Abstract
Plasma seed treatment has proven to be a useful technique for improving germination, growth dynamics, as well as plant resistance. In this paper, we studied the efficacy of soybean seeds treatment using various sources of cold atmospheric pressure plasma generated in air. We compared three types of plasma treatments: direct treatment with plasma generated by a diffuse coplanar surface barrier discharge (DCSBD), direct treatment with plasma generated by a multi-hollow surface dielectric barrier discharge, and indirect treatment using the gaseous products of plasma generated by an air plasma jet. The composition of plasma generated by each of the sources was analysed using optical emission spectroscopy and Fourier Transform Infrared spectroscopy (FTIR). Parameters of the plasma treatments have been optimized to improve soybean germination. Plasma-treated seeds were examined by the means of water contact angle, surface free energy and imbibition measurements, attenuated total reflectance FTIR (ATR-FTIR), x-ray photoelectron spectroscopy (XPS) and scanning electron microscope (SEM) analyses. Surface analysis by ATR-FTIR and XPS showed changes in the chemical bonds on the surface of the plasma-treated seeds, which led to an increase in wettability and imbibition. SEM analysis confirmed that the plasma treatment is non-invasive and does not cause changes in seed surface morphology. Among the studied sources, DCSBD proved to be the best suited for soybean seed treatment in terms of germination improvement as well as treatment time and energy efficiency.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
At present, the agricultural and food industries must cope with new challenges, due to the growing population. Current problems in agriculture that need to be addressed, in addition to global population growth, include environmental pollution, scarcity of agricultural land and climate change. Climate change is resulting in a significant reduction in crop yields. Approximately 95% of total annual losses in the agricultural sector are attributed to examples of extreme weather such as droughts, floods, heat waves and irregular rainfall, all directly related to climate change [1]. For these reasons, it is necessary to look for innovative approaches to food production as well as to food processing. Almost a third of the world's food production for human consumption is discarded or wasted [2]. Ecological strategies to increase crop yields and subsequently protect crops and food products, leading to reduced losses and extended shelf life, are therefore a major step towards globally sustainable food production.
One of the new strategies, which has received a lot of attention in recent years, is the use of the so-called Cold Atmospheric Pressure Plasma (CAPP) in agriculture. CAPP is a frequently used term for low-temperature, non-equilibrium plasma generated at atmospheric pressure. The effects of CAPP include seed quality improvement (activation of plant vitality, improvement of plant germination and resistance to adverse external influences) and inactivation of pathogenic microorganisms present on the seed surface (protection of seeds from fungal and bacterial infestation) [3,4,5,6,7,8,9]. The use of CAPP is advantageous due to simple integration of these systems directly into production and lower operating costs compared to low-pressure discharges. Other advantages of CAPP technologies include short processing time, the ability to treat heat-sensitive materials and materials of various shapes, and that it does not produce toxic waste. Thus, the use of plasma in agriculture presents an ecological alternative to typically used chemicals that have a negative impact on the environment.
Plasma treatments of seeds are largely focused on seed germination – the first crucial step in a plant's life cycle. After dispersal from the mother plant, the plant seeds reach a state of dormancy, which will ensure that germination in adverse environmental conditions is avoided. However, even if the conditions are favourable, many seeds do not germinate. It is therefore necessary to break the dormant state in order to initiate germination. In addition to external conditions such as temperature and humidity, dormancy and germination in the seeds is affected by an imbalance of plant hormones (phytohormones) [10]. The phytohormone abscisic acid is responsible for maintaining the state of dormancy, and the phytohormone gibberellin is responsible for its disruption [11, 12]. These hormones are produced in response to external factors. The application of CAPP, which consists of many reactive oxygen and nitrogen species (RONS), charged particles and radiation, acts on the seed as a factor that induces germination in various ways [10]. The possible effect of plasma on plant seeds is the partial disruption of the top layers of the seed coat, whereby reactive particles can interact directly with the internal tissue. Even if the surface is not visibly disrupted or damaged by the effect of plasma, changes in the chemical bonds on the surface can lead to an increase in surface hydrophilicity and water imbibition, which are necessary for seed germination [13,14,15].
Another possible mechanism of plasma-induced germination improvement is the activation of biochemical and molecular processes within the seed by plasma-generated RONS. Plasma generates different compositions of RONS depending on the working gases. These RONS can act as signalling molecules and initiate a germination cascade [10, 16]. The authors of the work [17] aiming to study the effect of plasma on the enzyme activity in peas have shown that low plasma doses act as a low stress factor, which has a paradoxically stimulating effect on seed germination, growth and development. Plasma-induced oxidative stress induces an adaptive response in the seed and contributes to a better resistance of the seeds to adverse conditions [18]. In the currently available literature, many authors agree that the physiological response to plasma treatment is not only specific to a particular species, but also depends on a particular variety [19,20,21].
Investigating the effects of plasma on plant seeds is a very complex task, connecting several scientific disciplines (plasma physics, plant physiology, genetics, microbiology). The evaluation of experimental results requires not only to verify the effect of plasma on plant material, but also to carry out the most comprehensive plasma diagnostics. Plasma-induced changes depend on the properties of the treated material (seed), the used plasma source, working gas composition, exposure time and other parameters. Therefore, it is necessary to examine and optimize the plasma treatment parameters for each type of plant seed separately and compare the efficiency of different plasma sources.
