Abstract
Urbanisation threatens species through habitat loss, isolation and fragmentation. Greenspace in urban landscapes often provides connectivity and habitat for wetland-dependent species. Accessible greenspace measures the combined effects of movement barriers (e.g., roads) and the total amount of greenspace on populations. I determined whether accessible greenspace was a better predictor of amphibian occupancy at ponds than the total amount of greenspace, and at what spatial scale. I also assessed the relative importance of habitat variables at the local pond-scale. Frog surveys were conducted at 65 ponds distributed along an urban–rural gradient over two breeding seasons in the greater Melbourne region, Australia. Accessible greenspace was mapped around ponds at varying spatial scales (250 m – 2000 m) using potential barriers to amphibian movement. The best-ranked multi-species occupancy model included the proportion of total greenspace within a 1000-m radius of a pond and four local-scale covariates (presence of mosquitofish, pond permanence, vertical pond walls and water conductivity). There was no support for models containing accessible greenspace. There was a positive relationship between the probability of community and species-level occupancy and total greenspace within 1000 m of a pond. There was a negative relationship between mean community occupancy and the presence of a vertical pond wall at a site. There was no apparent effect of barriers on occupancy, indicating that urban areas may be more permeable for movement by some species than predicted. Individual species responses demonstrated that management actions are required at fine and broad spatial scales to benefit amphibian communities in urban landscapes.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Urbanisation involves the widespread transformation of natural land surfaces, replacing vegetation and wetlands with roads, houses, buildings and other non-natural impervious surfaces, which subsequently fragments any remaining natural habitats into smaller patches (McKinney 2002; Theobald et al. 1997). Predicted trends show that by 2030 there will be an almost three-fold increase in global urban land cover from 2000 to 2030, corresponding with an increase of 5 billion people to the global urban population (Seto et al. 2011). Unsurprisingly, the rapid landscape transformation associated with urbanisation is driving increased rates of species extinctions, largely due to habitat loss and fragmentation, but also because of non-native species introductions (Czech et al. 2000; Grimm et al. 2008; Shochat et al. 2010; Vitousek et al. 1997). A key challenge in mitigating the effect of urbanisation on biodiversity is preserving habitat patches (e.g., greenspace) of sufficient size and quality to maintain viable remnant populations (Haaland and van den Bosch 2015; Hahs et al. 2009), given that most greenspaces are small, isolated and highly fragmented (Lepczyk et al. 2017).
Urban greenspace is defined as any vegetation found in the urban environment, including parks, open spaces and residential gardens (Haaland and van den Bosch 2015), although this definition can be extended to include urban ponds that are often embedded within greenspace (Oertli and Parris 2019). The importance of greenspace and associated wetlands for the persistence of wildlife in urban areas has been demonstrated for multiple taxa (Hamer et al. 2018; Matthies et al. 2017; Straka et al. 2016; Thornhill et al. 2017). Wetland-dependent organisms such as amphibians depend on inter-connected networks of aquatic and terrestrial habitats to facilitate movement among habitat patches within metapopulations (Semlitsch 2002). Ponds and wetlands provide breeding sites while non-breeding terrestrial habitats provide foraging and shelter sites, and both habitat types need to be linked to enable species to complete their complex life cycles through landscape complementation (Pope et al. 2000). Pond-breeding amphibian species have aquatic-developing larvae that metamorphose and disperse towards terrestrial habitats (Semlitsch 2002), and barriers that prevent dispersal (e.g., roads) can severely disrupt metapopulation dynamics (Hamer et al. 2015; Marsh et al. 2017). Over one-third of amphibian species are currently threatened by urbanisation due to habitat loss, isolation and fragmentation, and deteriorating habitat quality (Hamer and McDonnell 2008). Despite the myriad of threats faced by amphibians in urban areas, populations of some species can persist in urban ponds if surrounded by an adequate quantity and quality of greenspace that provides breeding habitats and landscape connectivity (Fuyuki et al. 2014; Hutto and Barrett 2021; Kruger et al. 2015; Li et al. 2018).
There is currently a call for greater understanding of the importance of urban greenspace for biodiversity, particularly from studies on less-studied taxa and that focus on responses to landscape and local-scale factors, underpinned by metapopulation theory (Lepczyk et al. 2017). Despite the novelty of many urban ecosystems and greenspaces (Aronson et al. 2017; Kowarik 2011), existing ecological theory (e.g., metacommunity) is considered sufficient for understanding how amphibian communities respond to urbanisation (Parris 2018). A metacommunity is a set of local communities that are linked by dispersal of multiple potentially interacting species (Leibold et al. 2004). Other ecological theories may equally apply in urban environments – for instance, the pond permanence gradient provides a basis for understanding how amphibian communities respond to pond drying and fish predation (Wellborn et al. 1996), although this will depend on the availability of certain pond types in urban areas, particularly ephemeral ponds which are often destroyed during urbanisation (Hamer and Parris 2013; Urban and Roehm 2018).
Research into the effect of fragmentation of urban greenspace on amphibian communities is likely to benefit by adopting approaches taken previously to investigate the impacts of linear barriers within circular buffers around breeding sites. In urban landscapes, the extent of terrestrial habitat around a pond, measured at distances up to 2000 m, usually correlates with an increase in amphibian species richness and occupancy (Gagné and Fahrig 2007; Green et al. 2021; Guderyahn et al. 2016; Smallbone et al. 2011). However, increases in road density around ponds invariably leads to reductions in terrestrial habitat and aquatic connectivity (and hence pond isolation), in addition to road effects and road mortality, all of which negatively impact amphibian communities (Beebee 2013; Parris 2006; Sutherland et al. 2010; Trombulak and Frissell 2000). Therefore, when assessing relationships between greenspace and amphibian distributions in urban landscapes it may be important to consider the availability and configuration of terrestrial habitat around a pond relative to the presence of linear barriers.
Accessible habitat measures the combined effects of linear barriers and habitat amount, defined as the amount of terrestrial habitat that can be reached by an amphibian from a focal habitat patch (e.g., pond) without crossing a road or other barrier, thereby acknowledging that habitat accessible from a focal patch without crossing a road is likely to be more important for maintaining metapopulation dynamics and landscape complementation than habitat on the other side of the road (Eigenbrod et al. 2008). In an urban context, accessible greenspace is therefore the amount of greenspace that can be accessed from a pond without crossing a movement barrier. Studies examining the relative effect of accessible habitat on amphibians have generally hypothesised that accessible habitat is the best predictor of the effects of habitat loss and roads for species for which roads are a major barrier to movement, and so there should be a stronger relationship between accessible habitat and species than total habitat amount (Eigenbrod et al. 2008; Hamer 2016, 2018). Barrier-based buffers are a similar method, using major impassable barriers to dispersal such as highways, railways and rivers to make predictions about amphibian pond occupancy compared to the area of terrestrial habitat measured in traditional circular buffers around a site (Zanini et al. 2008). While these studies examined relationships between accessible habitat and amphibian distribution in agricultural or peri-urban settings, no studies have assessed accessible habitat in highly-urbanised landscapes.
Here, I examined relationships between greenspace and amphibian community occupancy at ponds in an urban landscape using accessible greenspace. I predicted that (1) the proportion cover of accessible greenspace around a pond would be a better predictor of amphibian occupancy than the total amount of greenspace; (2) the proportion cover of greenspace within a 1000-m radius around a pond would be the best spatial scale for inference; and (3) local-scale habitat variables would be important for pond occupancy. Important relationships between greenspace measured within a 1000-m radius around a pond, local habitat quality and amphibian species richness and community composition were evident previously in the study area (Hamer and Parris 2011). I used hierarchical, multi-species occupancy modelling (MSOM) to estimate the probability of pond occupancy at a community-level scale and for individual frog species, thereby aiming to link metacommunity structure at ponds to the availability and accessibility of urban greenspace. The application of hierarchical multi-scale models to address questions of landscape ecology has been recommended in urban environments, given the fine spatial scales at which urban areas are fragmented (Norton et al. 2016). I summarise management interventions that could be applied in urban ponds and greenspace to increase the persistence of amphibian metacommunities and highlight the utility of using MSOM for monitoring amphibian communities in urban areas.
