Abstract
Malaria is an infectious disease caused by Plasmodium parasites and is transmitted among humans by female Anopheles mosquitoes. Climate factors have significant impact on both mosquito life cycle and parasite development. To consider the temperature sensitivity of the extrinsic incubation period (EIP) of malaria parasites, we formulate a delay differential equations model with a periodic time delay. We derive the basic reproduction ratio \(R_0\) and establish a threshold type result on the global dynamics in terms of \(R_0\), that is, the unique disease-free periodic solution is globally asymptotically stable if \(R_0<1\); and the model system admits a unique positive periodic solution which is globally asymptotically stable if \(R_0>1\). Numerically, we parameterize the model with data from Maputo Province, Mozambique, and simulate the long-term behavior of solutions. The simulation result is consistent with the obtained analytic result. In addition, we find that using the time-averaged EIP may underestimate the basic reproduction ratio.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
1 Introduction
Malaria is the most prevalent human vector-borne disease, with an estimated 214 million cases and about 438 thousand deaths worldwide in 2015 (World Health Organisation 2015). Malaria is caused by Plasmodium protozoan parasites and is transmitted among humans by the bites of female Anopheles mosquitoes. More precisely, female Anopheles mosquitoes pick up Plasmodium parasites in a blood meal taken from an infectious human host. The parasites then go through several developmental stages before they migrate to the mosquito salivary glands. Once in the salivary glands, the parasites can be transmitted to a susceptible human host when the mosquito takes another blood meal (Beier 1998; Beck-Johnson et al. 2013). The time parasites spent in completing its development within the mosquito and migrating to the salivary glands is known as the extrinsic incubation period (EIP).
Both the mosquito life cycle and the parasite development are strongly influenced by seasonally varying temperature. Understanding the role of temperature in malaria transmission is of particular importance in light of climate change (Ngarakana-Gwasira et al. 2014). The first mathematical model of malaria transmission was proposed by Ross (1911) and later extended by Macdonald (1957). Since then, a number of malaria models have been developed to study the climate impact on malaria transmission (see, e.g., Craig et al. 1999; Lou and Zhao 2010; Ngarakana-Gwasira et al. 2014; Wang and Zhao 2017; Wang and Zhao, and the references therein). Malaria parasites manipulate a host to be more attractive to mosquitoes via the chemical substances (Lacroix et al. 2005). Kingsolver (1987) proposed the first malaria model to account for the greater attractiveness of infectious humans to mosquitoes. Chamchod and Britton (2011) modeled such vector-bias effect in terms of the different probabilities that a mosquito arrives at a human at random and picks the human if he is infectious or susceptible. Recently, the vector-bias effect has also been incorporated into some climate-based malaria models (see, e.g., Wang and Zhao 2017; Wang and Zhao).
In our recent work, we developed a periodic vector-bias malaria model with incubation period and established the global dynamics in terms of the basic reproduction ratio. We remarked that the constant delay in the model may be modified to a time-dependent delay since the EIP is highly sensitive to temperature, and we left this interesting problem for future investigation (Wang and Zhao 2017). The aim of the current work is to solve this problem. We develop a delay differential equations model of malaria transmission in which the delay is periodic in time. To our knowledge, this is the first mosquito-borne disease model that takes into account the time-dependent delay. Several population models with time-dependent delays have been developed (see, e.g., Beck-Johnson et al. 2013; McCauley et al. 1996; Molnár et al. 2013; Nisbet and Gurney 1983; Omori and Adams 2011; Rittenhouse et al. 2016; Wu et al. 2015). However, little mathematical analysis is carried out to understand the asymptotic behavior of these models. Recently, Lou and Zhao (2017) studied the global dynamics of a host–macroparasite model with seasonal developmental durations by introducing a periodic semiflow on a suitably chosen phase space. We will use the theoretical approach developed in Lou and Zhao (2017) to analyze our model.
The rest of the paper is organized as follows. In the next section, we give the underlying assumptions and formulate the model. In the following section, we establish the threshold dynamics of the model in terms of the basic reproduction ratio. In Sect. 4, we carry out a case study for Maputo Province, Mozambique. A brief discussion concludes the paper.
2 Model Formulation
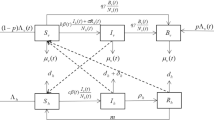
The purpose of this section is to formulate a mathematical model of malaria transmission that incorporates a temperature-dependent delay. We start with a brief introduction of the model of malaria transmission proposed by Wang and Zhao (2017). Since malaria is transmitted among humans by mosquitoes, we consider the dynamics of both human and mosquito populations. Let the state variables \(I_h(t)\), \(S_m(t)\), \(I_m(t)\) be the number of infectious humans, susceptible and infectious adult female mosquitoes at time t, respectively. We suppose that the total number of humans stabilizes at a constant value H. Then the number of susceptible humans at time t is \(H-I_h(t)\). We incorporate climate factors into the model by assuming that all the parameters related to mosquitoes are periodic functions. Let \(\beta (t)\) be the biting rate of mosquitoes. Let \(\mu (t)\) be the recruitment rate at which female adult mosquitoes emerge from larvae, and let \(d_m(t)\) be the death rate of mosquitoes. To depict the vector-bias effect, we introduce two parameters p and l, that is, the probabilities that a mosquito arrives at a human at random and picks the human if he is infectious and susceptible, respectively. Let \(\tau \) be the length of the EIP. The model of the reference (Wang and Zhao 2017) is governed by the following system of time-delayed differential equations:
We refer the readers to Wang and Zhao (2017) for more details about the derivation of model (1). To introduce the temperature-dependent incubation period, we use the arguments similar to those in Nisbet and Gurney (1983), Omori and Adams (2011). We consider the exposed compartment where mosquitoes are infected but not infectious yet. Let \(E_m(t)\) be the number of the exposed mosquitoes at time t, and M(t) be the number of newly occured infectious mosquitoes per unit time at time t. Then we have the following system:
where \(B(t,I_h(t),S_m(t))=\frac{b\beta (t)pI_h(t)S_m(t)}{pI_h(t)+l(H-I_h(t))}\). Let q be the development level of infection such that q increases at a temperature-dependent rate \(\gamma (T(t))=\gamma (t)\), \(q=q_E=0\) at the transition from \(S_m\) to \(E_m\), and \(q=q_I\) at the transition from \(E_m\) to \(I_m\). The variable q describes how complete the parasite developmental stages are in the mosquito (in other words, how complete the latency stage is). Let \(\rho (q,t)\) be the number of mosquitoes with development level q at time t. Then \(M(t)=\gamma (t)\rho (q_I,t)\).
