Abstract
Management of urban aquatic habitats for native wildlife, such as amphibians, is an important contemporary goal for many municipalities. However, our understanding of how local and landscape characteristics of urban aquatic habitat promote or inhibit amphibian occupancy and recruitment is limited. In this study, we examined amphibian community composition and occurrence patterns in ponds, wetlands, and swales of Gresham, Oregon. We collected occurrence data for five native amphibians: northwestern salamander (Ambystoma gracile), long-toed salamander (A. macrodactylum), Pacific chorus frog (Pseudacris regilla), northern red-legged frog (Rana aurora aurora), and rough-skinned newt (Taricha granulosa) as well as one non-native amphibian, the American bullfrog (Lithobates catesbeianus). One hundred sites were surveyed from 2007 to 2013. Local and landscape attributes were characterized for each site, and potential drivers of species occupancy were evaluated using a combination of multivariate approaches and generalized linear models. In general, percent impervious surface and distance to nearest forest patch, both associated with urbanization, were negatively correlated with site occupancy for all species. Non-native vegetation was also negatively associated with occupancy of three species (long-toed salamanders, Pacific chorus frogs, and northern red-legged frogs). In contrast, occupancy was positively correlated with pond depth and hydroperiod length for all species. We found evidence of two distinct groups of co-occurring amphibian species driven by habitat depth and hydroperiod. Finally, we report results of threshold analyses that examined species-specific habitat associations. This study describes urban habitat associations of a native amphibian community, identifies factors with positive, negative or mixed relationships with amphibian species, and is an important step in informing the management of urban aquatic habitat to promote persistence of native amphibians.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Human modification of the landscape for agricultural and developmental purposes has resulted in increasingly modified and isolated aquatic habitats worldwide, often to the detriment of native flora and fauna. As a result, urbanization is recognized as a major global threat to species diversity (Czech et al. 2000; Foley et al. 2005; Scheffers and Paszkowski 2012). Species with complex habitat requirements and life cycles, such as pond-breeding amphibians, are especially vulnerable to habitat loss and degradation (Becker et al. 2007). Wetlands and wet prairies in the Willamette Valley of the Pacific Northwest have declined significantly over the last 160 years (Titus et al. 1996). Depending on wetland type, historical cover has declined by 57 to 99.5 % since 1850 (ORBIC 2013). In urban areas, natural aquatic habitats are often isolated or are altered enough that their value as habitat for native wildlife is unclear (Vitousek et al. 1996; McKinney 2002). In addition, manmade aquatic habitats such as stormwater retention ponds and water treatment facilities are often required features of new developments and may offer important surrogate habitat to many native aquatic species, particularly as they may be the only still-water habitats available in highly modified environments (Scheffers and Paszkowski 2013). However, researchers have only recently begun to explore the role of manmade and altered natural aquatic habitats in the preservation of amphibian populations across urban landscapes (Birx-Raybuck et al. 2010; Brand and Snodgrass 2010; Hamer and Parris 2011). Characteristics such as water depth, hydroperiod, plant diversity and availability of refugia are important for a range of aquatic species but are rarely considered during the design and construction of facilities (where storing and filtering stormwater is the primary goal) or the management of remnant wetlands and ponds (Brand et al. 2010; Brown et al. 2012). Some urban aquatic habitats can even act as “ecological sinks” (McCarthy 2009) that attract breeding adults but fail to recruit juveniles due to inadequate hydrology or vegetative cover (Richter and Azous 2000). In addition, connectivity between aquatic and terrestrial habitats has been shown to be crucial for the survival and persistence of many amphibian species (Pope et al. 2000; Semlitsch and Bodie 2003). Such connectivity may be limited in highly urbanized environments.
Recently, some municipalities have begun including the creation of wildlife habitat as an equally important goal when designing stormwater facilities or mitigating impacts of city construction projects on aquatic habitats (e.g. City of Chicago 2006; City of Seattle 2015). Understanding how biotic and abiotic habitat characteristics affect amphibian communities in highly urbanized environments is critical for informing project designs that promote healthy amphibian populations, yet few studies have examined those relationships. The objective of this study was to further the current understanding of the types of urban aquatic habitats used by amphibians and identify patterns and potential environmental drivers for species occupancy over a suite of both within-wetland and landscape variables in Gresham, Oregon. The results from this study will help inform land management strategies that aim to incorporate the habitat needs of native amphibians at both the community and species level.
Materials and methods
Study area
This study was conducted within the city limits of Gresham, Oregon, USA (Multnomah County). Gresham is the fourth largest city in Oregon with a population of 109,000 people (US Census Bureau 2007) and is part of the greater metropolitan area of Portland, Oregon. Gresham is roughly 60.9 km2 with 41 % impervious or “built” surface. The local climate is cool-summer Mediterranean with an average rainfall of 110 cm mostly occurring between November and March. Average monthly temperatures range from 8 C (December) to 28 C (July and August).
Site selection
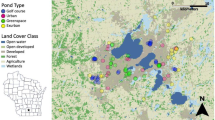
In Gresham, breeding habitat for aquatic amphibians is primarily concentrated in floodplain pockets near streams or in isolated stormwater facilities and ponds within developed areas. In order to assemble a comprehensive list of all aquatic habitats available to breeding amphibians in Gresham, we initially evaluated 234 ponds, wetlands and swales identified from the Fish and Wildlife Service’s 2003 National Wetland Inventory, the Portland-METRO region’s 2003 Local Wetland Inventory, a Gresham Watershed Division database of manmade ponds, water quality facilities, and swales (see Appendix 1 for database information; all spatial data were managed and queried in a geographic information system - ESRI 2011), and field investigations. We sought permission to access any sites identified on private lands. Over a 7-year period, we surveyed each site from 1 to 7 years each. Due to logistical constraints, not all sites were visited each year. Sites surveyed less than 3 years were discarded from analyses presented in this study. Sites were categorized as either “man-made” (stormwater ponds and swales built specifically for the detention and/or treatment of stormwater runoff) or “natural” (historical groundwater or floodplain wetlands that according to aerial imagery have been present since at least 1936 – the earliest aerial imagery available to us for analysis; Fig. 1).
