RS, Graça MAS, Santos AM, Medeiros AO, Santos PF, Nunes RF, Gonçalves JF (2016) Organic matter dynamics in a tropical gallery forest in a grassland landscape. Biotropica 48(3):301–310. https://doi.org/10.1111/btp.12308
Sales MA, Gonçalves JF, Dahora J, Medeiros AO (2014) Influence of leaf quality on microbial decomposition in a headwater stream in the Brazilian Cerrado: a 1-year study. Microb Ecol 69:84–94. https://doi.org/10.1007/s00248-014-0467-5
Sanders PF, Webster J (1980) Sporulation responses of some ‘aquatic hyphomycetes’ in flowing water. Trans Br Mycol Soc 74:601–605. https://doi.org/10.1016/s0007-1536(80)80062-3
Schoenlein-Crusius IH, Milanez AI (1989) Sucessão fúngica em folhas de Ficus microcarpa L. f. submersas no Lago Frontal situado no Parque Estadual das Fontes do Ipiranga, São Paulo. Rev Microbiol 20:95–101
Schoenlein-Crusius IH, Pires CLA, Milanez AI (1990) Sucessão fúngica em folhas de Quercus robur L. Carvalho submersas em um lago situado no município de Itapecerica da Serra, SP. Rev Microbiol 21:61–67
Schoenlein-Crusius IH, Milanez AI (1998) Fungal Succession on leaves of Alchornea triplinervia Spreng. M. Arg. submerged in a stream of an Atlantic rainforest in the State of São Paulo, Brazil. Rev Bras Bot 21:253–259. https://doi.org/10.1590/s0100-84041998000300003
Shearer CA, Webster J (1985) Aquatic hyphomycete communities in the river Teign. II. Temporal distribution patterns. Trans Br Mycol Soc 84(3):503–507. https://doi.org/10.1016/s0007-1536(85)80012-7
Shearer CA, Webster J (1991) Aquatic hyphomycete communities in the river Teign. IV. Twig colonization. Mycol Res 95(4):413–420. https://doi.org/10.1016/s0953-7562(09)80839-3
Shearer CA, Descals E, Kohlmeyer B, Kohlmeyer J, Marvanová L, Padgett D, Porter D, Raja HA, Schmit JP, Thorton HA, Voglymayr H (2007) Fungal biodiversity in aquatic habitats. Biodivers Conserv 16:49–67. https://doi.org/10.1007/s10531-006-9120-z
Sridhar KR, Sudheep NM (2010) Diurnal fluctuation of spores of freshwater hyphomycetes in two tropical streams. Mycosphere 1(2):89–101
Sridhar KR, Karamchand KS, Hyde KD (2010) Wood-inhabiting filamentous fungi in 12 high altitude streams of the Western Ghats by damp incubation and bubble chamber incubation. Mycoscience 51:104–115. https://doi.org/10.1007/s10267-009-0017-z
Tsui CKM, Hyde KD, Hodgkiss IJ (2001) Longitudinal and temporal distribution of ascomycetes and dematacious hyphomycetes on submerged wood in the Lam Tsuen River. J N Am Benthol Soc 20:601–615. https://doi.org/10.2307/1468086
Webster J (1975) Further studies of sporulation of aquatic hyphomycetes in relation to aeration. Trans Br Mycol Soc 641:119–127. https://doi.org/10.1016/s0007-1536(75)80082-9
Wong MKM, Hyde KD (2001) Diversity of fungi on six species of Gramineae and one species of Cyperaceae in Hong Kong. Mycol Res 105(12):1485–1491. https://doi.org/10.1017/s0953756201004695
Yanna, Ho WH, Hyde KD (2002) Fungal succession on fronds of Phoenix hanceana in Hong Kong. In: Hyde KD, Jones EBG (eds) Fungal succession. Fungal Diversity Springer, Netherlands, pp 183–209.
Funding
The authors are grateful to the “Programa de Pesquisa em Biodiversidade”—(PPBio Semi-arid/MCTI/CNPq) for financial support. POF thanks Coordenação de Aperfeiçoamento de Pessoal de Nível Superior—Programa de Doutorado Sanduíche no Exterior (CAPES-PDSE) for scholarship “Sciences Without Borders” (Proc. 99999.000984/2015-09) that enabled her stay at the Coastal Carolina University. POF also thanks “Programa de Pós-graduação em Botânica PPGBot/UEFS.” LFPG is grateful to CNPq for financial support (Proc. 303062/2014-2).
Author information
Authors and Affiliations
Corresponding author
Additional information
Section Editor: Dominik Begerow
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
ESM 1
(PDF 1267 kb)
Rights and permissions
About this article
Cite this article
Fiuza, P.O., Costa, L.A., Medeiros, A.O. et al. Diversity of freshwater hyphomycetes associated with leaf litter of Calophyllum brasiliense in streams of the semiarid region of Brazil. Mycol Progress 18, 907–920 (2019). https://doi.org/10.1007/s11557-019-01501-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11557-019-01501-6