Abstract
Aims
Soil salinization poses a significant challenge to agriculture. The practice of returning straw to the field has garnered increasing attention as a sustainable method to improve salinized land. This study aimed to investigate the impact of nitrogen application rates on rice straw decomposition, rice straw nutrient release, and rice yield in saline sodic rice field conditions.
Methods
The field experiment was designed with five nitrogen (N) fertilizer rates with 0 (N0), 90 (N1), 180 (N2), 270 (N3), and 360 kg N ha−1 (N4) under the condition of full return (8 t ha−1) of straw from sodic-saline paddy fields, nylon mesh bag filling method was used to study the decomposition process of rice straw. The field experiment was conducted in a completely randomized design with three replications.
Results
The results showed that the application of N fertilizer promoted the decomposition of rice straw and the release of C, N and P from the straw, but had no effect on the release of K from the straw. Compared with N0, the cumulative decomposition rates of rice straw in N1, N2, N3 and N4 treatments were significantly increased by 10.05%, 15.10%, 20.00% and 18.44%, respectively (two-year average). In addition, the highest rice yield was obtained in the N3 treatment.
Conclusion
Overall, the study suggests that applying 270 kg N ha−1 is the most effective in promoting rice straw decomposition, nutrient release, and increasing rice yield in sodic saline rice fields, offering valuable insights for optimizing N fertilizer application and maximizing farmer’s economic benefits.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
It is one of the greatest challenges to agriculture and livestock production with soil salinization in arid and semi-arid areas (Qadir et al. 2008). According to data published by FAO in 2015, the total area of saline soils is about 1 billion hectares globally (FAO and ITPS 2015). Salt-affected soils are characterized by high concentrations of dissolved mineral salts in their profiles, which can negatively impact crop production (Amini et al. 2016). The physical and chemical properties of Salt-affected soils are characterized by high salinity, high pH, poor soil structure, and limited nutrient content (Morton et al. 2019). The Songnen Plain in China has a soil area affected by salinization of more than 3.7 × 106 ha, which is one of the three major saline sodic soil areas in the world (Yang et al. 2016). Plants growing in this area are subjected to both Na+ toxicity and high pH stress caused by excess Na2CO3 and NaHCO3, which cause much more damage to plants than NaCl and Na2SO4 (Wang et al. 2018). Recent studies have shown that rice cultivation under adequate irrigation conditions is one of the most effective ways to increase land utilization, improve ecological environment and promote economic development in saline areas (Xu et al. 2020; Yuan et al. 2019). N can play both nutritional and osmotic roles in saline conditions (Song et al. 2019). The application of N fertilizer can effectively alleviate the saline stress suffered by rice and increase rice yield (Zhu et al. 2020). Therefore, N fertilizer is often used in large quantities and for a long time by farmers in rice cultivation in saline areas. However, in the saline sodic rice growing areas of the western Songnen Plain, this practice is not sustainable.
A challenge for many rice farmers is the nutrient deficiency of the soil and the high carbon deficit in the soil. However, the carbon-poor soils can effectively function as a sink with appropriate management strategies. While there are numerous complex and costly approaches, a simple and cost-effective strategy involves returning rice straw back to the soil. Straw is a significant biological resource. Straw return not only has multiple effects on the soil, such as promoting soil carbon accumulation, improving soil physicochemical properties, and enriching soil microbial diversity (Wang et al. 2021; Yu et al. 2020), but also improved crop photosynthesis to improve crop yield, while reducing N fertilizer application, and increasing farmers’ net income (Pan et al. 2022; Ran et al. 2022). However, the slow decomposition of straw, caused by the lack of nutrients in the soil, negatively impacts the growth of rice plants.In addition, the long winters and low temperatures in northeastern China have led to low acceptance of straw return by farmers, and large quantities of rice straw are burned directly in the fields. Straw burning causes nutrient losses of about 80% N, 25% P, and 21–33% K, and is accompanied by severe air pollution (Yan et al. 2019). How to promote straw return technology in northeast saline sodic areas is of important significance for soil improvement and reducing environmental pollution. Therefore, the clarification of the rules of straw decomposition is the basis of reasonable use of straw, and is also the key to the straw return technology in saline sodic rice fields.
Straw decomposition and nutrient release are influenced by soil and climatic factors because they are directly related to the activity of microbial decomposers (Wahdan et al. 2022; Li et al. 2020). In addition, the straw’s own C/N also heavily influences the process of straw decomposition and nutrient release (Zhang et al. 2022). When straw C/N < 20, straw decomposition is rapid, straw organic N mineralization, and soil N content increases; when straw C/N > 30, straw decomposition is slow, microbial sequestration of soil N is enhanced, and soil available N levels decrease (Christopher and Lal 2007; Truong et al. 2019). Rice straw is mainly composed of carbon rich materials such as cellulose, hemicellulose and lignin, and its C/N is much higher than 30 (Pan et al. 2017; Wu et al. 2014). The decomposition process of rice straw consumes a large amount of available N in the soil, which competes with crop growth for N and leads to crop yield reduction (Cheshire et al. 1999; Zhu et al. 2015). The application of mineral N fertilizer provides more inorganic N, which promotes the growth of microorganisms, thus accelerating the decomposition of straw and the release of nutrients (Zhao and Zhang 2018). Sinsabaugh et al. (2002) also showed that N fertilizer application promoted the activity of hydrolytic enzymes such as cellulase, which accelerated the decomposition of organic matter in straw. However, excessive N fertilizer application significantly reduced the abundance and diversity of functional genes in the carbon cycle and affected the decomposition of straw (Jing et al. 2021). At the same time, some studies have indicated that the emissions of greenhouse gases, including carbon dioxide and nitrous oxide, tend to rise as N application rates increase (Cao et al. 2021). Therefore, reasonable N fertilizer application is crucial for straw return into the field, especially in nutrient-poor saline sodic soils.
Related studies have shown that the combined use of inorganic fertilizer with straw incorporation has positive effects on rice roots, N fertilizer utilization, and photosynthetic characteristics in saline sodic rice fields (Ran et al. 2022; Meng et al. 2022). However, the characteristics of in situ straw decomposition and nutrient release by N fertilizer application are unknown. Therefore, this study was carried out to investigate the decomposition, nutrient release and the change pattern of organic components of rice straw under five N application rates in saline sodic rice fields. In this study, we aimed to (1) clarify the dynamics changes of decomposition and nutrient release of rice straw under different N fertilizer application rates; (2) seek N fertilization options for rapid straw decomposition and high grain yield under straw return conditions in saline rice growing areas; and (3) provides the basis for guiding the rational application of N fertilizer and realizing the maximization of farmers’ economic benefit.
Materials and methods
Site description
This study was conducted from October 2019 to October 2021 under saline paddy field conditions at the Saline Experimental Station of Jilin Agricultural University (N 45°35′58″-N 45°36′28″, E 123°50′27″-123°51′31″, 133.7 m a.s.l.), which is located in Sheli Town, Baicheng City, Jilin Province, China. The experimental station is located in the western part of the Songnen Plain in northeast China, which belongs to the overland of temperate continental monsoon climate and continental climate. The average annual temperature was 4.7 °C, the average annual precipitation was 413.7 mm, the average active cumulative temperature ≥ 10 °C was 2,996.2 °C, the average frost-free period was 144 d, and the average annual sunshine hours were 2915 h. The average monthly precipitation and average monthly temperature for the test years are shown in Fig. 1. According to the soil classification of the World Soil Resource Reference Base (IUSS Working Group 2014), the main soils in the experiment are of Solonetz type. The 0–20 cm soil was collected after the rice harvest in October 2019 to determine its physical and chemical characteristics (Table 1). Before the experiment, the newly reclaimed land was planted with rice for five consecutive years, and the straw produced from rice cultivation was burned on-site before spring plowing the following year.