Amphibian sampling
Our study included five native pond-breeding amphibian species found in the Gresham area: the northwestern salamander (Ambystoma gracile), the long-toed salamander (A. macrodactylum), the Pacific chorus frog (Pseudacris regilla), the northern red-legged frog (Rana aurora aurora), the rough-skinned newt (Taricha granulosa) and one non-native amphibian, the American bullfrog (Lithobates catesbeianus).
Egg mass surveys
Sites were surveyed for egg masses every 3–4 weeks between late January and mid-April, the primary breeding season of species in the region. We searched for egg masses near the edges of standing water and within vegetation in water up to 150 cm deep using protocols described in Corkran and Thoms (2006). All egg masses for each species were recorded; in addition, any adults seen or heard during field sampling were recorded.
Larval surveys
Larval surveys were also conducted every 3–4 weeks between late April and mid-July, or until the site dried for the season. At each larval survey visit, direct observations of free-swimming larvae were recorded and identified to species. In addition, a D-frame dip-net (30 cm diameter; 500 μm mesh) was used to sample each microhabitat within a site. All amphibians and fish were identified, counted and released. While American bullfrogs do not lay eggs until June/July in the study area, tadpoles often persist for 1 to 2 years at a site; thus, larval surveys paired with calling surveys remain a good method of determining the presence of American bullfrog breeding populations at a site.
Habitat characteristics
Each site was characterized by eight local variables describing the physical and biotic habitat within 10 m of the water’s edge. These variables were chosen due to their ease of collection, comparability across study systems, and their recognized importance for amphibian occurrence. We measured: pond age, aquatic footprint, percent shoreline cover, depth, presence of fish, percent floating and submerged vegetation, hydroperiod, and percent cover by non-native plants at each site. Age (Age) of built facilities was determined through reviewing as-built data collected from the Gresham Stormwater Division for created facilities. Examination of historical aerial photographs was used to determine the age of natural wetlands to a maximum of 78 years, because aerial photographs with coverage of the entire city dated 1935 are the earliest we were able to obtain. We determined aquatic habitat footprint (in m2; Area) as ordinary high water levels for each of selected sites using GIS shapefiles of aerial photographs from 2010 resolved to 1:2000. Total annual precipitation (TAP) in 2010 was representative of the TAP across the range of years covered by this study (26.71” total precipitation in 2013, 50.43” in 2012, and 37.09” in 2011, 46.18” in 2010, 30.50” in 2009, 27.12” in 2008, and 32.06” in 2007; NOAA 2016), and repeated site visits indicated that the aquatic habitat footprint did not significantly change over the 2007–2013 study interval. Percent shoreline cover (Cover) represents an ocular estimate of the amount of the shoreline within 10 m of the water’s edge covered by any material that could be used as refugia by amphibians moving to or away from a site (for example, rocks, logs, sticks and debris). Maximum pond depth at the deepest point (Depth) was measured during each amphibian sampling visit and then averaged for each site to produce an average depth variable. Presence of fish was recorded during egg mass and larval surveys (Fish). Percent floating and submerged vegetation (FloatSubVeg) represents an ocular estimate of the area of the aquatic footprint covered by these types of vegetation. Hydroperiod (HydroP) was calculated as the average number of days that the site held any amount of water from January 1 to July 15th (a maximum of 196 days); we rarely visited a site after July 15th in a given year. Finally, percent cover by non-native vegetation (NonNatVeg) represents an ocular estimate of the area within 10 m of the water’s edge covered by any non-native plant species.
Landscape characteristics
Site boundaries were determined from ordinary high water levels using the same procedure as when determining the aquatic footprint of the sites (described above). We quantified ten landscape-scale variables using land-use data and aerial photography (see Appendix 1 for details): percent total impervious surface (e.g. road or building) within a 200 m buffer (Built200); percent total impervious surface within a 1000 m buffer (Built1K); distance to nearest forest patch of sizes 50 m2 (For50), 200 m2 (For200), 500 m2 (For500), 1000 m2 (For1K), 2500 m2 (For2.5 K), and 5000 m2 (For5K); distance to other known wetland or pond site (NearSite) and distance to nearest perennial stream (NearStream). We characterized impervious surface and vegetative cover within both a 200 m and a 1000 m buffer around each site, because recent studies have determined that landscape characterization at these scales may provide the best prediction for species occupancy (Houlahan and Findlay 2003; Skidds et al. 2007; Ostergaard et al. 2008). Percent impervious surface (Built200 and Built1K) was calculated using regional tax lot and land use data (see Appendix 1 for dataset information). Distances from each site to the nearest pond and stream were calculated using straight line measurements derived from 2010 aerial photography (scale 1:2000). Finally, several of our target species are known to be dependent on forested habitats for aestivation and overwintering (Leonard et al. 1993; Corkran and Thoms 2006; Hayes et al. 2008). For that reason, we evaluated the minimum distance between each site and forest patches of different sizes (described above). Forest patch sizes were calculated using a regional land use dataset (see Appendix 1 for dataset information).
Statistical analyses
Data screening and transformations
To reduce uncertainty associated with low sampling effort, sites sampled less than 3 years total were excluded from the final dataset. To account for variable sampling effort, we calculated a proportional occurrence value for each site by dividing the number of years a species was observed by the number of years the site was sampled (observations/effort). Sites were examined for spatial autocorrelation using Moran’s I and Geary’s c, and sites within autocorrelated distance bins (200 m distance and less) were manually evaluated to determine whether inclusion in the final dataset was justified. Environmental variables were examined and were transformed (via log transformation) where applicable to achieve a more normal distribution. Environmental variables were also screened for collinearity, and highly correlated variables (r > 0.8) were generally not included in the same models.
Data analysis
Fisher’s exact test (Fisher 1922) was used to determine whether species-level occupancy of different site types (manmade versus naturally occurring wetlands) differed from expected. Multivariate statistics were used to explore patterns of amphibian community composition and to determine which, if any, local and/or landscape variables were closely related to community composition patterns. Community data included proportional occurrence data for all six amphibian species. We calculated assemblage dissimilarity using Bray-Curtis distance (Legendre and Legendre 2012) and applied nonmetric multidimensional scaling (NMDS) to visualize patterns of community composition. We also calculated correlation coefficients between the axes of the NMDS ordination and explanatory (local and landscape) variables to determine which explanatory variables were related to community composition. Only explanatory variables with correlation coefficients of r = |0.3| and above were considered. The NMDS analysis was performed using the statistical software R, version 2.15.0 (R Core Team 2013) with the ‘vegan’ package, version 2.0–3 (Oksanen et al. 2012).
