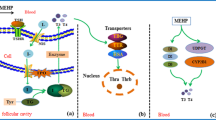
Abstract
Phthalates and bisphenols are two typical classes of endocrine-disrupting chemicals (EDCs) which cause endocrine disorder in humans and animals. Phthalates and bisphenols are suggested to be associated with thyroid dysfunction. However, the effects of combined exposure and the detailed mechanisms are yet poorly understood. We investigated the combined effects of di (2-ethylhexyl) phthalate (DEHP) and bisphenol A (BPA) on thyroid function during puberty. Female Sprague Dawley rats were gavaged from postnatal 28 to 70 days with a single or combined exposure of DEHP (0, 150, and 750 mg/kg/day) and BPA (0, 20, and 100 mg/kg/day) according to a 3 × 3 factorial design. The thyroid weights reduced after combined exposure to the highest dose of DEHP and BPA, which noted their adverse effects on thyroid. Additionally, DEHP could increase the number of follicular epithelial cells in thyroid. Both DEHP and in combination with BPA could disturb the levels of thyroid hormones in serum, such as TT3 and TT4. Meanwhile, the possible mechanism was also discussed in the present study. DEHP treatment induced a significant increase of phosphorylation of cAMP-response element binding protein (Creb) via estrogen receptor α (Esr1), while the upregulation was nullified by the concomitant presence of BPA. In conclusion, the complex action of DEHP/BPA mixture may disturb the thyroid hormone homeostasis, which ultimately would affect the development of thyroid during puberty.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Endocrine-disrupting chemicals (EDCs) widely exist in the environment, which have drawn much attention due to their adverse effects on multiple systems. Essentially, these EDCs can disrupt hormone balance by mimicking or blocking hormones (Gore et al. 2015), which would ultimately damage physiology function, such as endocrine and reproductive systems (Riaz et al. 2016) (Ma et al. 2017).
As typical EDCs, di-2-ethylhexyl phthalate (DEHP) and bisphenol A (BPA) are utilized in a range of consumer products, including personal care products, plastics, and food packaging, leading to widespread human exposure (Guo and Kannan 2012) (Kang et al. 2006) (Welshons et al. 2006). Meanwhile, their adverse effects are also widespreadly concerned (Hogberg et al. 2008) (Guidry et al. 2015). Previous studies demonstrated that phthalate exposure was associated with an increased risk of breast cancer (Ahern et al. 2019) (Lopez-Carrillo et al. 2010). Additionally, BPA exposure has adverse effects on the endocrine and reproductive systems (Markey et al. 2001) (Bilgi et al. 2019) (Li et al. 2019) and its prepubertal exposure may also elevate the susceptibility to mammary carcinogens (Leung et al. 2017). Recently, epidemiological evidence suggested that both DEHP and BPA might contribute to increase the risks of thyroid diseases, such as thyroid cancer and benign nodule (Liu et al. 2020) (Marotta et al. 2019) (Xie et al. 2017).
Thyroid hormones (THs) are essential endocrine messengers for the growth and development of virtually all vertebrates (Mendoza and Hollenberg 2017). The synthesis and secretion of THs are modulated by the hypothalamic-pituitary-thyroid (HPT) axis. As known thyronine (T4), triiodothyronine (T3), or thyroid stimulating hormone (TSH) may regulate the function of thyroid (Mondal et al. 2016). Accumulating evidences have shown the associations between phthalates exposure and altered TH levels (Johns et al. 2016) (Chuang et al. 2017) (Morgenstern et al. 2017) and thyroid functions (Meeker et al. 2007) (Miller et al. 2009) (Pan et al. 2017). Several studies have demonstrated that DEHP was negatively linked to the levels of total T3 (TT3), total T4 (TT4), and TSH in adults and pregnant women (Park et al. 2017) (Gao et al. 2017). Another study found that DEHP was negatively associated with the levels of free T3 (FT3) and free T4 (FT4) in children (Weng et al. 2017). Furthermore, an investigation reported that TSH was inversely correlated to the urinary BPA level across pregnancy (Aung et al. 2017). Other data indicated that BPA was positively associated with FT4 (Aker et al. 2016). While several studies demonstrated the adverse effect of DEHP or BPA single exposure on thyroid, few direct evidence of their combined effects on thyroid hormone homeostasis has been emphasized.