Monthly precipitation and temperature variations during the experiment. The meteorological data from http://www.meteomanz.com
Field experiment design
The field experiment used a completely randomized block design with three replications. Five N application rate treatments were set at a straw return rate of 8000 kg ha−1 (converted at a grain to straw ratio of 1:1.1 based on local rice yields). The stalks used in the study were obtained from fields that were managed in a consistent manner to maintain uniformity in their physical and chemical properties. The N application rates were as follows. 0, 90,180, 270 and 360 kg N ha−1 (hereafter referred to as N0, N1, N2, N3 and N4, respectively). The size of each subplot was 30 m2 (6 m×5 m). To avoid exchange of nutrients and water between plots, plots were separated by large ridges (0.6 m wide and 0.4 m high). Each plot had a separate irrigation and drainage outlet. The fertilizers applied in the trial were urea, calcium superphosphate, potassium chloride, and zinc sulfate heptahydrate. N fertilizer was applied in accordance with base fertilizer : tillering fertilizer : panicle fertilizer = 6:3:1. Potash (K2O) fertilizer was applied as a base and panicle fertilizer at a ratio of 6:4, at a rate of 75 kg ha−1. Phosphate fertilizer (P2O5) and zinc fertilizer (ZnSO4) were all applied as basal fertilizers at a rate of 50 kg ha−1 and 20 kg ha−1, respectively. The base fertilizer was applied on May 9, the tillering fertilizer was applied in late June, and the panicle fertilizer was applied in mid-July. The local rice variety " Baigeng 1” was planted in this experiment. Transplanting was carried out on May 20 at a density of 20 hills m−2 with a spacing of 16.5 × 30.0 cm and four seedlings per hill in both years. To prevent the phenomenon of salt return to the paddy fields, the paddy fields were flooded at a depth of 3–5 cm after transplanting until two weeks before the rice harvest. Diseases, insects and weeds were strictly controlled during the experiment.
After the rice harvest, rice straw was collected by hand from the fields. After natural air drying, the straw is cut into 5–7 cm long pieces with a straw chopper. The crushed rice straw was spread evenly in the harvested fields on October 17, 2019 and October 19, 2020, and then incorporated into the soil using a reverse stubble rotavator (1GFM-220, Lianyungang Xingan Machinery Manufacturing Co., Ltd., Jiangsu, China). Meanwhile, nylon mesh bag filling method was used to study the decomposition process of rice straw. Nylon mesh bags were 15 × 10 cm in size with an aperture of 0.15 mm. Each bag was put with 10.00 g of dried rice straw and then sealed with nylon thread. The initial nutrient and cellulose contents of rice straws in both years were measured before filling the nylon mesh bags, and the specific information is shown in Table 2. During the annual straw return in October, nylon mesh bags filled with straw were buried scattered among the rice fields, and the locations were marked in the fields. Each plot was buried with 24 bags of straw, at a depth of 15 cm, placed horizontally, and without overlapping. To prevent damage to the net bags by tilling the soil, the net bags were removed on May 9, 2020 and May 9, 2021, and placed in a container with soil, covered with a 15 cm thick layer of soil, and then the container was placed in the field. After the soil was rototilled, the net bags were put back in the field. The whole process lasted no more than 2 days. Three net bags containing rice straw were recovered from each plot on: (1) May 9 (during spring plowing); (2) June 9; (3) July 9; (4) August 9; (5) September 9; and (6) October 9 (after rice harvest). After recovery, the soil and root debris adhering to the straw were carefully removed with water, the mesh bag with the residual straw was dried at 70 °C for 48 h to a constant mass, and the straw was weighed and ground to measure the nutrient and cellulose content in the residual straw.
Sample analysis
Leaf area index (LAI) was determined according to the method described by Ran et al. (2023). Five rice plants with the same growth trend were selected from each plot (excluding side rows and sampling rows) based on the average number of tillers at mid-tillering (MT), panicle initiation (PI), heading (HD), filling (FI), and physiological maturity (PM) stages. All green leaves were manually removed, and the length (longest part) and width (widest point) of all green leaves were measured with a ruler. The leaf area of each leaf was then calculated (length-width coefficient method) with the formula: leaf area = leaf length × leaf width × 0.75. The leaf area index was calculated as follows:
Soil temperature was measured according to the method of Ding et al. (2017). After spring plowing, three right-angle geothermometers at a depth of 15 cm were buried in each plot. Measurements were taken every 10 days. Soil temperature values were read at 2:00, 8:00, 14:00, and 20:00 each time, and the average value was taken as the daily average temperature.
The decomposition rate, nutrient content and organic fraction (cellulose, hemicellulose, lignin) of the straw were determined using recovered straw bales. The straw decomposition rate was calculated using the mass loss method (Li and Zhong 2021). Its calculation formula is as follows:
where SD is the decomposition rate of straw (%), m0 is the original mass of straw (g), and mt is the residual mass of straw (g).
Straw C content was oxidized using potassium dichromate and then titrated with ferrous sulfate (Walkley and Black 1934). The straw total N content was digested by H2SO4-H2O2, and then determined by Kjeldahl N determination (Jia et al. 2019). The straw total phosphorus content was determined by the molybdenum antimony anti-colorimetric method (Lu 2000). The total K content of straw was digested using H2SO4-H2O2 and then determined by flame photometry (Lu 2000). The straw nutrient release rate was calculated according to the following equation:
wherehere NR is the nutrient release rate (%); m0 and mt are the initial and residual straw mass (g), respectively; c0 and ct are the nutrient contents (g kg−1) in the initial and residual straw, respectively.
The content of organic components (cellulose, hemicellulose and lignin) in rice straw was determined by a modified Van Soest detergent fiber method and the decomposition rate of organic components was calculated (Van-Soest 1963). The procedure was as follows: In the first step, 1.000 g (sieved through a 0.425 mm mesh) was weighed and extracted under neutral detergent (100 °C) for 60 min to obtain neutral detergent fiber (NDF). In the second step, 1.000 g (sieved through a 0.425 mm mesh) was weighed and extracted under acid detergent to obtain acid detergent fiber (ADF). In the third step, the acid detergent fiber was extracted and filtered with strong acid (5 mL, 72% H2SO4), and the non-extractable residue was dried at 60 °C and then ashed at 550 °C for 2 h. The acid detergent lignin (ADL) was obtained by the difference between the residue and the ashed material. The hemicellulose content of the straw residue was the difference between NDF and ADF, and the cellulose content was the difference between ADF and ADL. The decomposition rate of organic components (cellulose, hemicellulose and lignin) in straw was calculated as follows:
where OD is the decomposition rate of organic components in straw (%); m0 and mt are the mass of initial and residual straw (g), respectively; b0 and bt are the content of organic components in initial and residual straw (%), respectively.
Scanning electron microscopy (HITACHI S4800, Japan) was used to observe the microstructure of the straw surface. Rice straw samples were vacuum dried and gold sprayed, and imaged at an accelerating voltage of 5 kV with a magnification of 5000×.
Rice yield
Rice harvest was conducted in early October. In all plots, 2 × 2 m except for the border ones and the sampling area were collected for determining the yield per unit area with three replications. After threshing the rice, the empty deflated grains were blown clean and weighed and their moisture content was determined, and then converted to the corresponding yield at 14% moisture content.
Statistical analyses
All data collected were collated and calculated using Microsoft Excel 2019. Statistical analysis was conducted using one-way ANOVA of SPSS software (version 22.0, IBM SPSS Inc.). Significant differences between means were determined using the least significant difference (LSD) test with significance set at p < 0.05 (the data were normal distributed and variances homogenous). All data are presented as means with standard errors (SE). Graphs were drawn with Sigmaplot 14.0 software.
