Abstract
The aim of the study was to utilize the waste generated from Bullseye (Priacanthus hamrur) fish processing to recuperate proteins. Considering the cost-effectiveness, versatility, and color improvement after the pH shift method, proteins from this waste were extracted by the pH shift method using hydrochloric acid and sodium hydroxide. The properties of extracted proteins were studied in detail. During the protein solubility measurement, maximum solubilization was found at pH 3.0 (13.10 mg/mL) on the acidic side and pH 11.0 (14.25 mg/mL) on the alkaline side with a total yield of 51.62 ± 0.23 and 45.42 ± 0.29 (%), respectively. The process variables tested in this study showed a significant effect on protein solubility (p < 0.05). The protein content of the isolates extracted from the waste was 23.80 ± 0.49 and 22.48 ± 0.39% for acid and alkali processed isolates, which was significantly higher than the mince (19.46 ± 0.67%). pH shift processing of Bullseye proteins caused a significant reduction in its pigments, lipids, and myoglobin content (p < 0.05). Proteins extracted using alkali had significantly higher values for foaming stability, water holding capacity, and emulsion capacity than proteins extracted using acid. An overall assessment indicated that protein isolates obtained using alkali extraction were better in terms of textural attributes, gelling ability, and amino acid profile than protein isolates extracted using the acid process.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Globally, the fisheries and aquaculture industry generates approximately 150.5 billion USD through fish trade and significantly contributes to the world economy. Out of the total production of 177.8 million tonnes, 157.4 million tonnes were used for human consumption, whereas 20.4 million tonnes fish was destined for non-food purposes (FAO 2020). With the growing world population, interest in the animal protein source increased. Consequently overexploiting fisheries resources, including low-value and smaller size fish species, increased (Nolsøe and Undeland 2009; Kim and Venkatesan 2014).
The Bullseye fish family (Priacanthidae) comprises around 17 species distributed in tropical and subtropical marine waters. In India, the fishery has been emerging significantly as non-conventional fishery resource with the increase in commercial deep-sea trawling activity. As per the latest estimate, the landings of Priacanthids in India in 2020 was 22,107 tonnes (ICAR-CMFRI 2019). Among these, Priacanthus hamrur (Bullseye) is the most commonly exploited species. It is mainly used for preparing surimi and surimi-based products (Benjakul et al. 2002), which could produce large amounts of waste. Thus, it is crucial to design a technology that effectively recuperates functional proteins from fish wastes to fulfill the nutritional demand and reduce the ecology-related pressure on seafood processing.
The fish processing industry generates a considerable range of by-products and wastes, viz., 9–12% (heads), 12–18% (viscera), 1–3% (skin and fins), 15–20% (filleting waste), 9–15% (bones), and 5% (abdominal contents), which are almost as high as 57% (w/w) of the total captured fish weight (Rustad et al. 2011). Despite the rich nutritional profile, most of these are discarded in land-fills or into water steams without prior recovery of the nutrients (Lee et al. 2016), which causes wastage of high value nutrients, severe environmental pollution, and extra operational costs. One of the most acceptable ways to use these processing wastes is to retrieve the nutrients from the wastes to use them for human edible purposes. This would also address environmental pollution issues and reduce operational costs. However, the type of raw material, source, and process would determine the functionality and characteristics of the retrieved nutrient. Therefore, it is vital to optimize raw materials quality, procedures, and process parameters before applying them into food systems (Surasani 2018; Baraiya et al. 2023).
Over the years, numerous attempts have been made to recover nutrients from seafood processing waste, but not a single process was found efficient to address the problem related to seafood processing waste because of different technical hurdles. In order to address these issues, Hultin and Kelleher (1999) patented a technique known as isoelectric precipitation/pH-shift method/acid-alkaline solubilization, which was promising in isolating proteins from nonconventional complex marine sources. The isolate obtained by this process could be used as one of the food ingredients in producing protein based foods. Many studies have been published on recovery of protein from fish using this method with a higher recovery yields (with good functionality) than conventional surimi production process (Surasani 2018).
This process offers several benefits, including ambient storage conditions of the produced isolate powders, which could be used to develop value-added products in the food system (Shaviklo et al. 2012). Despite several studies on recuperating and characterizing the protein isolates from fishery waste, its skillful application in the food sector is minimal (Shaviklo et al. 2016; Surasani 2018). With this background, the following study was conducted to investigate the pH-based solubilization (pH 1.0 to 13.0) and recovery of Bullseye proteins from its processing waste and to characterize the obtained isolates.