Let J(q, t) be the flux, in the direction of increasing q, of mosquitoes with infection development level q at time t. Then we have the following equation (see, e.g., Kot 2001)
Since \(J(q,t)=\gamma (t)\rho (q,t)\), we have
For the \(E_m\) state, system (3) has the boundary condition
To solve system (3) with this boundary condition, we introduce a new variable
Let \(h^{-1}(\eta )\) be the inverse function of h(t), and define
In view of (3), we then have
This equation is identical in form to the standard von Foerster equation (see Nisbet and Gurney 1982). Let \(V(s)={\hat{\rho }} (s+q-\eta ,s)\). It follows from (4) that
Since \(\eta -(q-q_E)\le \eta \), we have
and hence,
Define \(\tau (q,t)\) to be the time taken to grow from infection development level \(q_E\) to level q by a mosquito who arrives at infection development level q at time t. Since \(\frac{\mathrm{d}q}{\mathrm{d}t}=\gamma (t)\), it follows that
and hence,
By a change of variable \(s=h(\xi )\), we then see that
It follows that
Define \(\tau (t):=\tau (q_I,t)\). We then obtain
Letting \(q=q_I\) in (5), we have
Taking the derivative with respect to t on both sides of (6), we obtain
Thus, there holds \(1-\tau '(t)>0\). In virtue of (6), it easily follows that if \(\gamma (t)\) is a periodic function, then so is \(\tau (t)\) with the same period. Substituting \(M(t)=\gamma (t)\rho (q_I,t)\) into system (2), we arrive at the following model system:
where all constant parameters are positive, and \(\mu (t)\), \(\beta (t)\), \(d_m(t)\), \(\tau (t)\) are positive, continuous and \(\omega \)-periodic functions t for some \(\omega >0\). The biological interpretations for parameters are listed in Table 1. It is easy to see that the function
is also \(\omega \)-periodic. Thus, model (7) can be written as \(u'(t)=F(t,u_t)\) with \(F(t+\omega ,\phi )=F(t,\phi )\) (see the proof of Lemma 2), and hence, it is an \(\omega \)-periodic functional differential system. Note that the term \(1-\tau '(t)\) is involved in the development rate from the \(E_m\) state to the \(I_m\) state, which is different from previous works with constant time delays [see, e.g., system (1) in Wang and Zhao (2017)].
3 Threshold Dynamics
In this section, we study the global dynamics of system (7). The basic reproduction ratio is a key threshold parameter providing information for disease risk and control (see, e.g., Diekmann et al. 1990; Driessche and Watmough 2002). There have been quite a few investigations on \(R_0\) analysis for population models in a periodic environment (see, e.g., Bacaër and Ait Dads 2012; Bacaër and Guernaoui 2006; Inaba 2012; Thieme 2009; Wang and Zhao 2008; Zhao 2017 and the references therein). In what follows, we will use the theory recently developed in Zhao (2017) to derive the basic reproduction ratio \(R_0\). Since the third equation of system (7) is decoupled from the other equations, it suffices to study the following system:
It is easy to see that the scalar linear periodic equation
has a unique positive \(\omega \)-periodic solution
which is globally attractive in \(\mathbb {R}\).
Linearizing system (8) at its disease-free periodic solution \((0,S_m^*(t),0)\), we then obtain the following system of periodic linear equations for the infective variables \(I_h\) and \(I_m\):
where \(a_{11}(t)=d_h+\rho \), \(a_{12}(t)=c\beta (t)\), \(a_{22}(t)=d_m(t)\), and
Let \(\hat{\tau }=\max _{0\le t\le \omega }\tau (t),\,\, C=C([-{\hat{\tau }},0],\mathbb {R}^2)\), \(C^+=C([-{\hat{\tau }},0],\mathbb {R}_+^2).\) Then \((C,C^+)\) is an ordered Banach space equipped with the maximum norm and the positive cone \(C^+\). For any given continuous function \(v=(v_1,v_2): [-{\hat{\tau }},\sigma )\rightarrow \mathbb {R}^2\) with \(\sigma >0\), we define \(v_t\in C\) by
for any \(t\in [0,\sigma ).\) Let \(F:\mathbb {R}\rightarrow \mathcal {L}(C,\mathbb {R}^2)\) be a map and V(t) be a continuous \(2\times 2\) matrix function on \(\mathbb {R}\) defined as follows:
Then the internal evolution of the infective compartments \(I_h\) and \(I_m\) can be expressed by
Let \({\varPhi }(t,s), t\ge s,\) be the evolution matrix of the above linear system. That is, \({\varPhi }(t,s)\) satisfies
and
where I is the \(2\times 2\) identity matrix. It then easily follows that
Let \(C_\omega \) be the ordered Banach space of all continuous and \(\omega \)-periodic functions from \(\mathbb {R}\) to \(\mathbb {R}^2\), which is equipped with the maximum norm and the positive cone \(C_\omega ^+:=\{v \in C_\omega : v(t) \ge 0 \) for all \(t \in \mathbb {R}\}\).
Suppose that \(v\in C_\omega \) is the initial distribution of infectious individuals. Then for any given \(s\ge 0\), \(F(t-s)v_{t-s}\) is the distribution of newly infectious individuals at time \(t-s\), which is produced by the infectious individuals who were introduced over the time interval \([t-s-{\hat{\tau }},t-s]\). Then \({\varPhi }(t,t-s)F(t-s)v_{t-s}\) is the distribution of those infectious individuals who newly became infectious at time \(t-s\) and remain in the infectious compartments at time t. It follows that
is the distribution of accumulative new infections at time t produced by all those infectious individuals introduced at all previous time to t.
Define a linear operator \(L : C_\omega \rightarrow C_\omega \) by
Following Zhao (2017), we define \(R_0=r(L)\), the spectral radius of L. Let \({\hat{P}}(t)\) be the solution maps of system (10), that is, \({\hat{P}}(t)\varphi =u_t(\varphi ), t\ge 0,\) where \(u(t,\varphi )\) is the unique solution of (10) with \(u_0=\varphi \in C\). Then \({\hat{P}}:={\hat{P}}(\omega )\) is the Poincaré map associated with linear system (10). Let \(r({\hat{P}})\) be the spectral radius of \({\hat{P}}\). By Zhao (2017, Theorem 2.1), we have the following result.
Lemma 1
\(R_0-1\) has the same sign as \(r({\hat{P}})-1\).
Let
Then we have the following preliminary result for system (8).
Lemma 2
For any \(\varphi \in W\), system (8) has a unique nonnegative bounded solution \(u(t,\varphi )\) on \([0,\infty )\) with \(u_0=\varphi \), and \(u_t(\varphi ):=(u_{1t}(\varphi ),u_{2t}(\varphi ),u_3(t,\varphi ))\in W\) for all \(t\ge 0\).
Proof
For any \(\varphi =(\varphi _1,\varphi _2,\varphi _3)\in W\), we define
Since \({\widetilde{f}}(t,\varphi )\) is continuous in \((t,\varphi )\in \mathbb {R}_+\times W\), and \({\widetilde{f}}(t,\varphi )\) is Lipschitz in \(\varphi \) on each compact subset of W, it then follows that system (8) has a unique solution \(u(t,\varphi )\) on its maximal interval \([0,\sigma _\varphi )\) of existence with \(u_0=\varphi \) (see, e.g., Hale and Verduyn Lunel (1993, Theorems 2.2.1 and 2.2.3)).
Let \(\varphi =(\varphi _1,\varphi _2,\varphi _3)\in W\) be given. If \(\varphi _i(0)=0\) for some \(i\in \{1,2\}\), then \({\widetilde{f}}_i(t,\varphi )\ge 0\). If \(\varphi _3=0\), then \({\widetilde{f}}_3(t,\varphi )\ge 0\). If \(\varphi _1(0)=H\), then \({\widetilde{f}}_1(t,\varphi )\le 0\). By Smith (1995, Theorem 5.2.1 and Remark 5.2.1), it follows that for any \(\varphi \in W\), the unique solution \(u(t,\varphi )\) of system (1) with \(u_0=\varphi \) satisfies \(u_t(\varphi )\in W\) for all \(t\in [0,\sigma _\varphi )\).