Methods
Study area
The study was conducted in the greater Melbourne region, Victoria, in south-eastern Australia, which includes the city of Melbourne. The region was first settled by European people in 1835 and had a human population of just over 4 million in 2010, which is predicted to reach 6.5 million by 2051 (Ives et al. 2013). Melbourne has a temperate climate, with an annual mean temperature of 15.5 °C and an annual mean precipitation of 639 mm, distributed evenly throughout the year (Stern 2005). The area of remnant wetlands in the inner and outer suburbs of Melbourne totals ca. 4500 ha, while only 2% and 16% of the inner-city area and outer suburbs of Melbourne, respectively, are still covered in remnant vegetation (McDonnell and Holland 2008). Habitat loss and fragmentation due to urbanisation since the latter half of the twentieth century has resulted in declining populations of several frog species in the region (Hamer and McDonnell 2010).
Study site selection
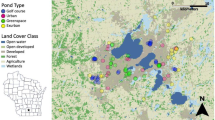
Sixty-five ponds (lentic waterbodies) were selected along an urban–rural gradient, stratified by the proportion of road cover in a 500-m radius around each pond, and by pond surface area (small: < 200 m2; and large: ≥ 200 m2). Site selection included a range of waterbody types including ornamental ponds in city parks and gardens, stormwater retention ponds, waterbodies in disused quarries, and semi-natural wetlands, varying greatly in size (2 – 51,104 m2). Nearest neighbour distances between ponds were between 2 m and 15.7 km (mean = 972 m; SD = 2607 m). See Fig. 1 for a map of the pond distribution relative to urban greenspace.
Frog surveys
Four frog surveys were conducted at the 65 ponds over two breeding seasons in spring, summer and autumn (November – December 2007, March – April 2008, August – September 2008 and November – December 2008). These dates corresponded with the breeding period of the 14 frog species recorded in the greater Melbourne region (Hero et al. 1991). It was assumed that the pond system was closed to local colonisation/extinction by frogs, despite extending over two years. The 65 ponds were assigned to one of 22 spatial clusters, each containing 1 – 17 wetlands, with each cluster comprising ponds ≤ 2 km apart that were not separated by major roads (i.e., barriers to dispersal). To eliminate bias in survey timing, the sequence that ponds were surveyed was randomised according to site cluster. Frog surveys comprised quiet listening at ponds for 5 min to detect calling male frogs (Scott and Woodward 1994). Frog species present in the Melbourne region have distinct mating advertisement calls and so there was minimal risk of making false positive detections. Visual encounter surveys were then conducted usually by one observer using a headlight to search for frogs in areas of aquatic vegetation, open water and around the shoreline, as well as the ground and vegetation within 10 m of the shoreline (Crump and Scott 1994). Frog surveys commenced approximately 30 min after sunset. The amount of time spent searching was proportional to pond size and habitat complexity. The number of frog species heard and seen was recorded, including life stage (metamorph, juvenile, sub-adult, adult). Tadpole sightings during frog surveys were not included in the results. Air temperature (wet bulb) was recorded at the start of each survey. Protocols to reduce the risk of spreading the amphibian chytrid fungus (Batrachochytrium dendrobatidis) were followed when conducting fieldwork (Phillott et al. 2010).
Accessible greenspace
Accessible greenspace (ACC_GREEN) was defined as the total area of land mapped as green open space within varying buffer radii extending around each site from the pond margin, that could be reached by a dispersing frog without having to cross main roads, highways, railways, rivers or dense inner-city urban matrix. Greenspace was mapped using a GIS spatial layer composed of parks and gardens, remnant patches of native vegetation, and recreational fields (Open Space 2002: Australian Research Centre for Urban Ecology 2003), many of which contain ponds and wetlands (Parris 2006). Greenspace was considered a potential measure of landscape connectivity, and both terrestrial and aquatic habitat availability (Hamer and Parris 2011), which amphibians require to sustain complex life cycles within metapopulations (Semlitsch 2002). I assumed that frog species in the study area are attempting to cross most roads, but that main roads and highways represent significant barriers to movement, with smaller two-lane roads having a weaker barrier effect due to a smaller physical footprint and potentially lower traffic volumes at night when frogs are most likely to disperse (Eigenbrod et al. 2008). I also assumed that railway lines and rivers (e.g., Yarra River) were completely impassable barriers for frogs, but also that highly-developed urban land contained a range of barriers (e.g., fences, walls, buildings, wide areas of open impervious surfaces) that constituted absolute barriers to amphibian movement (Joly et al. 2003; Zanini et al. 2008). To determine the relative importance of accessible greenspace in predicting the combined effects of habitat availability and natural and artificial barriers on amphibian occupancy, the total area of greenspace (GREEN) was also measured, ignoring barriers within buffers. Accessible and total greenspace were measured within buffers of varying radii extending from the pond margin – 250 m, 500 m, 1000 m and 2000 m – corresponding with distances either recommended for landscape analyses of biodiversity in urban ponds (Oertli and Parris 2019) or used in previous studies of amphibian populations in the greater Melbourne area (Hamer and Parris 2011; Heard et al. 2012). All GIS calculations were performed using QGIS v3.4.9 (QGIS Geographic Information System 2020). Main roads, highways and railways, and urban land cover were mapped using OpenStreetMap (© OpenStreetMap contributors).
Local habitat variables
Any assessment of amphibian distribution in urban areas should also consider habitat quality at local pond scales (Hamer and McDonnell 2008). Accordingly, local habitat variables were measured at ponds and included variables previously examined in amphibian research in the study area (Hamer and Parris 2011; Parris 2006). The proportion of the pond surface area covered by aquatic vegetation (i.e., emergent, submerged and floating vegetation; Aqveg) was recorded to measure vegetation availability and complexity. Emergent and submerged vegetation extended above or below the water surface, respectively, while floating vegetation included surface algae and macrophytes with free-floating roots or roots attached to the pond substrate. Predation by fish and hydroperiod can be strong drivers in structuring amphibian communities in ponds (Snodgrass et al. 2000). The predatory, non-native eastern mosquitofish (Gambusia holbrooki; hereafter Gambusia) eats the eggs and larvae of several Australian frog species (Pyke and White 2000). A previous investigation of breeding distribution in the study area found a negative relationship between the occurrence and abundance of tree frog (Litoria spp.) larvae and Gambusia, while there were positive associations with aquatic vegetation cover and ephemerality (Hamer and Parris 2013). The presence of Gambusia at a pond was determined during aquatic surveys conducted the morning after the frog surveys (see Hamer and Parris 2011). Hydroperiod (Permanence) was recorded at ponds throughout the study: ponds that were not observed to dry out were scored as permanent (1), whereas ponds that were dry during at least one site visit were scored as ephemeral (0). Pond surface area (Area) was measured at the high-water mark using GIS. Pond water electrical conductivity (EC; µS cm−1) was measured in 200-ml water samples collected from four equidistant points around the pond perimeter, ~ 1 m from the shoreline and at a depth of ~ 10 cm using a handheld electronic meter (Tracer Pocketester, LaMotte Company, Chestertown, Maryland, USA). The presence of a vertical pond wall (V_wall) was noted at sites, defined as a continuous (unbroken) vertical drop (~ 20–30 cm high) from the pond bank to the surface of the water, and usually constructed of concrete, stone or wood (Parris 2006). Some ponds had banks that were partly comprised of a vertical wall but the remainder was sloping; accordingly, these sites were not scored as having a (continuous) vertical pond wall. Vertical pond walls are likely to make a pond unsuitable for breeding by many ground-dwelling frogs that cannot climb steep surfaces; however, tree frogs are usually proficient at climbing vertical slopes. Hence, upon metamorphosis their offspring can climb out of the pond, whereas the metamorphs of ground-dwelling frogs cannot climb and may drown (Parris 2006).
Multi-species occupancy modelling
I used a community-level hierarchical modelling approach (i.e., multi-species occupancy modelling) to estimate the community-level and species-specific responses to the two landscape covariates, each measured at four different radii around a pond (ACC_GREEN, GREEN), and the six local habitat covariates (Aqveg, Area, Gambusia, Permanence, EC, V_wall). Using Bayesian inference, hierarchical community models estimate community-level parameters by estimating species-specific probabilities of occupancy and detection (Dorazio and Royle 2005; Dorazio et al. 2006). Crucially, multi-species occupancy models (MSOM) address the issue of imperfect species detection that often bias conventional statistics used to analyse community occurrence data by linking metacommunity structure to underlying environmental gradients (Mihaljevic et al. 2015). Occupancy models for individual species are linked together into a hierarchical model so that collectively they provide estimates of community-level responses to environmental covariates, which increases the precision of parameter estimates for infrequently-observed species (Dorazio et al. 2006; Kéry and Royle 2008). Data from species frequently detected at sites is shared across the community to improve the predictive ability of parameter estimates for rare species, which results in the mean estimates of data-poor species being drawn towards group averages (Link 1999).