In our work, we focused on investigating the effects of direct and indirect plasma treatment of soybean seeds using low-temperature plasma generated at atmospheric pressure in ambient air by three different plasma devices. We searched for optimal plasma treatment conditions in order to improve germination and growth parameters. We investigated the effect of the plasma treatment on changes in wettability, surface energy, chemical composition and surface morphology. We compared the parameters of the three plasma sources by analysing the chemical composition of their gaseous plasma products and determining their energy consumption for a treatment of a given amount of soybean seeds. When choosing the biological material, we took into consideration that soybean (Glycine max. L), a legume originating from East Asia, is the most important source of concentrated protein and vegetable oil among agricultural crops. It represents more than 50% of total world oilseed production [22]. The importance of soybean as an agricultural crop stem from its versatility, high yield and nutritional potential. Soybean foods for human consumption include tofu, soy milk, soy sauce, miso, tempeh and many more. Soybean meal, a by-product of soybean oil extraction, is used as feed. Soy is used not only in the food industry, but also in the production of pharmaceuticals, plastics, paper, cosmetics, etc. The potential use of soy as a biofuel opens new opportunities in the field of renewable energy sources [23,24,25]. Due to its ability to bind atmospheric nitrogen, which is typical for legumes, soy is also less dependent on nitrogen fertilizers than other agricultural crops.
Various works studied the effects of CAPP on soybean growth, germination, yield and nodulation enhancement [26,27,28]. However, there remains a lack of comparative studies differentiating between multiple plasma sources.
Materials and Methods
Plant Material
Dried soybeans (Glycine max L.; produced by Lagris, Slovakia, country of origin Czech Republic) were acquired from a superstore and stored in a fridge at 8 °C in the dark. Before each plasma treatment and/or measurement, the samples were taken out so that their temperature stabilized to room temperature.
Plasma Sources and Treatment
DCSBD Plasma Treatment
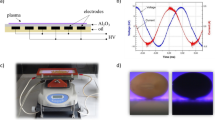
The direct plasma treatment was realised using a special type of dielectric barrier discharge with a coplanar electrode system configuration, the so-called DCSBD – Diffuse Coplanar Surface Barrier Discharge. As is the case with DBDs (Dielectric Barrier Discharges), plasma thermalization at atmospheric pressure is prevented due to the presence of a dielectric barrier in its electrode system and the use of alternating voltage. The schematic of a DCSBD electrode system designed for ambient air plasma generation is shown in Fig. 1A). The electrode system consists of parallel strip-line silver electrodes embedded below the surface of a ceramic plate (Al2O3, 96% purity). The discharge is fed by 15-kHz sinusoidal high voltage with an amplitude of up to 20-kV peak-to-peak supplied by a HV generator VF 700 (Lifetech Ltd., Brno, Czech Republic). DCSBD generates a thin layer (∼0.3 mm) of macroscopically homogeneous plasma at atmospheric pressure on the surface of a ceramic plate, with active plasma area of (20 × 8) cm2. The continuous operation of DCSBD at input power of 400 W is enabled by an effective cooling and insulating oil system. During seed treatment the DCSBD is placed on an orbital shaker PSU-10i (BIOSAN) to ensure rotational motion of the seeds (Fig. 2A). The detailed characteristic and properties of the DCSBD plasma source were reported previously [29].
Plasma treatment of the seeds (30 seeds per each treatment) was carried out in ambient air at exposure times of 15, 30 and 45 s. The seeds were placed into the plasma layer on top of the DCSBD ceramic plate and rotated at a rotary speed of 330 rpm. The movement of soybeans in the discharge area ensured a homogeneous plasma treatment of their surface. The input power of the DCSBD during the treatment was kept at 400 W.
MSDBD Plasma Treatment
The second studied plasma source was the MSDBD – Multi-hollow Surface Dielectric Barrier Discharge (Fig. 2B), a special type of DBD. MSDBD generates non-thermal plasma in a similar manner to DCSBD and additionally enables to use flowing gas. We used a commercial RPS30 multi-hollow device (ROPLASS, Czech Republic). The electrode system consists of two plane-parallel electrodes at a mutual distance of 0.5 mm, fully embedded in ceramic and perforated with 105 holes (Kyocera Japan). A schematic of the MSDBD electrode system is shown in Fig. 1B) and a more detailed description of MSDBD can be found in previous publications [30, 31]. The discharge is generated by sinusoidal alternating high voltage with amplitude of 13 kV (peak-to-peak) and frequency of approximately 27 kHz. As the input power gradually increases, filamentous surface micro-discharges ignite in the cavities. The total active area on which the plasma is generated is (18 × 19) mm2. The advantage of MSDBD over other types of DBD is that it can operate in gas flow mode (up to 20 lpm). The adjustable gas flow provides a great advantage because it ensures the distribution of active plasma particles further away from the ceramic surface and thus a better contact of the diffuse plasma with the surface of the treated material. As a result, the plasma generated by MSDBD is effective at greater distances (e.g., compared to DCSBD, which is only effective in the range of 0.1–0.3 mm from the surface of the discharge ceramic) and it also has the potential to treat structured surfaces.
Plasma treatment of seeds (15 seeds per each treatment) was carried out with the seeds placed directly on top of the discharge surface, while the device was rotating horizontally so that the samples were in motion. Ambient air was fed with a compressor using a RED-Y flow meter (Vögtlin Instruments AG) with a defined constant flow to the plasma source. The flow rate was 9 lpm and the exposure times were 15, 30 and 45 s. The input power of the MSDBD during the treatment was kept at 30 W.
Indirect Plasma Treatment by APJ
The third studied plasma source was a high-power DC air plasma jet (APJ) (Fig. 2C), as a member of atmospheric pressure plasma jets (APPJ), which is particularly effective in producing reactive nitrogen species. The plasma is generated between two electrodes separated by a copper neutrode. The copper cathode with a hafnium insert is connected to the negative polarity of the HV DC power supply with current feedback [32]. The discharge was maintained at an average power consumption of 300 W, a voltage of about 230 V, and a current of 1.3 A. The plasma jet was fed by filtered ambient air with a flow rate of 5 lpm.