We employed an information-theoretic approach (Burnham and Anderson 2002; Hobbs and Hilborn 2006) as an alternative to traditional hypothesis testing to compare and select models most supported by our data. We fit generalized linear models (GLMs) with a binomial probability distribution and a logit link to accommodate the proportional occurrence data. To avoid unstable models associated with few observations per variable, we aimed to retain a conservative minimum ratio of ten observations to each explanatory variable as recommend by Peduzzi et al. (1996). To achieve this we examined two sets of explanatory variables for each species: one with local explanatory variables (N = 8), and one with landscape explanatory variables (N = 10). Only species observed at >15 % of sites were included in GLM models as high rates of absences can lead to unstable models (Lemckert and Mahony 2011). Global models and their Akaike Information Criteria (AIC, Akaike 1973) were calculated using the package “MuMIn” (Bartoń 2014) in R. The difference between the AIC of a particular model and the AIC of the estimated best-fitting model (i.e. the model with the lowest AIC) is delta (Δ) AIC. Models with ΔAIC < 2 have strong support (Burnham and Anderson 2002). We thus retained models with ΔAIC < 2, and model weights for variables in supported models were summed to provide an overall weight for each variable (Burnham and Anderson 2002). A value of 1.0 indicates that the variable was included in every supported model, and values closer to 0 indicate the presence of the variable in only one or a few supported models. This procedure was performed for both local and landscape variables independently; the variables with the highest weight (up to n = 5) from both groups were then combined to evaluate local and landscape variables together (up to n = 10 environmental variables).
Models that include both species and environmental drivers together help elucidate relative strengths of drivers and help highlight complex interactions, but evaluating relationships between a single species and a single environmental variable may help elucidate true drivers of species occurrence and abundance. Single-species, single-variable models are also of interest to managers who may be forced to optimize only one or a few variables at a time. We examined single-species, single-variable relationships between proportional occurrence and a subset of environmental variables identified both by NMDS and GLM analyses as important. We also looked for the presence of thresholds in these relationships using an iterative procedure to estimate the breakpoints and slopes of multiple segmented relationships with the linear predictor, as implemented in the R package “segmented” (Muggeo 2008). Data were binned according to values of explanatory data. Equal deciles of explanatory data were used unless breaks by decile resulted in identical values. For example, built environment and forest distances (Built 200, Built1K, and Forest500) had > 10 % of the data with a value of 0. Binning by equal deciles would have resulted in multiple bins with values equal to 0. In those cases, we instead binned first by identical values, then by dividing the remaining data points in bins with counts as close to equal as possible. A weighted regression approach was then used in threshold detection in order to account for different sizes of bins. We also examined simple linear regression between bins of environmental and occurrence data (weighted where appropriate) to compare the fit of simple linear regression to the threshold models.
Results
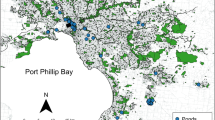
Our final dataset included 100 sampling sites (134 of the original 234 sites that were surveyed had less than 3 years of occupancy data and so were excluded from the local and landscape characteristic analysis; Table 1). Of the 100 sites included in this study, 21 were “naturally” occurring floodplain wetlands (though many have been altered to accept piped stormwater during high flows), whereas 79 were vegetated stormwater facilities such as ponds or swales, created expressly for the treatment and/or detention of stormwater (Fig. 2). None the species in this study were found to have a significant association with either “natural” wetlands or “man-made” stormwater facilities (Appendix 2; Appendix 3). High correlations between distances to forest patches of various sizes were observed. For this reason, only two forest patch sizes were retained: distance to a smaller minimum forest patch size of 500 m2 (correlated with distances to patch sizes of 50, 200, and 1000 m2; r ≥ 0.80), and distance to a larger minimum forest patch size of 5000 m2 (correlated with patch sizes 1000 and 2500 m2; r ≥ 0.82). Note that we found a significant correlation between hydroperiod and depth (r = 0.87), but we retained both variables as the mechanisms by which these affect species may be fundamentally different (Brooks and Hayashi 2002).
Nonmetric multidimensional scaling of proportional occurrences revealed two distinct groups of species. Group 1 included long-toed salamanders, Pacific chorus frogs, and northern red-legged frogs, with overlapping proportional occurrences that were generally positively associated with the first axis and negatively associated with the second axis (Fig. 3b, c, d; Group 1 members shown in gray). Group 2 included the northwestern salamander, the American bullfrog, and the rough-skinned newt (Fig. 3b, c, and d; Group 2 members shown in black). Species in Group 2 were not as tightly clustered as those in Group 1, but they had broad overlap and similar associations with both axes (negative for both axis 1 and 2). Proportional occurrences of both groups were positively related to Depth and HydroP, and all species were negatively related to Built200 and For500 (Fig. 3a). We also observed a negative correlation between members of Group 1 and both Fish and NonNatVeg (Fig. 3a). Group 2 was modestly and positively correlated with both Fish and NonNatVeg (Fig. 3a).
Two axes of a 2-dimensional nonmetric multidimensional scaling (NMDS) are shown. a) All study sites shown as gray points, and correlated environmental drivers (r ≥ |0.3| for at least one axis) are shown as arrows. b through d) Species proportional occupancies (number of years present / number years sampled) are coded by symbol size and color. Sample sites are represented as hollow circles, and proportional occurrences are colored either gray or black depending on species identity, shown in the panel legend. Completely full circles indicate sites where species were present during all years of sampling effort. Alternatively, sites with only small colored circles indicate that species occurrence for only one or a few years of sampling. Note that species with the same color across these three panels tend to overlap in multivariate space
Generalized linear models were used to examine relationships between environmental drivers and species proportional occurrences of northwestern salamanders, long-toed salamanders, Pacific chorus frogs, and northern red-legged frogs (Table 2). Note that American bullfrogs and rough-skinned newts were excluded from GLM modeling efforts due to a high proportion of absences across sites (>85 %). Generalized linear models revealed some consistent effects of environmental variables across species. For example, Built200 was strongly and negatively correlated with proportional occurrence of all four species. For500 was also negatively correlated with all species, but the relationship varied in strength across species. FloatSubVeg was negatively associated with all species except the northwestern salamander, although only modestly so. HydroP was strongly and positively correlated for three of four species (all except the northwestern salamander, which instead had a strong and positive correlation with Depth).