Materials and methods
Raw material
Bullseye (Priacanthus hamrur) fish (average size of 15–20 cm and 200 g weight) were procured from the major fish landing center in Mangalore, India, and transported aseptically to the laboratory in an insulated ice box and stored in ice until further used. All the preparatory and processing steps, including raw material processing, were performed at a temperature < 5 °C.
Protein solubility
Solubility curve
A pH series between 3.0 and 11.0 were adjusted to the homogenate with an interval of 1.0 pH using diluted acid and alkali (2 M), followed by its protein determination using biuret method (Robinson and Hogden 1940).
Solubility after isoelectric precipitation
Protein solubility post-isoelectric precipitation was tested using two pHs having the highest solubility (3.0 and 11.0) with a precipitation pH of 5.5. Protein solubility after the second centrifugation cycle was assessed using biuret method.
Effect of process variables on protein solubility
The influence of process variables, pH (pH 1–13), extraction time (5, 30, 60, 90, and 120 min), extraction temperature (4, 15, 25, 35, and 50 °C), homogenate weight to solvent ratio (1:20, 1:15, 1:10, 1:6, and 1:3), centrifugation speed (2000, 5000, and 8000 rpm), and stirring time (continuous stirring, stirring every 30 min, stirring every 15 min, and no stirring) on protein solubility was studied as per the methodology described by Surasani et al. (2017b).
Preparation of protein isolates
Protein isolates were prepared as per the methodology given by Surasani et al. (2017a). The ground waste was added with cold deionized water at ratio of 1:9 (1:9 w/v; 4 °C), followed by high-speed homogenization at 10,000 rpm for 60 s (2 × 30 s) (Ultra-Turrax, T 25, Janke and Kunkel GMBH and Co., Staufen, Germany). The prepared homogenate was adjusted to the desired pH (pH 3.0 and pH 11.0) using diluted NaOH and HCl with simultaneous addition of diluted acid/alkali to maintain the set pH for 30 min. The temperature of the homogenate (< 5 °C) during extraction was maintained using the flake ice. The homogenate was subjected to 1st centrifugation (Thermo Sorvall Legend XTR, Thermo Fisher Scientific, USA) at 10,000 × g for 20 min, followed by the separation of supernatant using a two layered muslin cloth. The resultant supernatant was subjected to precipitation at pH 5.5 using 2 M Hcl or NaOH, followed by the 2nd centrifugation at 10,000 × g for 20 min. The resultant sediment (isolate) was packed in a zip-lock bag and kept on ice in a cold room at 4 °C overnight for further use.
Protein recovery and preparing isolates
Protein recovery (theoretical and practical) was calculated as per the methodology described by Surasani et al. (2017a).
Characterization of isolates
The compositional analysis of raw material and Bullseye protein isolate was done using standard protocols described in AOAC (2000). Color values (L*, a*, and b* values) and the overall whiteness of the samples were determined according to the methodology of Chaijan et al. (2010). Total lipid content was determined as per the standard protocol given in AOAC (2000). Total pigment content was determined as per the method described by Rawdkuen et al. (2009). The total myoglobin content of samples was determined as per the procedure used by Chaijan et al. (2006). Surface active properties such as foaming capacity (FC), foaming stability (FS) (Sathe et al. 1982), emulsification capacity (EC), and water and oil holding capacity (WHC and OHC) (Foh et al. 2011) were determined using standard methods.
Protein gel preparation was done as per the procedure given by Kristinsson and Ingadottir (2006). Gel quality analysis, i.e., folding test (Kudo et al. 1973), gel strength (Reddy 2016), and expressible moisture (Feng and Hultin 2001), was performed as per the standard protocols. Amino acid profiling was done using a standard protocol as described by Bidlingmeyer et al. (1984). The amino acid content of Bullseye protein was measured and expressed as g/100 g protein.
Statistics analysis
All experiments were performed in triplicate unless otherwise stated. Results were expressed as mean ± standard deviation. All the results were subjected to a one-way analysis of variance (ANOVA), and significant difference was measured with Tukey post hoc (p < 0.05) using IBM SPSS (Version 20.0) statistical software package program.