Results
Leaf area index
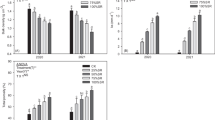
The leaf area index exhibited a pattern of initial increase followed by a decrease throughout the growth of rice, reaching its peak at the heading stage (Fig. 2). Throughout the rice growth stage, rice leaf area index gradually increased with increasing N fertilizer application rate, but there was no significant difference in leaf area index between N3 and N4 treatments. The application of N fertilizer significantly increased the leaf area index of rice from the panicle initiation stage to the physiological maturity stage compared to N0 treatment.
Effects of different N application rates on rice leaf area index (LAI) in saline sodic paddy field. Data are expressed as the mean ± standard error (n = 3). Different lowercase letters on the vertical bars of the same year indicate statistical significance between different fertilizer application at the p < 0.05 level. MT, PI, HD, FI and PM represent mid-tillering, panicle initiation, heading, filling and physiological maturity stages, respectively. A and B represent the years 2020 and 2021, respectively. N0, N1, N2, N3 and N4 were fertilized with 0, 90, 180, 270 and 360 kg ha−1 of N, respectively
Soil temperature
Soil temperature exhibited a pattern of increase followed by decrease during the period between spring plowing and rice harvest. The temperatures were higher from June to August, with a notable sharp decrease observed in September (Fig. 3). During the period from late June to early September, soil temperature gradually decreased with increasing N fertilizer application rate and was significantly lower in the N2, N3, and N4 treatments than in the N0 treatment. However, during the rest of the period, N fertilizer application had no significant effect on soil temperature.
Changes of soil temperature under different N application rates. Data are expressed as the mean ± standard error (n = 3). Different lowercase letters on the vertical bars of the same year indicate statistical significance between different fertilizer application at the p < 0.05 level. A and B represent the years 2020 and 2021, respectively. N0, N1, N2, N3 and N4 were fertilized with 0, 90, 180, 270 and 360 kg ha−1 of N respectively
Straw decomposition rate
The straw had a low decomposition rate (less than 10%) from when it was applied (mid to late October) until the following year’s spring ploughing (May 9). The decomposition of straw exhibited a pattern of initially fast decomposition from May to October, followed by a slower rate (Fig. 4). The overall straw decomposition rate was 55.27-66.07% in the first year of the experiment and 56.34-67.87% in the second year. The accumulated straw decomposition rates of N1, N2, N3 and N4 treatments were significantly higher by 10.05%, 15.10%, 20.00% and 18.44% (two-year average) compared to N0 treatment on October 9, but there was no significant difference between N3 and N4 treatments. However, he application of the N fertilizer had different effects on the daily decomposition rate of straw during rice growth. The average daily decomposition rates of straw for the N0, N1, N2, N3, and N4 treatments were 0.047, 0.057, 0.069, 0.073, and 0.073 g d−1(two-year average), respectively, between May 9 and July 9. And the average daily decomposition rates of straw in N0, N1, N2, N3, and N4 treatments were 0.025, 0.024, 0.023, 0.018, and 0.018 g d−1 (two-year average), respectively, between July 9 and September 9.
Effects of different N application rates on rice straw decomposition. Data are expressed as the mean ± standard error (n = 3). Different lowercase letters on the vertical bars of the same year indicate statistical significance between different fertilizer application at the p < 0.05 level. A represents October 2019 to October 2020; B represents October 2020 to October 2021. N0, N1, N2, N3 and N4 were fertilized with 0, 90, 180, 270 and 360 kg ha−1 of N, respectively
Dynamics of rice straw nutrient composition
Rice straw carbon release was mainly concentrated in the rice growing season (May-September), while straw carbon release was very slow in the non-rice growing season (October-May) (Fig. 5). During the rice growing season, carbon release from rice straw exhibited an initial rapid rate followed by a gradual slowdown. The overall carbon release from straw ranged from 50.09 to 60.92% in the first year of the experiment compared to 50.54-63.01% in the second year. In addition, the application of N fertilizer significantly increased the cumulative carbon release rate of rice straw. Compared with N0, the cumulative carbon release rate of straw increased by 11.13%, 17.34%, 23.14%, and 21.76% in N1, N2, N3, and N4 treatments (two-year average), but there was no significant difference between N3 and N4 treatments during the experiment.
Effects of different N application rates on carbon cumulative release rates of rice straw. Data are expressed as the mean ± standard error (n = 3). Different lowercase letters on the vertical bars of the same year indicate statistical significance between different fertilizer application at the p < 0.05 level. A represents October 2019 to October 2020; B represents October 2020 to October 2021. N0, N1, N2, N3 and N4 were fertilized with 0, 90, 180, 270 and 360 kg ha−1 of N, respectively
There was very little change in straw N release rate and C/N from straw application (mid to late October) to the following year before spring plowing (May 9) (Fig. 6). The straw N release rate was low during the experiment, ranging from 28.51 to 40.57% in the first year and 31.76–45.70% in the second year. Compared with N0, the cumulative release rate of straw N increased by 5.94%, 9.85%, 13.00%, and 11.5% in N1, N2, N3, and N4 treatments (two-year average), but there was no significant difference between N3 and N4 treatments during the experiment. The C/N in straw decreased continuously with time. After a complete rice growing season, the C/N in straw was 54.29–58.25. In addition, the application of N fertilizer reduced the C/N in residual straw. The C/N of residual straw in the N2, N3, and N4 treatments was significantly lower than that in the N0 treatment between July 9 and October 9. However, there was no significant difference in residual straw C/N among N2, N3, and N4 treatments throughout the experiment period.
Effect of different N application rates on N cumulative release rates (A and B) and C/N (C and D) of rice straw. Data are expressed as the mean ± standard error (n = 3). Different lowercase letters on the vertical bars of the same year indicate statistical significance between different fertilizer application at the p < 0.05 level. A and C represents October 2019 to October 2020; B and D represents October 2020 to October 2021. N0, N1, N2, N3 and N4 were fertilized with 0, 90, 180, 270 and 360 kg ha−1 of N, respectively
The release of P and K from rice straw was mainly concentrated in the non-rice growing season (October-May), while the release of P and K from straw entered a slow release stage after June 9 (Fig. 7). The cumulative P release rate from rice straw after a long winter until spring plowing (May 9) was about 65%. After rice harvest, straw P release rates were 74.58–80.52% (2020) and 78.28–83.58% (2021), respectively. N fertilizer application promoted the release of phosphorus from straw during the rice growing season. The cumulative P release rate from straw was significantly higher by 3.65%, 5.36%, 7.35%, and 6.90% (two-year average) in N1, N2, N3, and N4 treatments compared to N0 treatment on October 9, but there was no significant difference in P release rate from straw between N3 and N4 treatments. The K release rates in straw from straw application (mid-late October) to the following year before spring plowing (May 9) were 78.77-79.16% (2020) and 81.52–81.70% (2021). And the cumulative release of K from rice straw was as high as 92% on June 9 and then maintained between 93.45% and 96.01%. However, there was no significant effect of N fertilization on the release of K from straw during the experiment.
Effect of different N application rates on phosphorus (A and B) and potassium (C and D) cumulative release rates from rice straw. Data are expressed as the mean ± standard error (n = 3). Different lowercase letters on the vertical bars of the same year indicate statistical significance between different fertilizer application at the p < 0.05 level. A and C represents October 2019 to October 2020; B and D represents October 2020 to October 2021. N0, N1, N2, N3 and N4 were fertilized with 0, 90, 180, 270 and 360 kg ha−1 of N, respectively
The above results showed that the release of P and K in straw was faster, while the release of C and N was slower, and the sequence of nutrient release in straw was: K > P > C > N.