The structure of the MSOM followed the methods of (Dorazio et al. 2006), where the first level of hierarchy is based on an occupancy sub-model that assumed a true (but only partially observed) presence–absence matrix zi,j for species i = 1, 2,..., N at site j = 1, 2,..., J, where zi,j = 1 if species i was present at site j, and zi,j = 0 if the species was absent, and where zi,j ~ Bern(Ψi,j), and Ψi,j is the probability that species i occurs at site j. Because the state variable zi,j is usually not known with certainty, detection data xi,j,k is observed for species i at site j during sampling period k, which are also assumed to be Bernoulli random variables if species i is present. Thus, the data provided a three dimensional matrix xi,j,k for species i at site j for the kth sampling occasion. The second level of the model hierarchy was based on an observation sub-model, which specified that xi,j,k ~ Bern(θi,j,k × zi,j) where zi,j is the true occurrence matrix described above, and θi,j,k is the probability of detection for species i at site j for the kth sampling occasion. When species i was not present, detection was fixed to zero because zi,j = 0.
A series of MSOM were constructed of different covariates to examine relationships between accessible greenspace at multiple spatial scales and frog occupancy. Initially, intercorrelations were assessed among the landscape- and local-scale covariates using Pearson correlation coefficients (r) for pairwise comparisons of continuous variables, and Spearman rank correlation coefficients for binary variables (rs; ESM Table S1). Given the strong intercorrelations among landscape covariates (r > 0.4), each MSOM was limited to one landscape covariate. There was a strong correlation between pond area and the presence of Gambusia (rs = 0.609) and so each were not included in the same models. The proportion cover of aquatic vegetation was strongly correlated with all eight landscape covariates and was excluded from the landscape-scale models (Table S1). However, two models were run that included only local-scale covariates: Aqveg, V_wall, EC and either Area or Gambusia, to assess whether aquatic vegetation was more important than the landscape covariates. Pond permanence was strongly correlated with aquatic vegetation (rs = −0.404; Table S1) and so was not included in the local-scale covariate models. Given these constraints, a final set of 16 landscape models was examined, each containing one landscape covariate and four local-scale covariates, plus two models containing only local-scale covariates (Table 1). A categorical cluster term (Cluster) was added as a random effect in each model to account for spatial autocorrelation in pond occupancy because some ponds were within dispersal distance of their neighbouring ponds (Parris 2006). Spatial autocorrelation in wildlife observation data can result in biased estimation of model coefficients and diminished predictive performance (Wintle and Bardos 2006). Ponds were assigned into 22 spatial clusters (see “Frog surveys”). Each model had a maximum of five explanatory variables to eliminate the risk of overparameterisation that can result in inaccurate estimates of model coefficients (Harrison et al. 2018).
Three survey-specific covariates were also included in models to jointly estimate the probability of detection: number of days since 1 September (Days) separately for each survey year to account for variation in species detection since the start of the field surveys and corresponding to the beginning of the activity season for spring-breeding frogs; air temperature (Temp); survey year (Year: 1 = spring/summer 2007 – autumn 2008; 2 = spring/summer 2008). I assumed that the frog metacommunity would be closed over the two years during the study (i.e., no local colonisation or extinction), but added a year effect to account for potential differences in frog detection between Years 1 and 2. Survey date and air temperature influence the probabilities of detection for several frog species in the greater Melbourne area (Canessa et al. 2012).
Pond area and EC were log10(x)-transformed prior to analysis. Continuous covariates were standardised to have a mean = 0 and SD = 1. Standardised covariates allowed direct comparison of model coefficients so that the relative importance of each covariate could be determined according to the magnitude of the coefficient (Schielzeth 2010). Missing values of survey-specific covariates were replaced by the mean.
In the community-level of the hierarchical model, the species-specific parameters were linked to the broader metacommunity by estimating hyper-parameters (µ) that treated species-level parameters as random effects drawn from a normal distribution (Zipkin et al. 2009). For example, α1i ~ N(µα1, σα1) where α1i is the probability of occupancy for species i in response to site-specific covariate 1, µα1 is the mean community response (across species) to covariate 1, and σα1 is the standard deviation in α1 among species. Species’ responses in the metacommunity were assumed to be drawn from a common distribution where the species assessed have similar ecological requirements, and hence, species would have similar ecological responses to the environmental covariates (Pacifici et al. 2014).
Model parameters and community summaries were estimated using Bayesian inference with priors for the hyper-parameters drawn from a normal distribution (N[0, 2.25]) and uniform priors for the standard deviation of the estimates (U[0, 5]) (Guillera-Arroita et al. 2019). The cluster term was modelled using a uniform prior (U[0, 10]). I calculated the mean, standard deviation, and the 10th and 90th percentiles of the posterior distribution of the model coefficients, which represents a 90% Bayesian credible interval (BCI). I used a 90% BCI rather than 95% BCI given the inherently high variability in mean responses of animal communities to environmental covariates (Zipkin et al. 2009), which might otherwise obscure important relationships. Parameter estimates of covariates with a 90% BCI that did not overlap zero were considered to be clearly important, whereas estimates with a BCI overlapping zero had greater uncertainty. However, parameter estimates with ≥ 0.95 of the posterior distribution mass on one side of zero (either positive or negative) were also considered to be strongly influential. The probability of occupancy and detection were derived for both community-level and species-specific summaries using the log transformation: exp(α)/(1 + exp[α]).
Modelling was performed using the software program JAGS (version 4.3.0, Plummer 2013) called via the R2jags package (Su and Yajima 2015) from program R (version 3.6.1, R Core Team 2019). Each model was run using three replicate Markov chain Monte Carlo (MCMC) iterations to generate 100,000 samples from the posterior distribution of each model after discarding a ‘burn-in’ of 50,000 samples and a thinning rate of 5. The Gelman-Rubin statistics indicated there was acceptable convergence for all estimated parameters (i.e., \(\widehat{R}\) <1.05; Brooks and Gelman 1998; Gelman and Rubin 1992).
The 16 competing models of accessible greenspace versus total greenspace at each spatial scale, plus two local-scale covariate models, were ranked according to the relative fit of each model to the data, using the Widely Applicable Information Criterion (WAIC; Watanabe 2010). The WAIC is a fully Bayesian approach for estimating the computed log pointwise posterior predictive density of a model, which adds a correction for the effective number of parameters to adjust for model overfitting (Gelman et al. 2014). Similar to recommended rankings of the Akaike Information Criterion (AIC), the best-ranked models were considered to be those with a ΔWAIC < 2 (ΔWAIC = WAIC – minimum[WAIC]), while models with a ΔWAIC > 10 have essentially no support (Anderson 2008). Bayesian p-values were used to assess model fit and were calculated using the Freeman‐Tukey fit statistic (see Stolen et al. 2019). Bayesian p-values close to 0.5 indicate acceptable model fit (Gelman et al. 1996).
Results
Frog detections
A total of 10 frog species were detected (mean: 1.5 species site–1; SD: 1.5; range: 0 – 6), representing three taxonomic families (Limnodynastidae and Myobatrachidae: ground frogs; Pelodryadidae: tree frogs). The most frequently detected species was the southern brown tree frog (Litoria ewingii; naïve occupancy rate = 0.62), followed by the common eastern froglet (Crinia signifera; 0.31) and the striped marsh frog (Limnodynastes peronii; 0.23), whereas Peron’s tree frog (Lit. peronii) and the growling grass frog (Lit. raniformis) were detected at only one site (naïve occupancy rate = 0.02). A further three species were detected at < 5 sites: Victorian smooth froglet (Geocrinia victoriana), spotted grass frog (Lim. tasmaniensis) and Haswell’s froglet (Paracrinia haswelli). The remaining two species (southern bullfrog Lim. dumerilii and whistling tree frog Lit. verreauxii) were detected at nine and six sites, respectively (naïve occupancy rates: 0.14, 0.09). All species detected are native and have aquatic-developing larvae. Geocrinia victoriana deposits eggs in moist terrestrial microhabitats whereas the other species oviposit directly in water.