Generalized linear model models also revealed that some environmental variables differed in their effects by species. Age was strongly and negatively correlated with two species (long-toed salamander and the Pacific chorus frog) but had only a weak positive relationship with the northwestern salamander and was not retained in any northern red-legged frog models. Conversely, Built1K had a strong, positive correlation with two species (long-toed salamander and Pacific chorus frogs) but small and variable effects on the other two species (weak positive for northwestern salamanders; weak negative for northern red-legged frogs). Both Fish and For5K had modest to weak relationships that varied by species (negative for long-toed salamanders and Pacific chorus frogs; positive for northwestern salamanders, no relationship with northern red-legged frogs). NonNatVeg and NearStream also had modest relationships that varied by species (positive for northwestern salamanders, negative for all other species). Finally, NearSite and Cover had only modest contributions to one or two species, respectively.
Weighted linear regression and threshold analysis also excluded American bullfrogs and rough-skinned newts due to a high number of absences. Environmental variables considered included Depth, HydroP, Age, NonNatVeg, Built200, Built1K, and For500, identified as important both by NMDS and GLM approaches. Results revealed that both Depth and HydroP had significant relationships with all four species considered (Table 3). A significant, positive linear relationship between Depth and the northwestern salamander was identified. A depth threshold was identified for three of four species (long-toed salamander at 45.5 cm, Pacific chorus frog at 50.2 cm, and the northern red-legged frog at 58.6 cm). The relationship with Depth was positive up to the threshold and negative for depths greater than the threshold for all three species (Fig. 4b, d, and f). HydroP was positively and linearly related to the northwestern salamander, the long-toed salamander, and the Pacific chorus frog occurrence. A HydroP threshold was identified for northern red-legged frogs (180.3 days), where a strong and positive relationship between HydroP and proportional occurrence was observed for hydroperiods longer than the threshold (Fig. 4c). For500 had a negative, linear relationship with three of four species (non-significant for northern red-legged frogs). Built200 had a negative and significant linear relationship with Pacific chorus frogs and northern red-legged frogs. A Built200 threshold was identified for northwestern salamanders (25.4 %) that marked a transition from a negative to a roughly flat relationship (Fig. 4g). No significant relationship was observed between Built200 and long-toed salamanders. Built1K had a significant and negative relationship with northern red-legged frogs, and a threshold for Built1K was identified for Pacific chorus frogs (30.2 %) at which point the relationship transitioned from positive to negative (Fig. 4e). Age was only significantly related to one species (long-toed salamanders) for which a threshold from negative to positive was detected at 9.8 years (Fig. 4a). Finally, NonNatVeg was not significantly related to any species.
Plots of significant thresholds identified by species and environmental variable (Table 3). Bins of environmental variables are shown on the x-axis, and species are identified on the y-axis (note that species are constant across rows). Species proportional occurrences (averaged by bin) are shown as points. Piecewise linear regression models and 95 % CIs are shown as solid and dashed lines, respectively
Discussion
This study highlights local and landscape features that should be considered when creating and maintaining aquatic amphibian habitat in the urban ecosystem. Increased hydroperiod, water depth, impervious surfaces, non-native vegetation and forest patches were identified as important localized habitat features that affect occurrence across species. Hydroperiod and water depth are well known and widely recognized drivers of amphibian communities (Snodgrass et al. 1999; Snodgrass et al. 2000; Babbitt 2005). Our study shows this holds true of even highly modified environments despite the effects of urbanization and the presence of stressors that may decouple amphibian communities from their natural drivers. We stopped recording hydroperiod data on July 15 of each year, preventing us from defining a threshold for minimum wetted days; however, our data suggests that there is a positive correlation between all species and sites that stay wet until at least early summer. The northern red-legged frog in particular is often targeted for monitoring and conservation efforts, as it is listed by Oregon Department of Fish and Wildlife as “sensitive - vulnerable”, and in California and British Columbia as a “species of special concern” due to declining populations. In our study, the northern red-legged frog showed a threshold for hydroperiod at 180 days, suggesting that northern red-legged frogs are found more often at sites that stay wet until at least June 30 in a given year. Henning and Schiraton (2006) also found that hydroperiod affected northern red-legged frog presence where sites with “intermediate hydroperiods” (7 month inundation) had the highest abundance. In northwest Oregon and southwest Washington, northern red-legged frog eggs typically hatch late March-early April and reach metamorphosis 11–14 weeks later (Storm 1960; Brown 1975). Thus, management for this species should include providing breeding sites with a hydroperiod extending until late June in order to maximize survival to the juvenile stage.
Maximum water depth was an important variable for long-toed salamanders, Pacific chorus frogs, and northern red-legged frogs with each species showing a clear depth preference at 40–60 cm. This is likely related to hydroperiod, temperature, and these species’ ability to utilize ephemeral breeding ponds and metamorphose within one season (Nussbaum et al. 1983; Leonard et al. 1993). These species were observed at sites with depths that surpassed identified thresholds; however, at deep sites these species tended to deposit eggs and congregate closer to the shallow site margins (L.B. Guderyahn, pers. obs.). Pond margins are characterized by warmer water that promotes faster development and microhabitats that serve as potential refuge from predators (Bancroft et al. 2008). The northwestern salamander was the only native species to show an association with permanent water. This species typically overwinters at least once before metamorphosis (Watney 1941; Licht 1975) and several populations containing neotenic individuals (Licht 1992) that require permanent water have been found in Gresham (L. Guderyahn, unpublished data).
Additionally, we found two distinct groups of species driven largely by the hydroperiod and depth of aquatic habitat. Pacific chorus frogs, long-toed salamanders, and northern red-legged frogs were associated with ephemeral or intermittent sites, whereas northwestern salamanders, rough-skinned newts, and American bullfrogs showed a preference for perennial aquatic habitats. If promotion of species diversity is a goal across the landscape, management should include providing a variety of water depths and hydroperiods in order to increase the number of species likely to be supported. Alternatively, if the goal of a habitat project is to promote the existence and persistence of a specific species, such as the northern red-legged frog, a thorough understanding of species-specific needs is required. Our results provide an important first step in understanding how local and landscape factors affect amphibian diversity in urbanized environments. However, additional research is needed to understand how populations of sensitive species respond to management actions over time and across space.