To date, humans are concurrently exposed to a large number of chemicals through various routes (Suk et al. 2002). Previous studies have shown that BPA and DEHP in utero exposure exerted adverse effects on fetal male reproductive development and cord blood estradiol levels (Sunman et al. 2019), as well as phthalates and BPA exposure during in utero windows of susceptibility in relation to the testosterone concentrations and breast development in offspring girls (Watkins et al. 2017). Additionally, most data support the effects of bisphenol A and some phthalates on the development of obesity and type 2 diabetes mellitus (Stojanoska et al. 2017). Comparing with a single exposure, combined exposure to several chemicals may induce some entirely different effects. There is a concern that different EDCs act in synergy and may result in so-called cocktail effects. Anne Katchy et al. found that co-exposure to BPA and soy-based phytoestrogens could lead to additive estrogenic effects (Katchy et al. 2014). However, another study revealed that nonyl phenol (NP) and dibutyl phthalate (DBP) could disrupt the function of Sertoli cells and hormone levels in serum, while their mixture effects were mainly antagonistic (Hu et al. 2014). In concordance, other authors found that antagonism on the expression levels of genes was involved in pituitary-gonadal cross-talk after exposure to DEHP and polychlorinated biphenyls (PCBs) (Fiandanese et al. 2016). A recent study has also indicated antagonistic interactions of neurotoxicity after a combined exposure to lead (Pb) and DEHP (Li et al. 2018). Additionally, an in vitro experiment described that the combined effect of BPA and DBP might be antagonistic in the modification of TNF-α expression (Couleau et al. 2015). Cassandra D. Kinch revealed that exposure to environmental concentrations of the contaminants BPA, DEHP, nonylphenol, and fucosterol can lead to morphological defects of zebrafish embryos, which was distinct from individual contaminants, in a manner that cannot be explained by simple additive effects (Kinch et al. 2016). Besides, a mixture of phthalates and BPA presented in human amniotic fluid could disturb the human G protein-coupled receptor (RXFP2) function, and produce potential antagonistic effects that are not displayed by the compounds, individually (Suteau et al. 2020). Although DEHP could also prompt the upregulation of thyroid transcription factor-1 (TTf-1), TSHr, and the expression of NIS in thyroid tissue (Dong et al. 2017), as well as neonatal exposure to BPA disturbed the function of HPT axis in adult rats in estrus (Fernandez et al. 2018), through binding to TH receptors in a non-competitive pattern (Jung et al. 2007), no study to date has understood the deleterious impact of cocktails of DEHP and BPA in vivo on the thyroid. Accordingly, we supported the hypothesis that co-exposure to DEHP and BPA may exert an effect that differs from the ones of each disruptor alone on the thyroid, which is considered as one of the most important endocrine organs.
Collectively, in order to explore the combined effects of DEHP and BPA on thyroid hormone homeostasis in vivo, a 3 × 3 factorial design was used in the present study and several parameters such as thyroid weight, histological changes, and serum hormonal levels were investigated after exposure to DEHP and BPA in adolescent female rats. In addition, estrogen receptor pathway and other related indexes involved in the possible mechanism were also detected and analyzed. Hopefully, our current study may contribute to provide some scientific clues for evaluating the combined effects of DEHP and BPA on thyroid function, and highlight the necessity of eliminating the substance from plastic products.
Materials and methods
Animals
Sixty-three female Sprague Dawley rats (SPF grade) (three-week-old) were obtained from the Center for Experimental Animals at China Medical University (Shenyang, China) with a National Animal Use License number of SYXK-LN 2013-0001. All experiments and surgical procedures were approved by the Animal Use and Care Committee of China Medical University, which complies with the National Institutes of Health Guide for the Care and Use of Laboratory Animals. All efforts were made to minimize the number of animals and their suffering. Rats were housed at a temperature of 24 ± 1 C with 12-h light/12-h dark cycles and humidity 50–60%. Solid-bottomed polycarbonate cages with stainless steel wire-bar lids were used to house 2 rats per cage containing corn-cob bedding and nesting material. Food (Changsheng biotechnology, Shenyang, China) and tap water were provided ad libitum. Animals were housed for 1 week before being entered into the study.
Design and treatments
DEHP, BPA, and corn oil were purchased from Sigma-Aldrich (St. Louis, MO, USA). The study was conducted in 9 groups of 7 rats according to a 3 × 3 factorial design, and the detailed grouping and administration are shown in Table 1. The female rats were gavaged with vehicle (corn oil) or DEHP (0, 150, 750 mg/kg/day) and BPA (0, 20, 100 mg/kg/day) which was dissolved in corn oil until adulthood (endpoint: postnatal day 70) (Boughammoura et al. 2020) by a well-trained technician. The DEHP dose 1 (150 mg/kg/day) was equal to 1/200 of the half lethal dose of DEHP for rat, and DEHP 150 mg/kg/day also based on no-observed-adverse-effect-level (NOAEL) of 30 mg/kg/day in rats (David et al. 2000). Further, the DEHP dose 2 (750 mg/kg/day) was equal to 1/40 of the half lethal dose of DEHP for rat, and the DEHP 750 mg/kg/day was also known to be able to induce adverse impact in rats without causing systemic toxicity (Shelby 2006). Similarly, BPA dose 1 (20 mg/kg/day) was equal to 1/200 of the half lethal dose of BPA for rat, and also based on NOAEL of 5 mg/kg/day in rats while dose 2 (100 mg/kg/day) was equal to 1/40 of the half lethal dose of BPA for rat (US Environmental Protection Agency 2011). The intragastric administration was performed at a fixed time (8:30–9:30 a.m.) every day. All rats’ body weights were measured and recorded at the end of each week.
Sample collection and preparation
All animals were sacrificed under chloral hydrate anesthesia within 24 h after the last treatment. The blood samples were obtained from the aorta abdominal in each group after animals were anesthetized and then were allowed to clot. Serum was obtained by centrifugation at 3000 rpm for 15 min and then stored at − 80 C before hormonal detection. The thyroid samples were rapidly removed, weighted, and snap-frozen in liquid nitrogen or fixed in paraformaldehyde for later analysis.
HE staining and histological evaluation
Thyroids were fixed in paraformaldehyde, processed and trimmed, embedded in paraffin, sectioned to a thickness of 4 μm, and stained with Hematoxylin and Eosin Staining Kit (Beyotime, Shanghai, China) for microscopic examination.
Enzyme-linked immunoabsorbent assay
Total T4 (TT4), total T3 (TT3), free T4 (FT4), free T3 (FT3), and TSH levels in serum of rats were measured using the rat’s THs ELISA kits (Jiangsu Meilian Bioengineering Institute, China) according to the manufacturer’s instructions. All samples were run in duplicate.