Results and discussion
Effect of pH on protein solubility and recovery
The solubility of proteins at different pH differed significantly (p < 0.05). The protein solubility was maximum at pH 3.0 (13.10 ± 0.11 mg/mL) and pH 11.0 (14.25 ± 0.23 mg/mL). Generally, the higher protein solubility was achieved at pH 2.0 and 13.0, but the actual protein recovery percentage was maximum at pH 3.0 and 11.0. Surasani (2018) explained that the feasibility of any protein extraction process depends on the yield and protein recovery. The studies conducted by various researchers have documented that the maximum solubility of fish protein was in the pH range of 2.0–3.0 (acidic side) and 11.0–13.0 (alkaline side), while the least solubility was at the pH of 5.0–6.0.
According to Kristinsson et al. (2005), maximum protein solubility is essential to isolate protein from contaminants, while minimum solubility is essential in precipitation to achieve higher recovery of solubilized proteins. Protein surface charges turn more positive and negative, thus increasing solubility due to increased electrostatic repulsion above and below the isoelectric pH (Kelleher and Hultin 1999).
Higher yields for Bullseye frame waste protein isolate was obtained in the acidic solubilization process (36.44 ± 0.39%) than in the alkaline solubilization process (30.22 ± 0.59%) (p < 0.05). The theoretical recovery was more than the actual recovery with all the pH ranges tested. A similar pattern was observed during recovery through the second centrifugation step, and total process yield was 51.62 ± 0.23% and 45.42 ± 0.29% for pH 3.0 and 11.0, respectively (Table 1). Several factors influence the recovery yield of protein during the pH shift method such as different raw materials and processing methods (Undeland et al. 2002; Kristinsson and Ingadottir 2006; Nolsøe et al. 2007). The higher solubility of proteins was observed on the acidic side and larger sediment formation on the alkaline side (Nolsøe and Undeland 2009), which supports the findings of the current study. Our findings are also in line with the study by Surasani et al. (2017a, b) on protein recovery from pangas and rohu processing waste.
Effect of extraction time on solubility and recovery
The effect of extraction time (5–120 min) on protein solubility and recovery (%) is given in Fig. 1B. The protein solubility and recoveries at different extraction times were significantly different (p < 0.05). Maximum protein solubility (20.13 ± 0.16 mg/mL) and protein recovery (59.44 ± 0.35%) were recorded at 60 min, and a further increase in extraction time caused a decrease in solubility. The variation in protein solubility with different extraction times could be due to irregular diffusion of non-protein substances of homogenate that was observed in the present study and was well confirmed by the findings of previous researchers (Kahn et al.1974; Montecalvo et al. 1984; Surasani et al. 2020a, b, c).
Influence of pH (1 to 13) on Bullseye protein solubility and influence of different extraction variables on solubility of proteins from Bullseye fillet frame during alkaline (pH 11) solubilization. Note: Different superscripts in capital letters (A, B, and C) indicate significant difference (p < 0.05) between different treatments. W/V, weight of the homogenate/volume of the solvent
Effect of extraction temperature on protein solubility and recovery
The extraction temperature strongly influenced the protein solubility and recovery (p < 0.05). Protein solubility and recoveries improved from 9.59 ± 0.33 to 16.70 ± 0.44 mg/mL as extraction temperature increased from 4 to 50 °C (Fig. 1C). Surasani et al. (2017a,b) reported a similar pattern during their work on proteins from processing waste of pangas and rohu. In contrast, a reverse trend was observed by Kahn et al. (1974) and Montecalvo et al. (1984) during the recovery of proteins from flounder and squid.
Effect of homogenate to solvent ratio on solubility and recovery
The effect of homogenate weight to solvent volume ratio on Bullseye frame waste protein solubility is given in Fig. 1D. Protein solubility increased with the increasing homogenate weight to solvent volume ratio, significantly (p < 0.05), i.e., 1:20 to 1:3 (10.49 ± 0.39 to 24.90 ± 0.85 mg/mL). These findings are in agreement with the observations made by earlier researchers (Montecalvo et al.1984; Surasani et al. 2018a,b), who worked on protein extraction from fish waste. They stated that high ratios resulted in high viscous solutions and low ratios resulted in dilute solutions of large volumes, which may cause trouble during handling. Based on the observations in this study, it is more advantageous to use the weight of homogenate to solvent ratio of 1:6 for good protein recovery and easy handling.