Clearly, \(0\le u_1(t,\varphi )\le H\) for all \(t\in [0,\sigma _\varphi )\). In view of the second and third equations of system (8), we have
for all \(t\in [0,\sigma _\varphi )\). Thus, both \(u_2(t)\) and \(u_3(t)\) are bounded on \([0,\sigma _\varphi )\), and hence, Hale and Verduyn Lunel (1993, Theorem 2.3.1) implies that \(\sigma _\varphi =\infty .\) \(\square \)
For any given \(\varphi \in W,\) let \(u(t,\varphi )=(u_1(t),u_2(t),u_3(t))\) be the unique solution of system (8) satisfying \(u_0=\varphi \). Let
Then \((u_1(t),u_3(t))\) can be regarded as a solution of the following nonautonomous system:
It easily follows that w(t) satisfies
and system (12) has a unique positive \(\omega \)-periodic solution
which is globally attractive in \(\mathbb {R}\). Thus, system (11) has a limiting system:
Note that \(z(t)=(u_1(t),u_3(t), w(t))\) satisfies the following \(\omega \)-periodic system:
Clearly, system (8) is equivalent to (14). It suffices to study system (14). Let
We then have the following preliminary result for system (14).
Lemma 3
For any \(\varphi \in {\varOmega }\), system (14) has a unique solution \(z(t,\varphi )\) with \(z_0=\varphi \), and \(z_t(\varphi ):=(z_{1t}(\varphi ),z_2(t,\varphi ),z_3(t,\varphi ))\in {\varOmega }\) for all \(t\ge 0\).
Proof
For any \(\varphi \in {\varOmega }\), define
Since \(\hat{f}(t,\varphi )\) is continuous in \((t,\varphi )\in \mathbb {R}\times {\varOmega }\), and \(\hat{f}(t,\varphi )\) is Lipschitz in \(\varphi \) on each compact subset of \({\varOmega }\), it then follows that system (14) has a unique solution \(z(t,\varphi )\) with \(z_0=\varphi \) on its maximal interval \([0,\sigma _\varphi )\) of existence.
Let \(\varphi =(\varphi _1,\varphi _2,\varphi _3)\in {\varOmega }\) be given. If \(\varphi _1(0)=0\), then \(\hat{f}_1(t,\varphi )\ge 0\). If \(\varphi _i=0\) for some \(i=2, 3\), then \(\hat{f}_i(t,\varphi )\ge 0\). If \(\varphi _1(0)=H\), then \(\hat{f}_1(t,\varphi )\le 0\). By Smith (1995, Theorem 5.2.1 and Remark 5.2.1), it follows that for any \(\varphi \in {\varOmega }\), the unique solution \(z(t,\varphi )\) of system (14) with \(u_0=\varphi \) satisfies \(z_t(\varphi )\in {\varOmega }\) for all \(t\in [0,\sigma _\varphi )\).
Since system (12) has a globally attractive periodic solution K(t), it follows that \(z_3(t,\varphi )=w(t)\) is bounded on \([0,\sigma _\varphi )\), that is, there exists \(B>0\) such that \(w(t)\le B\) for all \(t\in [0,\sigma _\varphi ).\) In view of the second equation of system (14), we have
Hence, \(z_2(t,\varphi )=u_3(t)\) is also bounded on \([0,\sigma _\varphi )\). Then Hale and Verduyn Lunel (1993, Theorem 2.3.1) implies that \(\sigma _\varphi =\infty .\) \(\square \)
Let
Then we have the following result for system (13).
Lemma 4
For any \(\varphi \in Y(0)\), system (13) has a unique solution \(w(t,\varphi )\) with \(w_0=\varphi \), and \(w_t(\varphi ):=(w_{1t}(\varphi ),w_2(t,\varphi ))\in Y(t)\) for all \(t\ge 0\).
Proof
For any \(\varphi \in Y(0)\), define
Since f is continuous in \((t,\varphi )\in \mathbb {R}\times Y(0)\), and f is Lipschitz in \(\varphi \) on each compact subset of Y(0), it then follows that system (13) has a unique solution \(w(t,\varphi )\) with \(w_0=\varphi \) on its maximal interval \([0,\sigma _\varphi )\) of existence.
Let \(\varphi =(\varphi _1,\varphi _2)\in Y(0)\) be given. If \(\varphi _1(0)=0\), then \(f_1(t,\varphi )\ge 0\). If \(\varphi _2=0\), then \(f_2(t,\varphi )\ge 0\). If \(\varphi _1(0)=H\), then \(f_1(t,\varphi )\le 0\). By Smith (1995, Theorem 5.2.1 and Remark 5.2.1), it follows that the unique solution \(w(t,\varphi )\) of system (13) with \(w_0=\varphi \) satisfies \(w_t(\varphi )\in C([-{\hat{\tau }},0], [0,H])\times \mathbb {R_+}\).
It remains to prove that \(w_2(t)\le K(t)\) for all \(t\in [0,\sigma _\varphi )\). Suppose this does not hold. Then there exists \(t_0\in [0,\sigma _\varphi )\) and \(\epsilon _0>0\) such that
Since
there exists \(\epsilon _1\in (0,\epsilon _0)\) such that \(w_2(t)\le K(t)\) for all \(t\in (t_0,t_0+\epsilon _1)\), which is a contradiction. This proves that \(w_t(\varphi )\in Y(t)\) for all \(t\in [0,\sigma _\varphi ).\) Clearly, \(w_t(\varphi )\) is bounded on \([0,\sigma _\varphi ),\) and hence, Hale and Verduyn Lunel (1993, Theorem 2.3.1) implies that \(\sigma _\varphi =\infty .\) \(\square \)
Let
Lemma 5
For any \(\varphi \in G(0)\), system (13) has a unique solution \(v(t,\varphi )\) with \(v_0=\varphi \), and \(v_t(\varphi ):=(v_{1t}(\varphi ),v_2(t,\varphi ))\in G(t)\) for all \(t\ge 0\).
Proof
Let \({\bar{\tau }}=\min _{t\in [0,\omega ]}\tau (t)\). For any \(t\in [0,{\bar{\tau }}]\), since \(t-\tau (t)\) is strictly increasing in t, we have
and hence,
Therefore, we have the following ordinary differential equations for \(t\in [0,\bar{\tau }]\):
Given \(\varphi \in G(0)\), the solution \((v_1(t),v_2(t))\) of the above system exists for \(t\in [0,{\bar{\tau }}]\). In other words, we have obtained values of \(\psi _1(\theta )=v_1(\theta )\) for \(\theta \in [-\tau (0),{\bar{\tau }}]\) and \(\psi _2(\theta )=v_2(\theta )\) for \(\theta \in [0,{\bar{\tau }}]\). It is easy to see that \(v_1(t)\le H\) and \(v_2(t)\le K(t)\) for all \(t\in [0,\bar{\tau }]\).