Quantitative real-time PCR
Total RNA was extracted from the thyroid using a TRIzol® Reagent (Invitrogen Inc., Burlington, ON, Canada), followed by being reverse transcribed to cDNA with a HiScript II Q Select RT SuperMix for qPCR Kit (Vazyme Biotech, Nanjing, China) according to the manufacturer’s instructions. Primers specific for genes were designed and synthesized by Sangon Biotech (Shanghai, China). The primers are shown in Table 2. Real-time PCR was carried out using ChamQ Universal SYBR qPCR Master Mix (Vazyme Biotech, Nanjing, China) on a Light Cycler 480 II (Roche, Germany) PCR detection system according to the protocol provided by the manufacturer. Glyceraldehyde 3-phosphate dehydrogenase (Gapdh) was used as an internal reference due to previous successful application. The relative quantification of mRNA levels was performed using the comparative Ct method and formula 2−ΔΔCt (Bustin et al. 2009).
Immunohistochemistry
Sections of the thyroid gland were deparaffinized, rehydrated, and subjected to antigen retrieval. Antigen retrieval was performed in Tris-EDTA buffer by microwave for 15 min, slowly cooled down to room temperature. Endogenous peroxidase activity was blocked with hydrogen peroxide, and then the sections were incubated at 4 C overnight with the primary antibodies: rabbit antibody anti-Esr1 (1:200) and anti-phospho-Creb (1:200) (Affinity Biosciences, Cincinnati, OH, USA). After incubation with the primary antibodies, the sections were incubated with appropriate biotinylated immunoglobulin and avidin-biotin peroxidase complex. Using DAB (Maxim Biotechnologies, Fuzhou, China) complexes visualized the reaction product. The average optical density of immunohistochemistry in each sample was analyzed using Image-Pro Plus software. Each stained section was evaluated by a minimum of 5 randomly selected × 20 high-power fields for further statistical analysis.
Bioinformatics analysis
STITCH and chEMBL databases were used to identify the potential targets for the toxicity of DEHP and BPA, and Cytoscape software was used to construct an interaction network related to the molecular targets of DEHP and BPA. Gene oncology (GO) enrichment analysis and Kyoto Encyclopedia of Genes and Genomes (KEGG) signaling pathway enrichment analysis were performed on potential targets of DEHP and BPA using the Database for Annotation, Visualization, and Integrated Discovery (DAVID).
Statistical analysis
All experiments were performed at least in triplicate, and data were presented as the mean ± standard deviation (SD). Statistical analyses of the mean were performed by one-way ANOVA and least-significant difference (LSD) using the SPSS software, version 21.0 (SPSS, Inc., Chicago, IL, USA). Data transformation or nonparametric test was performed when the data cannot meet the homogeneity of the variance. Statistical significance was defined as P < 0.05.
Results
Effects of 6-week exposure to DEHP and BPA on body weight and thyroid coefficient
To evaluate the effect of DEHP and BPA on the growth and development of rats, hair growth, mental state, and body weight were observed. We found the rats in all groups survived well and no apparent differences in living status and hair growth were observed. Although an upward trend in body weight was displayed in each rat, no significant changes occurred among all groups. Notably, after 3 weeks, DEHP-exposed groups showed an increasing tendency in body weight compared with the control. On the contrary, BPA-exposed groups showed an decreasing tendency in body weight than the control (Fig. 1a). In addition, we measured the thyroid weight of each group in the end of the experiment and found that the coefficient of thyroid organ in the DEHP 750 + BPA 100 group was significantly lower than that of the control (P < 0.05), and also lower than that of the DEHP 750 group (P < 0.05) (Fig. 1b).
Histological changes in thyroid due to exposure to DEHP and BPA
HE staining was conducted to assess histological changes in the thyroid. The basic structure and function unit of thyroid tissue in rats is thyroid follicles, which present slightly spherical. Each follicle is composed of simple epithelial cells, termed follicular cells. A small number of larger C cells are located beside the follicle. The results showed that the numbers of follicular epithelial cells in the thyroid tissues of rats exposed to DEHP 750 (P < 0.05) or BPA 100 were higher than that of the control (Fig. 2a, b). Additionally, the thyroid follicular diameters of rats in DEHP 750, BPA 100, and DEHP 750 + BPA 100 groups show a lower tendency than those in the control, but no statistical difference was found (Fig. 2a, c).
Effects of DEHP and BPA on the histology of thyroids (n = 7). a Hematoxylin-eosin staining (H&E staining) of the thyroid gland (original magnification, × 40). Scale bar = 50 μm. b Quantification of thyrocytes staining in each group. c Quantification of thyroid follicular cavity diameter in each group. An asterisk indicates significantly different at P < 0.05
Exposure to DEHP and BPA altered THs and TSH levels in serum
ELISA data showed that TT3 level in serum of rats exposed to DEHP 150 was slightly increased, while a significant reduction was found when combined with BPA treatment (P < 0.05), and that of rats exposed to DEHP 750 + BPA 20 was significantly lower than that of rats exposed to DEHP 750 (P < 0.05) (Fig. 3a). Compared with the control, the serum TT4 level of rats in the DEHP 750, BPA 20, BPA 100, and the combined exposure groups was significantly reduced (P < 0.05). Notably the combined exposure to DEHP and BPA further reduced the TT4 level of rats comparing with the single exposure to DEHP (P < 0.05) (Fig. 3b). FT3, FT4, and TSH levels in serum of rats in each group showed no significant changes compared with those in the control group (Fig. 3c–e).