Effect of centrifugation speed on protein solubility and recovery
Centrifugation speed significantly affected protein solubility and recovery yields (Fig. 1E). With the increase in centrifugation speed from 2000 to 8000 rpm, protein solubility improved from 17.81 ± 0.68 to 21.27 ± 0.83 mg/mL, respectively. Solubility was minimum at low centrifugation speed, which could be due to insufficient centrifugal force that could not separate soluble protein from insolubilized matter, causing a heavy fluid-gel-like sediment. Kain et al. (2009) and Surasani et al. (2017a, 2020a) also reported similar observations in their study to assess the influence of process variable on protein recovery from rohu fish.
Effect of stirring on protein solubility and recovery
The protein solubility of isolates increased from 15.67 ± 0.55 to 21.59 ± 0.47 mg/mL with an increase in stirring time as shown in Fig. 1F. However, theoretical and actual recovery were significantly different in all the conditions (p < 0.05). The current finding suggests that stirring time did not show significant influence on protein solubility. Surasani et al. (2018a) also observed that stirring time did not show significant effect on protein solubility.
Characteristics of protein isolates
Proximate composition
The composition of raw material and its quality determine the percentage of protein recovery and protein characteristics. The proximate composition, i.e., moisture, protein, fat, and ash content of raw material used in the study was 73.66 ± 1.32, 19.46 ± 0.67, 1.52 ± 0.38, and 2.49 ± 0.41%, respectively (Table 2). The protein content of isolates extracted using acidic and alkaline processes was 22.48 ± 0.39 and 23.80 ± 0.49%, respectively. Surasani et al. (2017a, b) reported a protein content of 9.9 and 8.15% in rohu and pangas processing waste, while the protein isolates from pangas and rohu waste had a protein content of 17.86 and 20.45%, respectively. Baraiya et al. (2020) reported that the protein content of acid and alkali processed isolate obtained from pacu fish (freshwater species) was 21.01% and 22.87%, respectively.
Lipid reduction is another advantage of pH shift processed protein isolate, and lipid content of Bullseye filleting waste was reduced from 1.52 ± 0.38 to 0.24 ± 0.11% and 0.25 ± 0.12% in isolates processed from rohu and pangas waste. The lipid content of rohu and pangas protein isolates was 0.24 and 0.78% in comparison to its raw material with a fat content of 2.9 and 14.17%, respectively (Surasani et al. 2017a, b, 2018a). Earlier reports suggested that the lipids can be deposited in the bottom sediments during centrifugation, which can be removed as a pellet followed by a surfaced slayer as some neutral lipids are segregated into this layer (Nolsøe and Underland 2009; Vareltzis et al. 2012).
Color and whiteness
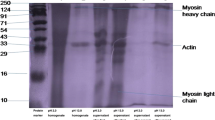
Color of food is one of the essential attributes in understanding the influence of processing effects and whiteness of fish meat or surimi is an important attribute that often determines its application in the market (Tabilo-Munizaga and Barbosa-Canovas 2004). The overall whiteness value was higher for acid-processed isolates (54.02 ± 1.02) as compared to alkali-processed isolate (48.85 ± 1.23) (Table 2). Current findings are supported by the results of Surasani et al. (2017a, b), who reported higher lightness values for acid-treated isolates (52.70) than alkaline-treated isolates (41.42) during rohu waste processing. The higher whiteness of acid-treated protein isolate might be due to efficient removal of pigments, myoglobin, hemoglobin, and melanin content (Panpipat and Chaijan 2016). The Maillard reaction could be the reason for less whiteness value in alkaline treated protein isolate, which also causes increased b* value (yellowness discoloration). These interpretations were supported by the findings reported by Kristinsson and Hultin (2003) and Kristinsson and Liang (2006) during acid/alkaline aided fish protein isolate extraction.
Lipid, pigment, and myoglobin content
The protein isolated through the acidic/alkaline method had significantly lower total lipid, myoglobin, and pigments than mince raw meat (Table 2). Among the two isolate groups, acid processed isolates had lower lipid content. Similarly, myoglobin and pigment also reduced from 285.87 ± 6.90 to 28.95 ± 1.77 mg/g and 180.97 ± 3.75 to 32.09 ± 1.23 ppm, respectively, in acid and alkali processed isolates. The reported lipid reduction was 50 to 90% in catfish, sardine, and tilapia protein isolate (Kristinsson et al. 2005; Batista et al. 2007). Myoglobin content was decreased significantly in the acid/alkaline method compared to traditional surimi processing (Rawdkuen et al. 2009). The elimination of myoglobin was observed to a greater extent in sardine and mackerel muscles during alkaline processing, and this efficiency is influenced by various factors such as fish species, muscle tissue type, the season of storage, and the washing method (Chaijan et al. 2006).