Decomposition rates of rice straw cellulose, hemicellulose, and lignin
The decomposition rates of cellulose, hemicellulose and lignin in rice straw under different treatments are shown in Fig. 8. The cumulative decomposition rates of cellulose, hemicellulose and lignin in straw gradually increased as the time of straw degradation increased. The cellulose, hemicellulose and lignin decomposition rates of straw were very low from the time of straw application until spring plowing (May 9). The decomposition rate of cellulose and hemicellulose of straw was faster between May 9 and July 9, while the degradation of cellulose and hemicellulose of straw gradually slowed down between July 9 and October 9. The decomposition rate of lignin was slow during the first month after spring plowing, while it was faster between June 9 and August 9. The cumulative decomposition rates of cellulose, hemicellulose and lignin in rice straw ranged from 43.25 to 55.09%, 51.45–62.24% and 28.68–43.28% (two-year average), respectively. This indicates that lignin in straw is more difficult to decompose than cellulose and hemicellulose. On October 9, the cumulative decomposition rates of cellulose, hemicellulose, and lignin of different treatments showed N3 > N4 > N2 > N1 > N0, but there was no significant difference in the decomposition rates of organic fractions of straw between N4 and N3 treatments during the experiment.
Effect of different N application rates on the cumulative release rates of cellulose (A and B), hemicellulose (C and D) and lignin (E and F) from rice straw. Data are expressed as the mean ± standard error (n = 3). Different lowercase letters on the vertical bars of the same year indicate statistical significance between different fertilizer application at the p < 0.05 level. A, C and E represent October 2019 to October 2020; B, D and F represent October 2020 to October 2021. N0, N1, N2, N3 and N4 were fertilized with 0, 90, 180, 270 and 360 kg ha−1 of N, respectively
SEM images of rice straw surface structure
According to the decomposition of straw, the two treatments with the lowest decomposition rate (N0) and the highest decomposition rate (N3) were selected, and the surface structure of straw was observed by Search Engine Marketing (SEM). The surface morphology of rice straw changed significantly during the decomposition process (Fig. 9). The surface of the original rice straw was flat and tightly structured, covered by a layer of wax and without hollows. On June 9, the rice straw began to show more obvious signs of decomposition, the flat surface of the straw was disrupted, the tight structure became loose, the epidermal hairs and other tissues on its outer surface had begun to fall off and decompose, and the wax-silica layer gradually became thinner. On July 9, large cavities appeared on the surface of the straw, and the epidermal tissue below its waxy-silicified layer had gradually appeared, and the cell wall and complex fiber structure had been obviously destroyed; the straw showed a fractured shape and uneven faults, and many pores of different sizes appeared on the surface. On October 9, the overall structure of the rice straw was severely damaged, and the basic tissues had been thoroughly decomposed, in which the hard-to-degrade waxy layer, silicified structure and large vascular bundles had disappeared, leaving only part of the highly lignified mechanical tissues. The above results showed that with the increase of decomposition time, the straw structure was severely damaged, the surface became rough, the fractures increased, the cavities increased, and the fiber bundles became loose and formed a near-net-like structure. Comparing N0 and N3 treatments, it is obvious that rice straw was more damaged under high N conditions, indicating that the decomposition effect of rice straw was significantly better under N application than without N treatment.
Rice yield
The application of N fertilizer significantly increased the rice yield in saline sodic paddy fields under straw return conditions (Fig. 10). Rice yield increased and then decreased with increasing N application rate, with the highest yield at N3 (270 kg N ha−1). However, there was no significant difference in rice yield between N2 (180 kg N ha−1) and N4 (360 kg N ha−1) treatments. The average two-year rice yield under N3 was 8.01 t ha−1. Rice yield was significantly higher in N3 by 10.06% (2020) and 11.13% (2021) compared to N4. This indicates that excessive N fertilizer application reduces rice yield.
Rice yield under different N application rates in 2020 (A) and 2021 (B). Data are expressed as the mean ± standard error (n = 3). Different lowercase letters on the vertical bars of the same year indicate statistical significance between different fertilizer application at the p < 0.05 level. N0, N1, N2, N3 and N4 were fertilized with 0, 90, 180, 270 and 360 kg ha−1 of N, respectively
Discussion
Effect of N fertilization on straw decomposition in saline sodic paddy fields
The process of straw decomposition is influenced by many factors, such as straw quality, soil temperature, soil properties, tillage practices, and landfill depth (Zhong et al. 2017; Min et al. 2022; Nakajima et al. 2015; Li et al. 2020). The most influential factors for the decomposition of rice straw are soil temperature and soil properties in sodic saline areas of northeast China (Zhang et al. 2019; Yan et al. 2019). The study revealed that the rate of rice straw decomposition was significantly low from the time of straw incorporation (mid-late October) until the following spring plowing on May 9. However, the decomposition of straw showed an initial rapid phase followed by a slower phase between May and October (Fig. 4). This is generally consistent with the study of Yan et al. (2019). This may be due to the fact that the experimental site is located in the western part of the Songnen Plain in northeast China, which has a long winter season (Fig. 1), with a deeper soil freeze and longer freezing time, resulting in slow decomposition of straw in winter (Yan et al. 2019). And as the temperature rises and the soil thaws, microbial activity strengthens, rice straw contains a large amount of easily decomposed organic matter, which is easily used by microorganisms, leading to faster decomposition (Chen et al. 2020; Yadvinder-Singh et al., 2010). As decomposition proceeds, the number of microorganisms in the soil decreases (Zhong et al. 2020). The easily decomposed organic matter in straw gradually decreases, and what remains is mainly crude fiber and lignin that are difficult to decompose, which are difficult to be easily used by microorganisms, leading to slow decomposition at a later stage (Sun et al. 2021; Jennifer et al. 2018). In addition, the cumulative straw decomposition rate was higher in 2021 than in 2020 in this study (Fig. 4). This may be due to the application of straw return could effectively improve soil aggregate structure and change microbial community composition as well as their metabolic activity, which in turn favors the accumulation of exogenous organic C in the tested soil (Zhang et al. 2019). Simultaneously, the N, phosphorus, and potassium content in the soil increased, while soil bulk density, pH, ECe, ENa, and ESP decreased, leading to an overall enhancement of the soil’s physical and chemical properties (Ran et al. 2022; Gao et al. 2024). Furthermore, the soil temperature in 2021 was slightly higher than in 2020 (Fig. 3), which facilitated straw decomposition in the second year.
After crop straw is returned to the soil, the straw decomposition process is accompanied by complex microbial activities (Bao et al. 2020). The present study showed that the application of N fertilizer promoted the decomposition of rice straw in saline sodic rice fields (Fig. 4), which was similar to the findings of Zeng et al. (2020). The carbon and N content of straw is crucial for straw decomposition, and usually the initial C/N of straw can be an important indicator to predict the decomposition dynamics of straw (Taylor et al. 1989). The appropriate C/N for microbial degradation of organic matter is 25, and either too high or too low C/N will affect the microbial decomposition of straw and the release of straw nutrients (Truong et al. 2019). Straw with a C/N higher than 25 is returned to the soil, creating a soil environment rich in carbon sources and poor in N sources for microorganisms. If the inorganic N supply in the soil N pool is insufficient, straw decomposition will be slowed down (Recous et al. 1995). The original C/N of rice straw was about 80 in this study (Table 2), much higher than 25, while the soil was extremely poor (Table 1) and could not provide a large source of N for microorganisms, which seriously affected the decomposition of straw. The application of N fertilizer provided a N source for microorganisms, thus accelerating the decomposition process of straw (Zhao and Zhang 2018). In addition, although microbial community composition and function were not affected by N application, N fertilizer application significantly increased the rate of straw microbial assembly, resulting in different decomposition rates of straw at different N application rates (Zhong et al. 2020).