For any \(t\in [{\bar{\tau }},2\bar{\tau }]\), we have
and hence, \(v_1(t-\tau (t))=\psi _1(t-\tau (t))\). Solving the following system of ordinary differential equations for \(t\in [{\bar{\tau }},2\bar{\tau }]\) with \(v_1({\bar{\tau }})=\psi _1(\bar{\tau })\) and \(v_2(\bar{\tau })=\psi _2(\bar{\tau })\):
We then get the solution \((v_1(t),v_2(t))\) on \([\bar{\tau },2\bar{\tau }]\). We also have \(v_1(t)\le H\) and \(v_2(t)\le K(t)\) for all \(t\in [\bar{\tau },2{\bar{\tau }}]\). Repeating this procedure for \(t\in [2\bar{\tau },3\bar{\tau }]\), \([3\bar{\tau },4\bar{\tau }]\),..., it then follows that for any \(\varphi \in G(0)\), system (13) has a unique solution \(v(t,\varphi )\) with \(v_0=\varphi \), and \(v_t(\varphi ):=(v_{1t}(\varphi ),v_2(t,\varphi ))\in G(t)\) for all \(t\ge 0\). \(\square \)
Remark 1
By the uniqueness of solutions in Lemmas 4 and 5, it follows that for any \(\psi \in Y(0)\) and \(\phi \in G(0)\) with \(\psi _1(\theta )=\phi _1(\theta )\) for all \(\theta \in [-\tau (0),0]\) and \(\psi _2=\phi _2\), we have \(w(t,\psi )=v(t,\phi )\) for all \(t\ge 0\), where \(w(t,\psi )\) and \(v(t,\phi )\) are solutions of system (13) satisfying \(w_0=\psi \) and \(v_0=\phi \), respectively. Similarly, we define
and
For any \(\psi \in {\varOmega }\) and \({\phi }\in {\varPi }\) with \({\psi }_1(\theta )={\phi }_1(\theta )\) for all \(\theta \in [-\tau (0),0]\) and \({\psi }_2={\phi }_2\), \({\psi }_3={\phi }_3\), we have \(z(t,{\psi })={\tilde{z}}(t,{\phi })\) for all \(t\ge 0\), where \(z(t,{\psi })\) and \({\tilde{z}}(t,{\phi })\) are solutions of system (14) satisfying \(z_0={\psi }\) and \({\tilde{z}}_0={\phi }\), respectively. It follows that \({\varPi }\) is positively invariant for system (14). For any \(\psi \in W\) and \({\phi }\in {\varPsi }\) with \({\psi }_1(\theta )={\phi }_1(\theta ), {\psi }_2(\theta )={\phi }_2(\theta )\) for all \(\theta \in [-\tau (0),0]\) and \({\psi }_3={\phi }_3\), we have \(u(t,{\psi })={\tilde{u}}(t,{\phi })\) for all \(t\ge 0\), where \(u(t,{\psi })\) and \({\tilde{u}}(t,{\phi })\) are solutions of system (8) satisfying \(u_0={\psi }\) and \({\tilde{u}}_0={\phi }\), respectively. It then follows that \({\varPsi }\) is positively invariant for system (8).
Let S(t) be the solution maps of system (13), that is, \(S(t)\varphi =v_t(\varphi )\), \(t\ge 0\), where \(v(t,\varphi )\) is the unique solution of system (13) with \(v_0=\varphi \in G(0)\). By similar arguments to those in Lou and Zhao (2017, Lemma 3.5), we have the following result.
Lemma 6
\(S(t): G(0)\rightarrow G(t)\) is an \(\omega \)-periodic semiflow in the sense that (i) \(S(0)=I\); (ii) \(S(t+\omega )=S(t)\circ S(\omega )\) for all \(t\ge 0\); (iii) \(S(t)\varphi \) is continuous in \((t,\varphi )\in [0,\infty )\times G(0)\).
Note that the linearized system of (13) at (0, 0) is
Let P be the Poincaré map of the linear system (15) on the space \(C([-\tau (0)\), 0], \(\mathbb {R})\times \mathbb {R}\), and r(P) be its spectral radius. Then we have the following threshold type result for system (13).
Lemma 7
The following statements are valid:
-
(i)
If \(r(P)\le 1\), then \(v^*(t)=(0,0)\) is globally asymptotically stable for system (13) in G(0).
-
(ii)
If \(r(P)>1\), then system (13) admits a unique positive \(\omega \)-periodic solution \({\tilde{v}}(t)=({\tilde{v}}_1(t),{\tilde{v}}_2(t))\) which is globally asymptotically stable for system (13) in \(G(0){\setminus }\{0\}\).
Proof
It follows from Remark 1 that S(t) maps G(0) into G(t), and \(S:=S(\omega ): G(0)\rightarrow G(\omega )=G(0)\) is the Poincaré map associated with system (13). By the continuity and differentiability of solutions with respect to initial values, it follows that S is differentiable at zero and the Frechét derivative \(DS(0)=P\).
For any given \(\varphi , \psi \in G(0)\) with \(\varphi \ge \psi \), let \({\bar{v}}(t)=v(t,\varphi )\) and \(v(t)=v(t,\psi )\) be the unique solutions of system (13) with \(v_0=\varphi \) and \(v_0=\psi \), respectively. Let \({\bar{\tau }}=\min _{t\in [0,\omega ]}\tau (t).\) Define
Since
we have
and hence, \(A(t)\ge B(t)\) for all \(t\in [0,{\bar{\tau }}]\). In view of \({\bar{v}}(0)=\varphi (0)\ge \psi (0)=v(0)\), the comparison theorem for cooperative ordinary differential systems implies that \({\bar{v}}(t)\ge v(t)\) for all \(t\in [0,{\bar{\tau }}].\)
Repeating this procedure for \(t\in [\bar{\tau },2\bar{\tau }], [2\bar{\tau },3\bar{\tau }],...\), it follows that \(v(t,\varphi )\ge v(t,\psi )\) for all \(t\in [0,\infty )\). This implies that \(S(t): G(0)\rightarrow G(t)\) is monotone for each \(t\ge 0\). Next we show that the solution map S(t) is eventually strongly monotone. Let \(\varphi >\psi \) and denote \(v(t,\varphi )=({\bar{y}}_1(t), {\bar{y}}_2(t))\), \(v(t,\psi )=(y_1(t),y_2(t))\).
Claim 1
There exists \(t_0\in [0,\bar{\tau }]\) such that \({\bar{y}}_2(t)>y_2(t)\) for all \(t\ge t_0\).
We first prove that \({\bar{y}}_2(t_0)>y_2(t_0)\) for some \(t_0\in [0,{\bar{\tau }}]\). Otherwise, we have \({\bar{y}}_2(t)=y_2(t)\) for all \(t\in [0,\bar{\tau }]\), and hence, \(\frac{\mathrm{d}{\bar{y}}_2(t)}{\mathrm{d}t}=\frac{\mathrm{d}y_2(t)}{\mathrm{d}t}\) for all \(t\in (0,\bar{\tau }).\) Thus, we have
Since \(\varphi >\psi \) and \(\varphi _2={\bar{y}}_2(0)=y_2(0)=\psi _2\), we have \(\varphi _1>\psi _1\). Then there exists an open interval \((a,b)\subset [-\tau (0),0]\) such that \(\varphi _1(\theta )>\psi _1(\theta )\) for all \(\theta \in (a,b)\). Let \(h(t)=t-\tau (t)\). Since \(h'(t)>0\), the inverse function \(h^{-1}(t)\) exists. It follows from (16) that \(y_2(t)=K(t)\) for all \(t\in (h^{-1}(a), h^{-1}(b))\), and hence,
which contradicts the fact that
Let
Since
we have
Since \({\bar{y}}_2(t_0)>y_2(t_0)\), the comparison theorem for ordinary differential equations (see Walter (1997, Theorem 4)) implies that \({\bar{y}}_2(t)>y_2(t)\) for all \(t\ge t_0\).