The mean estimated number of frog species (species richness) at a site was 2.5 (0.2 – 7.2) which is slightly higher than the naïve mean species richness. The mean probability of occupancy across all species was 0.311 (90% BCI: 0.175–0.494), whereas the mean probability of detection was 0.293 (90% BCI: 0.184–0.433). Mean estimated probabilities of occupancy varied markedly among the 10 frog species detected, ranging from 0.024 (90% BCI: 0.002–0.220) for Lit. peronii to 0.721 (0.421–0.913) for Lit. ewingii (Table 2). Mean estimated probabilities of detection were also highly variable, ranging from 0.085 (90% BCI: 0.011–0.361) for Lit. raniformis to 0.514 (0.360–0.668) for Lit. ewingii (Table 2).
Accessible greenspace
The barriers selected for delineating accessible greenspace substantially reduced the total amount of greenspace around ponds that could potentially be accessed via movement. There was a steeper decline in accessible greenspace area as buffer radii increased compared to the area of total greenspace. For example, there was a 77% decrease in the mean area of accessible greenspace from a 250-m to 2000-m radius around a pond, compared to a 60% decrease in total greenspace, while the mean area of accessible greenspace at a 2000-m radius was only 53% of the mean area of total greenspace (ESM Table S2).
Model inference and fit
The best-supported model included the proportion of total greenspace within a 1000-m radius of a pond, Gambusia, pond permanence, presence of a vertical pond wall and electrical conductivity (Model 1, WAIC = 3052.5; Table 1). There was also strong support for a model that included the proportion of total greenspace within 2000 m of a pond and the same local-scale variables (Model 2, WAIC = 3054.0; Table 1). There was no support for the remaining 14 landscape-scale covariate models (ΔWAIC > 10), including models containing the proportion cover of accessible greenspace (Table 1). There was no support for the two local-scale covariate models containing the proportion cover of aquatic vegetation (Table 1). Bayesian p-values for Models 1 and 2 indicated acceptable model fit (Model 1: p = 0.126; Model 2: p = 0.133). Model 1 was subsequently used to derive parameter estimates of occupancy and detection.
Community-level summary
There was a positive relationship between the mean estimated probability of occupancy across all frog species and the proportion cover of total greenspace within 1000 m of a pond (μα1; Table 3). However, the 90% BCI overlapped zero, indicating there was uncertainty associated with this estimate. Mean occupancy was predicted to increase from 0.251 at sites with no greenspace within 1000 m, to 0.507 at sites surrounded by 100% greenspace (Fig. 2). The standard deviation of the mean probability of occupancy was relatively low (σα1 = 1.025; Table 3), indicating there were consistently positive responses to total greenspace across the frog communities.
There was a relatively strong negative relationship between the mean occupancy across all frog species and the presence of a vertical pond wall at a site (μα4; Table 3). Mean occupancy was predicted to decrease from 0.323 (90% BCI: 0.175–0.494) at sites without a vertical pond wall, to 0.175 (0.067–0.317) at sites with a vertical pond wall. The standard deviation of the mean probability of occupancy was relatively high (σα4 = 3.335; Table 3) indicating that species had variable responses to the presence of a vertical pond wall.
There were no clear relationships between the mean probability of community occupancy and the presence of Gambusia, pond permanence or electrical conductivity, with 90% BCIs of all parameter estimates overlapping zero broadly (Table 3). However, the presence of Gambusia was included in the best-ranked models rather than pond area (Table 1), indicating that Gambusia had a greater influence on occupancy.
There was strong evidence of positive and negative spatial autocorrelation at each of two site clusters (ESM Fig. S1).
Species-specific summary
There were clear or influential positive relationships between the estimated probability of occupancy of Crinia signifera, Lit. ewingii and Paracrinia haswelli and the proportion cover of total greenspace within 1000 m (Table 4; Fig. 3). The predicted increase in occupancy was greatest for C. signifera where the mean was predicted to increase from 0.173 at sites with no greenspace within 1000 m, to 0.792 at sites surrounded by 100% greenspace (Fig. 3).
Predicted relationships between the species-specific estimates of the probability of occupancy of 10 frog species and the proportion cover of total greenspace within a 1000-m radius of a pond. Credible intervals are omitted for clarity. Species codes: Crisig = Crinia signifera; Geovic = Geocrinia victoriana; Limdum = Limnodynastes dumerilii; Limper = Lim. peronii; Limtas = Lim. tasmaniensis; Litewi = Litoria ewingii; Litper = Lit. peronii; Litran = Lit. raniformis; Litver = Litoria verreauxii; Parhas = Paracrinia haswelli
There were clear or influential negative relationships between the estimated probability of occupancy of C. signifera, Lim. dumerilii, Lim. tasmaniensis, Lit. peronii, Lit. verreauxii and Paracrinia haswelli and the presence of a vertical pond wall (Table 4). However, there was a strong positive relationship between pond occupancy by Lit. ewingii and a vertical pond wall (Table 4). Mean occupancy of C. signifera was predicted to decrease from 0.410 (90% BCI: 0.142–0.712) at sites without a vertical pond wall, to 0.019 (0.000–0.050) at sites with a vertical pond wall, whereas occupancy by Lit. ewingii increased from 0.681 (0.421–0.913) at sites with no vertical pond wall, to 0.877 (0.730–0.981) at sites with a vertical pond wall (Fig. 4). Limnodynastes peronii and Lit. ewingii were the only species detected at sites with a vertical pond wall.
Mean community and species-specific estimates of the probability of occupancy of 10 frog species according to the presence of a vertical pond wall at a site. Vertical bars are 90% Bayesian credible intervals. See Fig. 3 for species codes
There were mixed and ambiguous relationships between species-specific probabilities of occupancy and the presence of Gambusia, pond permanence and electrical conductivity (Table 4).
Detection probabilities
There was a clear positive relationship between the mean probability of detection across the community and air temperature (μβ2; Table 3), and clear relationships were evident for Lim. dumerilii, Lim. peronii, Lit. ewingii and P. haswelli (ESM Table S3). There was no clear relationship between mean community-level detection and either the number of days since 1 September (μβ1) or survey year (μβ3; Table 3). The only clear relationship between individual species detection and Year was evident for C. signifera, where detection was clearly higher in the second survey year (Table S3) when surveys commenced earlier in spring.
Discussion
I predicted there would be an important relationship between accessible greenspace and frog occupancy in an urban landscape, where accessible greenspace was a measure of the combined effects of habitat availability (i.e., total greenspace) and linear barriers (e.g., roads) on species occupancy. However, I found that the proportion cover of total greenspace measured at distances of 1000 and 2000 m from the pond margin was a better predictor of frog occupancy than accessible greenspace, suggesting that some species may be crossing the urban matrix and roads more successfully than initially predicted. There was a positive relationship between the cover of total greenspace and mean occupancy, highlighting the need for both landscape connectivity and terrestrial habitat to maintain amphibian communities. At the local (pond) scale, there was a strong negative relationship between the presence of a vertical pond wall at a site and mean community occupancy. The results concur with recent empirical studies highlighting the importance of urban greenspace for biodiversity in cities (Beninde et al. 2015).
The proportion cover of total greenspace was a better predictor of frog occupancy than accessible greenspace. While accessible habitat, or occupancy within barrier-based buffers, has been previously measured by assuming that highways and rivers are insurmountable barriers to the movement of amphibians (Eigenbrod et al. 2008; Hamer 2016; Zanini et al. 2008), here I assumed that main roads, railways and dense urban land also comprised an inhospitable matrix unsuitable for dispersal, thereby measuring accessible habitat at a finer spatial scale. The results therefore suggest that some species are successfully crossing the urban matrix and linear infrastructure, which is plausible given that connectivity of local amphibian populations in heterogeneous landscapes is less affected by landscape resistance than previously expected (Sinsch 2014). For instance, Limnodynastes peronii showed a neutral response to the proportion cover of total greenspace and this species adapts well to human-modified environments (Schell and Burgin 2003). Conversely, there were two ground frog species (Crinia signifera and Paracrinia haswelli) that had clearly positive relationships with total greenspace – both species were only detected at sites surrounded by large, contiguous fragments of remnant woodland, mainly in rural settings. Both species are relatively small-bodied frogs (30–35 mm; Cogger 2014) with presumably limited powers of dispersal (< 500 m) and dependent on terrestrial habitat surrounding ponds (Lauck 2005; Westgate et al. 2012), and thus would be particularly sensitive to habitat fragmentation and road effects (Rytwinski and Fahrig 2012). Tree frog species such as Litoria ewingii that were also strongly related to greenspace, can climb over small vertical barriers, and hence, are better adapted to disperse among fragmented patches of greenspace in inner city parks and gardens (i.e., urban adapters; McKinney 2002). Thus, interspecific differences in life history-traits and dispersal behaviour are likely to produce distinct patterns in occupancy of amphibian species along urban–rural gradients (Rubbo and Kiesecker 2005).