We also found that long-toed salamanders and Pacific chorus frogs showed moderate negative correlations to fish. This is a pattern reported elsewhere in the Pacific Northwest (Monello and Wright 1999; Bull and Marx 2002; Pearl et al. 2005), and may be due to a correlative effect such as the preference for ephemeral breeding sites and/or a direct effect of predation or competition avoidance behavior (Skelly 1995; Wellborn et al. 1996). Similarly, we found positive correlations between fish presence and American bullfrogs, northwestern salamanders and rough-skinned newts, and we suspect these correlations are driven by a shared requirement for sites with permanent water.
The American bullfrog has long been hypothesized to have a negative effect on populations of native amphibians, and studies have shown this relationship to be highly complex and dependent upon a host of wetland characteristics such as food availability, fish presence, and hydrologic regime (Kiesecker and Blaustein 1998; Adams 1999; Kiesecker et al. 2002). In our study, American bullfrogs were only found at eleven of the 100 sites. Furthermore, in five of those eleven sites, this species was found in only one of multiple years of surveys, suggesting that their presence may currently be limited in this urban study area. Studies that focus on the factors that affect and interact with American bullfrog presence in urban areas are needed to better understand the distribution of bullfrogs across the urban landscape and their relationship with native amphibian species.
In addition to modified habitat and invasive species, native amphibians in urban environments also must cope with contaminants in breeding habitats. Stormwater ponds in urban areas are built to collect, filter and treat high concentrations of metals, oils, and road salts and these contaminants may have negative impacts on wildlife using the ponds (Gallagher et al. 2011, 2014; Hatch and Blaustein 2003; Van Meter et al. 2011; Le Viol et al. 2012). While this study did not explore questions related to water chemistry, the urban nature of our sites suggests some level of toxins are likely present. We found no significant effect of habitat type (wetland vs water quality facility) on the presence of any species. However, the number of occurrences for some species in our study was small and may have limited our power to detect any significant preference for habitat type. Additionally, contaminant levels may not differ significantly between manmade versus naturally occurring breeding habitat in this study system. Thus, future work might aim to explore the importance of toxicity relative to habitat variables in the persistence of amphibians in urban environments.
Habitat connectivity and retention of forest habitat are also known to be important considerations when locating ponds to support breeding amphibians (Guerry and Hunter 2002; Semlitsch and Bodie 2003; Hayes et al. 2008). We found evidence that both the amount of impervious surface within 200 m of a breeding site and the distance to nearby forest patches had strong negative relationships with all species. For example, northwestern salamanders decreased to virtually no occurrences in ponds with over 25 % built surface in a 200 m radius buffer. These findings are consistent with Homan et al. (2004) who found significant thresholds for impervious cover for spotted salamanders and wood frogs at the 100 m–300 m spatial scale. Similarly, Ostergaard et al. (2008) found percent forest cover within 200 m of a pond to be positively correlated with northwestern salamanders and northern red-legged frogs. These findings suggest that reducing impervious cover within relatively small buffers around breeding sites and maintaining forest patches within that same buffer may have significant positive effects on species persistence in the urban landscape.
Interestingly, our study showed that the negative relationship with the amount of impervious cover within a 1000 m buffer was not strong for every species. The northwestern salamander, the northern red-legged frog and the long-toed salamander showed weak or insignificant relationships, and the Pacific chorus frog actually showed a moderately strong positive relationship. One explanation could be an inability of individuals to disperse among habitat patches in a fragmented urban landscape (Gibbs 1998; Parris 2006). Species may make disproportionate use of habitats closest to breeding sites, and as a result, may be less sensitive to land cover changes within the larger buffer. Sites located within riparian buffers or neighborhoods with a low proportion of impervious surfaces may also offer greater opportunities for foraging, overwintering and dispersal without individuals needing to make longer movements. A note should be made that our study only captured the total percent impervious within each buffer size, and did not include the spatial configuration of where that land cover type exists within the buffer. It is possible that the habitat nearest to the pond in the 1000 m buffers may not have had significant impervious surface even if the outer portions of the buffer zone did. Additional research is needed to look at movement and habitat use in urban environments so that we can move beyond correlative studies.
Transitioning non-native vegetation to native plant communities has been shown to improve habitat quality by increasing the number and ability of microhabitats to support native amphibians and other fauna throughout all life stages (Vitousek et al. 1996; Marzluff and Ewing 2001). Our finding of negative associations between long-toed salamanders, Pacific chorus frogs and northern red-legged frogs and the percent of non-native vegetation within 10 m of a site’s edge may suggest an inability of non-native vegetation to fulfill the habitat requirements for these species. However, non-native vegetation has also been correlated with sites that are more altered and have higher human presence or influence in general (McKinney 2001). Therefore, the negative correlations we found may be due to newly or highly disturbed or impacted habitats rather than pointing to a lack of habitat value by non-native species specifically. One additional observation made during the study was that lawn grasses (i.e. Kentucky bluegrass, ryegrass and fine fescue) were found to be very common around many of our man-made sites. While developers, neighbors, or land managers in urban areas may prefer lawn grass buffers around water quality facilities or protected wetlands for ease of maintenance and lawn aesthetic, the low profile of lawn grasses provides little cover for amphibians migrating to and from breeding sites, and the associated maintenance activities themselves may provide direct sources of mortality to metamorphs and migrating adults (i.e. lawn mowers, moving vehicles, etc.; Hartup 1996). If grass is the preferred vegetation type around these facilities, efforts should be made to allow grasses to grow taller to provide needed cover. In addition, precautions should be taken to minimize direct mortality through altering the timing of mowing or flushing adults and metamorphs from these areas before maintenance begins.