Claim 2
\({\bar{y}}_1(t)>y_1(t)\) for all \(t>t_0\).
We first prove that for any \(\epsilon >0\), there exists an open interval \((c,d)\subset [t_0,t_0+\epsilon ]\) such that \(H>{\bar{y}}_1(t)\) for all \(t\in (c,d)\). Otherwise, there exists \(\epsilon _0>0\) such that \(H={\bar{y}}_1(t)\) for all \(t\in (t_0,t_0+\epsilon _0).\) It then follows from the first equation of system (13) that \(0=-(d_h+\rho )H\), which is a contradiction. Let
Then we have
and hence,
Since \({\bar{y}}_1(t_0)\ge y_1(t_0)\), it follows from Walter (1997, Theorem 4) that \({\bar{y}}_1(t)>y_1(t)\) for all \(t>t_0\).
In view of Claims 1 and 2, we obtain
Since \(t_0\in [0,{\bar{\tau }}]\), it follows that
that is, \(v_t(\varphi )\gg v_t(\psi )\) for all \(t>{\bar{\tau }}+\tau (0)\). This shows that \(S(t): G(0)\rightarrow G(t)\) is strongly monotone for any \(t>{\bar{\tau }}+\tau (0)\).
For any given \(\varphi \gg 0\) in G(0) and \(\lambda \in (0,1)\), let \(v(t,\varphi )\) and \(v(t,\lambda \varphi )\) be the solutions of system (13) satisfying \(v_0=\varphi \) and \(v_0=\lambda \varphi \), respectively. Denote \(x(t)=\lambda v(t,\varphi )\) and \(z(t)=v(t,\lambda \varphi )\). As in the proof of Lemma 5, by the comparison theorem for ordinary differential equations, we have \(x(t)>0\) and \(z(t)>0\) for all \(t\ge 0\). Moreover, for all \(\theta \in [-\tau (0),0]\), we have
For any \(t\in [0,\bar{\tau }]\), we have \(-\tau (0)\le t-\tau (t)\le \bar{\tau }-\bar{\tau }=0\), and hence, \(z_1(t-\tau (t))=x_1(t-\tau (t))=\lambda \varphi _1(t-\tau (t))\). Thus, x(t) satisfies the following differential inequality:
for all \(t\in [0,\bar{\tau }]\). Since \(x(0)=z(0)\), it follows from the comparison theorem for ordinary differential systems (see Walter (1997, Theorem 4)) that \(x_1(t)<z_1(t)\) and \(x_2(t)<z_2(t)\) for all \(t\in (0,\bar{\tau }]\). By similar arguments for any interval \((n\bar{\tau },(n+1)\bar{\tau }]\), \(n=1,2,3,\cdots \), we can get \(x_1(t)<z_1(t)\) and \(x_2(t)<z_2(t)\) for all \(t>0\), that is, \(v(t,\lambda \varphi )\gg \lambda v(t,\varphi )\) for all \(t>0\). Therefore, \(v_t(\lambda \varphi )\gg \lambda v_t(\varphi )\) for all \(t>\tau (0)\).
Now we fix an integer \(n_0\) such that \(n_0\omega >{\bar{\tau }}+\tau (0)\). It then follows that \(S^{n_0}=S(n_0\omega ): G(0)\rightarrow G(0)\) is strongly monotone and strictly subhomogeneous. Note that \(DS^{n_0}(0)=DS(n_0\omega )(0)=P(n_0\omega )= P^{n_0}(\omega )=P^{n_0},\) and \(r(P^{n_0})=(r(P))^{n_0}.\) By Zhao (2003, Theorem 2.3.4 and Lemma 2.2.1) as applied to \(S^{n_0}\), we have the following threshold type result:
-
(a)
If \(r(P)\le 1,\) then \(v^*(t)=(0,0)\) is globally asymptotically stable for system (13) in G(0).
-
(b)
If \(r(P)>1\), then there exists a unique positive \(n_0\omega \)-periodic solution \({\tilde{v}}(t)= ({\tilde{v}}_1(t),{\tilde{v}}_2(t))\), which is globally asymptotically stable for system (13) in \(G(0){\setminus }\{0\}.\)
It remains to prove that \({\tilde{v}}(t)\) is also an \(\omega \)-periodic solution of system (13). Let \({\tilde{v}}(t)=v(t,\psi ).\) By the properties of periodic semiflows, we have \(S^{n_0}(S(\psi ))=S(S^{n_0}(\psi ))=S(\psi )\), which implies that \(S(\psi )\) is also a positive fixed point of \(S^{n_0}\). By the uniqueness of the positive fixed point of \(S^{n_0}\), it follows that \(S(\psi )=\psi \). So \({\tilde{v}}(t)\) is an \(\omega \)-periodic solution of system (13). \(\square \)
Next, we use the theory of chain transitive sets (see Hirsch et al. (2001) and Zhao (2003, Section 1.2)) to lift the threshold type result for system (13) to system (14).
Theorem 1
The following statements are valid:
-
(i)
If \(r(P)\le 1\), then the periodic solution (0, 0, K(t)) is globally asymptotically stable for system (14) in \({\varPi }\);
-
(ii)
If \(r(P)>1\), then system (14) admits a unique positive \(\omega \)-periodic solution \(({\tilde{v}}_1(t)\), \({\tilde{v}}_2(t)\), K(t)), which is globally asymptotically stable for system (14) in \({\varPi }{\setminus } (\{0\}\times \{0\}\times \mathbb {R}_+)\).
Proof
Let \({\tilde{P}}(t)\) be the solution maps of system (14), that is, \({\tilde{P}}(t)\varphi =z_t(\varphi ), t\ge 0,\) where \(z(t,\varphi )\) is the unique solution of system (14) with \(z_0=\varphi \in {\varPi }.\) Then \({\tilde{P}}:={\tilde{P}}(\omega )\) is the Poincaré map of system (14). Then \(\{{\tilde{P}}^n\}_{n\ge 0}\) defines a discrete-time dynamical system on \({\varPi }\). For any given \({\bar{\varphi }}\in {\varPi },\) let \({\bar{z}}(t)=(u_1(t),u_3(t),w(t))\) be the unique solution of system (14) with \({\bar{z}}_0= {\bar{\varphi }}\) and let \(\omega ({\bar{\varphi }})\) be the omega limit set of the orbit \(\{{\tilde{P}}^n({\bar{\varphi }})\}_{n\ge 0}\) for the discrete-time semiflow \({\tilde{P}}^n\).