Empirical studies investigating the relative resistance to gene flow of land cover types and barriers have found that urban habitats are generally more resistant than forest cover, and highways can be 7–8 times more resistant than secondary roads (Van Buskirk 2012). However, urban areas, highways and railways are not entirely resistant to amphibian movement as has been assumed in previous studies of landscape resistance that assigned resistance costs based on empirical data and literature (Ray et al. 2002; Safner et al. 2011). For instance, while studies have reported genetic differentiation of amphibian populations within urban landscapes (Hale et al. 2013; Noël et al. 2007; Wei et al. 2021), there are also studies that demonstrated a low degree of differentiation among populations in urbanised areas (Furman et al. 2016) or no direct connection between urbanisation or roads and genetic diversity, implying there is gene flow occurring across the fragmented landscape (Homola et al. 2019). Based on occupancy patterns, my study suggests that urban land and roads are not entirely impermeable to amphibian dispersal, but likely act as filters where some species are better adapted to move across different land cover types. Rivers are the most important natural element negatively affecting connectivity for amphibians (Covarrubias et al. 2021), and so it is likely that the Yarra River represents a significant dispersal barrier in the study area. Interspecific differences in movement patterns and life-history attributes are likely to result in unique spatial patterns of genetic structuring across the study landscape (Richardson 2012), and further investigations into the relative resistance of different land cover types for amphibian movement in the study area should include genetic sampling. Further studies into accessible habitat in the study area might also find a stronger relationship if only major barriers to amphibian movement are included (e.g., highways and freeways).
The results of this study corroborate the spatial scale used in a previous analysis of species richness of frog communities in the study area, reporting a strong positive relationship between the proportion cover of greenspace measured within a 1000-m radius around a pond and the number of species whose larvae were detected at a site (Hamer and Parris 2011). I found that an increase in the proportion of greenspace at distances of 1000 – 2000 m from a pond increases the probability of occupancy at both the community and species-specific levels and demonstrates that mass-effects are shaping metacommunity structure in the study area; i.e., immigration and emigration are likely to create differences in population size (or density) in different habitat patches (Leibold et al. 2004). Even relatively small-sized frog species (e.g., Paracrinia haswelli) may depend on connectivity to greenspace and terrestrial habitat up to 1000 m from ponds (Villaseñor et al. 2017). Greenspace around ponds may facilitate amphibian movement because these ponds have greater landscape connectivity to both terrestrial and aquatic habitats, which is necessary to maintain species metapopulations (Semlitsch 2002). For example, there was a strong correlation between the proportion of greenspace and the number of neighbouring ponds within a 1000-m radius around a site (r = 0.560). Thus, the number of neighbouring ponds is likely to increase around ponds with more greenspace, thereby increasing the probability of recolonisation should local extinction occur (Parris 2018). Moreover, there was positive spatial autocorrelation among 17 ponds distributed throughout a large contiguous patch of remnant woodland on the urban fringe, suggesting there is movement of species between ponds because the probability of occupancy depended on species occupancy at neighbouring ponds. Terrestrial habitats provide foraging and shelter habitats that are critical for the local persistence of amphibian populations (Pope et al. 2000; Semlitsch 2002), and terrestrial habitats provide egg-laying sites for Geocrinia victoriana (Cogger 2014), one of three species which only occurred in remnant woodland. Indeed, the distribution of some pond-breeding amphibians is more strongly related to landscape matrix quality than to wetland availability, probably because species require access to terrestrial habitats (Quesnelle et al. 2015).
The species-sorting perspective of metacommunity theory emphasises the importance of habitat quality in structuring species assemblages within metacommunities (Leibold et al. 2004). There was a strong negative relationship between community- and species-level occupancy and the presence of a vertical pond wall at a site. Previous studies of frog metacommunities in the study area have demonstrated a strong negative relationship between species richness and vertical pond walls (Parris 2006, 2018), corroborating the results of multi-species occupancy modelling in this study. The only frog species usually capable of occupancy at these ponds are tree frogs that can climb (e.g., Lit. ewingii); however, there were two tree frog species that had negative relationships with a vertical pond wall – Lit. peronii and Lit. verreauxii – but both species have historical distributions that do not overlap with the inner area of Melbourne where these ornamental pond types were located (Hamer 2011). It is therefore apparent that the presence of a vertical pond wall excludes many frog species (e.g., Crinia signifera, Lim. dumerilii) from occupying ponds in the study area; thus, the physical structure of ponds is likely to be an important filter of species assemblages in urban landscapes (Holzer et al. 2017).
There was a strong correlation between the proportion cover of greenspace within 1000 m of a pond and the proportion cover of aquatic vegetation (r = 0.519; ESM Table S1); however, there was no support for two local-scale models containing aquatic vegetation (Table 1), although this may be due to the reduced number of variables in the models. Nonetheless, aquatic vegetation provides crucial habitat complexity as shelter and foraging habitat for both frogs and tadpoles in the study area (Hamer and Parris 2011; Parris 2018), which may reduce the impact of fish predation on larvae (Hamer and Parris 2013). Despite the presence of Gambusia at a pond outranking pond area as a covariate in the MSOM, there were ambiguous relationships between Gambusia and the probability of occupancy, mirroring correlates of frog species richness in the study area (Hamer and Parris 2011). Similarly, there were ambiguous relationships between occupancy and pond permanence, although the lack of a relationship with hydroperiod might be due to a sampling bias towards permanent ponds (Hamer and Parris 2011). While increased water conductivity is generally associated with decreased species richness in the study area (Hamer and Parris 2011), there were no clear relationships with occupancy.
Application of MSOM in this study enabled clear predictions of habitat relationships for rare species detected at few sites (e.g., Paracrinia haswelli), thereby highlighting the utility of MSOM in the management of both common and rare species in urban areas. These models offer a powerful means of guiding the management of both individual species and entire community assemblages of species with similar ecological requirements (Zipkin et al. 2010) and adopting a community-level hierarchical modelling approach to species conservation in urban landscapes should be encouraged (e.g., Mata et al. 2014; Threlfall et al. 2017). While rare species detected in this study were only recorded at species-rich ponds, accounting for imperfect species detection and use of collective community data enabled inferences to be derived for these species and should improve future estimates of species richness, a response variable which has been recommended for monitoring both trends in frog communities and management actions in the greater Melbourne area (Hamer and Parris 2011). Rare species often show disproportionate responses to habitat change compared to common species (Zipkin et al. 2010), are frequently urban-sensitive species (McKinney 2002), and therefore worthy of attention for conservation management actions in urbanising landscapes.
In conclusion, this study adds to the growing body of evidence demonstrating the crucial importance of greenspace for maintaining biodiversity in urban landscapes. Increasing the area and connectivity of greenspaces are the most critical elements in promoting high levels of urban biodiversity, although habitat structure is also important (Beninde et al. 2015; Matthies et al. 2017; Nielsen et al. 2014). Amphibian communities, like other wetland-dependent taxa (e.g., freshwater turtles; Guzy et al. 2013), are no exception, and metapopulations require interconnections between and among wetlands and terrestrial habitats for persistence (Heard et al. 2012; Semlitsch 2002). Urban green spaces often support both high understorey vegetation cover and aquatic vegetation cover that is important for local biodiversity (Hamer and Parris 2011; Threlfall et al. 2017). It is therefore evident that management actions that increase vegetation cover, improve pond structure (e.g., remove vertical walls) and increase pond density and connectivity up to a radius of 2000 m around ponds, and eliminate non-native predatory fish, should therefore be undertaken in urban greenspaces to benefit a broad array of wetland-dependent organisms (Oertli and Parris 2019).
Availability of data and material
All data analysed during this study are included in this published article and its supplementary information files.
Code availability
Model code is available by contacting AJH.