Due to high temperatures of the plasma generated by the DC plasma jet, we used an alternative mode of sample treatment: the samples were not in direct contact with the plasma, but only with the long-living gaseous plasma products. The APJ discharge chamber was cooled from the outside by a system of pipes with flowing water. Gaseous plasma products flowed from the discharge chamber into the chamber in which the samples were placed. To ensure that the samples were not exposed to temperatures that could result in thermal damage, we measured the seeds' temperature during a treatment with a Fluke temperature probe. The maximum temperature reached after 5 min of APJ operation was 42.6 °C, which is within the safe range of temperatures for these biological samples. A schematic of the indirect APJ treatment system is shown in Fig. 1C) and its more detailed description can be found in previously published work [32].
Diagnostic Methods
Plasma Sources Diagnostics
OES Analysis
Optical emission spectroscopy (OES) was used to analyse active plasma components in the DCSBD and MSDBD plasma. As the treatment by APJ was indirect, radiation did not affect the samples. In the case of the DCSBD plasma radiation measurement the DCSBD was covered by a lid with a round quartz window to create an enclosed chamber with a gas inlet and outlet. The signal was captured by an optical fibre directed at the quartz window and analysed by a Stellar Net EP-2000 spectrometer in the (200–1100) nm range. The perpendicular distance between the fibre and the DCSBD ceramic was approximately 7 cm. A schematic diagram of the measurement setup can be found in our previous work [33]. The working gas (ambient air) was supplied to the chamber at a flow rate of 3 lpm. In the case of the MSDBD plasma radiation measurement the flow rate of the working gas was 9 lpm and the signal from the plasma source was fed using an optical fibre perpendicular to the MSDBD ceramic from the distance 3 cm.
FTIR Analysis
Studying the chemical composition of gaseous plasma products, which are specific for each plasma source, is important for understanding the effect of each active particle species on the treated surface. Gaseous plasma products generated by DCSBD, MSDBD and APJ were characterized by FTIR spectroscopy using a Bruker Optics Vector 22 spectrometer and a gas cell. IR absorbance spectra were measured in the range (4000–500) cm−1 with a resolution of 2 cm−1 or 4 cm−1. Measurements for each plasma source were performed in dynamic mode – the discharges were switched on for the whole time while the spectra were being acquired. The background spectrum was measured first, after 7 min of gas flowing into the chamber. A schematic diagram of the measurement setup for DCSBD can be found in our previous work [33]. Ambient air pumped by the compressor flowed through a plastic tube into the DCSBD reactor chamber at a flow rate of 3 lpm and then into the gas cell. The length of the tube transporting the plasma products into the gas cell was approximately 50 cm. In the case of MSDBD and APJ, their respective outlets (The MSDBD nozzle and the APJ silicon tube) were directly connected to the gas cell, without additional tubes.
After the discharge had been switched on for 3 min, the spectrum was measured. The measurements for DCSBD products consisted of 10 scans with a resolution of 2 cm−1.
Due to the maximum continuous operation time of MSDBD (45 s) the measurement time had to be reduced to fit into this timeframe, and thus 6 scans with a resolution of 4 cm−1 were taken. The spectra of APJ products were measured with the same number of scans and resolution as the spectra of MSDBD products.
Seeds Surface Diagnostics
The surface of soybean seeds was analysed before and after plasma treatment by several surface diagnostic methods and the effect of all three studied plasma sources on different surface properties was monitored. We studied wettability, surface free energy, morphology, as well as chemical bonds on the seeds’ surface.
Wettability and Surface Free Energy
Wettability and surface free energy (SFE) are characteristics that are linked to the seeds’ water uptake and germination. They can be evaluated by the means of contact angle measurements. These measurements were conducted using KRUSS Drop Shape Analyzer DSA30S with appropriate software for semi-automatic measurement of contact angles on curved surfaces. Liquid droplets of volume 2 μl were dropped on the seed surfaces and their projection was recorded with a high-resolution camera. On the acquired image, the curve of the seed surface where the droplet resided was approximated by a circle and the corresponding contact angle between the droplet and the surface was obtained. This measurement was carried out on 12 samples each time, the highest and the lowest value were removed, therefore the resulting contact angle for one set of samples was an average of 10 values. This process was repeated with three test liquids: deionized water, ethylene glycol and diiodomethane. SFE and its polar and dispersive components were then calculated by applying the Owens–Wendt-Rabel-Kaelble (OWRK) method on the average contact angles of these three liquids.
Imbibition
Imbibition measurements were performed to assess the effect of plasma treatment on the seeds’ ability to absorb water, which plays an important role in their subsequent growth process. Each sample set consisted of 50 seeds. Weighing of samples was performed on laboratory scales KERN ABT-220 (sensitivity 10–4 g).
The sample set was weighed on laboratory scales, treated with a plasma source and soaked in deionized water immediately after the plasma treatment. Every hour, the seeds were taken out of the water, excess water was removed using filter paper, and the seeds were weighed. After weighing, the seeds were put back into fresh deionized water. This process was repeated for the duration of 5 h. The water intake m% was calculated according to equation:
where wt is the actual weight after weighing at time t and w0 is the initial weight.
Germination
After plasma treatment, dry soybean seeds (30 seeds for each variant) were weighed on analytical scales and soaked in sterile distilled water for 1 h at room temperature. Imbibed seeds were blotted dry and wrapped in wet sterile filter paper. Rolls were cultivated in dark conditions in an incubator at the temperature (24 ± 2) °C for 5 days. During cultivation we counted the number of germinated seeds. We used this data to calculate germination percentage according to Abdul-Baki and Anderson (1973) [34]:
where n represents the number of seeds that germinated on a given day, N represents the total number of seeds.