The dependence of species on upland habitat for foraging, overwintering, and connectivity is well established (Nussbaum et al. 1983; Schuett-Hames 2004; Corkran and Thoms 2006; Hayes et al. 2008). Our study revealed a negative correlation between the distance to a forest patch and occurrence of all species. Additionally, we found stronger negative relationships between distances to small forest patch sizes (500 m2) than we did for the distances to larger forest patch sizes (5000 m2). This suggests that nearby, small forest patches are important habitat for species in an urban landscape. For example, the importance of upland forested overwintering grounds for northern red-legged frogs has been the subject of much research interest (Ritson and Hayes 2000; Schuett-Hames 2004; Hayes et al. 2008). Although northern red-legged frogs can migrate several kilometers in an urban area (Hayes et al. 2008), the resistance to movement presented by a highly developed landscape may be too great to overcome (Mathias 2008). The average distance to a 500 m2 forest patch for sites in our study was 44 m, which may be sufficiently short to allow migration from a pond through an urban landscape to forested upland. In contrast, the average distance to 5000 m2 forest patches was 225 m (up to 1869 m), which may be a more difficult distance to overcome in an urban landscape. Mathias (2008) proposes a 200 ha (2,000,000 m2) minimum forest patch size for northern red-legged frogs. However, forest patches of that size do not exist in most urban landscapes. This lack of large forest patches combined with our finding a positive association with forest patches as small as 50–500 m2 suggests that the minimum patch size needed may be much smaller, providing an attainable goal for land managers in urbanized environments. Forest patches with complex understory structure, high levels of woody debris (Aubry and Hall 1991; Haggard 2000; Schuett-Hames 2004), and a deep litter layer (Aubry 2000) are correlated with northern red-legged frog use, suggesting that the characteristics of forest patches are an important consideration in addition to size. Additional research to identify the size, shape and characteristics of forest patches required by northern red-legged frogs in urban areas is important for the persistence of this and other species.
Site age was correlated with the occurrence of Pacific chorus frogs and long-toed salamanders, both of which showed an affinity for sites less than 5 years old. Both of these species tend to be opportunistic breeders, with eggs and larvae often found in disturbed areas, such as newly formed (Leonard et al. 1993; Hamilton et al. 1998), recently disturbed (Corkran and Thoms 2006), and human influenced (Beneski et al. 1986; Llewellyn and Peterson 1998; Monello and Wright 1999) ponds. The trend found in our study may be a result of their ability to quickly colonize newly created or recently disturbed sites.
In areas where less aquatic habitat exists and significant barriers to accessing habitat patches occur, species are constrained to using the resources that are available to them and that best fit their preferences. Land managers and city planners are addressing habitat needs of amphibians and the impact of impervious surfaces through restoration, green infrastructure retrofits, improving habitat corridors, and new development standards (e.g. City of Chicago 2006; City of Seattle 2015). Reducing impervious cover around existing breeding sites as well as protecting sites that are currently adjacent to forest patches may have significant positive impacts on species occurrence. Additionally, restoration projects seeking to establish new habitat should carefully consider restoration goals and should seek to balance species-specific requirements with habitat characteristics beneficial to all wildlife. The results of this study are an important step to understanding how to manage for amphibian biodiversity as well as specific species requirements in urban areas. Additional research is required to add to our understanding of the mechanisms at work in these relationships and the role that movement corridors play in conserving urban amphibians.
References
Adams MJ (1999) Correlated factors in amphibian decline: exotic species and habitat change in western Washington. J Wildl Manag 63:1162–1171
Akaike H (1973) Information theory and an extension of the maximum likelihood principle. In: Petrov BN, Csaki F (eds) Second International Symposium on Information Theory. Akademiai Kiado, Budapest, pp 267–281
Aubry KB (2000) Amphibians in managed, second-growth Douglas-fir forests. J Wildl Manag 64:1041–1052
Aubry KB, Hall PA (1991) Terrestrial amphibian communities in the southern Washington Cascade Range. In: Ruggiero LF, Aubry KB, Carey AB, Huff MH (eds) U.S. Forest Service general technical report PNW-GTR-Pacific Northwest Research Station, USA. pp 326–338
Babbitt KJ (2005) The relative importance of wetland size and hydroperiod for amphibians in southern New Hampshire, USA. Wetl Ecol Manag 13(3):269–279
Bancroft BA, Baker NJ, Searle CL, Garcia TS, Blaustein AR (2008) Larval amphibians seek warm temperatures and do not avoid harmful UVB radiation. Behav Ecol 19:879–886
Bartoń K (2014) Package ‘MuMIn’: model selection and model averaging based on information criteria (AICc and alike). R package version 1.10.5. Manual: http://cran.r-project.org/web/packages/MuMIn/MuMIn.pdf
Becker CG, Fonseca CR, Haddad CFB, Batista RF, Prado PI (2007) Habitat split and the global decline of amphibians. Science 318:1775–1777
Beneski JT Jr, Zalisko EJ, Larsen JHJ (1986) Demography and migratory patterns of the eastern long-toed salamander, Ambystoma macrodactylum columbianum. Copeia 1986:398–408
Birx-Raybuck DA, Price SJ, Dorcas ME (2010) Pond age and riparian zone proximity influence anuran occupancy of urban retention ponds. Urban Ecosyst 13(2):181–190
Brand AB, Snodgrass JW (2010) Value of artificial habitats for amphibian reproduction in altered landscapes. Conserv Biol 24:295–301
Brand AB, Snodgrass JW, Gallagher MT, Casey RE, Van Meter R (2010) Lethal and sublethal effects of embryonic and larval exposure of Hyla versicolor to stormwater pond sediments. Arch Environ Contam Toxicol 58:325–331