Potential targets and pathway enrichment analysis of DEHP and BPA
STITCH and chEMBL databases were used to identify the potential targets for the toxicity of DEHP and BPA. We obtained 46 molecular targets of DEHP and 52 molecular targets of BPA. An interaction network containing 89 nodes was established using Cytoscape software (Fig. 4a). The Venn diagram showed that 14 targets are common to both compounds (Fig. 4b), which is considered as one of the important target sets of DEHP and BPA. Remarkably, the result suggested that Esr1 may act as a mediator in the interaction between DEHP and BPA. The Database for Annotation, Visualization, and Integrated Discovery (DAVID) was used to analyze the GO enrichment of potential targets of DEHP and BPA based on the terms of biological process (BP) and Kyoto Encyclopedia of Genes and Genomes (KEGG) signal pathway enrichment. From the consensus 47 biological processes, including 36 statistically significant biological process with P value < 0.05, 15 biological processes and KEGG signaling pathway with the smallest P value were chosen to display (Fig. 4c, d). We noticed that some biological processes may be related to the toxicity of DEHP and BPA, such as transcription regulation and steroid hormone–mediated signaling pathways, while KEGG signal pathway included steroid hormone biosynthesis, VEGF, estrogen receptor, thyroid hormone, prostate cancer, FoxO, and tumor and PI3K-AKT signaling pathway.
Effects of DEHP and BPA on Esr1-mediated transcriptional activation
Protein and mRNA levels of Esr1 were measured by immunohistochemical staining and RT-PCR in the study. Immunohistochemical results showed that Esr1 expression in the thyroid tissues of rats exposed to DEHP 750 and BPA 100 was higher, and DEHP 750 was significantly higher than that of the control (P < 0.05), while the expression level of Esr1 in the DEHP 750 + BPA 100 exposure group was significantly lower than that in the DEHP 750 and BPA 100 exposure group (P < 0.05) (Fig. 5a, b). Meanwhile, the Esr1 mRNA levels of rats exposed to DEHP 750 + BPA 100 were significantly lower than that of rats exposed to DEHP 750 (P < 0.05) (Fig. 5c). After the change of Esr1 was observed, the expression of its downstream p-Creb was further detected. Compared with the control, the expression of p-Creb in the DEHP 750 and BPA 100 exposed groups showed an increased way, and the DEHP 750 treatment was significant (P < 0.05) (Fig. 5d, e), while the mRNA levels of Creb in each group were not significantly changed (Fig. 5f).
Effects of DEHP and BPA on relative gene expression in the thyroid (n = 7). a Immunohistochemistry for Esr1 in the thyroid (original magnification, × 20). Scale bar = 200 μm. b The average optical density of Esr1 in the thyroid. c The mRNA expression of Esr1. d Immunohistochemistry for phosphorylation of Creb in the thyroid (original magnification, × 20). e The average optical density of phosphorylation of Creb in the thyroid. f The mRNA expression of Creb. An asterisk indicates significantly different at P < 0.05
Discussion
The present study focuses on the combined effects induced by two endocrine disruptor compounds, DEHP and BPA in female rats during puberty. Firstly, both DEHP and BPA have the potential to disrupt thyroid hormone homeostasis, while BPA showed a greater impact on serum TH levels than DEHP. Moreover, both DEHP and BPA individually could increase the number of follicular epithelial cells of thyroid. The combined effect of DEHP and BPA may depend on Esr1, further leading to the alteration of phosphorylation of Creb, and disturb the development of thyroid. No study to date has reported the interactions between DEHP and BPA on thyroid function indicated by in vivo experiment. In the present study, a 3 × 3 factorial design is used to analyze the complex effects of multiple EDCs on thyroid function, which can provide the opportunity to detect the interactions among multi-components (Collins et al. 2014). Since a 42-day in vivo experiment during puberty was used to clarify the toxicity of DEHP and BPA on thyroid development, a higher dose than human exposure was necessary. Hopefully, as a gross assessment of the short-term combined effects of DEHP and BPA on thyroid function, the present study may provide some references for our subsequent research, which contributes to our understanding of the adverse effect of EDCs mixture on human health during puberty.
Although the sequence of events that leads from puberty chemical exposure to altered dysfunction and even cancer is not completely understood, a weight of evidence is emerging that exposure to these chemicals during developmental periods produces persistent changes in growth, and functions in that tissue over the lifespan. Exposure to BPA and DEHP during puberty is of concern, based on numerous epidemiological studies reporting a range of effects associated with DEHP and BPA exposure, such as the effects related to neurobehavior, growth and development, and reproductive tissue dysfunction (Gore et al. 2015). In humans, thyroid hormone is important for normal development of the central nervous system, cardiovascular system, and other organs (Bernal 2007). Kim and Park reviewed that BPA can interfere with thyroid hormone synthesis, transport, and metabolism, and may affect thyroid function through several possible mechanisms of action (Kim and Park 2019). Furthermore, laboratory results found that neonatal exposure to BPA decreased the serum T4 and increased TSH levels in female rats (Fernandez et al. 2018); Dongjing et al. also reported a decreased tendency for total T4 levels and increased TSH levels in offspring with DEHP-treated pregnant female rats, while no change was found in total T3 (Dong et al. 2019). Other studies showed significantly reduced thyroid hormones (T3, T4) after DEHP exposure, whereas TSH was not affected (Liu et al. 2015). In the present study, along with the increasing dosage of DEHP or BPA, total T4 levels also decreased, especially at higher dose of BPA (100 mg/kg). When co-exposure to toxicants decreased the levels of total T3 and T4 in contrary to the single EDCs, the main effect analysis showed that BPA may have a greater impact on TH levels in serum than DEHP. TSH levels in serum were unaffected by DEHP and BPA which is consistent with the previous studies.