Since equation (12) has a unique positive \(\omega \)-periodic solution K(t), which is globally attractive, we have \(\lim _{t\rightarrow \infty }(w(t)-K(t))=0\), and hence, \(\lim _{n\rightarrow \infty }\) \(({\tilde{P}}^n({\bar{\varphi }}))_3\) \(=K(0)\). Thus, there exists a subset \(\tilde{\omega }\) of \(C([-\tau (0),0],[0,H])\times \mathbb {R}_+\) such that \(\omega ({\bar{\varphi }})=\tilde{\omega }\times \{K(0)\}.\)
For any \(\phi =(\phi _1,\phi _2,\phi _3)\in \omega ({\bar{\varphi }}),\) there exists a sequence \(n_k\rightarrow \infty \) such that \({\tilde{P}}^{n_k}({\bar{\varphi }})\rightarrow \phi \), as \(k\rightarrow \infty .\) Since \(u_{1n_k\omega }\le H\) and \(u_3(n_k\omega )\le w(n_k\omega ),\) letting \(n_k\rightarrow \infty ,\) we obtain \(0\le \phi _1\le H, 0\le \phi _2\le K(0).\) It then follows that \(\tilde{\omega }\subseteq C([-\tau (0),0],[0,H])\times [0,K(0)]=G(0).\) It is easy to see that
where S is the Poincaré map associated with system (13). By Zhao (2003, Lemma 1.2.1), \(\omega ({\bar{\varphi }})\) is an internally chain transitive set for \({\tilde{P}}^n\) on \({\varPi }\). It then follows that \(\tilde{\omega }\) is an internally chain transitive set for \(S^n\) on G(0).
In the case where \(r(P)\le 1\), it follows from Lemma 7 (i) that (0, 0) is globally asymptotically stable for \(S^n\) in G(0). By Zhao (2003, Theorem 1.2.1), we have \(\tilde{\omega }=\{(0,0)\}\), and hence, \(\omega ({\bar{\varphi }})=\{(0,0,K(0))\}.\) Then \({\tilde{P}}^n(\bar{\varphi })\rightarrow (0,0,K(0))\) as \(n\rightarrow \infty \). Clearly, (0, 0, K(0)) is a fixed point of \({\tilde{P}}\). This implies that statement (i) is valid.
In the case where \(r(P)>1\), by Lemma 7 (ii) and Zhao (2003, Theorem 1.2.2), it follows that either \(\tilde{\omega }=\{(0,0)\}\) or \(\tilde{\omega }=\{({\tilde{v}}_{10},{\tilde{v}}_2(0))\}\), where \({\tilde{v}}_{10}(\theta )={\tilde{v}}_1(\theta )\) for all \(\theta \in [-\tau (0),0]\). We further claim that \(\tilde{\omega }\ne \{(0,0)\}\). Suppose, by contradiction, that \(\tilde{\omega }=\{(0,0)\}\), then we have \(\omega ({\bar{\varphi }})=\{(0,0,K(0))\}\). Thus, \(\lim _{t\rightarrow \infty }(u_1(t),u_3(t))=(0,0)\), and for any \(\epsilon >0\), there exists \(T=T(\epsilon )>0\) such that \(|w(t)-K(t)|<\epsilon \) for all \(t\ge T\). Then for any \(t\ge T\), we have
Let \(r_\epsilon \) be the spectral radius of the Poincaré map associated with the following periodic linear system:
Since \(\lim _{\epsilon \rightarrow 0^+}r_\epsilon =r(P)>1\), we can fix \(\epsilon \) small enough such that \(r_\epsilon >1\). By similar result to Lemma 7 (ii), it follows that the Poincaré map of the following system
admits a globally asymptotically stable fixed point \(({\bar{u}}_{10},{\bar{u}}_3(0))\gg 0\). In the case where \({\bar{\varphi }}\in {\varPi }{\setminus } (\{0\}\times \{0\}\times \mathbb {R}_+)\), we have \((u_1(t),u_3(t))>0\) in \(\mathbb {R}^2\) for all \(t>0\). In view of (17) and (19), the comparison principle implies that
which contradicts \(\lim _{t\rightarrow \infty }(u_1(t),u_3(t))=(0,0).\) It then follows that \(\tilde{\omega }=\{({\tilde{v}}_{10}\),\({\tilde{v}}_2(0))\}\), and hence, \(\omega ({\bar{\varphi }})=\{({\tilde{v}}_{10},{\tilde{v}}_2(0),K(0))\}\). This implies that \(\lim _{t\rightarrow \infty }\) \(({\bar{z}}(t)-({\tilde{v}}_1(t)\), \({\tilde{v}}_2(t)\), \(K(t)))=(0,0,0).\) \(\square \)
By the definition of w(t), we have \(u_2(t-\tau (t))=(w(t)-u_3(t))e^{\int _{t-\tau (t)}^td_m(s)\mathrm{d}s}\). In the case where \(r(P)\le 1\), we have
It follows that \(\lim _{t\rightarrow \infty }(u_2(t)-S_m^*(t))=0.\) In the case where \(r(P)>1\), we have
where \({\hat{u}}_2(t):=e^{\int _{t-\tau (t)}^td_m(s)\mathrm{d}s}(K(t)-{\tilde{v}}_2(t))\) is a positive \(\omega \)-periodic function. Let \(x=h(t):=t-\tau (t)\). Then we have \(\lim _{x\rightarrow \infty }(u_2(x)-{\hat{u}}_2(h^{-1}(x)))=0\) and \(x+\omega =t+\omega -\tau (t)=t+\omega -\tau (t+\omega )=h(t+\omega )\). It follows that \({\hat{u}}_2(h^{-1}(x+\omega ))={\hat{u}}_2(t+\omega )={\hat{u}}_2(t)={\hat{u}}_2(h^{-1}(x))\). Replacing x by t, we have
where \({\hat{u}}_2(h^{-1}(t))\) is a positive \(\omega \)-periodic function.
As a straightforward consequence of Theorem 1, we have the following result for system (8).
Theorem 2
The following statements are valid for system (8):
-
(i)
If \(r(P)\le 1,\) then the disease-free periodic solution \((0,S_m^*(t),0)\) is globally asymptotically stable for system (8) in \({\varPsi }\);
-
(ii)
If \(r(P)>1,\) then system (8) admits a positive \(\omega \)-periodic solution \(({\tilde{v}}_1(t)\), \({\hat{u}}_2(h^{-1}(t))\), \({\tilde{v}}_2(t))\), which is globally asymptotically stable for system (8) in \({\varPsi }\setminus (\{0\}\times C([-\tau (0),0],\mathbb {R}_+)\times \{0\})\).
By the same arguments as in Lou and Zhao (2017, Lemma 3.8), we have \(r(P)=r({\hat{P}})\). Combining Lemma 1 and Theorem 2, we have the following result on the global dynamics of system (8).
Theorem 3
The following statements are valid for system (8):
-
(i)
If \(R_0\le 1\), then the disease-free periodic solution \((0,S_m^*(t),0)\) is globally asymptotically stable for system (8) in \({\varPsi }\);
-
(ii)
If \(R_0>1\), then system (8) admits a positive \(\omega \)-periodic solution \(({\tilde{v}}_1(t)\), \({\hat{u}}_2(h^{-1}(t))\), \({\tilde{v}}_2(t))\), which is globally asymptotically stable for system (8) in \({\varPsi }{\setminus }(\{0\}\times C([-\tau (0),0],\mathbb {R}_+)\times \{0\})\).