The data was analysed using Microsoft Excel (Microsoft Office 2013) and Statgraphic Centurion 19 (Statgraphics Technologies, Inc. The Plains, Virginia). Treatment effects were analysed by means of ANOVA multiple comparisons and comparisons between the mean values were considered significant at P < 0.05. All experimental data for the statistical analysis in this work are from at least three independent experiments.
SEM
Detailed micro-morphological characteristics of untreated and plasma-treated seeds were assessed by scanning electron microscopy (SEM), using SEM microscope Tescan Vega II SBH (Tescan, CZ), with an accelerating voltage of 30 kV and magnification 1000x. Before the microscopy, the samples were plated with gold using the SEM Coating System (BIO-RAD E52-5199) at pressure 8 Pa in argon. The thickness of the gold layer was 10 nm. The soybean samples subjected to this analysis were treated by each plasma source for 45 s.
ATR-FTIR
Changes in the chemical bonds of the soybean seeds’ surface due to interaction with plasma were studied by Attenuated Total Reflectance–Fourier Transform Infrared Spectroscopy (ATR-FTIR). The ATR-FTIR analysis was carried out using a Bruker Vector 22 FTIR spectrometer, with the addition of a single-reflection ATR accessory Pike MIRacle™ with a diamond crystal. The spectra were acquired in the range of 4000–500 cm−1, each consisting of 20 scans with 2 cm−1 resolution. The studied samples were treated by each plasma source for 45 s.
XPS
The chemical composition of the soybean surface was investigated by X-ray Photoelectron Spectroscopy (XPS). We used XPS as a suitable method for determining chemical composition of the surface as well as chemical bonds and electronic state of the individual elements on the surface. The XPS measurements were performed using the ESCALAB 250Xi device (Thermo Fisher Scientific, East Grinstead, United Kingdom at the Faculty of Science, Masaryk University in Brno). An X-ray beam with power of 200 W (650 microns spot size) was used. The survey spectra were acquired with a pass energy of 50 eV and energy step of 1 eV. High-resolution scans were acquired with a pass energy of 20 eV and energy step of 0.1 eV. In order to compensate for charges on the surface, an electron flood gun was used. The spectra were referenced to the hydrocarbon type C 1 s component set at a binding energy of 284.8 eV. Spectra calibration, processing and fitting routines were done using the Avantage software. Exposure times of the treated samples subjected to this analysis were 15 s and 45 s for DCSBD and MSDBD, 45 s and 5 min for APJ.
Results and Discussion
In the presented experimental results we use the following designation: D15, D30, D45 for the plasma treatment performed with DCSBD at exposure times 15 s, 30 s, 45 s; M15, M30, M45 for the plasma treatment performed with MSDBD at exposure times 15 s, 30 s, 45 s; and J45, J3, J5 for the indirect APJ treatment performed at exposure times of 45 s, 3 min, 5 min. Results of seeds without plasma treatment (reference samples) are denoted as ref.
OES analysis
Typical optical emission spectra of plasma generated using DCSBD and MSDBD in air at input power of 400 W for DCSBD and 30 W for MSDBD are shown in Fig. 3. The most intensive radiating systems in the atmospheric pressure air plasma are the nitrogen transitions: the second positive system of nitrogen N2(C-B), first negative system N2+(B-X), and first positive system N2(B-A). The NO(A-X) system is visible in the spectrum of DCSBD plasma only. OES measurements confirmed that the DCSBD plasma generated in air emits ultraviolet radiation in the UV regions: in UV-A (315 to 400 nm), UV-B (280 to 314 nm) and UV-C (10 to 279 nm). The DCSBD plasma vibrational temperatures determined by OES are approximately (2200–3500) K depending on the input power and the rotational temperatures are approximately 380 K, as was estimated by a more detailed spectroscopy study [35]. The order-of-magnitude temperature difference is evidence that the plasma generated in this way has a non-equilibrium character. Additionally, the measured rotational temperature does not correspond to the temperature of the plasma as a whole, which is significantly lower. One of the typical properties of the DCSBD plasma is that you can touch it with bare hands, which shows its “cold” nature. Moreover, since the plasma layer is 0.3 mm thin, the seeds move through the plasma, and heat is being transferred to the surrounding air, their temperature doesn’t reach levels at which thermal damage would occur.
FTIR Analysis
FTIR spectroscopy provides useful information on the composition of gaseous plasma products present in each seed treatment. The FTIR spectra of the three studied plasma sources are shown in Figs. 4, 5 and 6.
In the typical FTIR spectrum of DCSBD plasma gaseous products (Fig. 4), we can observe bonds belonging to HNO2 (at 1700 cm−1, 1270 cm−1, 891 cm −1, 852 cm−1), N2O (3482 cm−1, 2578 cm −1, 2220 cm−1, 1270 cm−1), NO (1850 cm−1, 1900 cm−1) and the most dominant peak attributed to NO2 (at 2916 cm−1, 1630 cm−1) [36, 37]. It can be noted that ozone peaks are missing in this spectrum. DCSBD plasma in air generates ozone, however at a higher input power its concentration declines rapidly. This is caused by the significant consumption of ozone in reactions due to the presence of humidity, UV radiation and elevated temperature [35]. Considering that plasma products are transported from the discharge chamber to the gas cell through a supply tube, ozone can also be depleted on its walls.
In the FTIR spectrum of products generated by MSDBD (Fig. 5), in comparison to the spectrum of DCSBD plasma, the peak attributed to ozone (1055 cm−1) dominates. We can also notice the presence of HNO3 (at 1700 cm−1, 1325 cm−1, 878 cm−1) and N2O5 (at 1720 cm−1, 1247 cm−1, 743 cm−1) [37,38,39], which did not appear in the DCSBD spectrum.