It is well known that DEHP and BPA may mimic or antagonize the effects of estrogens in target tissue, which could mediate main activities through the estrogen receptors (Casals-Casas and Desvergne 2011) (Tucker et al. 2018). Different compounds may induce the expression of different target genes via the same estrogen receptors (Katchy et al. 2014). Remarkably, women make up the majority of thyroid patients, and thyroid cancer is the most common malignancy tumor of the endocrine system, whose incidence is 2.9-times more common in women than in men (Rahbari et al. 2010). Increasing evidences suggested that estrogen may play an important role in the development and progression of thyroid cancer through estrogen receptors (Huang et al. 2014) (Fan et al. 2015). Similarly, our findings also suggested that thyroid gland may become a sensitive target of BPA and DEHP, and their exposure can affect the development of pubertal thyroid via disturbing the function of Esr1. Additionally, overall less is known about the exact mechanism of how DEHP and BPA affect the development of thyroid, bioinformatic analysis provided clues that Esr1 may act as an interaction mediator of DEHP and BPA, and compound-target-pathway enrichment analysis prompted us to focus on the estrogen signaling pathway in the present study. A significant reduction of mRNA expression of Esr1 was measured in the DEHP750 + BPA100–treated rats in comparison with BPA100 treatment. Then, the protein levels of Esr1 were detected, which showed a slight but not significant increase after DEHP or BPA alone treatment. Interestingly, the upregulation after exposure to DEHP or BPA was nullified by the concomitant presence of the other. It is clear that phosphorylate activation of the Creb through Esr1 has already been described in several models (Belcher et al. 2005) (Bouskine et al. 2009). Collectively, our data demonstrated that DEHP and BPA may change the levels of Esr1, further altered the levels of phosphorylated Creb, and ultimately effected the development of thyroid in adolescent female rats.
Conclusion
Together these data suggested that both DEHP and BPA have the potential to disrupt the thyroid hormone homeostasis, which might ultimately affect the thyroid function. Although the interaction of combined exposure is complex and not a simple summation of their single effect, our findings will deepen our understanding of the combined effects of DEHP and BPA on the thyroid development in adolescent female rats. Whether phthalates and bisphenols can induce the expression of related genes via a similar mechanism will be an interesting subject for the future exploration. Considering that humans are exposed to thousands of chemicals, a thorough analysis of combined effects of mixtures in humans needs to be undertaken to create a more reliable risk assessment of EDCs.
References
Ahern TP, Broe A, Lash TL, Cronin-Fenton DP, Ulrichsen SP, Christiansen PM, Cole BF, Tamimi RM, Sorensen HT, Damkier P (2019) Phthalate exposure and breast cancer incidence: a Danish nationwide cohort study. J Clin Oncol 37(21):1800–1809. https://doi.org/10.1200/JCO.18.02202
Aker AM, Watkins DJ, Johns LE, Ferguson KK, Soldin OP, Del Toro LVA, Alshawabkeh AN, Cordero JF, Meeker JD (2016) Phenols and parabens in relation to reproductive and thyroid hormones in pregnant women. Env Res 151:30–37
Aung MT, Johns LE, Ferguson KK, Mukherjee B, McElrath TF, Meeker JD (2017) Thyroid hormone parameters during pregnancy in relation to urinary bisphenol A concentrations: a repeated measures study. Environ Int 104:33–40. https://doi.org/10.1016/j.envint.2017.04.001
Belcher SM, Le HH, Spurling L, Wong JK (2005) Rapid estrogenic regulation of extracellular signal- regulated kinase 1/2 signaling in cerebellar granule cells involves a G protein- and protein kinase A-dependent mechanism and intracellular activation of protein phosphatase 2A. Endocrinology. 146(12):5397–5406. https://doi.org/10.1210/en.2005-0564
Bernal J (2007) Thyroid hormone receptors in brain development and function. Nat Clin Pract Endocrinol Metab 3(3):249–259. https://doi.org/10.1038/ncpendmet0424
Bilgi A, Abali R, Bilgi PT, Sahin M, Tuncdemir M, Boran AB (2019) The apoptotic effects of bisphenol A exposure on the rat ovary: an experimental study. Environ Sci Pollut Res Int. https://doi.org/10.1007/s11356-019-04487-6
Boughammoura S, Ben Mimouna S, Chemek M, Ostertag A, Cohen-Solal M, Messaoudi I (2020) Disruption of bone zinc metabolism during postnatal development of rats after early life exposure to cadmium. Int J Mol Sci 21(4). https://doi.org/10.3390/ijms21041218
Bouskine A, Nebout M, Brucker-Davis F, Benahmed M, Fenichel P (2009) Low doses of bisphenol A promote human seminoma cell proliferation by activating PKA and PKG via a membrane G-protein-coupled estrogen receptor. Environ Health Perspect 117(7):1053–1058. https://doi.org/10.1289/ehp.0800367
Bustin SA, Benes V, Garson JA, Hellemans J, Huggett J, Kubista M, Mueller R, Nolan T, Pfaffl MW, Shipley GL, Vandesompele J, Wittwer CT (2009) The MIQE guidelines: minimum information for publication of quantitative real-time PCR experiments. Clin Chem 55(4):611–622. https://doi.org/10.1373/clinchem.2008.112797
Casals-Casas C, Desvergne B (2011) Endocrine disruptors: from endocrine to metabolic disruption. Annu Rev Physiol 73:135–162. https://doi.org/10.1146/annurev-physiol-012110-142200
Collins LM, Dziak JJ, Kugler KC, Trail JB (2014) Factorial experiments: efficient tools for evaluation of intervention components. Am J Prev Med 47(4):498–504. https://doi.org/10.1016/j.amepre.2014.06.021
Couleau N, Falla J, Beillerot A, Battaglia E, D’Innocenzo M, Plancon S, Laval-Gilly P, Bennasroune A (2015) Effects of endocrine disruptor compounds, alone or in combination, on human macrophage-like THP-1 cell response. PLoS One 10(7):e0131428. https://doi.org/10.1371/journal.pone.0131428
David RM, Moore MR, Finney DC, Guest D (2000) Chronic toxicity of di(2-ethylhexyl)phthalate in rats. Toxicol Sci 55(2):433–443
Dong X, Dong J, Zhao Y, Guo J, Wang Z, Liu M, Zhang Y (2017) Effects of long-term in vivo exposure to di-2-ethylhexylphthalate on thyroid hormones and the TSH/TSHR signaling pathways in Wistar rats. https://doi.org/10.3390/ijerph14010044.