In the rest of this section, we derive the dynamics for the variable \(E_m(t)\) in system (7). It is easy to see that
In the case where \(R_0\le 1\), we have
It then follows from (20) that
In the case where \(R_0>1\), we have
By using the integral form (20), we obtain
Moreover, it is easy to verify that
is a positive \(\omega \)-periodic function. Consequently, we have the following result on the global dynamics of system (7).
Theorem 4
The following statements are valid for system (7):
-
(i)
If \(R_0\le 1\), then the disease-free periodic solution \((0,S_m^*(t),0,0)\) is globally asymptotically stable;
-
(ii)
If \(R_0>1\), then system (7) admits a unique positive \(\omega \)-periodic solution \(({\tilde{v}}_1(t)\), \({\hat{u}}_2(h^{-1}(t))\), \(E_m^*(t)\), \({\tilde{v}}_2(t))\), which is globally asymptotically stable for all nontrivial solutions.
4 A Case Study
In this section, we study the malaria transmission case in Maputo Province, Mozambique. Mozambique is a malaria-endemic country in sub-Saharan Africa. The topography and the climate of Maputo Province are favorable for malaria transmission. We will use the same values as those in Wang and Zhao (2017) for all the constant and periodic parameters except \(\tau (t)\). The constant parameter values are listed in Table 2. The values of p and l may vary from 0 to 1 and \(p\ge l\) (see Chamchod and Britton 2011; Kesavan and Reddy 1985; Lacroix et al. 2005). In the following simulations, we take \(p=0.8\) and \(l=0.6\).
The estimations for the periodic parameters \(\beta (t)\), \(d_m(t)\) and \(\mu (t)\) are given by
and
where \(k=5\times 1205709\).
According to Craig et al. (1999), the relationship between the EIP and the temperature is given by
where T is temperature in \(^{\circ }\)C, 111 is the total degree days required for parasite development, and 16 is the temperature at which the parasite development ceases. As in the case study of Wang and Zhao (2017), we take July 1 as the starting point. By using the monthly mean temperatures of Maputo Province (see Table 3, obtained from Climate Change Knowledge Portal website: http://sdwebx.worldbank.org/climateportal), we obtain the following approximation for the periodic time delay \(\tau (t)\) in CFTOOL (see Fig. 1):
To compute \(R_0\) numerically, we first write the operator L into the integral form of Posny and Wang (2014) by using the similar method to that in Lou and Zhao (2017). Since
we have
Let \(t-s-\tau (t-s)=t-s_1\). Since the function \(y=h(x)=x-\tau (x)\) is strictly increasing, the inverse function \(x=h^{-1}(y)\) exists. Solving \(t-s_1=h(t-s)\), we obtain \(s=t-h^{-1}(t-s_1), \mathrm{d}s_1=d(s+\tau (t-s))=(1-\tau '(t-s))\mathrm{d}s\quad \text {and}\quad \mathrm{d}s=\frac{1}{1-\tau '(h^{-1}(t-s_1))}\mathrm{d}s_1\). Therefore,
Define
and \(K_{12}(t,s)=e^{-\int _{t-s}^ta_{11}(r)\mathrm{d}r}a_{12}(t-s)\), \(K_{11}(t,s)=K_{22}(t,s)=0\). Then we can rewrite
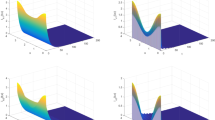
where \(G(t,s)=\sum _{j=0}^\infty K(t,j\omega +s)\). Consequently, we can use the numerical method in Posny and Wang (2014) to compute \(R_0\). We set \(\omega =12\) months. By using the obtained parameter values above, together with initial functions \(I_h(\theta )=337598\), \(S_m(\theta )=\)2,712,343, \(E_m(0)=1000\), \(I_m(0)=2000\) for all \(\theta \in [-\hat{\tau },0]\), we get \(R_0=3.1471>1\). In this case, the disease will persist and exhibit periodic fluctuation eventually (see Fig. 2). By employing some malaria control measures such as using insecticide-treated nets, spraying or clearance of mosquito breeding sites, if we can decrease the biting rate to \(0.7\beta (t)\), and increase the mosquito mortality rate to \(1.5d_m(t)\), then \(R_0=0.6591<1\). In this case, we observe that the infectious human population, the exposed and the infectious mosquito populations tend to 0, which means that the disease is eliminated from this area eventually (see Fig. 3). These numerical simulation results consist with the analytic results in the previous section.
Long-term behavior of the solution of system (7) when \(R_0=3.1471>1\)
Long-term behavior of the solution of system (7) when \(R_0=0.6591<1\)
We define the time-averaged EIP duration as
It follows that \([\tau ]=17.2500/30.4\) month. By using this time-averaged EIP duration and keeping all the other parameter values the same as those in Fig. 2, we obtain \(R_0=1.8540\), which is less than 3.1471 in Fig. 2. Figure 4 compares the long-term behaviors of the infectious compartments of model (7) under two different values of the EIP durations: the periodic \(\tau (t)\) and the constant \([\tau ]\). Figure 4a indicates that the use of the time-averaged EIP \([\tau ]\) may underestimate the number of infectious humans in Maputo. In Fig. 4b, we see that the amplitude of the periodic fluctuation of infectious mosquitoes is obviously smaller when \([\tau ]\) is used. In addition, the peak and the nadir of the periodic fluctuation of \(I_m\) are underestimated and overestimated, respectively.
Comparison of the long-term behaviors of the infectious compartments of model (7) under two different EIP durations (red curve: temperature-dependent EIP; blue curve: time-averaged EIP) (Color figure online)
5 Discussion
Malaria is strongly linked to climate conditions through the impact of climate on the vector and the parasite ecology. Of all the environmental conditions, temperature plays the most important role in malaria transmission. Both the mosquito Anopheles and the parasite Plasmodium are extremely sensitive to temperature. In particular, the duration of the EIP of Plasmodium is determined by temperature (see, e.g., Beck-Johnson et al. 2013; Ngarakana-Gwasira et al. 2014). An increasing number of malaria models have incorporated the effects of temperature on mosquito life cycle. However, none of the existing deterministic malaria models has taken into account the dependence of the EIP on temperature.
In this paper, we developed a malaria transmission model that, for the first time, incorporates a temperature-dependent EIP. The model is a system of delay differential equations with a periodic time delay. By using the theory recently developed by Zhao (2017), we derived the basic reproduction ratio \(R_0\). Incorporation of the periodic delay increases challenges for theoretical analysis. Fortunately, the work by Lou and Zhao (2017) throws light on mathematical analysis of delay differential system with periodic delays. Following the theoretical approach in Lou and Zhao (2017), we defined a phase space on which the limiting system generates an eventually strongly monotone periodic semiflow. By employing the theory of monotone and subhomogeneous systems and the theory of chain transitive sets, we established a threshold type result on the global dynamics in terms of the basic reproduction ratio \(R_0\): if \(R_0<1\), then malaria will be eliminated; if \(R_0>1\), then the disease will persist and exhibit seasonal fluctuation.
Using some published data from Maputo Province, Mozambique, and formula related to mosquito life cycle, we obtained estimations for all the constant and periodic parameters. We fitted the curve of the EIP for Maputo Province by appealing to the Detinova prediction curve. With the algorithm proposed by Posny and Wang (2014), we numerically calculated the basic reproduction ratio \(R_0\). The numerical simulation about the long-term behavior of solutions is consistent with the obtained analytic result. To compare our results with those for the constant EIP case, we also conducted numerical simulations for the long-term behavior of infectious compartments by using the time-averaged EIP. It turns out that the adoption of the time-averaged EIP may underestimate both the number of infectious humans and the basic reproduction ratio. Thus, the models incorporating the temperature-dependent EIP are more helpful for the control of the malaria transmission.