The FTIR spectrum of APJ (Fig. 6) shows, similarly to the spectrum of MSDBD, the presence of NO2, N2O5 and HNO3. Additionally, peaks belonging to N2O4, HNO2 and NO (1850 cm−1, 1900 cm−1) appear in the spectrum [37,38,39]. We were unable to precisely determine the position of some peaks around 1750 cm−1, 1600 cm−1 and 1260 cm−1, because the peaks overlapped. The main gaseous products of plasma generated in ambient air using the individual plasma sources are summarized in Table 1.
Wettability and Surface Free Energy
The wettability of the soybean seeds was evaluated using WCA measurements. As illustrated by the obtained results in Fig. 7, the largest decrease of WCA from the reference value was recorded on samples treated with DCSBD plasma, and a less pronounced decrease was observed on MSDBD and APJ treated samples. More detailed information on surface changes was provided by the measurement of SFE, where the proportion of dispersive and polar components was determined. The graph in Fig. 7 shows that after the plasma treatment, the polar part of SFE increases, which is responsible for the hydrophilization of the surface. This is a result of the interaction between the plasma particles and the surface, which leads to binding of oxygen containing groups on the reactive centres of the surface and thus the formation of polar groups.
Imbibition and Germination
The weight gain percentage due to water intake of the untreated samples and samples treated with DCSBD, MSDBD and APJ for 45 s is shown in Fig. 8. Surprisingly, the highest water intake during the entire measurement period was reached by the samples treated with APJ. After the first hour, the weight of the samples increased by 47%, which was more than the weight gain of the untreated samples after two hours (43%). After 5 h the weight gain increased to 110%. The samples treated with DCSBD performed almost as well (44% after the first hour and 108% after 5 h). Significantly lower values were achieved by the samples treated with MSDBD (92%) and the untreated samples (86%) after 5 h. The difference in water intake of the latter two was 11% after the first hour and decreased by half after 5 h. A longer measurement could show if this trend would continue and whether the untreated samples would reach better imbibition than the ones treated by MSDBD. These results are in agreement with similar results published in previous works [15, 19, 40], where plasma treatment also led to an increase in imbibition. According to many authors, increased water intake is closely related to plasma modifications of the seed surface caused by higher doses of plasma [15, 17, 41]. However, it should be noted that better imbibition does not guarantee improvement in germination and growth parameters. Increased water intake can be beneficial and may lead to faster germination, but in some cases, rapid water absorption can cause, on contrary, a reduction in the rate of germination or suffocation of the embryo.
The germination process of plasma treated soybean seeds was observed for 5 days. In Fig. 9 there can be seen a comparison of the germination dynamics of plasma-treated seeds and untreated seeds. Statistical processing shows that DCSBD treatment is more efficient even after a short treatment time (15 s), germination starts earlier (2nd day), probably due to the high power density of the plasma. MSDBD treatment is more efficient at a longer exposure time (45 s). Indirect treatment with APJ-generated products is less efficient, requiring longer treatment times. The effect on germination is dependent on the used plasma source as well as on exposure time. It is documented by application of cold low-pressure radiofrequency air plasma on wheat, lentil and bean seeds [15], by application of helium plasma discharge on soybean seeds [42] as well as by our previous work using DCSBD plasma generated in ambient air, oxygen and nitrogen applied on soybean seeds [43]. Inhibition of germination was recorded after application of plasma generated in nitrogen on soybean seeds for longer time (90 s, 120 s) [43].
SEM
By analysing the images acquired with the SEM microscope (Fig. 10), we can evaluate whether the plasma treatment using DCSBD, MSDBD and APJ affects the morphology of the soybean seed surfaces.
We do not observe any unambiguous differences in the effects of different plasma sources in the SEM images of individual samples (Fig. 10). The surface of the MSDBD and APJ treated samples may appear smoother than the surface of the other samples, but we cannot say whether this is a local or global property of the surface, because the soybean seed surfaces are characterized by significant irregularities. This may also be due to the MSDBD and APJ plasma products being blown onto the samples and thus removing the fine impurities on their surface with the gas flow. Most importantly, we do not observe any destructive effects of the plasma treatment on the surface morphology of the samples. Therefore, we can state that all three plasma sources are gentle on the processed material in this respect.
ATR-FTIR
To verify the changes in surface bonds induced by the plasma treatment, we investigated the ATR-FTIR spectra of reference soybean seed surface as well as the surfaces after the treatment using DCSBD, MSDBD and APJ at the same exposure time 45 s. In the ATR-FTIR spectra (Fig. 11) we observe the typical bands characteristic for carbohydrates, proteins and lipids derived from starch and cellulose. The wide peak between 3600 and 3000 cm−1 belongs to the stretching vibrations of O–H and N–H bands mostly from water, polysaccharides, and proteins [44, 45]. Carbohydrates are represented by the C–O, C–C, C–O–H bands (1200–800 cm−1), lipids and proteins by stretching vibrations of the C-H band (2950—2850 cm−1), proteins by the N–H band (3200 cm−1), C=O band (1640 cm−1), C–N and N–H bands (1550–1520 cm−1). After the plasma treatment, we do not observe any changes in the spectra of the samples that would indicate the destruction of these basic chemical bands belonging to the components forming the soybean seed coat. On the spectra of plasma-treated samples, a slight increase in intensities can be observed at 1740 cm−1, which belongs to the C=O–carbonyl group, and 1640 cm−1, which belongs to the O–H band. This indicates the attachment of polar oxygen groups on the surface. Since the depth of sample penetration of the FTIR-ATR is in the order of µm, which is very high compared to the depth of penetration of active substances from the plasma (tens of nanometres), with this method it is not possible to detect changes in chemical bonds on the surface at the nm level.