Dong J, Cong Z, You M, Fu Y, Wang Y, Wang Y, Fu H, Wei L, Chen J (2019) Effects of perinatal di (2-ethylhexyl) phthalate exposure on thyroid function in rat offspring. Environ Toxicol Pharmacol 67:53–60. https://doi.org/10.1016/j.etap.2019.01.012
Fan D, Liu SYW, van Hasselt CA, Vlantis AC, Ng EKW, Zhang H, Dong Y, Ng SK, Chu R, Chan ABW, du J, Wei W, Liu X, Liu Z, Xing M, Chen GG (2015) Estrogen receptor alpha induces prosurvival autophagy in papillary thyroid cancer via stimulating reactive oxygen species and extracellular signal regulated kinases. J Clin Endocrinol Metab 100(4):E561–E571. https://doi.org/10.1210/jc.2014-3257
Fernandez MO, Bourguignon NS, Arocena P, Rosa M, Libertun C, Lux-Lantos V (2018) Neonatal exposure to bisphenol A alters the hypothalamic-pituitary-thyroid axis in female rats. Toxicol Lett 285:81–86. https://doi.org/10.1016/j.toxlet.2017.12.029
Fiandanese N, Borromeo V, Berrini A, Fischer B, Schaedlich K, Schmidt J-S, Secchi C, Pocar P (2016) Maternal exposure to a mixture of di(2-ethylhexyl) phthalate (DEHP) and polychlorinated biphenyls (PCBs) causes reproductive dysfunction in adult male mouse offspring. Reprod Toxicol 65:123–132. https://doi.org/10.1016/j.reprotox.2016.07.004
Gao H, Wu W, Xu Y, Jin Z, Bao H, Zhu P, Su P, Sheng J, Hao J, Tao F (2017) Effects of prenatal phthalate exposure on thyroid hormone concentrations beginning at the embryonic stage. Sci Rep 7:13106. https://doi.org/10.1038/s41598-017-13672-x
Gore AC, Chappell VA, Fenton SE, Flaws JA, Nadal A, Prins GS, Toppari J, Zoeller RT (2015) EDC-2: the Endocrine Society’s second scientific statement on endocrine-disrupting chemicals. Endocr Rev 36(6):E1–E150. https://doi.org/10.1210/er.2015-1010
Guidry VT, Longnecker MP, Aase H, Eggesbo M, Zeiner P, Reichborn-Kjennerud T, Knudsen GP, Bertelsen RJ, Ye X, Calafat AM et al (2015) Measurement of total and free urinary phenol and paraben concentrations over the course of pregnancy: assessing reliability and contamination of specimens in the Norwegian Mother and Child Cohort Study. Environ Health Perspect 123(7):705–711. https://doi.org/10.1289/ehp.1408325
Guo Y, Kannan K (2012) Challenges encountered in the analysis of phthalate esters in foodstuffs and other biological matrices. Anal Bioanal Chem 404(9):2539–2554. https://doi.org/10.1007/s00216-012-5999-2
Hogberg J, Hanberg A, Berglund M, Skerfving S, Remberger M, Calafat AM, Filipsson AF, Jansson B, Johansson N, Appelgren M et al (2008) Phthalate diesters and their metabolites in human breast milk, blood or serum, and urine as biomarkers of exposure in vulnerable populations. Environ Health Perspect 116(3):334–339. https://doi.org/10.1289/ehp.10788
Hu Y, Wang R, Xiang Z, Qian W, Han X, Li D (2014) Antagonistic effects of a mixture of low-dose nonylphenol and di-n-butyl phthalate (monobutyl phthalate) on the Sertoli cells and serum reproductive hormones in prepubertal male rats in vitro and in vivo. PLoS One 9(3):e93425. https://doi.org/10.1371/journal.pone.0093425
Huang Y, Dong W, Li J, Zhang H, Shan Z, Teng W (2014) Differential expression patterns and clinical significance of estrogen receptor-alpha and beta in papillary thyroid carcinoma. BMC Cancer 14:383. https://doi.org/10.1186/1471-2407-14-383
Huang HB, Chuang CJ, Su PH, Sun CW, Wang CJ, Wu MT, Wang SL (2017) Prenatal and childhood exposure to phthalate diesters and thyroid function in a 9-year follow-up birth cohort study: Taiwan maternal and infant cohort study. Epidemiology 28:S10–S18. https://doi.org/10.1097/EDE.0000000000000722
Johns LE, Ferguson KK, McElrath TF, Mukherjee B, Meeker JD (2016) Associations between repeated measures of maternal urinary phthalate metabolites and thyroid hormone parameters during pregnancy. Environ Health Perspect 124:1808–1815. https://doi.org/10.1289/EHP170
Jung KK, Kim SY, Kim TG, Kang JH, Kang SY, Cho JY, Kim SH (2007) Differential regulation of thyroid hormone receptor-mediated function by endocrine disruptors. Arch Pharm Res 30(5):616–623
Kang J-H, Kondo F, Katayama Y (2006) Human exposure to bisphenol A. Toxicology. 226(2–3):79–89. https://doi.org/10.1016/j.tox.2006.06.009
Katchy A, Pinto C, Jonsson P, Nguyen-Vu T, Pandelova M, Riu A, Schramm K-W, Samarov D, Gustafsson J-A, Bondesson M, Williams C (2014) Coexposure to phytoestrogens and bisphenol A mimics estrogenic effects in an additive manner. Toxicol Sci 138(1):21–35. https://doi.org/10.1093/toxsci/kft271