In the present work, we only considered the extrinsic incubation period (EIP) in mosquitoes. Indeed, the malaria parasites also undergo the intrinsic incubation period (IIP) in human hosts, that is, the time elapsed between exposure to malaria parasites and when symptoms and signs are first apparent. In most cases, the IIP varies from 7 to 30 days. We may consider the EIP and the IIP simultaneously when developing malaria models in future.
As proposed by Ai et al. (2012), most of the existing malaria models consider only adult mosquitoes. In fact, there are four distinct development stages during a mosquito’s lifetime: egg, larva, pupa, and adult. While only adult mosquitoes are involved in malaria transmission, the dynamics of the first three aquatic stages have great impact on the mosquito population dynamics, and hence the disease transmission dynamics (see Ai et al. 2012). It is worthwhile to develop a mosquito-stage-structured malaria model with temperature-dependent incubation period. We leave this interesting problem as a future work.
References
Ai S, Li J, Lu J (2012) Mosquito-stage-structured malaria models and their global dynamics. SIAM J Appl Math 72(4):1213–1237
Bacaër N, Ait Dads EH (2012) On the biological interpretation of a definition for the parameter \(R_0\) in periodic population models. J Math Biol 65:601–621
Bacaër N, Guernaoui S (2006) The epidemic threshold of vector-borne diseases with seasonality. J Math Biol 53:421–436
Beck-Johnson LM, Nelson WA, Paaijmans KP, Read AF, Thomas MB, Bjornstad ON (2013) The effects of temperature on Anopheles mosquito population dynamics and the potential for malaria transmission. PLoS ONE 8(11):e79276. doi:10.1371/journal.pone.0079276
Beier JC (1998) Malaria parasite development in mosquitoes. Annu Rev Entomol 43:519–543
Chamchod F, Britton NF (2011) Analysis of a vector-bias model on malaria transmission. Bull Math Biol 73:639–657
Chitnis N, Hyman JM, Cushing JM (2008) Determining important parameters in the spread of malaria through the sensitivity analysis of a mathematical model. Bull Math Biol 70:1272–1296
Craig MH, Snow RW, le Sueur D (1999) A climate-based distribution model of malaria transmission in sub-Saharan Africa. Parasitol Today 15(3):105–111
Diekmann O, Heesterbeek JAP, Metz JAJ (1990) On the definition and the computation of the basic reproduction ratio \(R_0\) in the models for infectious disease in heterogeneous populations. J Math Biol 28:365–382
Hale JK, Verduyn Lunel SM (1993) Introduction to functional differential equations. Springer, New York
Hirsch MW, Smith HL, Zhao X-Q (2001) Chain transitivity, attractivity and strong repellors for semidynamical systems. J Dyn Differ Equ 13:107–131
Inaba H (2012) On a new perspective of the basic reproduction number in heterogeneous environments. J Math Biol 22:113–128
Kesavan SK, Reddy NP (1985) On the feeding strategy and the mechanics of blood sucking in insects. J Theor Biol 113:781–783
Kingsolver JG (1987) Mosquito host choice and the epidemiology of malaria. Am Nat 130:811–827
Kot M (2001) Elements of mathematical ecology. Cambridge University Press, Cambridge
Lacroix R, Mukabana WR, Gouagna LC, Koella JC (2005) Malaria infection increases attractiveness of humans to mosquitoes. PLoS Biol 3:e298
Lou Y, Zhao X-Q (2010) A climate-based malaria transmission model with structured vector population. SIAM J Appl Math 70(6):2023–2044
Lou Y, Zhao X-Q (2017) A theoretical approach to understanding population dynamics with seasonal developmental durations. J Nonlinear Sci 27:573–603
Macdonald G (1957) The epidemiology and control of malaria. Oxford University Press, London
McCauley E, Nisbet RM, De Roos AM, Murdoch WW, Gurney WSC (1996) Structured population models of herbivorous zooplankton. Ecol Monogr 66:479–501
Molnár PK, Kutz SJ, Hoar BM, Dobson AP (2013) Metabolic approaches to understanding climate change impacts on seasonal host-macroparasite dynamics. Ecol Lett 16:9–21
Ngarakana-Gwasira ET, Bhunu CP, Mashonjowa E (2014) Assessing the impact of temperature on malaria transmission dynamics. Afr Mat 25:1095–1112
Nisbet RM, Gurney WS (1982) Modelling fluctuating populations. The Blackburn Press, Newark
Nisbet RM, Gurney WS (1983) The systematic formulation of population models for insects with dynamically varying instar duration. Theor Popul Biol 23:114–135
Omori R, Adams B (2011) Disrupting seasonality to control disease outbreaks: the case of koi herpes virus. J Theor Biol 271:159–165
Posny D, Wang J (2014) Computing the basic reproductive numbers for epidemiological models in nonhomogeneous environments. Appl Math Comput 242:473–490
Rittenhouse MA, Revie CW, Hurford A (2016) A model for sea lice (Lepeophtheirus salmonis) dynamics in a seasonally changing environment. Epidemics 16:8–16
Ross R (1911) The prevention of malaria, 2nd edn. Murray, London
Smith HL (1995) Monotone dynamical systems: an introduction to the theory of competitive and cooperative systems. American Mathematical Society, Providence
Thieme HR (2009) Spectral bound and reproduction number for infinite-dimensional population structure and time heterogeneity. SIAM J Appl Math 70:188–211
van den Driessche P, Watmough J (2002) Reproduction numbers and sub-threshold endemic equilibria for compartmental models of disease transmission. Math Biosci 180:29–48
Walter W (1997) On strongly monotone flows. Ann Polon Math LXVI:269–274
Wang W, Zhao X-Q (2008) Threshold dynamics for compartmental epidemic models in periodic environments. J Dyn Differ Equ 20:699–717
Wang X, Zhao X-Q (2017) A periodic vector-bias malaria model with incubation period. SIAM J Appl Math 77:181–201
Wang X, Zhao X-Q. A climate-based malaria model with the use of bed nets (submitted)
World Health Organisation (2015) Global Malaria Programme, World Malaria Report
Wu X, Magpantay FMG, Wu J, Zou X (2015) Stage-structured population systems with temporally periodic delay. Math Meth Appl Sci 38:3464–3481
Zhao X-Q (2003) Dynamical systems in population biology. Springer, New York
Zhao X-Q (2017) Basic reproduction ratios for periodic compartmental models with time delay. J Dyn Differ Equ 29:67–82
Acknowledgements
We are very grateful to two anonymous referees for their careful reading and helpful suggestions which led to an important improvement of our original manuscript.
Author information
Authors and Affiliations
Corresponding author
Additional information
This work is supported in part by the NSERC of Canada.
Rights and permissions
About this article
Cite this article
Wang, X., Zhao, XQ. A Malaria Transmission Model with Temperature-Dependent Incubation Period. Bull Math Biol 79, 1155–1182 (2017). https://doi.org/10.1007/s11538-017-0276-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11538-017-0276-3