XPS
Additional information about the surface composition and chemical changes after plasma treatment of soybean seeds at a much smaller depth of the sample was obtained by XPS analysis (average analysis depth of about 3–10 nm depending on the nature of the material being analysed). Treatment times of 15 s and 45 s in the case of DCSBD and MSDBD, and 45 s and 5 min in the case of APJ were used for the XPS analyses. The survey XPS spectra of soybean seeds reveal mainly the presence of carbon, oxygen, and nitrogen. The chemical composition of the soybean seeds surfaces before and after plasma treatment collected in Table 2 shows higher amount of carbon atoms and lower amount of oxygen. In addition, nitrogen was also detected as a basic element of the surface, which was also expected from the natural composition of the seed coat, consisting mainly of lipids and carbohydrates derived from starch and cellulose, as was confirmed by ATR-FTIR analyses. The ratio between oxygen and carbon (Table 2) determined from the survey spectra is increased mainly in the case of DCSBD plasma treatment for 45 s indicating an increase in oxygen-containing bonds. The increased O/C ratio is attributed to the important oxidation occurring during the plasma treatment.
The presence of different carbon bonds was estimated by deconvolution of high-resolution C1s peaks. The chemical bonds were assigned to the C–C/C–H (284.8 eV), C–N/C–O (286.1 eV), C=O (287.8 eV) and COO (289.0 eV), summarized in Table 3. As can be seen from these results (Table 3, Fig. 12), after plasma treatment using DCSBD and MSDBD the oxygen-related chemical bonds C=O and COO increased, compared to the reference sample. The results showed good agreement with those observed using ATR-FTIR which also confirmed oxidation processes on the plasma treated surfaces.
Comparison of the Main Parameters of Plasma Sources
We characterized the three plasma sources used in this study by their corresponding parameters and typical properties (Table 4). Using FTIR spectroscopy of gaseous plasma products, we determined the main active species that interact with the seed surface. The main plasma effect on the physiological parameters of soybean seeds was evaluated by germination dynamics (Fig. 7). From these results it is evident that the optimal treatment time is 15 s in the case of DCSBD plasma treatment, 45 s in the case of MSDBD plasma treatment and an optimal treatment time was not clearly determined in the case of indirect APJ treatment. The effect on germination dynamics is in this case the least significant, and it turns out that longer exposure times of plasma products are necessary. In addition to the active components of the plasma, an important factor affecting germination is the presence of UV radiation. Using the OES measurements, we can say that both DCSBD and MSDBD plasma are dominated by nitrogen UV radiation in accordance with previous detailed studies [30, 35]. However, the indirect treatment by the APJ plasma source excluded the presence of UV radiation, so this factor did not affect the seeds. Based on the optimal treatment times needed to improve germination with individual sources and on the number of seeds that can be treated by the plasma sources at once, we determined the energy consumption required for treatment of 1 kg of seeds for each plasma source.
From the efficiency comparison, we see that the DCSBD plasma source appears to be the most advantageous, which is a consequence of the high power density and the large active area of the plasma. Thanks to this, even a short plasma treatment time is sufficient. The use of MSDBD is advantageous for comparing the plasma effect, the flow of the working gas creates a thicker plasma layer, but the device has a smaller active plasma area than DCSBD. Its advantage lies in its portability, which provides the convenience of testing samples at your location without transporting the entire device.
Conclusion
In our comparative study, we implemented plasma treatment of soybean seeds with three plasma sources in ambient air at atmospheric pressure, which differed in several properties, in one case it was an indirect treatment with plasma products without the presence of radiation. For each type of treatment, we determined the plasma composition, energy efficiency and the optimal treatment time, which was sufficient to improve germination. We also carried out seed surface diagnostics and determined changes in wettability and surface free energy of seeds due to changes in chemical bonds on the surface. It has been confirmed that plasma stimulates and improves seed germination, which is a consequence not only of the change in surface properties from hydrophobic to hydrophilic and the subsequent improvement of water absorption, but also of the further action of RONS and the induction of various biochemical processes and phytohormonal changes inside the seed.
In the case of plasma technology application directly in practice, the energy efficiency of this treatment is no less important than knowing the optimal parameters of plasma processing, the effect of the impact of plasma on its physical surface properties and physiological parameters such as germination and other growth parameters. Based on the obtained results from the compared plasma sources, we can conclude that the DCSBD proved to be the most efficient source, which also enables upscaling and rapid processing of a larger number of samples without damaging the biomaterial.
The use of cold atmospheric pressure plasma technology before sowing can be a promising technology that can bring benefits due to the improvement of germination and growth parameters without the consumption of chemicals and therefore without damaging the environment.
Availability of Data and Materials
The datasets generated during and analysed during the current study are available from the corresponding author on reasonable request.