References
Anderson DR (2008) Model based inference in the life sciences: a primer on evidence. Springer Science+Business Media, New York, New York, USA
Aronson MF, Lepczyk CA, Evans KL, Goddard MA, Lerman SB, MacIvor JS, Nilon CH, Vargo T (2017) Biodiversity in the city: key challenges for urban green space management. Front Ecol Environ 15:189–196. https://doi.org/10.1002/fee.1480
Australian Research Centre for Urban Ecology (2003) Open Space 2002 (ArcView Shapefile). Australian Research Centre for Urban Ecology, Melbourne, Australia
Beebee TJC (2013) Effects of road mortality and mitigation measures on amphibian populations. Conserv Biol 27:657–668. https://doi.org/10.1111/cobi.12063
Beninde J, Veith M, Hochkirch A (2015) Biodiversity in cities needs space: a meta-analysis of factors determining intra-urban biodiversity variation. Ecol Lett 18:581–592. https://doi.org/10.1111/ele.12427
Brooks SP, Gelman A (1998) General methods for monitoring convergence of iterative simulations. J Comput Graph Stat 7:434–455
Canessa S, Heard GW, Parris KM, McCarthy MA (2012) Integrating variability in detection probabilities when designing wildlife surveys: a case study of amphibians from south-eastern Australia. Biodivers Conserv 21:729–744. https://doi.org/10.1007/s10531-011-0211-0
Cogger HG (2014) Reptiles and Amphibians of Australia, 7th edn. CSIRO Publishing, Victoria, Australia
Covarrubias S, González C, Gutiérrez-Rodríguez C (2021) Effects of natural and anthropogenic features on functional connectivity of anurans: a review of landscape genetics studies in temperate, subtropical and tropical species. J Zool 313:159–171. https://doi.org/10.1111/jzo.12851
Crump ML, Scott NJ Jr (1994) Visual encounter surveys. In: Heyer WR, Donnelly MA, McDiarmid RW, Hayek LC, Foster MS (eds) Measuring and monitoring biological diversity. Standard methods for amphibians. Smithsonian Institution Press, Washington D.C., pp 84–92
Czech B, Krausman PR, Devers PK (2000) Economic associations among causes of species endangerment in the United States. Bioscience 50:593–601
Dorazio RM, Royle JA (2005) Estimating size and composition of biological communities by modeling the occurrence of species. J Am Stat Assoc 100:389–398. https://doi.org/10.1198/016214505000000015
Dorazio RM, Royle JA, Söderström B, Glimskär A (2006) Estimating species richness and accumulation by modeling species occurrence and detectability. Ecology 87:842–854. https://doi.org/10.1890/0012-9658(2006)87[842:esraab]2.0.co;2
Eigenbrod F, Hecnar SJ, Fahrig L (2008) Accessible habitat: an improved measure of the effects of habitat loss and roads on wildlife populations. Landsc Ecol 23:159–168. https://doi.org/10.1007/s10980-007-9174-7
Furman BLS, Scheffers BR, Taylor M, Davis C, Paszkowski CA (2016) Limited genetic structure in a wood frog (Lithobates sylvaticus) population in an urban landscape inhabiting natural and constructed wetlands. Conserv Genet 17:19–30. https://doi.org/10.1007/s10592-015-0757-6
Fuyuki A, Yamaura Y, Nakajima Y, Ishiyama N, Akasaka T, Nakamura F (2014) Pond area and distance from continuous forests affect amphibian egg distributions in urban green spaces: A case study in Sapporo, Japan. Urban For Urban Green 13:397–402. https://doi.org/10.1016/j.ufug.2013.11.003
Gagné SA, Fahrig L (2007) Effect of landscape context on anuran communities in breeding ponds in the National Capital Region, Canada. Landsc Ecol 22:205–215
Gelman A, Hwang J, Vehtari A (2014) Understanding predictive information criteria for Bayesian models. Stat Comput 24:997–1016. https://doi.org/10.1007/s11222-013-9416-2
Gelman A, Meng X-L, Stern H (1996) Posterior predictive assessment of model fitness via realized discrepancies. Stat Sin 6:733–760
Gelman A, Rubin DB (1992) Inference from iterative simulation using multiple sequences. Stat Sci 7:457–472
Green J, Govindarajulu P, Higgs E (2021) Multiscale determinants of Pacific chorus frog occurrence in a developed landscape. Urban Ecosyst 24:587–600. https://doi.org/10.1007/s11252-020-01057-4
Grimm NB, Faeth SH, Golubiewski NE, Redman CL, Wu J, Bai X, Briggs JM (2008) Global change and the ecology of cities. Science 319:756–760. https://doi.org/10.1126/science.1150195
Guderyahn LB, Smithers AP, Mims MC (2016) Assessing habitat requirements of pond-breeding amphibians in a highly urbanized landscape: implications for management. Urban Ecosyst 19:1801–1821. https://doi.org/10.1007/s11252-016-0569-6
Guillera-Arroita G, Kéry M, Lahoz-Monfort JJ (2019) Inferring species richness using multispecies occupancy modeling: Estimation performance and interpretation. Ecol Evol 9:780–792. https://doi.org/10.1002/ece3.4821
Guzy JC, Price SJ, Dorcas ME (2013) The spatial configuration of greenspace affects semi-aquatic turtle occupancy and species richness in a suburban landscape. Landsc Urban Plann 117:46–56. https://doi.org/10.1016/j.landurbplan.2013.04.011
Haaland C, van den Bosch CK (2015) Challenges and strategies for urban green-space planning in cities undergoing densification: A review. Urban For Urban Green 14:760–771. https://doi.org/10.1016/j.ufug.2015.07.009
Hahs AK, McDonnell MJ, McCarthy MA, Vesk PA, Corlett RT, Norton BA, Clemants SE, Duncan RP, Thompson K, Schwartz MW, Williams NSG (2009) A global synthesis of plant extinction rates in urban areas. Ecol Lett 12:1165–1173. https://doi.org/10.1111/j.1461-0248.2009.01372.x
Hale JM, Heard GW, Smith KL, Parris KM, Austin JJ, Kearney M, Melville J (2013) Structure and fragmentation of growling grass frog metapopulations. Conserv Genet 14:313–322. https://doi.org/10.1007/s10592-012-0428-9
Hamer AJ (2011) The herpetofauna of Melbourne: using past and present distributions to assess impacts of urbanisation. Vic Nat 128:162–173
Hamer AJ (2016) Accessible habitat delineated by a highway predicts landscape-scale effects of habitat loss in an amphibian community. Landsc Ecol 31:2259–2274. https://doi.org/10.1007/s10980-016-0398-2
Hamer AJ (2018) Accessible habitat and wetland structure drive occupancy dynamics of a threatened amphibian across a peri-urban landscape. Landsc Urban Plann 178:228–237
Hamer AJ, Harrison LJ, Stokeld D (2018) Terrestrial habitat and individual fitness increase survival of a freshwater turtle in an urban landscape. Urban Ecosyst 21:71–83. https://doi.org/10.1007/s11252-017-0708-8
Hamer AJ, Langton TES, Lesbarrerès D (2015) Making a safe leap forward: Mitigating road impacts on amphibians. In: van der Ree R, Smith DJ, Grilo C (eds) Handbook of road ecology. John Wiley & Sons, Chichester, pp 261–270
Hamer AJ, McDonnell MJ (2008) Amphibian ecology and conservation in the urbanising world: A review. Biol Conserv 141:2432–2449. https://doi.org/10.1016/j.biocon.2008.07.020
Hamer AJ, McDonnell MJ (2010) The response of herpetofauna to urbanization: Inferring patterns of persistence from wildlife databases. Austral Ecol 35:568–580. https://doi.org/10.1111/j.1442-9993.2009.02068.x
Hamer AJ, Parris KM (2011) Local and landscape determinants of amphibian communities in urban ponds. Ecol Appl 21:378–390. https://doi.org/10.1890/10-0390.1
Hamer AJ, Parris KM (2013) Predation modifies larval amphibian communities in urban wetlands. Wetlands 33:641–652. https://doi.org/10.1007/s13157-013-0420-2
Harrison XA, Donaldson L, Correa-Cano ME, Evans J, Fisher DN, Goodwin CED, Robinson BS, Hodgson DJ, Inger R (2018) A brief introduction to mixed effects modelling and multi-model inference in ecology. PeerJ 6:e4794. https://doi.org/10.7717/peerj.4794
Heard GW, Scroggie MP, Malone BS (2012) Classical metapopulation theory as a useful paradigm for the conservation of an endangered amphibian. Biol Conserv 148:156–166. https://doi.org/10.1016/j.biocon.2012.01.018