However, this study also found no significant difference in straw decomposition rates between 270 kg N ha−1 and 360 kg N ha−1. Zhang et al. (2017) found that soil microbial biomass C and N and relative abundance of fungi and bacteria increased under low N addition as microbial demand for N was alleviated. However, it decreases under high N addition due to toxic inhibition effects and the growth of microbial decomposers peaks at intermediate levels of N availability (Hogervorst et al. 2003; Jing et al. 2021). Therefore, high N fertilizer application does not continue to increase the rate of straw decomposition, which may be due to limitations on the activity of decomposing microorganisms (Berg and Mcclaugherty 2013). In addition, this study also found an interesting phenomenon that the average daily decomposition rate of straw in the N application treatment was lower than that in the N0 treatment during the mid and late stages of rice growth (July-September) (Fig. 4). This may be due to N can bind to the phenolic hydroxyl group of the lignin benzene ring in the straw residue, forming a more stable aniline and slowing down the decomposition of lignin (Xu et al. 2017). Additionally, the application of N fertilizer promoted the growth of rice in saline sodic fields, increased the leaf area of rice (Fig. 2), and formed a shading effect, resulting in lower soil temperature in the N application treatment than in the N0 treatment (Fig. 3), which in turn affected the average daily decomposition rate of straw in the middle and late stages of rice growth. Nakajima et al. (2015) also showed that the increase in soil temperature promoted the decomposition of rice straw.
Effect of N fertilizer application on nutrient release from rice straw in saline sodic paddy fields
Rice straw contains nutrients such as carbon, nitrogen, phosphorus and potassium (Table 2). Straw nutrient release is regulated by soil microbial-mediated immobilization, mineralization of organic matter, and humification (Vidal et al. 2021; Li et al. 2022). Since humification occurs mainly in recalcitrant straw, nutrient release from straw depends on the net balance between immobilization and mineralization mediated by soil microorganisms (Sajid et al. 2022; Vidal et al. 2021;). In the present study, the release of C, N, P and K from straw after one year of decomposition of 50.32–62.97%, 30.74–43.14%, 76.43–82.05% and 93.25–96.56%, respectively (K > P > C > N), which is in agreement with Yan et al. (2019). The highest release rate of potassium was found in straw, which may be due to the fact that most of the potassium in straw exists in water-soluble or ionic state and is easily soluble, and therefore released more rapidly (Yan et al. 2019). Usually, some of the phosphorus in straw constitutes a component of the cell membrane, and the remaining 60% of phosphorus is present in the ionic state in the straw (Sun et al. 2021). Therefore, the release of phosphorus is slower than that of potassium. Both carbon and N in straw are mainly present in the organic state that is difficult to decompose, resulting in a slow release rate. During straw decomposition, microbial activity intensifies and sequestered N, and straw total N content increases (Chaves et al. 2021). Therefore, the net N release rate is slower than the carbon release rate in straw. This is the primary reason for the notable decrease in the carbon to N ratio in the straw following N application (Fig. 6). As the straw decomposes, its composition stabilizes, leading to a gradual narrowing of this difference. In addition, this study also found that N fertilizer application promoted the release of C, N, and P nutrients from rice straw (Figs. 5, 6 and 7). In addition, this study found that the application of N fertilizer enhanced the release of C, N, and P in rice straw (Figs. 5, 6 and 7). This was further supported by the increased cumulative decomposition rate of straw with N fertilizer application (Fig. 4), as the decomposition of straw leads to nutrient release. As N fertilizer application increased, the levels of ammonia N and nitrate N in the soil also rose gradually (Ran et al. 2022). However, excessive N concentration may hinder the release of N from straw. Prior research has indicated that high N levels can impede microbial decomposition (Hogervorst et al. 2003), leading to the formation of more stable compounds when N combines with the phenol hydroxyl group of lignin benzene ring (Xu et al. 2017), thereby slowing the increase of organic carbon (Wu et al. 2019). Consequently, compared to N3, N4 was found to inhibit the release of C and N in straw (Figs. 5 and 6). In conjunction with the previous finding that excessive N application can increase greenhouse gas emissions (Cao et al. 2021), it can be inferred that excessive N application in conjunction with straw returning may enhance soil carbon and N mineralization, ultimately hindering soil carbon and N sequestration.
Lignin, hemicellulose and cellulose are important components of rice straw, and their decomposition is mainly influenced by the corresponding microorganisms (Deng et al. 2017). In the present study, the cumulative decomposition rates of hemicellulose and cellulose were higher than those of lignin at the same N level (Fig. 8), which is consistent with the results of Sun et al. (2021). This could be attributed to the complex structure of lignin, which is made up of carbohydrates connected by stable biochemical bonds with high molecular weight (Gupta et al. 2016). The intricate nature of lignin hinders extracellular enzymes from accessing the cell to participate in decomposition (Hashimah et al. 2013), while different microorganisms exhibit variations in selecting and utilizing carbon sources (Kuzyakov and Bol 2006). In addition, this study found that N fertilizer application significantly promoted the decomposition of hemicellulose, cellulose and lignin in straw (Fig. 8). The application of N fertilizer reduced the C/N of straw (Fig. 6) and also provided a N source for soil microorganisms, which was more favorable for straw decomposition (Zhao and Zhang 2018). At the same time, the application of N fertilizer enhanced the activity of hydrolytic enzymes associated with cellulose and hemicellulose, which in turn promoted the decomposition of cellulose and hemicellulose in straw (Sinsabaugh et al. 2002). Soil properties are also a key factor influencing the decomposition of straw. The physicochemical properties were extremely poor in the soil in this study, with pH > 8.5 (Table 1), and alkaline conditions promoted lignin degradation (Zhu et al. 2018). Zeng et al. (2020) also found that N fertilizer application promoted lignin decomposition in straw in a fluvo-aquic soil with a pH of 8.5. The presence of high N levels hinders the breakdown of cellulose, hemicellulose, and lignin in straw (Fig. 8). This aligns with the concept of suppressing straw decomposition and nutrient release in high N levels. Additionally, N can react with the phenolic hydroxyl group of lignin in straw residue, forming more stable compounds that slow down the degradation of lignin (Xu et al. 2017).
Rice straw is mainly composed of epidermis, mechanical tissues, vascular bundles and basic tissues, and decomposition occurs mainly because the thin-walled cells of basic tissues and the bast in vascular bundles are damaged (Sun et al. 2021; Li and Zhong 2021). Scanning electron microscopy (SEM) was able to visualize the structural changes in the appearance and the degree of damage during decomposition of the straw (Li et al. 2015). It was observed that the exterior cavities of the straw residue became larger and more numerous as the decomposition time increased. This was mainly due to the decomposition of a large amount of carbohydrates (including hemicellulose and cellulose), while lignin was more complex in structure and more difficult to be decomposed than other carbohydrates (Zeng et al. 2020). The straw in the N treatment was significantly more damaged than that in the non-N treatment (Fig. 9), further suggesting that N application promotes the decomposition of rice straw.
Effect of N fertilizer application on rice yield under the straw return condition in saline sodic rice fields
The real challenge in sodic saline areas is how to improve the soil at minimal cost while maximizing crop yield. Previous studies have shown that the application of biochar, desulfurized gypsum, aluminum sulfate, and compost can effectively improve the physicochemical properties of saline soils and increase the yield of crops (Mukhopadhyay et al. 2021). However, the cost of these improvement methods is high and it is difficult to achieve promotion. Straw contains a large number of nutrients required for crop growth (Table 2), which has a positive effect on soil improvement and is inexpensive (Wang et al. 2021). However, the incorporation of high C/N rice straw into the soil promoted a large uptake and utilization of soil effective N by microorganisms, leading to N competition between microorganisms and crops (Cheshire et al. 1999; Chaves et al. 2021). Therefore, straw return should be combined with an adapted N fertilizer application. This study showed that N fertilizer application under straw return conditions increased rice yield with the highest yield of 8.01 t ha−1 obtained at 270 kg N ha−1. This may be due to the fact that the application of N fertilizer promoted the decomposition and nutrient release of straw (Figs. 4, 5, 6, and 7), which improved the growth environment of the root system in the middle and late stages of rice growth, enhanced the nutrient uptake by the root system, and thus increased the rice yield (Ran et al. 2022). In addition, the application of excessive N fertilizer prolongs the nutritional growth stage of rice, reduces the efficiency of N fertilizer utilization, and leads to a decrease in rice yield (Wang et al. 2022).