Functional properties
Functionality is a total of functional features that determine protein-based product applications. The alkali-treated isolates were superior to acid-treated isolates in terms of FS, EC, WHC, and OHC, whereas FC was higher for acid processed isolate (68.30 ± 1.14 mL/100 mL) compared to alkali processed isolate (56.50 ± 1.34 mL/100 mL). The current findings are in agreement with the statement of Surasani et al. (2017a, b), who stated that these differences might be due to the differences in hydrophobic residues of proteins. Hultin and Kelleher (1999) stated that the protein structure is sensitive to extreme pH, resulting in less water holding capacity.
Properties of protein gels
Except for the color values, gels produced from acid processed isolates had poor textural attributes, i.e., expressible moisture, folding test, and gel strength, compared to gels made of alkaline extracted isolates (p < 0.05) (Table 3). Protein isolate gels had similar properties to raw protein isolates with respect to physical attributes. Acid processed isolates had high L* values, low a* and b* values than alkali processed isolates (p < 0.05). The whiteness value of protein isolates was (57.44 ± 1.23), relatively higher for acid processed protein isolates than alkali processed protein isolates (51.26 ± 0.75). Paker et al. (2015) also made similar observations during protein extraction from silver carp using the pH shift process.
The expressible moisture and gel strength values were higher in alkaline processed isolates than the acid processed isolate. During the folding test, all the specimens received a score of 5indicating a better gel networking. Surasani (2020b) reported that the gels prepared from acid-processed protein isolates were weaker than those from alkali extracted isolates during their investigations on rohu and pangas proteins. According to Chaijan et al. (2006), gelling properties differ due to the structural differences and its influence on the process of protein bonding. The acid aided method caused extreme denaturation of protein that resulted in poor networking of protein gels. The ability of proteins to form gels depends on raw material type (fish species), method of extraction, extraction time, etc. (Kristinsson and Liang 2006; Nolsøe et al. 2007).
Amino acid profiling
Amino acid profiling of protein isolate obtained through the pH shift method indicated the recovery of all the amino acids that were present in the homogenate (Fig. 2). The study revealed that the acidic/alkaline solubilization method did not show significant influence on amino acid recovery. It was observed that alkali aided processes recovered larger amount of amino acids from homogenate, which might be due to the connective tissues that were in the more extractable form during alkaline processing (Batista 1999). The amino acid composition of the isolates in the present study is in the range reported by the previous researchers (Dileep et al. 2016; Binsi et al. 2009; Surasani et al. 2017a, b). However, overall composition of amino acids and quality of protein is influenced by various factors such as fish species, processing method employed, and the solubilization pH used. The isolates obtained through acid/alkaline solubilization method contained amino acid concentrations above the suggested range for an adult consumption by FAO/WHO/UNO.
Amino acid profile of raw material and proteins recovered from Bullseye fillet frames during solubilization at pH 3.0 and 11.0. *Essential amino acid. ASP, aspartic acid; THR, threonine; SER, serine; GLU, glutamic acid; PRO, proline; GLY, glycine; ALA, alanine; CYS, cysteine; VAL, valine; MET, methionine; I LEU, isoleucine; LEU, leucine; TYR, tyrosine; PHE, phenylalanine; HIS, histidine; LYS, lysine; ARG, arginine; TRP, tryptophan
Conclusion
The protein isolates extracted from Bullseye filleting waste at pH 3.0 and pH 11.0 showed good recovery yield and recovery. It was found that the alkaline-aided processing is a suitable technique for protein recovery from marine fish processing waste with good functional properties. The pH shift method could significantly reduce lipid, pigment, and myoglobin content of isolates, resulting in whiter isolates with better stability. This technology could be well accepted for recovering proteins from marine fish processing waste. However, newer definite investigations should be encouraged to better understand protein isolate’s process variable and efficiency.