Brooks RT, Hayashi M (2002) Depth-area-volume and hydroperiod relationships of ephemeral (vernal) forest pools in southern New England. Wetlands 22:247–255
Brown HA (1975) Reproduction and development of the red-legged frog, Rana aurora, in northwestern Washington. Northwest Sci 49:241–252
Brown DJ, Street GM, Nairn RW, Forstner MRJ (2012) A place to call home: amphibian use of created and restored wetlands. Int J Ecol. doi:10.1155/2012/989872
Bull EL, Marx DB (2002) Influence of fish and habitat on amphibian communities in high elevation lakes in northeastern Oregon. Northwest Sci 76:240–248
Burnham KP, Anderson DR (2002) Model selection and multi-model inference: a practical information-theoretic approach. Springer, New York
City of Chicago (2006) Department of Planning and Development and Mayor Daley’s Nature and Wildlife Committee Nature and Wildlife Plan. Department of Planning and Development, Chicago
City of Seattle (2015) Seattle – municipal code. Municipal Code Corporation, Tallahassee
R Core Team (2013) R: a language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria. http://www.R-project.org/
Corkran CC, Thoms C (2006) Amphibians of Oregon, Washington and British Columbia: a field identification guide. Lone Pine, Auburn
Czech B, Krausmann PR, Devers PK (2000) Economic associations among causes of species endangerment in the United States. Bioscience 50:593–601
ESRI (2011) ArcGIS desktop: Release 10. Environmental Systems Research Institute, Redlands
Fisher RA (1922) On the interpretation of χ2 from contingency tables, and the calculation of P. J R Stat Soc 85(1):87–94
Foley JA, DeFries R, Asne GP, Barford C, Bonan G, Carpenter SR, Chapin FS, Coe MT, Daily GC, Gibbs HK, Helkowski JH, Holloway T, Howard EA, Kucharik CJ, Monfreda C, Patz JA, Prentice IC, Ramankutty N, Snyder PK (2005) Global consequences of land use. Science 309:570–574
Gallagher MT, Snodgrass JW, Ownby DR, Brand AB, Casey RE, Lev S (2011) Watershed-scale analysis of pollutant distributions in stormwater management ponds. Urban Ecosyst 14(3):469–484
Gallagher MT, Snodgrass JW, Brand AB, Casey RE, Lev SM, Van Meter RJ (2014) The role of pollutant accumulation in determining the use of stormwater ponds by amphibians. Wetl Ecol Manag 22(5):551–564
Gibbs JP (1998) Distribution of woodland amphibians along a forest fragmentation gradient. Landsc Ecol 13:263–268
Guerry AD, Hunter ML (2002) Amphibian distributions in a landscape of forests and agriculture: an examination of landscape composition and configuration. Conserv Biol 16:745–754
Haggard JG (2000) A radio telemetric study of the movement patterns of adult northern red-legged frogs (Rana aurora aurora) at freshwater lagoon, Humbolt County, California. M.A. Thesis. Humbolt State University, Arcata
Hamer AJ, Parris KM (2011) Local and landscape determinants of amphibian communities in urban ponds. Ecol Appl 21:378–390
Hamilton GB, Peterson CR, Wall WA (1998) Distribution and habitat relationships of amphibians on the Potlatch Corporation operating area in northern Idaho. Potlatch Corporation, Lewiston
Hartup BK (1996) Rehabilitation of native reptiles and amphibians in DuPage County, Illinois. J Wildl Dis 32:109–112
Hatch AC, Blaustein AR (2003) Combined effects of UV-B radiation and nitrate fertilizer on larval amphibians. Ecol Appl 13(4):1083–1093
Hayes MP, Quinn T, Richter KO, Schuett-Hames JP, Shean JS (2008) Maintaining lentic-breeding amphibians in urbanizing landscapes: the case study of the Northern red-legged frog (Rana aurora). In: Mitchell JC, Jung Brown RE, Bartholomew B (eds) Urban herpetology. Society for the Study of Amphibians and Reptiles, Salt Lake City, pp 445–461
Henning JA, Schiraton G (2006) Amphibian use of Chehalis River floodplain wetlands. Northwest Nat 87:209–214
Hobbs NT, Hilborn R (2006) Alternatives to statistical hypothesis testing in ecology: a guide to self teaching. Ecol Appl 16:5–19
Homan RN, Windmiller BS, Reed JM (2004) Critical thresholds associated with habitat loss for two vernal pool-breeding amphibians. Ecol Appl 14:1547–1553
Houlahan JE, Findlay CS (2003) The effects of adjacent land use on wetland amphibian species richness and community composition. Can J Fish Aquat Sci 60:1078–1094
Kiesecker JM, Blaustein AR (1998) Effects of introduced bullfrogs and smallmouth bass on microhabitat use, growth, and survival of native red-legged frogs (Rana aurora). Conserv Biol 12:776–787
Kiesecker JM, Chivers DP, Anderson M, Blaustein AR (2002) Effect of predator diet on life history shifts of red-legged frogs, Rana aurora. J Chem Ecol 28:1007–1015
Le Viol I, Chiron F, Julliard R, Kerbiriou C (2012) More amphibians than expected in highway stormwater ponds. Ecol Eng 47:146–154
Legendre P, Legendre L (2012) Developments in environmental modeling. In: Legendre P, Legendre L (eds) Numerical ecology, 3rd edn. Elsevier, Quebec, pp 275–277
Lemckert F, Mahony M (2011) The relationships among multiple-scale habitat variables and pond use by anurans in northern New South Wales, Australia. Herpetol Conserv Biol 5:537–547
Leonard WP, Brown HA, Jones LLC, McAllister KR, Storm RM (1993) Amphibians of Washington and Oregon. Seattle Audubon Society, Trailside Series. Seattle Audubon Society, Seattle
Licht LE (1975) Growth and food of larval Ambystoma gracile from a lowland population in southwestern British Columbia. Can J Zool 53:1716–1722
Licht LE (1992) The effect of food level on growth rate and frequency of metamorphosis and paedomorphosis in Ambystoma gracile. Can J Zool 70:87–93
Llewellyn RL, Peterson CR (1998) Distribution, relative abundance, and habitat associations of amphibians and reptiles on Craig Mountain, Idaho. Idaho Bureau of Land Management, Technical Bulletin 98–15, Boise, Idaho
Marzluff JM, Ewing K (2001) Restoration of fragmented landscapes for the conservation of birds: a general framework and specific recommendations for urbanizing landscapes. Restor Ecol 9:280–292
Mathias M (2008) Assessing functional connectivity for the northern red-legged frog (Rana aurora) in an urbanizing landscape. M.S. Thesis. University of Washington, Seattle