Kim MJ, Park YJ (2019) Bisphenols and thyroid hormone. Endocrinol Metab (Seoul) 34:340–348. https://doi.org/10.3803/EnM.2019.34.4.340
Kinch CD, Kurrasch DM, Habibi HR (2016) Adverse morphological development in embryonic zebrafish exposed to environmental concentrations of contaminants individually and in mixture. Aquat Toxicol 175:286–298. https://doi.org/10.1016/j.aquatox.2016.03.021
Leung Y-K, Govindarajah V, Cheong A, Veevers J, Song D, Gear R, Zhu X, Ying J, Kendler A, Medvedovic M, Belcher S, Ho SM (2017) Gestational high-fat diet and bisphenol A exposure heightens mammary cancer risk. Endocr Relat Cancer 24(7):365–378. https://doi.org/10.1530/ERC-17-0006
Li L, Li H, Qu P, Xiao M, Zhang G, Zhang Q, Cai Y, Jin C, Yang J, Wu S, Lu X (2018 Dec.) An antagonism joint action of lead and di-2-ethylhexyl phthalate explains an improved ability of learning and memory after combined exposure in weaning rats. Biol Trace Elem Res 191:126–134. https://doi.org/10.1007/s12011-018-1586-5
Li S, Jin Y, Zhao H, Jiang Y, Cai Z (2019) Evaluation of bisphenol A exposure induced oxidative RNA damage by liquid chromatography-mass spectrometry. Chemosphere. 222:235–242. https://doi.org/10.1016/j.chemosphere.2019.01.120
Liu C, Zhao L, Wei L, Li L (2015) DEHP reduces thyroid hormones via interacting with hormone synthesis-related proteins, deiodinases, transthyretin, receptors, and hepatic enzymes in rats. Environ Sci Pollut Res Int 22(16):12711–12719. https://doi.org/10.1007/s11356-015-4567-7
Liu C, Deng Y-L, Zheng T-Z, Yang P, Jiang X-Q, Liu E-N, Miao X-P, Wang L-Q, Jiang M, Zeng Q (2020) Urinary biomarkers of phthalates exposure and risks of thyroid cancer and benign nodule. J Hazard Mater 383:121189. https://doi.org/10.1016/j.jhazmat.2019.121189
Lopez-Carrillo L, Hernandez-Ramirez RU, Calafat AM, Torres-Sanchez L, Galvan-Portillo M, Needham LL, Ruiz-Ramos R, Cebrian ME (2010) Exposure to phthalates and breast cancer risk in northern Mexico. Environ Health Perspect 118(4):539–544. https://doi.org/10.1289/ehp.0901091
Ma S, Shi W, Wang X, Song P, Zhong X (2017) Bisphenol A exposure during pregnancy alters the mortality and levels of reproductive hormones and genes in offspring mice. Biomed Res Int. https://doi.org/10.1155/2017/3585809
Markey CM, Luque EH, Munoz De Toro M, Sonnenschein C, Soto AM (2001) In utero exposure to bisphenol A alters the development and tissue organization of the mouse mammary gland. Biol Reprod 65(4):1215–1223
Marotta V, Russo G, Gambardella C, Grasso M, La Sala D, Chiofalo MG, D’Anna R, Puzziello A, Docimo G, Masone S et al (2019) Human exposure to bisphenol AF and diethylhexylphthalate increases susceptibility to develop differentiated thyroid cancer in patients with thyroid nodules. Chemosphere. 218:885–894. https://doi.org/10.1016/j.chemosphere.2018.11.084
Meeker JD, Calafat AM, Hauser R (2007) Di(2-ethylhexyl) phthalate metabolites may alter thyroid hormone levels in men. Environ Health Perspect 115:1029–1034. https://doi.org/10.1289/ehp.9852
Mendoza A, Hollenberg AN (2017) New insights into thyroid hormone action. Pharmacol Ther 173:135–145. https://doi.org/10.1016/j.pharmthera.2017.02.012
Miller MD, Crofton KM, Rice DC, Zoeller RT (2009) Thyroid-disrupting chemicals: interpreting upstream biomarkers of adverse outcomes. Environ Health Perspect 117:1033–1041. https://doi.org/10.1289/ehp.0800247
Mondal S, Raja K, Schweizer U, Mugesh G (2016) Chemistry and biology in the biosynthesis and action of thyroid hormones. Angew Chem Int Ed Engl 55(27):7606–7630. https://doi.org/10.1002/anie.201601116
Morgenstern R, Whyatt RM, Insel BJ, Calafat AM, Liu X, Rauh VA, Herbstman J, Bradwin G, Factor-Litvak P (2017) Phthalates and thyroid function in preschool age children: sex specific associations. Environ Int 106:11–18. https://doi.org/10.1016/j.envint.2017.05.007
Bin HH, Pan WH, Chang JW, Chiang HC, Guo YL, JJK J, Huang PC (2017) Does exposure to phthalates influence thyroid function and growth hormone homeostasis? The Taiwan Environmental Survey for Toxicants (TEST) 2013. Environ Res 153:63–72. https://doi.org/10.1016/j.envres.2016.11.014