Implications for fertilizer management in saline sodic rice fields
Soils with low initial organic carbon concentrations have greater C sequestration potential because they are farther from their saturation level than soils with high organic carbon concentrations (Stewart et al. 2008). It is worth noting that saline sodic soils are extremely nutrient poor (Table 1), and have considerable C sequestration potential. Thus, carbon sequestration can be achieved while improving soil physicochemical properties through long-term straw return, especially in nutrient-poor saline soils. Our previous studies have also shown that straw return significantly improved the physicochemical properties of saline sodic soils and increased the sequestration of organic carbon in the soil (Ran et al. 2023). However, considering the phenomenon of competition for N between rice growth and straw decomposition, the reasonable application of N fertilizer is particularly important. The cumulative decomposition rate of rice straw was greatest and rice yield was highest at a N application rate of 270 kg ha−1. Therefore, we recommend that straw be applied in combination with 270 kg ha−1 of N fertilizer in saline sodic rice growing areas. Since most of the phosphorus and potassium in straw had been released before spring plowing (May 9), it basically satisfied the demand for phosphorus and potassium in the early stage of rice growth. Therefore, we recommend no or little application of phosphorus and potassium in the basal fertilizer process, and appropriately increase the proportion of phosphorus and potassium in the follow-up fertilizer process to meet the demand of rice growth. However, the N fertilizer requirement during straw decomposition may change as the number of years of straw return increases, soil physicochemical properties improve and soil nutrients accumulate, and soil N supply capacity increases. Therefore, it should be paid long-term attention to the straw decomposition and nutrient release patterns under different N application rates in future research to guide local farmers in a more reasonable application of N fertilizer under straw return.
Conclusion
The decomposition of rice straw in saline soil is a dynamic process. The results indicated that N application could enhance straw decomposition and the release of nutrients (C, N, P). This effect exhibited an increasing trend followed by a decreasing trend as N application increased, peaking at the N3 treatment. Similarly, rice yield demonstrated a corresponding trend with increasing N application. Therefore, the N3 (270 kg N ha−1) treatment could synergistically promote straw decomposition and enhance rice yield, ultimately contributing to an increase in farmers’ income.
Data availability
The data are available from the corresponding author on reasonable request.
References
Amini S, Ghadiri H, Chen CG, Marschner P (2016) Salt-affected soils, reclamation, carbon dynamics, and biochar: a review. J Soils Sediments 16:939–953
Bao YY, Feng YZ, Stegen JC, Wu M, Chen RR, Liu WJ, Zhang JW, Li ZP, Lin XG (2020) Straw chemistry links the assembly of bacterial communities to decomposition in paddy soils. Soil Biol Biochem 148:107866. https://doi.org/10.1016/j.soilbio.2020.107866
Berg B, Mcclaugherty C (2013) Decomposition, Humus formation, Carbon Sequestration. Springer, New York. https://doi.org/10.1007/978-3-030-59631-6
Cao YS, Shan Y, Wu PC, Zhang P, Zhang ZY, Zhao FL, Zhu TB (2021) Mitigating the global warming potential of rice paddy fields by straw and straw-derived biochar amendments. Geoderma 396:115081. https://doi.org/10.1016/j.geoderma.2021.115081
Chaves B, Redin M, Giacomini SJ, Schmatz R, Leonard J, Ferchaud F, Recous S (2021) The combination of residue quality, residue placement and soil mineral N content drives C and N dynamics by modifying N availability to microbial decomposers. Soil Biol Biochem 163:108434. https://doi.org/10.1016/j.soilbio.2021.108434
Chen QY, Niu B, Hu YL, Luo TX, Zhang GX (2020) Warming and increased precipitation indirectly affect the composition and turnover of labile-fraction soil organic matter by directly affecting vegetation and microorganisms. Sci Total Environ 714:136787. https://doi.org/10.1016/j.scitotenv.2020.136787
Cheshire MV, Bedrock CN, Williams BL, Chapman SJ, Solntseva I, Thomsen I (1999) The immobilization of nitrogen by straw decomposing in soil. Eur J Soil Sci 50(2):329–341. https://doi.org/10.1046/j.1365-2389.1999.00238.x
Christopher SF, Lal R (2007) Nitrogen management affects carbon sequestration in north American cropland soils. CRC Crit Rev Plant Sci 26:45–64. https://doi.org/10.1080/07352680601174830
Deng YY, Huang ZX, Ruan WQ, Zhao MX, Miao HF, Ren HY (2017) Co-inoculation of cellulolytic rumen bacteria with methanogenic sludge to enhance methanogenesis of rice straw. Int Biodeterior Biodegrad 117:224–235. https://doi.org/10.1016/j.ibiod.2017.01.017
Ding RX, Wang WY, Zhang Q (2017) Effect of straw mulching on soil respiration and its’ temperature sensitivity under different crop rotation systems. Chin J Eco-Agriculture 25(8):1106–1118 (in Chinese). https://doi.org/10.13930/j.cnki.cjea.170078
FAO and ITPS (2015) Status of the World’s Soil resources (SWSR) – main report. Food and Agriculture Organization of the United Nations and Intergovernmental Technical Panel on Soils, Rome, Italy
Gao JY, Zhao ZX, Zhang YT, Ran C, Geng YQ, Zhang Q, Du J, Guo LY, Shao XW (2024) Straw return with nitrogen fertilizer promotes rice yield through change in nutrient dynamics within soil aggregates in saline sodic soils. J Soils Sediments 24:928–945. https://doi.org/10.1007/s11368-023-03688-4
Gupta VK, Kubicek CP, Berrin JG, Wilson DW, Couturier M, Berlin A, Edivaldo Filho XF, Ezeji T (2016) Fungal enzymes for bio-products from sustainable and waste biomass. Trends Biochem Sci 41:633–645. https://doi.org/10.1016/j.tibs.2016.04.006
Hashimah ARN, Aini ARN, Suraini AA, Ali HM (2013) Production of ligninolytic enzymes by newly isolated bacteria from palm oil plantation soils. BioResources 8(4):6136–6150. https://doi.org/10.15376/biores.8.4.6136-6150
Hogervorst RF, Dijkhuis M, Schaar M, Berg MP, Verhoef HA (2003) Indications for the tracking of elevated nitrogen levels through the fungal route in a soil food web. Environ Pollut 126:257–266. https://doi.org/10.1016/S0269-7491(03)00186-6
IUSS Working Group (2014) World reference base for Soil resources 2014 international soil classification system for naming soils and creating legends for Soil maps. FAO, Rome
Jennifer MB, Kabir GP, Kathleen KT (2018) Litter chemistry influences decomposition through activity of specific microbial functional guilds. Ecol Monogr 88:429–444. https://doi.org/10.1002/ecm.1303