Data availability
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
References
AOAC (2000) Association of official and analytical chemists. 16th edn. Washington, DC
Baraiya KG, Raju CV, Surasani VKR, Lakshmisha IP, Kumar PA (2020) Optimization of the process for recuperating proteins from pacu processing waste: quantification and characterization of isolates. J Entomo Zoo Studi 4:710–719
Baraiya KG, Bojayanaik M, Surasani VKR et al (2023) Utilization of fish protein isolates to supplement oat-based cookies and assessment of end product quality. Environ Sci Pollut Res. https://doi.org/10.1007/s11356-023-27804-6
Batista I (1999) Recovery of proteins from fish waste products by alkaline extraction. Eur Food Res Technol 210:84–89. https://doi.org/10.1007/s002170050539
Batista I, Pires C, Nelhas R (2007) Extraction of sardine proteins by acidic and alkaline solubilisation. Food Sci Technol Int 13(3):189–194. https://doi.org/10.1177/1082013207079619
Benjakul S, Visessanguan W, Riebroy S, Ishizaki S, Tanaka M (2002) Gel-forming properties of surimi produced from bigeye snapper, Priacanthus tayenus and Priacanthus macracanthus, stored in ice. J Sci Food Agric 82:1442–1451. https://doi.org/10.1002/jsfa.1207
Bidlingmeyer BA, Cohen SA, Tarvin TL (1984) Rapid analysis of amino acids using precolumn derivatisation. J Chromatogr 336:93–104. https://doi.org/10.1016/S0378-4347(00)85133-6
Binsi PK, Shamasundara BA, Dileep AO, Badiib F, Howell NK (2009) Rheological and functional properties of gelatin from the skin of bigeye snapper (Priacanthus hamrur) fish: influence of gelatin on the gel-forming ability of fish mince. Food Hydrocoll 23:132–145. https://doi.org/10.1016/j.foodhyd.2007.12.004
Chaijan M, Benjakul S, Visessanguan W, Faustman C (2006) Physicochemical properties, gel-forming ability and myoglobin content of sardine (Sardinella gibbosa) and mackerel (Rastrelliger kanagurta) surimi produced by conventional method and alkaline solubilisation process. Eur Food Res Technol 222:58–63. https://doi.org/10.1007/s00217-005-0091-1
Chaijan M, Panpipat W, Benjakul S (2010) Physicochemical and gelling properties of short-bodied mackerel (Rastrelliger brachysoma) protein isolate prepared using alkaline-aided process. Food Bioprod Process 88:174–180. https://doi.org/10.1016/j.fbp.2009.11.003
Dileep AO, Shamasundar BA, Binsi PK, Badii F, Howell NK (2016) Composition, physicochemical and rheological properties of fresh bigeye snapper fish (Priacanthus hamrur) mince. J Food Biochem 36(5):577–586. https://doi.org/10.1111/j.1745-4514.2011.00592.x
FAO (2020) The state of world fisheries and aquaculture. Food and Agriculture Organization, Rome, p 2020
Feng YM, Hultin HO (2001) Effect of pH on the rheological and structural properties of gels of water-washed chicken-breast muscle at physiological ionic strength. J Agric Food Chem 49:3927–3935. https://doi.org/10.1021/jf001021f
Foh MBK, Wenshiu X, Amadou I, Jiang Q (2011) Influence of pH shift on functional properties of protein isolated of tilapia (Oreochromis niloticus) muscles and of soy protein isolate. Food Bioproc Tech 5(6):1–9. https://doi.org/10.1007/s11947-010-0496-0
Hultin HO, Kelleher SD (1999) U.S. patent no. 6,005,073. U.S. Patent and Trademark Office, Washington, DC
ICAR-CMFRI (2019) Annual report 2018–19. Indian Council of Agricultural Research, Central Marine Fisheries Research Institute, Chochi, India
Kahn L, Berk Z, Pariser ER, Goldblith SA, Flink IM (1974) Squid protein isolate: effect of processing conditions on recovery yields. J Food Sci 39:592–595. https://doi.org/10.1111/j.1365-2621.1974.tb02956.x
Kain RJ, Chen Z, Sonda TS, Abu-kpawoh JC (2009) Study on the effect of control variables on the extraction of peanut protein isolates from peanut meal (Arachis hypogaea L.). Am J Food Technol 4(1):47–55. https://www.cabdirect.org/cabdirect/abstract/20093022650. Accessed 14 Aug 2023
Kelleher SD, Hultin HO (1999) Functional chicken protein isolates prepared using low ionic strength, acid solubilization/precipitation. In Proc. 53rd Annual Reciprocal Meat Conference, Am. Meat Sci. Assoc., Columbus, Ohio, pp 76–81