Park C, Choi W, Hwang M, Lee Y, Kim S, Yu S, Lee I, Paek D, Choi K (2017) Associations between urinary phthalate metabolites and bisphenol A levels, and serum thyroid hormones among the Korean adult population - Korean National Environmental Health Survey (KoNEHS) 2012–2014. Sci Total Environ 584-585:950–957. https://doi.org/10.1016/j.scitotenv.2017.01.144
Rahbari R, Zhang L, Kebebew E (2010) Thyroid cancer gender disparity. Future Oncol 6(11):1771–1779. https://doi.org/10.2217/fon.10.127
Riaz N, Wolden SL, Gelblum DY, Eric J (2016) HHS public access. 118(24):6072–6078. https://doi.org/10.1002/cncr.27633.Percutaneous
Shelby MD (2006) NTP-CERHR monograph on the potential human reproductive and developmental effects of di (2-ethylhexyl) phthalate (DEHP). NTP CERHR MON.(18):v, vii-7, II–iii-xiii passim
Stojanoska MM, Milosevic N, Milic N, Abenavoli L (2017) The influence of phthalates and bisphenol A on the obesity development and glucose metabolism disorders. Endocrine 55:666–681. https://doi.org/10.1007/s12020-016-1158-4
Suk WA, Olden K, Yang RSH (2002) Chemical mixtures research: significance and future perspectives. Environ Health Perspect 110(Suppl):891–892. https://doi.org/10.1289/ehp.110-1241268
Sunman B, Yurdakok K, Kocer-Gumusel B, Ozyuncu O, Akbiyik F, Balci A, Ozkemahli G, Erkekoglu P, Yurdakok M (2019) Prenatal bisphenol A and phthalate exposure are risk factors for male reproductive system development and cord blood sex hormone levels. Reprod Toxicol 87:146–155. https://doi.org/10.1016/j.reprotox.2019.05.065
Suteau V, Briet C, Lebeault M, Gourdin L, Henrion D, Rodien P, Munier M (2020) Human amniotic fluid-based exposure levels of phthalates and bisphenol A mixture reduce INSL3/RXFP2 signaling. Environ Int 138:105585. https://doi.org/10.1016/j.envint.2020.105585
Tucker DK, Hayes Bouknight S, Brar SS, Kissling GE, Fenton SE (2018) Evaluation of prenatal exposure to bisphenol analogues on development and long-term health of the mammary gland in female mice. Environ Health Perspect 126(8):87003. https://doi.org/10.1289/EHP3189
US Environmental Protection Agency (2011) Bisphenol A action plan. Environ Heal
Watkins DJ, Sanchez BN, Tellez-Rojo MM, Lee JM, Mercado-Garcia A, Blank-Goldenberg C, Peterson KE, Meeker JD (2017) Phthalate and bisphenol A exposure during in utero windows of susceptibility in relation to reproductive hormones and pubertal development in girls. Environ Res 159:143–151. https://doi.org/10.1016/j.envres.2017.07.051
Welshons WV, Nagel SC, vom Saal FS. (2006) Large effects from small exposures. III. Endocrine mechanisms mediating effects of bisphenol A at levels of human exposure. Endocrinology 147(6 Suppl):S56–S69. https://doi.org/10.1210/en.2005-1159
Weng TI, Chen MH, Lien GW, Chen PS, Lin JCC, Fang CC, Chen PC (2017) Effects of gender on the association of urinary phthalate metabolites with thyroid hormones in children: a prospective cohort study in Taiwan. Int J Environ Res Public Health 14. https://doi.org/10.3390/ijerph14020123
Xie Y, Jiang L, Zhang X, Jiang F, Zhou Z, Zhang J (2017) Higher urinary bisphenol A concentration and excessive iodine intake are associated with nodular goiter and papillary thyroid carcinoma. Biosci Rep 37. https://doi.org/10.1042/bsr20170678
Funding
This study was supported by grants from the National Natural Science Foundation of China (81773470), National Cancer Climbing Fund (NCC201806B009), Liaoning Provincial Science and Technology Plan (2019-ZD-0595), and Liaoning Provincial Key Research and Development Program (No.2017225056).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
All experiments and surgical procedures were approved by the Animal Use and Care Committee of China Medical University, which complies with the National Institutes of Health Guide for the Care and Use of Laboratory Animals. All efforts were made to minimize the number of animals and their suffering.
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Responsible Editor: Mohamed M. Abdel-Daim
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Zhang, X., Zhao, Y., Cheng, C. et al. Combined effects of di (2-ethylhexyl) phthalate and bisphenol A on thyroid hormone homeostasis in adolescent female rats. Environ Sci Pollut Res 27, 40882–40892 (2020). https://doi.org/10.1007/s11356-020-09949-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-020-09949-w