Jia Y, Wang J, Qu Z, Zou D, Sha H, Liu H, Sun J, Zheng H, Wang J, Yang L, Zhao H (2019) Effects of low water temperature during reproductive growth on photosynthetic production and nitrogen accumulation in rice. Field Crop Res 242:107587. https://doi.org/10.1016/j.fcr.2019.107587
Jing H, Li J, Yan B, Wei F, Liu G (2021) The effects of nitrogen addition on soil organic carbon decomposition and microbial C-degradation functional genes abundance in a Pinus tabulaeformis forest. For Ecol Manag 489:119098. https://doi.org/10.1016/j.foreco.2021.119098
Kuzyakov Y, Bol R (2006) Sources and mechanisms of priming effect induced in two grassland soils amended with slurry and sugar. Soil Biol Biochem 38:747–758. https://doi.org/10.1016/j.soilbio.2005.06.025
Li JF, Zhong FF (2021) Nitrogen release and re-adsorption dynamics on crop straw residue during straw decomposition in an alfisol. J Integr Agric 20(1):248–259. https://doi.org/10.1016/S2095-3119(20)63238-5
Li YH, Bai YX, Pan CM, Li WW, Zheng HQ, Zhang JN, Fan YT, Hou HW (2015) Effective conversion of maize straw wastes into bio-hydrogen by two-stage process integrating H2 fermentation and MECs. Environ Sci Pollut Res 22:18394–18403. https://doi.org/10.1007/s11356-015-5016-3
Li DD, Zhao BZ, Olk DC, Zhang JB (2020) Soil texture and straw type modulate the chemical structure of residues during four-year decomposition by regulating bacterial and fungal communities. Appl Soil Ecol 155:103664. https://doi.org/10.1016/j.apsoil.2020.103664
Li Q, Wang L, Fu Y, Lin D, Hou M, Li X, Hu D, Wang Z (2022) Transformation of soil organic matter subjected to environmental disturbance and preservation of organic matter bound to soil minerals: a review. J Soils Sediments. https://doi.org/10.1007/s11368-022-03381-y
Lu R (2000) Analysis methods of Agrochemistry for soils. Chinese Agricultural Science and Technology, Beijing
Meng X, Ran C, Liu B, Zhao Z, Bai T, Zhao M, Cheng Z, Chen G, Geng Y (2022) Effect of straw return with nitrogen fertilizer on photosynthetic characteristics and yield of rice in soda saline-alkali rice paddy fields. Cereal Res Commun 51(2):509–526. https://doi.org/10.1007/s42976-022-00312-y
Min X, Song D, Ma M, Li H, Li S, Zou G, Du L, Liu J (2022) Effects of agronomic measures on decomposition characteristics of wheat and maize straw in China. Sustainability 14:12199. https://doi.org/10.3390/su141912199
Morton MJ, Awlia M, Al-Tamimi N, Saade S, Pailles Y, Negrão S, Tester M (2019) Salt stress under the scalpel–dissecting the genetics of salt tolerance. Plant J 97(1):148–163. https://doi.org/10.1111/tpj.14189
Mukhopadhyay R, Sarkar B, Jat HS, Sharma PC, Bolan NS (2021) Soil salinity under climate change: challenges for sustainable agriculture and food security. J Environ Manage 280:111736. https://doi.org/10.1016/j.jenvman.2020.111736
Nakajima M, Cheng W, Tang S, Hori Y, Yaginuma E, Hattori S, Hanayama S, Tawaraya K, Xu X (2015) Modeling aerobic decomposition of rice straw during the off-rice season in an andisol paddy soil in a cold temperate region of Japan: effects of soil temperature and moisture. Soil Sci Plant Nutr 62:90–98. https://doi.org/10.1080/00380768.2015.1121116
Pan M, Gan X, Mei C, Liang Y (2017) Structural analysis and transformation of biosilica during lignocellulose fractionation of rice straw. J Mol Struct 1127:575–582. https://doi.org/10.1016/j.molstruc.2016.08.002
Pan Y, Tung SA, Yang L, Wang Y, Zhou XB (2022) Effect of Straw Return and Nitrogen Application Rate on the photosynthetic characteristics and yield of Double-Season Maize. J Soil Sci Plant Nutr 22:660–673. https://doi.org/10.1007/s42729-021-00676-w
Qadir M, Tubeileh A, Akhtar J, Larbi A, Minhas PS, Khan MA (2008) Productivity enhancement of salt-affected environments through crop diversification. Land Degrad Dev 19:429–453. https://doi.org/10.1002/ldr.853
Ran C, Gao DP, Liu WY, Guo LY, Bai TQ, Shao XW, Geng YQ (2022) Straw and nitrogen amendments improve soil, rice yield, and roots in a saline sodic soil. Rhizosphere 24:100606. https://doi.org/10.1016/j.rhisph.2022.100606
Ran C, Gao DP, Bai TQ, Geng YQ, Shao XW, Guo LY (2023) Straw return alleviates the negative effects of saline sodic stress on rice by improving soil chemistry and reducing the accumulation of sodium ions in rice leaves. Agric Ecosyst Environ 342:108253. https://doi.org/10.1016/j.agee.2022.108253
Recous S, Robin D, Darwis D, Mary B (1995) Soil inorganic N availability: effect on maize residue decomposition. Soil Biology Biochem 27(12):1529–1538. https://doi.org/10.1016/0038-0717(95)00096-w
Sajid S, Zveushe OK, Dios VR, Nabi F, Lee YK, Kaleri AR, Ma L, Zhou L, Zhang W, Dong F, Han Y (2022) Pretreatment of rice straw by newly isolated fungal consortium enhanced lignocellulose degradation and humification during composting. Bioresour Technol 354:127150. https://doi.org/10.1016/j.biortech.2022.127150
Sinsabaugh RL, Carreiro MM, Repert DA (2002) Allocation of extracellular enzymatic activity in relation to litter composition, N deposition, and mass loss. Biogeochemistry 60(1):1–24. https://doi.org/10.1023/A:1016541114786
Song XD, Zhou GS, Ma BL, Wu W, Ahmad I, Zhu GL, Yan WK, Jiao XR (2019) Nitrogen application improved photosynthetic productivity, chlorophyll fluorescence, yield and yield components of two oat genotypes under saline conditions. Agronomy 9(3):115. https://doi.org/10.3390/agronomy9030115
Stewart C, Paustian K, Conant R, Plante A, Six J (2008) Soil carbon saturation: evaluation and corroboration by long-term incubations. Soil Biol Biochem 40:1741–1750. https://doi.org/10.1016/j.soilbio.2008.02.014
Sun L, Sun Z, Hu J, Yaa O-K, Wu J (2021) Decomposition characteristics, nutrient release, and Structural Changes of Maize Straw in Dryland Farming under Combined Application of Animal Manure. Sustainability 13:7609. https://doi.org/10.3390/su13147609
Taylor BR, Parkinson D, Parsons W (1989) Nitrogen and lignin content as predictors of litter decay rates: a microcosm test. Ecology 70(1):97–104. https://doi.org/10.2307/1938416
Truong T, Kristiansen P, Marschner P (2019) Influence of mulch C/N ratio and decomposition stage on plant N uptake and N availability in soil with or without wheat straw. J Plant Nutr Soil Sci 182:879–887. https://doi.org/10.1002/jpln.201900067
Van-Soest PJ (1963) Use of detergents in the analysis of fibrous feeds II. A rapid method for the determination of fiber and lignin. J Assoc off Anal Chem 49(4):546–551. https://doi.org/10.1093/jaoac/73.4.491
Vidal A, Klöffel T, Guigue J, Angst G, Steffens M, Hoeschen C, Mueller CM (2021) Visualizing the transfer of organic matter from decaying plant residues to soil mineral surfaces controlled by microorganisms. Soil Biol Biochem 160:108347. https://doi.org/10.1016/j.soilbio.2021.108347
Wahdan SFM, Ji L, Schädler M, Wu Y, Sansupa C, Tanunchai B, Buscot F, Purahong W (2022) Future climate conditions accelerate wheat straw decomposition alongside altered microbial community composition, assembly patterns, and interaction networks. ISME J 17:238–251. https://doi.org/10.1038/s41396-022-01336-2