Kim SK, Venkatesan J (2014) Introduction to seafood processing by-products. In: Kim SK (ed) Seafood processing by-products. Springer, New York. https://doi.org/10.1007/978-1-4614-9590-1_1
Kristinsson HG, Hultin HO (2003) Changes in conformation and subunit assembly of cod myosin at low and high pH and after subsequent refolding. J Agric Food Chem 51:7187–7196. https://doi.org/10.1021/jf026193m
Kristinsson H, Ingadottir B (2006) Recovery and properties of muscle proteins extracted from tilapia (Oreochromis niloticus) light muscle by pH shift processing. J Food Sci 1(3):E132–E141. https://doi.org/10.1111/j.1365-2621.2006.tb15626.x
Kristinsson HG, Liang Y (2006) Effect of pH-shift processing and surimi processing on Atlantic croaker (Micropogonias undulates). J Food Sci 71(5):304–312. https://doi.org/10.1111/j.1750-3841.2006.00046.x
Kristinsson H, Theodore AE, Demir N, Ingadottir B (2005) A comparative study between acid- and alkali-aided processing and surimi processing for the recovery of proteins from channel catfish muscle. J Food Sci 70(4):C298–C306. https://doi.org/10.1111/j.1365-2621.2005.tb07177.x
Kudo G, Okada M, Miyauchi D (1973) Gel-forming capacity of washed and unwashed flesh of some Pacific coast species of fish. Mar Fish Rev 32:10–15
Lee HJ, Lee GW, Yoon IS, Park SH, Park SY, Kim JS, Heu MS (2016) Preparation and characterization of protein isolate from yellowfin tuna Thunnus albacares roe by isoelectric solubilization/precipitation process. Fish Aquatic Sci 19:4. https://doi.org/10.1186/s41240-016-0014-z
Montecalvo J, Constantinides SM, Yang CST (1984) Optimization of processing parameters for the preparation of flounder frame protein product. J Food Sci 49:172–176. https://doi.org/10.1111/j.1365-2621.1984.tb13699.x
Nolsøe H, Undeland I (2009) The acid and alkaline solubilization process for the isolation of muscle proteins: state of art. Food Bioproc Tech 2:1–27. https://doi.org/10.1007/s11947-008-0088-4
Nolsøe H, Imer S, Hultin HO (2007) Study of phase separation by filtration instead of centrifugation affects protein yield and gel quality during an alkaline solubilization process- different surimi processing methods. Int J Food Sci 42:139–147. https://doi.org/10.1111/j.1365-2621.2006.01162.x
Paker I, Beamer S, Jaczynski J, Matak KE (2015) pH shift protein recovery with organic acids on texture and color of cooked gels. J Sci Food Agric 95(2):275–280. https://doi.org/10.1002/jsfa.6712
Panpipat W, Chaijan M (2016) Biochemical and physicochemical characteristics of protein isolates from bigeye snapper (Priacanthus tayenus) head by-product using pH shift method. Turkish J Fish Aquat Sci 16:41–50
Rawdkuen S, Sai-ut S, Khamsorn S, Chaijan M, Benjakul S (2009) Biochemical and gelling properties of tilapia surimi and protein recovered using an acid-alkaline process. Food Chem 112:112–119. https://doi.org/10.1016/j.foodchem.2008.05.047
Reddy SVK (2016) Effect of formulation and processing methods on the quality and acceptability of cutlets made from minced meat of pangas (Pangasius pangasius). SAARC J Agric 14(1):25–36. https://doi.org/10.3329/sja.v14i1.29573
Robinson HW, Hogden CG (1940) The biuret reaction in the determination of serum proteins; a study of the conditions necessary for the production of a stable color which bears a quantitative relationship to the protein concentration. J Bio Chem 135:707–725
Rustad T, Storro I, Slizyte R (2011) Possibilities for the utilization of marine by-products. Int J Food Sci Technol 46:2001–2014. https://doi.org/10.1111/j.1365-2621.2011.02736.x
Sathe SK, Deshpande SS, Salunkhe DK (1982) Functional properties of lupin seed (Supinus mutabilis) proteins and protein concentrates. J Food Sci 7:191–197. https://doi.org/10.1111/j.1365-2621.1982.tb10110.x
Shaviklo GR, Thorkelsson G, Arason S, Sveinsdottir K (2012) Characteristics of freeze-dried fish protein isolated from saithe (Pollachius virens). J Food Sci Technol 49(3):309–318. https://doi.org/10.1007/s13197-011-0285-4
Shaviklo AR, Moradinezhad N, Abolghasemi SJ, Motamedzadega A, Kamali- damavandi N, Rafipour F (2016) Product optimization of fish burger containing tuna protein isolates for better sensory quality during frozen storage stability. Turkish J Fish Aquat Sci 16:923–933