Hero J-M, Littlejohn M, Marantelli G (1991) Frogwatch field guide to Victorian frogs. Department of Conservation and Environment, East Melbourne, Australia
Holzer KA, Bayers RP, Nguyen TT, Lawler SP (2017) Habitat value of cities and rice paddies for amphibians in rapidly urbanizing Vietnam. J Urban Ecol 3:juw007–juw007. https://doi.org/10.1093/jue/juw007
Homola JJ, Loftin CS, Kinnison MT (2019) Landscape genetics reveals unique and shared effects of urbanization for two sympatric pool-breeding amphibians. Ecol Evol 9:11799–11823. https://doi.org/10.1002/ece3.5685
Hutto D Jr, Barrett K (2021) Do urban open spaces provide refugia for frogs in urban environments? PLoS One 16:e0244932. https://doi.org/10.1371/journal.pone.0244932
Ives CD, Beilin R, Gordon A, Kendal D, Hahs AK, McDonnell MJ (2013) Local assessment of Melbourne: the biodiversity and social-ecological dynamics of Melbourne, Australia. In: Elmqvist T, Fragkias M, Goodness J, Güneralp B, Marcotullio PJ, McDonald RI, Parnell S, Schewenius M, Sendstad M, Seto KC, Wilkinson C (eds) Urbanization, biodiversity and ecosystem services: challenges and opportunities: a global assessment. Springer, Dordrecht, The Netherlands, pp 385–407
Joly P, Morand C, Cohas A (2003) Habitat fragmentation and amphibian conservation: building a tool for assessing landscape matrix connectivity. C R Biol 326:S132–S139
Kéry M, Royle JA (2008) Hierarchical Bayes estimation of species richness and occupancy in spatially replicated surveys. J Appl Ecol 45:589–598. https://doi.org/10.1111/j.1365-2664.2007.01441.x
Kowarik I (2011) Novel urban ecosystems, biodiversity, and conservation. Environ Pollut 159:1974–1983. https://doi.org/10.1016/j.envpol.2011.02.022
Kruger DJD, Hamer AJ, Du Preez LH (2015) Urbanization affects frog communities at multiple scales in a rapidly developing African city. Urban Ecosyst 18:1333–1352. https://doi.org/10.1007/s11252-015-0443-y
Lauck B (2005) The impact of recent logging and pond isolation on pond colonization by the frog Crinia signifera. Pac Conserv Biol 11:50–56
Leibold MA, Holyoak M, Mouquet N, Amarasekare P, Chase JM, Hoopes MF, Holt RD, Shurin JB, Law R, Tilman D, Loreau M, Gonzalez A (2004) The metacommunity concept: a framework for multi-scale community ecology. Ecol Lett 7:601–613
Lepczyk CA, Aronson MFJ, Evans KL, Goddard MA, Lerman SB, MacIvor JS (2017) Biodiversity in the city: Fundamental questions for understanding the ecology of urban green spaces for biodiversity conservation. Bioscience 67:799–807. https://doi.org/10.1093/biosci/bix079
Li B, Zhang W, Shu X, Pei E, Yuan X, Wang T, Wang Z (2018) Influence of breeding habitat characteristics and landscape heterogeneity on anuran species richness and abundance in urban parks of Shanghai, China. Urban For Urban Green 32:56–63. https://doi.org/10.1016/j.ufug.2018.03.017
Link WA (1999) Modeling pattern in collections of parameters. J Wildl Manag 63:1017–1027. https://doi.org/10.2307/3802817
Marsh DM, Cosentino BJ, Jones KS, Apodaca JJ, Beard KH, Bell JM, Bozarth C, Carper D, Charbonnier JF, Dantas A, Forys EA, Foster M, General J, Genet KS, Hanneken M, Hess KR, Hill SA, Iqbal F, Karraker NE, Kilpatrick ES, Langen TA, Langford J, Lauer K, McCarthy AJ, Neale J, Patel S, Patton A, Southwick C, Stearrett N, Steijn N, Tasleem M, Taylor JM, Vonesh JR, Green D (2017) Effects of roads and land use on frog distributions across spatial scales and regions in the Eastern and Central United States. Divers Distrib 23:158–170. https://doi.org/10.1111/ddi.12516
Mata L, Goula M, Hahs AK (2014) Conserving insect assemblages in urban landscapes: accounting for species-specific responses and imperfect detection. J Insect Conserv 18:885–894. https://doi.org/10.1007/s10841-014-9696-7
Matthies SA, Rüter S, Schaarschmidt F, Prasse R (2017) Determinants of species richness within and across taxonomic groups in urban green spaces. Urban Ecosyst 20:897–909. https://doi.org/10.1007/s11252-017-0642-9
McDonnell MJ, Holland K (2008) Biodiversity. In: Newton PW (ed) Transitions: Transitioning to resilient cities. CSIRO Publishing, Melbourne, Victoria, Australia, pp 253–266
McKinney ML (2002) Urbanization, biodiversity, and conservation. Bioscience 52:883–890
Mihaljevic JR, Joseph MB, Johnson PTJ (2015) Using multispecies occupancy models to improve the characterization and understanding of metacommunity structure. Ecology 96:1783–1792. https://doi.org/10.1890/14-1580.1
Nielsen AB, van den Bosch M, Maruthaveeran S, van den Bosch CK (2014) Species richness in urban parks and its drivers: A review of empirical evidence. Urban Ecosyst 17:305–327. https://doi.org/10.1007/s11252-013-0316-1
Noël S, Ouellet M, Galois P, Lapointe FJ (2007) Impact of urban fragmentation on the genetic structure of the eastern red-backed salamander. Conserv Genet 8:599–606
Norton BA, Evans KL, Warren PH (2016) Urban biodiversity and landscape ecology: patterns, processes and planning. Curr Landsc Ecol Rep 1:178–192. https://doi.org/10.1007/s40823-016-0018-5
Oertli B, Parris KM (2019) Review: Toward management of urban ponds for freshwater biodiversity. Ecosphere 10:e02810. https://doi.org/10.1002/ecs2.2810
Pacifici K, Zipkin EF, Collazo JA, Irizarry JI, DeWan A (2014) Guidelines for a priori grouping of species in hierarchical community models. Ecol Evol 4:877–888. https://doi.org/10.1002/ece3.976
Parris KM (2006) Urban amphibian assemblages as metacommunities. J Anim Ecol 75:757–764
Parris KM (2018) Existing ecological theory applies to urban environments. Landsc Ecol Eng 14:201–208. https://doi.org/10.1007/s11355-018-0351-4
Phillott AD, Speare R, Hines HB, Skerratt LF, Meyer E, McDonald KR, Cashins SD, Mendez D, Berger L (2010) Minimising exposure of amphibians to pathogens during field studies. Dis Aquat Org 92:175–185. https://doi.org/10.3354/dao02162
Plummer M (2013) JAGS Version 3.4.0 user manual. https://sourceforge.net/projects/mcmc-jags/files/Manuals/
Pope SE, Fahrig L, Merriam HG (2000) Landscape complementation and metapopulation effects on leopard frog populations. Ecology 81:2498–2508
Pyke GH, White AW (2000) Factors influencing predation on eggs and tadpoles of the endangered green and golden bell frog Litoria aurea by the introduced plague minnow Gambusia holbrooki. Aust Zool 31:496–505
QGIS Geographic Information System (2020) Open Source Geospatial Foundation Project, version 3.4.9. http://qgis.osgeo.org
Quesnelle PE, Lindsay KE, Fahrig L (2015) Relative effects of landscape-scale wetland amount and landscape matrix quality on wetland vertebrates: a meta-analysis. Ecol Appl 25:812–825. https://doi.org/10.1890/14-0362.1
R Core Team (2019) R: A language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria. https://www.R-project.org/
Ray N, Lehmann A, Joly P (2002) Modeling spatial distribution of amphibian populations: a GIS approach based on habitat matrix permeability. Biodivers Conserv 11:2143–2165
Richardson JL (2012) Divergent landscape effects on population connectivity in two co-occurring amphibian species. Mol Ecol 21:4437–4451. https://doi.org/10.1111/j.1365-294X.2012.05708.x
Rubbo MJ, Kiesecker JM (2005) Amphibian breeding distribution in an urbanized landscape. Conserv Biol 19:504–511
Rytwinski T, Fahrig L (2012) Do species life history traits explain population responses to roads? A meta-analysis. Biol Conserv 147:87–98. https://doi.org/10.1016/j.biocon.2011.11.023
Safner T, Miaud C, Gaggiotti O, Decout S, Rioux D, Zundel S, Manel S (2011) Combining demography and genetic analysis to assess the population structure of an amphibian in a human-dominated landscape. Conserv Genet 12:161–173. https://doi.org/10.1007/s10592-010-0129-1