Walkley A, Black LA (1934) An examination of the Degtjareff method of determining soil organic matter, and a proposed modification of the chromic acid titration method. Soil Sci 37:29–38. https://doi.org/10.1097/00010694-193401000-00003
Wang H, Takano T, Liu S (2018) Screening and Evaluation of Saline–Alkaline Tolerant Germplasm of Rice (Oryza sativa L.) in Soda Saline–Alkali Soil. Agronomy 8:205. https://doi.org/10.3390/agronomy8100205
Wang YL, Wu PN, Mei FJ, Ling Y, Qiao YB, Liu CS, Leghari SJ, Guan XK, Wang TC (2021) Does continuous straw returning keep China farmland soil organic carbon continued increase? A meta-analysis. J Environ Manage 288:112391. https://doi.org/10.1016/j.jenvman.2021.112391
Wang JJ, Hussain S, Sun X, Zhang P, Javed T, Dessoky ES, Ren XL, Chen XL (2022) Effects of nitrogen application rate under straw incorporation on photosynthesis, productivity and nitrogen use efficiency in winter wheat. Front Plant Sci 13:862088. https://doi.org/10.3389/fpls.2022.862088
Wu W, Wang Z, Jin Y, Matsumoto Y, Zhai H (2014) Effects of LiCl/DMSO dissolution and enzymatic hydrolysis on the chemical composition and lignin structure of rice straw. Biomass Bioenergy 71:357–362. https://doi.org/10.1016/j.biombioe.2014.09.021
Wu PN, Wang YL, Li PF, Wang XN, Hou XQ (2019) Effects of straw returning combined with nitrogen fertilizer on spring maize yield and soil physicochemical properties under drip irrigation condition in Yellow River pumping irrigation area, Ningxia, China. Chin J Appl Ecol 30(12):4177–4185. https://doi.org/10.13287/j.1001-9332.201912.022(in Chinese)
Xu Y, Chen Z, Ding W, Fan J (2017) Responses of manure decomposition to nitrogen addition: role of chemical composition. Sci Total Environ 587–588:11–21. https://doi.org/10.1016/j.scitotenv.2017.02.033
Xu ZK, Shao TY, Lv ZX, Yue Y, Liu AH, Long XH, Zhou ZS, Gao XM, Rengel Z (2020) The mechanisms of improving coastal saline soils by planting rice. Sci Total Environ 703:135529. https://doi.org/10.1016/j.scitotenv.2019.135529
Yadvinder-Singh, Gupta RK, Jagmohan-Singh G-S, Gobinder-Singh, Ladha JK (2010) Placement effects on rice residue decomposition and nutrient dynamics on two soil types during wheat cropping in rice-wheat system in northwestern India. Nutr Cycl Agroecosyst 88:471–480. https://doi.org/10.1007/s10705-010-9370-8
Yan C, Yan SS, Jia TY, Dong SK, Ma CM, Gong ZP (2019) Decomposition characteristics of rice straw returned to the soil in northeast China. Nutrient CyclAgroecosyst 114:211–224. https://doi.org/10.1007/s10705-019-09999-8
Yang F, An FH, Ma HY, Wang ZC, Zhou X, Liu ZJ (2016) Variations on soil salinity and sodicity and its driving factors analysis under microtopography in different hydrological conditions. Water 8(6):227. https://doi.org/10.3390/w8060227
Yu C, Li Y, Mo RL, Deng W, Zhu ZX, Liu DB, Hu XM (2020) Effects of long-term straw retention on soil microorganisms under a rice–wheat cropping system. Arch Microbiol 202:1915–1927. https://doi.org/10.1007/s00203-020-01899-8
Yuan BF, Ma YT, Bao Y, Zhang JJ, Sun Q, Wang LC (2019) Effect of Rice Cultivation on ameliorating Soil Fertility of Soda saline-alkali soil in Western Jilin Province. J Soil Water Conserv 33(03):320–326 (In Chinese). https://doi.org/10.13870/j.cnki.stbcxb.2019.03.047
Zeng L, Zhang X, Zhang SQ, Wang XB, Liang GQ, Zhou W, Ai C, Zhang YQ (2020) Characteristics of decomposition, nutrient release and structure change of wheat straw in a fluvo-aquic soil under different nitrogen application rates. J Plant Nutr Fertilizers 26(9):1565–1577 (in Chinese). https://doi.org/10.11674/zwyf.20181
Zhang J, Ai Z, Liang C, Wang G, Xue S (2017) Response of soil microbial communities and nitrogen thresholds of Bothriochloa ischaemum to short-term nitrogen addition on the Loess Plateau. Geoderma 308:112–119. https://doi.org/10.1016/j.geoderma.2017.08.034
Zhang XF, Xin XL, Yang WL, Zhu AN, Ding SJ (2019) Short-term decomposition, turnover and retention of residue-derived carbon are influenced by the fertility level in a sandy loam soil. Geoderma 349:68–78. https://doi.org/10.1016/j.geoderma.2019.04.043
Zhang YH, Hu TL, Wang H, Jin HY, Liu Q, Chen Z, Xie ZB (2022) Nitrogen content and C/N ratio in straw are the key to affect biological nitrogen fixation in a paddy field. Plant Soil 481:535–546. https://doi.org/10.1007/s11104-022-05654-4
Zhao S, Zhang S (2018) Linkages between straw decomposition rate and the change in microbial fractions and extracellular enzyme activities in soils under different long-term fertilization treatments. PLoS ONE 13(9):e0202660. https://doi.org/10.1371/journal.pone.0202660
Zhong Y, Yan W, Wang R, Shangguan Z (2017) Differential responses of litter decomposition to nutrient addition and soil water availability with long-term vegetation recovery. Biol Fertil Soils 53:939–949. https://doi.org/10.1007/s00374-017-1242-9
Zhong Y, Liu J, Jia X, Shangguan ZP, Wang R, Yan WM (2020) Microbial community assembly and metabolic function during wheat straw decomposition under different nitrogen fertilization treatments. Biol Fertil Soils 56:697–710. https://doi.org/10.1007/s00374-020-01438-z
Zhu L, Hu N, Zhang Z, Xu J, Tao B, Meng Y (2015) Short-term responses of soil organic carbon and carbon pool management index to different annual straw return rates in a rice-wheat cropping system. CATENA 135:283–289. https://doi.org/10.1016/j.catena.2015.08.008
Zhu GL, Lu HT, Shi XX, Wang Y, Zhi WF, Chen XB, Liu JW, Ren Z, Shi Y, Ji ZY, Jiao XR, Ibrahim MEH, Nimir NEA, Zhou GS (2020) Nitrogen Management enhanced plant growth, antioxidant ability, and Grain Yield of Rice under salinity stress. Agron J 112(1):550–563. https://doi.org/10.1002/agj2.20013
Zhu Y, Liu J, Liao Y, Lv W, Ma L, Wang C (2018) Degradation of vanillin during lignin valorization under alkaline oxidation. Top Curr Chem 376:29. https://doi.org/10.1007/s41061-018-0208-1
Acknowledgements
This study was supported by the National key research and development program (2022YFD1500505) and the Science and Technology Development Program of Jilin Province (20240601059RC).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Competing interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Additional information
Responsible Editor: Alfonso Escudero.
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Jiayong Gao and Cheng Ran contributed equally to this study and share first authorship.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Gao, J., Ran, C., Hou, H. et al. Synergistic improvement of straw decomposition and rice yield in saline sodic paddy soils by rational nitrogen application. Plant Soil (2024). https://doi.org/10.1007/s11104-024-06918-x
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11104-024-06918-x