Surasani (2018) Acid and alkaline solubilization (pH shift) process: a better approach for the utilization of fish processing waste and by-products: review. Environ Sci Pollut Res 25(19):18345–18363. https://doi.org/10.1007/s11356-018-2319-1
Surasani VKR, Tyagi A, Kudre T (2017a) Recovery of proteins from rohu processing waste using pH shift method: characterization of isolates. J Aquat Food Prod Technol 26(3):356–365. https://doi.org/10.1080/10498850.2016.1186130
Surasani VKR, Khatkar SK, Singh S (2017b) Effect of process variables on solubility and recovery yields of proteins from pangas (Pangasius pangasius) frames obtained by alkaline solubilization method: characteristics of isolates. Food Bioprod Process 106:137–146. https://doi.org/10.1016/j.fbp.2017.09.008
Surasani VKR, Kudre T, Ballari RV (2018a) Recovery and characterization of proteins from pangs (Pangasius pangasius) processing waste obtained through pH shift processing. Environ Sci Pollut Res 25:11987–11998. https://doi.org/10.1007/s11356-018-1456-x
Surasani VKR, Mandal A, Pandey A (2018b) Utilization of freshwater mussel (Lamellidens marginalis) for the isolation of proteins through pH shift processing: characterization of isolates. Environ Sci Pollut Res 25(31):31497–31507. https://doi.org/10.1007/s11356-018-3154-0
Surasani VKR, Raju CV, Chandra MV, Uzair S, Lakshmisha IP (2020a) Effect of organic acid on recovery yields and characteristics of rohu (Labeo rohita) protein isolates extracted using pH shift processing. J Sci Food Agric 99(14):6546–6655. https://doi.org/10.1002/jsfa.9935
Surasani VKR, Raju CV, Uzair S, Chandra MV, Lakshmisha IP (2020b) Influence of protein isolates from pangas processing waste on physico-chemical, textural, rheological and sensory quality characteristics of fish sausages. LWT - Food Sci Tech 111:443–448. https://doi.org/10.1016/j.lwt.2019.108662
Surasani VKR, Raju CV, Shafiq U, Baraiya KG (2020c) Influence of frozen storage on properties of rohu and pangas protein isolates obtained from its processing waste. J Aquat Food Prod Technol 29(9):935–948. https://doi.org/10.1080/10498850.2020.1818908
Tabilo-munizaga G, Barbosa-canovas GV (2004) Color and textural parameters of pressurized and heat-treated surimi gels as affected by potato starch and egg white. Food Res Int 37:767–775. https://doi.org/10.1016/j.foodres.2004.04.001
Undeland I, Kelleher SD, Hultin HO (2002) Recovery of functional proteins from herring (Clupea harengus) light muscle by an acid or alkaline solubilization process. J Agric Food Chem 50(25):7371–7379. https://doi.org/10.1021/jf020199u
Vareltzis PK, Evaggelia P, Ntoumas D, Adamopoulos KG (2012) Process characteristics and functionality of sardine (Sardina pilchardus) muscle proteins extracted by a pH-shift method. Ann Food Sci Technol 13(2):132–143
Acknowledgements
The authors wish to express their sincere thanks to the Head, Department of Fish Processing Technology, College of Fisheries, KVAFSU, Mangalore, for their constant support during the work period. The author would also like to express their appreciation to Dr. D.B. Patil, the Director of Research and Dean of PG Studies at Kamdhenu University, Gandhinagar, for offering technical assistance to their institution.
Funding
Funding for the doctoral research was received from the University Grants Commission (UGC) in the form of a fellowship under the NFOBC (National Fellowship) scheme.
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. Material preparation, data collection, and analysis were performed by Baraiya Kirankumar Gopalbhai, Manjanaik Bojayanaik, Pravinkumar Vaghabhai Taral, Vijay Kumar Reddy Surasani, Elavarasan Krishnamoorthy, and Veena Shetty. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Competing interests
The authors declare no competing interests.
Additional information
Responsible Editor: Ta Yeong Wu
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Baraiya, K.G., Bojayanaik, M., Taral, P.V. et al. Utilizing Bullseye fish processing frame waste to produce edible proteins and quality assessment of the recovered proteins. Environ Sci Pollut Res (2023). https://doi.org/10.1007/s11356-023-29738-5
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11356-023-29738-5