Schell CB, Burgin S (2003) Swimming against the current: the brown striped marsh frog Limnodynastes peronii success story. Aust Zool 32:401–405
Schielzeth H (2010) Simple means to improve the interpretability of regression coefficients. Methods Ecol Evol 1:103–113. https://doi.org/10.1111/j.2041-210X.2010.00012.x
Scott NJ Jr, Woodward BD (1994) Surveys at breeding sites. In: Heyer WR, Donnelly MA, McDiarmid RW, Hayek LC, Foster MS (eds) Measuring and monitoring biological diversity. Standard methods for amphibians. Smithsonian Institution Press, Washington D.C., pp 118–125
Semlitsch RD (2002) Critical elements for biologically based recovery plans of aquatic-breeding amphibians. Conserv Biol 16:619–629
Seto KC, Fragkias M, Güneralp B, Reilly MK (2011) A meta-analysis of global urban land expansion. PLoS One 6:e23777. https://doi.org/10.1371/journal.pone.0023777
Shochat E, Lerman SB, Anderies JM, Warren PS, Faeth SH, Nilon CH (2010) Invasion, competition, and biodiversity loss in urban ecosystems. Bioscience 60:199–208. https://doi.org/10.1525/bio.2010.60.3.6
Sinsch U (2014) Movement ecology of amphibians: from individual migratory behaviour to spatially structured populations in heterogeneous landscapes. Can J Zool 92:491–502. https://doi.org/10.1139/cjz-2013-0028
Smallbone LT, Luck GW, Wassens S (2011) Anuran species in urban landscapes: Relationships with biophysical, built environment and socio-economic factors. Landsc Urban Plann 101:43–51. https://doi.org/10.1016/j.landurbplan.2011.01.002
Snodgrass JW, Bryan L Jr, Burger J (2000) Development of expectations of larval amphibian assemblage structure in southeastern depression wetlands. Ecol Appl 10:1219–1229
Stern H (2005) Climate. In: Brown-May A, Swain S (eds) The encyclopedia of Melbourne. Cambridge University Press, Melbourne, Australia, pp 147–154
Stolen ED, Oddy DM, Gann SL, Holloway-Adkins KG, Legare SA, Weiss SK, Breininger DR (2019) Accounting for heterogeneity in false-positive detection rate in southeastern beach mouse habitat occupancy models. Ecosphere 10:e02893. https://doi.org/10.1002/ecs2.2893
Straka TM, Lentini PE, Lumsden LF, Wintle BA, van der Ree R (2016) Urban bat communities are affected by wetland size, quality, and pollution levels. Ecol Evol 6:4761–4774. https://doi.org/10.1002/ece3.2224
Su Y, Yajima M (2015) Package ‘R2jags’: using R to run ‘JAGS’ Version 05–7
Sutherland RW, Dunning PR, Baker WM (2010) Amphibian encounter rates on roads with different amounts of traffic and urbanization. Conserv Biol 24:1626–1635. https://doi.org/10.1111/j.1523-1739.2010.01570.x
Theobald DM, Miller JR, Hobbs NT (1997) Estimating the cumulative effects of development on wildlife habitat. Landsc Urban Plann 39:25–36. https://doi.org/10.1016/S0169-2046(97)00041-8
Thornhill I, Batty L, Death RG, Friberg NR, Ledger ME (2017) Local and landscape scale determinants of macroinvertebrate assemblages and their conservation value in ponds across an urban land-use gradient. Biodivers Conserv 26:1065–1086. https://doi.org/10.1007/s10531-016-1286-4
Threlfall CG, Mata L, Mackie JA, Hahs AK, Stork NE, Williams NSG, Livesley SJ (2017) Increasing biodiversity in urban green spaces through simple vegetation interventions. J Appl Ecol 54:1874–1883. https://doi.org/10.1111/1365-2664.12876
Trombulak SC, Frissell CA (2000) Review of ecological effects of roads on terrestrial and aquatic communities. Conserv Biol 14:18–30
Urban MC, Roehm R (2018) The road to higher permanence and biodiversity in exurban wetlands. Oecologia 186:291–302. https://doi.org/10.1007/s00442-017-3989-y
Van Buskirk J (2012) Permeability of the landscape matrix between amphibian breeding sites. Ecol Evol 2:3160–3167. https://doi.org/10.1002/ece3.424
Villaseñor NR, Driscoll DA, Gibbons P, Calhoun AJK, Lindenmayer DB (2017) The relative importance of aquatic and terrestrial variables for frogs in an urbanizing landscape: Key insights for sustainable urban development. Landsc Urban Plann 157:26–35. https://doi.org/10.1016/j.landurbplan.2016.06.006
Vitousek PM, Mooney HA, Lubchenco J, Melillo JM (1997) Human domination of earth’s ecosystems. Science 277:494–499. https://doi.org/10.1126/science.277.5325.494
Watanabe S (2010) Asymptotic equivalence of Bayes cross validation and widely applicable information criterion in singular learning theory. J Mach Learn Res 11:3571–3594
Wei X, Huang M, Yue Q, Ma S, Li B, Mu Z, Peng C, Gao W, Liu W, Zheng J, Weng X, Sun X, Zuo Q, Bo S, Yuan X, Zhang W, Yang G, Ding Y, Wang X, Wang T, Hua P, Wang Z (2021) Long-term urbanization impacts the eastern golden frog (Pelophylax plancyi) in Shanghai City: Demographic history, genetic structure, and implications for amphibian conservation in intensively urbanizing environments. Evol Appl 14:117–135. https://doi.org/10.1111/eva.13156
Wellborn GA, Skelly DK, Werner EE (1996) Mechanisms creating community structure across a freshwater habitat gradient. Annu Rev Ecol Syst 27:337–363
Westgate MJ, Driscoll DA, Lindenmayer DB (2012) Limited influence of stream networks on the terrestrial movements of three wetland-dependent frog species. Biol Conserv 153:169–176. https://doi.org/10.1016/j.biocon.2012.04.030
Wintle BA, Bardos DC (2006) Modeling species-habitat relationships with spatially autocorrelated observation data. Ecol Appl 16:1945–1958
Zanini F, Klingemann A, Schlaepfer R, Schmidt BR (2008) Landscape effects on anuran pond occupancy in an agricultural countryside: barrier-based buffers predict distributions better than circular buffers. Can J Zool 86:692–699. https://doi.org/10.1139/z08-048
Zipkin EF, Andrew Royle J, Dawson DK, Bates S (2010) Multi-species occurrence models to evaluate the effects of conservation and management actions. Biol Conserv 143:479–484. https://doi.org/10.1016/j.biocon.2009.11.016
Zipkin EF, DeWan A, Andrew Royle J (2009) Impacts of forest fragmentation on species richness: a hierarchical approach to community modelling. J Appl Ecol 46:815–822. https://doi.org/10.1111/j.1365-2664.2009.01664.x
Acknowledgements
I thank staff from the Royal Botanic Gardens Victoria, Royal Botanic Gardens Cranbourne, University of Melbourne, Melbourne City Council and other local council staff for facilitating access to ponds. Kirsten Parris provided guidance on site selection and frog surveys. Amy Hahs provided access to GIS spatial layers. Geoff Heard and Eliza Poole assisted with fieldwork. The Baker Foundation provided generous support for fieldwork. Data analysis and writing was supported by the Balaton Limnological Research Institute. Fieldwork was approved by the University of Melbourne Animal Ethics Committee (register no. 0706488) and was conducted under research permit no. 10004319 (Victorian Department of Sustainability and Environment).
Funding
Funding for this research was provided by The Baker Foundation.
Author information
Authors and Affiliations
Contributions
AJH designed the study, acquired funding, collected and analysed the data, and wrote the manuscript.
Corresponding author
Ethics declarations
Ethics approval
Fieldwork was approved by the University of Melbourne Animal Ethics Committee (register no. 0706488) and was conducted under research permit no. 10004319 (Victorian Department of Sustainability and Environment).
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Conflicts of interest/Competing interests
There are no conflicts of interest or competing interests to declare.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Hamer, A.J. A multi-scale, multi-species approach highlights the importance of urban greenspace and pond design for amphibian communities. Urban Ecosyst 25, 393–409 (2022). https://doi.org/10.1007/s11252-021-01162-y
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11252-021-01162-y