McCarthy MA (2009) Using models to compare the ecology of cities. In: McDonnell MJ, Hahs A (eds) The ecology of city and towns: a comparative approach. Cambridge University Press, Cambridge, pp 112–125
McKinney ML (2001) Effects of human population, area, and time on non-native plant and fish diversity in the United States. Biol Conserv 100:243–252
McKinney ML (2002) Urbanization, biodiversity, and conservation. Bioscience 52:883–890
Monello RJ, Wright RG (1999) Amphibian habitat preferences among artificial ponds in the Palouse region of northern Idaho. J Herpetol 33:298–303
Muggeo VMR (2008) Segmented: an R package to fit regression models with broken-line relationships. R News 8(1):20–25
NOAA (2016) Annual Climatological Summary. National Centers for Environmental Information. Station: PORTLAND INTERNATIONAL AIRPORT, OR US COOP:356751. https://www.ncei.noaa.gov/
Nussbaum RA, Brodie ED Jr, Storm RM (1983) Amphibians and reptiles of the Pacific Northwest. Caxton, Caldwell
Oksanen J, Blanchet FG, Kindt R, Legendre P, Minchin PR, O’Hara RB, Simpson GL, Solymos P, Henry M, Stevens H, Wagner H (2012) Vegan: Community ecology package. R package version 2.0–3 Manual: http://cran.r-project.org/web/packages/vegan/vegan.pdf
Oregon Biodiversity Information Center (ORBIC) Biodiversity Data. Oregon Natural Heritage Information Center & NatureServe. Oregon Ecological Systems Database 2009. Available: http://orbic.pdx.edu/publications.html. July 6 2013
Ostergaard EC, Richter KO, West SD (2008) Amphibian use of stormwater ponds in the Puget lowlands of Washington, USA. In: Mitchell JC, Jung Brown RE, Bartholomew B (eds) Urban herpetology. Society for the Study of Amphibians and Reptiles, Salt Lake City, pp 259–270
Parris KM (2006) Urban amphibian assemblages as metacommunities. J Anim Ecol 75:757–764
Pearl CA, Adams MJ, Leuthold N, Bury RB (2005) Amphibian occurrence and aquatic invaders in a changing landscape: implications for wetland mitigation in the Willamette Valley, Oregon, USA. Wetlands 25:76–88
Peduzzi P, Concato J, Kemper E, Holford TR, Feinstein AR (1996) A simulation study of the number of events per variable in logistic regression analysis. J Clin Epidemiol 49:1373–1379
Pope SE, Fahrig L, Merriam HG (2000) Landscape complementation and metapopulation effects on leopard frog populations. Ecology 81:2498–2508
Richter KO, Azous AL (2000) Amphibian distribution, abundance, and habitat use. In: Azous A, Horner RR (eds) Wetlands and urbanization: implications for the future. Lewis Publishers, Boca Raton, pp 143–165
Ritson PI, Hayes MP (2000) Late season activity and overwintering in the northern red-legged frog (Rana aurora aurora). Final report to the US Fish and Wildlife Service. Portland State Office, Portland
Scheffers BR, Paszkowski CA (2012) The effects of urbanization on North American amphibian species: identifying new directions for urban conservation. Urban Ecosyst 15:133–147
Scheffers BR, Paszkowski CA (2013) Amphibian use of urban stormwater wetlands: the role of natural habitat features. Landsc Urban Plan 113:139–149
Schuett-Hames JP (2004) Northern red-legged frog (Rana aurora aurora) terrestrial habitat use in the Puget Sound lowlands of Washington. M.S. Thesis. Olympia, The Evergreen State College
Semlitsch RD, Bodie JR (2003) Biological criteria for buffer zones around wetlands and riparian habitats for amphibians and reptiles. Conserv Biol 17:1219–1228
Skelly DK (1995) A behavioral trade-off and its consequences for the distribution of Pseudacris treefrog larvae. Ecology 76:150–164
Skidds DE, Golet FC, Paton PW, Mitchell JC (2007) Habitat correlates of reproductive effort in wood frogs and spotted salamanders in an urbanizing watershed. J Herpetol 41:439–450
Snodgrass JW, Ackerman JW, Bryan AL Jr, Burger J (1999) Influence of hydroperiod, isolation, and heterospecifics on the distribution of aquatic salamanders (Siren and Amphiuma) among depression wetlands. Copeia 1999:107–113
Snodgrass JW, Komoroski MJ, Bryan AL Jr, Burger J (2000) Relationships among isolated wetland size, hydroperiod, and amphibian species richness: Implications for wetland regulation. Conserv Biol 14:414–419
Storm RM (1960) Notes on the breeding biology of the red-legged frog (Rana aurora aurora). Herpetologica 16:251–259
Titus, JH, Christy JA, VanderSchaaf D, Kagan JS, Alverson ER (1996) Native wetland, riparian, and upland plant communities and their biota in the Willamette Valley, Oregon - Phase I Project: Inventory and Assessment Report to Environmental Protection Agency, Region X, Seattle, Washington Willamette Basin Geographic Initiative Program
U.S. Census Bureau (2007) State & county quickfacts: Allegany County, N.Y. Retrieved Feburary 15, 2014, from http://quickfacts.census.gov
Van Meter RJ, Swan CM, Snodgrass JW (2011) Salinization alters ecosystem structure in urban stormwater detention ponds. Urban Ecosyst 14(4):723–736
Vitousek PM, D’Antonio CM, Loope LL, Westbrooks R (1996) Biological invasions as global environmental change. Am Sci 84:468–478
Watney GMS (1941) Notes on the life history of Ambystoma gracile. Copeia 1941:14–17
Wellborn GA, Skelly DK, Werner EE (1996) Mechanisms creating community structure across a freshwater habitat gradient. Annu Rev Ecol Syst 27:337–363
Acknowledgments
We would like to thank all of the City of Gresham Watershed Division for assisting the Natural Resource Program with collecting and assessing data on City ponds, wetlands and swales. In addition, City of Gresham Mapping Department assisted with GIS analyses and mapping. Special thanks to those who reviewed the manuscript and gave feedback: T. Brimecombe, C. Corkran, T. Curry, K. Holzer, K. Majidi, and Dr. J. Maser. M. Mims was supported by a National Science Foundation Graduate Research Fellowship (Grant No. DGE-0718124).
Author information
Authors and Affiliations
Corresponding author
Appendices
Appendix 1
Appendix 2
Appendix 3
Rights and permissions
About this article
Cite this article
Guderyahn, L.B., Smithers, A.P. & Mims, M.C. Assessing habitat requirements of pond-breeding amphibians in a highly urbanized landscape: implications for management. Urban Ecosyst 19, 1801–1821 (2016). https://doi.org/10.1007/s11252-016-0569-6
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11252-016-0569-6