References
Arora NK (2019) Impact of climate change on agriculture production and its sustainable solutions. Env Sustain 2:95–96. https://doi.org/10.1007/s42398-019-00078-w
Galanakis CM (2015) Food waste recovery: processing technologies and industrial techniques. Academic Press, San Diego
Waskow A, Howling A, Furno I (2021) Mechanisms of plasma-seed treatments as a potential seed processing technology. Front Phys 14(9):617345. https://doi.org/10.3389/fphy.2021.617345
Randeniya LK, De Groot GJJB (2015) Non-thermal plasma treatment of agricultural seeds for stimulation of germination, removal of surface contamination and other benefits: a review. Plasma Process Polym 12:608–623. https://doi.org/10.1002/ppap.201500042
Šimek M, Homola T (2021) Plasma-assisted agriculture: history, presence, and prospects—a review. Eur Phys J D 75:210. https://doi.org/10.1140/epjd/s10053-021-00206-4
Mitra A, Li YF, Klämpfl TG et al (2014) Inactivation of surface-borne microorganisms and increased germination of seed specimen by cold atmospheric plasma. Food Bioprocess Technol 7:645–653. https://doi.org/10.1007/s11947-013-1126-4
Bourke P, Ziuzina D, Boehm D et al (2018) The potential of cold plasma for safe and sustainable food production. Trends Biotechnol 36:615–626. https://doi.org/10.1016/j.tibtech.2017.11.001
Starek-Wójcicka A, Różyło R, Niedźwiedź I et al (2022) Pilot study on the use of cold atmospheric plasma for preservation of bread. Sci Rep 12:22003. https://doi.org/10.1038/s41598-022-26701-1
Waskow A, Avino F, Howling A, Furno I (2022) Entering the plasma agriculture field: an attempt to standardize protocols for plasma treatment of seeds. Plasma Process Polym 19:e2100152. https://doi.org/10.1002/ppap.202100152
Adhikari B, Adhikari M, Park G (2020) The effects of plasma on plant growth, development, and sustainability. Appl Sci 10:6045. https://doi.org/10.3390/app10176045
Chen K, Li GJ, Bressan RA et al (2020) Abscisic acid dynamics, signaling, and functions in plants. J Integr Plant Biol 62:25–54. https://doi.org/10.1111/jipb.12899
Schwechheimer C (2012) Gibberellin signaling in plants - the extended version. Front Plant Sci. https://doi.org/10.3389/fpls.2011.00107
Dubinov AE, Lazarenko EM, Selemir VD (2000) Effect of glow discharge air plasma on grain crops seed. IEEE Trans Plasma Sci 28:180–183. https://doi.org/10.1109/27.842898
Stolárik T, Henselová M, Martinka M et al (2015) Effect of low-temperature plasma on the structure of seeds, growth and metabolism of endogenous Phytohormones in Pea (Pisum sativum L.). Plasma Chem Plasma Process 35:659–676. https://doi.org/10.1007/s11090-015-9627-8
Bormashenko E, Grynyov R, Bormashenko Y, Drori E (2012) Cold radiofrequency plasma treatment modifies wettability and germination speed of plant seeds. Sci Rep 2:741. https://doi.org/10.1038/srep00741
Holubová L, Kyzek S, Ďurovcová I et al (2020) Non-thermal plasma—a new green priming agent for plants? Int J Mol Sci 21:9466. https://doi.org/10.3390/ijms21249466
Švubová R, Kyzek S, Medvecká V et al (2020) Novel insight at the effect of cold atmospheric pressure plasma on the activity of enzymes essential for the germination of Pea (Pisum sativum L. cv. Prophet) Seeds. Plasma Chem Plasma Process 40:1221–1240. https://doi.org/10.1007/s11090-020-10089-9
Kyzek S, Holubová Ľ, Medvecká V et al (2019) Cold atmospheric pressure plasma can induce adaptive response in pea seeds. Plasma Chem Plasma Process 39:475–486. https://doi.org/10.1007/s11090-018-9951-x
Šerá B, Špatenka P, Šerý M et al (2010) Influence of plasma treatment on wheat and oat germination and early growth. IEEE Trans Plasma Sci 38:2963–2968. https://doi.org/10.1109/TPS.2010.2060728
Meng Y, Qu G, Wang T et al (2017) Enhancement of germination and seedling growth of wheat seed using dielectric barrier discharge plasma with various gas sources. Plasma Chem Plasma Process 37:1105–1119. https://doi.org/10.1007/s11090-017-9799-5
Štěpánová V, Slavíček P, Kelar J et al (2018) Atmospheric pressure plasma treatment of agricultural seeds of cucumber (Cucumis sativus L) and pepper (Capsicum annuum L) with effect on reduction of diseases and germination improvement. Plasma Process Polym 15:e1700076. https://doi.org/10.1002/ppap.201700076
Pratap A, Kumar J, Mehandi S, Pandey VR (2016) In: Gupta SK (ed) Breeding oilseed crops for sustainable production: Opportunities and constraints, 1st edn. Academic Press, San Diego. https://doi.org/10.1016/C2013-0-19479-2
Özener O, Yüksek L, Ergenç AT, Özkan M (2014) Effects of soybean biodiesel on a DI diesel engine performance, emission and combustion characteristics. Fuel 115:875–883. https://doi.org/10.1016/j.fuel.2012.10.081
Canakci M, Van Gerpen JH (2003) Comparison of engine performance and emissions for petroleum diesel fuel, yellow grease biodiesel, and soybean oil biodiesel. Trans Am Soc Agric Eng 46:937–944. https://doi.org/10.13031/2013.13948
Canakci M (2005) Performance and emissions characteristics of biodiesel from soybean oil. Proc Inst Mech Eng Part D J Automob Eng 219:915–922. https://doi.org/10.1243/095440705X28736
Pérez-Pizá MC, Prevosto L, Grijalba PE et al (2019) Improvement of growth and yield of soybean plants through the application of non-thermal plasmas to seeds with different health status. Heliyon 5:e01495. https://doi.org/10.1016/j.heliyon.2019.e01495
Pérez-Pizá MC, Cejas E, Zilli C et al (2020) Enhancement of soybean nodulation by seed treatment with non–thermal plasmas. Sci Rep 10:4917. https://doi.org/10.1038/s41598-020-61913-3
Pérez Pizá MC, Prevosto L, Zilli C et al (2018) Effects of non–thermal plasmas on seed-borne Diaporthe/Phomopsis complex and germination parameters of soybean seeds. Innov Food Sci Emerg Technol 49:82–91. https://doi.org/10.1016/j.ifset.2018.07.009
Černák M, Černáková L, Hudec I et al (2009) Diffuse coplanar surface barrier discharge and its applications for in-line processing of low-added-value materials. EPJ Appl Phys 47:22806. https://doi.org/10.1051/epjap/2009131
Homola T, Krumpolec R, Zemánek M et al (2017) An array of micro-hollow surface dielectric barrier discharges for large-area atmospheric-pressure surface treatments. Plasma Chem Plasma Process 37:1149–1163. https://doi.org/10.1007/s11090-017-9792-z
Krumpolec R, Richter V, Zemánek M, Homola T (2019) Multi-hollow surface dielectric barrier discharge for plasma treatment of patterned silicon surfaces. Surf Interfaces 16:181–187. https://doi.org/10.1016/j.surfin.2019.01.014
Medvecká V, Omasta S, Klas M et al (2022) Plasma activated water prepared by different plasma sources: physicochemical properties and decontamination effect on lentils sprouts. Plasma Sci Technol 24:015503. https://doi.org/10.1088/2058-6272/ac3410
Tomeková J, Kyzek S, Medvecká V, Gálová E, Zahoranová A (2020) Infuence of cold atmospheric pressure plasma on pea seeds: DNA damage of seedlings and optical diagnostics of plasma. Plasma Chem Plasma Process 40:1571–1584. https://doi.org/10.1007/s11090-020-10109-8
Abdul-Baki AA, Anderson JD (1973) Vigor determination in soybean seed by multiple criteria 1. Crop Sci 13:630–633. https://doi.org/10.2135/cropsci1973.0011183x001300060013x
Trunec D, Navrátil Z, Tomeková J et al (2022) Chemical composition of gaseous products generated by coplanar barrier discharge in air and N2/O2 mixtures. Plasma Sources Sci Technol 31:115011. https://doi.org/10.1088/1361-6595/ac9c8f
Reuter S, Sousa JS, Stancu GD, Hubertus Van Helden JP (2015) Review on VUV to MIR absorption spectroscopy of atmospheric pressure plasma jets. Plasma Sources Sci Technol 24:054001. https://doi.org/10.1088/0963-0252/24/5/054001
Pavlovich MJ, Ono T, Galleher C et al (2014) Air spark-like plasma source for antimicrobial NOx generation. J Phys D Appl Phys 47:505202. https://doi.org/10.1088/0022-3727/47/50/505202
Mogili PK, Kleiber PD, Young MA, Grassian VH (2006) N2O5 hydrolysis on the components of mineral dust and sea salt aerosol: comparison study in an environmental aerosol reaction chamber. Atmos Environ 40:7401–7408. https://doi.org/10.1016/j.atmosenv.2006.06.048
Schmidt-Bleker A, Winter J, Bösel A et al (2015) On the plasma chemistry of a cold atmospheric argon plasma jet with shielding gas device. Plasma Sources Sci Technol 25:015005. https://doi.org/10.1088/0963-0252/25/1/015005
Zahoranová A, Henselová M, Hudecová D et al (2016) Effect of cold atmospheric pressure plasma on the wheat seedlings vigor and on the inactivation of microorganisms on the seeds surface. Plasma Chem Plasma Process 36:397–414. https://doi.org/10.1007/s11090-015-9684-z
Tong J, He R, Zhang X et al (2014) Effects of atmospheric pressure air plasma pretreatment on the seed germination and early growth of andrographis paniculata. Plasma Sci Technol 16:260–266. https://doi.org/10.1088/1009-0630/16/3/16
Li L, Jiang J, Li J et al (2014) Effects of cold plasma treatment on seed germination and seedling growth of soybean. Sci Rep 4:5859. https://doi.org/10.1038/srep05859
Švubová R, Slováková L, Holubová L et al (2021) Evaluation of the impact of cold atmospheric pressure plasma on soybean seed germination. Plants 10:177. https://doi.org/10.3390/plants10010177
Šerá B, Šerý M, Zahoranová A, Tomeková J (2021) Germination improvement of three pine species (Pinus) after diffuse coplanar surface barrier discharge plasma treatment. Plasma Chem Plasma Process 41:211–226. https://doi.org/10.1007/s11090-020-10128-5
Durak T, Depciuch J (2020) Effect of plant sample preparation and measuring methods on ATR-FTIR spectra results. Env Exp Bot 169:103915. https://doi.org/10.1016/j.envexpbot.2019.103915
Funding
Open access funding provided by The Ministry of Education, Science, Research and Sport of the Slovak Republic in cooperation with Centre for Scientific and Technical Information of the Slovak Republic. This work was supported by the Slovak Research and Development Agency under the contracts No. APVV-21-147 and APVV-19-0386 and by the Slovak Grant Agency for Science VEGA under the contract No. 1/0688/22. This publication is the result of support under the Operational Program Integrated Infrastructure for the project: Advancing University Capacity and Competence in Research, Development and Innovation (ACCORD, ITMS2014+:313021X329), co-financed by the European Regional Development Fund.
Author information
Authors and Affiliations
Contributions
Conceptualization: AZ; Methodology: SĎ, AZ; Experiments and analyses: SĎ, AZ, ĽS, MK, JT, PĎ, MS, DK; Writing – original draft preparation: SĎ, AZ; Writing – review and editing: SĎ, AZ, ĽS, MK, JT, PĎ, MS, DK; Funding acquisition: AZ. All authors have read and approved the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests as defined by Springer, or other interests that might be perceived to influence the results and/or discussion reported in this paper.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Ďurčányová, S., Slováková, Ľ., Klas, M. et al. Efficacy Comparison of Three Atmospheric Pressure Plasma Sources for Soybean Seed Treatment: Plasma Characteristics, Seed Properties, Germination. Plasma Chem Plasma Process 43, 1863–1885 (2023). https://doi.org/10.1007/s11090-023-10387-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11090-023-10387-y